AGRONOMY (AGRONOMIA)
Cutting from Campomanesia aurea O. Berg (Myrtaceae): The collection time of propagules and the effects of auxin
Estaquia de Campomanesia aurea O. Berg (Myrtaceae): Época de coleta de propágulos e efeito de auxina
Cutting from Campomanesia aurea O. Berg (Myrtaceae): The collection time of propagules and the effects of auxin
Revista Brasileira de Ciências Agrárias, vol. 13, no. 1, pp. 1-7, 2018
Pró-Reitoria de Pesquisa e Pós-Graduação - Universidade Federal Rural de Pernambuco
Received: 28 February 2017
Accepted: 24 October 2017
Published: 30 March 2018
ABSTRACT: Campomanesia aurea is a potentially ornamental native species of the Pampa Biome and its vegetative propagation may be an alternative for producing seedlings on a commercial scale. Thus, the goal of this study was to verify the feasibility of vegetative propagation of C. aurea collected at different times under the use of IBA. Cuttings were collected in an in situ in the Municipality of Barão do Triunfo, RS, Brazil, in autumn, winter and spring. IBA at concentrations of zero, 2,000, 4,000, 6,000 and 8,000 mg L-1 were tested. Rooting, callogenesis, leaf retention, mortality and root dry matter were analyzed. There was interaction between the doses of IBA and the collection time of cuttings in relation to rooting. In autumn, there was quadratic behavior with a maximum of 28% rooting with curve inflection at IBA doses above 6,500 mg L-1. The highest callogenesis and leaf retention rates and the lowest mortality rate have been observed in autumn. However, the highest rooting rate and the best root system quality have been observed in spring. We have therefore concluded that the vegetative propagation of C. aurea is feasible and the effects of IBA are dependent on the collection time of propagules.
Key words: commercial production, ornamental, Pampa Biome.
RESUMO: Campomanesia aurea é nativa do Bioma Pampa e apresenta potencial ornamental. A propagação vegetativa pode ser uma alternativa para produção de mudas da espécie em escala comercial. O objetivo deste trabalho foi avaliar a viabilidade de propagação vegetativa de C. aurea coletadas em diferentes épocas utilizando AIB. As estacas foram coletadas in situ, no Município de Barão do Triunfo, RS, no outono, inverno e primavera. Foram testadas as concentrações de AIB de zero, 2.000, 4.000, 6.000 e 8.000 mg L-1. Avaliou-se o enraizamento, calogênese, retenção foliar, massa seca de raiz e mortalidade. Houve interação entre as doses de AIB e a época de coleta das estacas para enraizamento. Houve comportamento quadrático para o outono com máximo de 28% de enraizamento, havendo inflexão da curva em doses superiores a 6.500 mg L-1 de AIB. No outono houve maior calogênese, retenção de folhas e menor mortalidade, entretanto, na primavera verificou-se maior enraizamento e melhor qualidade do sistema radicular. Conclui-se que a propagação vegetativa de C. aurea é viável, sendo que, o efeito de AIB é dependente da época de coleta dos propágulos.
Palavras-chave: produção comercial, ornamental, Bioma Pampa.
Introduction
The Pampa Biome is well-known for its great variety of species. There are more than 2,150 gymnosperms and angiosperms (Boldrini, 2015) and at least 250 species distributed in 54 families have already been identified as potentially ornamental for floral art and landscaping (Stumpf et al., 2012). Considering the commercial scale production, such potential gives innovative opportunities for nurseries and landscapers, therefore promoting the floriculture productive chain as well as the propagation, valorization and preservation of biodiversity and the reduction of extractivism (Stumpf et al., 2015).
The seedling production of Pampa native species is even more relevant when considering the gradual loss of the biome’s area caused by agricultural monoculture expansion, increased silviculture activity with exotic species and due to at least 500 endemic plant species that may be extinct by meadow suppression (Vélez-Martin et al., 2015).
Campomanesia aurea is a Pampa Biome native species popularly known as guabirobinha-do-campo or araçá-rasteiro (Lorenzi et al., 2006). It is potentially ornamental because of its small size, irregular shape, and intense and aromatic flowering. It can be used either in massifs and borders of gardens or as an outdoor vase plant (Stumpf, 2009). It also offers edible fruits (Reitz, 1977; Lorenzi et al., 2006).
It is found in grassy or shrub fields of southern Brazil, mainly in the state of Rio Grande do Sul. It goes as far as the state of Paraná where it occurs in steppe and open areas of Mixed Ombrophilous Forest (Reitz, 1977; Lima et al., 2011). As an essentially heliophilic species, it occurs both in well-drained soils and swamp banks. Its shoot system develops on a xylopodium from which four or more buds branch out sometimes forming dense clumps (Reitz, 1977).
C. aurea is naturally propagated by seeds (Lorenzi et al., 2006), but the vegetative propagation may be an alternative for producing seedlings on a commercial scale. Cutting is the vegetative propagation form commonly used for several ornamental species as it shortens the seedling formation period and guarantees its genetic homogeneity (Ingels, 2010).
