Morfofisiologia Vegetal
Flooding avoidance Triplaris gardneriana Wedd. (Polygonaceae): growth and morpho-anatomical aspects
Evitação do alagamento em Triplaris gardneriana Wedd. (Polygonaceae): crescimento e aspectos morfo-anatômicos
Flooding avoidance Triplaris gardneriana Wedd. (Polygonaceae): growth and morpho-anatomical aspects
Acta Scientiarum. Biological Sciences, vol. 38, no. 3, pp. 341-346, 2016
Universidade Estadual de Maringá
Received: 08 March 2016
Accepted: 23 June 2016
Abstract: The aim of this study was to analyze the effect of flooding in Triplaris gardneriana Wedd, cultivated in drained soil (control) and in flooded condition. The experiment was developed in a greenhouse, using plants with 90 days after the emergency. The response to treatment was evaluated at 0, 30, 60 and 90 days. Growth measurements were made, such as biomass allocation, relative growth rate (RGR). Adventitious roots were not measured only observed, as well as the development of hypertrophied lenticels. The RGR was continuously reduced along the 90 days in flooding conditions for the roots, stem and leaves, compared to control. The flooding of the substrate caused alterations such as: increasing of the cortex width and diameter of the central cylinder of root and increasing the diameter of the vessel element of the root and stem. Results show that T. gardneriana remains under stress when submitted to flooding. Therefore, the production of structures as lenticels, aerenchyma and adventitious roots, structures related to the avoidance of this type of stress, were key factors for the maintenance and survival of T. gardneriana.
Keywords: Pantanal, hypoxia, RGR, flooding.
Resumo: O objetivo deste trabalho foi analisar o efeito do alagamento em Triplaris gardneriana Wedd, cultivadas em condições de solo drenado (controle) e solo alagado. O experimento foi desenvolvido em casa de vegetação, utilizando-se plantas com 90 dias após a emergência. As respostas ao tratamento foram avaliadas para os períodos de 0, 30, 60 e 90 dias. Foram feitas medidas de crescimento, como alocação de biomassa, taxa de crescimento relativo (TCR). Raízes adventícias não foram medidas, apenas observadas, assim como o desenvolvimento de lenticelas hipertrofiadas. A TCR foi continuamente reduzida durante os 90 dias em condições de inundação para as raízes, o caule e as folhas em relação ao controle. O alagamento do substrato provocou alterações anatômicas como aumento da largura do córtex e do diâmetro do cilindro central da raiz e aumento do diâmetro dos elementos de vaso do caule e da raiz. Nossos resultados indicam que T. gardneriana permanece em estresse quando submetida a inundações. No entanto, a produção de estruturas relacionadas à evitação deste tipo de estresse foi chave para a manutenção e sobrevivência de T. gardneriana.
Palavras-chave: Pantanal, hipoxia, TCR, alagamento.
Introduction
The flooding process can be a high stress factor, playing a limiting role in the distribution of the arboreal vegetation (Lüttge, 1997) , once the excess of water interferes in the vigor and growth of the non-adapted plants (Crawford, 1992; Schueler & Holanda, 2000).
The restrictions to plant development induced by flooding result from the low availability of oxygen in the roots, causing changes in the metabolism and concentration of phytotoxins, besides the increase of anaerobic decomposition of
the organic matter and the availability of potentially toxic mineral substances (Schluter & Crawford, 2001; Visser, Voesenek, Vartapetian, & Jackson, 2003; Bailey-Serres & Voesenek, 2008).
Thus, the wetlands are occupied by plants tolerant to variable periods of flooding, because these plants compensate the anaerobic conditions of the soil by developing protection mechanisms, reducing the harmful effects in conditions of hypoxia and anoxia (Benz, Rhode, & Cruzan, 2007). Such mechanisms consist in morphological (Parolin et al. 2004) and physiological adaptations (Armstrong, Brandle, & Jackson, 1994).
However, the sensitivity to flooding can vary with the type, duration and intensity of the stress, as well as the stage of development of the plant. Even just considering the tropical plants, there is great diversity of responses in arboreal species to the seasonal flooding of the soil (Parolin et al. 2004; Duarte et al. 2005; Ferreira, Piedade, Junk, & Parolin, 2007).
