CROP PRODUCTION
Interaction between increased CO2 and temperature enhance plant growth but do not affect millet grain production
Interaction between increased CO2 and temperature enhance plant growth but do not affect millet grain production
Acta Scientiarum. Agronomy, vol. 44, e53515, 2022
Editora da Universidade Estadual de Maringá - EDUEM
Received: 04 May 2020
Accepted: 17 August 2020
ABSTRACT. : The intergovernmental panel on climate change predicts a progressive increase in atmospheric CO2 concentration and temperature; however, their effects on cereals have been shown for a limited number of species. This study evaluates the effects of increased CO2 concentration and temperature separately and combined on millet growth and grain production in open-top chambers where the microclimate was adjusted to the following conditions: ambient CO2 and temperature; CO2 enriched (~ 800 ppm) and ambient temperature; ambient CO2 and higher temperature (+3ºC); and CO2-enriched and higher temperature. For each treatment, two chambers were used, each containing 15 7 L pots. Each pot received five seeds at the beginning of the experiment and thinning to one plant per pot at 15 days after sowing. Ten plants were harvested from each chamber 65 days after sowing and the plant height, the number of leaves and the longest root length as well as shoot and root biomass were measured. The remaining plants were harvested 130 days after sowing to evaluate grain production. The results indicate that high CO2 levels did not affect plant growth and biomass. On the other hand, plants subjected to high temperature grew 7% taller than those grown under ambient temperature. Contrastingly, plants submitted to both elevated CO2 and temperature were 19% taller and had 22% more shoot biomass than plants under ambient CO2 and temperature. However, grain production did not change in any of the environmental conditions. We provide evidence that millets are tolerant of the predicted climate changes and that grain production potential may not be affected.
Keywords: climate change, C4 plants, grain production, adaptive responses.
Introduction
Carbon dioxide (CO2) concentration in the atmosphere has increased since the industrial revolution. In the mid-eighteenth century, it was 280 ppm (Intergovernmental Panel on Climate Change [IPCC], 2013), increasing to 403 ppm in 2016 (World Meteorological Organization [WMO], 2017), and the forecast is between 700 and 1010 ppm by 2100 (IPCC, 2013). The predicted high level of CO2 may lead to a substantial temperature increase of up to 4.8ºC by the end of this century (Sage & Kubien, 2007; IPCC, 2013). Several studies have shown that plants will respond directly to the increase in atmospheric CO2 and temperature (Friend, 2010; Martinez, Oliveira, Mello, & Alzate-Marin, 2015). The CO2 is assimilated in photosynthesis and its concentration increase in the atmosphere may influence carbon uptake and assimilation rate and therefore plant growth (Shine, Fuglestvedt, Hailemariam, & Stuber, 2005; Leakey, 2009; Gray & Brady, 2016).
Temperature also has an important role in plant physiology due to the close relationship with transpiration, respiration and photorespiration (Sage & Kubien, 2007). However, above the optimum temperature range, metabolic activity may become unfeasible, leading to losses in plant growth, reproduction and survival (Streck, 2005; Gray & Brady, 2016). The effect of CO2 and temperature may vary according to the plant photosynthetic mechanism of CO2 fixation (Liang, Xia, Liu, & Wan, 2013; Martinez et al., 2015). Generally, in the absence of limitations by nutrients or water, plants with C3 metabolism increase biomass accumulation in response to a rise in CO2 concentration (Liang et al., 2013; Bishop, Betzelberger, Long, & Ainsworth, 2014; Gray & Brady, 2016). In contrast, an atmospheric CO2 concentration increase is expected to have little or no direct effect on RuBisCO activity in C4 plants and consequently in the CO2 assimilation of these species (Leakey, 2009). Because C4 plants can concentrate CO2 in RuBisCO active sites, they are also less bound to suffer carbon losses from photorespiration (Sage & Kubien, 2003; Martinez et al., 2015). In fact, they reach maximum rates of CO2 assimilation in higher temperatures (> 25 to 30ºC), when compared to C3 plants (below 20ºC; Ehleringer, Cerling, & Helliker 1997; Sage & Kubien, 2003).
