Biotecnologia
Extraction and structural properties of Acanthophora muscoides (Rhodophyceae) extracellular matrix sulfated polysaccharides and their effects on coagulation
Extração e propriedades estruturais de polissacarídeos sulfatados de matriz extracelular de Acanthophora muscoides (Rhodophyceae) e seus efeitos na coagulação
Extraction and structural properties of Acanthophora muscoides (Rhodophyceae) extracellular matrix sulfated polysaccharides and their effects on coagulation
Acta Scientiarum. Technology, vol. 38, no. 3, pp. 273-282, 2016
Universidade Estadual de Maringá
Received: 21 December 2014
Accepted: 15 March 2016
Abstract: Acanthophora muscoides (Rhodophyta) contains structurally heterogeneous sulfated polysaccharides (Am-SPs) with pharmacological importance; however, its matrix SPs composition has not been still extensively investigated. This study sequentially extracted and compared the structural features and the in vitro anticoagulant effects of the Am-SPs. Papain-extraction sequence yielded Am.E-1, Am.E-2 and Am.E-3 containing differences among the relative proportions of sulfate (26.18-33%) and hexoses (42.02-60.67%) based on chemical analyses. One- (1H) and two-dimensions (1H/13C) nuclear magnetic resonance experiments showed very complex Am-SPs composed of alternating 4-linked-α-galactopyranosyl units and 3-linked-β-galactopyranosyl units presenting variable sulfation, CH3 substitutions and 3,6-anhydro-α-L-galactose units and pyruvated-D-galactose residues, respectively, typical of agarocolloids. Different chromatographic profiles (DEAE-cellulose) were observed, with fractions (Am I, Am II and Am III eluted with 0.5, 0.75 and/or 1 M of NaCl, respectively) revealing charge density patterns and distinct mobility to heparin by agarose-electrophoresis and, when analyzed by polyacrylamide-electrophoresis, a dispersive migration and similar mobility as chondroitin-6-sulfate for Am I fractions were noted. Regarding the activated partial thromboplastin time test, fractions had no virtually anticoagulation (1.47→3.07 IU mg-1) in comparison with 193 IU mg-1 heparin. Therefore, Am-SPs show significantly lower anticoagulation than heparin.
Keywords: Rhodophyceae, cell-wall, polyanionics, structural chemistry, clotting.
Resumo: Acanthophora muscoides (Rhodophyta) contém polissacarídeos-sulfatados (Am-PSs) heterogêneos estruturalmente com importância farmacológica. Entretanto, ainda carece de investigação quanto à composição de PSs da sua matriz. Os Am-PSs foram extraídos sequencialmente e comparados às características estruturais e aos efeitos anticoagulantes in vitro. A sequência de extração com papaína rendeu Am.E-1, Am.E-2 e Am.E-3 contendo diferenças entre as proporções relativas de sulfato (26,18-33%) e hexoses (42,02-60,67%) baseadas nas análises químicas. Experimentos de ressonância magnética nuclear uni-/bi-dimensionais mostraram Am-PSs muito complexos compostos de unidades alternantes de α-galactopiranosil 4-ligada e de unidades β-galactopiranosil 3-ligada, apresentando sulfatação variável, substituições de CH3 e de unidades de 3,6-anidro-α-L-galactose e de resíduos de D-galactose-piruvatada, respectivamente, típicos de agarocoloides. Foram observados perfis cromatográficos (DEAE-celulose) diferentes, com frações (Am I, Am II e Am III, eluídas com 0,5; 0,75 e/ou 1 M de NaCl, respectivamente) revelando graus de densidade de carga e mobilidade distinta à heparina por eletroforese em agarose. Eletroforese em poliacrilamida apresentou frações Am I com migração dispersiva e mobilidade similar ao condroitim-6-sulfato. Observaram-se, ainda, frações destituídas praticamente de anticoagulação (1,47→3,07 UI mg-1) comparadas à heparina (193 UI mg-1), segundo teste do tempo de tromboplastina parcial ativada. Portanto, Am-PSs mostram anticoagulação significativamente inferior que o da heparina.
Palavras-chave: Rhodophyceae, parede celular, polianiônicos, química estrutural, coágulo.
Introduction
Marine macroalgae (seaweeds) contain structurally unique metabolites (e.g., carbohydrates, lectins and lipids) (Mayer, Rodriguez, Berlinck, & Hamann, 2007; Miguel et al., 2014) recognized to have a broad spectrum of functionalities for scientific studies (e.g., pharmaceutics) (Cardozo et al., 2007; Fidelis et al., 2014; Wu, Shiu, Hsieh, & Tsai, 2016), industrial applications (e.g., foods and cosmetics) (Cardozo et al., 2007; Prajapati, Maheriya, Jani, & Solanki, 2014), and in biotechnological field (e.g., tissue engineering and drug delivery) (Pereira & Costa-Lotufo, 2012) due to their pronounced physical (e.g., gelling and stabilizing) and biological (e.g., anticoagulation and anti-inflammation) abilities of considerable interest toward polymer industry worldwide (Cardozo et al., 2007; Rodrigues, Vanderlei, Quinderé, Fontes, & Benevides, 2010; Pomin, 2012; Prajapati et al., 2014).
