Research Article
Water deficit affects leaf non-structural carbohydrates and biomass partitioning in chickpea1
Déficit hídrico afeta carboidratos não estruturais foliares e partição de biomassa em grão-de-bico
Water deficit affects leaf non-structural carbohydrates and biomass partitioning in chickpea1
Pesquisa Agropecuária Tropical, vol. 54, e79442, 2024
Escola de Agronomia/UFG
Received: 31 May 2024
Accepted: 19 September 2024
Published: 11 October 2024
Funding
Funding source: CNPq
Contract number: 312854
Funding
Funding source: Capes
Contract number: 88887.688626/2022-00
Funding statement: To the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), for the productivity grant to the researcher Marcio Mesquita (312854); and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (Capes), for the scholarship to Elen P. P. Bento-da-Silva (88887.688626/2022-00).
ABSTRACT: Water deficit induces morphophysiological adjustments in plants, what includes changes in biomass partitioning, non-structural carbohydrates and their ratios, reducing stomatal conductance and photosynthesis, and affecting the synthesis of transitory starch in leaves. This study analyzed the impact of soil water tensions on leaf non-structural carbohydrates and biomass partitioning during chickpea cultivation. The BRS Aleppo and BRS Toro genotypes were grown at soil water potentials of -30, -50, -70 and -90 kPa throughout the cycle. The water status, shoot and root dry mass, non-structural carbohydrate content and carbohydrate ratios were assessed. Decreasing the soil water potential reduced the relative water content and shoot dry mass of both genotypes. However, the BRS Toro´s root dry mass increased with the reduction in the soil water potential, as did the root:shoot ratio, in both genotypes. The genotypes also showed variations in the contents of soluble carbohydrates and starch in leaves, which increased with the reduction in the soil water potential, except for starch in leaves of the BRS Toro. These findings, especially variations in the carbohydrate ratios, suggest that the starch turnover is essential in chickpeas´ resilience to water deficit.
KEYWORDS: Cicer arietinum L, soil water potential, starch.
RESUMO: O déficit hídrico induz ajustes morfofisiológicos nas plantas, o que inclui mudanças na partição de biomassa, carboidratos não estruturais e em suas proporções, reduzindo a condutância estomática e a fotossíntese, e afetando a síntese de amido transitório nas folhas. Este estudo analisou o impacto de tensões de água no solo sobre os carboidratos não estruturais das folhas e a partição de biomassa, durante o cultivo de grão-de-bico. Os genótipos BRS Aleppo e BRS Toro foram cultivados utilizando-se potenciais hídricos do solo de -30, -50, -70 e -90 kPa, durante todo o ciclo. Foram avaliados o estado hídrico, massa seca da parte aérea e raízes, conteúdo de carboidratos não estruturais e proporções de carboidratos. A diminuição do potencial hídrico do solo reduziu o conteúdo relativo de água e a massa seca da parte aérea de ambos os genótipos. No entanto, a massa seca da raiz de BRS Toro aumentou com a redução do potencial hídrico do solo, assim como a relação raiz:parte aérea, em ambos os genótipos. Os genótipos também apresentaram variações nos teores de carboidratos solúveis e amido nas folhas, que aumentaram com a redução do potencial hídrico do solo, com exceção do amido nas folhas da BRS Toro. Essas constatações, especialmente as variações nas proporções de carboidratos, sugerem que o turnover do amido é essencial para a resiliência do grão-de-bico ao déficit hídrico.
PALAVRAS-CHAVE: Cicer arietinum L, potencial hídrico do solo, amido.
INTRODUCTION
Chickpea (Cicer arietinum L.) is a legume rich in protein and dietary fiber. For this reason, it has a high nutritional value (Jukanti et al. 2012). Throughout its growth cycle, it requires little water (up to 400 mm), responds well to dry and mild climates and is suitable for rainfed and semi-arid regions (Nascimento et al. 2016). Its deep root system enables water absorption from deeper soil layers, evidencing its high adaptability (Queiroga et al. 2021).
During the growth cycle, water availability significantly affects the development of chickpea plants and may hinder their yield (Queiroga et al. 2021, Silva et al. 2021); however, a moderate water deficit may not directly hinder its development (Kurdali et al. 2002). Thus, understanding the effects of water restrictions on chickpea physiology and biochemistry is crucial for developing effective crop management strategies, such as reducing the water use and enhancing the crop´s resistance to drought, which will in turn promote a sustainable production.
Drought may result in substantial yield losses (Foti et al. 2021). It affects the plants structure and function, inhibits growth, alters biomass partitioning, reduces leaf gas exchanges and changes signaling pathways and gene expression. These alterations induce metabolic adjustments, including osmolyte accumulation, that seek to maintain water absorption (Ozturk et al. 2021).
