Aquicultura e Recursos Pesqueiros
Impact of Brazilian fish species at early developmental stages on plankton communities and water chemical parameters
Impacto de espécies de peixes brasileiros em estágios iniciais de desenvolvimento sobre a comunidade planctônica e parâmetros químicos da água
Impact of Brazilian fish species at early developmental stages on plankton communities and water chemical parameters
Acta Scientiarum. Biological Sciences, vol. 38, no. 3, pp. 263-272, 2016
Universidade Estadual de Maringá
Received: 30 May 2016
Accepted: 21 July 2016
Abstract: Effect of larvae of Astyanax altiparanae (AA), Piaractus mesopotamicus (PM), Leporinus obtusidens (LO) and Prochilodus lineatus (PL) on plankton communities and water quality were evaluated. Twenty500 L tanks were used into a completely randomized design with five treatments and four replications, containing 350 individuals of each species per tank. One treatment control had no larvae (CT). Every six days, were collected periphyton and water to determine the concentrations of chlorophyll and total phosphorus. Plankton samples (20 mm mesh net) were taken every three days. Fish affected plankton communities, which showed lower densities of total zooplankton in AA tanks than in LO tanks. Increased density of rotifers and reduced density of copepods were found in AA, PM and PL tanks in relation to the CT. Higher density of testate amoebae was observed in AA, PM and LO tanks. As to biovolume, higher values for total plankton were registered in the CT and LO tanks compared with AA, PM and PL tanks; higher values for rotifers were observed in PL tanks than in the CT. Copepod biovolume was higher in CT and LO tanks. Biovolume of cladoceran and testate amoebae were not affected. Plankton´s chlorophyll concentration was higher in tanks with fish. Higher concentrations of dissolved oxygen were found in AA and PM tanks. It can be concluded that juveniles of these fish species differently affect the plankton community and chemical parameters of the water.
Keywords: Astyanax altiparanae, fish larvae, plankton interaction, Leporinus obtusidens, Piaractus mesopotamicus, Prochilodus lineatus.
Resumo: Avaliou-se o efeito das fases iniciais de desenvolvimento de Astyanax altiparanae (AA), Piaractus mesopotamicus (PM), Leporinus obtusidens (LO) e Prochilodus lineatus (PL) nas comunidades planctônicas e a qualidade da água. Foram utilizados 20 tanques (500 L) em um delineamento inteiramente casualizado com cinco tratamentos e quatro repetições, onde foram estocados 350 indivíduos de cada espécie por tanque. Foi mantido um tratamento sem larvas (CT). A cada seis dias, foram retiradas amostras do perifíton e da água para determinar as concentrações de clorofila e do fósforo total. Foram realizadas coletas de plâncton (rede 20 mm) a cada três dias. Houve efeito dos peixes sobre a comunidade planctônica com menores densidades de zooplâncton total nos tanques com AA do que naqueles com LO. Observou-se aumento das densidades de rotíferos e redução das de copépodes nos tanques com AA, PM e PL em relação ao CT. Observaram-se maiores densidades de tecamebas nos tanques com AA, PM e LO. Observaram-se também valores superiores do biovolume para o plâncton total no CT e LO em comparação a AA, PM e PL, constatando-se maiores índices de rotíferos nos tanques com PL em relação ao CT. O biovolume dos copépodes foi mais elevado nos tanques CT e LO. O biovolume de cladóceros e tecamebas não foi afetado. A concentração de clorofila do plâncton foi maior nos tanques com peixes. Maiores teores de oxigênio dissolvido foram obtidos com AA e PM. Conclui-se que a presença das formas jovens dessas espécies de peixe afeta de forma diferenciada a comunidade planctônica e os parâmetros químicos da água.
Palavras-chave: Astyanax altiparanae, larva de peixe, plâncton, Leporinus obtusidens, Piaractus mesopotamicus, Prochilodus lineatus.
Introduction
Adult and juvenile fish influence planktonic communities in natural environments, reducing
densities and/or the average population size of some organisms and consequently, enabling the population growth of other (Kissic, 1987; Guest,
Drenner, Threlkeld, Martin, & Smith, 1990; Gerking, 1994; Karus, Paaver, Agasild, & Zingel, 2014; Sass et al., 2014). Selective visual predation by fish is an important mechanism structuring zooplankton communities (Keefe, Brewer, & Dodson, 1998), affecting the biomass, species composition and size of organisms (Guest et al., 1990; Böing, Wagner, Voigt, Deppe, & Benndorf, 1998; Milstein, Vandenberg, & Harpaz, 2006). The consumption of large filtering zooplankton reduces the impact of grazing upon phytoplankton, which in turn allow for the increase of its biomass and promote changes in the composition of this community (Milstein, Hepher, & Teltch, 1988; Qin, Madon, & Culver, 1995; Böing et al., 1998; Esteves, 1998; Matheus & Barbiere, 1999; Pegano, Saint-Jean, Arfi, Bouvy, & Guiral, 1999) and consequently increase in values of total chlorophyll (Beklioglu & Moss, 1995; Qin et al., 1995). On the other hand, there is an increased density of small sized zooplankton (Kissic, 1987; Beklioglu & Moss, 1995; Qin et al., 1995). Thus, the effects of fish predation on zooplankton communities are differently expressed in lake ecosystems, which can cause changes in the diversity and density of zooplanktonic species, the composition and biomass of phytoplankton and the physical and chemical conditions of the environment.
In lakes with high densities of zooplanktivorous fish, we observe low density of large zooplankton, such as cladoceran, and the dominance of small organisms, such as rotifers. On the other hand, in lakes with low densities of these fish, we find the opposite (Johannsson & O’Gorman, 1991; Esteves, 1998; Sass et al., 2014). The feeding strategy used by a particular species of fish usually promotes more effective capture of a specific group of zooplankton, favoring the groups not captured, since they will be able to proliferate, due to the reduction of predators, competitors and/or greater availability of food. High abundance of larvae and juveniles of gizzard shad (Dorossoma cepedianum) leads to a reduction in the densities of total zooplankton in mesotrophic environments, which interferes with the recruitment of sport fishes in four reservoirs. During the recruitment of larvae and at high densities of juvenile D. cepedianum, downstream and upstream of four Ohio reservoirs, there is a sharp decrease in density and increase in biomass of total zooplankton, which was associated with the consumption of small zooplankton (Bremigan & Stein, 1999).
Shifts in the dominant zooplankton species lead to changes in the phytoplankton (Watson, Bayne, Devries, & Williams, 2003; Hunt & Matveevb, 2005; Soares & Hayashi, 2005). Lower biomass of phytoplankton in environments with high biomass of Daphnia galeata were observed by Urabe (1990), and Böing et al. (1998) reported lower biovolume of phytoplankton with increasing clearance rate by D. galeata. These authors also observed the increase in the average size of cells and colonies of the phytoplankton, related to the grazing pressure by this cladoceran.
