AGRONOMY (AGRONOMIA)
Selectivity of pre-emergent herbicides in sweet potato genotypes
Seletividade de herbicidas pré-emergentes para genótipos de batata-doce
Selectivity of pre-emergent herbicides in sweet potato genotypes
Revista Brasileira de Ciências Agrárias, vol. 13, no. 1, pp. 1-8, 2018
Pró-Reitoria de Pesquisa e Pós-Graduação - Universidade Federal Rural de Pernambuco
Received: 22 March 2017
Accepted: 15 January 2018
Published: 30 March 2018
ABSTRACT: Sweet potato is sensitive to weeds and a chemical control is needed for high yield production. This work describes the evaluation of three sweet potato genotypes grown in pre-treated soil with clomazone, linuron, metribuzin mechanical weed control (MWC) and without weed control. Visual injury and yield of genotypes were examined, as well as production of branches and roots in fresh and dry biomass. Herbicide residues at root samples were determined by the LC-MS/MS method. The genotypes exhibited a different behaviour under clomazone, metribuzin and linuron applications. Metribuzin provided a yield decrease in all plants. Pre-treated soil with clomazone and mechanical control provided plants more productive. Linuron residues showed values of up to 0.327 ng g-1 in sweet potato roots.
Key words: chemical control, Ipomoea batatas (L.) Lam., LC-MS/MS, minor crops.
RESUMO: Assim como todas as plantas de exploração agrícola, a batata-doce é sensível à interferência imposta por plantas daninhas, o que justifica estudos sobre o controle químico. Objetivou-se com o trabalho avaliar a sensibilidade de três genótipos de batata-doce aos herbicidas clomazone, linuron e metribuzin. Para isso, foi montado um experimento a campo e as plantas foram submetidas a três métodos de controle de plantas daninhas: a aplicação dos três herbicidas em pré-emergência, o controle mecânico e o não controle. Foram avaliadas a intoxicação visual, o desenvolvimento e a produtividade, bem como a presença dos herbicidas nas raízes utilizando-se de cromatografia líquida de alta eficiência acoplada à espectrometria de massas. O crescimento e acúmulo de biomassa pelas plantas foi influenciado pelo genótipo e pelo método de controle. O metribuzin proporcionou diminuição de rendimento em todas as plantas, por outro lado, o clomazone proporcionou plantas mais produtivas ou semelhantes ao controle mecânico. A metodologia de quantificação de herbicidas foi positiva para o linuron, sendo detectados até 0,327 ng g-1 nas raízes das plantas.
Palavras-chave: controle químico, Ipomoea batatas (L.) Lam., LC-MS, minor crops.
Introduction
The sweet potato (Ipomoea batatas (L.) Lam.) is a Convolvulaceae plant cultivated in more than one hundred countries. The roots are used for human consumption and animal feed. Branches can be used as cattle feed as well. In addition, sweet potatoes present a great potential to be incorporated into the production of ethanol because they show higher fuel productivity than common crops such as sugarcane and corn (Ziska et al., 2009, Bennett, 2015; Pietrosemoli et al., 2016).
The use of sweet potato crops for animal feed as well as energy production requires high matter production and, consequently, cultivation in large areas. Thus, among all the phytosanitary problems that interfere in the production of the crops, the occurrence of weeds should be highlighted because it may lead to yield reduction of up to 85% (Seen et al., 2003). Likewise, mechanical control causes damage in the parts of interest and, for large areas, it is hard, slow and unfeasible (Harker & O’ Donovan, 2013).
The use of chemical weed control in sweet potato crops still demands further studies because there are many management methods, high genetic variability between genotypes, herbicide selectivity, as well as root contamination by herbicides. However, some studies showed that chemical weed control is recommended, mainly in cases in which an increase of crop production has been reported after it was used in other countries (Harker & O’ Donovan, 2013).
In Brazilian laws, there is no report of herbicides for weed control in sweet potato crops. Clomazone, linuron and metribuzin are effective pre-emergent herbicides that could be applied in sweet potato crops (Harrison and Jackson, 2011; Meyers et al., 2013). The tolerance of these herbicides is influenced by the genotype and medium soil residue of approximately 90 days (Lugo-Torres and Díaz, 2007; Shaner, 2014), which is enough time for crops to grow and fully occupy the area, thus avoiding weed interference (Ravi and Saravanan, 2012).
Sweet potato species have evolved genetically and have a high number of genotypes. In Brazil, there are hundreds of sweet potato genotypes that present high genetic variability and potential use in the production of forage and ethanol, either directly or as progenitors in genetic improvement programs (Torquato-Tavares et al., 2017). Nonetheless, for sweet potato crops, there are no reports on herbicides for weed control.
Some pre-emergent herbicides present high efficiency in weed control but they remain in the plants, which hence become toxic to consumers. Therefore, clomazone, linuron and metribuzin have been determined in food by LC-MS/MS methods. In some cases, they are quantified even in crops which they are not registered for. (Bakırcı et al., 2014; Walorczyk et al., 2015).