The specie of the family Polygonaceae, Triplaris gardneriana Weed is a tree commonly found in Pantanal, Center-West of Brazil, subject to regular and intense flooding, and which remains partially submerged most of the year (Damasceno-Junior, Semir, Santos, & Leitão-Filho, 2005). These flooding conditions expose T. gardneriana species to distinct microhabitats with a greater period of flooding, resulting in a high tolerance degree to this condition.
Once that T. gardneriana is distributed in natural and seasonally flooded areas of Pantanal, our hypothesis is that the flooded soil induces responses that enable this specie to tolerate and/or avoid the stress imposed by flooding. This study evaluated whether: (1) Seedlings of T. gardneriana kept in water saturation conditions of the soil survive and present changes in growth; (2) Plants of T. gardneriana kept in flooded soil produce structures related to the stress avoidance to hypoxia. By understanding the adaptive mechanisms of plants, we enrich the knowledge on the strategies of plants to flooding, particularly at a phase of seedlings/saplings, critical stage for the establishment of plants (Junk, 1993).
Material and methods
Plant material and treatments
Seeds of T. gardneriana were collected in 15 matrixes approximately 10 km apart, in order to avoid the collection of seeds from a single population, sub-region of Abobral and Miranda (19°30′S, 57°04′W), Mato Grosso do Sul, Brazil. The fruits from the distinct matrixes were homogenized to obtain a single lot, and the seeds were set to germinate in Styrofoam trays containing organic soil (60% pinus bark, 15% fine grade vermiculite, 15% superfine grade and 10% humus).
A total of 90 plants were used and transferred at 10 days and around 3 cm tall to plastic vases of 3.5 liters (one plant per vase) containing organic soil, with no addition of fertilizers. After a period of 30 days acclimatization in a greenhouse, the experiment started, in a totally random design, with 45 plants kept in flooded soil and 45 control plants in drained soil. The plants flooded for 90 days were kept with a 2 cm water level above ground, and the control plants were irrigated by aspersion twice a day.
Plant growth
At 0, 30, 60 and 90 days after the flooding of the soil, the number of new leaves were counted and length of the stem was measured with the aid of a measuring tape and the diameter of the stem in the region of the lap with a digital caliper. For the study of plant growth and development, ten control plants and ten plants flooded were separated in roots, stem and leaf and were used in the beginning of the experiment and after 30, 60 and 90 days of flooding. These organs were dried in an oven at 70°C for 72 hours, time enough to reach constant weight. Each part of the plant was weighed in a semi-analytical scale, and the total biomass was calculated by the sum of the mentioned biomass categories. From this value, the relative growth rate (RGR) was calculated according to the procedures indicated by Benincasa (2003).
The leaf area (LA) of the first completely expanded leaf was obtained after 30, 60 and 90 days of the treatments. The LA was obtained through photography from a digital camera, Sony brand model W110, using a millimeter sheet as measurement referential. The images were processed using the program Sigma Scan Pro v. 5.0, Jandel Scientific (1991). From the values of LA and the dry mass of leaves, the specific leaf area (SLA) was obtained according to the formula described by Benincasa (2003).
Anatomy
For the anatomical study, five plants were separated from each treatment, fixated in FAA 50 (Johansen, 1940) at 90 days experiment. Free hand transversal cuts were made in the root and lap of the stem with the aid of a steel blade, the sections were clarified in sodium hypochlorite 20%, stained with safranin/astra blue (Kraus & Arduin, 1997), and fixed in glycerin 50% (Johansen, 1940).
The sections were analyzed in a light microscope and with the aid of a micrometric ocular the stem’s cortex and the cortex of the pivoting root were analyzed. Thus, we obtained the width of the cortex, diameter central cylinder and the diameter of the vessel elements. The differential qualitative anatomical characters, obtained for the control and flooded plants were documented in a Leica DC 300F system of microscope image capture.
Statistical analysis
ANOVA was used to establish variance and the comparison between medium values used Tukey’s test at 5% of probability. Statistical analyses were performed in R version 3.0.2 (R Development Core Team, 2013).
Results
Plants of T. gardneriana survived the 90 days of experiment and exhibited a considerable tolerance to flooding. There were no significant differences between plants growth with 0, 30 and 90 days of flooding (p > 0.05). The length of the stem and diameter of the stem’s base and the number of leaves were greater in the control individuals (Table 1).
