ABSTRACT: The Bacillus thuringiensis bacterium has demonstrated an effective potential in the control of several agricultural pests, among them nematodes. This study aimed to standardize and establish a methodology of bioassays of B. thuringiensis and Meloidogyne incognita with the use of resorcinol, select B. thuringiensis strains toxic to M. incognita in vitro and molecularly identify the presence of the cry6 gene in B. thuringiensis strains. Second-stage juveniles were subjected to resorcinol doses, verifying that the concentration of 0.2 % did not cause mortality and provided the Cry6A toxin ingestion. Thereafter, 16 B. thuringiensis strains were tested in the presence or absence of resorcinol, resulting in mortality rates of 4-36 %. Among the B. thuringiensis strains analyzed for the presence of the cry6 gene, only one was detected (S1617).
KEYWORDS: Ingestion inducer, cry6 gene, nematode.
RESUMO: A bactéria Bacillus thuringiensis tem demonstrado potencial no controle de diversas pragas agrícolas, dentre elas os nematoides. Objetivou-se padronizar e estabelecer uma metodologia de bioensaios de B. thuringiensis e Meloidogyne incognita com o uso de resorcinol, selecionar estirpes de B. thuringiensis tóxicas a M. incognita in vitro e identifcar molecularmente a presença do gene cry6 em estirpes de B. thuringiensis. Juvenis de segundo estágio foram submetidos a doses de resorcinol, verifcando-se que a concentração de 0,2 % não causou mortalidade e proporcionou a ingestão da toxina Cry6A. Após isso, 16 estirpes de B. thuringiensis foram testadas na presença ou não de resorcinol, resultando em mortalidades de 4-36 %. Dentre as estirpes de B. thuringiensis analisadas quanto à presença do gene cry6, detectou-se apenas uma (S1617).
PALAVRAS-CHAVE: Indutor de ingestão, cry6 gene, nematoide.
Research Article
Selection of Bacillus thuringiensis strains toxic to Meloidogyne incognita
Seleção de estirpes de Bacillus thuringiensis tóxicas a Meloidogyne incognita
Received: 09 June 2022
Accepted: 23 September 2022
Published: 27 October 2022
Agricultural losses caused by nematodes are increasingly frequent in Brazil. Nematodes of the Meloidogyne genus constitute the most important group of phytoparasites in the world, with more than 90 species described (Karssen & Moens 2006) and more than 3,000 host plants, among cultivable plants and weeds, representing a threat to the world agricultural production (Perry et al. 2009, Xiang et al. 2018). In Brazil, Meloidogyne incognita (Kofoid and White 1919), Chitwood, 1949 (races 1, 2, 3 and 4), M. javanica and M. arenaria predominate, being present in any type of soil, mainly in sandy soils and with temperatures above 25 ºC (Pinheiro et al. 2010).
The main symptom of M. incognita infection is the formation of typical galls in the roots of susceptible host plants (Xiang et al. 2018), resulting in a lower absorption of water and nutrients and presenting weakened plants, of smaller stature, withered at certain times of the day, and with low yield (Priya et al. 2011,Zeng et al. 2018, Sikandar et al. 2019). These nematodes are endophytic, sedentary and have a fast reproduction rate. Therefore, their management depend on crop rotation with non-host plants, what is not interesting from the producer’s point of view. Chemical control has been the main control strategy used; however, it presents variable results in the field, high costs and negative impacts on the environment (Engelbrecht et al. 2018) and, as such, many of these products that had a non-tolerable level of toxicity to non-target organisms were withdrawn from use (Oka 2014, Soltanzadeh et al. 2016). In addition, biological control has shown considerable efficacy results by several phytopathogenic nematode control microorganisms and by different forms of action (Engelbrecht et al. 2018, Oliveira et al. 2019, Ramalakshmi et al. 2020).
Among bacteria, Bacillus has a great potential in agricultural pest control, since it is a microorganism present in various ecological niches, isolated from various environments, especially the soil, with a direct association with nematodes (Li et al. 2007). In the Bacillus genus, B. thuringiensis (Bt) Berliner, 1915, represents the most successful commercial biopesticide in the biological control market, due to the great diversity of isolated and identified strains. In addition to the spectrum of action that encompasses several agricultural pests, nematodes and public health vectors, B. thuringiensis has an easy mass reproduction with the use of different technologies and specific action for its target pests, being harmless to humans, vertebrates, beneficial plants and insects, as well as completely biodegradable (Bravo 2018, Gutierrez et al. 2019).
Cry proteins produced by B. thuringiensis are classified into families from Cry1 to Cry78, based on their amino acid sequence identity (Crickmore et al. 2018). Among these known Cry toxin families, Cry5, Cry6, Cry12, Cry13, Cry14 and Cry21 demonstrated nematicidal activity (Guo et al. 2008). Transgenic plants that express the cry5b (Li et al. 2008) and cry6a (Li et al. 2007) genes significantly reduce the reproductive capacity of M. incognita and the number of galls in plant roots. Thus, the isolation and sequencing of cry genes should be encouraged in the search for new toxins that can be used in the biological control of pests or in the genetic transformation of plants.