Propagation by cutting is possible because the plant cells are able to convert their differentiated condition into meristem and form adventitious roots (Hartmann, 2011). However, such process depends on a number of factors, including the mother plant’s genotype, physiological condition and hormonal balance, which are greatly influenced by seasonal variation. Therefore, there is a collection time more appropriate to each species and environmental condition (Fachinello et al., 2005; Xavier et al., 2013).
The exogenous application of growth regulators may be necessary to enable the vegetative propagation of some species. In addition, it may have an effect on the root system’s quality, uniformity and speed of growth (Hartmann, 2011; Xavier et al., 2013).
Auxins are the most used growth regulators to promote adventitious rooting. Among them, the indole-3-butyric acid (IBA) is widely recommended because it shows high chemical stability, low mobility and no toxic effect in a large dosage range, being therefore effective in promoting rooting in a great variety of species (Hartmann, 2011; Xavier et al., 2013). However, the application of exogenous auxin causes a stimulatory effect in rooting up to a certain dosage, depending on the species and the tissue’s endogenous concentration from which it has an inhibitory effect (Fachinello et al., 2005).
In this context, the goal of our study was to analyze the feasibility of vegetative propagation of C. aurea collected at different times under the use of IBA.
Material and Methods
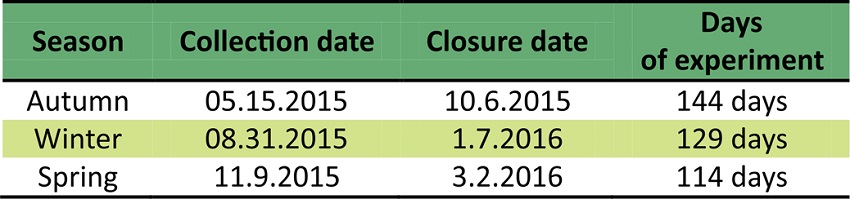
The collections of the propagating material were carried out in an in situ located in the municipality of Barão do Triunfo, RS, Brazil, at latitude 30º18’S and longitude 51º50’W. It shows a wavy to strongly-wavy relief reaching 300 m mean altitude and its annual temperature and precipitation averages 17°C and 1,350 mm, respectively. According to the Köppen classification, its climate type ranges from humid temperate (Cf) to humid subtropical (Cfa) so that there are two well-defined seasons. Collections were carried out on May 15th (autumn), August 31st (winter) and November 9th (spring), 2015. Climatic data are shown in Figure 1.
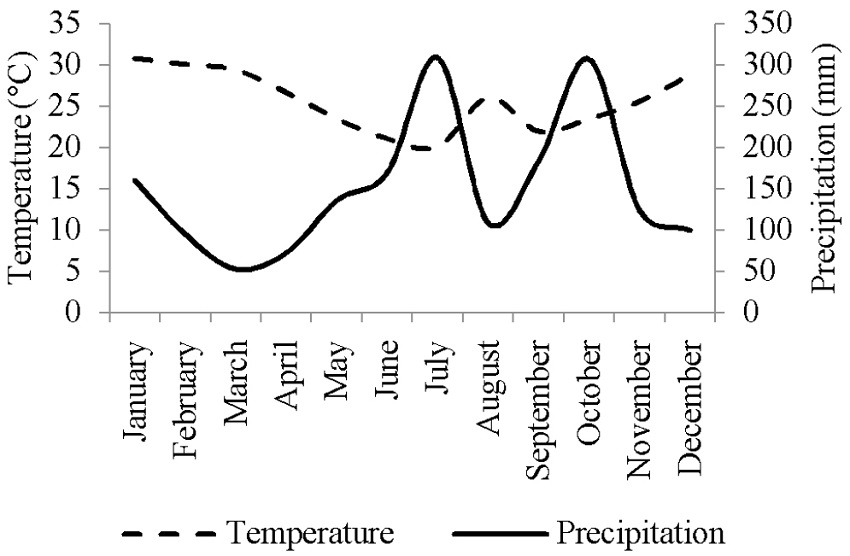
Figure 1
Climatic data of average maximum temperature (°C) and total precipitation (mm) in 2015 obtained from the station of the City of Porto Alegre, RS, Brazil.
Branches from at least 50 specimens were collected, wrapped in a wet newspaper and placed in plastic bags in order to minimize water loss during transportation. Only the apical parts of the branches were used for the cuttings. Branches were cut up to seven centimeters from the leaves insertion and the four apical leaves were saved. A bevel cut at the basal end of each stem was performed and cuttings were immersed in hydro alcoholic IBA solutions at concentrations of zero (control), 2,000, 4,000, 6,000 and 8,000 mg L-1 for five seconds. They were immediately transferred to a 50 cell bedding plant tray (100 mL per cell) filled with carbonized rice hulls. Cuttings were immersed about one centimeter deep in the substrate and trays were moved to a protected environment with an intermittent irrigation system by nebulization and air relative humidity above 90%.
