Original research
Serum cortisol concentration in the Colombian creole horse1
Concentración del cortisol sérico en el caballo criollo colombiano
Concentração de cortisol sérico no cavalo crioulo colombiano
Serum cortisol concentration in the Colombian creole horse1
Revista Colombiana de Ciencias Pecuarias, vol. 30, no. 3, 2017
Facultad de Ciencias Agrarias, Universidad de Antioquia
Received: 22 July 2016
Accepted: 13 December 2016
Abstract
Background: Cortisol is indicative of adaptation to physiological factors, disease and environmental stress in equines. Cortisol recently gained attention for its prognostic value in critical disease and as an indicator of fitness and athletic performance.
Objective: To determine baseline serum cortisol (SC) morning concentrations in the Colombian creole horse (CCH).
Methods: Blood serum was taken from 62 CCH of both genders with and average age of 8.8 years (± 5.3), which were clinically healthy and had different performance levels. Results: The overall SC average determined by sandwich ELISA was 13.3 ± 7.4 mg/dL. No significant differences were attributed to gender, age, or performance.
Conclusion: This concentration can be used as a reference value for CCH maintained in low physical activity.
Keywords: adrenocorticotrophic hormone+ equine+ hypothalamic-pituitary-adrenal axis+ stress.
Resumen
Antecedentes: El cortisol es un indicador de adaptación a factores de estrés fisiológico, patológico y ambiental en equinos. Recientemente, esta hormona ha ganado interés por su valor en la prognosis de enfermedades críticas y como indicador del nivel de acondicionamiento y rendimiento atlético. Objetivo: Determinar la concentración del cortisol sérico (CS) matinal para el caballo criollo colombiano (CCC).
Métodos: Se tomó suero sanguíneo de 62 CCC de ambos sexos, edad promedio 8,8 años (± 5,3), clínicamente saludables, divididos en grupos según edad, sexo y tipo de actividad física.
Resultados: La media del CS general determinada por ELISA tipo sándwich fue 13,3 ± 7,4 μg/dL. No hubo diferencias significativas atribuibles a género, edad o desempeño.
Conclusión: Esta concentración puede ser utilizada como valor de referencia en CCC que desarrollen actividades de baja exigencia física.
Palabras clave: enESeje hipotalámico-pituitário-adrenal, equino, estrés, hormona adrenocorticotrófica.
Resumo
Antecedentes: O cortisol nos equinos considera-se um hormônio indicador da adaptação a fatores de estresse fisiológico, patológico e ambiental. Recentemente, tem incrementado o interesse por seu valor prognóstico em doenças críticas e como indicador de acondicionamento e rendimento atlético.
Objetivo: Determinar a concentração do cortisol sérico (SC) matinal no cavalo crioulo colombiano (CCC).
Métodos: Soro sanguíneo foi coletado de 62 CCC, de ambos sexos, idade média de 8,8 anos (± 5,3), clinicamente sadios, divididos em grupos segundo idade, sexo e tipo de atividade física.
Resultados: A média geral da SC determinada pela ELISA tipo sanduiche foi 13,3 ± 7,4 μg/dL. Não houve diferenças significativas atribuíveis ao sexo, idade ou desempenho.
Conclusão: A concentração determinada pode ser utilizada como valor de referência no CCC que desenvolvem atividades de baixa exigência física.
Palavras chave: enPTeixo hipotalâmico-pituitário-adrenal, equino, estresse, hormônio adrenocorticotropina.
Introduction
Cortisol concentration in equines has been used to determine stress generated by illness, pain, and lack of welfare. Recently, cortisol has gained interest due to its prognostic value in the critical phases of gastrointestinal disease (Mair et al., 2014), respiratory disease (Shaba et al., 2014), and clinical cases of sepsis in equine (Hart et al., 2009; Armengou et al., 2013), and as an indicator of fitness and athletic performance in sport horses (Kedzierski et al., 2014). Therefore, this substance is considered the hormone of adaptation to physiological, pathological, and environmental stress factors.
The hypothalamic-pituitary-adrenal axis (HPA) activation generates signals that are interpreted and integrated by the hypothalamus, which subsequently produces and releases corticotropin-releasing hormone (CRH) into the hypothalamic-hypophyseal portal vessels. The CRH acts upon type 1 CRH receptors on the surface of corticotropic cells of the anterior pituitary to systemically produce and release adrenocorticotropic hormone (ACTH). The ACTH interacts with melanocortin-2 receptors on the surface of the adrenal gland cortex cells to synthesize and secrete cortisol. Cortisol secretion derived from physiological stress ensures an adequate level in the plasma and controls its own production by negative feedback on HPA components (Hart and Barton, 2011).
