Abstract: Previous investigations have tried to prove the effectiveness of interventions that seek to improve rest in elderly people. Neurofeedback (NF) is an intervention aimed to train self-regulation of brain functions, which, in other populations, has proven to be beneficial for rest quality as well. The objective of this experimental study was to determine the effect of NF on sleep quality in elderly women that stay in nursing homes or institutions that care for the elderly by comparing the scores of 14 participants. No statistically significant differences were observed between the NF group and the control group; however, sleep quality improved in the NF group and alpha frequency increased significantly in participants after the intervention. Although there is evidence of modification in physiological indicators and increased sleep quality in the NF group, it is likely that because of the living conditions of the participants, the effects have not been powerful enough to manifest themselves in the sleep quality evaluation.
Keywords:neurofeedbackneurofeedback,elderlyelderly,sleepsleep,institutionalizedinstitutionalized,womenwomen.
Resumen: Diversas investigaciones han tratado de probar la efectividad de las intervenciones para mejorar el descanso en las personas mayores. Neurofeedback (NF) es una intervención destinada a entrenar la autorregulación de las funciones cerebrales, una condición que es beneficiosa con otras poblaciones para mejorar el descanso. Se realizó un estudio experimental con 14 adultas mayores institucionalizadas para evaluar el efecto de NF en la calidad del sueño. Al comparar las puntuaciones, no se observaron diferencias estadísticamente significativas entre el grupo NF y el grupo control. Sin embargo, se observó mejora en la calidad del sueño en el grupo NF y se demostró que después de la intervención las participantes aumentaron significativamente la frecuencia alfa. Aunque existe evidencia de modificación en los indicadores fisiológicos e incremento de la calidad del sueño en el grupo NF, es probable que, debido a las condiciones de las participantes, los efectos no hayan sido lo suficientemente potentes como para manifestarse en la evaluación de la calidad del sueño.
Palabras clave: neurofeedback, adultas mayores, sueño, institucionalizado, mujeres.
Artículos
The effect of neurofeedback on sleep quality amongst elderly women in nursing homes
Efecto de neurofeedback sobre la calidad de sueño en adultas mayores institucionalizadas
Received: 23 January 2020
Accepted: 22 April 2020
Currently, the concern for studying and identifying physical and psychological phenomena during the last years of life has become increasingly important due to an escalation of life expectancy, shown in the significant growth of the elderly population worldwide (WHO, 2016). Ageing is a progressive and inevitable process in the life of humans, consisting of a multiplicity of changes, amongst which is the gradual decrease of physiological activity. How these changes manifest in the last stage of life will depend on individual and environmental variables, such as sex, age, health habits, and medical history (Rallabandi et al., 2020). The deterioration associated with ageing constitutes a health condition with a high impact on the population; one of the frequently present variables that may affect such deterioration is the quality of sleep (Crowley, 2011).
The elderly experience changes in the sleep-wake cycle patterns, which produce modifications in sleep structure and quality: latency time increases, as well as the frequency and duration of night-time awakenings; stages 1 and 2 of non-REM (Rapid Eye Movements) phases become longer while stages 3 and 4 (deep sleep), along with REM phases, become shorter (Bombois et al., 2010). Sleep disturbances are one of the most frequent geriatric syndromes. As one gets older, sleep alterations increase significantly and up to 57% of them can be attributed to non-biological factors. Primary sleep disorders (obstructive apnea syndrome sleep, restless legs syndrome, among others) can be related to psychological conditions such as depression or anxiety. On the other hand, poor sleep quality can be a risk factor for medical conditions such as cardiovascular and respiratory diseases, as well as cognitive impairments. Nevertheless, there is no clear relationship between the pathogenesis of mild cognitive impairment (MCI) and dementia, particularly Alzheimer's disease (AD). Custodio (2017) estimates that nearly 40% of patients with AD have alterations in continuous spontaneous ventilation.
