Articulos
Flat sheet membrane elaboration by TIPS method using palm oil as solvent and its application in membrane distillation
Elaboración de membranas planas de polietileno por el método TIPS usando aceite de palma como solvente y su aplicación en destilación con membranas
Flat sheet membrane elaboration by TIPS method using palm oil as solvent and its application in membrane distillation
Ingeniería y competitividad, vol. 19, no. 1, pp. 60-65, 2017
Facultad de Ingeniería, Universidad del Valle
Received: 04 November 2015
Accepted: 04 January 2016
Abstract: Palm oil (PO) was used as new solvent for the elaboration of low density polyethylene (LDPE) flat sheet membranes via TIPS method. Phase diagram LDPE/PO was determined, and the effect of parameters such as polymer content (20, 30% w/w), temperature of coagulation bath (0, 25°C) and polymer size (#12, #6 mesh) on thickness, pore size, water vapor permeability and water intrusion pressure of the LDPE membranes was studied. The only parameter that significantly affects the membrane characteristics was polymer content (exempting thickness membrane). By an increase of polymer content, pore size and water vapor permeability decrease (2.7 and 1.2 times, respectively), while water intrusion pressure increases (>1.2 times). Membranes with a thickness of less than 400 µm and pore sizes between 2.3 and 0.8 µm were obtained. Membranes with 0.8 µm pore size were selected to concentrate ethanol-water and glucose solutions by vacuum membrane distillation. Membranes permeated 0.0667 and 0.0508 mol.m-2.s-1, respectively in 2 h of experimentation. Moreover, a 2.1 concentration factor was found using ethanol-water solutions.
Keywords: Membranes, palm oil, phase diagram, polyethylene, TIPS.
Resumen: Se estudió el uso de aceite de palma (AP) como solvente novedoso para la elaboración de membranas planas de polietileno de baja densidad (LDPE) utilizando el método TIPS. Se construyó el diagrama de fases de la mezcla LDPE/AP y se estudió el efecto del contenido de polímero (20 y 30% p/p), la temperatura de baño de coagulación (0 y 25°C) y el tamaño de partícula del polímero (malla #12 y #6) sobre el espesor, el tamaño de poro, la permeabilidad de vapor de agua y la presión de intrusión de las membranas. El contenido de polímero fue la única variable que influyó significativamente en las características evaluadas (exceptuando el espesor). Con el aumento del contenido de polímero, el tamaño de poro y la permeabilidad de vapor de agua disminuyen (2.7 y 1.2 veces, respectivamente), mientras que la presión de intrusión aumenta (>1.2 veces). Se obtuvieron membranas de espesor inferior a 400µm y tamaños de poro entre 2.3 y 0.8 µm. Las membranas de 0.8 µm fueron seleccionadas para concentrar soluciones acuosas de etanol y de glucosa utilizando destilación con membranas en vacío. Se encontraron permeabilidades de 0.0667 y 0.0508 mol.m-2.s-1, respectivamente. Adicionalmente, se encontró un factor de concentración de 2.1 en soluciones etanol-agua.
Palabras clave: Aceite de palma, diagrama de fases, membranas, polietileno, TIPS.
1. Introduction
Membrane distillation (MD) is an interesting technique due to its high potential in the recovery of substances related to applications in water purification and food industry, among others (Mohammadi & Akbarabadi, 2005; Song et al., 2012; Alkhudhiri et al., 2012). In last years, industry and research groups have shown an increased interest about this technique, concerning new applications, transfer phenomena and membrane elaboration for MD (Drioli et al., 2015). MD uses, as driving force, the difference in the vapor pressure between both sides of the membrane, so that volatile compounds pass through the porous membrane, usually hydrophobic (Al-Asheh et al., 2006; Mohammadi & Akbarabadi, 2005; Benavides-Prada, 2013). Membrane hydrophobicity prevents the passage of the solution (aqueous) through its pores, resulting in the formation of a liquid-vapor interface on the membrane surface (Lawson & Lloyd, 1997; Mohammadi & Akbarabadi, 2005; Dao et al., 2013). Additionally, it is important to note that the operating pressure should not exceed the value of the membrane intrusion pressure, which causes fluid passage through the membrane (Saffarini et al., 2012).