The capture of food-organisms by fish, particularly with respect to early stages of development (larvae and juveniles) depends on a variety of environmental factors, such as light and turbidity (Fermin & Seronay, 1997; Soares & Hayashi, 2005, Rieger & Summerfelt, 1997; Portella et al., 2014), as well as factors related to interaction between predator and prey, such as size, ability to escape, palatability and prey availability, the visual and swimming efficiency of predator and development of sensory system (Nunn, Tewson, & Cowx, 2012; Portella et al., 2014). The interaction between two or more of these factors may affect the development, growth, survival and behavior of juvenile fish (Fermin & Seronay, 1997; Rieger & Summerfelt, 1997; Behr, Neto, Tronco, & Fontana, 1999, Soares & Hayashi, 2005), and the effect may vary depending on the biology of each species, since there are differences in the type of environment needed for each species, varying even for the different stages of development. Notwithstanding, the feeding ecology of young fishes, particularly larvae and 0+ juveniles, is poorly understood compared with older individuals. Indeed, there are a large number of species for which there is no information on their foraging ecology during the larval and juvenile periods. (Nunn et al., 2012).
Given the above, under the hypothesis: the eating habits of different of Brazilian native fish in early developmental stages can change / affect the quality of water and the plankton community of experimental tanks of fish farming, this study aimed to evaluate if the quality of water and the plankton community may be affected in experiments populated by larvae of Brazilian native fish. The specific objectives are: 1) To evaluate the influence of the larvae of species Astyanax altiparanae, Piaractus mesopotamicus, Leporinus obtusidens and Prochilodus lineatus on the water quality of experimental tanks; 2) To evaluate the influence of the larvae of the species A. altiparanae, P. mesopotamicus, Leporinus obtusidens and P. lineatus on the structure and abundance of planktonic communities of experimental tanks.
Material and methods
The experiment was conducted between January and February 2001, for forty days. Five treatments were distributed in a completely randomized design. The larvae used were produced by induced spawning, and stocked in the same tanks for 48-72 hours after hatching. Each tank contained 350 larvae of the four species P. mesopotamicus, A. altiparanae, L. obtusidens and P. lineatus. The stages of development were considering according to the degree of flexion of the terminal region of the notochord and development of the fins, following the terminology proposed by Kendall, Ahlstrom, and Moser (1984), into pre-flexion, flexion, and post-flexion larval stages, and the juvenile stage. so individuals of the four fish species were still in the larval stage during the experimental phase.
Fiber cement tanks (n=20), coated with non-toxic water-based paint, with a capacity of 500-L, were cleaned, exposed to the sun for disinfection during two days, and supplied with water from an artesian well and treated (ratio 1: 1), added with 15 mL of sodium thiosulfate to inactivate chlorine. Seven days before stocking with larvae, each tank was fertilized with 1.5 g of inorganic fertilizer (NPK) and inoculated with water from lakes of the Paraná River basin for the proliferation of planktonic organisms. Each tank also received weekly applications of 0.75 g of NPK.
Tanks were covered with 50% shading net to avoid predators. Macrophytes, Eichhornia crassipes and Pistia stratiotes, were placed at a ratio of 1: 1 so as to cover 50% of the surface of each tank; this area was delimited by a wire tied to the sides of the tank. All tanks of each treatment were provided with wooden supports containing 20 glass slides (2.5 x 7.0 cm), set in the middle of the tanks at 25 cm depth, so as to accompany the development of periphyton community. Every three days, plankton samples were collected by taking five 250mL-subsamples at 30 cm depth at different points inside the tanks, which formed a composite sample from which we removed 250 mL to filter through a 20 mm net, and fixed in a solution of 2% formaldehyde for later counting. At least 30 individuals of each species were measured for length and width, except the spines and antennae, according to Ruttner-Kolisko (1977), in order to determine the biovolume. The identification of planktonic organisms was performed according to Hino and Tundisi (1977), Pontin (1978), Lewis (1979), Needham and Needham (1982), Sendacz and Kubo (1982), Picelli-Vicentim (1987).
Water samples (500 mL) were collected in polyethylene vials, with 0.50 mL sulfuric acid, at each tank (30 cm deep) for the determination of total phosphorus concentration, by use of spectrophotometer, according Golterman, Clymo, and Ohmstad (1978). Other water samples (500-mL) were taken from each tank (30 cm deep) and filtered for the determination of plankton chlorophyll concentration. For determining the periphyton community chlorophyll, both surfaces of one glass slide (12.5 cm2 area) were scraped, using a blade wrapped with aluminum foil and distilled water. Samples for determination of the plankton and periphyton chlorophyll were filtered through fiberglass microfilter (47 nm diameter) with the aid of a vacuum pump. Chlorophyll concentration was determined according to Golterman et al. (1978).
On a daily basis, temperature in the tanks was measured in the morning (8 hours) and afternoon (17 hours) with a mercury thermometer, and every three days, we measured pH (pH meter F1002 Bernauer), conductivity (Conductivity Meter F1000 Bernauer) and dissolved oxygen (meter YSI, model 55-12FT) by means of digital equipment. At the end of the experimental period, we performed the counting and weighing of all fish in each tank and 30 individuals from each experimental unit were individually weighed and measured.
In the end of the experimental period all the fishes were taken from each tank counted and weighed (semi-analytical balance Mars Model Al- 500 precision of 0.01). Were collected 20 fish per tank that were packed in polyethylene vials with 4% formalin for subsequent measurements of the total length from the beginning of the snout to the end of the tail under Wild Heerbrugg M5A Stereo Microscope with millimeter ruler and of total wheight (analytical balance Sartoruis with 0.0001 precision).
The variables analyzed consisted of the data obtained from the analyses of the plankton communities during the experimental period, the values of physical and chemical parameters and chlorophyll of plankton and periphyton. The effect of juveniles of different fish species on the biotic and abiotic factors was tested by a multivariate analysis of variance (MANOVA, p > 0.05) and analysis of variance (ANOVA, p > 0.05) through the protected ANOVA protocol. In case of significant differences, we used a Tukey’s test at 5% probability. Statistical analyses referring to planktonic organisms were run with log transformed data, due to the occurrence of zero values, in the computer software STATISTICA release 7. The computer software STATISTICA release 7 was used to create the artwork for figures.
Results
Mean values of biomass, weight and survival of fish in early development and at the end of the experimental period are shown in Table 1. Water temperature during the experimental period ranged from 22.10 to 30.40°C.

Mean values of length, width and biovolume of principal zooplankton (Testate amoebae, rotifers, copepods, cladocerans) organism found in the tanks with young forms of the four fish species are listed in Table 2.
