Química
Oxidative stability of biodiesel blends derived from waste frying oils
Estabilidade oxidativa de blendas de biodiesel de fritura
Oxidative stability of biodiesel blends derived from waste frying oils
Acta Scientiarum. Technology, vol. 39, no. 3, pp. 375-378, 2017
Universidade Estadual de Maringá

This work is licensed under Creative Commons Attribution 4.0 International.
Received: 08 October 2015
Accepted: 23 February 2016
Abstract: The high cost of biodiesel production is mainly linked to the price of raw material.This factor has favored the use of alternative fats and oils such as those used in frying. Since biodiesel can be obtained from several vegetable and animal raw materials, the physicochemical characteristics of the fuel may vary considerably. One of these characteristics is the fatty acid composition. It directly affects the oxidative stability of biodiesel, which can be impaired when the fuel undergoes exposure to sunlight, metals, oxygen and high temperatures. In order to improve the oxidative stability of biodiesels produced from waste frying oil some studies involving blends of different raw materials have been carried out. In this sense, this work aimed to assess the characteristics resulting from the blending of soybean waste frying oil with other waste biodiesels in what concerns to oxidation. The blends of fatty materials were obtained by means of a 2² factorial design. The induction periods of biodiesel blends were enough to meet the ASTM D6751 standard. Swine fat was responsible for the increase in the induction period values.
Keywords: pequi oil, swine fat, transesterification, waste biodiesel.
Resumo: O alto custo de produção do biodiesel está ligado principalmente ao preço da matéria-prima e este fator tem favorecido a utilização de óleos e gorduras alternativos como os de fritura. Uma vez que o biodiesel pode ser obtido a partir de diversas matérias-primas vegetais e animais, as características físico-químicas do combustível são bastante variáveis, dentre elas a composição em ácidos graxos que afeta diretamente a estabilidade oxidativa do biocombustível. A estabilidade oxidativa de biodiesel pode ser prejudicada quando o combustível sofre exposição à luz solar, metais, oxigênio e altas temperaturas, principalmente. A fim de melhorar a estabilidade oxidativa de biodieseis produzidos a partir de óleos e gorduras residuais de fritura, vem sendo realizados alguns estudos que envolvem blendas de diferentes matérias-primas. Neste sentido o presente trabalho tem como objetivo avaliar as características resultantes da blendagem de biodiesel residual de soja com outros biodieseis residuais do ponto de vista oxidativo. As blendas dos materiais graxos foram obtidas a partir de planejamento fatorial 2². Os períodos de indução das blendas de biodiesel foram suficientes para atender à norma ASTM D6751, sendo que a gordura suína foi responsável pelo aumento destes valores.
Palavras-chave: óleo de pequi, gordura suína, transesterificação, biodiesel residual.
Introduction
Due to various factors, such as the price of raw materials and production costs, biodiesel does not compete economically with its fossil similar (Ong, Mahlia, Masjuki, & Honnery, 2012). In order to reduce these costs, alternative raw materials, such as waste frying oils, have been used (Aghabarari, Dorostkar, & Martinez-Huerta, 2014; Cremonez et al., 2015). Among the raw materials, soybean and canola oils stand out for having large amounts of free fatty acids and unsaturated carbon chains (Serrano et al., 2014), which compromise their oxidative stability, especially when it comes to biodiesel storage for a prolonged period of time (Serrano, Bouaid, Martínez, & Aracil, 2013). The use of raw material whose predominant carbon chains are saturated provides more stable biodiesels, where as mono- and polyunsaturated chains lead to biodiesels that are more prone to oxidation (Serrano et al., 2014).
The oxidative stability of biodiesel is normally affected by its exposure to oxygen, ultraviolet irradiation, metal contamination, moisture, dyes, some enzymes and high temperatures (Knothe & Dunn, 2003), moreover, it is also influenced by ester composition (saturated and unsaturated). Therefore, it is a feature that varies with each raw material. This degradation may occur during storage or in the engine system, what can affect its performance (Bouaid, Martinez, & Aracil, 2009). To ensure the oxidative stability, certain rules were adopted in order to maintain a standard of quality, such as the ASTM D6751, which determines that the induction period must be more than 3h in Rancimat equipment.
Aiming to improve the standards and the oxidative stability of biodiesel, studies using blends of oil biodiesels and waste frying oils have been carried out to optimize the mixture of fatty acids of these raw materials (Giakoumis, 2013; Ramos, Fernández, Casas, Rodríguez, & Pérez, 2009). In this sense, this study aims to assess the oxidative stability resulting from the blending of soybean waste frying oil with other waste biodiesels.
Material and methods
Frying process
In this work we used soybean oil (S), pequi oil (Caryocar brasiliense Camb.) (P) and swine fat (F) to fry potato chips (Solanum tuberosum L., cv. Monalisa) at a temperature of 200ºC during 5 hours.