Some of the most commonly used materials for the production of hydrophobic porous membranes are polytetrafluoroethylene (PTFE), polypropylene (PP), polyvinylidene fluoride (PVDF) and polyethylene (PE) (Dao et al., 2013). Among these materials, the PE stands out as a polymer easy to produce, stable chemically (resistant to strong acids, strong bases, soft oxidants and reducing agents) and inexpensive (Elyashevich et al., 2005; Zuo et al., 2016). An important aspect to consider in the development of membranes is the selection of a method to control the structure of the resulting membrane. Depending on this, specific pore sizes and porosities will be obtained, factors affecting the performance of the membrane (Gozálvez, 1998; Palacio, 1998; Park & Kim, 2014).
Among the methods used for the preparation of porous polymeric membranes, TIPS (Thermally Induced Phase Separation) method is the most studied because it allows a better control of the membrane structure compared with other methods and also due to its high reproducibility (Mulder, 1996; Matsuyama et al., 1998; Shang et al., 2003; Lin et al., 2009; Park & Kim, 2014). TIPS method includes the dissolution of a polymer in a solvent of low molecular weight and low volatility at elevated temperatures to form a homogeneous mixture. Then molded into the desired shape and cooled to induce separation of the mixture into two phases: a solid phase (rich in polymer) constituting the membrane, and a liquid phase (poor in polymer) (Porter, 1990; Mulder, 1996; Gozálvez, 1998; Palacio, 1998; Pinnau et al., 2000; Elyashevich et al., 2005).
The porous structure results after removing the solvent from the membrane by using an extracting agent, insoluble with the polymer (Mulder, 1996; Caplan et al., 1997; Gozálvez, 1998; Liang et al., 2013). In the elaboration of PE membranes by TIPS method, they have been identified the following variables that affect the final characteristics of membranes: polymer molecular weight (Matsuyama et al., 2003), polymer/solvent composition (Zhang et al., 2008) and temperature of the coagulation bath (Liu et al., 2011). Water has been the most common fluid used for the coagulation bath (Matsuyama et al., 2003; Zhang et al., 2008; Zhang et al., 2010).
Besides that, it is necessary to obtain a phase diagram (Temperature vs. Polymer content) of the polymer/solvent. This diagram give information about the behavior that mixture would have during phase separation: important to explain the morphology of the resultant membrane (Caplan et al., 1997; Yave et al., 2005; Liang et al., 2013).
Considering this, the solvent plays an important role since it is involved in the final morphology (pore size and structure) and properties of the membrane. Previous studies have reported development of PE membranes by the TIPS method using mainly the following solvents: liquid paraffin (LP) (Matsuyama et al., 2003; Liu et al., 2011), di-isodecyl phthalate (DIDP) (Matsuyama et al., 2003), di-octyl phthalate (DOP) (Jeon & Kim, 2007; Zhang et al., 2008) and di-phenyl ether (DPE) (Zhang et al., 2010). However, their use is limited because they are toxic substances and environmentally unfriendly (Cui et al., 2013; Park & Kim, 2014). For that reason, friendly solvents have been introduced, such as mineral oil (Matsuyama et al., 2002a), tri-ethyl-ol-propane tris(2-ethylhexanoate) (TEPTEH) (Park & Kim, 2014) and vegetable oils like soybean oil (Kim et al., 1995; Jeon & Kim, 2007; Tang et al. 2010).
Given the importance of finding sustainable solvents to the membrane elaboration processes, this paper proposed palm oil (PO) as a new solvent, due to its no toxicity and good thermal stability. In addition, for the case of Colombia, palm oil has the advantage of availability and lower costs compared with other vegetable oils. Tang et al. (2010) concluded in their study that for natural solvents, the most important parameter affecting phase diagram, and so membrane process elaboration, is the molecular weight. About this aspect, palm oil has a fatty acids average molecular weight of 285.9 g/mol (CORPODIB, 2006), near of soybean oil molecular weight, 290 g/mol (Tang et al., 2010). Moreover, palm oil has a smoke point (275 °C) higher to that of soybean oil (240 °C), indicating a further thermal stability and making possible the use of palm oil in process working at high temperature, like in this application (Ortiz-López, 2014). It doesn´t exist studies dealing with palm oil for the elaboration of hollow fiber porous membranes, and it is important to make experimental that allow to confirm this hypothesis. In addition, the phase diagram of LDPE/PO mixture was built. The effect of polymer content in the mixture and temperature of coagulation bath in the pore size, water vapor permeability and intrusion pressure of LDPE flat membranes was studied. Finally, the performance of these membranes in vacuum membrane distillation (VMD) was evaluated.