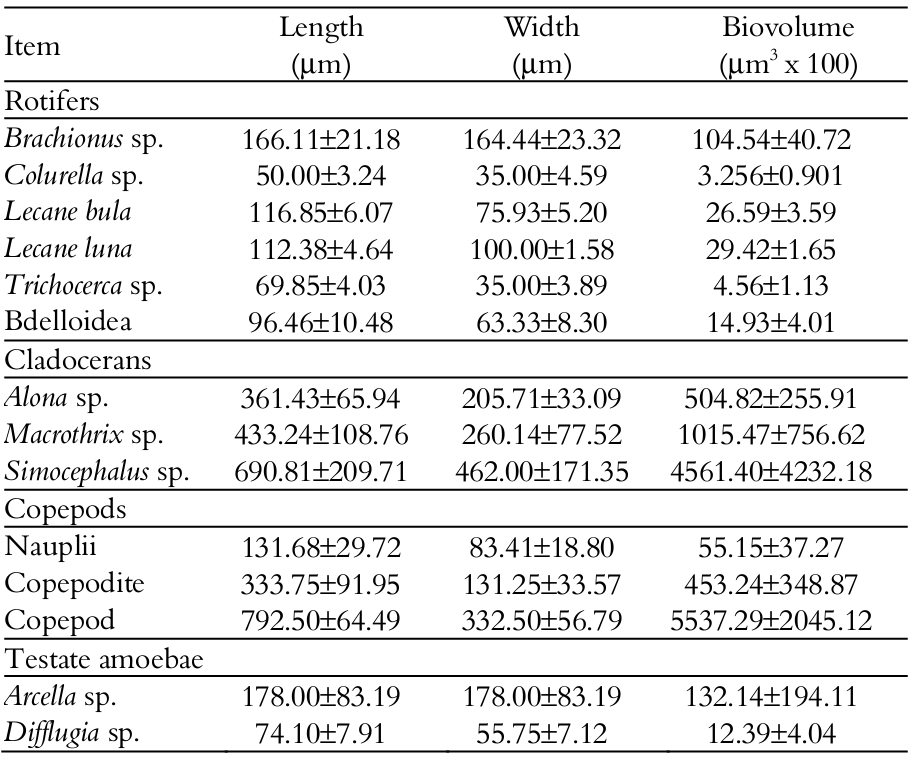
It was observed effect of fish species (MANOVA, F=12.80; p < 0.001) and experimental days (MANOVA, F=25.90; p < 0.001), but no interaction between treatment and days (MANOVA, F=1.08; p > 0.280) on the mean density values of zooplankton organisms. ANOVA evidenced effect of species (F = 12.8, p < 0.001), days (F = 25.9; p < 0.001) and planktonic organisms (F = 282.8, p < 0.001), without interaction between these three factors (F = 1.08, p < 0.280). Higher values of total zooplankton density (Tukey’s test; p < 0.05) were found in tanks with L. obtusidens than with A. altiparanae, but both did not differ from the other treatments. Lower values of rotifer density (Tukey’s test; p < 0.05) were verified in the control tank than in tanks with fish. Considering cladocerans, there was no effect (ANOVA, p > 0.05) of treatments (Figure 1) on the density of these micro-crustaceans.
The highest densities of copepods were observed in the control tank and L. obtusidens tanks (Tukey’s test, p < 0.05), with the lowest densities found in A. altiparanae tanks, different from those in P. mesopotamicus and P. lineatus tanks (Figure 1a). With regard to testate amoebae, higher densities were verified in L. obtusidens tanks than in control tank and P. lineatus tanks, however, the values in L. obtusidens tanks were not different from tanks with A. altiparanae and P. mesopotamicus (Tukey’s test, p < 0.05). The density of these organisms in the control was lower than that obtained with A. altiparanae, P. mesopotamicus and L. obtusidens, though not differing from P. lineatus.
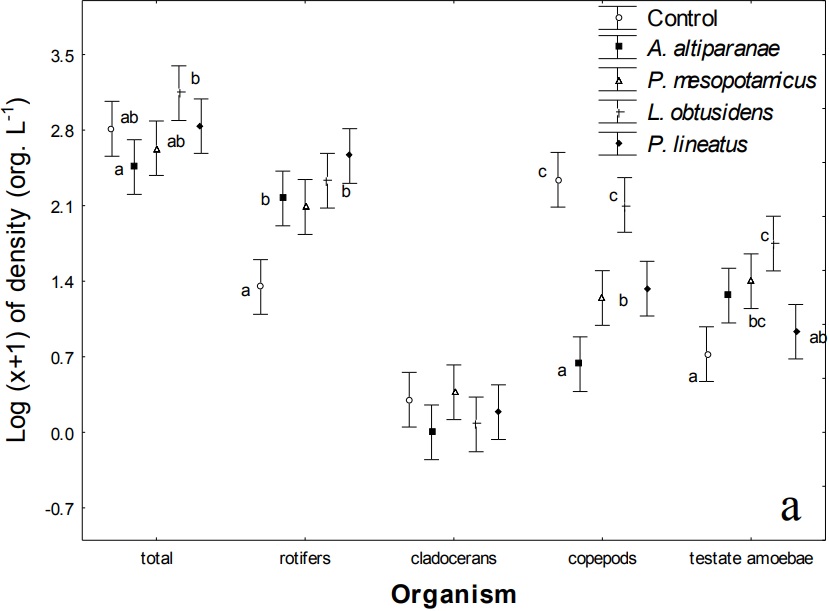
Figure 1
Density of the zooplankton groups (a) and dissolved oxygen (mg L-1), pH and electrical conductivity (mS cm1) (b) in the control tank and tanks stocked with larvae of Astyanax altiparanae, Piaractus mesopotamicus, Leporinus obtusidens and Prochilodus lineatus. Bars within a group followed by the same letter are not significantly different (Tukey’s test; p < 0.05)
Figure 1 (Cont.)
Density of the zooplankton groups (a) and dissolved oxygen (mg L-1), pH and electrical conductivity (mS cm1) (b) in the control tank and tanks stocked with larvae of Astyanax altiparanae, Piaractus mesopotamicus, Leporinus obtusidens and Prochilodus lineatus. Bars within a group followed by the same letter are not significantly different (Tukey’s test; p < 0.05).
Mean biovolume values of zooplankton organisms were affected by treatments (MANOVA, F=23.9, p < 0.000) and days (MANOVA, F=21.2, p < 0.000), but there was no interaction between these parameters (MANOVA, F=1.0, p > 0.348). In relation to the total zooplankton biovolume, higher values were registered in the control and in L. obtusidens tanks, which are higher than those observed in the tanks with A. altiparanae, P. mesopotamicus and P. lineatus (Tukey’s test; p < 0.05). We observed lower values than those obtained with A. altiparanae than with P. lineatus, but both were not different (ANOVA, p > 0.05) from that observed for P. mesopotamicus. The rotifer biovolume values were higher in P. lineatus tanks compared with the control (Tukey’s test; p < 0.05), and the values of both treatments were not different from that obtained for altiparanae, P. mesopotamicus and L. obtusidens (Figure 2). No effects of treatments were detected on the biovolume of cladocerans and testate amoebae (Tukey’s test; p < 0.05).
