Artículos

Esta obra está bajo una Licencia Creative Commons Atribución-SinDerivar 4.0 Internacional.
Recepción: 21 Abril 2021
Aprobación: 15 Mayo 2021
Publicación: 10 Junio 2021
DOI: https://doi.org/10.5281/zenodo.5651290
Abstract: Background: Febrile neutropenia define as an oral fever of more than 38.3°C or ≥ 38.0°C sustained for more than one hour, with absolute neutrophil count (ANC) should be <500/μL. FN associated with high rate of disease indisposition, mortality, affect chemotherapy price and doses, and leading to chemotherapy course interruptions, as well as affects the chemotherapy potency. Aim of study: This study was aimed to estimate the patients that have cancer and newly diagnosed with febrile neutropenia Patients and methods: A retrospective study that included one hundred and fifty patients already diagnosed with cancer who attended the outpatient clinic of Babylon Cancer Treatment Center, from 1st of January 2020 to 1st of January 2021. Results: From a total of 150 patients, age of patients ranged from 20-85 years, with mean age of 62.3±10.17 years, 63(42%) were males and 87(58%) were females. Breast cancer form 40%, as the most common solid cancers presented with febrile neutropenia, while head and neck cancer form 2%. We found 20% blood culture positive, and E. coli most bacterial isolated (40%), while 8.66% was positive urine culture and E. coli was form 84.6% also most commonly urine bacterial isolated, and only 2.66% sputum culture was positive and S. pneumonia is most common bacterial isolated (50%). 73.34% of patients presented with fever without localizing symptoms, while 12.66% of patients presented with respiratory system related symptoms. We found day 8th after last cycle of chemotherapy was the most day of starting fever (28.66%). Conclusion:FN is a leading cause of cancer related death, early management is required to decrease rate of mortality.
Keywords: febrile neutropenia, presentation, solid tumor.
Resumen: Antecedentes: la neutropenia febril se define como una fiebre oral de más de 38,3°C o ≥38,0°C sostenida durante más de una hora, con un recuento absoluto de neutrófilos (RAN) que debe ser <500/μL. La FN se asocia con una alta tasa de indisposición de la enfermedad, mortalidad, afecta el precio y las dosis de la quimioterapia, y conduce a interrupciones del curso de quimioterapia, así como también afecta la potencia de la quimioterapia. Objetivo del estudio: Este estudio tuvo como objetivo estimar los pacientes que tienen cáncer y recién diagnosticados con neutropenia febril Pacientes y métodos: Un estudio retrospectivo que incluyó a ciento cincuenta pacientes ya diagnosticados con cáncer que asistieron a la clínica ambulatoria del Centro de Tratamiento del Cáncer de Babylon, del 1 de enero de 2020 al 1 de enero de 2021. Resultados: De un total de 150 pacientes, la edad de los pacientes osciló entre 20 y 85 años, con una edad media de 62,3±10,17 años, 63 (42%) eran varones y 87 (58%) eran mujeres. El cáncer de mama forma el 40%, ya que los cánceres sólidos más comunes se presentan con neutropenia febril, mientras que el cáncer de cabeza y cuello forma el 2%. Encontramos un 20% de hemocultivo positivo, y E. coli la mayoría de las bacterias aisladas (40%), mientras que el 8,66% fue un urocultivo positivo y la E. coli fue del 84,6% también más comúnmente aislada de bacterias en la orina, y solo el 2,66% del cultivo de esputo fue positivo. y S. pneumonia es la bacteria aislada más común (50%). El 73,34% de los pacientes presentó fiebre sin síntomas localizadores, mientras que el 12,66% de los pacientes presentó síntomas relacionados con el sistema respiratorio. Descubrimos que el octavo día después del último ciclo de quimioterapia fue el que tuvo más días de inicio de la fiebre (28,66%). Conclusión: La FN es una de las principales causas de muerte relacionada con el cáncer, por lo que se requiere un manejo temprano para disminuir la tasa de mortalidad.
Palabras clave: neutropenia febril, presentación, tumor sólido.