Algae bioprospection for novel potentially active natural chemicals have revealed innumerable sulfated polysaccharides (SPs) with health-related benefits (Cardozo et al., 2007; Rodrigues et al., 2012; Rodrigues et al., 2013), generating biotechnological innovation (Prajapati et al., 2014) and economical impact (Cardozo et al., 2007; Pereira & Costa-Lotufo, 2012). These marine glycans of high molecular weights (Pomin, 2012; Fidelis et al., 2014) are abundantly available in the cell-walls and intercellular space as structural components of the algae (Estevez, Ciancia, & Cerezo, 2008; Rodrigues, Torres, Alencar, Sampaio, & Farias, 2009; Miguel et al., 2014). Anticoagulation (Rodrigues et al., 2009; Rodrigues et al., 2013; Mourão, 2015), antinociception and anti-inflammation (Araújo et al., 2012; Rodrigues et al., 2012; Wu et al., 2016) are some of their most important pharmacological properties. Although also occurring in other living organisms (Aquino, Landeira-Fernandez, Valente, Andrade, & Mourão, 2005; Dantas-Santos et al., 2012; Chang, Lur, Lu, & Cheng, 2013), sulfated galactans (mainly carrageenan and agar) (Rhodophyceae) (Estevez et al., 2008; Mourão, 2015), fucan or fuicodan (Phaeophyceae) (Leite et al., 1998; Pereira & Costa-Lotufo, 2012) and other structurally diverse SPs found in Chlorophyceae (Rodrigues et al., 2013; Rodrigues et al., 2014) are the main classes of these biomolecules present in nature. Yield and physical-chemical and biological properties of SPs depend 1) on the morphology of the thallus; 2) the tissue under study (Estevez et al., 2008; Miguel et al., 2014); 3) the use of different protocols and algal species for extraction (Rodrigues et al., 2009; Rodrigues et al., 2011; Araújo et al., 2012); 4) climatic factors; and 5) growth and/or the reproducible cycle of the algae (Zvyagintseva et al., 2003; Cardozo et al., 2007; Pereira & Costa-Lotufo, 2012).
SPs from red seaweeds have highly complex structures varying among different species (Estevez et al., 2008; Maciel et al., 2008; Fenoradosoa et al., 2009; Mourão, 2015) due to their different patterns on the sulfate position, linkage, side-chain substitutions and presence of methyl ethers or anhydro sugars (Cardozo et al., 2007; Maciel et al., 2008; Barros et al., 2013). They are homogeneous with basis on their backbone constitution, composed of alternating units of 3-linked β-galactopyranosyl (G-units) and 4-linked α-galactopyranosyl (D-units) or 3,6-anhydro-α-galactopyranpose (AnGal-units), with variable density in the sulfated group. According to the stereochemistry, specifically galactans with 4-linked-α-galactose residues of the L-series are termed agarans and those of the D-series are termed carrageenans (or DL-hybrids) (Cardozo et al., 2007; Maciel et al., 2008; Prajapati et al., 2014). Such variations in the structural composition of SPs, especially those with high 3,6-anhydrogalactose content and the position of sulfation on their chains, would determine their physical and biological properties (Cardozo et al., 2007). On the other hand, the red algae cell-wall matrix SPs structural and functional features have been still few understood at level of research and their applications (Estevez et al., 2008; Rodrigues et al., 2009; Amorim et al., 2012).
The incidence of diseases related to heart and blood have significantly increased in recent decades worldwide (Pomin, 2012). Although crucial in clinical thrombotic events, unfractionated heparin (UHEP), a 1,4-linked linear glycosaminoglycan variously sulfated consisting of β-D-glucosamine (N-sulfated or acetylated) and uronic acid (β-D-glucuronic acid or α-L-iduronic acid) residues, and extracted from animal tissues, manifests extensive bleeding side effects (mainly hemorrhage) (Nader et al., 2001). Additionally, the alarming report of UHEP that has been contaminated with glycosaminoglycan oversulfated chondroitin sulfate (inducing hypotension) and some fatal events during the “UHEP crisis” (Pomin, 2012) have caused a pressing need to search novel specific glycans from renewable sources displaying analogies with UHEP (Leite et al., 1998; Athukorala, Jung, Vasanthan, & Jeon, 2006; Dantas-Santos et al., 2012; Pomin, 2012; Rodrigues et al., 2013; Mourão, 2015; Ustyuzhanina et al., 2016).
The genus Acanthopohra J. V. F. Lamouroux (Rhodomelaceae, Ceramiales) of red algae shows a discontinuous pattern of global distribution. Extracts containing agar-type polysaccharides have been examined for antiviral and anticoagulant effects (Duarte et al., 2004). Agar for industrial use has been extracted from Gracilariaceae, Gelidiaceae and Phyllophoraceae families, using different protocols (Cardozo et al., 2007; Maciel et al., 2008; Amorim et al., 2012). Brazilian samples of A. muscoides (Linnaeus) Bory de Saint-Vicent contained three SPs fractions (Am I, Am II and Am III), of which Am II manifested antinociception, anti-inflammation (Quinderé et al., 2013), and antithrombotic effects devoid of the serpin-dependent action (Quinderé et al., 2014), as well as attenuated inflammation in atherogenesis in vivo (Quinderé et al., 2015). Herein, we sequentially extracted and compared the yield and physical and chemical features; it also structural investigation of its SPs was performed along the extractions, using nuclear magnetic resonance analytical method. Further, its polysaccharidic fractions have been assessed for its in vitro effects on coagulation.
Material and methods
Yield, chemical composition and structural analysis of A. muscoides crude SPs extracts
Samples of A. muscoides were manually collected by hand from the mesolittotal region from Pacheco Beach (Ceará State, Caucaia) on the Northwestern coast of Brazil. Water temperature, pH and salinity were measured at the site, with values of 29°C, 8.1 and 35%, respectively. Then, from plastic bags, they were taken to the Carbohydrates and lectins laboratory (CarboLec), Federal University of Ceará, Brazil. After collection, salt, macroscopic epiphytes and sand were removed from algal samples with tap water and, finally, carefully rinsed with distillated water, macerated in liquid nitrogen and stored at 20°C until use (Quinderé et al., 2013). A voucher specimen (n. 46093) was deposited in the Herbarium Prisco Bezerra in the Department of Biology, Federal University of Ceará, Brazil. The analyses of the A. mucoides SPs were performed at Connective Tissue laboratory, Federal University of Rio de Janeiro, Brazil.