The search for toxic proteins, regardless of the target, undergoes in vitro pathogenicity bioassays. In the case of nematodes, it is necessary to ingest the proteins to know their toxicity. In this way, in order to induce the ingestion of proteins, the root extract of plants, phenolic compounds such as resorcinol (Zhang et al. 2012) or carbamoylcholine chloride (Adam et al. 2008) have been used, or tested on model microorganisms such as the free-living bacteriophage nematode Caenorhabditis elegans (Montalvão et al. 2018, Verduzco-Rosas et al. 2021).
Resorcinol is a phenolic compound described as a neurostimulant known to induce the impulse of the stylet along with the accumulation of esophageal gland secretions, allowing the absorption of substances present in the solution by the nematode (Jaubert et al. 2002, Williamson & Gleason 2003). Huang et al. (2006) used resorcinol to stimulate dsRNA uptake during the RNAi (interfering RNA) process in vitro in second stage juveniles (J2) of M. incognita.Zhang et al. (2012), on the other hand, perfected the bioassay protocol involving B. thuringiensis and M. hapla using resorcinol as an inducer of protein intake to replace root exudates of tomato plants; but the dose used in these assays was not compatible with M. incognita. However, adverse effects were reported when stimulants were used to induce the intake of substances, from changes in mobility (Goto et al. 2010) to even the death of nematodes (Adam et al. 2008).
Therefore, this study aimed to standardize a methodology of bioassays of B. thuringiensis with M. incognita and the use of resorcinol, select strains of B. thuringiensis toxic to M. incognita in in vitro assays and molecularly identify the presence of the cry6 gene in strains of B. thuringiensis with potential to be used in the biocontrol of nematodes.
The experiments were carried out from 2017 to 2018, at the Embrapa Recursos Genéticos e Biotecnologia (Brasília, DF, Brazil). Sixteen strains of Bacillus thuringiensis (Bt) belonging to the Invertebrate Bacteria Collection were used: S09, S26, S53, S906, S1295, S1577, S1617, S1615, S1620, S1930, S2493, S2538, S2548, S2557, S2558 and S2560.
The strains were grown using Embrapa medium (Monnerat et al. 2007) at 28 ± 2 ºC for 72 h, in a rotary incubator at 200 rpm. After this culture period, the morphology of the strains was analyzed with a phase contrast microscope to observe the presence of spores and protein crystals.
The initial inoculum of Meloidogyne incognita race 3 was provided by the Embrapa. The nematodes were multiplied in tomato (Solanum lycopersicum L. cv. Santa Clara) for 3 months in a greenhouse. The eggs were obtained using the technique of Hussey & Barker (1973), modified by Boneti & Ferraz (1981). Surface disinfestation of the eggs was performed in a laminar flow chamber (Zuckerman & Brzeski 1966). The eggs were suspended in 30 mL of 0.12 % chlorhexidine gluconate solution along with antibiotics (10 μg mL-1 of erythromycin and 2.5 g L-1 of streptomycin) for 30 min. Then, they were centrifuged (3 min; 360 g) and, after that, the supernatant was discarded and the eggs present in the pellet were suspended in sterile distilled water. The procedure was repeated twice in a sterile environment, where the eggs were placed in a modified Baermann funnel (Flegg 1967) for incubation. The M. incognita J2 individuals that hatched within 24-48 h were collected for use in the experiments.
The aforementioned strains were used in the in vitro pathogenicity test. In 6-cell plates, 1,680 μL of the nematode suspension obtained from the hatching chamber were added, standardized with approximately 45 juveniles J2 of M. incognita and 320 μL (16 %) of the bacterial suspensions of Bt, totaling 2 mL, with the J2 incubated only in sterile distilled water in the control treatment. The plates were sealed with plastic flm and kept in a BOD type chamber at 25 ºC, in the absence of light, for 48 h. After the incubation period, the evaluation was performed, adding 10 µL of 1 N NaOH (Xiang & Lawrence 2016) to the solution, in order to help diferentiate the paralyzed or immobile nematodes from those that were actually dead. The counts were performed under an optical microscope and Petters slide, determining the percentages of dead J2.
In order to obtain the non-lethal concentration of resorcinol, tests were performed with different doses of the product on the obtained J2, as previously described. For this, different solutions of resorcinol diluted in distilled water (v/v) were prepared in pre-established proportions. Given the regression curve generated by the average of the tests, it was found that the concentration of 0.2 % did not cause toxicity to the nematodes. From this moment on, new assays were carried out to evaluate whether this concentration would provide the ingestion of the Cry6Aa protein, described as a nematicide (Wei et al. 2003, Guo et al. 2008, Palma et al. 2014), obtained from a genetically transformed strain of Bt.