For the reasons mentioned above, this work describes the evaluation of three sweet potato genotypes growing in pre-treated soil with clomazone, linuron and metribuzin. The herbicides residues in the root samples were determined by the LC-MS/MS method.
Material and Methods
Field experiment
This study was carried out under field conditions at the Federal University of Jequitinhonha and Mucuri Valleys (UFVJM), Diamantina, Minas Gerais, Brazil (located at 18°17’31.8”S and 43°51’19.6”W). The climate, according to Köppen’s classification, is Cwa. The experiment was carried out from May to October 2016 and the average monthly values of precipitation and temperature were the following [precipitation (mm) and temperature (oC), respectively]: May (9.6 and 18.2), June (3.4 and 17.1), July (14.4 and 18.1), August (13.6 and 18.4), September (5.0 and 20.4) and October (79.2 and 22.1).
The evaluated genotypes were collected from the germplasm bank of UFVJM. The samples evaluated were UFVJM21, UFVJM27 and UFVJM31. Branches segments were selected with eight knots for seedling production. They were planted in trays with 72 pyramidal cells of 121.2 cm3. Then trays were filled with Quartizarenic Neosol soil as substrate and the seedlings were developed for 35 days.
The substrate presented the following characteristics: pH (H2O): 6.3. P and K (mg dm-3): 152.5 and 277.0, respectively. Ca, Mg, Al, H+Al (cmolc dm-3): 4.2, 0.9, 0.1 and 2.7, respectively; sand, silt, clay and organic matter (%): 70.0, 21.0, 9.0, and 2.4, respectively. The area was prepared in order to form ridge with 1.0 m width and 0.4 m height. Nitrogen (urea), phosphorus (single superphosphate) and potassium (KCl) (20, 40 and 20 kg ha-1) were applied and incorporated before branch transplantation. In addition, side dressing 20 (N) and 20 (K) kg ha-1 were applied 45 days after transplanting (DAT).
One day before transplanting, the soil was irrigated and the herbicides clomazone (Gamit®), linuron (Afalon SC®) and metribuzin (Sencor 480®) were applied at the doses of 720, 675 and 360 g ha-1 (i.a.), respectively. The treatments were applied using a CO2 constant pressure backpack sprayer with a spray tip type (model Teejet XR8004) calibrated at a pressure of 100 KPa. Untreated plots were protected and the weather conditions were favourable to herbicide application.
Each plot consisted of a set of 10 plants spaced 0.3 meters apart. A distance of 1.0 meter was maintained between each plot. The experiment was arranged in a randomized block design with four repetitions in a Split-plot scheme. The plants were evaluated in five plots composed by clomazone, linuron and metribuzin treatments; mechanical weed control (MWC) and no weed control, while the genotypes were allocated to the sub-plots.
The symptoms of visual injury in the plants were evaluated at 7, 14, and 45 days after planting (DAP) according to European Weed Research Council (EWRC) (1964). Branch length of the plants was evaluated at 14, 45, 55 and 145 DAP. At 180 DAP, the production of branches and roots in fresh and dry biomass was determined. They were dried in an forced air chamber at 65 °C for 72 hours, and then they were weighted. In addition, weed population and weed density were evaluated at 55 DAP.
Herbicide determination in the roots using LC-MS/MS
Quantification of residual herbicides in root samples was performed with a Thermo Scientific TSQ Quantum Access Max LC-ESI-MS/MS system. The optimized MS conditions methods were spray voltage, +4 kV; capillary temperature, 300°C; vaporizer temperature, 300°C; drying-gas flow-rate, 8 L h-1.
An ACE C18 column (150 × 2.1 mm, 3 μm) was used for separation of herbicides at 30°C. The mobile phase was composed of a mixture of acetic acid/water (0.1%) and acetonitrile in gradient mode [time (minutes)-acetic acid: acetonitrile]: 0-80:20, 5-20:80, 10-80:20 and 14-80:20. The flow rate was 0.4 mL min-1.
Sweet potato root samples were crushed in an analytical mill; 1.0g was extracted with 10mL ethyl acetate; 5.0g of anhydrous sodium sulfate was added to remove water traces. The samples were centrifugated at 4.000rpm for 15 minutes. The organic phase was evaporated and dissolved in methanol.
The extract was cleaned up at a Lichrolut RP-18 cartridge which was conditioned with 4.0mL of methanol and 4.0 mL of acetic acid solution at pH 4.0. The sample was applied to the cartridge then washed with 4.0 mL of acetic acid solution at pH 4.0. The cartridge was submitted to vacuum drying for removal of water traces. The sample was eluted with 4.0 mL acetonitrile. Nitrogen flux was applied to the sample until dryness. The sample was dissolved in 2.0mL of methanol and filtered in a 0.2µm PTFE syringe filter, and 10 μL was injected for LC-MS/MS analysis.