2. Methodology
2.1 Materials
It was used low density polyethylene (LDPE) supplied by CARLIXPLAST Ltda., palm oil supplied by Oleoflores Inc. (acidity 0.1% -oleic acid equivalent-; peroxide content 0.82 mEq O2/kg; refractive index 1.465; density 0.911 g/mL; iodine index 57.9 g Iodine/100g; saponification index 196.24 mgKOH/g) and n-hexane R.A. by Merck Inc. Additionally, glucose syrups (55°Brix) were supplied by PROMITEC SANTANDER S.A.S and ethanol 96% by Suquin Ltda.
2.2 Preparation of LDPE / palm oil samples
Samples of LDPE/PO (20 g) with different compositions were prepared at 180°C and mechanically stirred until complete homogenization (Liu et al., 2011; Matsuyama et al., 2003). Then, mixtures were left at room temperature until solidification. These samples were employed to construct the phase diagram.
2.3 Phase diagram determination
The crystallization temperature was determined by DSC (Differential Scanning Calorimetry) analysis, equipment DSC-2010 (TA Instrument). A 10 mg sample of each mixture was subjected to temperatures within the range 25-200°C, at 10°C/min. The crystallization temperature corresponded to the value where the exothermic peak appears during the mixture cooling (Matsuyama et al., 1998; Matsuyama et al., 2002b; Matsuyama et al., 2003; Yin et al., 2007; Lin et al., 2009).
To determine the temperature where phase separation occurs, samples were taken on a slide with black background, that was placed in a muffle furnace at 180°C, reducing gradually the system temperature 1°C/min to detect visually any turbidity in the mixture (Liu et al., 2011; Matsuyama et al., 2003). This test was performed by triplicate for each composition.
2.4 LDPE membrane elaboration.
The assembly used is shown in Figure 1. This system is composed of an aluminum support plate with a sink to collect the excess of polymer-solvent mixture. It also has a space in which a rectangular aluminum mold (200x150x1mm) is assembled and where the mixture is deposited. Additionally, this mold has a channel-shaped machining in order to facilitate heat transfer. A seal plate of known mass (200x150x5mm), allows to close the mold so that the mixture remains covered, exerting a pressure of 260 Pa to obtain membrane with a thickness less than 350 µm.
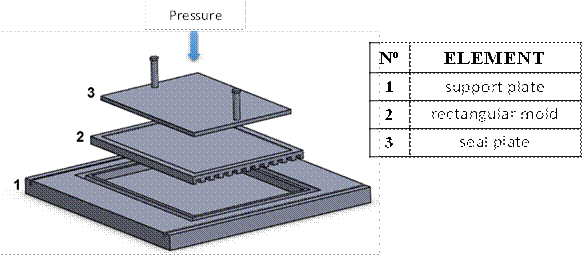
Figure 1
Flat membrane elaboration system.
The membrane elaboration process begins with the preparation of the LDPE/PO mixture at 180°C, stirring at 15 rpm during 12 h to complete homogenization. Next, a quantity of the mixture is deposited in the assembly described above (Figure 1), which should be at 180°C. Then, the mixture is subjected to a thermal shock using a distilled water bath (by immersion during 2 min). The membrane, carefully removed from the assembly, is immersed in a bath of hexane (250 ml) for 12 h to extract the oil contained in it; this procedure is done twice. Finally, the obtained membrane is dried in an oven at 50°C for 4 h. To study the effect of polymer content in the mixture on the selected characteristics, values of 20 and 30% w/w were used, and to study the effect of coagulation bath temperature, values of 0 and 25 °C were used.
2.5 Membrane characterization
2.5.1 Membrane structure, thickness and pore diameter
The structure and morphology of the cross section of the membranes were observed by Scanning Electron Microscope (SEM) QUANTA 650 FEG with 10 kV accelerating voltage. The thickness and the average pore size of each membrane were taken from pores selected randomly from the cross section (30 readings).
2.5.2 Water vapor permeability
Membranes were deposited in the system designed by Gutiérrez-Gutiérrez et al. (2015). This system consists of two stainless steel cells of equal volume (1 591. 2 cm3) connected by a hole with 2.5 cm in diameter (area of 19.63 cm2). Silica was used in the permeate cell to reduce humidity (RH <23%), and distillated water was introduced in the feed cell to maintain it saturated with water vapor. Relative humidity and dry bulb temperature were measured every 15 min for both cells using a digital hygrometer DT321S brand BRIXCO during 180 min. With that information, it was determined the water vapor mass flux of the permeated through the membrane.