Normally, cortisol secretion follows a circadian rhythm pattern, with elevated peaks of production in the equine during the morning between 6:00- 10:00 a.m.; cortisol production falls to its lowest level between 6:00-9:00 p.m. (McCue, 2002; Nagel et al., 2012), which implies variable plasma levels during the day. The circadian cycle can be affected by factors such as exercise, mating, training, stressors (Pell and McGreevy, 1999), sleep patterns, and individual activities. Cortisol response to these factors is immediate and proportional to the level of stress and rapidly surpasses the normal plasma concentration (Alexander and Irvine, 1998).
The cortisol concentration presents fluctuations over short periods of time and depends on the adaptation of the horse to its environment (Irvine and Alexander, 1994). Additionally, there is evidence in some countries of a seasonal dynamic and variations between annual periods in the same geographic region (Schreiber et al., 2008; Borer-Weir et al., 2013). Therefore, environmental and climatological factors, and the presence of insects cause transient alterations in the diurnal cortisol-releasing pattern (Bohák et al., 2013).
The aim of this study was to determine the concentration of serum cortisol (SC) for the Colombian creole horse (CCH) based on the specific characteristics of temperament, horse raising, environmental conditions, and type of activity performed by the animals.
Materials and methods
This study was approved by the Ethics Committee for Animal Experimentation of Universidad de Antioquia, Colombia (act 1062016).
Animals
A total of 62 CCH of both genders were used. The average age of horses, selected by convenience, was 8.8 ± 5.3 years, and were clinically healthy, based on history and general clinical examination. The animals were kept in stables with bedding of wood shavings, and feed forage, pellets, and water ad libitum.
The animals were divided into age groups (A = between 1 and 5 years old, B = between 6 and 10 years old, and C = greater than 11 years old), gender groups (non-pregnant females and males), and according to the type of physical activity or work performed (A1 = education at a riding school, B1 = exhibition and horse trading, and C1 = recreation in a theme park).
Location and environmental conditions
Samples were taken between March 30 and April 8, 2015. Individuals were located in the city and metropolitan area of Medellin (Antioquia, Colombia). The climatic conditions for these dates were a mean temperature of 23.8 °C (18 to 27.95 °C) and relative humidity was 58%.
Sample collection
Blood samples were taken by jugular venipuncture with pre-cleaning and antisepsis of the site. The samples were collected in vacuum tubes without additives to achieve clot formation. The samples were centrifuged at 1,500 rpm for 10 minutes to facilitate separation and serum removal. The serum was transferred to a cryovial and frozen at -20 °C prior to the analysis. Aliquots of each serum sample were prepared to avoid disruption by repeated defrosting. All samples were collected in the morning between 8:00 a.m. and 11:00 a.m. with minimal handling and containment of the horse.
Determination of the cortisol concentration
The validation of a commercial sandwich ELISA kit for the measurement of cortisol (AccuBind®, Monobind Inc., CA, USA) was performed using six kit calibrators and three commercial cortisol controls (QSure® Multi-Ligand Control Tri-Level, Monobind Inc., CA, USA). The ELISA plate was read at a wavelength between 450-630 nm in a conventional reader (Stat Fax 303® Plus Microstrip Reader, Awareness Technology Inc., FL, USA). The SC concentration values were given in μg/dL. According to information from the supplier, this kit can detect cortisol concentrations from 0.366 μg/dL to 95 μg/dL. This range was validated with known concentrations.
Statistical analysis
The SC concentration data in all groups were subjected to the Kolmogorov-Smirnov test for normality using a statistical package (RStudio®, RStudio Inc., MA, USA). Because it did not obey a normal distribution, the data were normalized using the «standardize» function of the Microsoft Excel 2013® software (Microsoft office 2013®, Microsoft Corporation, WA, USA). Subsequently, we applied Tukey’s test using the SPSS® statistical software V 3.0 (IBM®, NY, USA) to compare the means between age groups and types of physical activity or work groups. The T-test was used to compare the means between genders. The confidence level used for both tests was 95%.
Finally, an analysis of the position-determining percentiles (10, 25, 50, 80, and 97) was performed for the cortisol concentration values using the SPSS® statistical software V 3.0.