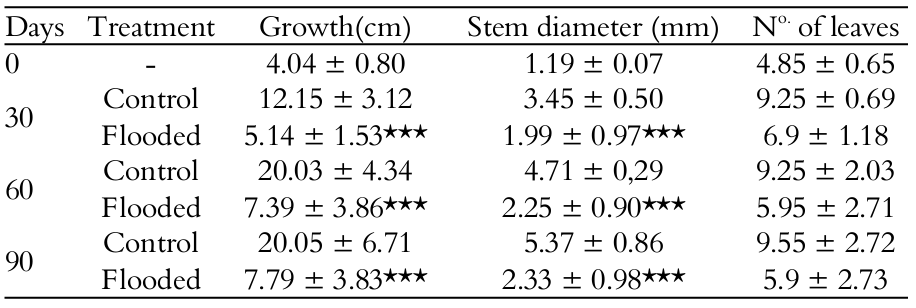
Triplaris gardneriana presented chlorosis at 30 days of flooding. The RGR was lower in flooding conditions, presenting significantly lower values (p < 0.05) for the root, stem and leaves (Table 2).
Compared to the control plants, the values of LA were significantly inferior in the flooded plants after 30 and 90 days of flooding (Figure 1). The flooding for 30 and 90 days induced increase in the SLA in plants of T. gardneriana (Figure 1) kept in flooded soil when compared to the control plants.
There were changes in the percentage of dry biomass of T. gardneriana kept under flooding (Figure 2). The flooded plants of T. gardneriana invested 35% of biomass in leaves, 33% roots and 32% stem; and the control plants invested 29% in leaves, 25% in stem and 46% in roots.
Flooding induced the formation of adventitious roots, longitudinal fissures and hypertrophy of lenticels with a mass of spongy tissue in the lap region after 10 days of flooding in T. gardneriana (Figure 3).