Despite the atmospheric CO2 increase being associated with temperature increase (IPCC, 2014), most of the studies have considered such factors separately (Sá, Negreiros, Fernandes, Dias, & Franco, 2014; Gray & Brady, 2016). In truth, they affect plants jointly (IPCC, 2014). In C3 plants under CO2 addition and temperature increase, there is a growth reduction if compared to those only exposed to high CO2 concentrations (Gray & Brady, 2016). For C4 plants, the responses to such conditions are still varied and indefinite (Leakey et al., 2006). Some studies reported increases in shoot and root biomass production, while others did not (Vu, Allen Jr., & Gesch, 2006; Ruiz-Vera, Siebers, Drag, Ort, & Bernacchi, 2015; Bordignon, Faria, França, & Fernandes, 2019).
Pearl Millet (Pennisetum glaucum [L.] R. Br.) (Poaceae) is an annual warm climate plant from Africa (Bezançon et al., 2009), and together with corn, sorghum and sugarcane, it is among the most cultivated C4 plants worldwide in tropical regions (Leakey, 2009; Yadav & Rai, 2013). Millet in Brazil is processed almost exclusively for animal food (Pereira Filho et al., 2003). Furthermore, millet is an interesting species to be used as crop cover in tropical areas, particularly in the Cerrado (Brazilian Savannah) (Rosolem, Calonego, & Foloni, 2005). However, considering all the challenges imposed by climate change on biomass, it is essential to understand the effects it may have on millet and how it may influence the application of this planting practice. After all, one of the determinants for the success of no-till systems in tropical regions is the amount of straw accumulated on the soil surface (Rosolem et al., 2005). Therefore, this study aimed at testing the following hypotheses about the effects of CO2 and temperature increase on millet: i) high CO2 concentration under ambient temperature will not affect growth and grain production, irrespective of the CO2 level; ii) warmer temperatures will have a positive effect on growth and grain production; and iii) the positive effect of warmer temperatures on growth and grain production will be more evident on plants subjected to higher CO2 concentration.
Material and methods
Plant material and experimental design
Pennisetum glaucum seeds from cultivar ‘BRS 1501’ supplied by Embrapa Milho e Sorgo were used for this experiment. They were cultivated in open-top chambers (OTC) in a greenhouse at the Federal University of Minas Gerais, Belo Horizonte, state of Minas Gerais, Brazil. The chambers had aluminium structures covered by PVC plastic according to Aidar et al. (2002), modified by the inclusion of electrical resistances to increase the temperature inside the chambers, and internal volume of 1.53 m3.
To simulate the conditions foreseen by IPCC for 2100 (IPCC, 2013; 2014) the chambers were calibrated and monitored by sensors to keep the following treatments: Control (Ctrl, ambient CO2 and temperature); +T (ambient CO2 and increased air temperature at 3ºC above ambient); +C (high CO2 at ~ 800 ppm and ambient temperature); and +C+T (high CO2 and temperature). Eight open-top chambers were used, two for each tested treatment. The CO2 sources were from pressurised cylinders containing 99.8% CO2, 58.3 KgF cm-2. The atmospheric CO2 concentration in the chambers was constantly monitored through an auto CO2 meter SBA-4 OEM® (PP Systems). The air moisture content inside the chambers was not controlled.
The experiment was conducted under a natural photoperiod. Measurements of CO2, temperature and air relative humidity were stored every 15 min in a computer through a Remote Integrated Control System application (RICS 3.7, Evco), which also regulated temperature and CO2 concentration in the heated and high CO2 chambers, to keep experimental conditions stabilised relative to the non-heated, ambient CO2 chambers. All chambers contained temperature sensors that performed comparisons every 15 min between those exposed to ambient air temperature and those exposed to +3ºC and automatically activated the resistance, elevating temperature by ~ 3ºC in the high temperature simulating chambers. Temperature averages (ºC) during the experiment were higher in the +C+T treatment, followed by the +T treatment, the +C treatment, and the Ctrl (Figure 1A). Average relative humidity (%) was lower in the +C+T treatment, followed by the +T treatment, the +C treatment, and the Ctrl (Figure 1B).