The recovery tests were carried out in spiked samples at three levels: 0.5; 1.0 and 2.0 µg g-1 by adding 5.0 µg mL-1 of standard solution in methanol at root samples following the procedure of Titato et al. (2007).
Data analysis
Tukey’s test at significance level of 5% was applied to compare average branch length and visual injury was performed, while considering the split-split-plot design and the evaluations over time allocated in sub-sub-plot group (Laurindo et al., 2015).
Results and Discussion
For visual injury, a regression study with graphical representation was performed through multivariate clustering, with a fit quadratic model for each of the sub-plots using the lm function. The fitted model coefficients for each of the sub-plots were subjected to multivariate analysis. Tocher’s optimization method was applied for grouping the curves (Cruz et al, 2014).
There was significance of the factors isolated form and of the interaction. Analysing the branches growth and according to the grouping of regression curves obtained by Tocher’s test the formation four groups were observed. Figure 1 shows the grouping curves by Tocher’s test for the growth of branches in the three sweet potato genotypes.
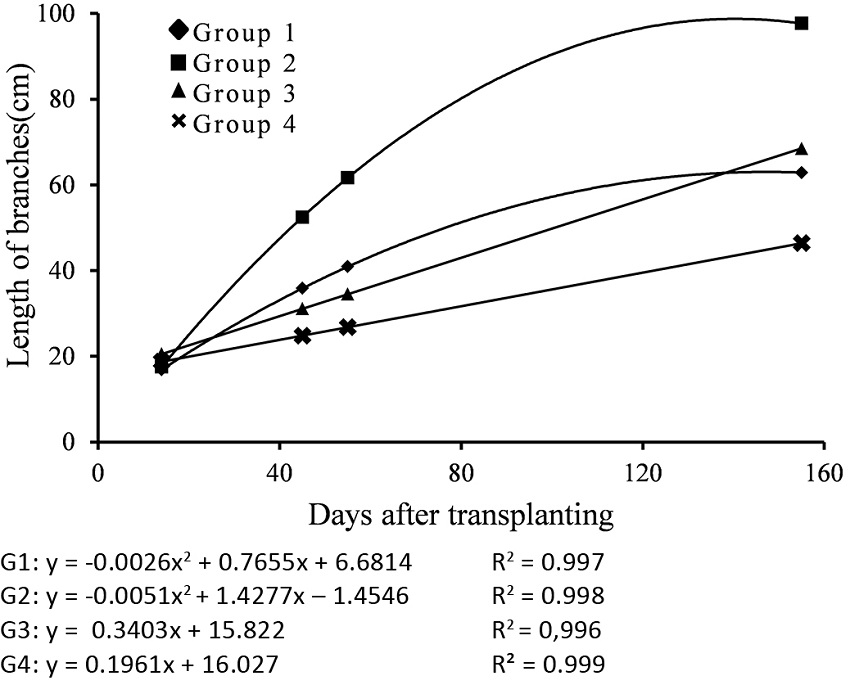
Figure 1
Grouping curves by Tocher’s test for the growth of branches in three sweet potato genotypes, subjected to five weed control types on assessments at 14, 45, 55 and 145 days after transplanting. G1: mechanical control, metribuzin and no control for all plants; linuron for UFVJM21 and UFVJM31. G2: clomazone for UFVJM21 and UFVJM31. G3: clomazone for UFVJM27. G4: linuron for UFVJM27.
Group 1 (quadratic fit) presented the largest number of treatments including mechanical weed control, use of metribuzin and no control method (for all genotypes) and linuron to the UFVJM21 and UFVJM31 genotypes. Group 2 (quadratic fit) includes UFVJM21 and UFVJM31, only when developed in soil treated with clomazone. Similarly, Group 3 (linear fit) was composed of UFVJM27 in soil treated with clomazone. Finally, Group 4 (linear fit) was composed of UFVJM27 genotype that developed in soil treated with linuron. In the clomazone treatment, the UFVJM21 and UFVJM31 genotypes presented the highest growth of branches, whose length was approximately 110% higher than that of plants treated with linuron and 67% higher compared with the other treatments.
When visual injury data is analyzed, it can be seen that the plants presented similar growing with the mechanical weed control, no weed control, linuron and metribuzin treatments (Figure 1). This is due to the fact that there is high genetic variability among all the genotypes. In addition, the decrease in branch growth was influenced by intoxication resulting from competition between herbicides and weeds. Also, mechanical weed control caused injuries in the plants.
Thus, the clomazone treatment for UFVJM21 and UFVJM31 genotypes must be highlighted in comparison to the other groups (Figure 1). This fact is probably due to efficient weed control caused by the herbicide and its low toxicity to the crop, as reported by Lugo-Torres and Diaz (2007) and Meyers et al. (2013).