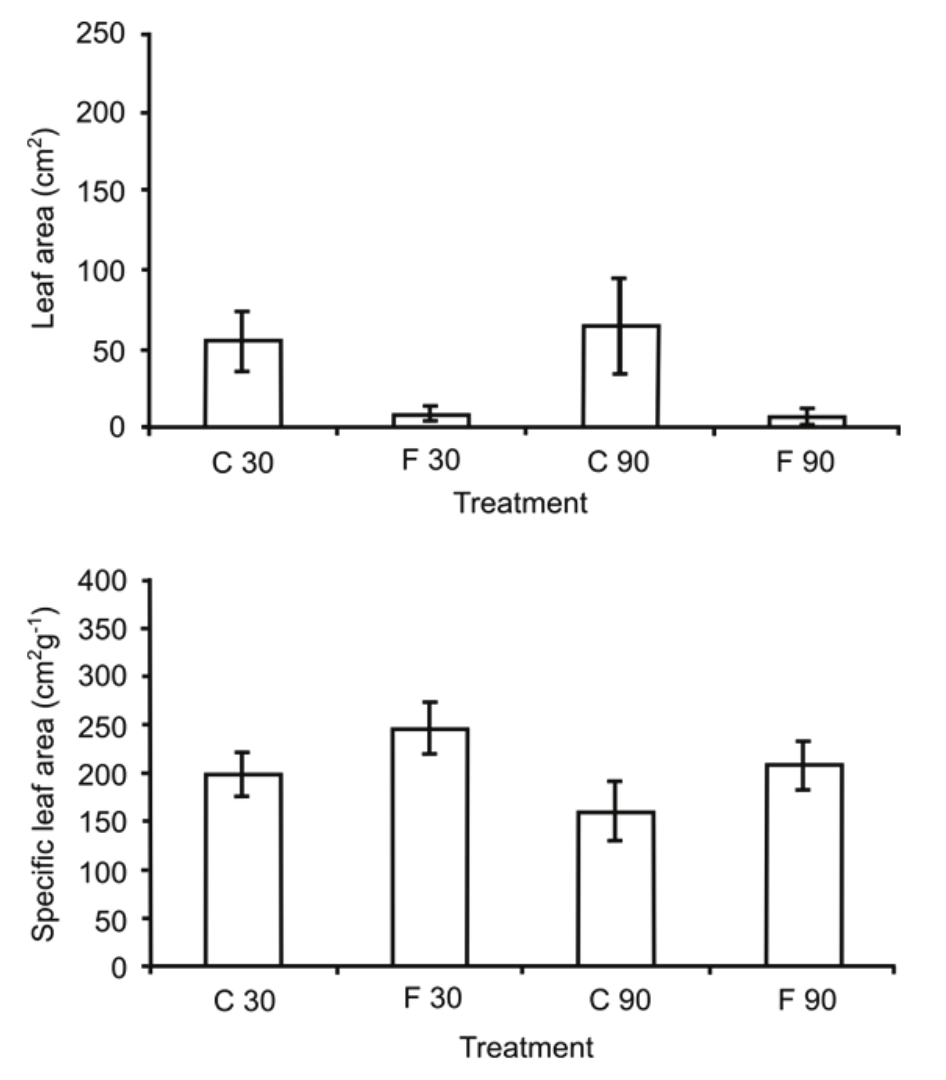
Figure 1
Means of leaf area (p < 0.001, n = 10) and specific leaf area (p < 0.05, n = 10) of Triplaris gardneriana under control (C) and flooded plants (F) at 30 and 90 days
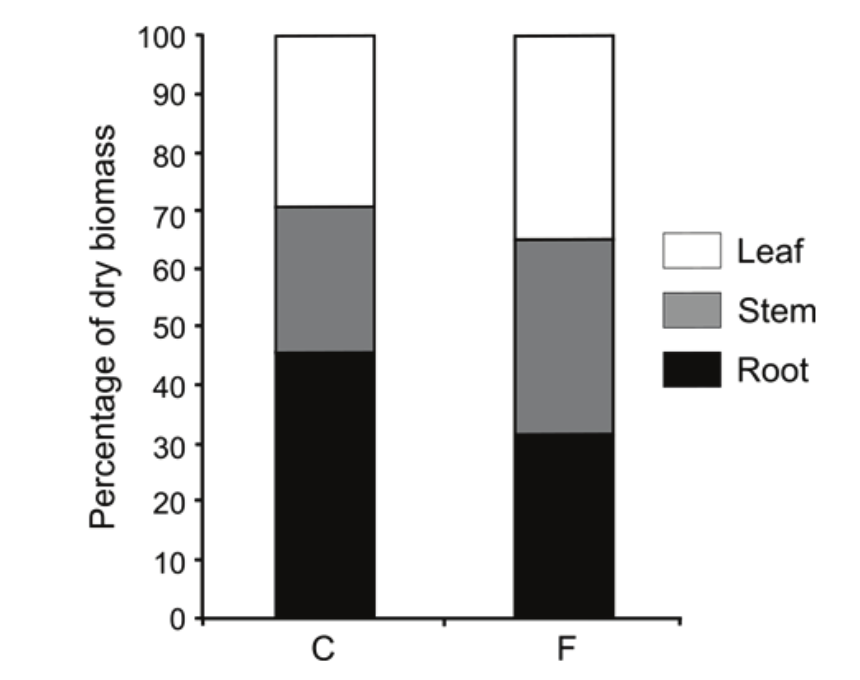
Figure 2
Percentage of dry biomass of the vegetative organs in the control plants (C) and flooded plants (F) of Triplaris gardneriana at 90 days of the experiment
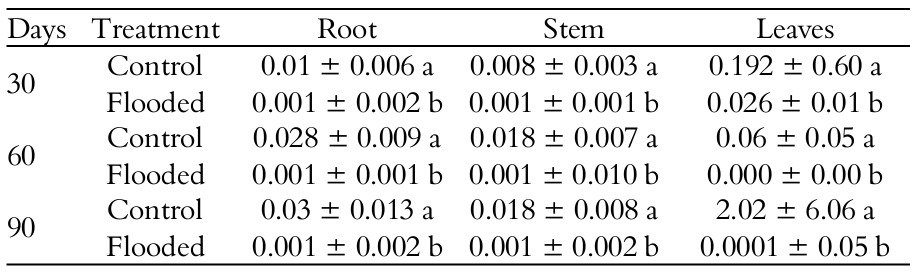
In the parenchyma cells of roots in plants kept in flooded soil, there was a decrease in the starch reserve (data not shown). The analysis of the transversal sections of the stem and root of T. gardneriana showed that the flooding of the substrate induced the decrease of some anatomical structures (Table 3) such as: width of the cortex and diameter of the central cylinder of root and diameter of the vessel element of root and stem.