Osmolytes such as glucose, fructose and sucrose, along with the polysaccharide starch, are non-structural carbohydrates. These compounds are crucial for metabolism, transport and interactions with other organisms (Hartmann & Trumbore 2016). Starch is an osmotically inactive polysaccharide composed of glucose units; however, water deficit may induce plants to hydrolyze starch into osmotically active glucose (Thalmann & Santelia 2017). In leaves, photosynthesis produces a transitory starch pool, which is daily hydrolyzed to meet photoassimilate requirements at night and at low CO2 assimilation periods (Thalmann & Santelia 2017). Starch levels in source or in sinks are metabolic coordinators crucial for plant production (Sulpice et al. 2009). Additionally, non-structural carbohydrate partitioning between roots and shoots and their ratios are potential biomarkers for plant production (Purdy et al. 2015).
This study hypothesizes that reducing soil water potentials throughout chickpea cultivation increases the root overshoot biomass and, at the grain-filling phase, hydrolyzes transitory starch in leaves, thus raising the soluble carbohydrate levels. Moreover, chickpea genotypes significantly affect biomass partitioning and non-structural carbohydrates modulation under different soil water tensions.
Thus, this study aimed to assess the effects of soil water deficits throughout the crop cycle on non-structural carbohydrates in leaves and biomass partitioning of two chickpea genotypes suitable for dryland farming.
MATERIAL AND METHODS
The experiment was conducted at the Instituto Federal Goiano, in Urutaí, Goiás state, Brazil (17º29´10´S and 48º12´38´W), from December 2020 to March 2021. BRS Toro, BRS Aleppo, BRS Cristalino and BRS Cícero seeds, provided by the Embrapa Hortaliças, were pre-treated with carbendazim. Five seeds were sown at 3-cm depth in 10-L polyethylene pots containing a 3:1 mixture of soil and coarse sand. The seedlings were transplanted into individual pots, arranged in rows and spaced 50 cm apart. The soil was fertilized at planting with 250 kg ha-1 of single superphosphate, 160 kg ha-1 of potassium chloride and 60 kg ha-1 of nitrogen. At 20 days after emergence, the soil was fertilized with 20 kg ha-1 of urea.
A split-plot randomized block design, in a 4 x 4 factorial scheme, was used, with five replicates. Each replicate comprised two planting lines and a drip line between them. Soil water potential treatments were assigned to plots and genotypes to subplots. The pots were irrigated up to the field capacity for the first 20 days after emergence using a drip system. Then, irrigation was adjusted to reach the soil water potentials of -30 kPa, -50 kPa, -70 kPa and -90 kPa, until the end of the crop cycle. At the grain-filling phase, only BRS Aleppo and BRS Toro provided samples suitable for biochemical analysis and, therefore, were included in this study.
The relative water content in fully expanded leaves was determined according to Barrs & Weatherley (1962) on the 90th day post-germination. After harvest, the shoots and roots were carefully removed from the pots, cleaned and oven-dried under forced ventilation at 50 ºC, until constant weight. The root and shoot dry mass were determined gravimetrically, and then the root:shoot ratio was calculated. On the same day, fully expanded leaves were collected, wrapped in aluminum foil and frozen in liquid nitrogen. In the lab, enzymes were inactivated by microwave drying for 1 min, and the leaves were then oven-dried at 50 ºC, for 24 hours, and then ground to a fine powder.
Soluble carbohydrates were extracted according to Costa et al. (2021) by extraction steps with 80 % ethanol and distilled water. Total soluble carbohydrates were quantified using the phenol-sulphuric method (DuBois et al. 1956), and glucose was the reference standard. The extraction residue of soluble carbohydrates was oven-dried at 50 ºC, for 48 hours, for starch extraction and quantification (Amaral et al. 2007), using thermostable alpha-amylase and amyloglucosidase. The glucose released after enzymatic incubations was determined using glucose oxidase and peroxidase reagent (GOD/POD). Absorbance was read with a microplate spectrophotometer at 490 nm. The soluble carbohydrate and starch content were expressed as mg g-1 of dry mass.
Inositols, glucose, fructose and sucrose levels were determined by high-performance anion exchange chromatography with pulsed amperometric detection (HPAEC/PAD), using a CarboPac PA1 column attached to the chromatograph (ICS 5000, Dionex). The mobile phase was sodium hydroxide (100 mM) eluted at 1 mL min-1, for 20 min. Due to the inability to separate the inositol (Purdy et al. 2023), "inositol" referred to the peak that co-eluted with myo-inositol. Using calibration curves generated in the Chromeleon software, sugars were determined. Glucose and fructose were combined to calculate the hexose amounts, and the glucose:fructose, sucrose:hexoses, hexoses:starch and sucrose:starch ratios were calculated.