Introduction:
Febrile neutropenia (FN) is define myelo suppressed patients with fever, even though an extensive variety of temperature and neutrophil cutoff have been used to recognize such incidents1,2. FN define as an oral fever of more than 38.3°C or ≥ 38.0°C sustained for more than one hour, with absolute neutrophil count (ANC) should be <500/μL. or <1000/μL with fast decrease <500/μL3,4. Febrile neutropenia associated with high rate of disease indisposition, mortality, affect chemotherapy price and doses, and also leading to chemotherapy course interruptions4,5, as well as affects the chemotherapy potency6,7.
The frequency of FN arising during the first cycle of treatment is around (23%–36%)., although most patients complained from mild events, the frequency of severe complications range (25–30%) and mortality (9–12%)9. While the progressions in helpful care of patients have cancer development, decrease death due to FN from 40% in 1970 to (1-3%) currently10,11. Most local12, national13, and international14-17 guideline groups have suggested that routine primary FN prophylaxis be used for treatments with an FN risk 20 %13.
Prompt empirical therapy with broad-spectrum antibiotics is standard of care and lethality is 10% in adult patients17,18. FN remains the important cause of emergency hospitalization19. Immediately must be sending for full blood count to determine the neutrophil count beside other investigations is essential in guiding early management20,21. When patients at low-risk, may be treated in outpatient with oral empirical antibiotic, while high-risk patients need hospital admission for IV antibiotic medications22. Infections due to Candida species may occur later in the course of neutropenia, particularly as a result of gastrointestinal (GI) mucositis22,23. The NCCN Guidelines Panel advises that fluoroquinolone prophylaxis (levofloxacin is preferred) be considered in patients with an expected duration of neutropenia (ANC˂1000 neutrophils/μL) for more than7 days. This is in agreement with the recommendations of the recent IDSA guidelines for the use of antimicrobial agents in neutropenic patients with cancer24,25. This study was aimed to estimate the patients that have cancer and newly diagnosed with febrile neutropenia to help them and medical staff for proper management and decrease rate of morbidity and mortality.
Patients and methods
A retrospective study that included 150 patients already diagnosed with cancer who attended the outpatient clinic of Babylon Cancer Treatment Center, from 1st of January 2020 to 1st of January 2021. All patients had underlying solid malignancy with absolute neutrophil count ≤0.5 x10/L. plus either: Body temperature ≥38.3 C for a single measure or ≥38.0 C sustained over one hour following chemotherapy.
All patients in this study were sent for the following tests: Complete blood count with WBC differential, Chest X-ray (PA view), GUE and Microbiological tests: Blood culture and sensitivity to identification of blood by using routine usual laboratory methods, other culture and sensitivity such as sputum, urine and stool cultures (when appropriate). For blood culture, 2 blood samples were sent acquired from two different peripheral blood vessels; 5 ml was aspirated for each sample.
Ethical consideration: The study was conducted in accordance with ethical principles that have their origin in the declaration of Helsinki. It was carried out with patients verbal and analytical approval before the sample was taken. The study protocol and the subject information and consent from were reviewed and approved by the local ethics committee.
Statistical analysis done by SPSS 22 frequency and percentage used for categorical data, mean and SD for continuous data.
Results
The age of patients ranged from 20 - 85 years, with mean age of 62.3±10.17 years, age groups are shown in table 1. Of the total 150 patients included in the study 63 (42%) were males and 87 (58%) were females as shown in figure 1.
Table 1: Frequency distributions of age for patients have solid malignancy with febrile neutropenia
Frequency distributions of age for patients have solid malignancy with febrile neutropenia
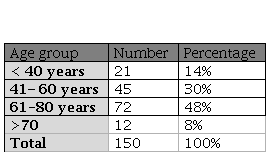
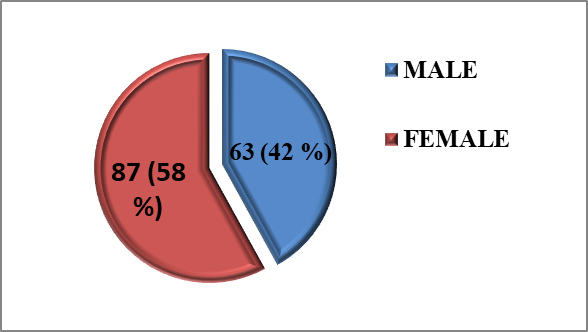
Figure 1:
Frequency distributions of males and females among patients have FN and solid malignancy.