Papain-assisted extraction (6 hours, 60°C) in 100 mM sodium acetate buffer (pH 5.0) containing cysteine and EDTA (both 5 mM) (Quinderé et al., 2013) was sequentially applied to obtain crude SPs extracts from five grams of the dehydrated algal tissue (Rodrigues et al., 2009). Lyophilized crude SPs extracts (12 mg) were then dissolved in 8 mL of 50 mM sodium acetate buffer (pH 5.0) and applied to DEAE cellulose column (1.2 × 12 cm) equilibrated with the same solution. The column was developed using a stepwise gradient of 0 to 1 M NaCl at 0.25 M intervals in the same solution. The flow rate of the column was 2.5 mL min-1 and fractions were collected and analyzed for SPs using the metachromatic assay containing dimethylmethylene blue with an Amersham Biosciences Utrospec 1100 spectrophotometer at 525 nm. The metachromatic fractions were then dialyzed and freeze-dried. The crude SPs yields (%) were calculated as the percentage of dry matter. Quantitative determination of sulfate, total sugars and contaminant proteins of the SPs fractions were carried out. The purity of the SPs fractions was checked by agarose gel electrophoresis procedure. These experimental protocols were performed, as previously described (Quinderé et al., 2013). Polyacrylamide gel electrophoresis was also performed to estimate the molecular weights of the SPs fractions by comparison with the electrophoretic mobility of standard glycosaminoglycans unfractionated heparin (UHEP, ~ 14 kDa), chondroitin-4-sulfate (C-4-S, ~ 40 kDa), chondroitin-6-sulfate (C-6-S, ~ 60 kDa) and dextran sulfate (Dex, ~ 8 kDa) (Quinderé et al., 2014).
Nuclear magnetic resonance (1H and 13C NMR) spectroscopy
1H and 13C, one-dimensional and two-dimensional spectra, of the crude SPs extracts from the red seaweed A. muscoides were recorded using a Bruker DRX 600 MHz apparatus with a triple resonance probe. About 10 mg of each sample was dissolved in 0.6 mL 99.9% deuterium oxide (Cambridge Isotope Laboratory, Cambridge, MA). All spectra were recorded at 35°C with HOD suppression by presaturation. The 1D 1H-NMR spectra were recorded using 16 scans and inter-scan delay set to 1 s. The 1H/13C edited-HSQC spectra (128 scans) were run with 1024 × 300 points and globally optimized alternating phase rectangular pulses (GARP) for decoupling. Chemical shifts are displayed relative to external trimethylsilylpropionic acid at 0 ppm for 1H and relative to methanol for 13C. All spectra were processed using the SpinWorks 3.1.8 software package (Quinderé et al., 2014).
Activated partial thromboplastin time (APTT) assay
The APTT assay was performed using normal human plasma (obtained from 10 different donors at the University Hospital Clementino Fraga Filho - UHCFF), following manufacturer’s specifications. For this, a mixture of 100 μL of citrated normal human plasma and concentration of SPs (1 mg mL-1) was incubated with 100 μL of APTT reagent (kaolin bovine phospholipid reagent). After 2 min of incubation at 37°C, 100 μL of 20 mM CaCl2 was added to the mixtures, and the clotting time was recorded in a coagulometer (Amelung KC4A). Unfractionated heparin (UHEP) with 193 international units per mg (IU mg-1) of polysaccharide was used as the standard. All the tests were performed in triplicate.
Results and discussion
The red alga A. muscoides SPs were isolated and compared to examine their abundance and their structural features. Samples of this species were sequentially digested three times with papain (6 hours, 60°C), followed by precipitation with CPC and ethanol to obtain crude SPs extracts (Am.E-1, Am.E-2 and Am.E-3) for a total extraction yield of 23.47% from the dehydrated algal tissue (25°C) and lyophilization procedure. According to Table 1, Am.E-1 had highest extraction yield (17%) with SPs levels of 60.67 and 33% for total sugars and sulfate, respectively, similarly to those values found previously by Quinderé et al. (2013).
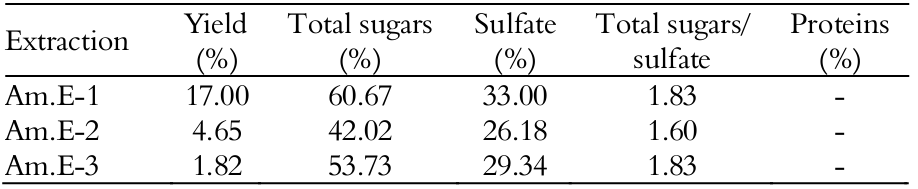
The crude SPs extracts yields (%) were calculated as the percentage of dry matter; - Not detected
Total extraction yields of Am.E-2 and Am.E-3 ranged from 1.82 to 4.65% composing 26.18-29.34% sulfate and 42.02-53.73% total sugars, respectively; however, these values were not identical when compared to those of Am.E-1. Nevertheless, on the basis of the total sugars/sulfate ratio, the compositional analysis within all extracts was similar along its wall-matrix as a result of same charge/mass ration (Fidelis et al., 2014), although exhibiting different availably in terms of total yield (Rodrigues et al., 2009).
Values of yield and composition vary among species of the order Ceramiales as a result of heterogeneity of these biopolymers, such as in Acanthophora spicifera (3.6-6.6% yield; 27-51% total hexoses and 8-20% sulfate, respectively) (Duarte et al., 2004). Sulfate ranges among different algae families, such as Gracilariaceae, Gelidiaceae and Phyllophoraceae (4-30%) (Estevez et al., 2008; Maciel et al., 2008; Amorim et al., 2012), which are the main raw material supplies for the world’s agar industry (Cardozo et al., 2007). Herein, values would reflex a disorganization of the algal tissue because proteolysis attack the extracellular matrix and cleave the interaction between SPs and proteins (Rodrigues et al., 2011; Fidelis et al., 2014), especially on the surface texture from mucilage deposition on the cell-wall, as observed on vegetative cells in red algae, in which the SPs could be mainly located (Estevez et al., 2008; Miguel et al., 2014); thus, leading to obtain novel biologically active SPs (Rodrigues et al., 2009). Papain-enzyme action, as demonstrated by the Bradford’ method, also removed contaminant proteins in all analyzed raw samples (Rodrigues et al., 2011; Araújo et al., 2012; Quinderé et al., 2013; Rodrigues et al., 2014).