Sleep is the state of uniform rest in an organism, characterized by low levels of physiological activity (blood pressure and respiration) and a lower response to external stimulation. During this state, there is a recovery process associated with the relationship between physical well-being, mood regulation, concentration and memory, as long as the quantity and quality of sleep hours are adequate for the individual (Durán et al., 2014). It is possible to monitor relevant strategies to facilitate sleep according to age and daily activities (Irish et al., 2015) to ensure that both the quantity and quality of sleep facilitates the recovery process. Research has been done in this area. For example, Ho and Siu (2018) made a systematic review of the different interventions from an occupational therapy approach to improve sleep quality, in which they reported on two studies with older adults (Gutman et al., 2016; Leland et al., 2016). All participants reported favourable results in terms of decreased insomnia.
In the first study, Gutman et al. (2016) at the Columbia University Medical Center in the U.S, used a special pillow and bracelet designed for meditation, in addition to sleep hygiene techniques, to research their influence on sleeping habits. Twenty-nine participants (healthy adults, between 25 and 65 years old) were randomly assigned to each group [pillow (. = 10), meditation (. = 9), and sleep hygiene (. = 10)]. At the beginning of the study, all of the participants used a 7-day sleep hygiene regimen to reduce poor sleep habits. Then for 14 days, 10 participants used the pillow and sleep hygiene method, nine used the meditation and sleep hygiene method, and 10 continued the sleep hygiene method only. They used the General Sleep Disturbance Scale (GSDS), and the Pittsburgh Sleep Quality Index (PSQI), to assess perceived sleep quality; an actigraphy accelerometer that measures the length of time required to fall asleep and a sleep journal. The meditation group experienced statistically greater time asleep than both the pillow (. < .006, . = 1.87) and sleep hygiene groups (. < .03, . = 1.80). The pillow group experienced statistically fewer night-time awakenings than the meditation (.< .04, . = −1.53) and sleep hygiene (. < .004, . = −1.43) groups. No differences were found between groups in perceived sleep quality, length of time needed to fall asleep, and fatigue level the next day.
In the second study, conducted in Los Angeles in the U.S by Leland et al. (2016) interventions were used based on occupational therapy and a randomized controlled trial was done to explore baseline napping and sleeping patterns, as well as 6-month changes in these outcomes in an ethnically diverse group of community-living older adults with 217 participants, predominantly female, (119 participants in the experimental group and 98 in the control group). The intervention involved content regarding healthy lifestyle behaviours including sleep. To foster the adoption and maintenance of lifestyle changes, participants received as many as 10 individual 1-hour sessions with an occupational therapist in their home or community. They created three variables that measured change during sleep or napping. These included: (1) changes in night-time sleeping, (2) changes in nap duration, and (3) change in total sleep in 24 hr. These change measures were calculated by subtracting responses to baseline sleep measures from responses to follow-up measures. They suggest that participating in an occupation-based lifestyle intervention may be related to enhanced sleep because the significant difference between the groups was seen in total sleep duration during the follow-up, in which the adults no longer napped (p < .05). For experimental participants who no longer napped, the loss of daytime sleep minutes was compensated for by an average increase of 48 minutes of night-time sleep, resulting in a net gain in total sleep time of 9 minutes.
Another alternative for sleep improvement is neurofeedback (NF). It trains self-regulation skills in the person through computerized technology, intending to correct irregularities in brain wave patterns associated with mental health and/or cognitive problems (Hammond, 2011). Through acquired self-regulation, the subject can experience non-invasive brain training that allows constant stimulation by a learned behaviour that eventually increases rest quality.
Hammer (2011) carried out a pilot study of Z-Score SMR and Individualized Protocols by Neurofeedback in eight adults with insomnia in Portland in the U.S. The mean age of the participants was 49.63 years old and included 5 women and 3 men. Their symptoms included woken after sleep onset, woken too early, sleep onset latency and non-restorative sleep. It was a randomized, single-blind trial, with a parallel-group, in which both groups received 15 20-minute sessions of neurofeedback. During post-treatment, all participants were classified as normal sleepers according to the scores of the Insomnia Severity Index (ISI) and Pittsburgh Sleep Quality Inventory (PSQI). However, there were no significant differences in the findings between the two groups, so they recommended a future large-scale study that could use the Z-Score SMR protocol, which is considerably less burdensome.