Higher biovolume of copepods was found in the control tank and in tanks stocked with L. obtusidens (Tukey’s test, p < 0.05). Also, greater biovolume of these microcrustaceans was observed in P. lineatus tanks, compared with tanks with A. altiparanae, and these were not different from that obtained for P. mesopotamicus tanks.
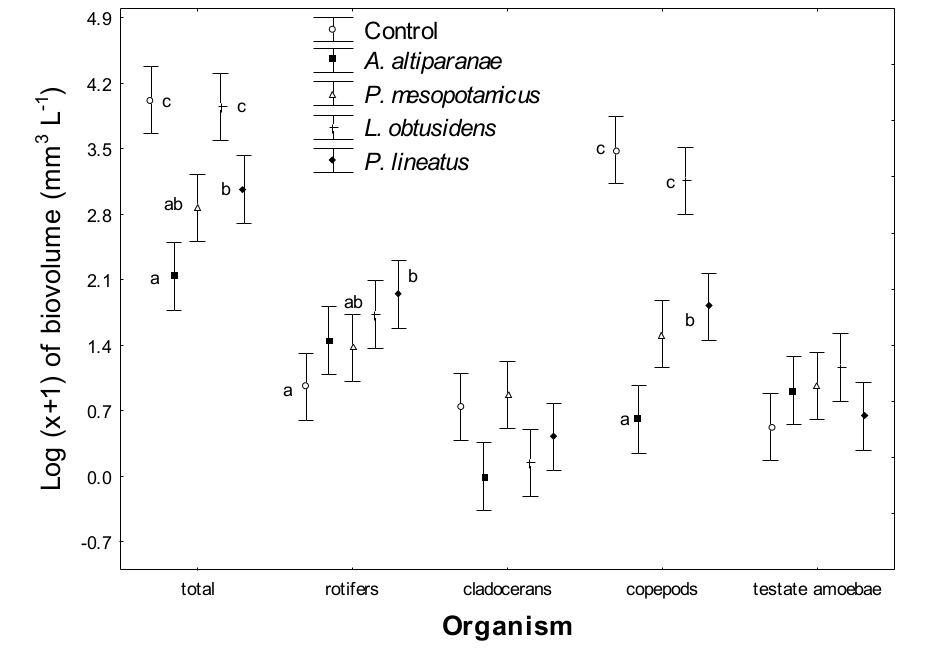
Figure 2
Biovolume of zooplankton groups in the control tank and tanks stocked with larvae of Astyanax altiparanae, Piaractus mesopotamicus, Leporinus obtusidens and Prochilodus lineatus. Bars followed by the same letter in the same group are not significantly different (Tukey’s test, p < 0.05)
Mean biovolume values of planktonic organisms during the experimental period are illustrated in Figure 3. There was the predominance of copepods in the control tank and in L. obtusidens tank. The biovolume of copepods in the control was higher (ANOVA, F=30.0; Tukey’s test, p < 0.05) than in tanks with A. altiparanae in all samplings from the 15th day and higher than those obtained in tanks with P. mesopotamicus from the 15th through the 24th day. Biovolume of copepods in L. obtusidens tanks was higher than in tanks stocked with A. altiparanae and P. mesopotamicus on the days 15, 18 and 27, and higher than in those of A. altiparanae on the days 30 and 33. The values of this attribute in P. lineatus tanks, except for the 33rd day when it was lower than in the control, were not different from the other treatments. As for rotifers, a lower biovolume was found in the control, compared with L. obtusidens tanks on the 33rd day (ANOVA, F=5.462; Tukey’s test, p < 0.05), without differences between treatments in the other collections.
We observed higher values of biovolume of cladocerans (ANOVA, F = 3.716, Tukey test, p < 0.05) in tanks with P. mesopotamicus on the days 12 and 15 and higher values of biovolume of testate amoebae in tanks with fish of this species (ANOVA, F = 3.456, Tukey’s test; p < 0.05) at 24th and 27th days compared with the other treatments.
Regarding the values of dissolved oxygen, pH and electrical conductivity (Table 3 and Figure 1b), we observed effects of treatments (MANOVA, F = 15.0, p < 0.001) and days (MANOVA, F = 41.0; p < 0.001) but no interaction between these factors (MANOVA, F = 1.0; p > 0.813). pH and electrical conductivity were not affected by treatments. However, there were differences in the values of dissolved oxygen in the tanks with A. altiparanae and L. obtusidens, with values superior to those observed in the tanks with P. mesopotamicus, P. lineatus and in the control (Tukey’s test, p < 0.05).
Values of dissolved oxygen, pH and electrical conductivity showed variations during the study period (Figure 4) in all treatments, but for pH and conductivity, there were no differences between the treatments in the same collection (ANOVA, p > 0.05). Higher values of dissolved oxygen were found in the tanks with A. altiparanae and L. obtusidens, in relation to the other treatments on the 15th day (Tukey’s test, p < 0.05). Also, lower values of this parameter were observed in the control and P. lineatus tanks on the days 21 and 24, and lower values in A. altiparanae and P. mesopotamicus tanks on the 36th day (Tukey’s test, p < 0.05).
The plankton chlorophyll concentration in the total study period (Table 3 and Figure 5) was affected by the different treatments (ANOVA, F = 4.50, p > 0.02), with the L. obtusidens tanks presenting higher values than P. lineatus tanks and control (Tukey test; p < 0.05). Values of plankton chlorophyll in tanks with L. obtusidens and P. lineatus were not different from those observed in tanks with P. mesopotamicus and A. altiparanae, however, the value of this variable in the control was inferior to all the treatments with fish (Tukey test; p < 0.05). In turn, the periphyton chlorophyll concentration in this period was influenced by different treatments; higher values were found in tanks with P. lineatus and L. obtusidens compared with the control tank, but the concentrations of periphyton chlorophyll in tanks of these three treatments were not significantly different from those observed in tanks stocked with A. altiparanae and P. mesopotamicus. As to the plankton chlorophyll, higher values were verified in L. obtusidens tanks in relation to the control, and both showed values similar to the other treatments. Values of total phosphorus were not affected by the treatments analyzed (Figure 5). Regarding the values of plankton and periphyton chlorophyll and total phosphorus throughout the experimental period (Figure 6), there were no differences in the values of total phosphorus and periphyton chlorophyll (ANOVA, p > 0.05). The values of plankton chlorophyll showed variations over time, mainly in the control tank and P. mesopotamicus tanks. Still, no differences between treatments were observed on the same date.