Normality and homoscedasticity were assessed using the Shapiro-Wilk and Bartlett tests, respectively. Two-way Anova and Tukey (p d 0.05) tests were performed for all parameters. All tests were conducted using the Easyanova package (Arnhold 2013). The graphs were plotted using the ggplot2 package (Wickham 2009). Statistical analyses were performed in the R 4.1.0 software (R Development Core Team 2021).
RESULTS AND DISCUSSION
Both genotypes had a significant relative water content reduction at -90 kPa (Table 1; Figure 1A). BRS Toro reduced less the levels than BRS Aleppo. Soil water deficit triggers numerous plant adjustments, including reduced relative water content, which is a tissue hydration measure (Bashir et al. 2021). Importantly, chickpea genotypes with higher drought tolerance generally show low relative water content reductions (Bindra et al. 2021, Tiwari et al. 2023), suggesting this trait as a selection criterion for drought-resistant chickpea crops (Talebi et al. 2013).
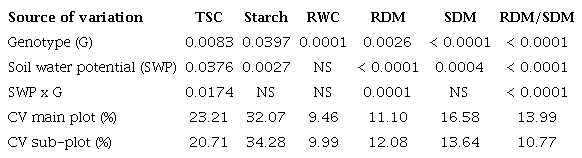
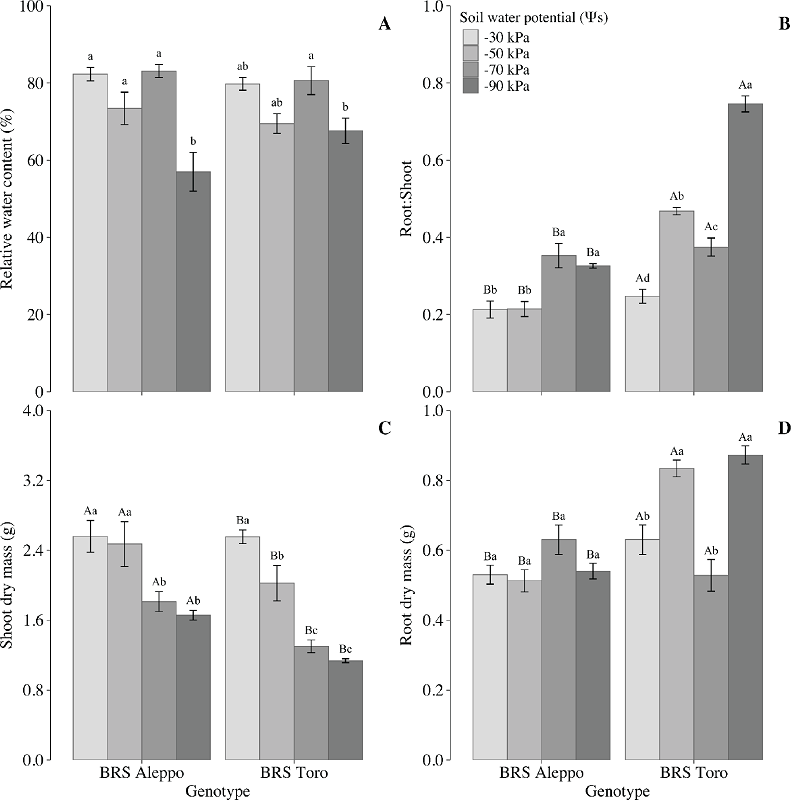
Figure 1
Effect of soil water deficits on leaf relative water content (A), root:shoot ratio (B), shoot dry mass (C) and root dry mass (D) of two chickpea genotypes. Uppercase letters mean significant differences between genotypes, and lower-case letters mean significant differences among soil water potentials by the Tukey test (p < 0.05). Means ± SE (n = 5).
The decrease in relative water content means alterations in cell turgor, which reduce growth, a typical response to drought (Bashir et al. 2021). However, the roots and shoots response to water deficit are different with the suppression of shoot activity and growth to conserve water and increase the roots´ metabolic activity and growth to optimize water absorption (Gargallo-Garriga et al. 2014).
The reduction in soil water potential decreased the shoot dry mass of both chickpea genotypes (Figure 1). This was noted from -70 kPa in BRS Aleppo and from -50 kPa in BRS Toro (Figure 1C). Conversely, the BRS Toro root dry mass increased at -50 kPa and -90 kPa, while the BRS Aleppo root dry mass remained unaffected. This modified the root:shoot ratio, with BRS Aleppo increasing at -70 and -90 kPa and BRS Toro peaking at -90 kPa (Figure 1B).