Results
The overall mean and the minimum and maximum values of the morning SC concentrations determined by sandwich ELISA in 62 CCH are shown in Table 1. The cortisol concentration determined in this equine population was 13.3 ± 7.4 μg/dL.

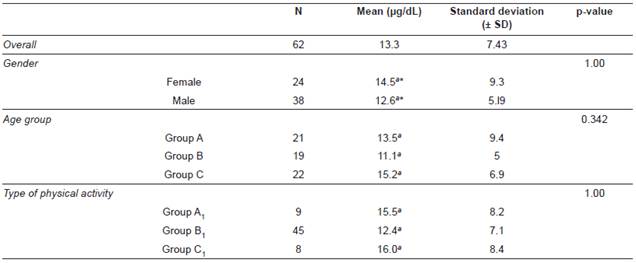

The mean cortisol concentration and comparisons between groups are described in Table 2. Mean comparison between age groups (A, B, and C) and type of activity or work (A1, B1, and C1) showed no significant differences according to Tukey’s test. Similarly, the gender comparison was not different according to the T-test.
Measures for the total number of SC concentration results are described in Table 3. We expect that SC measurements performed in a group of CCH will be equal to or less than 17.26 mg/dL in 80% of the samples.
Discussion
This study reports SC concentrations of young and adult female and male CCH. These values are of interest because no comparative studies exist for other breeds or for the conditions established in this study. Similarly, it is important to know blood cortisol concentration because it contributes to the physiological characterization of CCH. Furthermore, SC helps to define reference values to develop evidence-based methods for the diagnosis and approaches to diseases and situations that compromise the HPA axis. However, it is important to note that these results were derived from a population selected for convenience, and therefore it is necessary to consider the statistical power of the data since less than 40 horses were evaluated per group.
The average cortisol concentration in this study revealed no major differences with the levels obtained for Thoroughbreds in other countries (13.16 and 12.17 μg/dL; Fazio et al., 2008; Kedzierski et al., 2014). However, the horses sampled in previous studies were physically strained, in contrast to the animals in the present study where samples were taken prior to start the daily routine. Therefore, our value could be relatively inflated due to external and inherent factors, considering that the horses were clinically healthy. Similarly, the mean cortisol value of this study was higher than the reference values in the North American literature (maximum value: 9.0 μg/dL; Douglas 1999; Kaneko et al., 2008; Haffner et al., 2010), although those studies did not describe physiological and environmental conditions, breed characteristics, age, and gender of the animals. This difference reaffirms that SC variations is due to HPA susceptibility to exogenous and endogenous factors compromising the circadian cycle dynamics characteristic of the species. Apparently, external factors have great influence on this variation because the literature describes different reference values for the same breed in different geographical locations (Schreiber et al., 2008).
The SC concentration (13.3 mg/dL) determined in this study under resting conditions and sampled with minimal handling intended that these values were minimally affected by physical stress. The increase observed could be due to other environmental and individual factors related to the temperament of CCH. However, studies in other breeds conducted with samples of the same nature and collected with similar methods reported values lower than the values obtained here. Therefore, cortisolemia level in equines should be interpreted with caution by considering the acute or chronic course of stress, degree of adaptation of the horse to the handling situation, environmental conditions, sampling time, and health conditions.
Nonetheless, the mean cortisol concentration determined in this CCH population was lower than that in a group of healthy Spanish horses (37.6 μg/dL; Martos et al., 2003). However, it was not possible to compare the values from each study because Martos et al. (2003) did not report the management, environmental conditions, type of activity, and breed characteristics. Therefore, the apparent hypercortisolemia in these two populations of clinically normal horses in the absence of endocrine and/or metabolic alterations indicates a multifactorial variability in the synthesis and secretion of the hormone. This should be considered for disease and stress states as well as welfare conditions.
Although most studies on cortisol did not report the minimum and maximum values, it was possible to demonstrate a broad range for the CCH (4.2 and 48.1 μg/dL). However, according with the percentile analysis only 20% of the population was distant from the mean concentration value and only a minority (3%) was higher than 34.9 μg/dL. Consequently, the location analysis of cortisol concentration reported that 80% of the population presented a value equal to or less than 17.26 μg/dL, which is close to the overall mean. Therefore, this value can be useful and should be taken into account during the interpretation of CCH results.
Contrary to acute stress, a horse under chronic stress can inhibit or stimulate cortisol production, which affects the normal response to later acute stress episodes (Alexander et al., 1988). A significant increase in SC is observed in cases of acute inflammation and colic episodes in equines (Martos et al., 2003; Mair et al., 2014). Conversely, a decrease in the concentration of this analyte is evident in cases of chronic inflammation and social stress. Therefore, the broad range for cortisol values in the CCH should be interpreted with caution and with the aforementioned considerations.