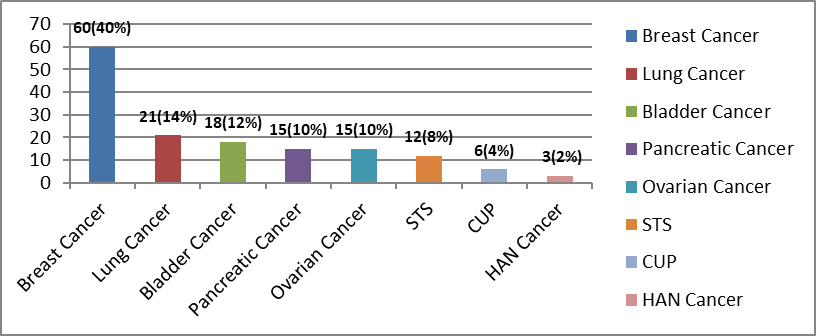
Figure 2:
Frequency distribution of Febrile Neutropenia among solid malignancy.
Frequency distributions of blood, sputum and urine culture results among patients have solid malignancy with febrile neutropenia.
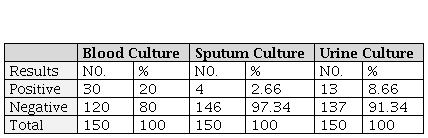
frequency distribution of bacterial type isolated from blood, urine and sputum cultures among patients with solid malignancy and febrile neutropenia.
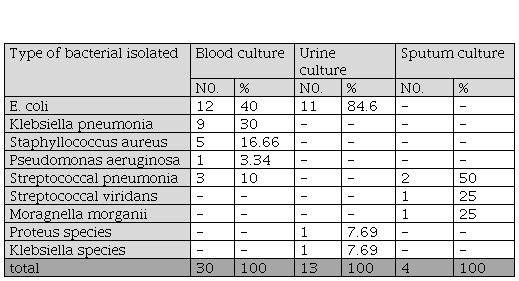
Frequency distribution of system related clinical symptoms in patients with solid malignancy.
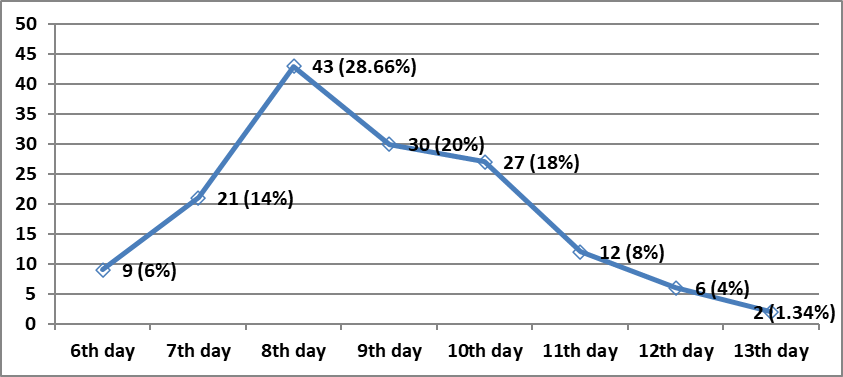
Figure 3
fever onset day after starting last cycle of chemotherapy in patients with febrile neutropenia.