Cuttings were analyzed for rooting, callus formation, leaf retention, root dry matter and mortality. Only cuttings with visible roots of any size were considered as rooted. Callus formation was also verified by eye, regardless root formation, and considered if the cuttings base showed healing structures formed by white parenchyma cells. The period of time to perform the analysis in each collection time was determined based on the verification of the rooting observed under the trays, on leaf retention and cuttings survival (Table 1).
Leaf retention was analyzed by counting the number of leaves after the experiment period so as to calculate its rate in relation to the initial number.
Root dry matter was obtained by drying the material at 65°C up to constant weight and weighing it on analytical balance. The resulting value was divided by the number of rooted cuttings of each sample and expressed in mg.
We have used a completely randomized design with a 3 x 5 factorial arrangement (three seasons and five IBA doses) with four replications of 20 cuttings each, making a total of 400 cuttings for each collection time.
Data were submitted to analysis of variance and for each significant result the means were compared by the Tukey test for collection time and by regression for IBA doses. Rooting, callus formation, leaf retention and root dry matter data have not met the assumption of normality and were thus respectively transformed to √x, √x + 5, arcosen √(x)/100 and √x (results are shown in their original values). Mortality data have not met the normality of variances even after transformation and were therefore analyzed by the non-parametric statistics through the Kruskal-Wallis test. The Spearman correlation of the variables’ residues was then performed. Sigma Plot 11.0 and Costat 6.4 programs were used for statistical analysis.
Results and Discussion
Data statistical analysis has shown interaction between IBA concentrations and collection time for rooting and callus formation (p ≤ 0.25). However, no regression equation could be adjusted for callogenesis, so data have been analyzed only according to collection time (p < 0.01). For other variables, only the collection time factor has statistically differed (Table 2).
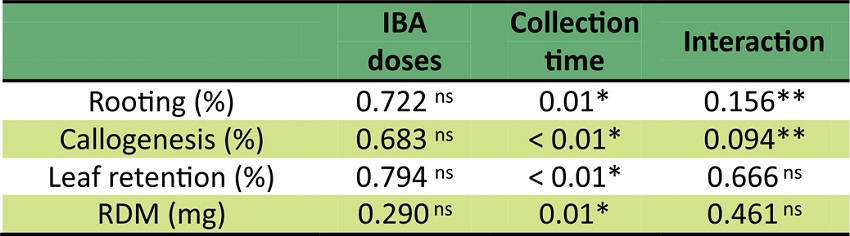
ns not significant; * significant at 1% error probability; ** significant at 20% error probability.
Only cuttings collected in autumn have shown a significant regression for rooting, showing a quadratic behavior. The inflection point has occurred at IBA concentrations above 6,500 mg L-1 with rooting estimated at just over 28% (Figure 2).
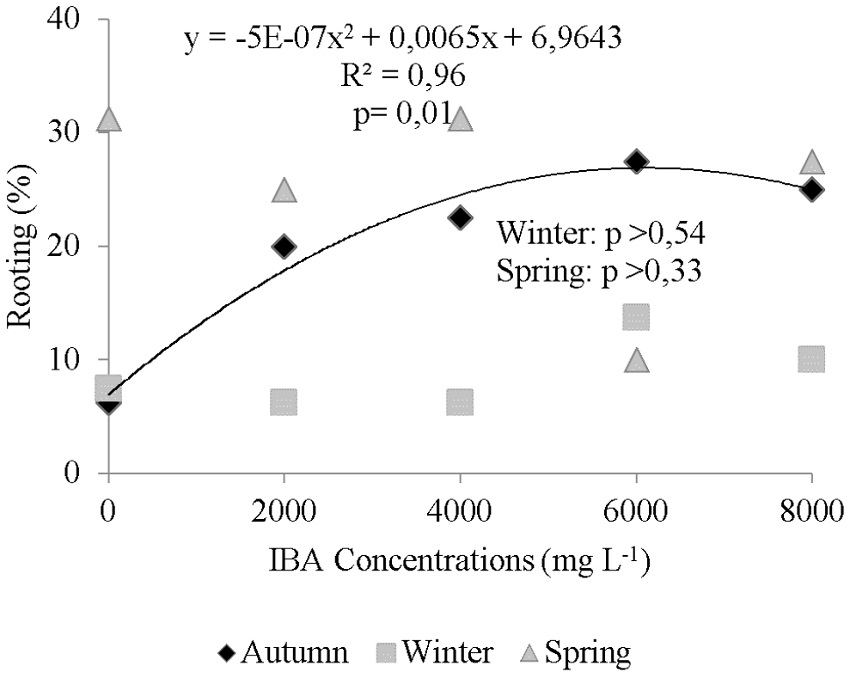
Figure 2
Rooting of cuttings from Campomanesia aurea collected at different times and treated at different doses of indolebutyric acid (IBA).