Tang et al. (2015) implemented an intervention using neurofeedback, with auditory and visual reinforcement, in the same direction. They trained 13 subjects from the community sites in Philadelphia and Seattle in the U.S to self-administer a 30-minute visual and auditory stimulation program (using glasses and headphones) before bedtime for one month and asked them to record their sleep patterns in a diary. Participants included eight adults over 88-years-old, without cognitive impairment (displaying an average of 29 MMSE points) and moderate difficulties in sleep quality assessed by the Pittsburgh Sleep Quality Index (average of 9.9 PSQI points). They intended to modify brain waves towards deep relaxation and sleep status, although no record of them was made. At the end of the intervention, the severity of insomnia decreased to below threshold and 63% of participants no longer met the criteria for insomnia (PSQI ≤ 7).
In Mexico within 20 years, the country's older adult population has steadily increased. The elderly population has practically doubled (10.1 million), especially amongst women as they represent 53.5% of the older adult population (5.4 million), and although most live with their families, a large number also live in nursing homes or institutions that care for the elderly (INEGI, 2014), within which the effectiveness of interventions to improve sleep quality has not been proven. Consequently, it seemed relevant to ask if there would be a difference in the quality of sleep between women who stay in nursing home-type institutions who receive NF and those who do not. Therefore, the objective of the present investigation is to research the effect of NF on the quality of sleep in these elderly women.
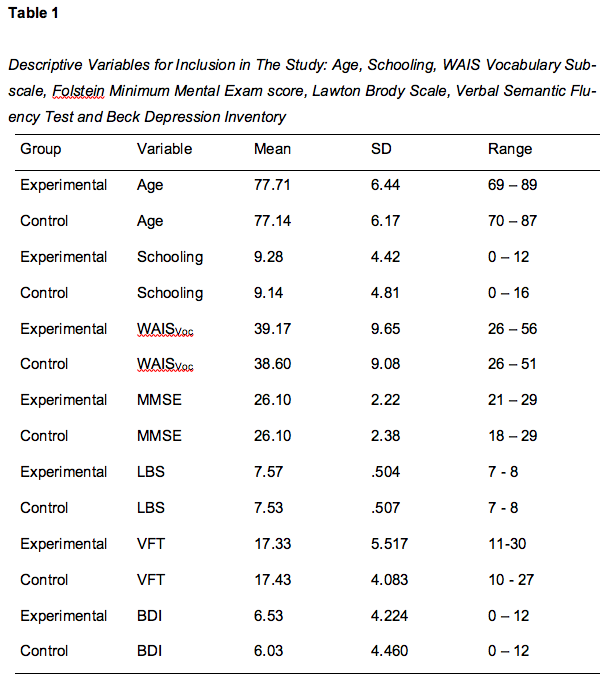
The objective of this research was to evaluate the effects of an intervention program based on neurofeedback amongst elderly women who live in institutions, which care for the elderly, in Mexico City. An experimental study was conducted involving 14 women (mean age was 77.42 ± 2.43 years and schooling was 9.21 ± 1.67 years). Participants did not suffer from neurological, psychiatric or chronic-degenerative diseases that impeded their independence (assessed by Lawton Brody Scale, score ≥ 7 and semantic verbal fluency test, score> 10). They did not present drug and/or alcohol addiction, nor depression symptoms (score <12) based on the Beck Depression Inventory (Beck, 1987). Also, participants did not experience dementia symptoms (score ≥ 18) according to the Mini-Mental State scale of Folstein et al. (1975). Participants’ understanding of verbal instructions was evaluated by the vocabulary subscale of the Wechsler Adult Intelligence Scale (score ≥ 26) (Wechsler, 2012). Their vision was normal or corrected to normal (assessed by the Snellen chart). The descriptive variables for inclusion in the study are shown below in Table 1.
Participants were randomly assigned (matched by age and schooling) to the control group and the experimental group. They were informed of the scope and limitations of the study and were assured that their participation was confidential, voluntary, and they were free to withdraw from the study at any time. They then signed the consent form and gave their informed consent. At the end of the study, people assigned to the control group were invited to take the intervention program if they wanted. This was done to ensure no participant was disadvantaged in the course of the study and since our sample was elderly women, which can be considered a vulnerable population we were mindful of ensuring that they were treated with respect and dignity throughout the research process, and no harm came to them.