The low values (5.82 μg/dL) reported by Haffner et al. (2010) in Mongolian horses may be due to changes in the dynamics of cortisol production compared with the values observed in this study. It should be taken into account that Mongolian horses are subjected to extreme physical demands under adverse climatic and feeding conditions (Haffner et al., 2004). This possibly explains an adaptation to the pressure, chronicity, and nature of the stimuli of the HPA (Broom, 2006) for coping with stress factors (Freymond et al., 2015). However, the frequencies and prevalence of derived abnormal and stereotypical behaviors have not been described in these animals.
The SC concentration determined by radioimmunoassay and reported by Mair et al. (2014) in horses with colic surgery (15.68 ± 8.27 μg/dL) was close to the mean and range obtained by sandwich ELISA in the CCH without manifestation of acute abdomen or history of recent disease. This pattern confirms the need to use reference values adjusted to populations with different conditions of management, environment and type of activity. Additionally, particular patterns are required by the work performed (i.e. a certain degree of dorsoventral hyperflexion of the neck is induced for greater showiness and elegance in CCH). This behavior has been associated with significant increase of stress-related cortisol in other breeds (McGreevy et al., 2010; Christensen et al., 2014; Smiet et al., 2014). Although this phenomenon has not been evaluated in the Colombian horse, it might occur in a similar way.
In addition to its variable nature, cortisol is also susceptible to differences in results according to the processing technique and sample type. One study estimated a difference of 1.57 μg/dL between ELISA and chemiluminescence, suggesting greater accuracy for the latter. However, this comparison was made in different populations without considering all the variables (Armengou et al., 2013). The present study did not aim to compare techniques; however, validation with commercial controls and the similarity of SC in CCH to other horses from the same region (unpublished data) support the reliability of the technique used.
Measurements of cortisol in equines are performed in plasma, serum, saliva, tears, feces, and hair (Kirschbaum et al., 2000; Comin et al., 2012; Bohák et al., 2013; Monk et al., 2014; Nuñez et al., 2014). Recently, cortisol determination in saliva has been used to monitor the stress effect because it minimizes the effect of sample collection on cortisol concentration. However, studies report a high correlation and no significant difference in total plasma cortisol in its respective phases (Hart et al., 2011; Kedzierski et al., 2014). The type of sample used in this study is supported by the absence of a reference value in this substrate because blood cortisol is found in both its free form and bound to proteins in different proportions, as was previously reported. Determination of total cortisol as reference values in CCH encourages future work aimed at determining this hormone in other types of samples.
This study found no significant difference between groups, although female values (C and C1) were higher. These findings partially agree with results by Douglas (1999) where cortisol was not affected by gender, age, breed, or gestation. However, the influence of pregnancy was not tested in the CCH females. Contrary to the report by Douglas (1999), recent studies showed variation according to gestational age and post-partum period because HPA maturation in the foal/colt occurs days before and weeks after birth (Nagel et al., 2012).
The present study showed that cortisol concentration at rest was less variable in horses older than one year, which contrast to neonates in their first two hours of life (Nagel et al., 2015). However, there are reports indicating a reduction in sensitivity to the negative feedback of cortisol with increasing age (Gust et al., 2000), which may explain the upward trend in group C. A report with healthy neonates and 6-hour-old CCH (3.42 ± 1.47 μg/dL; Moreno, 2009) showed a lower value than that observed in this study, which coincides with the results by Hart et al. (2011) comparing basal cortisol in neonates and healthy adults considering that large variations occur immediately after birth.
Cortisol concentration in equines is associated with the level of physical or athletic demands and the intensity of stressful situations before, during, and after developing an activity (Alexander et al., 1988; Schmidt et al, 2010; Ferlazzo et al., 2012; Kedzierski et al., 2014). The lack of difference between CCH groups regarding type of activity occurred because animals were sampled at rest before performing specific activities. The effect of stabling and social stress may have influenced cortisol levels in all 62 CCH, which encourages us to conduct studies under different management systems in the same geographic region to verify the hormone dynamics.
Finally, given the multifactorial variability of cortisol in the horse, Douglas (1999) proposed evaluating the rhythmicity of the circadian cycle using morning and evening measurements. The determination of the rate of change through this method requires knowledge of the basal concentration. Therefore, future studies should search for alterations in the circadian cycle in CCH to obtain more reliable individual interpretations and to elucidate the behavior of this hormone in stress situations.