The averages of CO2 concentration (ppm), temperature (ºC), and relative humidity (%) in the treatments were for (1) the Ctrl 407.0 ppm of CO2, 22.95ºC and 60.1% of relative humidity (RH); (2) +C+T: 771.5 ppm, 25.95ºC and 49.65% of RH; (3) +C: 783.0 ppm, 24.0ºC and 59.1% RH; and (4) +T: 386.5 ppm, 26.65ºC and 54.7% of RH.
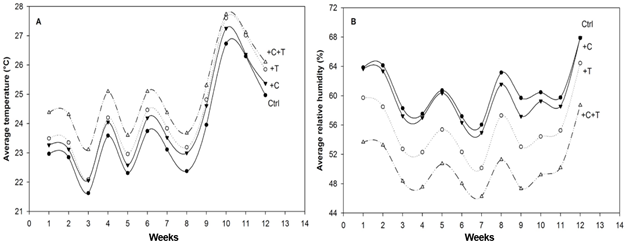
Figure 1
Average air temperature (ºC) and relative humidity (%) in open-top chambers exposed to the following environmental conditions: (1) Ctrl (ambient CO2 and temperature); (2) +T (ambient CO2 and elevated air temperature at 3ºC above ambient); (3) +C (high CO2 ~ 800 ppm and ambient temperature) and (4) +C+T (high CO2 and temperature).
PVC pots of 32.5 cm height and 7 L volume filled with Terral® commercial sterilised substrate were used. For each treatment, 30 pots (15 per chamber, a total of 120 pots) were numbered, identified, and in each, five seeds were planted. The pots were placed in the open-top chambers and irrigated daily with 300 mL of distilled water (Sá et al., 2014) and 50 mL full-strength Hoagland nutrient solution (Hoagland & Arnon, 1950) every three days. Fifteen days after planting, thinning was performed and only one plant remained in each pot. To avoid possible environmental differences among chambers, pots were exchanged weekly among chambers of the same treatment.
Plant growth measurements
Plant height and number of leaves were measured weekly from 22 to 63 days after planting. To determine the plants' relative growth rate (RGR) in the interval between 22 and 63 days after planting, the following expression was used, according to McGraw and Garbutt (1990): RGR = (lnB2-lnB1)/(t2-t1), where: ln, natural logarithm; B, average total height; t, time. After the panicle development phase (65 days of growth), 20 plants of each treatment were randomly selected and removed from the pots and separated into roots, stems, leaves, and panicles. Roots were washed in running water using a sieve and the length of the longest root (cm) was measured using a measuring tape.
After millet maturation, about 130 days of growth, we verified the same number of panicles per plant for each treatment (average [± standard error]: Ctrl = 3.8 ± 0.57; +C = 3.6 ± 0.4; +T = 3.8 ± 0.3; +C+T = 3.7 ± 0.37). We collected the main panicle from 40 plants (10 plants for each treatment) to evaluate the total grain production of the main panicle per plant. Panicles were weighed on an analytical scale (0.0001 g). Subsequently, grains were removed from panicles, placed in a forced-air oven at 60ºC and weighed after reaching constant mass (Vu & Allen Jr., 2009). Afterward, we calculated the total grain mass in the main panicle.