For the three herbicide treatments, clomazone and linuron caused the lowest intoxication rates and the plants were affected 14 days after transplanting. Otherwise, metribuzin caused the highest intoxication symptoms in the second evaluation period, when the plants showed more than 30% of visual injury (Table 1).
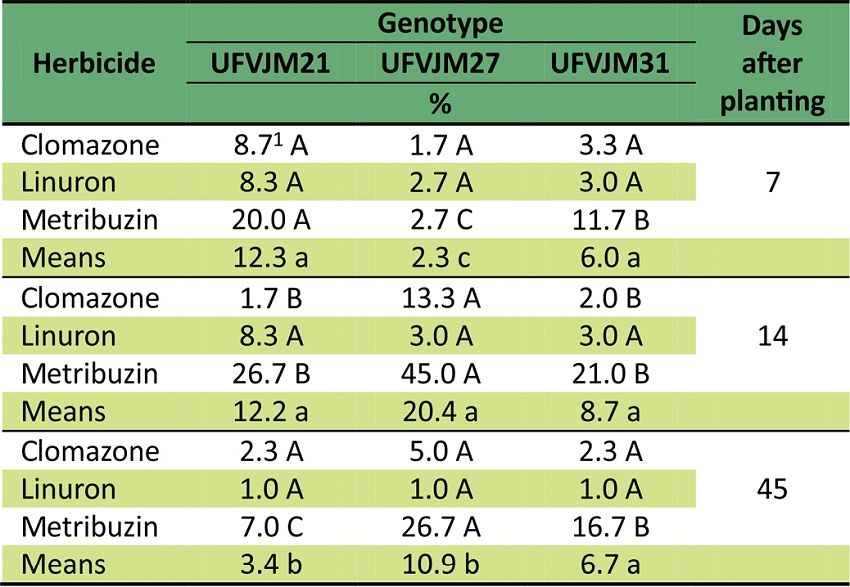
10%: plant with no herbicide visual injury. 100%: dead plant. Means followed by same letter, minuscule vertically and capital horizontally, do not differ by Tukey’s test (p ≥ 0.05). CV1: 61.4%; CV2: 59.7; CV3: 61.3
Linuron intoxication was low and similar among the three genotypes evaluated, although it caused an emphasis on the growth curves (Figure 1). On the other hand, metribuzin intoxication was influenced by genotype and evaluation period, especially UFVJM27, which presented the highest damage at 14 and 45 days after transplanting (Table 1). Thus, the UFVJM27 genotype was that most sensitive to metribuzin.
The experimental area presented high diversity and density of weeds. Nine different weed species were identified in the plots with no herbicides. Plant density ranged from 95 to 498 plants m-2 (Figure 2A). Despite such high density, herbicides controlled over 98% of the weeds, especially clomazone, which allowed the emergence of only four of nine identified species (Figure 2B).
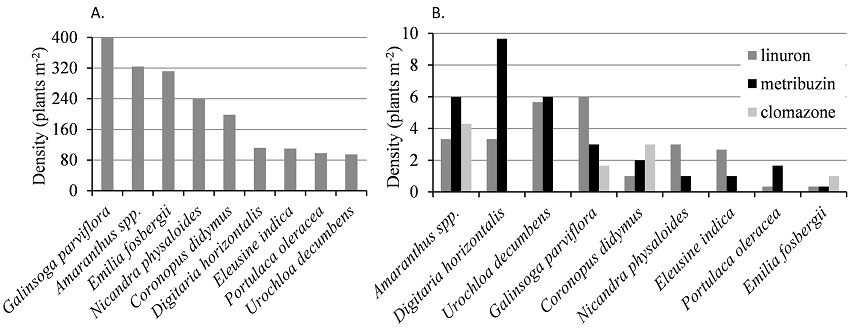
Figure 2
Weed density in sweet potato cropping area at 55 days after transplanting: (A) Without herbicides. (B) With pre-emergent herbicides.
Galinsoga parviflora had the highest number of individuals in untreated plots. However, Amaranthus spp., Coronopus didymus, Digitaria horizontalis, Emilia fosberguii, Nicandra physaloides, Portulaca oleracea and Urochloa decumbens (Figure 2A) were also reported to have high density. The same species appeared in plots with linuron and metribuzin applications (Figure 2B), but at low densities.
Table 2 shows the yield of the three sweet potatoes genotypes submitted to five weed control methods and harvested after 180 days.