Figure 3
Triplaris gardneriana after 90 days of experiment. (A) T. gardneriana grown in drained soil. (B) T. gardneriana grown in flooded soil, with development of hypertrophied lenticels (arrow 1) and adventitious roots (arrow 2)

Discussion
Although our results indicate that the T. gardneriana plants are capable of surviving for considerable periods of flooding, such condition seems to be prejudicial to the development. Flooding reduced the growth of the plants studied in all of the morphological variables measured, which can be a reflection of oxygen reduction for the roots, preventing the plants from absorbing water and nutrients and from synthesizing hormones as cytokines (Jackson, 1993).
The results of LA and SLA indicate increase in thickness of leaves in detriment of the expansion of the leaf area in the flooded plants. These responses can be important for the water economy of the plant (Batista, Medri, Bianchini, Medri, & Pimenta, 2008). Some studies pointed water deficit in flooded plants due to the lower water absorption caused by modifications in permeability of membranes in the cells of the roots, in consequence of the hypoxic environment, resulting in lower water conductivity in the roots (Else, Coupland, Dutton, & Jackson, 2001). The duration of flooding affected the biomass of the leaf, stem and root of the flooded plants, since it slowed the productivity in response to the flooding, compromising the growth of vegetative organs. According to Lobo and Joly (1995) the species tolerant to flooding have the capacity of maintaining biomass of the aerial part during the flooding period, as seen for T. gardneriana during the 90 days of experiment. However, flooding is a condition of stress for these species, since there was a deviation from the optimum (Larcher, 2004), indicated by the significantly different values between the control and flooded plants.
The decrease in RGR of the leaf, root and stem of T. gardneriana kept in flooded soil may be showing the lowest quantity of energy available, and this decrease is caused by the deviation in the metabolic pathways of glucose to fermentation pathways, which results in the reduction of energy levels (Crawford & Braendle, 1996). That may lead to the loss of biomass, as response to the reduction in availability of carbohydrates due to change from aerobic to anaerobic respiration, which is energetically less efficient than the aerobic route (Crawford & Braendle, 1996).
The formation of adventitious roots, aerenchyma and hypertrophied lenticels is strongly associated with the survival of T. gardneriana. Such responses are common in species tolerant to flooding of the soil (Parolin, 2001) and described as enablers of oxygen transportation to the roots, contributing to the recovery and maintenance of aerobic respiration in flooded plants (Ferreira, Piedade, Franco, & Junk, 2009; Suralta & Yamauchi, 2008; Colmer, 2003; Finlayson, 2005). In addition, hypertrophied lenticels can enable the elimination of potentially toxic components produced during anaerobiosis, such as ethylene, ethanol and acetaldehyde (Kozlowski, 1997). Similar responses were reported for several aquatic species or the ones that live in flooded areas, such as Myriophyllum spicatum (Xie, Ren, & Li, 2007), Himatanthus sucuuba (Ferreira et al., 2009) and Calophyllum brasiliense (Oliveira & Joly, 2010). According to Armstrong (1968) the capacity presented by several species of surviving under such conditions depends partially or totally on the capacity of production and on the activity of these structures.
Triplaris gardneriana showed lower quantity of starch in flood plants. According to Kolb et al., (1998) the decrease in starch reserves of the cortical parenchyma during flooding can be related to the large demands of carbohydrates required for the development of the observed morpho-anatomical structures, in addition to the need of maintaining the anaerobic metabolism, which demands high energy rate (Koslowski, 1997; Crawford & Brandle, 1996). Species such as Lithraea molleoides (Medri et al., 2007) and Guibourtia hymenifolia (Santiago & Paoli, 2007) have also presented lower quantity of starch reserve in plants kept in flooded soil.
The lower diameter of the vessel elements of the main root and stem in flooded plants of T. gardneriana can be related to the process of avoiding embolism and ensuring the water flow, once the flooding can reduce the absorption of water by the plant (Coutts, 1981).
