Ciência, Tecnologia de Alimentos e Engenharia de Alimentos
Influence of the nutrient concentrations of whey on ethanol and biomass production and COD reduction
Influência da concentração de nutrientes do soro na produção de etanol, biomassa e redução de DQO
Influence of the nutrient concentrations of whey on ethanol and biomass production and COD reduction
Acta Scientiarum. Technology, vol. 39, 05, pp. 533-541, 2017
Universidade Estadual de Maringá

This work is licensed under Creative Commons Attribution 4.0 International.
Received: 18 October 2015
Accepted: 01 September 2016
Abstract: The present study aimed at the use of cheese whey as substrate for ethanol production by Kluyveromyces marxianus. The effect of the initial cheese whey concentration (M1 57.60; M2 45.60; M3 32.50 and M4 18.80 g L-1) on the alcoholic fermentation was studied. After sterilization, the in natura media were incubated at 30ºC for 48 hours, and then lactose, protein, ethanol, cell growth and chemical oxygen demand were analyzed. According to the results, M1 medium (the highest concentration) showed a greater production and volumetric productivity of ethanol 16.90 and 1.26 g L-1 hour-1, respectively, and resulted in the greatest cell production and volumetric productivity of 5.80 and 0.40 g L-1 hour-1, respectively. However, in terms of ethanol yield, the most satisfactory result was obtained using the M3 medium, pro 82.30% in 12 hours of fermentation. With respect to the COD, the organic substance content of M4 medium was reduced by 82.28%.
Keywords: bioprocess, biofuel, Kluyveromyces marxianus, environment.
Resumo: O presente estudo teve como objetivo realizar o aproveitamento do soro de queijo, obtido a partir da produção de queijo, utilizado como substrato para produção de etanol por meio de Kluyveromyces marxianus. Foi estudado o efeito da concentração inicial de soro (M1 57,60; M2 45,60; M3 32,50 e M4 18,80 g L-1) sobre a fermentação alcoólica. Após esterilização, os meios que se apresentaram in natura foram incubados a 30ºC por 48h, sendo realizadas análises de lactose, proteínas, etanol, crescimento celular e demanda química de oxigênio. De acordo com os resultados, o meio com maior concentração de lactose inicial (M1) apresentou maior produção e produtividade volumétrica de álcool, 16,90 e 1,26 g L-1 h-1, respectivamente, consumo de lactose, 96,20%, e maior crescimento e produtividade celular, 5,80 e 0,40 g L-1 h-1, respectivamente. Quanto ao rendimento etanólico final, o mais satisfatório ocorreu no meio M3 com 82,30% em 12h. Já a carga orgânica foi reduzida em até 82,28% no meio M4.
Palavras-chave: bioprocessos, biocombustível, Kluyveromyces marxianus, meio ambiente.
Introduction
Climate change combined with strategic needs for energy production has led to the production of alternative fuels, preferably from renewable sources (Rocha & Buckeridge, 2009, Harish, Ramaiah, & Uppuluri, 2015).
The amount of research related to biofuel production has been significant in recent years, especially with respect to the depletion of fossil fuel reserves such as oil and its derivatives, considered as a major contributor to global warming (Gupta & Demirbas, 2010, Todhanakasem, Sangsutthiseree, Areerat, Young, & Thanonkeo, 2014). Despite advances in the area of sustainable energy, the processing of raw materials and the use of technologies for the recovery and use of co-products and wastes with energetic interest still present technical challenges.
Bioethanol produced by carbohydrate fermentation from agricultural raw materials is one of the most important biotechnological products on the world scenario, and to respond to the increasing demand, it is essential to find ways to optimize the production of this biofuel (Todhanakasem et al., 2014, Vincenzi et al., 2014, Harish et al., 2015).
Due to industrial growth and development, there is a great potential for the generation of polluting waste, mainly due to its physical and chemical characteristics and the presence of organic compounds that can cause serious environmental damage if improperly disposed of in the environment (Banaszewska, Cruijssen, Claassen, & Van der Vorst, 2014).
In this context, dairy products represent an important sector of the food industry, from both the economic and social points of view. The main by-product is whey, or cheese whey, which can cause serious environmental problems due to its high organic load from biodegradable matter, having a Chemical Oxygen Demand (COD) of about 50,000 to 80,000 mg L-1 (Schultz, Chang, Hauck, & Syldatk, 2006, Vincenzi et al., 2014).