In the experimental group, each participant attended 30 sessions, at the rate of five weekly sessions of approximately 40 minutes each, divided into blocks of 10 minutes of training and two minutes of rest interspersed, by placing three electrodes during the experiment (Cz, according to international system 10-20, for signal registration, the lobe of the right ear for reference and lobe of the left ear in the function of the ground). The increase in power of alpha frequency (8-14 Hz) was also reinforced.
In the sessions, we projected images in the form of puzzles, in such a way that if the power of the alpha frequency increased, a new piece appeared until the image was completed. Participants had to follow directions to maintain their brain activity to complete images that were formed. Throughout each session, a member of the research team stayed with them, asking how they felt and verbally reinforcing the progress they had. To maintain the motivation of the participants, they were given a written recognition in session 10 and a visual stimulation breath trainer was included on the screen, consisting of a bar with a circle that marked the optimal rhythm of breathing when performing neurofeedback training. In session 20, their effort was verbally recognized, and they were told that they were in the last phase of training.
From session 25, discontinuation of visual reinforcement was gradually applied. They were allowed to see the visual stimulus half the time during each block and in the second half the screen was turned off, this way they regulated without the image and they continued to be verbally motivated.
The control group received standard-of-care intervention as a benchmark for improvement. This involves daily visits and group artistic and activation activities. A member of the research group came to their room and for 30 minutes chatted with the participant about her day and activities, following up on their participation in groups where they produced guided crafts, attendance at religious services, in case of religious practice, walking with friends in the garden or participating in the weekly video club, where a film was screened and discussed.
To evaluate the dependent variable, the PSQI (Buysse et al., 1989) was used in two applications, before and after the intervention. The PSQI is a questionnaire that provides a global sleep quality score through the evaluation of seven hypothetical components. In the Mexican population, it has a reliability coefficient of 0.78 and significant correlation coefficients of 0.53 to 0.77 between the components and the total sum, consisting of two factors: sleep quality and sleep duration (Jiménez-Genchi et al., 2008).
We used a laptop, a 17” television, a Pro Comp 2 registration system from Thought Technology Ltd, Montreal Canada, controlled by Biograph Infiniti software, an EEG-Z sensor (pre-amplified electroencephalography sensor with built-in impedance control) and reusable cup electrodes made of a gold plate for EEG. The EEG signals were recorded, processed, filtered and represented using the BioGraph Infiniti EEG Software. Pro Comp 2 Infiniti hardware (Thought Technology Ltd; Montreal, Quebec) sent the signal to the computer. The EEG used for feedback was sampled at 256 Hz/sec and sent to the computer by the A/D converter. For the registration, a monopolar electrode connection was used. The impedance was kept below 10KΩ and the artefact rejection thresholds were set to suspend feedback when eye movements, or some motor activity, caused EEG fluctuations. The amplitude values were measured continuously in microvolts (μV), peak to peak, according to the recommendations made in previous reports (Gruzelier et al., 2006).
Result-scoring and statistical analysis were blind. Data analysis was carried out with a Mann-Whitney U test, performed to analyze differences in the PSQI score pre and post between the experimental group and the control group. Because of the sample size, the data does not behave according to the normal curve. To compare the results between before and after treatment in the experimental group and determine if there is a significant difference, the PSQI test was used. In addition, in the physiological measurements, which were only made to the experimental group, variance analysis was carried out for repeated measurements between the power of frequency alpha in four cuts during the experiment (baseline, day 10, day 20 and day 30).
When comparing the results between the two applications of PSQI, no statistically significant differences (z = -1,286, p> 0.05) were found between the participants of the control group and the experimental group.
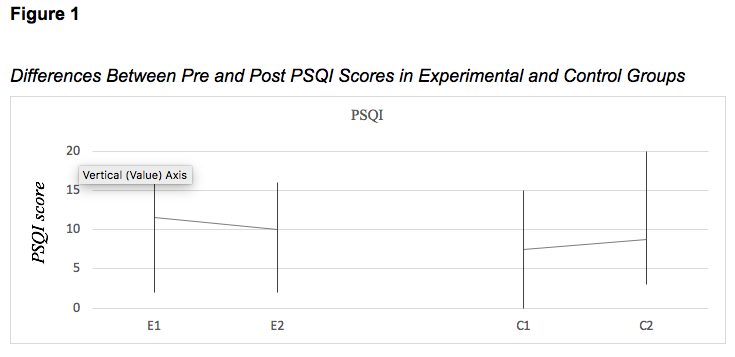
However, when comparing the PSQI scores pre and post in the experimental group, a statistically significant difference was found, using a T-test (t(10)= 5.25, p<.01). Furthermore, a decrease in the test score indicates an improvement in the quality of sleep of the participants.