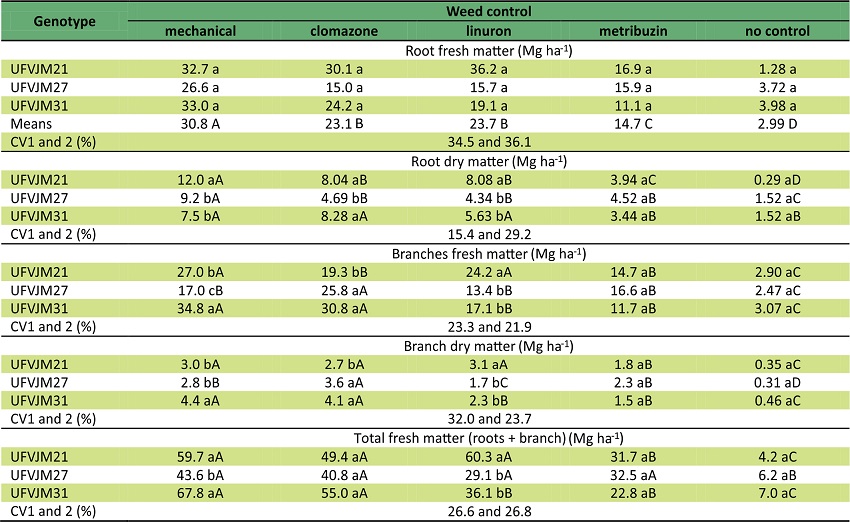
Means followed by same letter, minuscule vertically and capital horizontally, do not differ by Tukey’s test at 5% of significance.
MWC was the most efficient of the five methods, with average production yield equivalent to 30.8 t ha-1 for the three genotypes (Table 2). Genotypes grown in soil treated with clomazone and linuron showed productivity values 24% lower than the mechanical weed control. For metribuzin, productivity values were 52% lower. In the herbicide treatments, sweet potato grew with weeds throughout the entire cycle, and productivity decrease was equivalent to 90%. There was no significative difference between genotypes (Table 2).
The analysis of root dry matter content showed a significant difference between chemical weed control (CWC) and MWC. In a comparison of the data of the UFVJM21 and UFVJM27 genotypes, MWC showed much higher rates of root dry matter than CWC but for the the UFVJM31 genotype, there was no significant difference for root dry matter data between MWC and CWC for clomazone and linuron (Table 2).
The UFVJM21 genotype presented high values for root dry matter data in the linuron treatment while MWC showed the highest weight value of 3.34 tons. This cultivar presented higher root dry matter content than UFVJM27 in comparison with the clomazone treatment. In the metribuzin treatment, there was no significant difference between CWC and no weed control. However, the presence of weeds in the area decreased root dry matter to the values of 11.7, 7.7 and 6.0 Mg ha-1 for UFJM21, UFJM27 and UFJM31 genotypes, respectively, when compared with MWC (Table 2).
As regards branch productivity, the UFVJM21 genotype presented the largest material accumulation in the linuron treatment and MWC. For the UFVJM27 genotype, the best performance was 25.8 Mg ha-1 in the clomazone treatment. And for the UFVJM31 genotype, productivity was hardly influenced by the control method and was equivalent to 32.8 Mg ha-1 in the clomazone treatment and MWC, equal to 14.3 Mg ha-1 in the linuron and metribuzin treatments and equal to 3.0 Mg ha-1 in the no weed control treatment (Table 2).
An evaluation of branch production in MWC for the UFVJM31 genotype showed the highest yield, and it was 205% higher than that of UFVJM27. However, these two genotypes presented similar production in CWC and in no weed control. The linuron and metribuzin treatments were harmful to branch production for the UFVJM31 genotype. However, the clomazone treatment showed the highest value for the UFVJM27 genotype because the hoeing process often damages the plants (Table 2). Dry matter accumulation values in the aerial part were also influenced by genotype and control method. The UFVJM21 genotype presented a reduction in this parameter in the metribuzin treatment and in no weed control. The UFVJM31 genotype presented the highest in MWC. The clomazone treatment presented the best results among CWC (Table 2).
For total yield data, the UFVJM21 genotype was impaired in the metribuzin treatment and when there was no weed management. For the UFVJM27 genotype, the decrease in this parameter was significant only where weed management was not performed. In addition, for the UFVJM31 genotype, the highest yields were found in MWC and in the clomazone treatment. When the three genotypes were compared with MWC, the UFVJM27 genotype presented the lowest yield for the linuron treatment and in the presence of weeds. This herbicide also provided lower productivity for the UFVJM31 genotype (Table 2).
The action of clomazone in plants is associated with the activity of the enzyme phytoene-dhesidrogenase. It also involves the destruction of chlorophyll, reduction of protein synthesis and damage to plastids. Intoxication symptoms include yellowing of leafs followed by stoppage of growth. For linuron and metribuzin, the typical symptoms include chlorosis, necrosis and growth stoppage (Shaner, 2014).
In this study, the plants showed all intoxication symptoms, except for growth stoppage. Moreover, no intoxication symptoms in the assessments done at 90 days after planting. No death was reported 14 days after transplanting. It means that plants are tolerant to applied herbicides, which corroborates other studies on cultivar under different environmental conditions (Harrison and Jackson 2011; Meyers et al., 2013).
In the clomazone treatment, the UFVJM21 and UFVJM31 genotypes presented the best performance in branch growth. However, the genotypes grown in the presence of weeds showed branch growth similar to those without interference. That could be explained by competition, which has stimulated etiolation depending on different light perception (Leduc et al., 2014), and by other stimulus caused by interference (Ravi and Saravanan, 2012).