Whey is the byproduct resulting from the precipitation and removal of the casein from milk during cheese production, retaining about 55% of the milk nutrients. It is comprised of approximately 4.5% lactose, 0.6 to 0.8 % soluble proteins (albumins and globulins), 0.03 to 0.01% fat, 0.5 to 0.8% minerals (mainly sodium chloride and calcium phosphate) and 0.20 to 0.80% lactic acid (Carvalho, Prazeres, & Rivas, 2013, Murari, Moraes, Bueno, & Del Bianchi, 2013).
Only part of the cheese whey industrially generated is used to produce other products such as whey powder, protein concentrate and several dairy beverages. Consequently, much of the whey produced throughout the world is still incorporated into the dairy wastewater (Guimarães, Teixeira, & Domingues, 2010, Carvalho et al., 2013), being the main source of environmental pollution generated by this sector.
A promising alternative for a satisfactory and effective utilization of cheese whey is its use as a substrate for the growth of microorganisms able to use lactose as their source of carbon and energy, leading to the generation of commercially important bio-products such as ethanol (Guimarães et al., 2010, Murari et al., 2013).
From the microorganisms isolated and selected for this purpose, the yeast Kluyveromyces spp. is the one showing the greatest suitability, especially the genus K. marxianus. This genus shows an ample metabolic diversity and a wide range of biotechnological applications (Fonseca, Heinzle, Wittmann, & Gombert, 2008), such as the production of enzymes, ribonucleotides, oligosaccharides, oligopeptides, heterologous protein, aromatic compounds, ethanol and biomass for the food industry (Van Ooyen et al., 2006).
Many authors have reported inhibitory effects associated with these culture media due to the high lactose concentrations present in the whey during fermentation. These problems are generally attributed to the osmotic sensitivity (dueto the high lactose concentrations), and low tolerance to ethanol and high salt concentrations of some of the isolated lactose fermenting strains (Grubb & Mawson, 1993, Zafar & Owais, 2005).
Thus this study aimed to produce bioethanol from whey containing different lactose concentrations using the yeast Kluyveromyces marxianus CCT 4086, and evaluate the influence of the initial substrate concentration as a limiting factor for the development of the strain, and the reduction of the whey polluting potential.
Material and methods
Microorganism and maintenance
The yeast Kluyveromyces marxianus CCT 4086 was obtained from the Biochemistry and Microbiology Department at UNESP - Rio Claro (São Paulo State), and propagated in Potato Dextrose Agar (PDA) at 30ºC for 24 hours in a BOD incubator. It was then stored in a refrigerator at 5°C.
Cheese whey
Whey, obtained from the Milk and Dairy Products Laboratory of the Department of Food Technology and Engineering, Ibilce/Unesp, after the preparation of Fresh Minas cheese, was filtered using a vacuum pump, sterilized in an autoclave at 121°C for 10 min and stored at 4°C until used.
Preparation of the inoculum
Eight mL aliquots of cell suspension obtained from the stock culture (maintained in tubes containing [in g L-1]: 10.0 peptone, 10.0 yeast extract and 30.0 lactose, corrected to an optical density of 0.8 at 600 nm) were added to 250 mL conical flasks containing 100 mL culture medium composed of lactose (3 g L-1), yeast extract (1 g L-1), (NH4)2 SO4 (0.1 g L-1), KH2PO4 (0.10 g L-1), MgSO4 (0.1 g L-1) and CaCl2 (0.2 g L-1). The flasks were incubated at 35°C and 100 rpm for 24 hours.
After the incubation period, the medium was centrifuged at 3000 g for 20 min. The pellet was washed in 0.1% peptone water and the procedure repeated once more Murari et al. (2013), adapted from Silveira, Passos, Mantovani, and Passos (2005).
Whey fermentation at different lactose concentrations
Whey fermentation was tested with different nutrient concentrations, in order to evaluate the initial substrate concentration (mainly nutrients such as lactose and protein) as a limiting factor for growth of the yeast. Table 1 shows the nutrient concentrations of the whey tested.