2.5.3 Water Intrusion Pressure
The assembly used is shown in Figure 2. Distilled water is fed to membrane module from the feed tank using dry compressed air, which puts pressure on the liquid. The exerted pressure is determined with a manometer and adjusted by using a valve connected to the dry air supply. In this assembly, pressure did not exceed 1.3 bar taking into account that VMD technique does not work with transmembrane pressures higher than 1 bar (full vacuum).
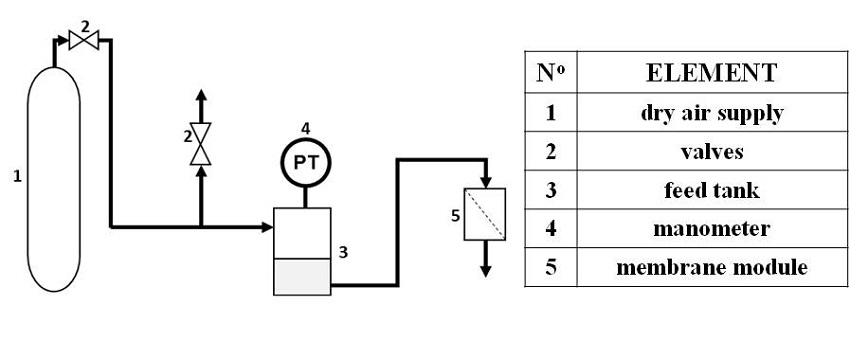
Figure 2
Water intrusion pressure system.
2.5.4 Vacuum membrane distillation (VMD)
VMD tests were carried out with ethanol aqueous solutions (8.2 mol%) and glucose-syrups aqueous solutions (10 °Brix) to evaluate the performance of the LDPE flat membranes. The solution to be treated is fed to membrane module (effective diameter: 3.45 cm), using a peristaltic pump, at 0.6 gpm from a feed tank having magnetic stirring and temperature control. The permeate (water/ethanol or water vapors, as appropriate) is directed to the condenser using a vacuum pump and is accumulated in a trap coupled to it (see Figure 3).
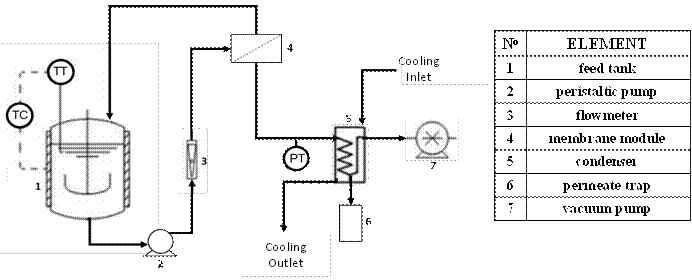
Figure 3
Scheme of the Vacuum Membrane Distillation system used in this study. Cotamo-De la Espriella et al. (2015).
3. Results and discussion
3.1 Phase diagram
The phase diagram obtained for LDPE-palm oil mixtures is shown in Figure 4. In this diagram, the binodal curve (point cloud data) fell with polymer content in the mixture, while the crystallization curve has a slight increase. It is important to note that the binodal curve is located above the crystallization curve in polymer content range of 20 to 80% w/w. This suggests that the interaction between LDPE and palm oil is weak, which leads to L-L phase separation prior to crystallization of the polymer during cooling. This implies the formation and growth of the polymer poor phase as droplets around the polymer-rich phase, which ends when the polymer is solidified by a heat shock. After solvent extraction, structures in cell shape or lace (round interconnected pores) with high porosity will be obtained (Caplan et al., 1997; Pinnau et al., 2000; Liu et al., 2011; Song et al., 2012). Considering that, it is expected that the elaborated membranes present porous structures with cell shape within the range of the studied polymer content; characteristics that make these membranes attractive to use in MD (Song et al., 2012; Liu et al., 2011; Dao et al., 2013).
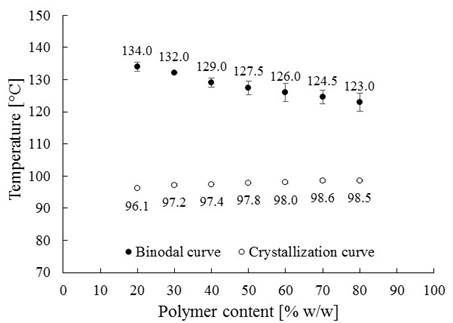
Figure 4
Phase Diagram for LDPE- palm oil mixtures. *Average deviation bars (not visible for crystallization points).
3.2 Membrane morphology
Figure 5 shows the morphology of the cross section of a membrane produced with a LDPE content of 30% w/w and a coagulation bath temperature of 25°C.