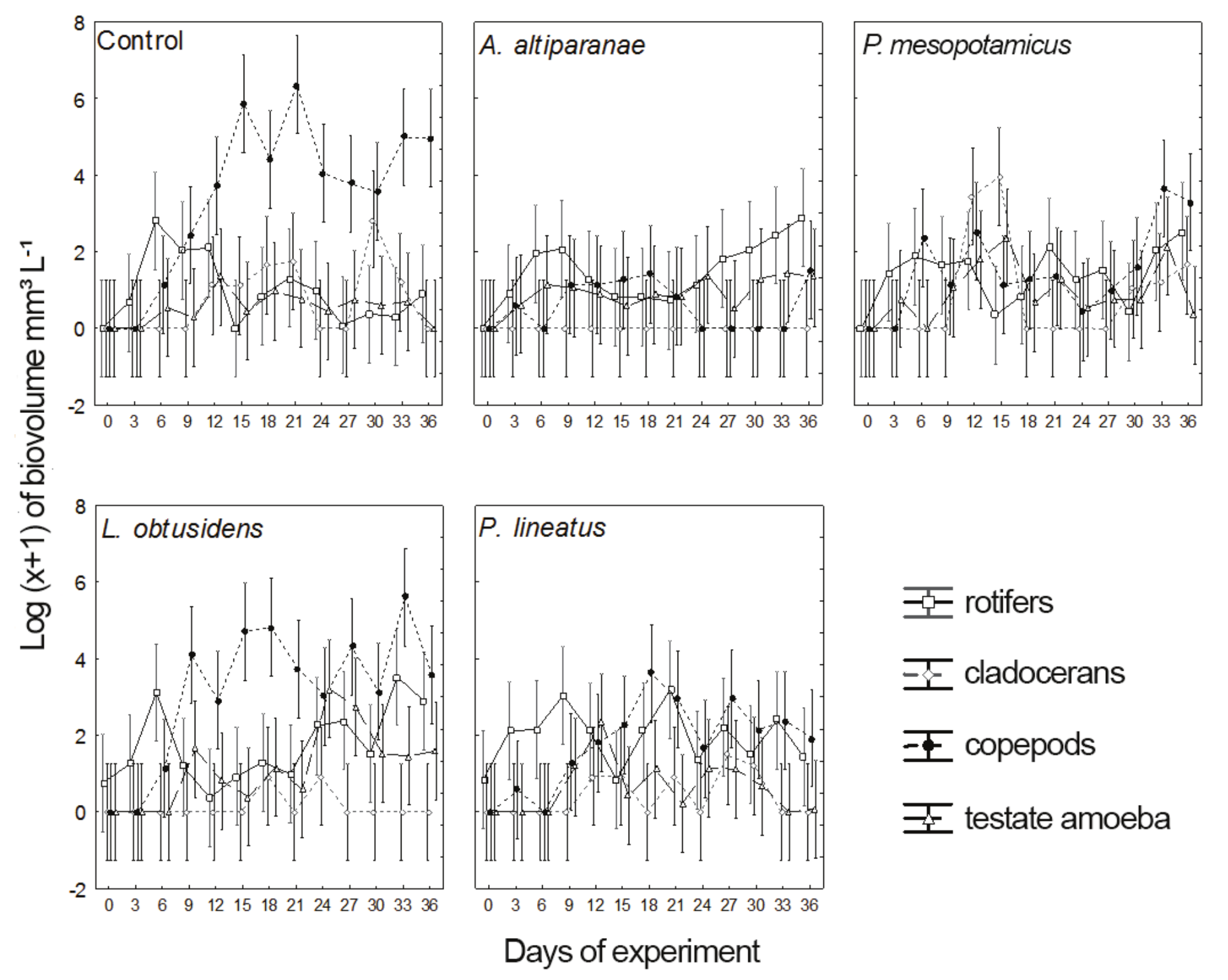
Figure 3
Biovolume of zooplankton groups (rotifers, copepods, cladocerans and testate amoebae) over time in the control tank and tanks stocked with larvae of Astyanax altiparanae, Piaractus mesopotamicus, Leporinus obtusidens and Prochilodus lineatus
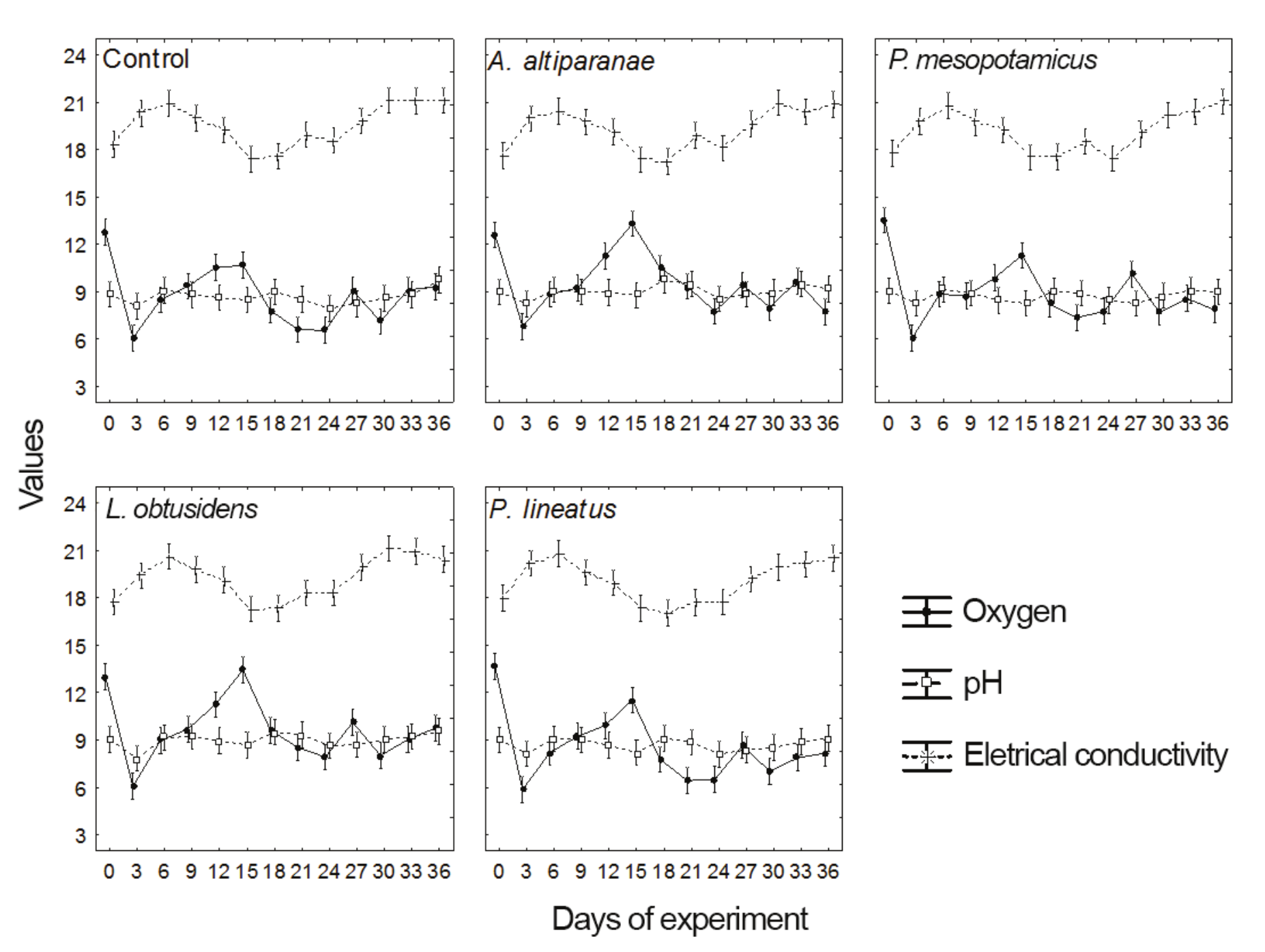
Figure 4
Oxygen (mg L-1), pH and electrical conductivity (mS cm-1) in the control tank and tanks with larvae of Astyanax altiparanae, Piaractus mesopotamicus, Leporinus obtusidens and Prochilodus lineatus along the experimental period. Bars followed by the same letter are not significantly different (Tukey’s test; p < 0.05)
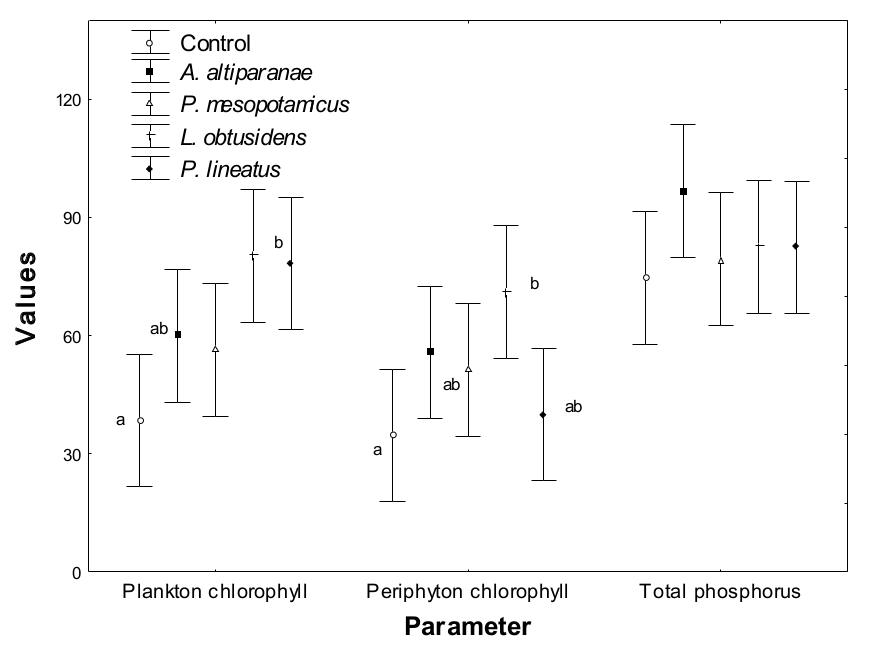
Figure 5
eriphyton (mg cm-1) and plankton chlorophyll (mg L-1) and phosphorus (mg L-1) in the control tank and tanks with larvae of Astyanax altiparanae, Piaractus mesopotamicus, Leporinus obtusidens and Prochilodus lineatus. Bars followed by the same letter are not significantly different (Tukey’s test, p < 0.05)
Discussion
The individuals in early development stages of the four fish species, in general, affected the composition of the plankton community and also the total plankton, both in terms of density and biovolume. Lower densities of rotifers in the control tank, compared with tanks with fish, and lower values of biovolume of this same group, compared with tanks with L. obtusidens and P. lineatus, may be related to the impact of fish on larger zooplankton (cladoceran and copepods), thus reducing the competition for food. This result corroborates whith argued that juvenile fish affect plankton, reducing the densities of some organisms and consequently, enabling the population growth of other species, altering the zooplankton structure (Gerking, 1994, Qin et al., 1995; Keefe et al., 1998; Böing et al., 1998; SASS et al., 2014). An association between consumption of larger sized zooplankton organisms with reduced grazing on phytoplankton and consequent increase in biovolume of this community has been described by several authors (Matheus & Barbiere, 1999; Pegano et al., 1999; Hunt & Matveevb, 2005).
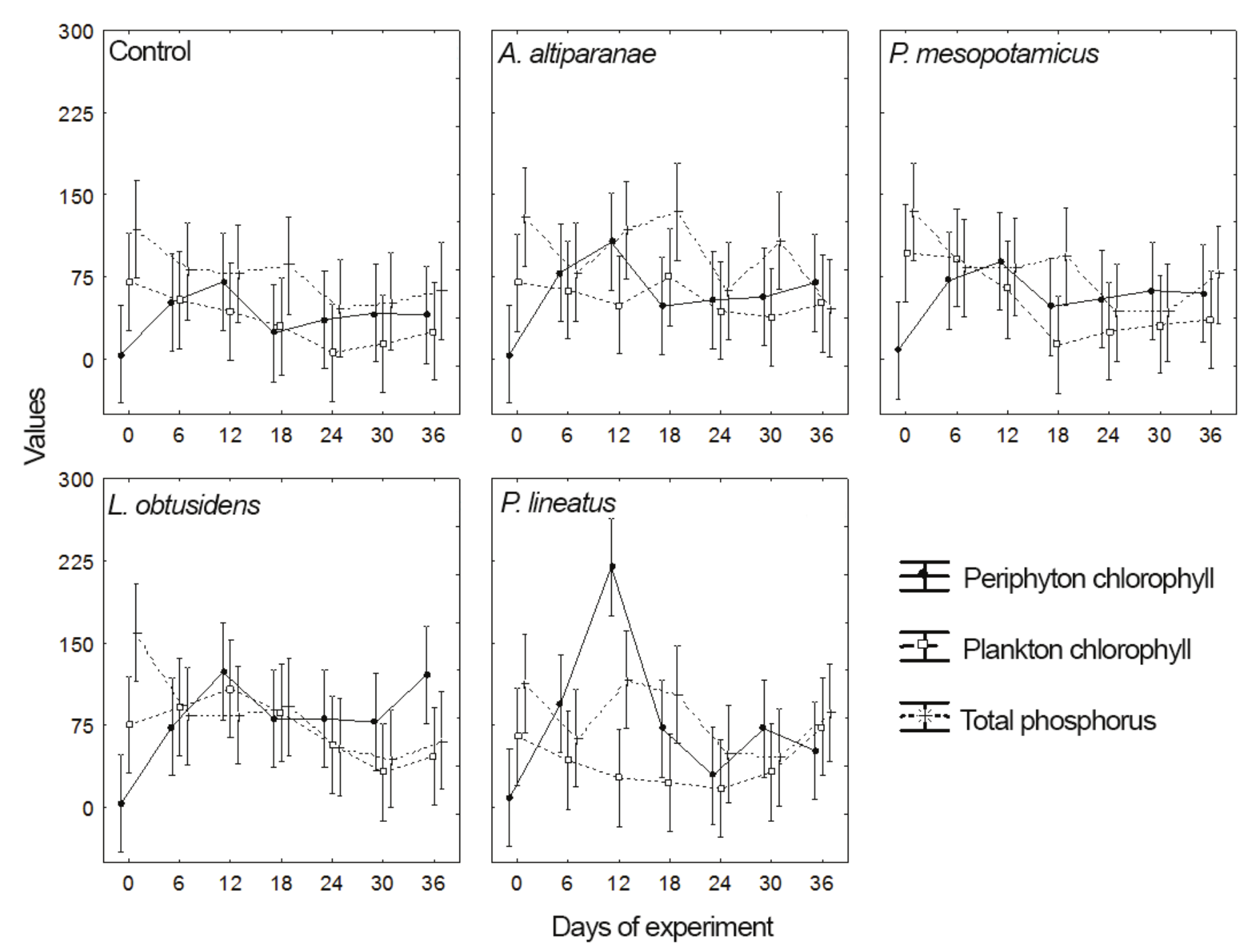
Figure 6
Periphyton chlorophyll (mg cm-1), plankton chlorophyll (mg L-1) and total phosphorus (mg L-1) in the control tank and tanks stocked with Astyanax altiparanae, Piaractus mesopotamicus, Leporinus obtusidens and Prochilodus lineatus, over the experimental period
The effect of the larvae of different fish species on the assemblage of copepods was not observed by Guest et al. (1990), which differs from the results obtained here in, which may be related to larval diet in both studies. On the other hand, these authors observed effects of larvae of different fish species on cladocerans, different from that found in the present study, in which the density and biovolume of cladocerans was not different between tanks with different species and even the control tank.