Conclusion
The survival of the plants during the 90 days experiment is an indication of the maintenance of the minimal metabolic rate, resulting in the survival of the plants. The results show faster responses to flooding for the species T. gardneriana, as it is demonstrated with the production of lenticels and adventitious roots at 10 days of experiment, with a greater biomass and growth. These responses can be related to the plant’s distribution in the field since they occupy areas with longer flooding. Therefore, this species present adaptive mechanism that characterizes its local distribution, with greater exposure to flooding, although the energy production generated is larger during the normoxia period.
Acknowledgements
The authors would like to thank the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior/CAPES for the Master’s degree grant given to Vanessa Pontara. Bueno and Pontara wish to express gratitude to the Coordenadoria de Pesquisa da Pró-Reitoria de Pesquisa and the Rede de Sementes do Pantanal, for their support during the field work that made this research project possible.
References
Armstrong, W. (1968). Oxygen diffusion from the roots of woods species. Physiologia Plantarum, 21(3), 539-543.
Armstrong, W., Brandle, R., & Jackson, M. B. (1994). Mechanisms of flood tolerance in plants. Acta Botanica Neerlandica, 43(4), 307-358.
Bailey-Serres, J., & Voesenek, L. A. C. J. (2008). Flooding stress: acclimations and genetic diversity. Annual Review of Plant Biology, 59, 313-319.
Batista, C. U. N., Medri, M. E., Bianchini, E., Medri, C., & Pimenta, J. A. (2008). Tolerância à inundação de Cecropia pachystachya Trec. (Cecropiaceae): aspectos ecofisiológicos e morfoanatômicos. Acta Botânica Brasílica, 22(1), 91-98.
Benincasa, M. M. P. (2003). Análise do crescimento de plantas: noções básicas (2a ed.). Jaboticabal, SP: Funep.
Benz, R. B., Rhode, M. J., & Cruzan, B. M. (2007). Aerenchyma development and elevated alcohol dehydrogenase activity as alternative responses to hypoxic soils in the Piriqueta caroliniana complex. American Journal of Botany, 94(4), 542-550.
Colmer, T. D. (2003). Long-distance transport of gases in plants: a perspective on internal aeration and radial oxygen loss from roots. Plant, Cell & Environment, 26(1), 17-36.
Coutts, M. P. (1981). Effect of waterlogging on dormant sitka spruce seedlings. Annals of Botany, 47(6), 747-753.
Crawford, R. M. M. (1992). Oxygen availability as ecological limit to plant distribution. In M. Bergon, & A. H. Fitter (Eds.), Advances in ecological research (p. 93-185). London, UK: Academic Press.
Crawford, R. M. M., & Braendle, R. (1996). Oxygen deprivation stress in a changing environment. Journal Experimental of Botany, 47(2), 145-159.
Damasceno-Junior, G. A., Semir, J., Santos, F. A. M., & Leitão-Filho, H. F. (2005). Structure, distribution of species and inundation in a riparian forest of Rio Paraguai, Pantanal, Brazil. Flora (Jena), 200(2), 119-135.
Duarte, H. M., Geβler, A., Scarano, F. R., Franco, A. C., de Mattos, E. A., Nahm, M., ... Lütge, U. (2005). Ecophysiology of six selected shrub species in different plant communities at the periphery of the Atlantic Forest of SE- Brazil. Flora, 200(5), 456-476.
Else, M. A., Coupland, D., Dutton, L., & Jackson M. B. (2001). Decreased root hydraulic conductivity reduces leaf water potential, initiates stomatal closure and slows leaf expansion in flooded plants of castor oil (Ricinus communis) despite diminished delivery of ABA from the roots to shoots in xylem sap. Physiologia Plantarum, 111(1), 46-54.
Ferreira, C. S., Piedade, M. T. F., Franco, A. C., & Junk, W. J. (2009). Adaptive strategies to tolerate prolonged flooding in seedlings of floodplain and upland populations of Himatanthus sucuuba, a Central Amazon tree. Aquatic Botany, 90, 246-252.
Ferreira, C. S., Piedade, M. T. F., Junk, W. J., & Parolin, P. (2007). Floodplain and upland populations of Amazonian Himatanthus sucuuba: Effects of flooding on germination, seedling growth and mortality. Environmental and Experimental Botany, 60(3), 477-483.
Finlayson, C. M. (2005). Plant ecology of Australia’s tropical floodplain wetlands: a review. Annals Botany, 96, 541- 555.