Extracts Am.E-1, Am.E-2 and Am.E-3, which showed to be soluble in D2O solution, were further examined for their structural features by NMR experiments (1D 1H and 2D 1H-13C edited-HSQC) (Figure 1). The 1H NMR physical technique offers valuable information of polysaccharides (Estevez et al., 2008; Maciel et al., 2008; Prajapati et al., 2014) and was conducted at 35°C temperature due to the good solubility of the polysaccharide (Quinderé et al., 2014). However, very complex spectra showing several unresolved peaks were recorded and the 1H-signals for each native extract were displayed from δH 5.6 to 1.6 ppm (Figures 1A, B and C), as based on Quinderé et al. (2014) who previously analyzed a native fraction from this same algal species with high degree of structural heterogeneity, typical for algae SPs (Maciel et al., 2008; Fenoradosoa et al., 2009; Rodrigues et al., 2014).
Although showing poorly resolved spectra, it was possible to infer resonances associated to the presence of α-1H- and β-1H-anomeric signals attributed to protons of (1,4)-linked galactopyranose and anhydrogalactose at down-field region and of (1,3)-linked galactopyranoses at up-field region (Maciel et al., 2008; Fenoradosoa et al., 2009), respectively. Spectra also presented two singlets at δH 3.7 ppm related to 2-O-methyl groups (Fenoradosoa et al., 2009) and another signal at δH 1.7 ppm indicative of CH3 groups (Rodrigues et al., 2014), which would correspond, respectively, the chemical shifts of a methyl-esterified C2 position, as well as from the CH3 groups themselves, as confirmed in another investigation (Quinderé et al., 2014). An attempt to assign the occurrence of pyruvic acid in polysaccharidic extracts composition was further suggested with basis on variable intensity resonance signals around δH 1.7 ppm (Fenoradosoa et al., 2009).
![1D 1H (A, B and C) and 2D HSQC (D) NMR spectra of the
crude SPs extracts (Am.E-1[A], Am.E-2 [B] and Am.E-3 [C], [D]) from the red
seaweed Acanthophora muscoides. 1H
signals resonate at down and up fields regions and the labels 2-O-CH3 and CH3
indicate, respectively, methyl-esterified C-2 position, and from the CH3 groups
themselves. 1H-13C signals also reveal pyruvated (Pyr)-D-galactose residues.](../303246074002_gf1.png)
Figure 1
1D 1H (A, B and C) and 2D HSQC (D) NMR spectra of the crude SPs extracts (Am.E-1[A], Am.E-2 [B] and Am.E-3 [C], [D]) from the red seaweed Acanthophora muscoides. 1H signals resonate at down and up fields regions and the labels 2-O-CH3 and CH3 indicate, respectively, methyl-esterified C-2 position, and from the CH3 groups themselves. 1H-13C signals also reveal pyruvated (Pyr)-D-galactose residues.
Regarding the two dimensional 1H-13C edited-HSQC spectra of Am.E-1, Am.E-2 and Am.E-3, of which Am-3 showed a better resolution spectrum among themselves (Figure 1D), at spectral windows of expansions δH/C 5.4-1.3/106-16 ppm, they were not clearly liable for identification of the polysaccharidic residues. The lack of typical signals for galactans in the current study would reinforce the previous 1H NMR results due to diverse unresolved peaks variable intensity presenting among the analyzed samples (Figures 1A, B and C). The main anomeric chemical shifts values at δH/C 4.74/101 ppm were tentatively assigned to the H-1/C-1 of β-D-galactose (β-D-Gal) (resonating at up-field regions and belonging to the 4-linked units) linked to 6-O-sulfate-α-L-galactopyranose (6-O-Sul-α-L-Gal) (δH1/C1 5.3/102 ppm) (Duarte et al., 2004) or to 3,6-α-L-anhydrogalactose (3,6-α-L-AGal) (δH1/C1 5.22/98.1 ppm) (Barros et al., 2013) that resonated at down-field regions and belonged to the 3-linked units. Estevez, Ciancia, and Cerezo (2008) evaluated the system of SPs from the red seaweed Gymnogongrus torulosus and observed that hot-water extractions would result in both carrageenan and agaran, but presenting the highest amount of agaran-structures.
Due to the association between the intensity signals attributed to methyl hydrogens (around δH 1.4-1.6 ppm) and the L-galactose residue (δH 5.3 ppm), it was denoted in this study the presence of pyruvated (Pyr)-D-galactose residues in the 1H-13C edited-HSQC spectral value of A. muscoides galactans based on Barros et al. (2013), and this structural feature was in accordance to the Acanthophora genus (Duarte et al., 2004), consistent with 1H NMR spectral data (Figures 1A, B and C), since pyruvic acid has not been frequently found in agarophytes (Cardozo et al., 2007). Furthermore, spectral data revealed signals representative of 2-O-CH3, 6-SO3- and 6-unsubstituted from 1H-2 and 13C-2 pairs due to a methyl-esterified site showing methylation pattern possibly attached to O-6 of D-galactose (Duarte et al., 2004; Barros et al., 2013; Quinderé et al., 2014), as was also previously suggested (Figures 1A, B and C). It has been widely described the degree of substitution of methyl ether at C-6 varying among species (Cardozo et al., 2007; Maciel et al., 2008: Prajapati et al., 2014). Therefore, studies on the ultrastructure of these polymers would contribute for the better understanding of the cell-wall matrix architecture in the seaweeds (Estevez et al., 2008; Prajapati et al., 2014; Miguel et al., 2014).
Physical and chemical features would also reflect the A. muscoides wall-matrix polysaccharide composition
The fractionation of a sample of each native crude extract containing SPs (Am.E-1, Am.E-2 and Am.E-3), obtained from A. muscoides by ion exchange chromatography on DEAE-cellulose procedure by step elution with NaCl, is shown in Figure 2.