Statistically significant differences were found [Time: F (0.05,3,54) = 12.5, p <0.0001; Frequency: p> 0.05; Interaction: F (0.05,6,54) = 2,595, p <0.05] with respect to power of frequency alpha obtained from the neurofeedback treatment. These show an increase on day 30 for the baseline.
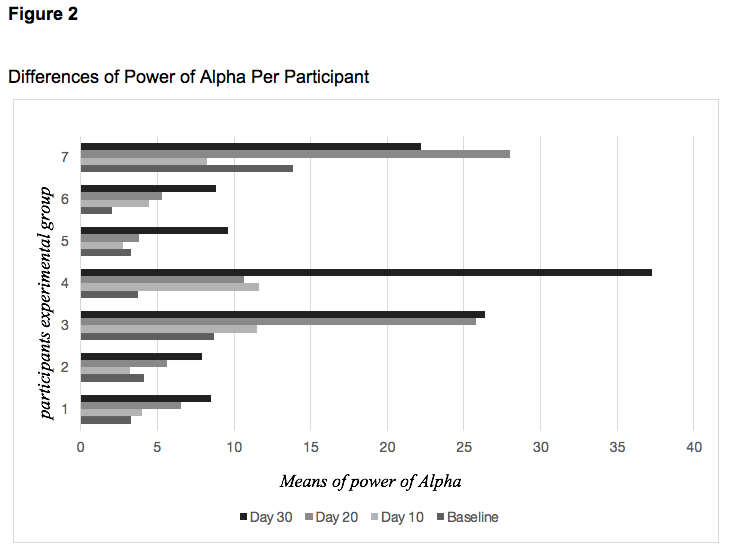
The alpha frequency has been commonly associated with states of relaxation and rest that can help induce and maintain sleep (Knaut et al., 2019), so the results obtained in the physiological measurement of the participants of the experimental group indicated that their biological disposition towards rest increased after neurofeedback training.
The manifestation of a change in alpha frequency between the baseline and 30th session may be attributed to the repetition of training, which favours the change in the electroencephalographic functioning. Repetitions become an essential variable since change does not manifest in the same way in the 10 or 20 sessions cut. To think about the benefit of this, it is interesting to consider that other studies in older adults have shown that enhancing slow oscillation leads to a memory enhancement (Ladenbauer et al., 2016; Westerberg et al., 2015).
Although on the other hand, Schabus et al. (2017) raises the importance of demonstrating long-term benefits beyond the placebo effect in NF-based interventions and found that with less than 20 sessions the desired results were obtained. However, it is important to consider that the participants of this research have an average age of 77.7 years, so the results obtained could be explained because the learning process necessary to achieve the effect of NF may be diminished as an expected effect of ageing. They also argue that the observed effect could be due to the relationship established with the researcher, for which it sought to control this variable in the study by visits with the same duration and interaction that were made daily to the control group. However, it is necessary to methodologically strengthen this intervention by using the NF placebo to determine the actual efficacy, as they put it in their conclusions.
The change in alpha frequency is consistent with the change of PSQI score in the experimental group and previous research data which demonstrated that the increase in SMR (12-15Hz) through neurofeedback induces and increases the appearance of sleep spindles (Hoedlmoser et al., 2008; Sterman, 1996;Zotou et al., 2017) and improves sleep parameters such as decreased sleep onset latency and increased sleep duration (Arns et al., 2014;Cortoos et al., 2010; Hoedlmoser et al., 2008).
Nevertheless, it is necessary to consider that this preliminary experiment has limitations that do not allow for the findings to be generalized. Ros et al. (2019) published a consensus on the reporting and experimental design of clinical and cognitive-behavioural neurofeedback studies (CRED-nf checklist); the main limitation being the lack of placebo treatment for the control group, because the inclusion of a control group that receives sham neurofeedback provides an opportunity to use a double-blind design instead of a single-blind design and would have allowed us to obtain more reliable results in which the influence of unmeasured variables, such as motivation, suggestion or positive relationship with the experimenter, could have been ruled out. We acknowledge this oversight but still believe that this study provides valuable data. Lovato et al. (2019) recommends that research on sleep quality and neurofeedback should be done using multi-site studies utilizing experimental designs where patients are randomized to receive treatment, placebo treatment or no treatment.