There was no adjustment of the regression for the application of exogenous auxin for the rooting analysis of cuttings collected in winter and spring, which have averaged 8.75 and 25.00% rhizogenesis, respectively. Cuttings from C. aurea collected in the summer have also not respond to the application of IBA, averaging 28% rooting (Emer et al., 2016).
Several factors, such as genotype, collection time and the endogenous concentration of phytohormones influence the response to the application of growth regulators in cuttings. Cuttings from C. adamantium have also shown the effect of collection time on the response to the application of exogenous auxins. In addition, woody cuttings from C. adamantium have shown a higher rooting rate (57% maximum rooting for cuttings collected in May by failing to use plant regulator) than herbaceous cuttings (Martins et al., 2015). Similar effects have been observed for woody cuttings from jabuticaba trees (Plinia cauliflora), which have shown higher rates than herbaceous propagules with no response to the application of IBA (Sasso et al., 2010).
The opposite behavior has been observed for C. aurea, since herbaceous cuttings collected in spring have shown a higher rooting rate than the more lignified ones collected in winter. The high degree of lignification may have hindered the action of the IBA, therefore resulting in a low rooting rate during winter, such as reported by Franzon et al. (2004) for cuttings from mountain guava trees (Acca sellowiana).
For pitanga trees (Eugenia uniflora), a species from the same family, only cuttings collected from young plants (38%) with no response to the application of IBA have shown rooting. In that case, authors have highlighted the effect of the mother plant’s juvenility on the results (Lattuada et al., 2011). The mother plant’s juvenility is a factor that also may have influenced on the results we have found for cuttings from C. aurea since propagules have been collected from plants in a spontaneous population, so there is naturally greater heterogeneity in the ontogenetic age of plants, which may have contributed to the variations we have observed in the cuttings rooting and the absence of response to exogenous auxin. The ontogenetic age of plants may be a decisive factor for the rooting of species that hardly propagate by cutting (Xavier at al., 2013).
Applying IBA in cuttings from a mini garden of pitanga trees formed by seedlings also did not result in greater rooting (Peña et al., 2015). The same results have been observed in cuttings from Malaleuca trees (Melaleuca alternifolia) (Silva et al., 2012), guava trees (Psidium guajava) (Colombo et al., 2008) and Myrceugenia exsucca (Vidal et al., 2010).
It is noticed that results may vary within cultivars of the same species, as once observed for guava trees (P. guajava). Cuttings from the Paluma cultivar treated at 2,000 mg L-1 of IBA have shown a higher rooting rate (73%) than the ones not treated with IBA. This effect has not been observed for the Século XXI cultivar (Zietemann & Roberto, 2007).
However, other species from the family Myrtaceae have shown a positive effect related to the application of IBA. For example, cuttings from camu camu trees (Myrciaria dubia) have shown 58% rooting at 200 mg L-1 (Delgado & Yuyama, 2010) and the highest rooting rate (41%) in cuttings from Blepharocalyx cruckshanksii was observed at 2,000 mg L-1 (Vidal et al., 2010).
For callus formation, there was statistical difference among collection times (Table 3). In the collection carried out in autumn, 87.25% cuttings have shown callogenesis, but only 18.82% cuttings have formed callus in spring. In the collection carried out in summer, 27% cuttings have shown callus formation, with no influence of the different doses of IBA (Emer et al., 2016).
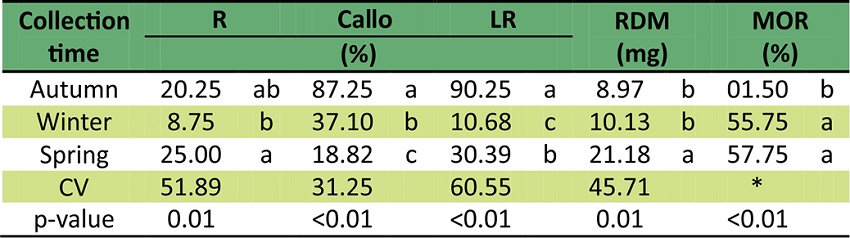
The means followed by the same letter in each column have not differed from each other by the Tukey test at 5% error probability. * Kruskal Wallis.
Callus formation is commonly observed in cuttings under favorable conditions for the formation of adventitious root and it has been associated with difficult-rooting species. However, its development is a precursor of root formation for some species, but for others it may hinder rooting (Hartmann, 2011). As already reported for C. aurea (Emer et al., 2016), rooted cuttings with and without callus were observed in all collection times, which, in this case, indicates that callus formation is not necessarily a precursor of root formation, as one can notice by the low correlation between these two variables (0.32).
For pitanga trees (E. uniflora), the highest callus formation rate was observed in cuttings collected from adult plants by failing to use IBA (Lattuada et al., 2011). The application of IBA has not influence callus formation in cuttings from mountain guava trees (A. sellowiana) and guava trees (P. guajava) (Tavares et al., 1995; Franzon et al., 2004; Colombo et al., 2008). The highest callus formation rate in cuttings from guava trees (P. guajava) has been observed in the collection time with the lowest rooting rate (Tavares et al., 1995). Similarly, the highest rooting rate in cuttings from C. aurea was observed in the collection time with the lowest callus formation rate.