Data analysis
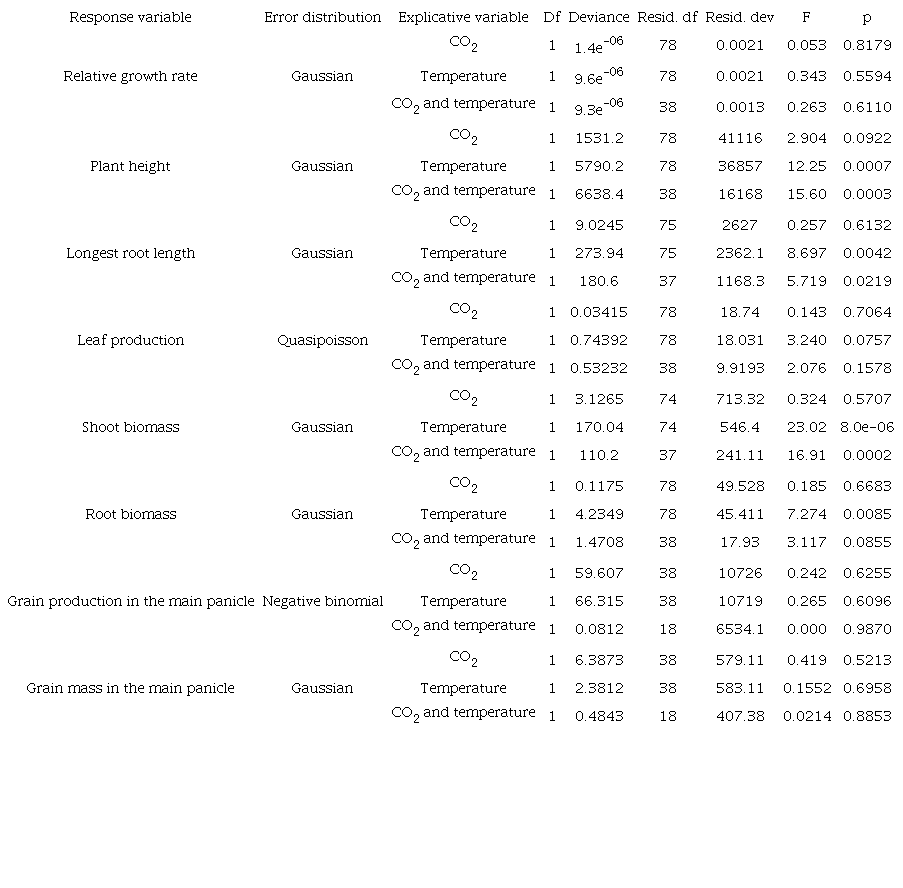
The analyses were performed in R software (R Core Team, 2018). Generalised linear models (GLM) were built with adequate distribution error and corrections of distribution performed whenever necessary (Crawley, 2013). The test of each GLM was done through an analysis of variance (Crawley, 2013) and the results with p < 0.05 were considered statistically significant.
To investigate the effect of CO2 increase, the factors high CO2 (+C) and combined high CO2 and temperature (+C+T) were grouped into the category ‘+C ambient’ and compared to the category ‘C ambient’, compounded of the high temperature (+T) and control (Ctrl) factors. Likewise, to investigate the effect of temperature increase, the factors high temperature (+T) and combined high CO2 and temperature (+C+T) were grouped into the category ‘+T ambient’ and compared to the category ‘T ambient’, the compound of the high CO2 (+C) and control (Ctrl) factors. Thus, the explicative variable was composed of treatments +C ambient vs C ambient, +T ambient vs T ambient and +C+T ambient vs Ctrl. Relative growth rate, plant height, number of leaves, longest root length, shoot biomass, mass and number of grains in the main panicle were used as variable responses.
Results and discussion
Millets grown under increased CO2 concentration had similar relative growth rates, cumulative numbers of leaves formed, numbers of grains produced, grain mass in the main panicle, and total biomass per plant compared to those exposed to ambient CO2 (p > 0.05, Table 1). These results corroborate our first hypothesis that plant growth and grain production in millets are not expected to change under CO2 enrichment in OTC chambers. Likely, the millet under the increased atmospheric CO2 was not enough to further increase RuBisCO activity already operating at its maximum rate of catalysis (Edwards & Walker, 1983; Barnaby & Ziska, 2012).
Contrastingly, millets subjected to elevated temperature had an increase in height (6.75% higher, p = 0.0007, Table 1, Figure 2A), length of the longest root (5.82% higher, p = 0.0042, Table 1, Figure 2B), shoot biomass (8.90% higher, p < 0.001, Table 1, Figure 2C), and root biomass (5.82% higher, p = 0.0008, Table 1, Figure 2D). However, the elevated temperature did not statistically affect grain production (p = 0.6096) and grain mass in the main panicle (p = 0.6858, Table 1). These responses under elevated temperature partially corroborate our assumption that millet grown in warmer temperatures presents higher growth and grain production. In general, extremely high temperatures during the reproductive stage affect grain and/or fruit formation (Hatfield et al., 2011). However, the expected temperature rise for the next century from global warming could not affect grain production, although it could favour millet growth. Yet, this scenario of temperature increase has already been observed during El Niño events and intensified by climate changes (IPCC, 2013). It is known that C4 plants may have a greater capacity for photosynthetic acclimation to high temperature (Yamori, Hikosaka, & Way, 2014), which can result in a modest increase in photosynthetic rate and plant biomass (Dwyer, Ghannoum, Nicotra, & von Caemmerer, 2007).