In conclusion, the average morning SC value for the CCH was estimated to be 13.3 ± 7.43 μg/dL. This concentration can be used as a reference value in CCH of either gender (non-pregnant females) independent of the age group (provided they exceed one year of age) for activities with low physical demands, such as recreation, saddling, or reproduction.
Acknowledgements
The authors would like to acknowledge the staff at Test Clinical Veterinary Laboratory (Medellin, Colombia) for their support in the purchase of immunological kits and the logistics for developing the procedures.
Conflicts of interest
The authors declare they have no conflicts of interest with regard to the work presented in this report
References
Alexander SL, Irvine CH. The effect of social stress on adrenal axis activity in horses: The importance of monitoring corticosteroid binding globulin capacity. J Endocrinol 1998; 157:425-32.
Armengou L, Jose-Cunilleras E, Ríos J, Cesarini C, Viu J, Monreal L. Metabolic and endocrine profiles in sick neonatal foals are related to survival. J Vet Intern Med 2013; 27:567-575.
Bohák Z, Szabó F, Beckers J, Melo de Sousa N, Kutasi O, Nagy K, Szenci O. Monitoring the circadian rhythm of serum and salivary cortisol concentrations in the horse. Dom Anim Endocrinolo 2013; 45:38-42.
Borer-Weir KE, Menzies-Gow NJ, Bailey SR, Harris PA, Elliott J. Seasonal and annual influence on insulin and cortisol results from overnight dexamethasone suppression tests in normal ponies and ponies predisposed to laminitis. Equine Vet J 2013; 45:688-693.
Broom DM. Behaviour and welfare in relation to pathology. Appl Anim Behav Sci 2006; 97:78-83.
Comin A, Veronesi MC, Montillo M, Faustini M, Valentini S, Cairoli F, Prandi A. Hair cortisol level as a retrospective marker of hypothalamic-pituitary-adrenal axis activity in horse foals. Vet J 2012; 194:131-132.
Christensen JW, Beekmans M, Van Dalum M, Van Dierendonck Effects of hyperflexion on acute stress responses in ridden dressage horses. Physiol Behav 2014; 128:39-45.
Douglas R. Circadian cortisol rhythmicity and equine Cushing´s- like disease. J Equine Vet Scien 1999; 19:684-687.
Fazio E, Medica P, Aronica V, Grasso L, Ferlazzo A. Circulating β-endorphin, adrenocorticotrophic hormone and cortisol levels of stallions before and after short road transport: Stress effect of different distances. Acta Vet Scand 2008; 50:1-7.
Ferlazzo A, Medica P, Cravana C, Fazio E. Circulating b-endorphin, adrenocorticotropin, and cortisol concentrations of horses before and after competitive show jumping with different fence heights. J Equine Vet Scien 2012; 32:740-746.
Freymond B, Bardou D, Briefer EF, Bruckmaier R, Fouché N, Fleury J, Maigrot L, Ramseyer L, Zuberbühler K, Bachmann I. The physiological consequences of crib-biting in horses in response to an ACTH challenge test. Physiol Behav 2015; 151:121-128.
Gold JR, Divers TJ, Barton MH, Lamb SV, Place NJ, Mohammed HO, Bain FT. Plasma adrenocorticotropin, cortisol, and adrenocorticotropin/cortisol ratios in septic and normal-term foals. J Vet Intern Med 2007; 21:791-796.
Gust DA, Wilson ME, Stocker T, Conrad S, Plotsky PM, Gordon TP. Activity of the hypothalamic-pituitary-adrenal axis is altered by aging and exposure to social stress in female rhesus monkeys. J Clin Endocrinol Metab 2000; 85:2556-2563.
Haffner JC, Juergens T, Yagaan B, Mendjargal A, Yandag G. Mongolian horses: training and racing. J Equine Vet Scien 2004; 24:5-8.
Haffner JC, Fecteau KA, Eiler H, Tserendorj T, Hoffman RM, Oliver JW. Blood steroid concentrations in domestic Mongolian horses. J Vet Diagn Invest 2010; 22:537-543.
Hart KA, Slovis N, Barton M. Hypothalamic-pituitary-adrenal axis dysfunction in hospitalized neonatal foals. J Vet Intern Med 2009; 23:901-912.
Hart KA, Barton MH. Adrenocortical insufficiency in horses and foals. Vet Clin Equine 2011; 27:19-34.