For leaf retention, there was no interaction between IBA doses and the collection times. There were statistical differences among collection times only. Cuttings collected in autumn have shown the highest leaf retention rate (90.25%), followed by cuttings collected in spring (30.39%) and winter (10.68%). The low leaf retention observed in winter may have been caused by the specific specie feature, which naturally lose part of the leaves in this period, as observed in situ. Seasonal changes like photoperiod and temperature affect the senescence of deciduous species as the synthesis and transport of auxin in the leaves decrease and ethylene levels increase, therefore causing the fall of the leaves (Taiz & Zieger, 2013).
In our experiment, leaf retention has not been hindered by increased doses of IBA. The same was observed in cuttings from guava trees (P. guajava) (Zietemann & Roberto, 2007; Colombo et al., 2008; Yamamoto et al., 2010), but the opposite has been reported for young cuttings from pitanga trees, in which leaf retention has been linearly hindered by increased doses of IBA (Lattuada et al., 2011).
The retention of leaves, especially of lower leaves, is important for the rooting and survival of cuttings from clones of Eucalyptus grandis x E. urophylla probably due to the high photosynthetic rate that is provided by a greater leaf area retained in the cuttings (Souza et al., 2013).
Leafless cuttings from guava trees (P. guajava) have not shown root formation and consequently died (Santoro et al., 2010). The same effect has been observed in herbaceous cuttings from jabuticaba trees (P. cauliflora), where only the propagules that have retained their leaves during the whole period of experiment have formed callus and/or root. However, woody cuttings from jabuticaba trees (P. cauliflora) have formed root even when there were loss of leaves (Sasso et al., 2010).
Leaves play an important role in the rooting of cuttings because they produce carbohydrates used in the root process besides being sources of auxins and cofactors that support rooting (Fachinello et al., 2005; Hartmann, 2011). In addition, leaves are primarily responsible for transpiration, which allows the water to move through the xylem so as to control the rate of ascent of sap and water absorption (Kerbauy, 2013). In this sense, leaf retention and adequate water conditions for the root bed are important to keep the cuttings hydrated as water stress causes the inhibition of cell division, protein synthesis and photosynthesis (Taiz & Zieger, 2013).
Although the cuttings collected in spring have retained only 30% of their leaves, they have shown the highest rooting rate (25%) among all times of collection. We believe that several factors have contributed to this result, such as the endogenous hormonal balance and the phenological stage of mother plants, as well as environmental factors.
Cuttings collected in periods of intense vegetative growth show high physiological juvenility, low degree of lignification and high vascular cambium activity, therefore presenting a higher rhizogenesis ability for difficult-rooting species (Fachinello et al., 2005). It is also possible that the higher accumulation of endogenous auxins produced by leaves and sprouting stem buds may have contributed to the high rooting rate and the absence of effects of IBA observed in spring.
In this sense, cuttings from peach trees have shown a higher tryptophan content in autumn and winter than in spring and summer. Decreased tryptophan levels suggest its conversion into indoleacetic acid (IAA), which has correlated with the high rooting rates (Dutra et al., 2002). In line with this information, the application of tryptophan has caused an effect on the rooting and survival of mini-cuttings from difficult-rooting clones of Eucalyptus grandis x E. urophylla (Goulart et al., 2011).
The physiological stage of mother plants used for collecting propagules is especially important for difficult-rooting species, and flowering works as a drainage of photoassimilates therefore hindering the rooting of cuttings (Hartmann, 2011). Cuttings collected from flowering mother plants of C. aurea during spring have shown higher results than those collected during other seasons, but the rooting rate would probably be higher if there were no flowers. According to Vidal et al. (2010), the ability to flower indicates the early state of maturity and the ability to form adventitious organs is decreased. This idea is supported by results from former studies carried out with pitanga trees (E. uniflora) and jabuticaba trees (P. cauliflora), in which collecting cuttings at the beginning or in the end of the breeding season have hindered the rooting process (Sasso et al., 2010; Lattuada et al., 2011).
For the experiment carried out in winter, we have observed that, in the average of the treatments, 13.5% of cuttings have shown new buds (data not given - a characteristic practically missing from the other seasons. This fact may be related to the low rooting rate obtained in the winter, as cuttings reserves have possibly been translocated to bud formation therefore hindering root development. As explained by Hartmann (2011), when buds are formed before root they result in competition of photoassimilates being detrimental to root formation.
Rooting may also have been influenced by the climatic conditions prevailing in the greenhouse. There is a complex interaction between temperature, photoperiod and the level of auxin and other hormones responsible for promoting rhizogenesis (Hartmann, 2011).