Millets simultaneously subjected to elevated CO2 and warmer temperatures grew 18.9% more (p = 0.0003, Table 1, Figure 2E), had a 9.4% increase in the length of the longest root (p = 0.0219, Table 1, Figure 2F), and 22.02% in shoot biomass. However, grain production and grain mass of the main panicle under increased CO2 and temperature did not vary relative to plants under ambient CO2. Considering the scenario predicted for climate changes in terms of CO2 and temperature increases, this millet cultivar would not be affected. Plant growth rate and development are dependent on the optimal average temperatures for each species (Hatfield et al., 2011). However, for most plant species, as the atmospheric temperature rises beyond the ideal, the losses in crop yields accelerate instead of falling at a rate proportional to the temperature increment (Hatfield & Prueger, 2015). Maximum growth and yields for corn, soy and cotton are reached with temperatures between 29 and 32ºC but decrease dramatically with any increase beyond this temperature interval (Schlenker & Roberts, 2009). It is feasible to consider that temperature increases from 23 to 26-27ºC would expose millets to a temperature close to the optimum range for CO2 assimilation. According to Streck (2005), the two most important metabolic functions in determining the extent of plant growth are CO2 assimilation and water use efficiency, as indicated for C4 grasses also grown in OTC chambers (Faria, Marabesi, Gaspar, & França, 2018), the same conditions in which millet was grown.
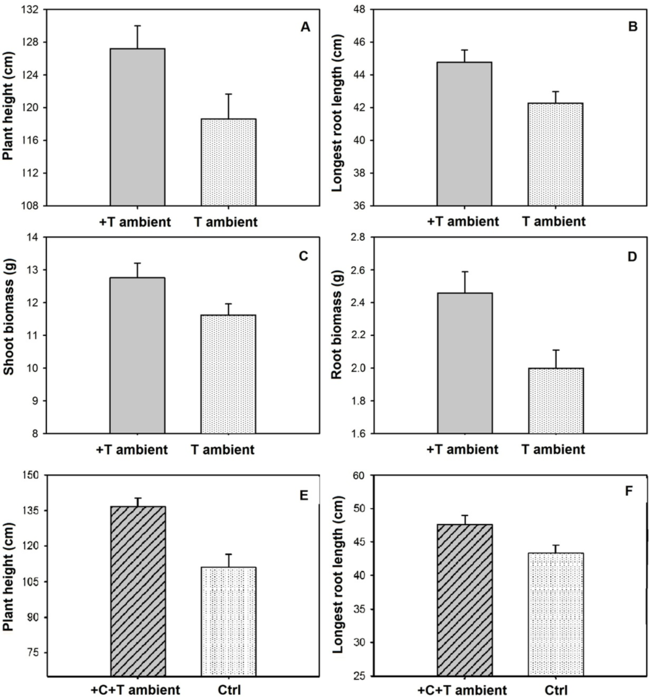
Figure 2
Comparison of growth parameters of millet in open-top chambers. 1. Average (± standard error) of height (cm); (A) length of the longest root (cm); (B) shoot biomass (g); (C) and root biomass (g); (D) subjected to elevated temperature (+T ambient, 3ºC above ambient temperature) and ambient temperature (T ambient). 2. Average (± standard error) of height (cm); (E) and length of the longest root (cm); (F) of millet under both elevated CO2 and temperature (+C+T ambient) and ambient CO2 and temperature (Ctrl).
The more significant growth in shoot height and biomass in millets could be an advantage for this cultivar since it is mainly used as forage for feeding cattle and other animals and as silage (Pereira Filho et al., 2003). Indeed, keeping the millet straw on the soil surface provides a considerable reserve of nutrients that can easily become available for the next harvest (Gassen & Gassen, 1996). The longest root length was also higher under these changed environmental conditions. Moreover, the high lignin content of straws could allow an increase in carboxylic and humic acids, making the soil less susceptible to compaction and erosion, improving the structure and stabilisation of soil aggregates (Lanzanova et al., 2007). These changes also promote greater efficiency and a sustainable no-tillage system needed in the face of climate change scenarios (Lanzanova et al., 2007).