Figure 5
SEM images for LDPE membrane cross section. Membrane elaborated with polymer content of 30% w/w and Coagulation bath temperature of 25 °C. (a) 500x (b)10000x.
A global cross section view is shown in Figure 5 (a), while Figure 5 (b) shows a more detailed zoom of this section. In this work, membrane thickness of 280 ± 30 µm were obtained. They were superior to those reported for commercial-ones such as Polypropylene (171,8 µm) and PTFE (253,5 µm). In these images, cell type porous structures are observed, representatives of L-L phase separation prior to crystallization of the polymer, supporting the idea described above.
3.3 Pore diameter
Figure 6 shows the morphology of the cross section of the membranes prepared with polymer contents (20 and 30% w / w) and coagulation bath temperatures (0 and 25 ° C) defined above. The pore sizes obtained are shown in Figure 7 as a function of polymer content and coagulation bath temperature. From these results, it is possible to see a reduction of pore size (2.8 times) with increasing polymer content in the mixture from 20 to 30% w/w; a decrease can also be observed in SEM images of Figure 7. This is because, according to the obtained phase diagram, with the increasing in the polymer content of the mixture (from 20 to 30% w/w LDPE ), the spacing between the binodal curve and crystallization becomes smaller (8.2% reduced). This trend in the binodal line has also been found in other studies that elaborate polyethylene membranes. Jeon & Kim (2007) and Park & Kim (2014) reported decreases in the cloud point when increasing the polymer content in the mixture (from 20 to 60% w / w) of 49 and 142°C, respectively. All previous authors used high density polyethylene and different solvents to form the polymer mixture: Jeon & Kim (2007) worked with soybean oil as a solvent and Park & Kim (2014) used triethylolpropane tris (2-ethylhexanoate) (TEPTEH). This implies greater difficulty in the formation and growth of droplets of polymer-poor phase during phase separation, creating a smaller pore size (Zhang et al., 2008). Additionally, no influence of the coagulation bath temperature in the pore size to the evaluated conditions (0 and 25 ° C) is evidenced. At these temperatures, and considering mixtures were at 180°C, energy transfer rates are highs, traducing in rapid cooling and similar times for the growth of the forming-pores droplets, which leads to similar pore diameters.
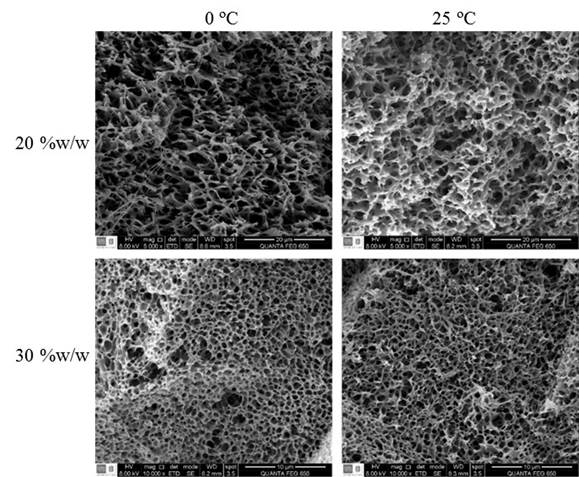
Figure 6
SEM images for LDPE membrane cross section. Membrane elaborated with polymer content of 20% w/w, 5 000x. Membranes with 30% w/w, 10000x.
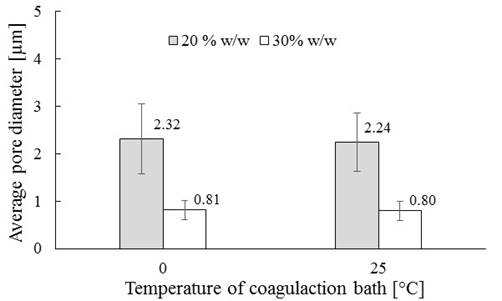
Figure 7
Average pore diameter of the membrane vs. Coagulation bath temperature. *Standard deviation bars.
3.4 Water vapor permeability
The results obtained for the flux of water vapor permeate through the membrane are presented in Figure 8. In this figure, a reduction of water vapor flux (1.3 times) when increasing the polymer content is found. This is due to the smaller pores obtained by increasing the polymer content; a higher content of polymer in the mixture involves a smaller volume occupied by the solvent in the membrane, which generates smaller pores after being removed (and possibly, membranes with lower porosity). These smaller pores increase the flow resistance and, with this, a decreasing in permeability (Park & Kim, 2014). Furthermore, no influence of the coagulation temperature bath in the permeability of membranes was evidenced (similar to water vapor permeate flux). As mentioned above, this is related to the pore diameter of the membrane, which did not change significantly; making the passage of water vapor through it to be unaffected.
