Research Article
Antimicrobial activity of Lafoensia pacari phenolics-containing extract against common bean phytopathogens
Atividade antimicrobiana de extrato fenólico de Lafoensia pacari contra ftopatógenos de feijoeiro
 Waléria Ramos Nogueira de Souza walfarmagyn@gmail.com; prisciladiasfarmacia@gmail.com; mtbara@gmail.com
Waléria Ramos Nogueira de Souza walfarmagyn@gmail.com; prisciladiasfarmacia@gmail.com; mtbara@gmail.com
Antimicrobial activity of Lafoensia pacari phenolics-containing extract against common bean phytopathogens
Pesquisa Agropecuária Tropical, vol. 52, e72726, 2022
Escola de Agronomia/UFG
Received: 04 May 2022
Accepted: 18 August 2022
Published: 23 September 2022
ABSTRACT: Lafoensia pacari represents an alternative to control phytopathogens and common bean (Phaseolus vulgaris L.) diseases. This study aimed to investigate the chemical composition of L. pacari phenolics-containing extract obtained from leaves and assess its in vitro and in vivo inhibitory activity against Curtobacterium flaccumfaciens pv. flaccumfaciens, Xanthomonas axonopodis pv. phaseoli, Xanthomonas fuscans subsp. fuscans, Xanthomonas sp., Colletotrichum lindemuthianum, Fusarium oxysporum and Macrophomina phaseolina. The microbial susceptibility screening and the extract’s potential to reduce the anthracnose severity and the common bacterial blight were examined. Eleven phenolic compounds were identified, eight of which were described for the first time in L. pacari leaves: gentisic, caffeic, p-coumaric, protocatechuic and vanillic acids, luteolin, naringenin and quercetin. The L. pacari extract completely inhibited bacterial growth at the concentration of 0.62-1.25 %, as well as the mycelial growth of the C. lindemuthianum (BRM 007626) isolate at the concentration of 5 % (v/v). Under greenhouse conditions, the extract led to the control of the common bacterial blight caused by X. axonopodis pv. phaseoli (BRM 25302) in the Pérola bean cultivar and to a low efficiency in the control of anthracnose caused by C. lindemuthianum (BRM 007447) in the IPA 7419 cultivar.
KEYWORDS: Phaseolus vulgaris L., Colletotrichum lindemuthianum, Curtobacterium flaccumfaciens, Xanthomonas axonopodis.
RESUMO: Lafoensia pacari representa uma alternativa para controlar fitopatógenos e doenças do feijoeiro (Phaseolus vulgaris L.). Objetivou-se investigar a composição química de extrato fenólico obtido de folhas de L. pacari e avaliar sua atividade inibitória in vitro e in vivo contra Curtobacterium flaccumfaciens p v. flaccumfaciens, Xanthomonas axonopodis pv. phaseoli, Xanthomonas fuscans subsp. fuscans, Xanthomonas sp., Colletotrichum lindemuthianum, Fusarium oxysporum e Macrophomina phaseolina. Foram investigados a triagem da suscetibilidade microbiana e o potencial de redução da severidade da antracnose e do crestamento bacteriano comum pelo extrato. Onze compostos fenólicos foram identificados, sendo oito descritos pela primeira vez em folhas de L. pacari: ácidos gentísico, cafeico, p-cumárico, protocatecuico e vanílico, luteolina, naringenina e quercetina. O extrato de L. pacari inibiu completamente o crescimento bacteriano na concentração de 0,62-1,25 %, bem como o crescimento micelial do isolado de C. lindemuthianum (BRM 007626) na concentração de 5 % (v/v). Em condições de casa-de-vegetação, o extrato resultou no controle do crestamento causado por X. axonopodis p v . phaseoli (BRM 25302) na cultivar de feijão Pérola e em baixa eficiência no controle da antracnose causada por C. lindemuthianum (BRM 007447) na cultivar IPA 7419.
PALAVRAS-CHAVE: Phaseolus vulgaris L., Colletotrichum lindemuthianum, Curtobacterium flaccumfaciens, Xanthomonas axonopodis.
INTRODUCTION
Brazil is the third largest producer of common bean (Phaseolus vulgaris L.) in the world (Silva 2020), a staple food and key protein source in the country’s traditional diet. The crop has a high socioeconomic value, as it is part of small, medium and large-scale production systems that are widely distributed across the country (Silva 2020).
Beans may be affected by several diseases that can generate considerable losses to producers, among them Curtobacterium wilt, common bacterial blight, anthracnose, charcoal rot and Fusarium wilt (Wendland & Lobo Junior 2018).
Bacteriosis caused by Curtobacterium flaccumfaciens pv. flaccumfaciens colonizes and obstructs xylem vessels, leading to leaf wilting, yellowing or burning, vascular darkening, dwarfism and death of common bean (Valdo et al. 2016). The common bacterial spot caused by Xanthomonas axonopodis pv. phaseoli (Smith) Dye and Xanthomonas fuscans subsp. fuscans may cause losses of up to 100 % in the bean production (Paiva et al. 2020). Infections by Xanthomonas spp. cause a broad range of nonspecific symptoms, making their identification difficult, and water-soaked spots evolve into necrosis on leaves, wilting, rotting, hypertrophy, hyperplasia, blights, dieback and cankers (Catara et al. 2021).