These results reveal genotype-dependent root responses to drought in chickpea (Ramamoorthy et al. 2017). They have practical implications, since root biomass is a crucial trait related to drought tolerance in chickpea crops (Asati et al. 2022). As root biomass is the numerator for determining the root:shoot ratio, an increased or minimally reduced root dry mass leads to a higher root:shoot ratio, a typical response to drought (Gargallo-Garriga et al. 2014). Such an adjustment is a drought-avoidance strategy, and evidences a shift in dry mass toward the roots, what potentially improves water uptake (Kou et al. 2022).
Non-structural carbohydrates are fundamental for plant metabolism, including respiration, water balance, growth, reproduction and defense (Hartmann & Trumbore 2016). In this study, genotype and soil water potential affected the soluble carbohydrate levels in chickpea leaves. BRS Aleppo had higher total soluble carbohydrates levels (p = 0.0376) (Figure 2A). As the soil water potential decreased, the soluble carbohydrate levels peaked at -70 kPa in BRS Aleppo. In BRS Toro, soluble carbohydrates peaked at -90 kPa, evidencing a clear relationship with soil water potential.
Figure 2
Effects of soil water potential on leaf total soluble carbohydrates (TSC; A) and starch content (B) in two chickpea genotypes. Uppercase letters represent significant differences between genotypes, and lower-case letters represent significant differences among soil water potentials by the Tukey test (p < 0.05). Means ± SE (n = 5). DM: dry mass.
The predominance of soluble carbohydrates over starch and the increase in soluble carbohydrate levels under reduced soil water potential stresses the non-structural carbohydrate, in relation to drought response, a common phenomenon in plants (Rosa et al. 2009), including other chickpea genotypes (Basu et al. 2007a, Basu et al. 2007b, Mafakheri et al. 2011). Intriguingly, Farjam et al. (2014) reported a decrease in leaf total soluble carbohydrates in chickpea under drought, underscoring the complexity of this response.
The leaf starch content showed a significant variation between the chickpea genotypes (p = 0.0027). BRS Aleppo had the highest starch content, especially at -70 kPa (Figure 2B). Typically, the leaf starch content decreases with low leaf water content (Basu et al. 2007a, Basu et al. 2007b).
In leaves, soluble carbohydrates are primary for metabolism and, as sucrose, for transport to sinks (Zepeda et al. 2022). When the leaf´s ability to assimilate carbon is limited, such as during water deficit, soluble sugar contents increase, possibly due to starch hydrolysis (Zhang et al. 2022). In fast-growing crops, the starch accumulation in vegetative tissues negatively correlates with biomass production (Sulpice et al. 2009, Purdy et al. 2015), stressing the association between low starch content and higher root dry mass in BRS Toro.
A non-structural carbohydrate content represents the balance between production by photosynthesis and consumption for growth and maintenance (Dietze et al. 2014). Environmental conditions may change the carbohydrate allocation and the plant´s development (Hartmann & Trumbore 2016). Under drought and other abiotic stresses, plants increase their soluble carbohydrate content for osmoregulation and cell signaling, enabling them to adjust to a shifting environment (Valluru & Van den Ende 2011).
Although not directly involved in osmotic regulation, starch is crucial for energy storage (Zepeda et al. 2022). In periods with lower photosynthetic activity, plants use stored starch for energy and carbon to maintain growth and metabolism, decreasing the leaf starch content (Hasan et al. 2023). The differences observed in this study, that is, BRS Toro maintaining a constant leaf starch content and increasing the root dry mass, and BRS Aleppo increasing starch at -70 kPa and an unaltered root dry mass, suggest that genotypes allocate non-structural carbohydrates differently under water deficit.
Regarding the individual sugar content, there was no significant difference in inositol content between the two chickpea genotypes at the soil water potentials (Table 2). Although they did not vary in this study, inositols are vital for plant stress responses, development and cell signaling (Valluru & Van den Ende 2011).