Temperature has an important regulatory function in plant metabolism and affects the rooting of cuttings (Xavier et al., 2013). Rhizogenesis involves mitotic divisions with energy expenditure that originate from several chemical reactions, which speed and efficiency depend on temperature (Martins et al., 2015). Low temperatures decrease cuttings metabolism, making the rooting process to take longer or failing to provide adequate conditions for root induction, development and growth (Xavier et al., 2013). We have confirmed this fact since cuttings collected in autumn and winter have shown lower rooting rates than those collected in spring in spite of being kept in the greenhouse for a longer period.
For the root system quality, which is given by the root dry matter variable, there was significant variation only as far as the collection time is concerned. No influence of the IBA doses has been observed. The cuttings collected in spring have shown the best root development. In many cases, applying exogenous auxin does not cause an effect on the rooting of cuttings, but improves the root system quality by increasing the number and length of roots, as reported by many authors in the literature. That was not the case for cuttings from C. aurea.
Increased root system quality has been observed in mini-cuttings from pitanga trees (E. uniflora) in which increasing the dose of IBA up to 2,500 mg L-1 has resulted in a higher number of roots, root length and root dry matter per mini-cutting (Peña et al., 2015). Lattuada et al. (2011) have found similar results for the same species in an experiment where the application of IBA up to 2,400 mg L-1 has provided a quadratic response for root dry matter (Lattuada et al., 2011).
Applying IBA has also improved the root system quality of cuttings from camu camu trees (M. dubia) and guava trees (P. guajava) (Colombo et al., 2008; Delgado & Yuyama, 2010). We have verified the same result in cuttings from C. aurea. Yet no influence of the doses of IBA has been observed on the root system quality of cuttings from Melaleuca trees (M. alternifolia) (Silva et al., 2012).
A high root development rate provides the cuttings with conditions to maintain their feasibility and may directly influence the development of seedlings in the nursery and later in the field establishment due to its high capacity to absorb water and nutrients (Santoro et al., 2010; Silva et al., 2012). In this sense, cuttings collected in spring not only show a higher rooting rate but possibly a better performance when transplanted to field as their root system quality is increased.
The high mortality rates of cuttings collected in winter (55.75%) and spring (57.75%) has statistically differed from the ones collected in autumn, since most have survived (98.5%). However, such a high survival rate did not result in higher rooting. The cuttings collected in winter and spring have shown similar mortality results in spite of statistically differing for the variables rooting and leaf retention.
Mother plants usually show a less active metabolism and a high concentration of tissue reserves in the winter. Since this species naturally loses part of the leaves during this period, what kept cuttings alive even with low leaf retention rate was possibly the branch reserves.
Only little more than 42% of the cuttings collected in spring have survived in spite of showing higher leaf retention rate than the ones collected in winter. In this case, leaves seem to be more important for metabolic activity and therefore essential for the cuttings survival. In addition, the high water content in the cuttings collected in spring has resulted in a greater susceptibility to adverse environmental conditions, which explains the high mortality rate observed in this season.
In this sense, a negative correlation (-0.78) between leaf retention and cuttings mortality has thus been observed, as already reported for cuttings from C. aurea (Emer et al., 2016).
The carbohydrate content of the propagules is a source of energy and carbon and therefore important for synthesizing substances required for root formation, generally correlating positively with the rooting and survival of cuttings (Xavier et al., 2013). The obtained results confirm the fact that intense vegetative growth seasons show low carbohydrate content while vegetative resting plants and more lignified branches have higher reserve content (Fachinello et al., 2005).
No effect of IBA has been observed on the survival of cuttings from mountain guava trees (A. sellowiana). The cuttings collected from the basal portion of the branches have shown the highest survival rate (15%) while apical cuttings have not survived. Authors have explained such result by the small amount of reserves accumulated in the apical portion in relation to the basal portion (Franzon et al., 2004).
No differences in the mortality rate of cuttings from melaleuca trees (M. alternifolia) have been observed regardless the dosage of IBA, indicating the absence of toxic effect from the phytoregulator on this species (Silva et al., 2012). Similar results have been found in cuttings from guava trees (P. guajava) and M. exsucca, where the IBA doses have not influenced the survival of cuttings (Zietemann & Roberto, 2007; Colombo et al., 2008; Vidal et al., 2010). The opposite has been observed in cuttings from pitanga trees (E. uniflora), where increased IBA doses have been detrimental to the survival of cuttings (Lattuada at al., 2011). Similar results have been observed in cuttings from guava trees (P. guajava) treated with a hydro alcoholic IBA solution (Yamamoto et al., 2010).
The highest mortality rate of cuttings from B. cruckshanksii has been observed under the highest IBA doses, which have also caused the highest rooting rate (Vidal et al., 2010).
Even though the rooting results we have found are considered low for a commercial crop, they demonstrate the ability of adventitious root formation in cuttings from C. aurea. By considering the lack of studies regarding this species, especially related to propagation, as well as its potentially ornamental, medicinal and fruitful use, and the lack of native seedlings from the Pampa Biome for recovering degraded areas, our results are promising, taking into account that propagules were collected in a native population. Thus, with simple management practices, such as the reduction of the time between collection and cutting, the selection of genotypes, and the rejuvenation and balanced fertilization of mother plants is possible to achieve more satisfactory results.