Fungal diseases also pose risk hazards for the common bean. Anthracnose, whose causal agent is the Colletotrichum lindemuthianum (Sacc & Magn.) Scribner fungus, is a disease that affects bean crops located in regions with temperatures of 13-26 ºC, associated with high relative humidity caused by rainfall or frequent irrigation. It causes necrotic lesions on veins and the underside of leaves, as well as shoots, and may result in losses of up to 100 % in the bean production (Barbosa & Gonzaga 2012).
Fusarium wilt has as its causal agent the Fusarium oxysporum Schlecht. f. sp. phaseoli fungus, which survives in the soil as chlamydospores and infects the host through natural or artificial wounds, blocking the plant’s xylem vessels and causing yellowing that may result in the death of the leaves and of the whole plant (Cardoso 1990). Another widespread disease in beans is charcoal rot caused by Macrophomina phaseolina (Tassi) Goid. (1947), a soil fungus found in places with temperatures of 24-27 ºC and which produces gray stem rot (Hagedorn & Inglis 1986).
Alternative treatments for diseases that can affect bean crops should be investigated, mainly because agrochemicals may be related to human health problems and environmental damage (Rongai et al. 2019). In addition, bioproducts offer new horizons in biotechnology by promoting industrial and environmental sustainability (Varjani et al. 2020). In this context, the search for bioproducts based on medicinal plants is a promising alternative in the control of plant diseases and phytopathogens (Carvalho et al. 2021, García-Pérez et al. 2021, Itako et al. 2021).
A plant native to the Cerrado (Brazilian Savanna) that reportedly possesses antimicrobial activity is Lafoensia pacari A. St.-Hil. (Lythraceae) (Silva et al. 2018), whose leaves have secondary metabolites such as tannins, favonoids and other phenolic compounds (Carneiro et al. 2016, Pereira et al. 2018). The potential of these polyphenols for antimicrobial activity is known through damage mechanisms to cell structure (Rongai et al. 2018, Silva et al. 2018, Tocci et al. 2018).
Thus, this study aimed to assess the phenolic chemical composition of standardized extract of L. pacari via liquid chromatography-high resolutiontandem mass spectrometry (LC-HRMS) and investigate its antibacterial and antifungal activities against common bean pathogens.
MATERIAL AND METHODS
Lafoensia pacari leaves were collected from trees cultivated at the Agência Goiana de Assistência Técnica, Extensão Rural e Pesquisa Agropecuária (16º36’19”S, 49º15’48”W and 710 m of altitude), in Goiânia, Goiás state, Brazil, in May 2016. A voucher specimen was deposited at the Universidade Federal de Goiás herbarium (nº. UFG-47581).
The leaves were dried at room temperature and grounded by a knife mill. To yield the extract, 500 g of plant material were percolated in 10 L of 70 % hydroethanolic solution (v/v). The ethanol was then evaporated in a rotary evaporator (Buchi R-220 SE), under reduced pressure (30 rpm; 40 ºC; 600 bar), until the solid content reached ± 25 %. The L. pacari phenolics-containing extract was standardized in punicalagin (Garcia et al. 2021) by high performance liquid chromatography using a Waters® model HPLC Alliance with a photodiode array detector. Chromatographic separations were conducted via Waters® C18 column (5 µ; 250 × 4.6 mm; Agilent), at a 257 nm wavelength, elution mode gradient, with the mobile phase consisting of acetonitrile (A) and 0.001 mol L-1 phosphoric acid:0.01 mol L-1 monobasic potassium phosphate (B): 0-20 min: 8:92 (A:B %); 20-25 min: 18:82 (A:B %). The flow rate was 0.8 mL min-1, with a 10 µL injection volume, and the column was maintained at 25 ºC. The external standard of punicalagin (purity ≥ 98.5 %; Sigma-Aldrich®) was used at a 0.5 mg mL-1 concentration.
The chemical composition of the extract was analyzed by liquid chromatography-high resolution-tandem mass spectrometry (LC-HRMS). Chromatographic separations were conducted on a Liquid Chromatograph Thermo Scientific® Ultimate 3000, using the ACE® C18 column (3 μm; 100 x 4.6 mm) coupled with a Q-Exactive Orbitrap High Resolution Mass Spectrometer, Thermo Scientific®, with H-ESI source, operating on positive and negative modes and using spray voltage of 4 kV, capillary temperature of 350 ºC, auxiliary gas temperature of 300 ºC and scan range (m/z) of 150-700. Gradient elution was obtained via deionized acidified water with 0.1% formic acid (mobile phase A; v/v) and methanol acidified with 0.1 % formic acid (mobile phase B; v/v): 0-10 min: 93:7 (A:B %); 10-20 min: 70:30 (A:B %); 20-25 min: 50:50 (A:B %); 25-28 min: 30:70 (A:B %); 28-30 min: 20:80 (A:B %); 30-33 min: 100 (B %), kept constant for 3 min, and 93:07 (A:B %) in 2 min, kept constant for 2 min. The flow rate was 0.3 mL min1, with a 10 μL injection volume and column temperature at 20 ºC. For the fragmentation study, a parallel reaction monitoring (PRM) experiment was performed with different energies of collision (EC).