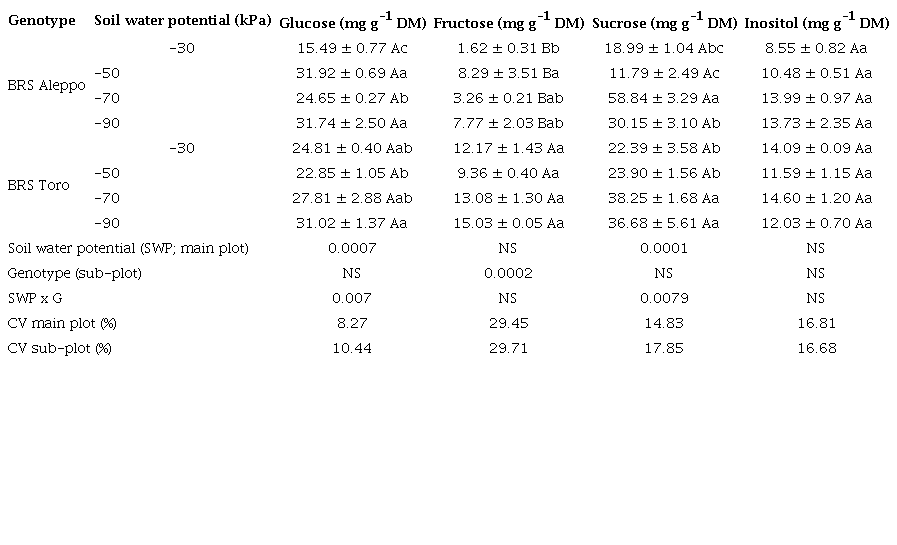
Monosaccharides are intermediates in carbohydrate metabolism used for biosynthesis or resulting from hydrolysis (Hartmann & Trumbore 2016). Among monosaccharides, glucose is a key regulator of a variety of plant cellular and developmental processes (Saksena et al. 2020). Free glucose contents were equivalent in both genotypes (p = 0.6221) and influenced by soil water potential (p = 0.0007) (Table 2). In BRS Aleppo, the free glucose was lower at -30 kPa. In BRS Toro, the free glucose levels were higher at -90 kPa, when compared to -50 kPa (Table 2). However, Basu et al. (2007b) reported that, despite the increase in free glucose at lower soil water potentials, the low glucose levels in chickpea genotypes do not significantly contribute to osmotic adjustment or maintaining relative water content.
The studied genotypes differed for fructose contents (p = 0.0002) (Table 2). BRS Toro had the highest fructose content, but the soil water potential did not affect it (Table 2). Conversely, BRS Aleppo had the highest fructose content at -50 kPa and the lowest at -30 kPa.
Free glucose levels were two to eight times greater than fructose levels. The glucose:fructose ratio, a carbohydrate metabolism indicator, varied between genotypes (p < 0.0001) and according to soil water potential (p = 0.0008) (Table 3). BRS Aleppo had a higher ratio, especially at -30 kPa and -70 kPa (Table 3). A ratio close to 1 suggests that glucose and fructose levels mainly result from sucrose metabolism, as this disaccharide consists of both monosaccharides. The observed higher ratios indicate a glucose predominance over fructose, potentially linked to a high rate of starch turnover (Purdy et al. 2015).
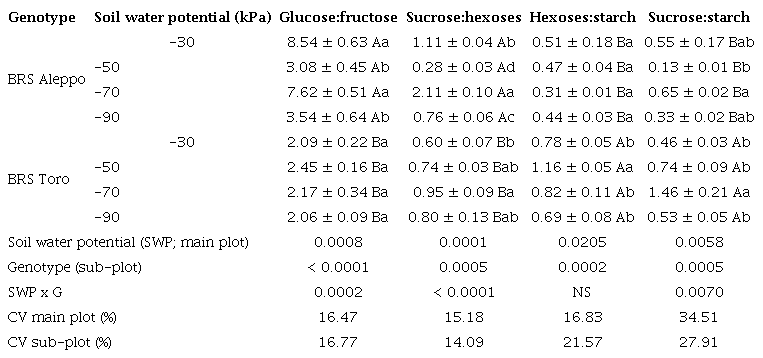
Contrary to fructose, the sucrose levels were similar in both genotypes (p = 0.889) (Table 2) and impacted by soil water potential (p = 0.0001) (Table 2). The sucrose content in BRS Aleppo peaked at -70 kPa, and the minimum was at -50 kPa. BRS Toro had maximum sucrose contents at both -70 kPa and -90 kPa (Table 2).
A low water availability usually increases sucrose and decreases plant starch (Zahoor et al. 2017, Hasan et al. 2023). However, the sucrose and starch levels in BRS Aleppo increased at -70 kPa (Figure 2; Table 2). Sucrose is fundamental for plant survival and production, translocating from leaves (sources) to active sinks, such as roots and reproductive organs (Jeandet et al. 2022). In the present study, sucrose accumulated in leaves at low soil water potential, suggesting restrictions in export. If photosynthesis remained operating in water restriction, it would lead to non-structural carbohydrate accumulation in leaves, evidencing the chickpea plants´ resilience to lower soil water potential.