Conclusion
It is concluded that the vegetative propagation of Campomanesia aurea is feasible and the effects of IBA are dependent on the collection time of propagules. Spring is the most promising collection time both for the rooting rate and the root system quality.
Acknowledgements
The authors thank to the CNPq, CAPES and FAPERGS for granting scholarships and financial resources for the development of this study.
Literature Cited
Boldrini, I.; Overbeck, G.; Trevisan, R. Biodiversidade de plantas. In: Pillar, V. P.; Lange, O. (Eds.). Os Campos do Sul. Porto Alegre: Rede Campos Sulinos - UFRGS, 2015. p. 51-60.
Colombo, L.A.; Tazima, Z. H.; Mazzini, R. B.; Andrade, G.A.; Kanayama, F.S.; Baquero, J.E.; Auler, P.A. M.; Roberto, S.R. Enraizamento de estacas herbáceas da seleção 8501-1 de goiabeira submetidas a lesão na base e a concentrações de AIB. Semina: Ciências Agrárias, v. 29, n.3, p. 539-546, 2008. https://doi.org/10.5433/1679-0359.2008v29n3p539.
Delgado, J.P.M.; Yuyama, K. Comprimento de estaca de camu-camu com ácido indolbutírico para a formação de mudas. Revista Brasileira de Fruticultura, v. 32, n. 2, p. 522-526, 2010. https://doi.org/10.1590/S0100-29452010005000066.
Dutra, L.F.; Kersten, E.; Fachinello, J.C. Época de coleta, ácido indolbutírico e triptofano no enraizamento de estacas de pessegueiro. Scientia Agrícola, v. 59, n. 2, p. 327-333, 2002. https://doi.org/10.1590/S0103-90162002000200019.
Emer, A.A.; Schafer, G.; Avrella, E.D.; Delazeri, M.; Veit, P.A.; Fior, C.S. Influence of indolebutyric acid in the rooting of Campomanesia aurea semihardwood cuttings. Ornamental Horticulture, v.22, n.1, p. 94-100, 2016. https://doi.org/10.14295/oh.v22i1.855.
Fachinello, J.C.; Hoffmann, A.; Nachtigal, J.C. Propagação de plantas frutíferas. Brasília: Embrapa Informação Tecnológica, 2005. 221p.
Franzon, R.C.; Antunes, L.E.C.; Raseira, M.C.B. Efeito do AIB e de diferentes tipos de estaca na propagação vegetativa da goiabeira-serrana (Acca sellowiana Ber). Revista Brasileira Agrociência, v. 10, n. 4, p. 515-518, 2004. https://periodicos.ufpel.edu.br/ojs2/index.php/CAST/article/view/1036/934. 10 Fev. 2017.
Goulart, P.B.; Xavier, A.; Dias, J.M.M. Efeito dos cofatores hidroquinona, prolina e triptofano no enraizamento de miniestacas de clones de Eucalyptus grandis x E. urophylla. Revista Árvore, v. 35, n. 5, p. 1017-1026, 2011. https://doi.org/10.1590/S0100-67622011000600007.
Hartmann, H.T., Kester, D.E., Davies, F.T.; Geneve, R.L. Plant propagation: principles and practices. 8.ed. Upper Saddle River: Prentices Hall, 2011. 915 p.
Ingels, J.E. Ornamental horticulture: science, operations, & management. 4.ed. New York: Delmar, 2010. 687 p.
Kerbauy, G.B. Fisiologia vegetal. 2.ed. Rio de Janeiro: Guanabara Koogan, 2013. 431 p.
Lattuada, D.S.; Spier, M.; Souza, P.V.D. Pré-tratamento com água e doses de ácido indolbutírico para estaquia herbácea de pitangueiras. Ciência Rural, v.41, n. 12, p. 2073-2079, 2011. https://doi.org/10.1590/S0103-84782011001200006.
Lima, D.F.; Goldenberg, R.; Sobral, M. O gênero Campomanesia (Myrtaceae) no estado do Paraná, Brasil. Rodriguésia, v. 62, n. 3, p. 683-693, 2011. https://doi.org/10.1590/2175-7860201162313.
Lorenzi, H.; Bacher, L.; Lacerda, M.; Sartori, S. Frutas brasileiras e exóticas cultivadas (de consumo in natura). São Paulo: Instituto Plantarum de Estudos da Flora, 2006. 768 p.
Martins, W.A.; Mantelli, M.; Santos, S.C.; Costa Netto, A.P.; Pinto, F. Estaquia e concentração de reguladores vegetais no enraizamento de Campomanesia adamantium. Revista de Ciências Agrárias, v. 38, n. 1, p. 58-64, 2015. http://www.scielo.mec.pt/scielo.php?script=sci_arttext&pid=S0871-018X2015000100009&lng=pt&nrm=iso. 04 Fev. 2017.