The other significant limitation is the small sample size and the lack of following up. We consider that in this first approach to researching older women that stay in nursing homes or institutions that care for the elderly, under the present conditions, it was possible to observe the importance of continuing this line of research, strengthening the methodological conditions of the study. However, no differences were observed in the PSQI scores between groups, with which we can infer that, although there were changes in the participants, it does not necessarily reflect the effect of the neurofeedback intervention. This phenomenon may be attributed to several factors, such as the sex of the participants, being at a nursing home and the nature of participation during the sessions.
Regarding the sex of the participants, a higher incidence of difficulties associated with sleep has been found in women over 65 years old. Ayalon et al. (2004) described female predominance in the manifestation of sleep disturbances in ageing. Some of the factors that may be associated to this are biological events such as decreased REM sleep, increased awakenings, and reduced sleep efficiency, as well as behavioural conditions such as increased number of naps, eating habits, and poor physical activity during the day.
In the same direction, Arber and Bote (2006) found that there is a higher prevalence of sleep-related disorders in people who do not live with their family. People who maintain an adequate family network say they feel protected by these links and have a lower percentage of sleep problems than those who have less intense social support or live alone (Arber & Bote, 2006). In this way, the research concludes that living in a family context provides the elderly with a protective factor against sleep disorders. In the case of the participants of our study, the condition of institutionalization hinders both the perception of a close family network and the implementation of sleep hygiene guidelines since they experience environmental factors like lighting, wake-up time or noise that cannot be controlled by individuals.
Although these results suggest some impact of neurofeedback on increased sleep quality, methodological differences from other studies shall not remain unmentioned. Regarding the modality of participation, it is noteworthy that in the study by Tang et al. (2015) elderly people were motivated to carry out actions aimed at improving the quality of sleep such as using the equipment provided by researchers in their private homes and keeping track of their activity in sleep diaries. In the case of the participants of the present investigation, they were not asked to modify their behaviour or to carry out any proactive action to improve their rest to evaluate the effect of the intervention on the physiological measurement without the risk of bias from attributing the probable changes to conditions already proven in previous investigations.
It is important to point out that the results of this preliminary study encourage us to investigate further. Wexler and Thibault (2018) argue that sometimes research on technology equipment that seeks to positively impact health can make hasty conclusions about its effectiveness, and as researchers we are aware of this.
It is probable that, along with the neurofeedback-based intervention, behavioural sleep-hygiene training should also be implemented, allowing for lasting and observable changes amongst individuals.
Our findings illustrate that the neurofeedback intervention increased alpha frequency scores associated with sleep and rest in the elderly in nursing homes or institutions where they care for the elderly; however, albeit our initial findings are promising, caution is needed in their interpretation. These results should be replicated in larger, more controlled clinical trial settings, which also include a control group of no treatment, to ensure the validity of the results. In future research, a comparison between neurofeedback, mock feedback and a non-intervention condition could help specify the role of neurofeedback and sleep quality amongst elderly individuals.
Qualitative changes in elderly sleeping are the mean frequency of the slow waves, showing a slowing in average frequency in older relative to younger adults. Carrier et al. (2011) describes this effect across most EEG derivations. It is likely that due to the conditions of the participants (sex, distance from the family network, and degree of participation) the effects have not been powerful enough to manifest themselves in the PSQI when the results of the experimental group were compared to the control group, although the experimental group did show a change. Additionally, since an extra hour of sleep during the day reduces the amount of slow-wave sleep during the night (Krauchi et al., 2006), controlling this variable in the groups could help identify both favourable and unfavourable guidelines, which could improve the quality of sleep in elderly adults.
The National Council of Science and Technology (CONACYT; Grant 450753, Intern number 609436) supported this work. The funding sources had no role in the study design; the collection, analysis or interpretation of the data; the writing of the manuscript; or the decision to submit the paper for publication.