Hart KA, Barton MH, Ferguson DC, Berghaus R, Slovis NM, Heusner GL, Hurley DJ. Serum free cortisol fraction in healthy and septic neonatal foals. J Vet Intern Med 2011; 25:345-355.
Irvine CH, Alexander SL. Factors affecting the circadian rythm in plasma cortisol concentrations in the horse. Domest Anim Endocrinol 1994; 11:227-238.
Kaneko JJ, Harvey JW, Bruss ML. Clinical biochemistry of domestic animals. 6th ed, Elsevier Inc. 2008, 916p.
Kedzierski W, Cywinska A, Strzelec K, Kowalik S. Changes in salivary and plasma cortisol levels in purebred Arabian horses during race training session. Anim Sci J 2014; 85:313-317.
McCue PM. Equine Cushing’s disease. Vet Clin Equine 2002; 18:533-543.
Mair TS, Sherlock CE, Boden LA. Cortisol concentrations in horses with colic. Vet J 2014; 201:370-377.
Martos N, Ayala I, Hernández J, Gutiérrez C. Determinación de los niveles plasmáticos de cortisol en diferentes patologías de los équidos. An Vet (Murcia) 2003; 19:129-140.
McGreevy P, Harman A, McLean A, Hawson L. Over-flexing the horse’s neck: A modern equestrian obsession? J Vet Behav 2010; 5:180-186.
Monk C, Hart K, Berghaus R, Norton N, Moore P, Myrna K. Detection of endogenous cortisol in equine tears and blood at rest and after simulated stress. Vet Ophthalmol 2014; 17:53-60.
Moreno FE. Niveles de cortisol en el neonato equino, en sus primeras 6 horas de vida. Trabajo de grado CES. Medellín, Colombia. 2009; . p.44
Nagel C, Erber R, Bergmaier C, Wulf M, Aurich J, Möstl E, Aurich C. Cortisol and progestin release, heart rate and heart rate variability in the pregnant and postpartum mare, fetus, and newborn foal. Theriogenology 2012; 78:759-767.
Nagel C, Erber R, Ille N, Wulf M, Aurich J, Möstl E, Aurich C. Heart rate and salivary cortisol concentrations in foals at birth. Vet J 2015; 203:250-252.
Nuñez C, Adelman J, Smith J, Gesquiere L, Rubenstein D. Linking social environment and stress physiology in feral mares (Equus caballus): Group transfers elevate fecal cortisol levels. Gen Comparative Endocrinol 2014; 196:26-33.
Pell SM, McGreevy PD. A study of cortisol and beta-endorphin levels in stereotypic and normal thoroughbreds. Appl Anim Behav Sci 1999; 64:81-90.
Place NJ, McGowan CM, Lamb SV, Schanbacher BJ, McGowan T, Walsh DM. Seasonal variation in serum concentrations of selected metabolic hormones in horses. J Vet Intern Med 2010; 24:650-654.
Pritchett LC, Ulibarri C, Roberts MC, Schneidera RK, Sellon DC. Identification of potential physiological and behavioral indicators of postoperative pain in horses after exploratory celiotomy for colic. Appl Anim Behav Sci 2003; 80:31-43.
Shaba JJ, Behan Braman A, Robinson N.E. Plasma cortisol concentration increases within 6 hours of stabling in RAO-affected horses. Equine Vet J 2014; 46:642-644.
Schmidt A, Möstl E, Wehnert CH, Aurich J, Müller J, Aurich CH. Cortisol release and heart rate variability in horses during road transport. Hormon Behav 2010; 57:209-215.
Schreiber, CM, Stewart, AJ; Behrend, EN, Wright J, Kemppainen R, and Busch, KA. Seasonal variation in diagnostic tests for pituitary pars intermedia dysfunction in normal aged geldings. J Vet Intern Med 2008; 22:734.
Smiet E, Van Dierendonck M, Sleutjens J, Menheere P, Van Breda E, Boer D, Back W, Wijnberg I, Van der Kolk J. Effect of different head and neck positions on behaviour, heart rate variability, and cortisol levels in lunged Royal Dutch Sport horses. Vet J 2014; 202:26-32
Notes
Author notes
* Corresponding author: José Ramón Martínez Aranzales, Escuela de Medicina Veterinaria, Facultad de Ciencias Agrarias, Universidad de Antioquia, Calle 70 No. 52-21, Medellín Colombia. Email: jose.martinez@udea.edu.co