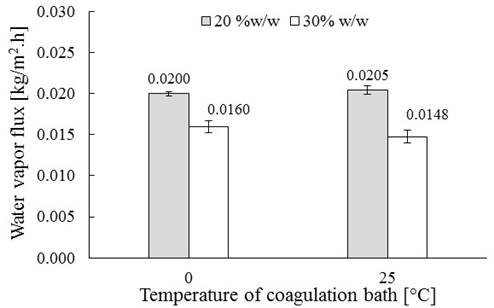
Figure 8
Water vapour flux through the membrane vs. Temperature of coagulation bath. *Average deviation bars.
3.5 Water intrusion pressure
According to the intrusion pressure tests, membranes elaborated with polymer content of 30% w/w did not permeate water at the maximum pressure evaluated (1.3 bar), while those made with 20% w/w polymer permeated at 1.1 bar. This is related to pore size reduction by increasing the polymer content in the mixture. This causes an increased resistance to the passage of water through the membrane (Cui et al., 2013). Therefore, a smaller pore size, a higher intrusion pressure will be required. Intrusion pressures of 3.6, 4.4 and 5.2 bar were found for commercial membranes of polypropylene with 0.45 µm of pore diameter, and PTFE with 0.45 and 0.2 µm of pore diameter, respectively; these values can change for a same pore diameter depending of membrane characteristics like thickness and porosity.
3.6 VMD performance
LDPE membranes prepared with 30% w/w of polymer mixtures were selected to concentrate aqueous solutions of ethanol and glucose syrups by VMD technique. These membranes have an average pore size of 0.8 µm and thickness of 300 µm. The parameters used for both cases are presented in Table 1.

For ethanol separation from aqueous solutions, the elaborated membranes permeated a total molar flux of 0.0667 mol.m-2.s-1 during 2 h of experimentation, with a concentration factor (permeate concentration/feed concentration) of 2.1. Similarly, when working with aqueous solutions of glucose syrups, elaborated membranes presented a molar flux for permeate of 0.0508 mol.m-2.s-1 during 2 h of experimentation. Differences in permeability found with the two studied cases could be explained by differences in composition of solutions used (etanol-water and diluted glucose syrups), and vapors permeated through the membrane (ethanol-water and only water, respectively). The poor permeability evidenced for the membranes (water flux < 0.0667 mol.m-2.s-1), in comparison with other permeabilities reported in literature (0.33 mol.m-2.s-1, PTFE membrane and pore diameter of 0.45 µm) (Cotamo-De la espriella et al., 2015), is related to its characteristics (thickness and porosity), factors that increase the resistance to flow through them (Cotamo-De la espriella et al., 2015). Although these results show the applicability in MD of membranes developed in the present study using palm oil as a solvent, it is necessary to continue working on improving its features in order to increase permeability.
4. Conclusions
Palm oil was used as a new solvent for LDPE membrane elaboration by applying the TIPS method. The phase diagram of LDPE palm-oil mixture was determined for the first time. Its characteristics provide information about the formation of cell type porous structures because of the separation L-L, prior to solidification of the polymer; the type of structure was verified through SEM images. Membrane characterization by SEM, water vapor permeability and intrusion pressure were carried out using the elaborated LDPE membranes, showing the influence of the polymer content in the pore size, which affects the performance of the membrane, such as permeability and water intrusion pressure. The performance of the elaborated membranes to separate ethanol from ethanol aqueous solutions and water from diluted glucose syrups by VMD technique was evaluated. Results show the capability of these membranes to separate these volatile compounds but with low permeability rates. These values are associated with the thickness and porosity of the membranes, characteristics that increase the resistance to flow through them. It is necessary to keep improving the characteristics of the membranes, essentially related to the thickness and porosity.
5. Acknowledgments
The authors thank the Vicerrectoría de Investigación y Extensión of Universidad Industrial de Santander, the company Promitec Santander SAS and the Administrative Department of Science, Technology and Innovation - COLCIENCIAS for their support in the realization of this work, which is part of the project entitled "Increasing in the productivity of concentrated glucose syrups derived from starch hydrolysates of cassava by using unconventional methods" (code. 1102-5022-7720).
6. References
Al-Asheh, S., Banat, F., Qtaishat, M., & Al-Khateeb, M. (2006). Concentration of sucrose solutions via vacuum membrane distillation. Desalination 195 ( 1), 60-68.