Discussion
In this study among 150 patients with febrile neutropenia following chemotherapy, we found positive blood culture result was 30 (20%), this result was consistent to what had been reported by Hughes WT et al28 who reported that the incidence of positive blood culture reaching to 20% and also this study was consistent with Al-Zubaidy AS et al29 who found that the incidence was 18%, while our result disagree to what had been reported by Abhayakumar SM et al30 that reported in their study a lower incidence of positive blood culture reaching to 12%, this difference might be due to better infection control and different in sample size. Regarding urine culture, in our study we find the urine culture was positive in 13(8.66%) and was negative in 137(91.33%), this study was similar to that of Abhayakumar SM et al30 who reported slightly a lower incidence of positive urine culture and also consistent with Salam et al29 who reported the positive urine culture in 4%. Among the patients of this study, 2.66% were found to be positive sputum culture, this result showed an agreement with that of Al-Zubaidy AS et al29 who found that the results of sputum culture in solid malignancy was (0%). In addition, the result of this study was disagreed with that of Abhayakumar SM et al30 who reported a higher incidence of positive sputum culture reaching to 9.7%. This difference might be due to smaller sample size in our study. Regarding the overall culture positivity results in our study were obtained in 31.32% (20% for blood cultures, 8.66% for urine cultures and 2.66% for sputum cultures) which was comparable to the results obtained by Abhayakumar SM et al30 and Al-Zubaidy AS et al29 were found 25% and 22% overall culture positivity, respectively. We found the predominant bacterial pathogens were gram negative bacteria forming 73.33 % for blood culture and 100% of urine culture and gram positive bacteria forming 26.66% in blood culture, 0% in urine culture and 100% for sputum culture. We noted a higher gram negative infection probably due to absence of use of central venous catheter and low frequency of ciprofloxacin used as prophylactic treatment, this result in agreement to what had been reported by Al-Zubaidy AS et al29 who stated the predominant bacterial pathogens were gram negative bacteria forming 81.81% and gram positive bacteria identified only in 18.18%. In addition, this results were consistent with the Safdar A et al31 who also found that gram negative microbe isolates were most predominant accounted 63.3% while gram positive accounted only 36.7%, we noted in both our study and Safdar A et al31 study that gram negative bacteria were more common than gram positive bacteria but in different percentage while Naurois JD et al32 found that positive cultures were typically gram positive bacteria in 70%, this also this might be related to the use of central venous catheter and use of prophylactic treatment. Regarding the type of bacterial infection, E.coli was the most common type of gram negative bacteria and staphylococcus aureus was the only type of gram positive bacteria identified in blood culture, and also E coli was the most common type of bacteria identified in urine culture , these result agreed with the result in study mentioned above conducted by Abhayakumar SM et al30 who also reported that E. coli was the most common type of gram negative bacteria and staphylococcus aureus was the most common type of gram positive bacteria identified in blood culture also E coli was the only type of bacteria identified in urine culture. Among the patients of this study, 73.34% patients presented clinically, with fever without localizing signs or symptoms and this result is comparable with slightly higher incidence to what has been reported by Oktay Y et al33 who reported higher incidence reaching to 60%, while Rolston KV et al34 who found 40% of patients presented clinically, with fever without localizing signs or symptoms. Also in our study, 26.65% patients presented clinically, with fever and system related clinical symptoms and most commonly 12.66% with respiratory symptoms such as dyspnea or cough followed by 8.66% with gastrointestinal symptoms (as diarrhea) and 5.33% patient with genitourinary symptoms (as dysuria), and these result differed from the result reported by Rolston KV et al23 who found that gastrointestinal symptoms (diarrhea) was the most common symptom present in (29.5%) followed by respiratory symptom (20%) , sinus and ear infection (5%), skin and soft tissue infections (4.5%) and urinary tract symptoms(2%). In our study we found the onset of fever occur most commonly on 8th day followed by 9th day after starting last cycle of chemotherapy that causing febrile neutropenia. This result was consistent with Al-Zubaidy AS et al29who found that the most common day of fever onset occur on 9th day followed by 8th day post chemotherapy cycle.
Conclusion:
FN is a leading cause of cancer related death. High index of suspicion is essential, and early treatment with antibiotics depending on local area susceptibility and sensitivity is needed to decrease rate of morbidity and mortality and hospital stay.
References
1. Seneviratne N, Yeomanson D, Phillips R. Short-course antibiotics for chemotherapy-induced febrile neutropaenia: retrospective cohort study. Arch Dis Child 2020; 105:881–885.
2. Herd F, Bate J, Chisholm J, et al. Variation in practice remains in the UK management of paediatric febrile neutropenia. Arch Dis Child 2016; 101:410–1.
3. Cooksley T, Font C, Scotte F, Escalante C, Johnson L, Anderson R, Rapoport B. Emerging challenges in the evaluation of fever in cancer patients at risk of febrile neutropenia in the era of COVID-19: a MASCC position paper. Support Care Cancer. 2021 Feb;29(2):1129-1138.
4. Ba Y, Shi Y, Jiang W, et al. Current management of chemotherapy-induced neutropenia in adults: key points and new challenges: Committee of Neoplastic Supportive-Care (CONS), China Anti-Cancer Association Committee of Clinical Chemotherapy, China Anti-Cancer Association. Cancer Biol Med. 2020;17(4):896-909.