Transesterification
Biodiesel was obtained by applying 70 g of oil or waste frying fat in the basic and homogeneous reaction of methanol transesterification for 30 minutes under constant agitation of 300 rpm and temperature of 55°C. The molar ratio alcohol/fatty material was fixed in excess of 6:1 and 1% (w/w) NaOH catalyst as a function of the mass of the fatty material, in accordance with Kumar, Tiwari and Garg (2013) and Miladinović, Krstić, Tasić, Stamenković and Veljković (2014). We performed triplicates ofthe transesterification reactions with all fatty materials. The reaction mixture was centrifuged at 3000 rpm for 10 min. to separate the phases. Then, the esters were washed with distilled water at 55°C for 10 min. and centrifuged one more time. The biodiesel was subjected to heating (100 ± 5°C) for 10 min. for the removal of excess moisture and alcohol (adapted from Geris et al., 2007).
Blends
After the transesterification of the fatty materials the biodiesels were blended, respecting the ratio exposed in Table 1
| Treatment | Proportion | Soybean | Pequi | Swine Fat |
| 1 | 10 : 0 : 0 | + | - | - |
| 2 | 8 : 2 : 0 | + | + | - |
| 3 | 8 : 0 : 2 | + | - | + |
| 4 | 6 : 2 : 2 | + | + | + |
| 5 | 8 : 1 : 1 | Central | Central | Central |
Characterization
Conversion
In order to determine the percentage of conversion to methyl esters, we used a Perkin Elmer gas chromatograph (Clarus 680) equipped with a flame ionization detector (FID) at 250°C and a split injection system at 250°C with a ratio of 1:50 for an injection volume of 2 µL. The carrier gas flow was 1.5 mL min.-1. We used a capillary column (Perkin Elmer - Elite Wax, 30 mx 0.25 mm x 0.5 um.)
Oxidative stability
To determine the induction period, we used an air blowing rate of 10h-1, which precedes the step of propagationin radical chain reactions. The oxidative stability of fatty materials was performed using Rancimat equipment, model 873 (Metrohm® - Herisau / Switzerland) according to the official standard for the determination of oxidative stability by accelerated test (EN 14112).
Results and discussion
Transesterification conversions
Table 2 shows the conversion of the transesterification of raw materials used in the study. Even when dealing with raw materials derived from frying, a quite satisfactory conversionof oil/fat was achieved (over 81%).
| Oil/Fat | Conversion (%) |
| Soybean | 89.46 |
| Pequi | 86.04 |
| Swine Fat | 81.05 |
The high conversion rate of the transesterification process emphasizes that less noble raw materials can be used without further issues, provided that they are at least subjected to some type of filtering/cleaning process. In addition to the environmental gain, the use of oils and fats derived from frying can generate economic benefits due to their low cost.
Oxidative stability of blends
After 5 hours of frying, the induction periods were sufficient to achieve the minimum time of 3h, specified by the American standard ASTM D6751, but insufficient to achieve the minimum of 6h, determined by the European standard EN 14112. Figure 1 depicts the response surface generated by the factorial design employed.
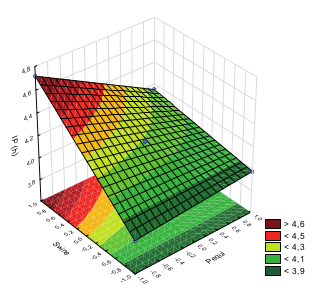
Figure 1
Figure 1. Response surface of the oxidative stability of the biodiesel blends. Equation IP = 4.19 - 0.1225Pequi* + 0.25Swine Fat*-0.16Pequi*Swine Fat*. Fcalc/Ftab= 17.068. R² = 0.9978.
Figure 1 shows that the use of biodiesel from swine fat in the blend with soybean biodiesel provided an increase in the induction period and, consequently,in the oxidative stability of the blend. Trial 3 (SF) showed the highest induction period, reaching 4.72h (Figure 2), showing that the use of swine fat biodiesel was responsible for the increased oxidative stability of the soybean biodiesel (17.4%).
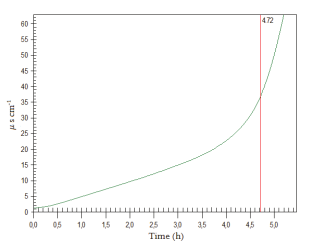
Figure 2
Figure 2. Induction period of the SF blend by means of the Rancimat method.
One of the most important factors in the study of the oxidative stability of biodiesel is the composition of the fatty acids, since it varies according to each raw material. Such importance is due to the mechanisms involved in oxidation, which are facilitated when the presence of unsaturated fatty acids is high. According to Yakoob, Narayanan, Padikkaparambil, Unni and Akbar (2014), as the number of unsaturation increases, so does the susceptibility of the ester to oxidation. Oils and fats with a higher presence of unsaturated fatty acids are preferred.