Alkhudhiri, A., Darwish, N., & Hilal, N. (2012). Membrane distillation: A comprehensive review. Desalination 287 (1), 2-18.
Benavides-Prada, O., Guevara-Lastre, C., Barón-Núñez, F., Barajas-Ferreira, C., Ortiz-Basurto, R.I., Torrestiana-Sánchez, B., & Muvdi-Nova, C. (2013). Vacuum membrane distillation: modeling and analysis for recovery of ethanol from ethanol/water solutions. CT&F -Ciencia, Tecnología y Futuro 5 (2), 47-60.
Caplan, M.R., Chiang, C.Y., Lloyd, D.R., & Yen, L.Y. (1997). Formation of microporous Teflon ® PFA membranes via thermally induced phase separation. Journal of Membrane Science 130 (1-2), 219-237.
CORPODIB (Corporación para el desarrollo Industrial de la Biotecnología y producción limpia). (2006). Aspectos económicos de la implementación de la tecnología de producción de biodiesel a partir de aceite de palma. Bogotá, Colombia.
Cotamo-De la espriella, R., Barón-Núñez, F., & Muvdi-Nova, C. (2015). Study of the operating conditions influence in vacuum membrane distillation and sweeping gas membrane distillation for the recovering of ethanol from aqueous mixtures. CT&F -Ciencia, Tecnología y Futuro 6 (2), 69-80.
Cui, Z., Hassankiadeh, N.T., Lee, S.Y., Lee, J.M., Woo, K.T., Sanguineti, A., Arcella, V., & Lee, Y.M. (2013). Poly(vinylidene fluoride) membrane preparation with an environmental diluent via thermally induced phase separation. Journal of Membrane Science 444, 223-236.
Dao, T.D., Mericq, J.P., Laborie, S., & Cabassud, C. (2013). A new method for permeability measurement of hydrophobic membranes in Vacuum Membrane Distillation process. Water Research 47 (6), 2096-2104.
Drioli, E., Ali, A., & Macedonio, F. (2015). Membrane distillation: Recent developments and perspectives. Desalination 356 (1), 56-84.
Elyashevich, G.K., Olifirenko, A.S., & Pimenov, A.V. (2005). Micro and nanofiltration membranes on the base of porous polyethylene films. Desalination 184 (1), 273-279.
Gozálvez, J.M. (1998). Fabricación de membranas de acetato de celulosa aptas para ósmosis inversa y nanofiltración mediante el método de inversión de fase. Tesis Doctoral, Departamento de Ingeniería Química y Nuclear, Universidad Politécnica de Valencia, Valencia, España.
Gutiérrez-Gutiérrez, M.I., Morales-Mendivelso, D., Muvdi-Nova, C., & Cháves-Guerrero, A. (2015). Síntesis y caracterización de membranas híbridas a partir de quitosan, polivinil alcohol y sílice para su aplicación en deshidratación de gases. ITECKNE 12 (1), 33-43.
Jeon, M.Y., & Kim, C.K. (2007). Phase behavior of polymer/diluent/diluent mixtures and their application to control microporous membrane structure. J. Membr. Sci. 300 (1-2), 172-181.
Kim, J.J., Hwang, J.R., Kim, U.Y., & Kim, S.S. (1995). Operation parameters of melt spinning of polypropylene hollow fiber membranes. J. Membr. Sci. 108 (1-2), 25-36.
Lawson, K.W., & Lloyd, D.R. (1997). Membrane distillation Review. Journal of Membrane Science 124 (1), 1-25.
Liang, H.Q., Wu, Q.Y., Wan, L.S., Huang, X.J., & Xu, Z.K. (2013). Polar polymer membranes via thermally induced phase separation using a universal crystallizable diluent. Journal of Membrane Science 446, 482-491.
Lin, Y.K., Chen, G., Yang, J., & Wang, X.L. (2009). Formation of isotactic polypropylene membranes with bicontinuous structure and good strength via thermally induced phase separation method. Desalination 236 (1-3), 8-15.
Liu, S., Zhou, C., & Yu, W. (2011). Phase separation and structure control in ultra-high molecular weight polyethylene microporous membrane. Journal of Membrane Science 379 (1-2), 268-278.
Matsuyama, H., Berghmans, S., Batarseh, M., & Lloyd, D. (1998). Effects of thermal history on anisotropic and asymmetric membranes formed by thermally induced phase separation. Journal of Membrane Science 142 (1), 27-42.