![Separation of SPs (Am.E-1 [A], Am.E-2 [B] and Am.E-3
[C]) from the red seaweed Acanthophora muscoides by
DEAE-cellulose. Fractions were collected and checked by metachromasia using
1,9-dimethylmethylene blue (n¾n) and phenol-H2SO4 (¡¾¡). NaCl stepwise (-). Agarose [D] and [E] polyacrylamide
gels electrophoreses. Fractions (Am-1, Am-2 and Am-3), chondroitin-6-sulfate (C-6-S,
60 kDa), chondroitin-4-sulfate (C-4-S, 40 kDa), dextran sulfate (DexS, 8 kDa) and
unfractionated heparin (UHEP, 14 kDa) present on gels were stained with 0.1%
toluidine blue.](../303246074002_gf2.png)
Figure 2
Separation of SPs (Am.E-1 [A], Am.E-2 [B] and Am.E-3 [C]) from the red seaweed Acanthophora muscoides by DEAE-cellulose. Fractions were collected and checked by metachromasia using 1,9-dimethylmethylene blue (n¾n) and phenol-H2SO4 (¡¾¡). NaCl stepwise (-). Agarose [D] and [E] polyacrylamide gels electrophoreses. Fractions (Am-1, Am-2 and Am-3), chondroitin-6-sulfate (C-6-S, 60 kDa), chondroitin-4-sulfate (C-4-S, 40 kDa), dextran sulfate (DexS, 8 kDa) and unfractionated heparin (UHEP, 14 kDa) present on gels were stained with 0.1% toluidine blue.
The elution profiles regard to the three different SPs fractions (Am I, Am II and Am III) were obtained, with 0.5, 0.75 and 1 M of NaCl, respectively, and were noticed for Am.E-1 and Am.E-3. Interestingly, only two major peaks, eluted from the DEAE-cellulose column with 0.5 and 0.75 M of NaCl, respectively, were obtained for Am.E-2. In addition, the highest total sugars dosage was found in Am II, eluted with 0.75 M of NaCl (Am.E-1), in relation to its metachromasia and fractions Am I and Am III (Figure 2A). Similar tendency was observed for the Am.E-2 and Am.E-3 extracts-derived DEAE-cellulose chromatograms, although revealing more relatively low sugars-containing SPs fractions (Figures 2B and C). These results suggested that the polysaccharides were capable of variably interacting with the DEAE-cellulose column during the papain-extraction sequence (Leite et al., 1998; Athukorala et al., 2006; Rodrigues et al., 2009; Araújo et al., 2012). Obviously, differences in the relative yields of the SPs fractions were also observed, as listed in Table 2, and 0.75 M NaCl fractions (Am II) had highest SPs yields ranging from 46.7 to 56.7%, within all other isolated A. muscoides SPs fractions among the papain-assisted extractions as a consequence of heterogeneity in availably of these algal biomaterials (Leite et al., 1998; Rodrigues et al., 2011; Araújo et al., 2012).
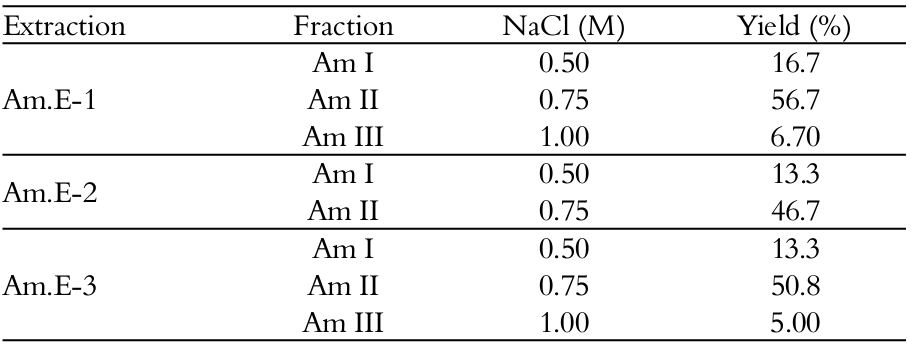
This differentiation in the yield (Tables 1 and 2), as well as in the content of total sugars (Figure 2) varying among extractions could perhaps justify the previous NMR results due to variable intensity of 3,6-anhydrogalactose residues (Figure 1), reflecting also the heterogeneity of these polymers. Duarte et al. (2004) evaluated three SPs-containing raw extracts obtained exhaustively with water (cold and hot) from the red seaweed A. spicifera. When they were fractionated by DEAE-Sephadex column by step elution with NaCl (0.25 → 4 M), the authors suggested the tendency of 3,6-anhydrogalactose and/or xylose-rich SPs to interact with the column, and themselves revealing a dispersion in these residues.
Agarose gel electrophoresis and PAGE procedures are shown in Figures 2D and E respectively, and similar electrophoretic profiles were revealed along the extraction sequence. For agarose analysis, Am-1 and Am-2 exhibited single, homogeneous and coincident metachromatic bands, while Am-3 presented a very clear band on gel; in addition, all of them had higher mobility than UHEP due to a different structural conformation and charge/mass ratio between SPs (Dietrich & Dietrich, 1976; Fidelis et al., 2014). This contrasted with a previous study by Quinderé et al. (2013) who formerly reported the A. muscoides SPs physical and chemical characterization by agarose gel when Am-1 did not appear on gel and Am-3 had an intense metachromasia, as well as differing from other studied algae species SPs (Athukorala et al., 2006; Rodrigues et al., 2009; Rodrigues et al., 2011; Araújo et al., 2012).Thus, it was speculated that this algal species would perhaps biochemically change its matrix polysaccharide composition according to the collection period (Zvyagintseva et al., 2003; Cardozo et al., 2007; Pereira & Costa-Lotufo, 2012).
Regarding the PAGE procedure, there was no electrophoretic migration of the SPs fractions during the performance of three consecutive extractions, for any standard glycosaminoglycan observed, as a result of their high molecular masses (> 100 kDa) (Pomin, 2012; Fidelis et al., 2014; Mourão, 2015), except the 0.5 NaCl fraction (Am-1) that presented polydispersion as chondroitin-6-sulfate. This electrophoretic profile was evidenced along the crude SPs extractions. Therefore, the observed characteristics of the sulfated glycans of A. muscoides cell wall would also reflect in terms of molecular masses (Duarte et al., 2004). Further studies should be performed to analyze extensively the physical and chemical system of SPs present in other species of the order Ceramiales (Estevez et al., 2008).