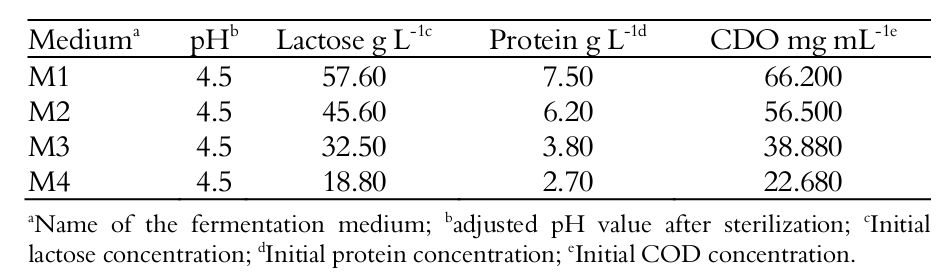
Fermentation medium
Aliquots of 15% of the standardized inoculum were added to the in nature culture media (M1, M2, M3 and M4) under sterile conditions and incubated in a BOD incubator at 35°C for 48 hours. Samples were taken during fermentation for the analysis of lactose, protein, ethanol, cell growth and chemical oxygen demand (COD). The experiments and analyses were carried out in triplicate.
Analytical techniques
The biomass concentration was determined from the dry weight, where the yeast cells were centrifuged at 3000 g for 30 min. and washed with distilled water, repeating the procedure once more. They were then placed in an oven at 105°C and weighed periodically to constant weight (Association of Official Analytical Chemists [AOAC], 1995).
The reducing sugar (lactose) content was determined using the 3.5-dinitrosalicylic acid method described by Miller (1959) with fermentation time. The standard curve was prepared using serial dilutions of a pure lactose solution.
The soluble protein concentration was determined by Lowry’s method as modified by Hartree, using BSA (Bovine Serum Albumin) as the standard (Hartree, 1972).
The ethanol content was determined by gas chromatography (Hewlett Packard Model 5890 series II equipped with a SPB-35 column and flame ionization detector). The running conditions included heating the column to 40-100ºC at a heating rate of 20ºC min-1, injection and detection temperatures of 230ºC and a carrier gas of nitrogen with a split rate of 1:5.
The COD (Chemical Oxygen Demand) was measured according to American Public Health Association (Apha, 1995), using the closed reflux and colorimetric method. The concentrations of lactose, protein and COD were determined at the beginning and end of fermentation.
The sample data were submitted to an analysis of variance (ANOVA) and the averages compared by Tukey’s test using the Kramer Graphpad Instat program (Rutgers University, Camden, New Jersey, USA). The treatments were considered significant at p < 0.05.
Determination of fermentation parameters
The parameters used to evaluate the performance of the ethanol fermentation were: substrate of product yield coefficient, volumetric productivity of ethanol and theoretical conversion of the lactose to ethanol (Equation 1, 2 and 3, respectively).
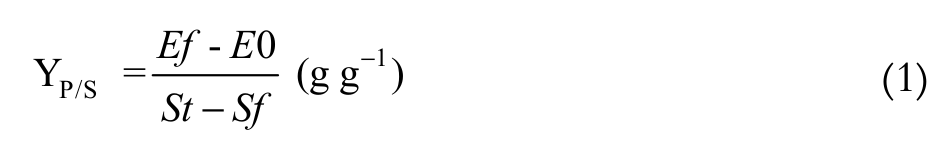 (1)
(1)
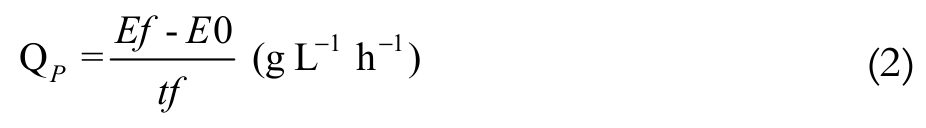 (2)
(2)
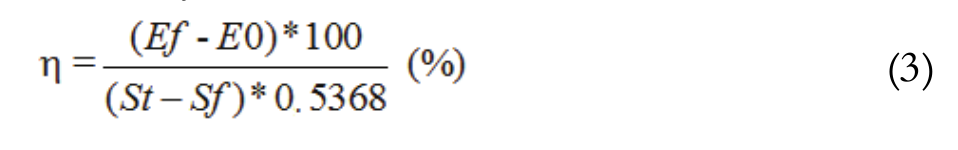 (3)
(3)The parameters used to evaluate the fermentation performance in the biomass production were: Productivity and Substrate to biomass yield conversion (Equation 4 and 5, respectively).