To identify the phenolic compounds, a standard mixture solution at a 50 μg mL-1 concentration was prepared. The phenolic compound standards (Sigma-Aldrich®) were: gallic acid (≥ 98.5 %), protocatechuic acid (≥ 97 %), gentisic acid (≥ 98 %), cafeic acid (≥ 98 %), p-coumaric acid (≥ 98 %), vanillic acid (≥ 97 %), ellagic acid (≥ 95 %), catechin (≥ 98 %), epicatechin (≥ 90 %) and the favonoids rutin (≥ 94 %), quercetin (≥ 95 %), naringenin (≥ 95 %), luteolin (≥ 98 %) and kaempferol (≥ 90 %). The data were processed by the Xcalibur™ software.
For the biological assays, a screening was carried out with some of the phytopathogens most commonly found in the common bean (Table 1), provided by the official collection of multifunctional and phytopathogenic microorganisms of the Embrapa Arroz e Feijão.
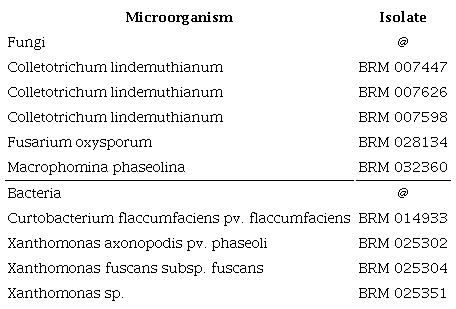
The in vitro inhibitory activity of the extract on bacterial growth was conducted on 100-mm
Petri dishes containing sucrose, casein, potassium phosphate, magnesium sulfate agar for X. axonopodis and nutrient sucrose agar for C. flaccumfaciens. The extract was added at concentrations of 5, 2.5, 1.25 and 0.62 % (v/v). Aliquots of 100 μl of bacterial suspensions were used to inoculate approximately 104 CFU mL-1 of the culture medium. The plates were incubated at 25 ºC for three days. The bacterial growth inhibitory activity was monitored by counting the colony-forming units (CFU) with a magnifying glass. The control consisted of pure bacterial suspensions on the culture medium (Côrtes et al. 2012) and the experiments were performed in quadruplicates.
The in vitro antifungal activity of the extract on mycelial growth was conducted on 100-mm Petri dishes containing potato-dextrose-agar (PDA). The extract was added at concentrations of 5, 2.5, 1.25 and 0.62 % (v/v). A 5-mm disc of fresh PDA medium containing fungal mycelium of C. lindemuthianum, M. phaseolina or F. oxysporum was inoculated and then incubated at a controlled temperature of 21 ºC, with a light cycle (12 h) and dark (12 h) photoperiod by artificial light (fuorescent lamp). The colony diameters were measured with a manual caliper at the seventh and fourteenth days of incubation for C. lindemuthianum and M. phaseolina and at the seventh day for F. oxysporum. The control consisted of fungi inoculated in BDA (Côrtes et al. 2012) and the experiments were performed in quadruplicates. For the in vitro assays, the sample unit consisted of a Petri dish with four replicates for each microorganism species.
To evaluate the potential of 1.25 % (v/v) of the extract in reducing the severity of common bacterial blight (Rava 1984) in the common bean, an experiment was carried out in a greenhouse, with the following treatments: a) seeds of Pérola bean cultivar were treated with the L. pacari extract and then planted in factory-sterilized disposable cups filled with a prepared substrate (Maxfertil®). Four seeds of each cultivar were planted in five cups and assessed after 14 days under greenhouse conditions. Daily irrigation was performed by dripping without quantification of water volume; b) other seeds of the Pérola bean cultivar were treated with 1.25 % (v/v) of the L. pacari extract and then planted as aforementioned. After 15 days of growth, the plants were inoculated with a bacterial suspension (X. axonopodis pv. phaseoli - BRM 025302) prepared at a concentration of 108 CFU mL-1. With the aid of needles, micro-lesions were made on the limb of leaves and then inoculated with the bacterial suspension by hand spraying. After 1 h of inoculation, the plants were treated again with 1.25 % (v/v) of L. pacari extract and transferred to a nebulization room at a temperature of 21 ºC. Daily irrigation was performed by dripping without quantification of water volume. After seven days, the common bacterial blight symptoms were assessed using a general grade scale of 1 to 6 degrees of severity: 1 - no symptoms; 2 - beginning of yellowing; 3 - injury itself; 4 - lesion in the border area between the leaf cuts; 5 - injury in the area beyond the leaf cut; 6 - advanced symptoms throughout inoculated leaf (Rava 1984). Controls containing sterile distilled water were performed and treatments were done in quadruplicates.