The chickpea genotypes differed for sucrose:hexoses ratio (p = 0.0008), influenced by the soil water potentials (p = 0.0001) (Table 3). BRS Aleppo had higher sucrose:hexoses ratios than BRS Toro, peaking at -70 kPa, whereas those for BRS Toro increased as the soil water potential decreased (Table 3). These results align with other studies that reported starch and sucrose catabolism under stress, increasing reducing sugars in chickpea (Basu et al. 2007a, Basu et al. 2007b, Chandel & Sharma 2023).
The hexoses:starch ratio differed for the chickpea genotypes (p = 0.0002) and was affected by the soil water potentials (p = 0.0205) (Table 3). BRS Toro had the highest ratio and peaked at -50 kPa, while BRS Aleppo showed no variation among the soil water potentials.
BRS Toro had a higher sucrose:starch ratio (p = 0.0005), influenced by the soil water potentials (p = 0.0058), peaking at -70 kPa for both genotypes. These ratios result from the dynamics between energy requirements (sucrose and hexoses) and energy availability (starch). Values lower than 1 suggest the gradual hydrolysis of reserves as the soil water potential decreases.
CONCLUSIONS
-
1. Reducing the soil water tensions during chickpea cultivation changes the plant water status, represented by a decrease in the leaf relative water content, which induces a change in the dry matter partitioning by increasing the root:shoot ratio and modulates the leaf non-structural carbohydrate levels by increasing soluble carbohydrates associated with leaf starch turnover;
-
2. Differential carbohydrate allocation among chickpea genotypes in response to water deficit has potential implications for selecting genotypes for environments with low water availability.
ACKNOWLEDGMENTS
To the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), for the productivity grant to the researcher Marcio Mesquita (312854); and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (Capes), for the scholarship to Elen P. P. Bento-da-Silva (88887.688626/2022-00).
REFERENCES
AMARAL, L. I. V.; GASPAR, M.; COSTA, P. M. F.; AIDAR, M. P. M.; BUCKERIDGE, M. S. Novo método enzimático rápido e sensível de extração e dosagem de amido em materiais vegetais. Hoehnea, v. 34, n. 4, p. 425-431, 2007.
ARNHOLD, E. Pacote em ambiente R para análise de variância e análises complementares. Brazilian Journal of Veterinary Research and Animal Science, v. 50, n. 6, p. 488-492, 2013.
ASATI, R.; TRIPATHI, M. K.; TIWARI, S.; YADAV, R. K.; TRIPATHI, N. Molecular breeding and drought tolerance in chickpea. Life, v. 12, n. 11, e1846, 2022.
BARRS, H.; WEATHERLEY, P. A re-examination of the relative turgidity technique for estimating water deficits in leaves. Australian Journal of Biological Sciences, v. 15, n. 3, p. 413-428, 1962.
BASHIR, S. S.; HUSSAIN, A.; HUSSAIN, S. J.; WANI, O. A.; NABI, S. Z.; DAR, N. A.; BALOCH, F. S.; MANSOOR, S. Plant drought stress tolerance: understanding its physiological, biochemical and molecular mechanisms. Biotechnology & Biotechnological Equipment, v. 35, n. 1, p. 1912-1925, 2021.
BASU, P. S.; ALI, M.; CHATURVEDI, S. K. Osmotic adjustment increases water uptake, remobilization of assimilates and maintains photosynthesis in chickpea under drought. Indian Journal of Experimental Biology, v. 45, n. 3, p. 261-267, 2007a.
BASU, P. S.; BERGER, J. D.; TURNER, N. C.; CHATURVEDI, S. K.; ALI, M.; SIDDIQUE, K. H. M. Osmotic adjustment of chickpea (Cicer arietinum) is not associated with changes in carbohydrate composition or leaf gas exchange under drought. Annals of Applied Biology, v. 150, n. 2, p. 217-225, 2007b.
BINDRA, S.: SINGH, I.: SINGH, S.: KUSHWAH, A.: GILL, B. S.: SALARIA, S.; KAPOOR, K.; GREWAL, S. K.; BHARADWAJ, C.; NAYYAR, H.; SINGH, S. Use of morpho-physiological and biochemical traits to identify sources of drought and heat tolerance in chickpea (Cicer arietinum). Crop and Pasture Science, v. 72, n. 10, p. 801-814, 2021.
CHANDEL, S. S.; SHARMA, K. D. Down-regulation of carbohydrate metabolic pathway genes lowers sucrose and starch content in chickpea leaves under high temperature stress. National Academy Science Letters, v. 46, n. 5, p. 445-449, 2023.
COSTA, N. B.; BEZERRA, G. A.; PINHEIRO-FILHO, G. O.; MORAES, M. G. Distribution of non-structural carbohydrates in the vegetative organs of upland rice. Ciência e Agrotecnologia, v. 45, e008721, 2021.