To this end, the mortality of nematodes was evaluated after 48 h in contact with the bacterial suspension of Bt (Cry6Aa), at a concentration of 16 %, 10 % of resorcinol (0.2 %) and 74 % of nematode suspension containing approximately 45 juveniles J2 per mL. In the control treatment, the J2 were incubated in sterile distilled water and the material incubated and evaluated after 48 h. After standardization, all B. thuringiensis strains were retested in the presence of resorcinol, following this methodology.
To determine the non-toxic dose of resorcinol, regression analysis was performed with increasing doses of the product and mortality rate, using the average of 3 trials and with 4 repetitions for each dose.
The mean mortality data of M. incognita with the use of B. thuringiensis strains associated or not with resorcinol were submitted to analysis of variance and compared by the Scott-Knott test at the level of 5 % for the formation of groups between the tested treatments.
DNA from the strains was extracted using the methodology described by Bravo et al. (1998), with the bacteria cultured using the Embrapa solid medium (Monnerat et al. 2007) for 16 h, at 30 ºC. After this period, for each sample, the bacterial growth was collected with a loop and transferred to a properly identified 1.5-mL polyethylene tube containing 200 μL of sterile ultra-purified water (MiliQ). The samples were then homogenized in a Vortex apparatus and frozen at -80 ºC for 20 min, boiled in bain-marie at 100 ºC for 10 min, and finally incubated on ice for 2 min. The obtained supernatant was used for reactions, then 5 μL of the DNA from each strain were transferred to a 0.2-mL polyethylene tube containing 0.5 μM of the oligonucleotide, 0.2 mM dNTP, 1X Ta q buffer and 2.5 U of Ta q DNA polymerase (5.0 U), totaling a final volume of 25 μL. The characteristics of the initiator used for the reaction are described in Table 1. The PCR program used for the amplification was 94 ºC for 5 min, 35 cycles of 94 ºC for 1 min, annealing of 48.5 ºC for 1 min, 72 ºC for 1 min and 30 s, as well as a final extension of 72 ºC for 5 min.

For the analysis of the result, 25 μL of the PCR product mixed with 5 μL of 10X running buffer in 1.5 % agarose gel were applied. The electrophoresis run was done in 1X TBE buffer (Tris base; boric acid; 0.5 M EDTA; pH 8.0). After electrophoresis, the gel was stained with ethidium bromide at 1 μg mL-1 for 20 min, and decolored in distilled water for 15 min. The gel was visualized in a transluminator under UV light and photographed in a photo-documenter.
Figure 1 shows the percentage of resorcinol concentration associated with M. incognita, considering a maximum mortality of 15 % in the tests. Mortality values increased as the product concentration increased. Adam et al. (2008) also found that resorcinol doses greater than 0.5 % cause lethality in M. javanica J2, what was also observed for M. incognita during this assay.
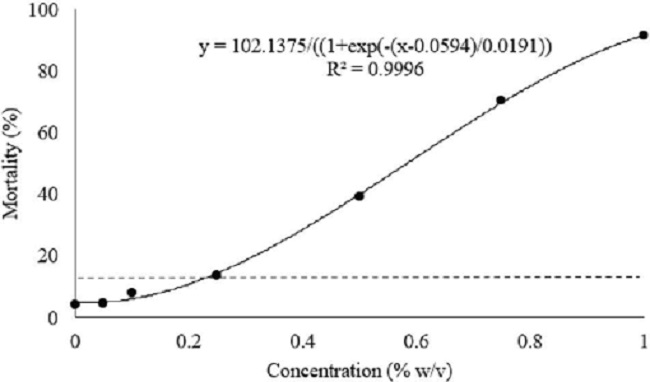
Considering the prescribed mortality limit, a value of 0.2 % was adopted for subsequent validation tests. From this point, it was tested if this selected concentration would maintain the neurostimulant properties of the product. In the following trials, where resorcinol was associated with the bacterial suspension of a genetically modified B. thuringiensis strain, expressing only the nematicidal protein Cry6Aa, there was a 3.5-fold higher mortality, when compared to the treatment of the same strain without resorcinol (Table 2). On the other hand, the control treatment with resorcinol alone presented only 4.67 % of mortality, well below the 15 % pre-established as a mortality limit for the control.

The presence of resorcinol stimulated the intake of nematicidal proteins that were in the solution, improving the nematicidal activity, considering that there was a 3.5-fold increase in mortality. Zhang et al. (2012) obtained similar results for M. hapla, verifying an increase in the nematicidal activity with resorcinol, when compared to tomato root exudates.