To evaluate the potential of the L. pacari extract on reducing the anthracnose severity (Chaves 1980), the IPA 7419 and Pérola bean cultivars were used. An amount of 150 mL of 5 % (v/v) extract were manually sprayed, and seeds were planted in sterile styrofoam trays previously disinfected with 20 % sodium hypochlorite, containing 16 rows with eight cells each. A total of eight rows were filled with a prepared substrate (Maxfertil®), four rows for each cultivar, one seed per cell, with row spacing between them. After planting, the trays were kept in a greenhouse under 21 ºC. Daily irrigation was performed by dripping without quantification of water volume. At the ninth day of growth, 1.2 x 106 conidia mL-1 of C. lindemuthianum BRM 007447 were inoculated by manual spray for each pair of trays (Rava et al. 1993).
The treatments were performed with 5 % (v/v) of L. pacari extract as it follows: a) after 4 h of conidia inoculation, the plants of both cultivars were manually sprayed with 150 mL of the extract, and the seedlings were transferred to a nebulization room at 21 ± 1 ºC and evaluated after seven days. Daily irrigation was performed by dripping, without quantification of water volume; b) at the ninth day of bean growth of both cultivars (as previously mentioned), 1 h prior to the inoculation of fungal suspension, the plants were manually sprayed with 150 mL of the extract. After 4 h, 150 mL of the extract were sprayed again. The trays were transferred to a nebulization room at 21 ± 1 ºC, for seven days. Daily irrigation was performed by dripping, without quantification of water volume, when anthracnose symptoms were assessed using a general grade scale from 1 to 9 degrees of severity: 1 - no symptoms; 2 - up to 1 % of veins showing necrotic spots, perceptible only on the underside of leaves; 3 - higher frequency of leaf symptoms described in the previous grade, up to 3 % of veins affected; 4 - up to 1 % of veins showing necrotic spots, noticeable on both sides of leaves; 5 - higher frequency of leaf symptoms described in the previous grade, up to 3 % of veins affected, on both sides of leaves; 6 - necrotic spots on veins noticeable on both sides of leaves and presence of some lesions on stems, branches and petioles; 7 - necrotic patches on most veins and much of adjacent mesophyll tissue that ruptures. Presence of abundant lesions on stalks, branches and petioles; 8 - necrotic spots on almost all veins, causing ruptures, defoliation and reduced plant growth. Very abundant lesions on stalks, branches and petioles; 9 - most plants dead (Ciat 1990).
Controls containing sterile distilled water were used and the treatments were done in quadruplicates.
For the greenhouse trials, the experimental unit was represented by a cup containing one plant, with five replications.
RESULTS AND DISCUSSION
The standardization of plant extracts is important to reduce the chemical composition variability, which affects bioactivity (Rongai et al. 2019). As regards the extraction process used, we initially opted for the 70 % (v/v) hydroethanolic solution, to favor the presence of polar compounds such as favonoids, phenolic acids and punicalagin (Carneiro et al. 2016). Once the extract was obtained, quantification of a chemical marker was necessary for its standardization. Strategies for expressing the plant extract concentration by means of a compound already identified in this species and related to the biological activity being researched have been already reported (Pereira et al. 2018). Punicalagin was then used as a marker compound in the extract standardization. The analysis of the HPLC chromatogram of the L. pacari extract showed two peaks (Figure 1), representing α- or β-anomeric hydrogens in hydrolysable tannins such as punicalagin. Following the punicalagin quantification, the content of this marker in the L. pacari extract was 45.86 % ± 3.45.
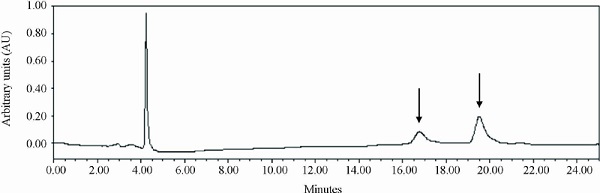
Figure 1
Chromatographic profle at 254 nm of Lafoensia pacari phenolics-containing extract. Column C18 (5 µ; 250 × 4.6 mm), mobile phase in a gradient system. Punicalagin α and β anomers (arrows).
Through LC-HRMS, a total of 11 chemical compounds, including ellagic, gallic, gentisic, caffeic, p-coumaric, protocatechuic and vanillic phenolic acids, as well as the favonoids luteolin, naringenin, quercetin and kaempferol, were identified (Table 2). Eight compounds were first identified in Lafoensia pacari leaves: gentisic, cafeic, p-coumaric, protocatechuic and vanillic acids, luteolin, naringenin and quercetin.