Sweet potato presents high capacity to compete against weeds, and it has a fast initial growth and the ability to suppress the development of several weed species. These characteristics are related to agronomic environment and variability (Harrison and Jackson, 2011). Another factor is the production of allopathic compounds which reduces the negative effects of weeds (Xuan et al., 2012). Metribuzin was the most toxic herbicide to the plants and presented the lowest yield values. For UFVJM21 and UFVJM31, those two factors combined could be responsible for lower genotype yield when growing in an area treated with metribuzin.
In the experiment, weed presence and herbicide intoxication decreased values for roots biomass and yield biomass. Thus, for branch productivity of the UFVJM27 genotype, the presence of weeds decreased yield productivity by 90%. Moreover, CWC presented higher yield productivity and it is more efficient and economical than MWC (Cedergreen et al., 2009). Table 3 shows the MS parameters for each herbicide.
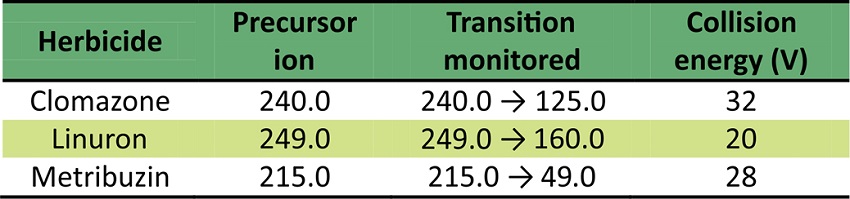
The coefficients of determination, after construction of recovery curves, were greater than 0.99 and the linear equations were: y = - 6740.81 + 5905.51x (clomazone); y = 226973 + 2.27736e + 006x (linuron) and y = -72822.7 + 28129.7x (metribuzin). The recovery values for the herbicides are satisfactory, as shown in Table 4.
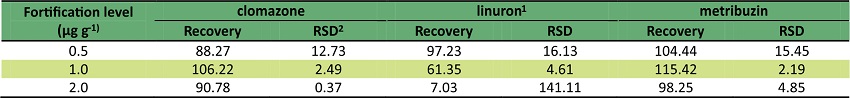
1Levels 2 and 3 were considered unsatisfactory. 2relative standard deviation.
The recovery values were satisfactory for the three herbicides. There were not any traces of metribuzin and clomazone in the root samples. Linuron traces presented average values equal to 0.2711 ng g-1 (Table 5). Solubility values of clomazone and metribuzin are higher than that of linuron. Another point of view is that both herbicides are less persistent than linuron (Shaner, 2014). These facts explain the presence of linuron in the root samples. Furthermore, among the three genotypes, the UFVJM27 genotype presented the highest concentration of linuron residue.
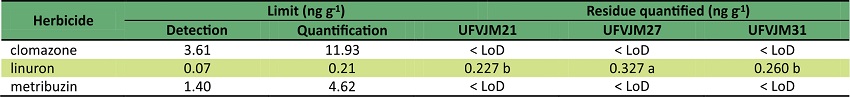
Means followed by same letter do not differ by Tukey’s at 5% of significance. < LoD: lower than the detection limit.
Herbicide loss in soil occurs because there is a great amount of sand present in the study area; in addition, other factors such as biodegradation, pH, organic matter content and soil fertility contribute to decrease herbicide concentration in the rhizospheric region (Shaner, 2014). Sweet potato roots absorbed linuron from soil with no degradation. Linuron’s half-life in the soil ranges from two to five months (Shaner, 2014). Corn and wheat tolerate linuron in the soil with no absorption. By contrast, soybean, tomato and pea absorb it in large quantities and store it in roots and in other parts (Pascal-Lorber, 2010).
Starch is the major component of sweet potato roots and the average value found in this work was 20% at dry basis. For sweet potato genotypes from Sri Lanka, values were reported to range from 33-to-64% (Senanayake, 2013). Starch content in plants is related to the tolerance capacity of herbicides. Some plants have an affinity for enzymes related to the metabolism of starch which is retained together with this reserve component (Jain et al., 2015).
In order to commercialize high quality food while following food safety protocols, the World Health Organization (WHO, 2017) establishes a maximum residue level (MRL) of herbicides in food. These values are based on an agreement between more than 160 countries worldwide through Codex Alimentarius. Similarly, in Brazil, through the Brazilian Health Surveillance Agency (Jardim & Caldas, 2012), residual monitoring of phytosanitary products is conducted in agricultural foods. MRL values for linuron in corn, carrots and cassava present values of 0.3; 0.3 and 1.0 mg kg-1, respectively. These values are higher than the value determined in this work for sweet potato.
Conclusions
The three sweet potato genotypes presented different behaviors under clomazone, metribuzin and linuron in pre-emergent applications.