Matsuyama, H., Kim, M.M., & Lloyd, D. (2002a). Effect of extraction and drying on the structure of microporous polyethylene membranes prepared via thermally induced phase separation. Journal of Membrane Science 204 (1-2), 413-419.
Matsuyama, H., Maki, T., Teramoto, M., & Asano, K. (2002b). Effect of polypropylene molecular weight on porous membrane formation by thermally induced phase separation. Journal of Membrane Science 204 (1-2), 323-328.
Matsuyama, H., Okafuji, H., Maki, T., Teramoto, M., & Kubota, N. (2003). Preparation of polyethylene hollow fiber membrane via thermally induced phase separation. Journal of Membrane Science 223 (1-2), 119-126.
Mohammadi, T., & Akbarabadi, M. (2005). Separation of ethylene glycol solution by vacuum membrane distillation (VMD). Desalination 181 (1-3), 35-41.
Mulder, M. (1996). Basic Principles of Membrane Technology. Segunda Edición. Holanda: Kluwer Academic Publishers.
Ortiz-López, S.A. (2014) Investigación de los derivados de la palma africana, sus beneficios y aplicación en la gastronomía. Tesis de grado previa a la obtención del título de Administrador Gastronómico, Carrera de Gastronomía, Facultad de Turismo, Hotelería y Gastronomía, Universidad Tecnológica Equinoccial, Quito, Ecuador.
Palacio, L. (1998). Caracterización estructural y superficial de membranas microporosas. Tesis Doctoral, Departamento de Termodinámica y Física Aplicada, Universidad de Valladolid, Valladolid, España.
Park, M.J., & Kim, C.K. (2014). Fabrication of polyethylene microporous membranes using triethylolpropane tris(2-ethylhexanoate) as a novel diluent by a thermally induced phase separation process. Journal of Membrane Science 449, 127-135.
Pinnau, I., & Freeman, B.D. (2000). Membrane Formation and Modification. Primera Edición. Washington D.C.: American Chemical Society.
Porter, M.C. (1990). Handbook of Industrial Membrane Technology. Primera Edición. Park Ridge, NJ: Noyes Publications.
Saffarini, R., Arafat, H., & Thomas, R. (2012). Influence of pore structure on membrane wettability in membrane distillation. In Proceedings of the 6th Jordan International Chemical Engineering Conference, Amman, Jordan, p. 303-307.
Shang, M., Matsuyama, H., Teramoto, M., Lloyd, D.R., & Kubota, N. (2003). Preparation and membrane performance of poly(ethylene-co-vinyl alcohol) hollow fiber membrane via thermally induced phase separation. Polymer 44 (24), 7441-7447.
Song, Z., Xing, M., Zhang, J., Li, B., & Wang, S. (2012). Determination of phase diagram of a ternary PVDF/c-BL/DOP system in TIPS process and its application in preparing hollow fiber membranes for membrane distillation. Separation and Purification Technology 90, 221-230.
Tang, N., Jia, Q., Zhang, H., Li, J., & Cao, S. (2010). Preparation and morphological characterization of narrow pore size distributed polypropylene hydrophobic membranes for vacuum membrane distillation via thermally induced phase separation. Desalination. 256 (1-3), 27-36.
Yave, W., Quijada, R., Serafini, D., & Lloyd, D.R. (2005). Effect of the polypropylene type on polymer-diluent phase diagrams and membrane structure in membranes formed via the TIPS process: Part I. Metallocene and Ziegler-Natta polypropylenes. Journal of Membrane Science 263 (1-2), 146-153.
Yin, J., Lv, R., Zhou, J., Du, Q.G., & Zhong, W. (2007). Preparation of EVOH microporous membranes via thermally induced phase separation using binary solvents. Chinese Journal of Polymer Science 25 (4), 379-386.
Zhang, H., Zhou, J., Zhang, X., Wang, H., Zhong, W., & Du, Q. (2008). High density polyethylene-grafted-maleic anhydride low-k porous films prepared via thermally induced phase separation. European Polymer Journal 44 (4), 1095-1101.
Zhang, C., Bai, Y., Sun, Y., Gu, J., & Xu, Y. (2010). Preparation of hydrophilic HDPE porous membranes via thermally induced phase separation by blending of amphiphilic PE-b-PEG copolymer. Journal of Membrane Science 365 (1-2), 216-224.
Zuo, J., Bonyadi, S., & Chung, T.S. (2016). Exploring the potential of commercial polyethylene membranes for desalination by membrane distillation. Journal of Membrane Science 497 (1), 239- 247.