![Phenolic acids and favonoids identified in Lafoensia pacari
phenolics-containing extract by LC-HRMS, in negative mode
([M-H]-).](../253070366041_T2_tabla.png)
The presence of polyphenols such as ellagic acid, kaempferol, quercetin glycosides and punicalagin were identified in L. pacari by electrospray ionizationmass spectrometric detection (ESI-MS) (Pereira et al. 2018). Carneiro et al. (2016) identified punicalagin by electrospray ionization time-of-flight mass spectrometry (ESI-TOF-MS).
Phenolic acids are known to own antifungal and antibacterial activity through a damage mechanism (Osorio et al. 2010, Veloz-García et al. 2010, Tocci et al. 2018, García-Pérez et al. 2021). It is interesting to note that plant extracts have a phytocomplex that contributes to the biological activity detected (Tocci et al. 2018, Rongai et al. 2019). It is thus suggested that secondary metabolites present in the L. pacari extract may explain, by association, its antimicrobial activity.
Bioactive compounds from plants are potentially useful tools to control phytopathogenic bacteria and fungi. In this research, the L. pacari extract ranged from 5 % (22.93 mg g1 of punicalagin) to 2.5 % (11.46 mg g1), 1.25 % (5.73 mg g1) and 0.625 (2.87 mg g1). The results showed a differential inhibition of bacterial and mycelial growth of fungi by the extract, with bacteria being more sensitive.
The in vitro inhibitory activity of the L. pacari extract on mycelial and bacterial growth showed a complete inhibition of C. flaccumfaciens and Xantomonas isolates at the 1.25 % concentration. At 0.62 %, the Xantomonas sp. (BRM 025351) growth was inhibited by 90 % (Figure 2).
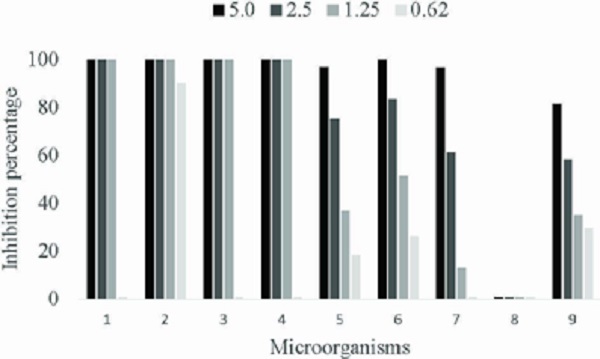
Figure 2
In vitro inhibition percentage of bacterial and mycelial growth by Lafoensia pacari phenolicscontaining extract (%). 1 - Xanthomonas axonopodis BRM 025302; 2 - Xanthomonas sp. BRM 025351; 3 - Xanthomonas fuscans BRM 025304; 4 - Curtobacterium flaccumfaciens BRM 014933; 5 - Colletotrichum lindemuthianum BRM 007447; 6 - Colletotrichum lindemuthianum BRM 007626; 7 - Colletotrichum lindemuthianum BRM 007598; 8 - Macrophomina phaseolina BRM 032360; 9 - Fusarium oxysporum BRM 028134.
According to Valentini et al. (2010), diseases caused by bacteria represent an agricultural hazard in various economic crops. The Curtobacterium wilt caused by Curtobacterium flaccumfaciens pv. Flaccumfaciens, a pathogen recently found in Brazil and subsequently detected in several bean-producing regions (Valentini et al. 2010), has become a threat to the cultivation of bean (P. vulgaris). Measures are hence needed to prevent the spread of such pathogens. According to Catara et al. (2021), the Xanthomonas genus infects a wide range of crops, resulting in global economic and environmental impacts on seed, plant and food trades.
The inhibitory activity of the L. pacari extract on mycelial growth only showed a complete inhibition for the C. lindemuthianum isolated BRM 007626 at the concentration of 5 % (22.93 mg g-1 of punicalagin). The other two races of this fungus were inhibited by about 96 % at this concentration. It is suggested that these different sensitivities to the extract may be due to the genetic and structural variability of C. lindemuthianum. Fusarium was inhibited by 81 % at the extract concentration of 5 % and M. phaseolina was resistant to the concentrations under analysis (Figure 2).
It must be pointed out that we have been unable to find studies on extracts of this medicinal plant as applied to agricultural purposes. However, we have found other plant extracts that have phenolics, such as Caesalpinia cacalaco, which shows in vitro fungistatic activity against C. lindemuthianum mycelial growth for common bean (Veloz-García et al. 2010). Osorio et al. (2010) reported a high efficiency of phenolic extracts from Larrea tridentata, Carya illinoensis and Punica granatum in inhibiting the mycelial growth of Colletotrichum truncation, Colletotrichum coccodes and Fusarium species. Rongai et al. (2018) reported that punicalagins may form pore-like structures in the cell wall of Botrytis cinerea, which can alter physiological transmembrane gradients and lead to fungal cell death. Polyphenols' antimicrobial action mechanisms are also due to their redox behavior and antioxidant capacity (Chiorcea-Paquim et al. 2020).