Moreover, the relationship of density and biovolume of total zooplankton with the consumption of algae is evidenced in this work, given the lower values of plankton chlorophyll in control and L. obtusidens tanks, which contained higher densities and biovolume of total zooplankton, and where it must have occurred higher consumption of algae by zooplankton organisms, thereby reducing the plankton chlorophyll concentration.
The highest values of plankton chlorophyll in tanks stocked with A. altiparanae, P. mesopotamicus and P. lineatus, compared with the control, are probably related to the lower densities of copepods in these tanks. Likewise, Beklioglu & Moss (1995) and Sarvala, Helminem, Saarikari, Salonem, & Vuorio (1998) observed that the decline in populations of large zooplankton organisms leads to higher values of chlorophyll, due to the lower impact of grazing. In this way, predation by fish determines the abundance of herbivorous zooplankton, which regulates phytoplankton biomass (Sarvala et al., 1998).
Besides, lower densities of copepods in the tanks with A. altiparanae and P. mesopotamicus, compared with control and L. obtusidens tanks, along the experimental period, and may be related to the consumption by fish larvae as they grow. According to Soares & Hayshi (2005), some species start to consume larger organisms as they grow, while other consume smaller items with algae (diatoms as the main item) and a more varied diet (rotifers, ostracods and algae), respectively.
Studies on the relationships of larvae and juveniles of Dorossoma cepedianum with zooplankton and recruitment of sport fish in four reservoirs by Bremigan and Stein (1999) and with culture of Stizostedion vitreum by Qin et al., (1995) reported that the high abundance of juveniles of these fish reduces the densities of total zooplankton. This was also observed in this study with juveniles of A. altiparanae, P. mesopotamicus and P. lineatus.
The highest values of plankton chlorophyll in tanks with fish and dissolved oxygen in the tanks with A. altiparanae and L. obtusidens indicate that the impact of larvae on zooplankton promotes changes in these factors. Guest et al. (1990) observed changes in Secchi depth in tanks with different fish species. These results are similar to those of Starling & Rocha (1990) and Watson et al. (2003) who observed an increase of algae in tanks with zooplanktivorous fish in relation to the control. Considering the total phosphorus, Guest et al. (1990) found no differences in tanks with different fish species.