The herbicides decreased root fresh matter yield. However, clomazone and linuron did not cause a decrease of total fresh matter yield for UFVJM21 and UFVJM27.
Linuron did not affect the yield of branches for UFVJM21, whereas clomazone did not affect the yield of branches in UFVJM27 and UFVJM31. Moreover, metribuzin caused a reduction in all parameters. The values of linuron residues were up to 0.327 ng g-1 in root samples.
Acknowledgement
The authors acknowledge the financial support provided by CNPq, CAPES and FAPEMIG.
Literature Cited
Bakırcı, G.T.; Acay, D.B.Y.; Bakırcı, F.; Ötleş, S. Pesticide residues in fruits and vegetables from the Aegean region, Turkey. Food Chemistry, v.160. n.1, p.379-392, 2014. https://doi.org/10.1016/j.foodchem.2014.02.051.
Bennett, B.C. Subsistence Farming in Southern Florida (1840-1940): The Importance of Sweet Potato-In Memory of Daniel F Austin. Economic Botanic, v.169, n.3, p.185-198, 2015. https://doi.org/10.1007/s12231-015-9312-0.
Cedergreen, N.; Felbyb, C.; Porter, J.R.; Streibig, J.C. Chemical stress can increase crop yield. Field Crops Research, v.114, n.1, p.54-57, 2009. https://doi.org/10.1016/j.fcr.2009.07.003.
Cruz, C.D.; Carneiro, P.C.S.; Regazzi, A.J. Modelos biométricos aplicados ao melhoramento genético. Viçosa: Universidade Federal de Viçosa, 2014. 668p.
Davies, J.; Caseley, J.C. Herbicide safeners: A review. Pesticide Science, v.55, n.3, p.1043-1058, 1999. https://doi.org/10.1002/(SICI)1096-9063(199911)55:11.
European Weed Research Council - EWRC. Report of 3rd and 4rd meetings of E.W.R.C. Cittee of methods in weed research. Weed Research, Bachlaan, v. 4, n. 1, p. 88, 1964.
Harker, K.N.; O’donovan, J.T. Recent Weed Control, Weed Management, and Integrated Weed Management. Weed Technology, v.27, n.1, p.1-11, 2013. https://doi.org/10.1614/WT-D-12-00109.1.
Harrison Jr., H.F.; Jackson, D.M. Response of two sweet potato cultivars to weed interference. Crop Protection, v.30, n.2, p.1291-1296, 2011. https://doi.org/10.1016/j.cropro.2011.05.002.
Harrison Jr., H.F.; Jackson, D.M. Greenhouse Assessment of Differences in Clomazone Tolerance among Sweetpotato Cultivars. Weed Technology , v.25, n.3, p.501-505, 2011a. https://doi.org/10.1614/WT-D-10-00132.1.
Jain, R.; Jain, N.; Kumar, A. Structural Prediction, Glucose-1-Phosphate Interaction and Influence of Broad Leaves Herbicides on Spinach Leaves α-glucan Phosphorylase: An in Silico Study. Bioscience Bioengineering, v.3, n.2, p.51-59, 2015. https://doi.org/10.13189/bb.2015.030401.
Jardim, A.N.O.; Caldas, E.D. Brazilian monitoring programs for pesticide residues in food - Results from 2001 to 2010. Food Control, v.25, n.4, p.607-616, 2012. https://doi.org/10.1016/j.foodcont.2011.11.001.
Kelly, S.T.; Shankle, M.W.; Miller, D.K. Efficacy and Tolerance of Flumioxazin on Sweetpotato (Ipomoea batatas). Weed Technology , v.20, n.2. p.334-339, 2006. https://doi.org/10.1614/WT-04-330R1.1.
Laurie, S.M.; Maja, M.N.; Ngobeni, H.M.; Du Plooy, C.P. Effect of Different Types of Mulching and Plant Spacing on Weed Control, Canopy Cover and Yield of Sweet Potato (Ipomoea batatas (L.) Lam). American Journal Expimerimental Agriculture, v.5, n.5, p.450-458, 2015. https://doi.org/10.9734/AJEA/2015/12404.
Laurindo, B.S.; Laurindo, R. D. F.; Azevedo, A.M.; Nick, C.; Silva, D.J.H.; Mizubuti, E.S.G. Seleção de acessos de tomateiro resistentes à pinta-preta pela análise de agrupamento das curvas de progresso da doença. Pesquisa Agropecuária Brasileira, v. 50, n. 4, p. 106-114, 2015. https://doi.org/10.1590/S0100-204X2015000200002.
Leduc, N.; Roman, H.; Barbier, F.; Péron, T.; Huché-Thélier, L. Review Light Signaling in Bud Outgrowth and Branching in Plants. Plants, v.3, n.2, p.223-250, 2014. https://doi.org/10.3390/plants3020223.