It is important to report data that corroborate those of the present study, regarding the inhibitory concentration of punicalagin. Rongai et al. (2019) selected a pomegranate (Punica ganatum L.) genotype with punicalagin content (α- and (β-anomers) of 4.767 mg g-1, close to the concentration of 1.25 % used here (5.73 mg g-1), and observed a 50 % inhibition of the F. oxysporum mycelial growth. In the present study, at the concentration of 1.25 % of the L. pacari extract, there was an inhibition of 35 % for the F. oxysporum BRM 028134 and of 51.2 % for the C. lindemuthianum BRM 007626.
Following the study, tests were performed to investigate the in vivo performance of the L. pacari extract in reducing the severity of common bacterial blight. Only one bacterium, selected at random, was tested, since the four investigated were completely inhibited at concentrations between 0.62 and 1.25 % of the extract in the in vitro tests.
In the first treatment, characteristic symptoms of common bacterial blight were observed, resulting in a general score of 4, which did not difer from the control. In the second treatment, characteristic symptoms of common bacterial blight were not observed, resulting in a general score of 1. The L. pacari extract, in the experimental conditions used in the second treatment, was promising, as regards reducing the severity of the common bacterial blight caused by X. axonopodis pv. phaseoli (BRM 025302).
Once again, it is necessary to highlight the gap in the literature of in vivo studies that use the L. pacari extract as a means to control this disease. However, it is believed that the phenolics found in the extract (Table 2) may be responsible for the recorded biological activity, as reported in the literature (Tocci et al. 2018, Rongai et al. 2019, García-Pérez et al. 2021).
According to Catara et al. (2021), given future climate change scenarios that predict an increase in epidemic severity and a geographical expansion of pathogens, the pressure on agrifood systems will become even more significant. Thus, we have demonstrated the potential of the L. pacari extract to control the common bacterial blight in common bean (as the second treatment) and recommend further studies to better investigate such potential in relation to this disease.
Lastly, an experiment was conducted to investigate the in vivo potential of the L. pacari extract in reducing the severity of anthracnose caused by Colletotrichum. The C. lindemuthianum BRM 007447 was selected, which is the most common isolate associated to anthracnose in common bean in Brazil. The L. pacari extract weakly reduced the disease severity for the IPA 7419 cultivar (susceptible to the disease) in the first and second treatments, in which the overall scores were 7 and 6, respectively. The overall score was 9 for the control. For the Pérola cultivar (mildly susceptible to the disease), the L. pacari extract did not reduce the disease severity in either treatment. The overall score was similar to the control.
No studies with the L. pacari extract aiming to control anthracnose in common bean were identified. However, phenolics from Caesalpinia cacalaco showed an antimicrobial activity against Colletrotrichum lindimuthianum (Veloz-García et al. 2010). These authors verified an in vitro fungistatic activity against this fungus and a good protection against anthracnose severity in in vivo assays. In this study’s HPLC analysis, gallic acid and tannic acid were identified (data not shown).
In the applied experimental conditions, the L. pacari extract was not promising for reducing the severity of anthracnose caused by the C. lindemuthianum BRM 007447. Further studies should be carried out to identify the appropriate dose that controls the disease, or even other experimental conditions tested to evaluate the extract potential in controlling anthracnose in common bean. It should be noted that in its chemical composition there are phenolic acids, favonoids and tannins, whose antimicrobial activities have been described (Tocci et al. 2018, Rongai et al. 2019, García-Pérez et al. 2021). Thus, bioproducts ofer promising perspectives for the control of plant diseases and phytopathogens.
CONCLUSIONS
- 1. For the first time, eight compounds were identified in Lafoensia pacari leaves: gentisic, caffeic, p-coumaric, protocatechuic and vanillic acids, luteolin, naringenin and quercetin;
- 2. The data showed a major in vitro antibacterial activity of the L. pacari phenolics-containing extract, especially against Curtobacterium flaccumfaciens pv. flaccumfaciens, Xanthomonas axonopodis pv. phaseoli, X. fuscans subsp. fuscans and Xanthomonas sp. In addition, they showed the in vivo potential of this extract in controlling the common blight caused by X. axonopodis pv. phaseoli (BRM 25302) in the Pérola bean cultivar;
- 3. The L. pacari extract is not efficient in controlling anthracnose in common bean.
ACKNOWLEDGMENTS
This study was supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (Capes). The authors thank the Centro Regional para o Desenvolvimento Tecnológico e Inovação (CRTI) of the Universidade Federal de Goiás (UFG), for the LC-HRMS analysis, and also the Agência Goiana de Assistência Técnica, Extensão Rural e Pesquisa Agropecuária (Emater), for kindly providing the Lafoensia pacari plants.
REFERENCES
BARBOSA, F. R; GONZAGA, A. C. O. Informações técnicas para o cultivo do feijoeiro-comum na região central-brasileira: 2012-2014. Santo Antônio de Goiás: Embrapa Arroz e Feijão, 2012. (Documentos, 272).