This study showed that when a group of organisms was suppressed by juveniles of a certain fish species, others stood out, which was more evident for rotifers and copepods. This result differs from that observed by Guest et al. (1990), in which there was a marked reduction of all zooplankton groups after the breeding period in tanks with Promoxis annularis alone or associated with D. cepadium. Although density values of total zooplankton of the control tank was not different from the values observed for tanks with fish, the biovolume values of the total zooplankton were higher than those in tanks with A. altiparanae, P. mesopotamicus and P. lineatus, which indicates that the zooplankton in the control tank was formed by larger organisms than other treatments. Predation of larger organisms by fish must have led to this change in the community.
Conclusion
The larvae of A. altiparanae, P. mesopotamicus, L. obtusidens and P. lineatus affect the zooplankton community, altering the composition as to the proportions of the comprising groups, as well as to the total values, with a reduction of copepods in the presence of larvae of A. altiparanae, P. mesopotamicus and P. lineatus and increase in rotifers with L. obtusidens, all occurring as a result of abiotic factors. Thus indicating that there are differences in function of species with different feeding habits in influence on communities of planktonic organisms.
References
Behr, E. R., Neto, J. R., Tronco, A. P., & Fontana, A. P. (1999). Influência de diferentes níveis de luminosidade sobre o desempenho de larvas de jundiá (Rhamdia quelen) (Quoy e Gaimard, 1824) (Pisces Pimelodidae). Acta Scientiarum. Biological Science, 21(2), 325-330.
Beklioglu, M., & Moss, B. (1995). The impact of pH on the interactions among phytoplankton algae, zooplankton and perch (Perca fluviatilis) in shalow, fertile lake. Freshwater Biology, 33(3), 97-509.
Böing, W., Wagner, A., Voigt, H., Deppe, T., & Benndorf, J. (1998). Phytoplankton responses to grazing by Daphnia galeata in the biomanipulated Bautzen Reservoir. Hydrobiologia, 389(1), 101-114.
Bremigan, M., & Stein R. A. (1999). Larval gizzard shad success, juvenile effects, and reservoir productivity: toward a framework for multi-system management. Transactions American Fisheries Society, 128(6), 1106-1124.
Esteves, F. A. (1998) Fundamentos de limnologia (2a ed.). Rio de Janeiro, RJ: Interciência/ Finep.
Fermin, A. C., & Seronay, G. A. (1997). Effects of different illumination levels on zooplankton abundance, feeding periodicity, growth and survival of Asian sea bass, Lates calcarifer (Bloch), fry in illuminate floating nursery cages. Aquaculture, 157(3-4), 227-237.