SPs from A. muscoides discretely delay the APTT
The in vitro anticoagulantion of the SPs fractions from the red seaweed A. muscoides was measured by testing samples on the APTT standard plasma clotting assay stimulated by a mixture of contact activators and phospholipids (Nader et al., 2001). As shown in Table 3, the plasma sample treated with SPs fractions discretely extended the clotting time (32.07 ± 0.03-66.67 ± 0.57 s) compared to that of the control sample (31.9 ± 0.23 s). The highest anti-clotting potencies were recorded for the 0.75 M NaCl fractions (3.07 (Am.E-1) -3.06 (Am.E-3) IU mg-1, respectively) when only at a high concentration of SPs (1 mg mL-1) was capable of doubling the normal APTT values in relation to a 193 IU mg-1UHEP standard. No change in the prothrombin time was noted (data not shown). Values revealed inefficacy of this system of glycans in the intrinsic and/or common and extrinsic coagulation pathways inhibition (Duarte et al., 2004; Athukorala et al., 2006; Rodrigues et al., 2010; Rodrigues et al., 2011; Araújo et al., 2012).
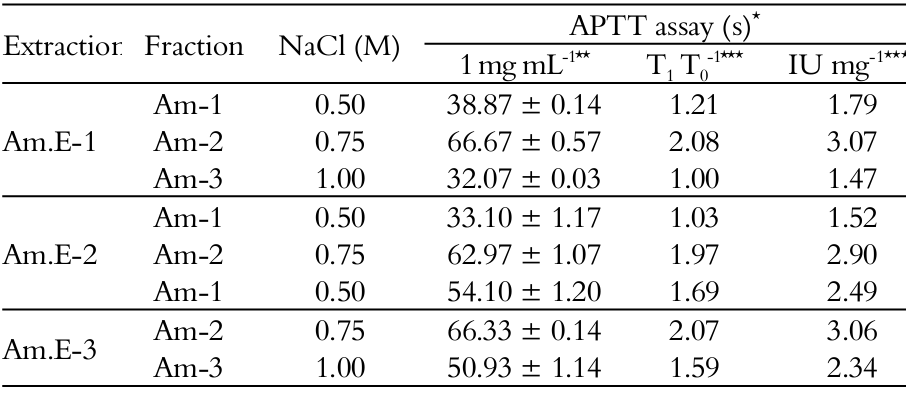
NaCl – Sodium Chloride; *Activated partial thromboplastin time (APTT); **SPs concentration to prolong the APTT in seconds; ***Anticoagulant effect expressed as T1 T0-1; ****Anticoagulant effect expressed in international units (IU) per mg of SPs (IU mg-1); UHEP (193.00 IU mg-1; 0.010 mg mL-1; APTT: 41.83 ± 0.75 s); Control: 31.9 ± 0.23 s (n = 3).
SPs from seaweeds exert complex effects in blood coagulation and several studies have been conducted to identifying novel anticoagulants (Leite et al., 1998; Duarte et al., 2004; Athukorala et al., 2006; Rodrigues et al., 2009; Fidelis et al., 2014; Mourão, 2015; Ustyuzhanina et al., 2016). Sugar type, sulfation pattern, anomeric configuration, glycosidic linkage and molecular mass make a great change in their anticoagulant actions (Pomin, 2012). The variable density in the sulfated group on their chains would result in specific bindings with plasma proteins to their molecular interactions in the haemostatic system (Pomin, 2012; Rodrigues et al., 2013; Mourão, 2015; Ustyuzhanina et al., 2016).
Agar has relatively low sulfate content compared to other anticoagulant SPs analyzed in different algae species and it is traditionally used in food and cosmetic applications as gelling agent due to its high 3,6-anhydro-α-galactose content (Cardozo et al., 2007). The system of SPs found in Ceramiales order, when analyzed by RMN analytical method, is primarily agaran structures presenting a high degree of chemical modifications and methyl ether substitutions with high anhydro-sugar content (Duarte et al., 2004; Estevez et al., 2008), as observed in the current study (Figure 2). 3,6-anhydro-α-galactose-rich SPs frequently have no strong anticoagulant effect (Cardozo et al., 2007; Quinderé et al., 2014), when measured by APTT assay, as was also previously reported for the SPs from the red alga A. spicifera (Duarte et al., 2004).
Results were actually intriguing because A. muscoides exhibited similar structural (Figure 1) and physical and chemical profiles (Table 1 and Figure 2), which were supported by APTT assay since modest anticoagulant effect was also detected in plasma samples treated with SPs fractions during the papain-assisted extraction (Table 3), and could be the result of its serpin-independent anticoagulant mechanism (Quinderé et al., 2014). This reinforced the hypothesis of same cell wall matrix polysaccharide (Duarte et al., 2004) in this algal species, based on total sugars/sulfate ratio as revealed in Table 1, although in different yield proportions (Tables 1 and 2) (Rodrigues et al., 2009). In addition, papain-extraction sequence allowed the solubilization of the SPs in water, as well as the addition of calcium chloride reagent to display the APTT assay did not modify the evaluation by in vitro testing (Araújo et al., 2012), since the water solubility of seaweeds SPs depends essentially on the levels of sulfate groups and associated cations, like calcium, which contribute to the viscosity of solutions or strength of gels formed by SPs (Cardozo et al., 2007; Prajapati et al., 2014).
Significantly lower anti-clotting effects of the A. mucoides SPs could not be correlated to other biological systems, since antinociceptive and anti-inflammatory responses in rodents, as previously reported in another study (Quinderé et al., 2013), were displayed 27-fold higher than the concentration tested on APTT assay in the current study due to a lack of information on the structure-function relationship (Pomin, 2012). More recently, an antithrombotic SPs fraction isolated from this same algal species (Quinderé et al., 2014) also reduced macrophage chemotaxis in atherosclerotic mice by impacting on cell recruitment and tissue factor content (Quinderé et al., 2015). Overall, our combined observations could reflect important biochemical aspects in the cell wall architecture polysaccharide composition of A. muscoides. However, further studies on the composition of these biomaterials in different periods of the year with possible impacts on their bioactivities should be conducted (Pereira & Costa-Lotufo, 2012).