Several studies suggest a drop in grain production for plants growing under elevated CO2 concentration and temperature (Streck, 2005; Ruiz-Vera et al., 2015). However, unlike Zea mays (Ruiz-Vera et al., 2015), the grain production of millet was not affected negatively by the treatments. The temperature increase may affect the reproductive development of plants anticipating reproductive events or causing damages by heat in reproductive structures (Gray & Brady, 2016). The lack of negative effects in grain production is likely related to the ‘BRS 1501’ millet cultivar used in this study, which is considered good for grain production and as a cover crop (Pereira Filho et al., 2003; Rosolem et al., 2005).
Conclusion
We provide evidence that the increased CO2 concentration or temperature or both conditions predicted by IPCC could positively affect biomass accumulation of Pennisetum glaucum without affecting grain production. This increased biomass production represents a greater amount of straw that can assist in covering the soil and recycling nutrients or as forage for animals. This species can, therefore, be resistant to climate change and can be an alternative crop for coverage of soil and as a cover crop in a new environmental scenario.
Acknowledgements
We thank L. Tameirão, A. R. Cordeiro, M. B. Matias, M. S. M. Castro, C. Rago and A. P. Faria for experimental assistance and two anonymous reviewers for their valuable comments and suggestions. This research was supported by Fundação de Amparo à Pesquisa do Estado de Minas Gerais (Fapemig), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (Capes). We also thank the Embrapa Milho e Sorgo for seed supply
References
Aidar, M. P. M., Martinez, C. A., Costa, A. C., Costa, P. M. F., Dietrich, S. M. C., & Buckeridge, M. S. (2002). Effect of atmospheric CO2 enrichment on the establishment of seedlings of Jatobá, Hymenaea Courbaril L. (Leguminosae, Caesalpinioideae). Biota Neotropica, 2(1), 1-10. DOI: https://doi.org/10.1590/S1676-06032002000100008
Barnaby, J. Y., & Ziska, L. H. (2012). Plant responses to elevated CO2. In eLS (p. 1-10). Chichester, EN: John Wiley & Sons, Ltd.
Bezançon, G., Pham, J.-L., Deu, M., Vigouroux, Y., Sagnard, F., Mariac, C., ... Chantereau, J. (2009). Changes in the diversity and geographic distribution of cultivated millet (Pennisetum glaucum (L.) R. Br.) and sorghum (Sorghum bicolor (L.) Moench) varieties in Niger between 1976 and 2003. Genetic Resources and Crop Evolution, 56(2), 223-236. DOI: https://doi.org/10.1007/s10722-008-9357-3
Bishop, K. A., Betzelberger, A. M., Long, S. P., & Ainsworth, E. A. (2014). Is there potential to adapt soybean (Glycine max Merr.) to future [CO2]? An analysis of the yield response of 18 genotypes in free-air CO2 enrichment. Plant, Cell & Environment, 38(9), 1765-1774. DOI: https://doi.org/10.1111/pce.12443
Bordignon, L., Faria, A. P., França, M. G. C., & Fernandes, G. W. (2019). Osmotic stress at membrane level and photosystem II activity in two C4 plants after growth in elevated CO2 and temperature. Annals of Applied Biology, 174(2), 113-122. DOI: https://doi.org/10.1111/aab.12483
Crawley, M. J. (2013). The R book. New York, NY: John Wiley & Sons.
Dwyer, S. A., Ghannoum, O., Nicotra, A., & von Caemmerer, S. (2007). High temperature acclimation of C4 photosynthesis is linked to changes in photosynthetic biochemistry. Plant, Cell & Environment , 30(1), 53-66. DOI: https://doi.org/10.1111/j.1365-3040.2006.01605.x
Edwards, G., & Walker, D. A. (1983). C3, C4. mechanisms, and cellular and environmental regulation, of photosynthesis. Los Angeles, CA: University of California Press.