DIETZE, M. C.; SALA, A.; CARBONE, M. S.; CZIMCZIK, C. I.; MANTOOTH, J. A.; RICHARDSON, A. D.; VARGAS, R. Nonstructural carbon in woody plants. Annual Review of Plant Biology, v. 65, n. 1, p. 667-687, 2014.
DUBOIS, M.; GILLES, K. A.; HAMILTON, J. K.; REBERS, P. A.; SMITH, F. Colorimetric method for determination of sugars and related substances. Analytical Chemistry, v. 28, n. 3, p. 350-356, 1956.
FARJAM, S.; SIOSEMARDEH, A.; KAZEMI-ARBAT, H.; YARNIA, M.; ROKHZADI, A. Response of chickpea (Cicer arietinum L.) to exogenous salicylic acid and ascorbic acid under vegetative and reproductive drought stress conditions. Journal of Applied Botany and Food Quality, v. 87, n. 1, p. 80-86, 2014.
FOTI, C.; KALAMPOKIS, I. F.; ALIFERIS, K. A.; PAVLI, O. I. Metabolic responses of two contrasting lentil genotypes to peg-induced drought stress. Agronomy, v. 11, n. 6, e1190, 2021.
GARGALLO-GARRIGA, A.; SARDANS, J.; PÉREZ-TRUJILLO, M.; RIVAS-UBACH, A.; ORAVEC, M.; VECEROVA, K.; URBAN, O.; JENTSCH, A.; KREYLING, J.; BEIERKUHNLEIN, C.; PARELLA, T.; PEÑUELAS, J. Opposite metabolic responses of shoots and roots to drought. Scientific Reports, v. 4, n. 1, e6829, 2014.
HARTMANN, H.; TRUMBORE, S. Understanding the roles of nonstructural carbohydrates in forest trees: from what we can measure to what we want to know. New Phytologist, v. 211, n. 2, p. 386-403, 2016.
HASAN, M. D. M.; ALABDALLAH, N. M.; SALIH, A. M.; AL-SHAMMARI, A. S.; ALZAHRANI, S. S.; AL LAWATI, A. H.; JAHAN, M. S.; RAHMAN, M. A.; FANG, X. W. Modification of starch content and its management strategies in plants in response to drought and salinity: current status and future prospects. Journal of Soil Science and Plant Nutrition, v. 23, n. 1, p. 92-105, 2023.
JEANDET, P.; FORMELA-LUBOICSKA, M.; LABUDDA, M.; MORKUNAS, I. The role of sugars in plant responses to stress and their regulatory function during development. International Journal of Molecular Sciences, v. 23, n. 9, e5161, 2022.
JUKANTI, A. K.; GAUR, P. M.; GOWDA, C. L. L.; CHIBBAR, R. N. Nutritional quality and health benefits of chickpea (Cicer arietinum L.): a review. British Journal of Nutrition, v. 108, n. S1, p. S11-S26, 2012.
KOU, X.; HAN, W.; KANG, J. Responses of root system architecture to water stress at multiple levels: a meta-analysis of trials under controlled conditions. Frontiers in Plant Science, v. 13, e1085409, 2022.
KURDALI, F.; AL-AIN, F.; AL-SHAMMA, M. Nodulation, dry matter production and N2 fixation by fababean and chickpea as affected by soil moisture and potassium fertilizer. Journal of Plant Nutrition, v. 25, n. 2, p. 355-368, 2002.
MAFAKHERI, A.; SIOSEMARDEH, A.; BAHRAMNEJAD, B.; STRUIK, P. C.; SOHRABI, Y. Effect of drought stress and subsequent recovery on protein, carbohydrate contents, catalase and peroxidase activities in three chickpea (Cicer arietinum) cultivars. Australian Journal of Crop Science, v. 5, n. 10, p. 1255-1260, 2011.
NASCIMENTO, W. M.; SILVA, P. P.; ARTIAGA, O. P.; SUINAGA, F. A. Grão-de-bico. In: NASCIMENTO, W. M. (ed.). Hortaliças leguminosas. Brasília, DF: Embrapa, 2016. p. 89-118.
OZTURK, M.; TURKYILMAZ-UNAL, B.; GARCÍA-CAPARRÓS, P.; KHURSHEED, A.; GUL, A.; HASANUZZAMAN, M. Osmoregulation and its actions during the drought stress in plants. Physiologia Plantarum, v. 172, n. 2, p. 1321-1335, 2021.
PURDY, S. J.; FUENTES, D.; RAMAMOORTHY, P.; NUNN, C.; KAISER, B. N.; MERCHANT, A. The metabolic profile of young, watered chickpea plants can be used as a biomarker to predict seed number under terminal drought. Plants, v. 12, n. 11, e2172, 2023.