Peña, M.L.P.; Zanette, F.; Biasi, L.A. Época de coleta e ácido indolbutírico no enraizamento de miniestacas de pitangueira. Semina: Ciências Agrárias , v. 36, n. 5, p. 3055-3068, 2015. https://doi.org/10.5433/1679-0359.2015v36n5p3055.
Reitz, P. R. Flora ilustrada catarinense. Itajaí: Herbário Barbosa Rodrigues, 1977. 158 p.
Santoro, P.H.; Mikami, A.Y.; Souza, S.G.H.; Roberto, S.R. Influência de folhas e lesões na base de estacas herbáceas no enraizamento de goiabeira da seleção 8501-9. Semina: Ciências Agrárias , v. 31, n. 2, p. 289-294, 2010. https://doi.org/10.5433/1679-0359.2010v31n2p289.
Sasso, S.A.Z.; Citadin, I.; Danner, M.A. Propagação de jabuticabeira por estaquia. Revista Brasileira de Fruticultura v. 32, n. 2, p. 577-583, 2010. https://doi.org/10.1590/S0100-29452010005000054.
Silva, R.C.; Antunes, M.C.; Roveda, L.F.; Carvalho, T.C.; Biasi, L.A. Enraizamento de estacas de Melaleuca alternifolia submetidas a diferentes reguladores vegetais. Semina: Ciências Agrárias , v. 33, n.5, p. 1643-1652, 2012. https://doi.org/10.5433/1679-0359.2012v33n5p1643.
Souza, C.C.; Xavier, A.; Leite, F.P.; Santana, R.C.; Leite, H.G. Padrões de miniestacas e sazonalidade na produção de mudas clonais de Eucalyptus grandis Hill X E. urophylla S. T. Black. Revista Árvore, v. 37, n. 1, p. 67-77, 2013. https://doi.org/10.1590/S0100-67622013000100008.
Stumpf, E.R.T. Cores e formas no Bioma Pampa: plantas ornamentais nativas. Pelotas: Embrapa Clima Temperado, 2009. 272 p.
Stumpf, E.R.T.; Heiden, G.; Iganci J.R.V.; Barbieri, R.L.; Corrêa L.B.; Perleberg, T.D.; Romano C.M.; Fischer, S.Z.; Neitzke, R.S. Prospecting native ornamental plants in the Brazilian pampa for use in landscaping and floral art. Acta Horticulturae, v. 937, p. 1161-1166, 2012. https://doi.org/10.17660/ActaHortic.2012.937.145.
Stumpf, E.R.T.; Silva, P.S.; Romagnoli, I.D.; Fischer, S.Z.; Mariot, M.P. Espécies nativas que podem substituir as exóticas no paisagismo. Ornamental Horticulture, v. 21, n. 2, p. 165-172, 2015. https://doi.org/10.14295/aohl.v21i2.663.
Taiz, L.; Zeiger, E. Fisiologia vegetal. 5.ed. Porto Alegre: Artmed, 2013. 918 p.
Tavares, M.S.W.; Kersten, E.; Siewerdt, F. Efeitos do ácido indolbutírico e da época de coleta no enraizamento de estacas de goiabeira (Psidium guajava L.). Scientia Agricola, v. 52, n. 2, p. 310-317, 1995. https://doi.org/10.1590/S0103-90161995000200018.
Vélez-Martin, E.; Rocha, C.H.; Blanco, C.; Azambuja, B.O.; Hasenack, H.; Pillar, V.P. Conversão e fragmentação. In: Pillar, V.P.; Lange, O. (Eds.). Os Campos do Sul. Porto Alegre: Rede Campos Sulinos - UFRGS , 2015. p.125-132.
Vidal, M.L.; Delgado, P.S.; Barra, E.H.; Peña-Cortés, F. Propagación vegetativa de Myrceugenia exsucca y Blepharocalyx cruckshanksii, especies dominantes del bosque pantanoso de la Depresión Intermedia de la región de La Araucanía, Chile. Bosque, v. 31, n. 3, p. 247-251, 2010. https://doi.org/10.4067/S0717-92002010000300009.
Xavier, A.; Wendling, I.; Silva, R.L. Silvicultura clonal: princípios e técnicas. 2.ed. Viçosa: Editora da UFV, 2013. 279 p.
Yamamoto, L.Y.; Borges, R.S.; Sorace, M.; Rachidi, B.F.; Ruas, J.M.F.; Sato, O.; Assis, A.M.; Roberto, S.R. Enraizamento de estacas de Psidium guajava L. ‘Século XXI’ tratadas com Ácido Indolbutírico veiculado em talco e álcool. Ciência Rural, v. 40, n. 5, p.1037-1042, 2010. https://doi.org/10.1590/S0103-84782010000500006.
Zietemann, C.; Roberto, S.R. Efeito de diferentes substratos e épocas de coleta no enraizamento de estacas herbáceas de goiabeira, cvs. Paluma e Século XXI. Revista Brasileira de Fruticultura , v. 29, n. 1, p. 31-36, 2007. https://doi.org/10.1590/S0100-29452007000100009.