Ehleringer, J. R., Cerling, T. E., & Helliker, B. R. (1997). C4 photosynthesis, atmospheric CO2, and climate. Oecologia, 112, 285-299. DOI: https://doi.org/10.1007/s004420050311
Faria, A. P., Marabesi, M. A., Gaspar, M., & França, M. G. C. (2018). The increase of current atmospheric CO2 and temperature can benefit leaf gas exchanges, carbohydrate content and growth in C4 grass invaders of the Cerrado biome. Plant Physiology and Biochemistry, 127, 608-616. DOI: https://doi.org/10.1016/j.plaphy.2018.04.042
Friend, A. D. (2010). Terrestrial plant production and climate change. Journal Experimental Botany, 61(5), 1293-1309. DOI: https://doi.org/10.1093/jxb/erq019
Gassen, D. N., & Gassen, F. R. (1996). Plantio direto: o caminho do futuro. Passo Fundo, RS: Aldeia Sul.
Gray, S. B., & Brady, S. M. (2016). Plant developmental responses to climate change. Developmental Biology, 419(1), 64-77. DOI: https://doi.org/10.1016/j.ydbio.2016.07.023
Hatfield, J. L., & Prueger, J. H. (2015). Temperature extremes: Effect on plant growth and development. Weather and Climate Extremes, 10(Part A), 4-10. DOI: https://doi.org/10.1016/j.wace.2015.08.001
Hatfield, J. L., Boote, K. J., Kimball, B. A., Ziska, L. H., Izaurralde, R. C., Ort, D., … Wolfe, D. (2011). Climate impacts on agriculture: implications for crop production. Agronomy Journal, 103(2), 351-370. DOI: https://doi.org/10.2134/agronj2010.0303
Hoagland, D. R., & Arnon, D. I. (1950). The water-culture method for growing plants without soil. Berkeley, CA: California Agricultural Experiment Station.
Intergovernmental Panel on Climate Change [IPCC]. (2013). Summary for policymakers. In T. F. Stocker, D. Qin, G.-K. Plattner, M. Tignor, S. K. Allen, J. Boschung, … P. M. Midgley (Eds.), Climate change 2013: the physical science basis. contribution of working group i to the fifth assessment report of the intergovernmental panel on climate change (p. 1-30). New York, NY: Cambridge University Press.
Intergovernmental Panel on Climate Change [IPCC]. (2014). Climate change 2014: synthesis report. contribution of working groups i, ii and iii to the fifth assessment report of the intergovernmental panel on climate change. Switzerland, CH: IPCC.
Lanzanova, M. E., Nicoloso, R. S., Lovato, T., Eltz, F. L. F., Amado, T. J. C., & Reinert, D. J. (2007). Atributos físicos do solo em sistema de integração lavoura-pecuária sob plantio direto. Revista Brasileira de Ciência do Solo, 31(5), 1131-1140. DOI: https://doi.org/ 10.1590/S0100-06832007000500028
Leakey, A. D. B. (2009). Rising atmospheric carbon dioxide concentration and the future of C4 crops for food and fuel. Proceedings of the Royal Society Biological Sciences, 276(1666), 2333-2343. DOI: https://doi.org/10.1098/rspb.2008.1517
Leakey, A. D. B., Uribelarrea, M., Ainsworth, E. A., Naidu, S. L., Rogers, A., Ort, D. R., & Long, S. P. (2006). Photosynthesis, productivity, and yield of maize are not affected by open-air elevation of CO2 concentration in the absence of drought. Plant Physiology, 140(2), 779-790. DOI: https://doi.org/10.1104/pp.105.073957
Liang, J., Xia, J., Liu, L., & Wan, S. (2013). Global patterns of the responses of leaf-level photosynthesis and respiration in terrestrial plants to experimental warming. Journal of Plant Ecology, 6(6), 437-447. DOI: https://doi.org/10.1093/jpe/rtt003
Martinez, C. A., Oliveira, E. A. D., Mello, T. R. P., & Alzate-Marin, A. L. (2015). Respostas das plantas ao incremento atmosférico de dióxido de carbono e da temperatura. Revista Brasileira de Geografia Física, 8(esp.), 635-650. DOI: https://doi.org/10.26848/rbgf.v8.0.p635-650
McGraw, J. B., & Garbutt, K. (1990). The analysis of plant growth in ecological and evolutionary studies. Trends in Ecology & Evolution, 5(8), 251-254. DOI: https://doi.org/10.1016/0169-5347(90)90065-L
Pereira Filho, I. A., Ferreira, A. S., Coelho, A. M., Casela, C. R., Karam, D., Rodrigues, J. A. S., ... Waquil, J. M. (2003). Manejo da cultura do milheto. Sete Lagoas, MG: Embrapa Milho e Sorgo.