Vegetable oils and fats generally have greater oxidative stability than those of animal origin due to the presence of natural antioxidants such as carotenoids, flavonoids and tocopherols. The increased oxidative stability of biodiesel with swine fat can be explained in two ways: the first explanation shows that the vegetable oil refining processes are responsible for the removal of natural antioxidants, as described by Serrano et al. (2013). The second assumption proves that a higher number of saturated chains is responsible for the increased oxidative stability, as obtained in the blending of soybean oil esters with swine fat esters. Table 3 shows a comparison of the average composition of fatty acids
| Fatty Acid Composition (%) | Soybean | Pequi | Swine Fat |
| Palmitic (C16:0) | 11.2 | 31.4 | 25.69 |
| Palmitoleic (C16:1) | 0.2 | 0.7 | 2.82 |
| Stearic (C18:0) | 3.3 | 2.2 | 14.50 |
| Oleic (C18:1) | 23.3 | 47.3 | 40.88 |
| Linoleic (C18:2) | 52.6 | 15.6 | 12.93 |
| Linolenic (C18:3) | 6.7 | 0.7 | - |
Due to the high concentration of saturated fatty acids in swine fat-based biodiesel, the oxidative process may have been hampered by the temperature used in the frying process. Freire et al. (2012) showed that the oxidation by temperature is initiated at temperatures around 120°C in esters of unsaturated fatty acids. Therefore, it can be said that the oxidative process was achieved with greater intensity withthe use of raw materials with higher presence of unsaturated fatty acids.
Increased oxidative stability in blends of waste biodiesels is highly desirable, as it reduces the cost of biofuel manufacture from less noble raw materials when the conversions are satisfactorily achievable.
Conclusion
In this study, the induction periods of biodiesels produced from soybean oil, pequi oil and swine fat used in frying for 5 hours were sufficient to reach the minimum time of 3h determined by the American standard ASTM D6751. The use of swine fat in blends with soybean waste frying oil resulted in a significant increase in the initial induction period. This gain is essential to reduce biodiesel production costs, since waste oils and fats are less noble materials. Blending factors can also bestudied further in order to optimize the gains of oxidative stability.
References
Aghabarari, B., Dorostkar, N., & Martinez-Huerta, M. V. (2014). Synthesis of biodiesel from Nigella sativa seed oil using surfactant-Bronsted acidic-combined ionic liquid as catalyst. Fuel Processing Technology, 118(1), 296-301.
Bouaid, A., Martinez, M., & Aracil, J. (2009). Production of biodiesel from bioethanol and Brassica carinata oil: oxidation stability study. Bioresource Technology, 100(7), 2234-2239.
Cremonez, P. A., Feroldi, M., Nadaleti, W. C, De Rossi, E., Feiden, A., De Camargo, M. P., ...Klajn, F. F. (2015). Biodiesel production in Brazil: current scenario and prospectives. Renewable and Sustainable Energy Reviews, 42(1), 415-428.
Freire, L. M. S., Santos, I. M., Carvalho Filho, J. R., Cordeiro, A. M. T. M., Soledade, L. F. B., Fernandes Jr, V.J., ... Souza, A. G. (2012). Influence of the synthesis process on the properties of flow and oxidative stability of biodiesel from Jatropha curcas biodiesel. Fuel, 94(1), 313-316.
Geris, R., Santos, N. A. C., Amaral, B. A., Maia, I. S., Castro, V. D., & Carvalho, J. R. M. (2007). Biodiesel de soja – reação de transesterificação para aulas práticas de química orgânica. Química Nova, 30(5), 1369-1373.
Giakoumis, E. G. (2013). A statistical investigation of biodiesel physical and chemical properties, and their correlation with the degree of unsaturation. Renewable Energy, 50(1), 858-878.
Knothe, G., & Dunn, R.O. (2003). Dependence of oil stability index of fatty compounds on their structure and concentration and presence of metals. Journal of the American Oil Chemists Society, 80(10), 1021-1026.
Kumar, R., Tiwari, P., & Garg, S. (2013). Alkali transesterification of linseed oil for biodiesel production. Fuel, 104(1), 553-560.
Miladinović, M. R., Krstić, J. B., Tasić, M. B., Stamenković, O. S., & Veljković, V. B. (2014).A kinetic study of quicklime-catalyzed sunflower oil methanolysis. Chemical Engineering Research and Design, 92(9), 1740-1752.
Ong, H. C., Mahlia, T. M. I., Masjuki, H. H., & Honnery, D. (2012). Life cycle cost and sensitivity analysis of palm biodiesel production. Fuel, 98(1), 131-139.
Ramos, M. J., Fernández, C. M., Casas, A., Rodríguez, L., & Pérez, A. (2009). Influência da composição de ácidos graxos de matérias-primas em propriedades do biodiesel. Bioresource Technology, 100(1), 261-268.
Serrano, M., Bouaid, A., Martínez, M., & Aracil, J. (2013). Oxidation stability of biodiesel from different feedstocks: influence of commercial additives and purification step. Fuel, 113 (1), 50-58.
Serrano, M., Oliveros, R., Sánchez, M., Moraschini, A., Martínez, M., & Aracil, J. (2014). Influence of blending vegetable oil methyl esters on biodiesel fuel properties: oxidative stability and cold flow properties. Energy, 65(1), 109-115.
Yakoob, Z., Narayanan, B. N., Padikkaparambil, S., Unni, K. S., & Akbar, P. M. (2014). A review on the oxidation stability of biodiesel. Renewable and Sustainable Energy Reviews, 35(1), 136-153.
Author notes
michaelferoldi@gmail.com