5. Pettengell R, Schwenkglenks M, Leonard R, Bosly A, Paridaens R, Constenla M, et al. Neutropenia occurrence and predictors of reduced chemotherapy delivery: results from the INC-EU prospective observational European neutropenia study. Support Care Cancer. 2008; 16(11):1299–309.
6. Lyman GH, Michels SL, Reynolds MW, Barron R, Tomic KS, Yu J. Risk of mortality in patients with cancer who experience febrile neutropenia. Cancer. 2010; 116(23):5555–63.
7. Aapro MS, Bohlius J, Cameron DA, Lago LD, Donnelly JP, Kearney N, et al. 2010 update of EORTC guidelines for the use of granulocyte-colony stimulating factor to reduce the incidence of chemotherapy-induced febrile neutropenia in adult patients with lymphoproliferative disorders and solid tumours. Eur J Cancer. 2011; 47(1):8–32.
8. Bayonas AC, FonsecaPJ, Castro EM, et al. SEOM clinical practice guideline: management and prevention of febrile neutropenia in adults with solid tumors (2018). Clinical and Translational Oncology (2019) 21:75–86.
9. Rapoport BL, Aapro M, Paesmans M, van Eeden R, Smit T, Krendyukov A, et al. Febrile neutropenia (FN) occurrence outside of clinical trials: occurrence and predictive factors in adult patients treated with chemotherapy and an expected moderate FN risk. Rationale and design of a real-world prospective, observational, multinational study. BMC Cancer. 2018; 18: 917.
10. Phillips B, Selwood K, Lane SM, et al. Variation in policies for the management of febrile neutropenia in United Kingdom children’s cancer Study Group centres. Arch Dis Child 2007; 92:495–8.
11. Chisholm JC, Dommett R. The evolution towards ambulatory and day-case management of febrile neutropenia. Br J Haematol 2006; 135:3–16.
12. Clemons, M. et al. A multicentre, randomised trial comparing schedules of G-CSF (filgrastim) administration for primary prophylaxis of chemotherapy-induced febrile neutropenia in early stage breast cancer. Annals of Oncology, 2020,31, 951–957.
13. Fernandes R, Mazzarello S, Stober C, Ibrahim MFK, Dudani S, Perdrizet K, Majeed H, Vandermeer L, Shorr R, Hutton B, Fergusson D, Gyawali B, Clemons M. Primary Febrile Neutropenia Prophylaxis for Patients Who Receive FEC-D Chemotherapy for Breast Cancer: A Systematic Review. J Glob Oncol. 2018 Sep;4:1-8.
14. Crawford J, Becker PS, Armitage JO, Blayney DW, Chavez J, Curtin P, Dinner S, Fynan T, Gojo I, Griffiths EA, Hough S, Kloth DD, Kuter DJ, Lyman GH, Mably M, Mukherjee S, Patel S, Perez LE, Poust A, Rampal R, Roy V, Rugo HS, Saad AA, Schwartzberg LS, Shayani S, Talbott M, Vadhan-Raj S, Vasu S, Wadleigh M, Westervelt P, Burns JL, Pluchino L. Myeloid Growth Factors, Version 2.2017, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2017 Dec;15(12):1520-1541.
15. Aapro MS, Bohlius J, Cameron DA, Dal Lago L, Donnelly JP, Kearney N, Lyman GH, Pettengell R, Tjan-Heijnen VC, Walewski J, Weber DC, Zielinski C; European Organisation for Research and Treatment of Cancer. 2010 update of EORTC guidelines for the use of granulocyte-colony stimulating factor to reduce the incidence of chemotherapy-induced febrile neutropenia in adult patients with lymphoproliferative disorders and solid tumours. Eur J Cancer. 2011 Jan;47(1):8-32.
16. Smith TJ, Bohlke K, Lyman GH, Carson KR, Crawford J, Cross SJ, Goldberg JM, Khatcheressian JL, Leighl NB, Perkins CL, Somlo G, Wade JL, Wozniak AJ, Armitage JO; American Society of Clinical Oncology. Recommendations for the Use of WBC Growth Factors: American Society of Clinical Oncology Clinical Practice Guideline Update. J Clin Oncol. 2015 Oct 1;33(28):3199-212.
17. Kuderer NM, Dale DC, Crawford J, Cosler LE, Lyman GH. Mortality, morbidity, and cost associated with febrile neutropenia in adult cancer patients. Cancer. 2006 May 15;106(10):2258-66.