Lugo-Torres, M.L.L.; Díaz, M. Weed control in sweet potato. Journal of Agriculture of the Universtiy of Puerto Rico, v.91, n.3. p.1-11. 2007. http://revistas.upr.edu/index.php/jaupr/article/view/3283/2798. 21 Mar. 2017.
Meyers, S.L.; Jennings, K.M.; Monks, D.W. Herbicide-Based Weed Management Programs for Palmer Amaranth in Sweetpotato. Weed Technology , v.27, n.2, p.331-340, 2013. https://doi.org/10.1614/WT-D-12-00036.1.
Oliveira, A.C.B.; Sediyama, M.A.N.; Sediyama, T.; Finger, F.L.; Fruz, C.D. Variabilidade genética em batata-doce com base em marcadores isoenzimáticos. Horticultura Brasileira, v. 20, n. 4, p. 576-582, 2002.
Pascal-Lorber, S.; Alsayeda, H.; Jouanin, I.; Debrauwer, L.; Canlet, C.; Laurent, F. Metabolic Fate of [14C]Diuron and [14C]Linuron in Wheat (Triticum aestivum) and Radish (Raphanus sativus). Journal of Agriculture andFood Chemistry , v. 58, n.20, p.10935-10944, 2010. https://doi.org/10.1021/jf101937x.
Peel, M.C.; Finlayson, B.L.; McMahon, T.A. Updated world map of the Köppen-Geiger climate classification. Hidrology and Earth System Science, v.11, n.2, p.1633-1644, 2007. https://doi.org/10.5194/hess-11-1633-2007.
Pietrosemoli, S.; Moron-Fuenmayor, O.E.; Paez, A.; Villamide, M.J. Effect of including sweet potato (Ipomoea batatasLam) meal in finishing pig diets on growth performance, carcass traits and pork quality. Animal Science Journal, v.87, n.10, p.1281-1290, 2016. https://doi.org/101111/asj12546.
Ravi, V.; Saravanan, R. Crop physiology of sweetpotato; fruit, vegetable and cereal science and biotechnology. http://www.globalsciencebooks.info/Online/GSBOnline/images/2012/FVCSB_6(SI1)/FVCSB_6(SI1)17-29o.pdf. 21 Mar. 2017.
Seen, J.E.; Creamer, N.G.; Monks, D.W. Critical Weed-Free Period for ‘Beauregard’ Sweetpotato (Ipomoea batatas). Weed Technology , v.17, n.2, p.686-695, 2003. https://doi.org/10.1614/WT02-089.
Senanayake, A.S.; Ranaweera, K.K.D.S.; Gunaratne, A.; Bamunuarachchi, A. Comparative analysis of nutritional quality of five different cultivars of sweet potatoes in Sri Lanka. Food Science and Nutrition, v.1, n.4, p.284-291, 2013. https://doi.org/10.1002/fsn3.38.
Shaner, D.L. Herbicide Handbook. Lawrence: Weed Science Society of America, 2014. 430p.
Titato, G.M.; Bicudo, R.C.; Lanças, F.M. Optimization of the ESI and APCI experimental variables for the LC/MS determination of s-triazines, methylcarbamates, organophosphorous, benzimidazoles, carboxamide and phenylurea compounds in orange samples. Journal of Mass Spectrometry, v.42, n.10, p.1348-1357, 2007. https://doi.org/10.1002/jms.1310.
Torquato-Tavares, A.; Nascimento, I.R.; Pascual-Reyes, I.D.; Santana, W.R.; Silveira, M.A. Potential for sweet potato (Ipomoea batatas (L.) Lam.) single crosses to improve ethanol production. Revista Chapingo, v.23, n.1, p.59-74, 2016. https://doi.org/10.5154/r.rchsh.2016.05.013.
Walorczyk, S.; Drożdżyński, D.; Kierzek, R. Determination of pesticide residues in samples of green minor crops by gas chromatography and ultra-performance liquid chromatography coupled to tandem quadrupole mass spectrometry. Talanta, v.132, n.2, p.197-204, 2015. https://doi.org/10.1016/j.talanta.2014.08.073.
World Health Organization - WHO. Codex Alimentarius International Food Standards. Pesticides. http://www.fao.org/fao-who-codexalimentarius/thematic-areas/pesticides/en/#c452840. 07 Feb. 2017.
Xuan, T.D.; Toyama, T.; Khanh, T.D.; Tawata, S.; Nakagoshi, N. Allelopathic interference of sweet potato with cogongrass and relevant species. Plant Ecology, v.213, n.3, p.1955-1961, 2012. https://doi.org/10.1007/s11258-012-0136-z.
Ziska, L.H.; Runion, G.B.; Tomecek, M.; Prior, S.A.; Torbet, H.A.; Sicher, R. An evaluation of cassava, sweet potato and field corn as potential carbohydrate sources for bioethanol production in Alabama and Maryland. Biomass and Bioenergy, v.33, n.11, p.1503-1508, 2009. https://doi.org/10.1016/j.biombioe.2009.07.014.