Jackson, M. B. (1993). Are plant hormones involved in root to shoot communication? Advances in Botanical Research, 19,103 -187.
Jandel Scientific. (1991). User’s Maual. San Rafael, CA.
Johansen, D. A. (1940). Plant Microtechnique. Tata MacGraw-Hill, Bombay.
Junk, W. J. (1993). Wetlands of tropical South-America. In Whigham, D., Hejny, S., & Dykyjova, D. (Eds.), Wetlands of the World (p. 679-739). Dordrechet: Kluwer Academic Publishers.
Kolb, R. M., Medri, M. E., Bianchini, E., Pimenta, J. A., Giloni, P. C. & Correa, G. T. (1998). Anatomia ecológica de Sebastiania commersoniana (Baillon) Smith & Downs (Euphorbiaceae) submetida ao alagamento. Revista Brasileira de Botânica, 21, 305-312.
Kozlowski, T. T. (1997). Reponses of woody plants to flooding and salinity. Tree Physiology Monograph, 1(7), 1-29.
Kraus, J. E., & Arduin, M. (1997). Manual básico de métodos em morfologia vegetal. Rio de Janeiro, RJ: Universidade Federal Rural do Rio de Janeiro.
Larcher, W. (2004). Ecofisiologia vegetal. São Carlos, SP: Rima.
Lobo, P. C., & Joly, C.A. (1995). Mecanismos de tolerância a inundação de plantas de Talauma ovata St. Hil. (Magnoliaceae), uma espécie típica de matas de brejo. Revista Brasileira de Botânica, 18(2), 177-183.
Lüttge, U. (1997). Physiological Ecology of Tropical Plants. Berlin; Heidelberg, DO: Springer-Verlag.
Medri, M. E., Ferreira, A. C., Kolb, R. M., Bianchini, E., Pimenta, J. A., Davanso-Fabro, V. M., & Medri, C. (2007). Alterações morfoanatomicas em plantas de Lithraea molleoides (Vell.) Engl. submetidas ao alagamento. Acta Scientiarum. Biological Science, 29(1), 15-22.
Oliveira, V. C., & Joly, C. A. (2010). Flooding tolerance of Calophyllum brasiliense Camb. (Clusiaceae): morphological, physiological and growth responses. Trees, 24(1), 185-193.
Parolin, P. (2001) Senna reticulata, a pioneer tree from Amazonian varzea floodplains. The Botanical Review 67(2), 239-254.
Parolin, P., De Simone, O., Haase, K., Waldhoff, D., Rottenberger, S., Kuhn, U., … Junk, W.J. (2004). Central Amazon floodplain forests: tree survival in a pulsing system. The Botanical Review, 70(3), 357-380.
R Core Team (2013). R: A language and environment for statistical computing. Vienna, AU. Retrieved from http://www.R-project.org/
Santiago, E. F., & Paoli, A. A. S. (2007). Respostas morfológicas em Guibourtia hymenifolia (Moric.) J. Leonard (Fabaceae) e Genipa americana L. (Rubiaceae), submetidas ao estresse por deficiência nutricional e alagamento do substrato. Revista Brasileira de Botânica, 30(1), 131-140.
Schluter, U., & Crawford, R. M. M. (2001). Long-term anoxia tolerance is leaves of Acorus calamus L. and Iris pseudacorus L. Journal of experimental Botany, 52(364), 2213-2225.
Schueler, T. R., & Holland, H. K. (2000). Practice of watershed protection. Wetter is not always better flood tolerance of woody species (Technical Note from Watershed Protection Techniques, 1). Ellicott City, MD: Center for Watershed Protection.
Suralta, R. R., & Yamauchi, A. (2008). Root growth, aerenchyma development, and oxygen transport in rice genotypes subjected to drought and waterlogging. Environmental Experimental of Botany, 64(1), 5-82.
Visser, E. J. W., Voesenek, L. A. C. J., Vartapetian, B. B., & Jackson, M. B. (2003). Flooding and plant growth. Annals of Botany, 91(2), 107-109.
Xie, W., Ren, B., & Li, F., (2007). Morphological and physiological responses to sediment type and light availability in roots of the submerged plant Myriophyllum spicatum. Annals of Botany, 100(7), 1517-1523.
Author notes
vanessapontara@gmail.com