Gerking, S. D. (1994). Larval feeding. In S. D. Gerking (Ed.), Feeding Ecology of fish (p. 139-170). San Diego, CA: Academic Press.
Golterman, H. L., Clymo, R. S., & Ohmstad, M. A. M. (1978). Methods for physical and chemical analysis of fresh waters. Oxford, UK: Blackwell Scientific.
Guest, W. C., Drenner, R. W., Threlkeld, S. T., Martin, F. D., & Smith, J. D. (1990). Effects of gizzard shad and threadfin shad on zooplankton and young-of-year white crappie production. Transactions American Fisheries Society, 119(3), 529-536.
Hino, K., & Tundisi, J. G. (1977). Atlas de algas da Represa do Broa. São Carlos, SP: UFSCar.
Hunt, R. J., & Matveevb, V. F. (2005). The effects of nutrients and zooplankton community structure on phytoplankton growth in a subtropical Australian reservoir: an enclosure study. Limnologica, 35(1-2), 90-101.
Johannsson, O. E., & O’Gorman, R. (1991). Roles of predation, food, and temperature in structuring the epilimnetic zooplankton populations in lake Ontario, 1981-1986. Transactions American Fisheries Society, 120(2), 193-208.
Karus, K., Paaver, T., Agasild, H., & Zingel, P. (2014). The effects of predation by planktivorous juvenile fish on the microbial food web. European Journal of Protistology, 50(2), 109-121.
Keef, T. C. O., Brewer, M. C., & Dodson, S. I. (1998). Swimming behavior of Daphnia: its role in determining predation risk. Journal of Plankton Research, 20(5), 973-984.
Kendall Jr., A. W., Ahlstrom, E. H., & Moser, H. G. (1984). Early life history stages of fishes and their characters. In H. G. Moser, W. J. Richards, D. M. Cohen, M. P. Fahay, A. W. Kendall Jr., & S. L. Richardson (Eds.), Ontogeny and systematics of fishes: based on International Symposium dedicated to the memory of Elbert Halvor Ahlstrom (Special publication/American Society of Ichthyologists and Herpetologists, 1, p. 11-22). Lawrence, KS: American Society of Ichthyologists and Herpetologists.
Kissick, L. A. (1987). Prey selectivity and feeding periodicity of logperch larvae in Acton Lake, Ohio. Environmental Biology of Fishes, 20(2),155-160.
Lewis Jr., W. M. (1979). Zooplankton community analysis: studies on a tropical system. New York, NY: Springer Verlag.
Matheus, C. E., & Barbieri, G. (1999). Interações entre os peixes e as comunidades fito e zooplanctônicas em tanques de piscicultura: bases teóricas para o manejo. Boletim Técnico do Instituto Pesca, 27(1), 1-13.
Milstein, A., Hepher, B., & Teltch, B. (1988). The effect of fish species combination in fish ponds on plankton composition. Aquaculture Fish Management, 19(2), 127-137.
Milstein, A., Valdenberg, A., & Harpaz, S. (2006). Fish larvae: zooplankton relationships in microcosm simulations of earthen nursery ponds. II. Brackish water system. Aquaculture International, 14(5), 431-442.
Needham, P. R., & Needham, J. G. (1982). Guía para el estudio de los seres vivos de las aguas dulces. Barcelona, ES: Editorial Reverté.
Nunn, A. D., Tewson, L. H., & Cowx, I. G. (2012). The foraging ecology of larval and juvenile fishes. Reviews in Fish Biology and Fisheries, 22(2), 377-408.
Pegano, M., Saint-Jean, L., Arfi, R., Bouvy, M., & Guiral, D. (1999). Zooplankton food and grazing impact in eutrophic brackish-water tropical pond (Côte d’Ivoire, West Africa). Hydrobiologia, 390(1), 83-98.
Picelli-Vicentim, M. M. (1987). Chlorococcales planctônicas do Parque Regional do Iguaçu, Curitiba, Estado do Paraná. Revista Brasileira de Biologia, 47, 57-85.
Pontin, R. M. (1978). A key to the freshwater planktonic and semi-planktonic rotifers of the British Isles (Scientifical Publications, 38). Cumbria, UK: Freshwater Biological Association.
Portella, M. C., Jomori, R. K., Leitão, N. J., Menossi, O. C. C., Freitas, T. M., Kojima, J. T., Lopes, T. S., ... & Carneiro, D. J. (2014). Larval development of indigenous South American freshwater fish species, with particular reference to pacu (Piaractus mesopotamicus): a review. Aquaculture, 432, 402-417.
Qin, J., Madon, S. P., & Culver, D. A. (1995). Effect of larval walleye (Stizostedion vitreum) and fertilization on the plankton community: implications for larval fish culture. Aquaculture, 130(1), 51-65.
Rieger, P. W., & Summerfelt, R. C. (1997). The influence of turbidity on larval walleye, Stizostedion vitreaum, behavior and development in tank culture. Aquaculture, 159(1-2), 19-32.
Ruttner-Kolisko, A. (1977). Suggestions for biomass calculation of plankton rotifers. Archiv für Hydrobiologie–Beiheft Ergebnisse der Limnologie, 8(1), 71-76.
Sarvala, J., Helminem, H., Saarikari, V., Salonem, S., & Vuorio, K. (1998). Relations between planktivorous fish abundance, zooplankton and phytoplankton in three lakes of differing productivity. Hydrobiologia, 363(1), 81-95.
Sass, G. G., Hinz, C., Erickson, A. C., Mcclelland, N. N., Mcclelland, M. A., & Epifanio, J. M. (2014). Invasive bighead and silver carp effects on zooplankton communities in the Illinois River, Illinois, USA. Journal of Great Lakes Research, 40(4), 911-921.
Sendacz, S., & Kubo, E. (1982). Copepoda (Calanoida e Cyclopoida) de reservatórios do Estado de São Paulo. Boletim do Instituto de Pesca, 9(1), 51-89.
Soares, C. M., & Hayash, C. (2005). Interactions of freshwater fish fry with the plankton community. Boletim do Instituto de Pesca, 31(2), 175-187.
Starlig, F. L. R., & Rocha, A. J. N. (1990). Experimental study of the impact of planktivorous fishes on plankton community and eutrophication of a tropical Brazilian reservoir. Hydrobiologia, 200/201(1), 581-591.
Urabe, J. (1990). Stable horizontal variation in the zooplankton community structure of a reservoir maintained by predation and competition. Limnology and Oceanography, 35(8), 1703-1717.
Watson, D. L., Bayne, D. R., Devries, D. R., & Williams, J. (2003). Influence of gizzard shad on phytoplankton size and primary productivity in mesocosms and earthen ponds in the southeastern U.S. Hydrobiologia, 495(1), 17-32.
Author notes
cmsoares.miro@gmail.com