CARDOSO, J. E. Doenças do feijoeiro causadas por patógenos de solo. Santo Antônio de Goiás: Embrapa-CNPAF, 1990. (Documentos, 30).
CARNEIRO, C. C.; SANTOS, S. C.; LINO JUNIOR, R. S.; BARA, M. T. F.; CHAIBUB, B. A.; REIS, P. R. M.; CHAVES, D. A.; SILVA, A. J. R.; SILVA, L. S.; SILVA, D. M.; CHEN-CHEN, L. Chemopreventive effect and angiogenic activity of punicalagin isolated from leaves of Lafoensia pacari A. St.-Hil. Toxicology and Applied Pharmacology, v. 310, n. 1, p. 1-8, 2016.
CARVALHO, J. C. B.; CHAIBUB, A. A.; SOUSA, K. C. I.; BRITO, D. C.; FILIPPI, M. C. C.; KATO, L.; ARAUJO, L. G. Efficiency of a new Waitea circinata extract against rice pathogens. Pesquisa Agropecuária Tropical, v. 51, e66916, 2021.
CATARA, V.; CUBERO, J.; POTHIER, J. F.; BOSIS, E.; BRAGARD, C.; DERMIĆ, E.; HOLEVA, M. C.; JACQUES, M. A.; PETTER, F.; PRUVOST, O.; ROBÈNE, I.; STUDHOLME, D. J.; TAVARES, F.; VICENTE, J. G.; KOEBNIK, R.; COSTA, J. Trends in molecular diagnosis and diversity studies for phytosanitary regulated Xanthomonas. Microorganisms, v. 9, n. 4, p. 862-893, 2021.
CENTRO INTERNACIONAL DE AGRICULTURA TROPICAL (CIAT). Informe anual 1988: programa de frijol. Cali: CIAT, 1990. (Documento de trabajo, 72).
CHAVES, G. La antracnosis. In: SCHWARTZ, H. F.; GÁLVEZ, G. E. Problemas de producción del fríjol: enfermedades, insectos, limitaciones, edáficas y climáticas de Phaseolus vulgaris. Cali: CIAT, 1980. p. 37-54.
CHIORCEA-PAQUIM, A. M.; ENACHE, T. A.; SOUZA, E. G. de; OLIVEIRA-BRETT, A. M. Natural phenolic antioxidants electrochemistry: towards a new food science methodology. Comprehensive Reviews in Food Science and Food Safety, v. 19, n. 4, p. 1680-1726, 2020.
CÔRTES, M. V. C. B.; LIMA, D. C. S.; SILVA-LOBO, V. L.; FILIPPI, M. C. C.; PRABHU, A. S. Inibição do desenvolvimento micelial de Magnaporthe oryzae por metabólito extracelular produzido por Sarocladium oryzae. Santo Antônio de Goiás: Embrapa Arroz e Feijão, 2012. (Boletim de pesquisa e desenvolvimento, 37).
GARCIA, S. A. S.; ROCHA, P. B. R.; SOUZA, B. S.; PAZ, A. T. S.; NEGRIS, A. L. C.; MARRETO, R. N.; CONCEIÇÃO, E. C.; BARA, M. T. F.; TAVEIRA, S. F. Enhanced skin permeation of punicalagin after topical application of pluronic micelles or vesicles loaded with Lafoensia pacari extract. Planta Medica, v. 88, n. 6, p. 479-488, 2021.
GARCÍA-PÉREZ, P.; AYUSO, A.; LOZANO-MILO, E.; PEREIRA, C.; DIAS, M. I.; IVANOV, M.; CALHELHA, R. C.; SOKOVIC, M.; FERREIRA, I. C. F. R.; BARROS, L.; GALLEGO, P. P. Phenolic profling and in vitro bioactivities of three medicinal Bryophyllum plants. Industrial Crops and Products, v. 162, e113241, 2021.
HAGEDORN, D. J.; INGLIS, D. A. Handbook of bean diseases. Madison: University of Wisconsin, 1986.
ITAKO, A. T.; TOLENTINO JUNIOR, J. B.; RADUAN, J. L. F. P.; MATTOS, A. P.; SANTOS, K. L.; CIOTTA, M. N. Efect of essential oils on the development of Colletotrichum sp. fungus in fragments of Feijoa sellowiana fruits. Acta Scientiarum Biological Sciences, v. 43, e53055, 2021.
OSORIO, P.; FLORES, M.; HERNÁNDES, D.; VENTURA, J.; RODRÍGUEZ, R.; AGUILAR, C. N. Biological efficiency of polyphenolic extracts from pecan nuts shell (Carya Illinoensis), pomegranate husk (Punica granatum) and creosote bush leaves (Larrea tridentata Cov.) against plant pathogenic fungi. Industrial Crops and Products, v. 31, n. 1, p. 153-157, 2010.