18. Carmona-Bayonas A, Fonseca PJ, Font C, Martínez-García J, Torrella F, Urrego E, et al. Bacteremia in adult cancer patients with apparently stable febrile neutropenia: data from a cohort of 692 consecutive episodes from a single institution. J Community Support Oncol. 2014; 12(9):312–20.
19. Koenig C, Schneider C, Morgan JE, et al. Interventions aiming to reduce time to antibiotics (TTA) in patients with fever and neutropenia during chemotherapy for cancer (FN), a systematic review. Supportive Care in Cancer (2020) 28:2369–2380.
20. Carmona-Bayonas A, Gmez J, Gonzlez-Billalabeitia E, Canteras M, Navarrete A, Gonzvez ML, et al. Prognostic evaluation of febrile neutropenia in apparently stable adult cancer patients. Br J Cancer. 2011; 105(5):612–7.
21. Lymanand GH and Rolston KV. How We Treat Febrile Neutropenia in Patients Receiving Cancer Chemotherapy, Journal of oncology practice, 2010 vol. 6 no. 3149-152.
22. Freifeld AG, Bow EJ, Sepkowitz KA, Boeckh MJ, Ito JI, Mullen CA, Raad II, Rolston KV, Young JA, Wingard JR; Infectious Diseases Society of America. Clinical practice guideline for the use of antimicrobial agents in neutropenic patients with cancer: 2010 update by the infectious diseases society of america. Clin Infect Dis. 2011 Feb 15;52(4):e56-93.
23. Rolston KV. The Infectious Diseases Society of America 2002 guidelines for the use of antimicrobial agents in patients with cancer and neutropenia: salient features and comments. Clin Infect Dis. 2004 Jul 15;39 Suppl 1:S44-8.
24. Freifeld AG, Bow EJ, Sepkowitz KA, et al. Clinical practice guideline for the use of antimicrobial agents in neutropenic patients with cancer: 2010 update by the infectious diseases society of america. Clin InfectDis 2011; 52:e56-93.
25. Crawford J, Caserta C, Roila F, GroupObotEGW. Hematopoietic growth factors applications in high risk patient with febrile neutropenia: ESMO Clinical Practice Guidelines for the. Annals of Oncology.2010; 21:v248–51.
26. Moon H, Choi YJ, Sim SH, et al. Validation of the Clinical Index of Stable Febrile Neutropenia (CISNE) model in febrile neutropenia patients visiting the emergency department. Can it guide emergency physicians to a reasonable decision on outpatient vs. inpatient treatment? PLoS ONE 13(12): e0210019.
27. Klastersky J, Paesmans M, Aoun M, Georgala A, Loizidou A, Lalami Y, et al. Clinical research in febrile neutropenia in cancer patients: Past achievements and perspectives for the future. World Journal of Clinical Infectious Diseases. 2016; 6(3):37.
28. Hughes WT, Armstrong D, Bodey GP, et al. Guidelines for the use of antimicrobial agents in neutropenic patients with cancer". Clin. Infect. Dis. 2002, 34 (6): 730–51.
29. Al-Zubaidy AS, Jumaah SS, Radhi QR. Bacteriological diagnosis of febrile neutropenia in oncology teaching Hospital. The Iraqi postgraduate medical journal. Vol. 18, No. 1, 2019, PP. 37-42.
30. Abhayakumar SM, Govindbabu K, Jayashree RS et al. Management of febrile neutropenia in solid organ malignancies following chemotherapy. Journal of Cancer Research and Therapeutics, Vol. 10, No. 3, July-September, 2014, pp. 540-543.
31. Safdar A, Rodriguez GH, Balakrishnan M et al. Changing trends in etiology of bacteremia in patients with cancer. Eur J Clin Microbiol Infect Dis 2006;25:522-6.
32. de Naurois J, Novitzky-Basso I, Gill MJ, Marti FM, Cullen MH, Roila F; ESMO Guidelines Working Group. Management of febrile neutropenia: ESMO Clinical Practice Guidelines. Ann Oncol. 2010 May;21 Suppl 5:v252-6.
33. OktayY, Filiz G, Hafize Y, et al. Evaluation of febrile neutropenic episodes in adult patients with solid tumors, molecular and clinical oncology, 2016 Mar; 4(3):379–382.