Conclusion
Papain digestion sequence of Brazilian samples of Acanthophora muscoides (Rhodophyta) reveals a good strategy for biochemical and structural featuring of the sulfated polysaccharides present in its cell-wall extracellular matrix, when analyzed by nuclear magnetic resonance experiments and agarose and polyacrylamide gels electrophoreses procedures. Polysaccharides modify discretely the normal human plasma by classical coagulation method (APTT), as relevant for additional investigations concerning their use in other biological systems, since the structural composition does not drastically change the anti-clotting response in comparison with unfractionated heparin. Findings indicate new focus to gain insight in the study of Rhodomelaceae.
Acknowledgements
We thank to the funding from Capes/PNPD, Faperj, CNPq and MCT.
References
Amorim, R. N. S., Rodrigues, J. A. G., Holanda, M. L., Quinderé, A. L. G., Paula, R. C. M., Melo, V. M. M., & Benevides, N. M. B. (2012). Antimicrobial effect of a crude sulfated polysaccharide from the seaweed Gracilaria ornata. Brazilian Archives of Biology and Technology, 55(2), 171-181.
Aquino, R. S., Landeira-Fernandez, A. M., Valente, A. P., Andrade, I. R., & Mourão, P. A. S. (2005). Occurrence of sulfated galactans in marine angiosperms: evolutionary implications. Glycobiology, 5(1), 11-20.
Araújo, I. W. F., Rodrigues, J. A. G., Vanderlei, E. S. O., Paula, G. A., Lima, T. B., & Benevides, N. M. B. (2012). Iota-carrageenans from Solieria filiformis (Rhodophyta) and their effects in the inflammation and coagulation. Acta Scientiarum. Technology, 34(2), 127-135.
Athukorala, Y., Jung, W. K., Vasanthan, T., & Jeon, Y. J. (2006). An anticoagulative polysaccharide from an enzymatic hydrolysate of Ecklonia cava. Carbohydrate Polymers, 66(2), 184-191.
Barros, F. C. N., Silva, D. C., Sombra, V. G., Maciel, J. S., Feitosa, J. P. A., Freitas, A. L. P., & Paula, R. C. M. (2013). Structural characterization of polysaccharide obtained from red seaweed Gracilaria caudata (J Agardh). Carbohydrate Polymers, 92(1), 598-603.
Cardozo, K. H. M., Guaratini, T., Barros, M. P., Falcão, V. R., Tonon, A. P., Lopes, N. P., ... Pinto, E. (2007). Metabolites from algae with economical impact. Comparative Biochemistry and Physiology - Part C, 146(1-2), 60-78.
Chang, C. W., Lur, H. S., Lu, M. K., & Cheng, J. J. (2013). Sulfated polysaccharides of Amillariella mellea and their anti-inflammatory activities via NF-κB suppression. Food Research International, 54(1), 239-245.
Dantas-Santos, N., Gomes, D. L., Costa, L. S., Cordeiro, S. L., Costa, M. S. S. P., Trindade, E. S., ... Rocha, H. A. O. (2012). Freschwater plants synthesize sulfated polysaccharides: Heterogalactans from water hycinth (Eicchornia crassipes). International Journal of Molecular Sciences, 13(1), 961-976.
Dietrich, C. P., & Dietrich, S. M. C. (1976). Electrophoretic behaviour of acidic mucopolysaccharides in diamine buffers. Analytical Biochemistry, 70(2), 645-647.
Duarte, M. E. R., Cauduro, J. P., Noseda, D. G., Noseda, M. D., Gonçalves, A. G., Pujol, C. A., ... Cerezo, A. S. (2004). The structure of the agaran sulfate from Acanthophora spicifera (Rhodomelaceae, Ceramiales) and its antiviral activity. Relation between structure and antiviral activity in agarans. Carbohydrate Research, 339(2), 335-347.
Estevez, J. M., Ciancia, M., & Cerezo, A. S. (2008). The system of sulfated galactans from the red seaweed Gymnogongrus torulosus (Phyllophoraceae, Rhodophyta): Location and structural analysis. Carbohydrate Polymers, 73(4), 594-605.
Fenoradosoa, T. A., Delattre, C., Laroche, C., Wadouachi, A., Dulong, V., Picton, L., ... Michaud, P. (2009). Highly sulphated galactan from Halymenia durvillei (Halymeniales, Rhodophyta), a red seaweed of Madagascar marine coasts. International Journal of Biological Macromolecules, 45(2), 140-145.
Fidelis, G. P., Camara, R. B. G., Queiroz, M. F., Costa, M. S. S. P., Santos, P. C., Rocha, H. A. O, ... Costa, L. S. (2014). Proteolysis, NaOH and ultrasound-enhanced extraction of anticoagulant and antioxidant sulfated polysaccharides from the edible seaweed, Gracilaria birdiae. Molecules, 19(11), 18511-18526.
Leite, E. L., Medeiros, M. G. L., Rocha, H. A. O., Farias, G. G. M., Silva, L. F., Chavante, S. F., ... Nader, H. B. (1998). Structure and pharmacological activities of a sulfated xylofucoglucuronan from the alga Spatoglossum shroederi. Plant Science, 132(2), 215-228.
Maciel, J. S., Chaves, L. S., Souza, B. W. S., Teixeira, D. I. A., Freitas, A. L. P., Feitosa, J. P. A., ... Paula, R. C. M. (2008). Structural characterization of cold extracted fraction of soluble sulfated polysaccharide from red seaweed Gracilaria birdiae. Carbohydrate Polymers, 71(4), 55-565.
Mayer, A. M. S., Rodriguez, A. D., Berlinck, R. G. S., & Hamann, M. T. (2007). Marine pharmacology in 2003-4: Marine compounds with anthelmintic antibacterial, anticoagulant, antifungal, anti-inflammatory, antimalarial, antiplatelet, antiprotozoal, antituberculosis, and antiviral activities; affecting the cardiovascular, immune and nervous systems, and other miscellaneous mechanisms of action. Comparative Biochemistry and Physiology - Part C, 145(4), 553-581.