R Core Team. (2018). R: a language and environment for statistical computing. Vienna, AT: R Foundation for Statistical Computing.
Rosolem, C. A., Calonego, J. C., & Foloni, J. S. S. (2005). Potassium leaching from millet straw as affected by rainfall and potassium rates. Communications in Soil Science and Plant Analysis, 36(7-8), 1063-1074. DOI: https://doi.org/10.1081/CSS-200050497
Ruiz-Vera, U. M., Siebers, M. H., Drag, D. W., Ort, D. R., & Bernacchi, C. J. (2015). Canopy warming caused photosynthetic acclimation and reduced seed yield in maize grown at ambient and elevated [CO2]. Global Change Biology, 21(11), 4237-4249. DOI: https://doi.org/10.1111/gcb.13013
Sá, C. E. M., Negreiros, D., Fernandes, G. W., Dias, M. C., & Franco, A. C. (2014). Carbon dioxide-enriched atmosphere enhances biomass accumulation and meristem production in the pioneer shrub Baccharis dracunculifolia (Asteraceae). Acta Botanica Brasilica, 28(4), 646-650. DOI: https://doi.org/10.1590/0102-33062014abb3329
Sage, R. F., & Kubien, D. S. (2003). Quo vadis C4? An ecophysiological perspective on global change and the future of C4 plants. Photosynthesis Research, 77(2-3), 209-225. DOI: https://doi.org/10.1023/A:1025882003661
Sage, R. F., & Kubien, D. S. (2007). The temperature response of C3 and C4 photosynthesis. Plant, Cell & Environment , 30(9), 1086-1106. DOI: https://doi.org/10.1111/j.1365-3040.2007.01682.x
Schlenker, W., & Roberts, M. J. (2009). Nonlinear temperature effects indicate severe damages to U.S. crop yields under climate change. Proceedings of the National Academy of Sciences, 106(37), 15594-15598. DOI: https://doi.org/10.1073/pnas.0906865106
Shine, K. P., Fuglestvedt, J. S., Hailemariam, K., & Stuber, N. (2005). Alternatives to the global warming potential for comparing climate impacts of emissions of greenhouse gases. Climatic Change, 68, 281-302. DOI: https://doi.org/10.1007/s10584-005-1146-9
Streck, N. A. (2005). Climate change and agroecosystems: the effect of elevated atmospheric CO2 and temperature on crop growth, development, and yield. Ciência Rural, 35(3), 730-740. DOI: https://doi.org/10.1590/S0103-84782005000300041
Vu, J. C. V., & Allen Jr., L. H. (2009). Stem juice production of the C4 sugarcane (Saccharum officinarum) is enhanced by growth at double-ambient CO2 and high temperature. Journal of Plant Physiology , 166(11), 1141-1151. DOI: https://doi.org/10.1016/j.jplph.2009.01.003
Vu, J. C. V., Allen Jr., L. H., & Gesch, R. W. (2006). Up-regulation of photosynthesis and sucrose metabolism enzymes in young expanding leaves of sugarcane under elevated growth CO2. Plant Science, 171(1), 123-131. DOI: https://doi.org/10.1016/j.plantsci.2006.03.003
World Meteorological Organization [WMO]. (2017). WMO greenhouse gas bulletin (GHG Bulletin) - No.13: The state of greenhouse gases in the atmosphere based on global observations through 2016. Geneva, CH: WMO.
Yadav, O. P., & Rai, K. N. (2013). Genetic improvement of pearl millet in India. Agricultural Research, 2, 275-292. DOI: https://doi.org/10.1007/s40003-013-0089-z
Yamori, W., Hikosaka, K., & Way, D. A. (2014). Temperature response of photosynthesis in C3, C4, and CAM plants: temperature acclimation and temperature adaptation. Photosynthesis Research , 119, 101-117. DOI: https://doi.org/10.1007/s11120-013-9874-6
Author notes
*Author for correspondence. E-mail: gw.fernandes@gmail.com