Of the 16 strains of B. thuringiensis tested against M. incognita in the presence or absence of resorcinol as an inducer of protein intake (Table 3), it was found that, in the absence of resorcinol, 8 strains (S1615, S1577, S1617, S2538, S1620, S1295, S53 and S2493) were statistically equal to the control treatment, not presenting satisfactory mortality results. Seven other strains presented intermediate results, difering statistically from the control group and presenting an intermediate mortality rate between 8.81 and 17.04 % (S09, S2548, S2557, S2558, S2560, S26 and S906). Finally, the highest mortality obtained without the use of resorcinol was achieved by the S1930 strain, with 35.7 %.
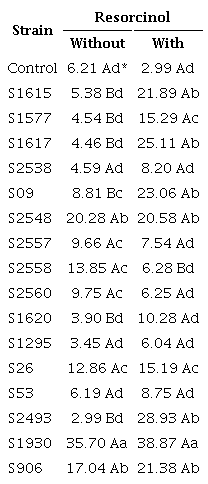
By analyzing the column of the treatments tested with the addition of resorcinol, it is clear that, in general, the mortality rates in the presence of resorcinol were higher than those found in the absence of the product, or statistically equal, except for the S2557 strain, which had its mortality reduced in the presence of the product, probably interfering negatively in some substance produced by the strain.
The S2493 strain, which presents the nematicidal gene cry6, which encodes the Cry6 toxin and presented a low mortality rate (2.99 %), stood out in the absence of resorcinol, appearing in the intermediate control group (28.93 %). With the use of resorcinol, taking into account that only the bacterial suspension was used, without any type of concentration and purification technique aiming to obtain only the crystals produced by the strain, it is assumed that the mortality result could be much higher.
Another strain that showed a large increase (greater than 5 times) in mortality was S1617. Subsequently, it was detected that the strain showed a positive result for the PCR product with the cry6 primer, indicating that this is the reason for the mortality expansion.
The strain that caused the highest percentage of mortality in the in vitro tests was S1930, not being influenced by the presence of resorcinol, although its use caused a higher mortality. This strain also did not present the PCR product with the cry6 primer previously tested. It can be seen, then, in comparison with the strains that presented the nematicidal protein, that, in the presence of resorcinol, there is a large increase in mortality, as observed in the strains S2493 and S1617, indicating that the probable cause of mortality for the strain S1930 was not a Cry protein, but some other toxic metabolite produced by the strain, which was later verified with the sequencing of this strain.
Zheng et al. (2016) demonstrated that 120 spore-forming Bacillus strains from different families share virulence factors, what may contribute to their nematicidal capacity. B. thuringiensis, B. cereus, B. subtilis, B. pumilus, B. frmus, B. toyonensis, Lysinibacillus sphaericus, Brevibacillus laterosporus and B. brevis were highly nematicidal, with the first of them demonstrating the highest activity.
Jouzani et al. (2008) observed in vitro that the concentration of spore-crystal mixtures of 22 selected strains provided an average mortality 10 % higher in M. incognita with a concentration 2 times the standard. The strains with the best performances presented toxicity of 77 and 81 %, respectively, for the combination of spores and crystals.
Verduzco-Rosas et al. (2021), testing 310 strains of B. thuringiensis, using C. elegans as a model, found that 10 strains showed toxicity results in spore and crystal concentration. Of these, two strains produced damage to the body and intestine of the nematode. Molecular analysis also revealed the presence of cry5B, cry14A, cry21A and cyt1A genes and the new cry gene.
The results revealed a very low occurrence of the nematicidal gene cry6 in the selected strains. As previously mentioned, only the S1617 strain presented the cry6 gene. According to the methodology used in the present study, only the detection of the cry6 gene was sought; thus, it cannot be said that the used strains are not producers of other nematicidal proteins.
Of the 70 Bt isolates tested by Jouzani et al. (2008), using 12 primers specific for the nematicidal genes cry5, cry6, cry12, cry13, cry14 and cry21, the authors found 22 isolates (31.5 %) which contain at least one cry gene active against nematodes. Strains containing the cry6 gene were the most abundant and represent 22.8 % of the isolates.
On the other hand, Bravo et al. (1998), analyzing multiplex PCR-based with general and specific primers, found a total of 496 samples of B. thuringiensis, which were isolated from 503 soil samples collected from the five macro-regions of Mexico, of which the proteins are nematicidal and were analyzed in this study (Cry5, Cry12, Cry13 and Cry14), where no strains with the cry5, cry12, cry13, cry14 or cry21 genes were found.
Although soil is the main source of most B. thuringiensis isolates, and they still have the ability to present several cry genes in their molecular profile, their geographical and ecological distribution are very variable, that is, it is not possible to establish a correlation between geographical distribution and frequency of occurrence of cry genes of nematicidal activity.
This study was partially financed by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (Capes) (Finance code 001).