PURDY, S. J.; MADDISON, A. L.; CUNNIFF, J.; DONNISON, I.; CLIFTON-BROWN, J. Non-structural carbohydrate profiles and ratios between soluble sugars and starch serve as indicators of productivity for a bioenergy grass. AoB Plants, v. 7, eplv032, 2015.
QUEIROGA, V. P.; GIRÃO, Ê. G.; ALBUQUERQUE, E. M. B. Grão de bico (Cicer arietinum L.): tecnologias de plantio e utilização. Campina Grande: AREPB, 2021.
R DEVELOPMENT CORE TEAM. R: a language and environment for statistical computing. Vienna: R Foundation for Statistical Computing, 2021.
RAMAMOORTHY, P.; LAKSHMANAN, K.; UPADHYAYA, H. D.; VADEZ, V.; VARSHNEY, R. K. Root traits confer grain yield advantages under terminal drought in chickpea (Cicer arietinum L.). Field Crops Research, v. 201, n. 1, p. 146-161, 2017.
ROSA, M.; PRADO, C.; PODAZZA, G.; INTERDONATO, R.; GONZÁLEZ, J. A.; HILAL, M.; PRADO, F. E. Soluble sugars-metabolism, sensing and abiotic stress: a complex network in the life of plants. Plant Signaling and Behavior, v. 4, n. 5, p. 388-393, 2009.
SAKSENA, H. B.; SHARMA, M.; SINGH, D.; LAXMI, A. The versatile role of glucose signaling in regulating growth, development and stress responses in plants. Journal of Plant Biochemistry and Biotechnology, v. 29, n. 4, p. 687-699, 2020.
SILVA, K. F.; MORAES, D. H. M.; MESQUITA, M.; OLIVEIRA, H. F. E.; NASCIMENTO, W. M.; BATTISTI, R.; FLORES, R. A. Water requirement and crop coefficient of three chickpea cultivars for the edaphoclimatic conditions of the Brazilian Savannah biome. Irrigation Science, v, 39, n. 5, p. 607-616, 2021.
SULPICE, R.; PYL, E.-T.; ISHIHARA, H.; TRENKAMP, S.; STEINFATH, M.; WITUCKA-WALL, H.; GIBON, Y.; USADEL, B.; POREE, F.; PIQUES, M. C.; VON KORFF, M.; STEINHAUSER, M. C.; KEURENTJES, J. J. B.; GUENTHER, M.; HOEHNE, M.; SELBIG, J.; FERNIE, A. R.; ALTMANN, T.; STITT, M. Starch as a major integrator in the regulation of plant growth. Proceedings of the National Academy of Sciences, v. 106, n. 25, p. 10348-10353, 2009.
TALEBI, R.; ENSAFI, M. H.; BAGHEBANI, N.; KARAMI, E.; MOHAMMADI, K. Physiological responses of chickpea (Cicer arietinum) genotypes to drought stress. Environmental and Experimental Biology, v. 11, n. 1, p. 9-15, 2013.
THALMANN, M.; SANTELIA, D. Starch as a determinant of plant fitness under abiotic stress. New Phytologist, v. 214, n. 3, p. 943-951, 2017.
TIWARI, P. N.; TIWARI, S.; SAPRE, S.; BABBAR, A.; TRIPATHI, N.; TIWARI, S.; TRIPATHI, M. K. Screening and selection of drought-tolerant high-yielding chickpea genotypes based on physio-biochemical selection indices and yield trials. Life, v. 13, n. 6, e1405, 2023.
VALLURU, R.; VAN DEN ENDE, W. myo-inositol and beyond: emerging networks under stress. Plant Science, v. 181, n. 4, p. 387-400, 2011.
WICKHAM, H. ggplot2: elegant graphics for data analysis. 2. ed. New York: Springer, 2009.
ZAHOOR, R.; DONG, H.; ABID, M.; ZHAO, W.; WANG, Y; ZHOU, Z. Potassium fertilizer improves drought stress alleviation potential in cotton by enhancing photosynthesis and carbohydrate metabolism. Environmental and Experimental Botany, v. 137, n. 1, p. 73-83, 2017.
ZEPEDA, A. C.; HEUVELINK, E.; MARCELIS, L. F. M. Non-structural carbohydrate dynamics and growth in tomato plants grown at fluctuating light and temperature. Frontiers in Plant Science, v. 13, e968881, 2022.
ZHANG, X.; YANG, H.; DU, T. Osmotic adjustment of tomato under mild soil salinity can enhance drought resistance. Environmental and Experimental Botany, v. 202, e105004, 2022.