PAIVA, B. A. R.; WENDLAND, A.; TEIXEIRA, N. C.; FERREIRA, M. A. S. V. Rapid detection of Xanthomonas citri pv. fuscans and Xanthomonas phaseoli p v. phaseoli in common bean by loop-mediated isothermal amplification. Plant Disease, v. 104, n. 1, p. 198-203, 2020.
PEREIRA, L. O. M.; VILEGAS, W.; TANGERINA, M. M. P.; ARUNACHALAM, K.; BALOGUN, S. O.; ORLANDI-MATTOS, P. E.; COLODEL, E. M.; MARTINS, D. T. D. Lafoensia pacari A. St.-Hil.: wound healing activity and mechanism of action of standardized hydroethanolic leaves extract. Journal of Ethnopharmacology, v. 219, n. 1, p. 337-350, 2018.
RAVA, C. A. Patogenicidade de isolados de Xanthomonas campestres pv. phaseoli. Pesquisa Agropecuária Brasileira, v. 19, n. 4, p. 445-448, 1984.
RAVA, C. A.; MOLINA, J.; KAUFFMANN, M.; BRIONES, I. Determinación de razas fisiológicas de Colletotrichum lindemuthianum en Nicaragua. Fitopatologia Brasileira, v. 18, n. 3, p. 388-391, 1993.
RONGAI, D.; SABATINI, N.; PULCINI, P.; DI MARCO, C.; STORCHI, L.; MARRONE, A. Efect of pomegranate peel extract on shelf life of strawberries: computational chemistry approaches to assess antifungal mechanisms involved. Journal of Food Science and Technology, v. 55, n. 7, p. 2702-2711, 2018.
RONGAI, D.; PULCINI, P.; DI LERNIA, G.; NOTA, P.; PREKA, P.; MILANO, F. Punicalagin content and antifungal activity of different Pomegranate (Punica ganatum L.) genotypes. Horticulturae, v. 5, n. 3, p. 52-61, 2019.
SILVA, O. F. Dados de conjuntura da produção de feijão comum (Phaseolus vulgaris L.) e caupi (Vigna unguiculata (L.) Walp) no Brasil (1985 a 2019). Santo Antônio de Goiás: Embrapa Arroz e Feijão, 2020.
SILVA, T. C.; ZARA, A. L. S. A.; SÁ, F. A. S.; BARA, M. T. F.; AVILA, R. I.; COSTA, C. R.; VALADARES, M. C.; SANTOS, A. S.; FREITAS, V. A. Q.; SILVA, M. R. R. Antifungal potential of punicalagin against Cryptococcus neoformans species complex. Revista do Instituto de Medicina Tropical de São Paulo, v. 60, e60, 2018.
TOCCI, N.; WEIL, T.; PERENZONI, D.; NARDUZZI, L.; MADRIÑÁN, S.; CROCKETT, S.; NÜRK, N. M.; CAVALIERI, D.; MATTIVI, F. Phenolic profle, chemical relationship and antifungal activity of Andean Hypericum species. Industrial Crops and Products, v. 112, n. 1, p. 32-37, 2018.
VALDO, S. C. D.; WENDLAND, A.; ARAÚJO, L. G.; MELO, L. C.; PEREIRA, H. S.; MELO, P. G.; FARIA, L. C. Diferential interactions between Curtobacterium flaccumfaciens pv. flaccumfaciens and common bean. Genetics and Molecular Research, v. 15, n. 4, egmr15048712, 2016.
VALENTINI, G.; GUIDOLIN, A. F.; BALDISSERA, J. N. C.; COIMBRA, J. L. M. Curtobacterium flaccumfaciens pv. flaccumfaciens: etiologia, detecção e medidas de controle. Biotemas, v. 23, n. 4, p. 1-8, 2010.
VARJANI, S. J.; TAHERZADEH, M. J.; KHANAL, S. K.; PANDEY, A. New horizons in biotechnology: advances in sustainable industrial and environmental bioprocesses and bioproducts. Industrial Crops and Products, v. 158, n. 15, e113000, 2020.
VELOZ-GARCÍA, R.; MARÍN-MARTÍNEZ, R.; VELOZ-RODRÍGUEZ, R.; RODRÍGUEZ-GUERRA, R.; TORRES-PACHECO, I.; GONZÁLEZ-CHAVIRA, M. M.; ANAYA-LÓPEZ, J. L.; GUEVARA-OLVERA, L.; FEREGRINO-PÉREZ, A. A.; LOARCA-PIÑA, G.; GUEVARA-GONZÁLEZ, R. G. Antimicrobial activities of cascalote (Caesalpinia cacalaco) phenolics-containing extract against fungus Colletotrichum lindemuthianum. Industrial Crops and Products, v . 31, n. 1, p. 134-138, 2010.
WENDLAND, A.; LOBO JUNIOR, M. Integrated management of common bean diseases. In: CAMPOSVEGA, R.; BASSINELO, P. Z.; OOMAH, B. D. Integrated management of common bean diseases. New York: Nova Science, 2018. p. 115-142.