Miguel, T. B. A., Schmidt, E. C., Bouzon, Z. L., Nascimento, F. E. P., Cunha, M., Pereda, S. F., ... Sampaio, A. H. (2014). Morphology, ultrastructure and immunocytochemistry of Hypnea cervicornis and Hypnea musciformis (Hypneaceae, Rhodophyta) from the coastal Waters of Ceará, Brazil. Journal of Microscopy and Ultrastructure, 2(2), 104-116.
Mourão, P. A. S. (2015). Perspective on the use of sulfated polysaccharides from marine organisms as a source of new antithrombotic drugs. Marine Drugs, 13(5), 2770-2784.
Nader, H. B., Pinhal, M. A. S., Baú, E. C., Castro, R. A. B., Medeiros, G. F., Chavante, S. F., ... Dietrich, C. P. (2001). Development of new heparin-like compounds and other antithrombotic drugs and their interactions with vascular endothelial cells. Brazilian Journal of Medical and Biological Research, 34(6), 699-709.
Pereira, R. C., & Costa-Lotufo, L. V. (2012). Bioprospecting for bioactives from seaweeds: Potential, obstacles and alternatives. Brazilian Journal of Pharmacognosy, 22(4), 894-905.
Pomin, V. H. (2012). Fucanomis and galactanomics: Current status in drug discovery, mechanisms of action and role of the well-defined structures. Biochimica et Biophysica Acta, 1820(12), 1971-1979.
Prajapati, V. D., Maheriya, P. M., Jani, G. K., & Solanki, H. K. (2014). Carrageenan: A natural seaweed polysaccharide and its applications. Carbohydrate Polymers, 105(25), 97-112.
Quinderé, A. L. G., Benevides, N. M. B., Pelli, G., Lenglet, S., Burger, F., Carbone, F., … Montecucco, F. (2015). Treatment with sulphated galactan inhibits macrophage chemotaxis and reduces intraplaque macrophage content in atherosclerotic mice. Vascular Pharmacology, 71(null), 84-92.
Quinderé, A. L. G., Fontes, B. P., Vanderlei, E. S. O., Queiroz, I. N. L., Rodrigues, J. A. G., Araújo, I. W. F., ... Benevides, N. M. B. (2013). Peripheral antinociception and anti-edematogenic effect of a sulfated polysaccharide from Acanthophora muscoides. Pharmacological Reports, 65(3), 600-613.
Quinderé, A. L. G., Santos, G. R. C., Oliveira, N. M. C. G., Glauser, B. F., Fontes, B. P., Queiroz, I. N. L., ... Mourão, P. A. S. (2014). Is the antithrombotic effect of sulfated galactans independent of serpins? Journal of Thrombosis and Haemostasis, 12(1), 43-53.
Rodrigues, J. A. G., Araújo, I. W. F., Paula, G. A., Lima, T. B., Bessa, E. F., & Benevides, N. M. B. (2011). Carragenana da epífita Hypnea musciformis obtida do cultivo experimental de Solieria filiformis em Flecheiras, Estado do Ceará, Brasil. Acta Scientiarum. Technology, 33(2), 137-144.
Rodrigues, J. A. G., Neto, E. M., Santos, G. R. C., Paula, R. C. M., Mourão, P. A. S., & Benevides, N. M. B. (2014). Structural analysis of a polysaccharidic fraction obtained from the coenocytic green seaweed Caulerpa cupressoides var. lycopodium. Acta Scientiarum. Technology, 36(23), 203-210.
Rodrigues, J. A. G., Neto, E. M., Teixeira, L. A. C., Paula, R. C. M., Mourão, P. A. S., & Benevides, N. M. B. (2013). Structural features and inactivation of coagulation proteases of a sulfated polysaccharidic fraction from Caulerpa cupressoides var. lycopodium (Caulerpaceae, Chlorophyta). Acta Scientiarum. Technology, 35(4), 611-619.
Rodrigues, J. A. G., Torres, V. M., Alencar, D. B., Sampaio, A. H., & Farias, W. R. L. (2009). Extração e atividade anticoagulante dos polissacarídeos sulfatados da alga marinha vermelha Halymenia pseudofloresia. Revista Ciência Agronômica, 40(2), 224-231.
Rodrigues, J. A. G., Vanderlei, E. S. O., Quinderé, A. L. G., Fontes, B. P., & Benevides, N. M. B. (2010). Polissacarídeos sulfatados isolados das clorofíceas Caulerpa racemosa e Caulerpa cupressoides - extração, fracionamento e atividade anticoagulante. Acta Scientiarum. Biological Sciences, 32(2), 113-120.
Rodrigues, J. A. G., Vanderlei, E. S. O., Silva, L. M. C. M., Araújo, I. W. F., Queiroz, I. N. L., Paula, G. A., ... Benevides, N. M. B. (2012). Antinociceptive and anti-inflammatory activities of a sulfated polysaccharide isolated from the green seaweed Caulerpa cupressoides. Pharmacological Reports, 64(2), 282-292.
Ustyuzhanina, N. E., Bilan, M. I., Gerbs, A. G., Ushakova, N. A., Tsvetkova, E. A., Dmitrenok, A. S., ... Nifantiev, N. E. (2016). Anticoagulant and antithrombotic activities of modified xylofucan sulfate from the brown alga Punctaria plantaginea. Carbohydrate Polymers, 136(1), 826-833.
Wu, G. J., Shiu, S. M., Hsieh., M. C., & Tsai, G. J. (2016). Anti-inflammatory activity of a sulfated polysaccharide from the brown alga Sargassum cristaefolium. Food Hydrocolloids, 53(1), 16-23.
Zvyagintseva, T. N., Shevchenko, N. M., Chizhov, A. O., Krupnova, T. N., Sundukova, E. V., & Isakov, V. V. (2003). Water-soluble polysaccharides of some far-eastern brown seaweeds. Distribution, structure, and their dependence on the developmental conditions. Journal of Experimental Marine Biology and Ecology, 294(1), 1-13.
Author notes
arieviloengpesca@yahoo.com.br