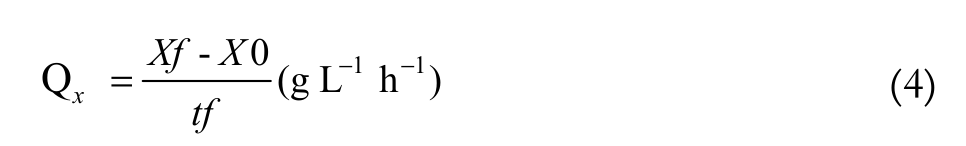 (4)
(4)
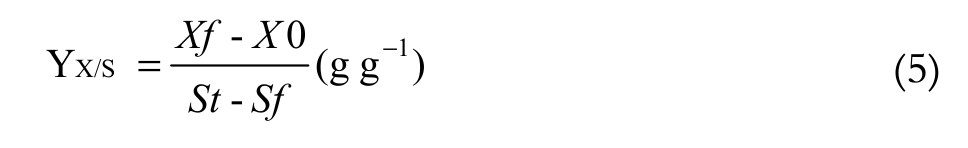 (5)
(5)where:
Ef = final product concentration (g L-1);
E0 = initial product concentration (g L-1);
tf = total fermentation time (hours);
St= total substrate concentration (g L-1);
Sf = final substrate concentration (g L-1); 0.5368 is the mass conversion.
The stoichiometry is 1 lactose to 4 ethanol;
Xf = final biomass concentration (g L-1);
X0 = initial biomass concentration (g L-1).
The consumption of lactose (Equation 6), protein (Equation 7) and the average speed of substrate consumption (Equation 8) were determined as:
 (6)
(6)
 (7)
(7)
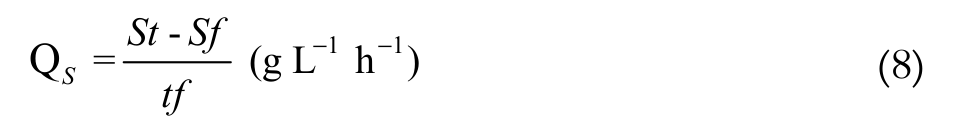 (8)
(8)where:
Sf, Pf = final substrate and protein concentrations (g L-1);
St, Pt = total substrate and protein concentrations (g L-1);
tf = total fermentation time (hours).
The chemical oxygen demand was calculated as Equation 9:
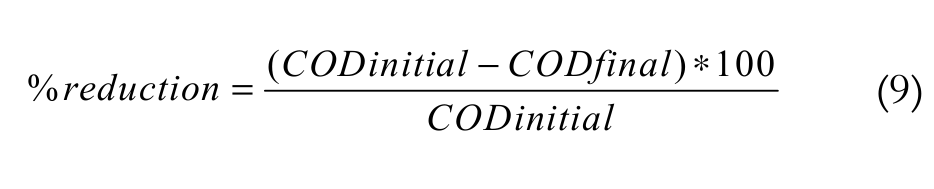 (9)
(9)Results and discussion
Table 1 shows the characteristics of the culture media M1, M2, M3 and M4 with respect to pH and the carbohydrate, protein and COD concentrations before fermentation by Kluyveromyces marxianus CCT 4086. The pH of the media was adjusted to 4.5 after sterilization, because this is the optimum value for the yeast development used in this study.
Lactose and protein consumption
With respect to lactose consumption (Figure 1A), after 48 hours of fermentation, culture medium M1 had a lactose concentration of 2.18 g L-1, followed by M2 2.24, M3 2.06 and M4 1.20 g L-1, representing total lactose consumptions of 96.20, 95.10, 93.66 and 93.60% respectively. There was no significant difference in the final substrate consumption (p > 0.05) amongst the media tested in this experiment. Similar works carried out by Zoppellari and Bardi (2013) and by Koushki, Jafari, and Azizi (2012) obtained final lactose consumptions in the most concentrated media of 94.57 and 96.20% respectively, also using the yeast of the genus Kluyveromyces in whey. These authors also carried out anaerobic fermentations with initial lactose concentrations of 44.21 and 49 g L-1 at temperatures of 37 and 30º C, respectively.
Regarding the highest average speed of substrate consumption (PS) obtained, the M1, M2 and M3 media showed the highest results of 0.74; 0.65 and 0.60 g L-1 h-1 (p > 0.05), respectively, in 12 hours of fermentation, whereas M4 only reached 0.30 g L-1 h-1 in 10 hours of fermentation. This showed that the higher the substrate concentration in the medium, the higher the consumption rate by the yeast.
The protein contents were determined (Figure 1B), and the M1 and M2 media (higher initial protein concentrations, see Table 1) showed higher protein consumption (76.00 and 68.75% respectively), with no significant difference between them (p < 0. 05). The M3 and M4 media showed consumptions of 73.68 and 66.00% protein, respectively (p < 0.05).
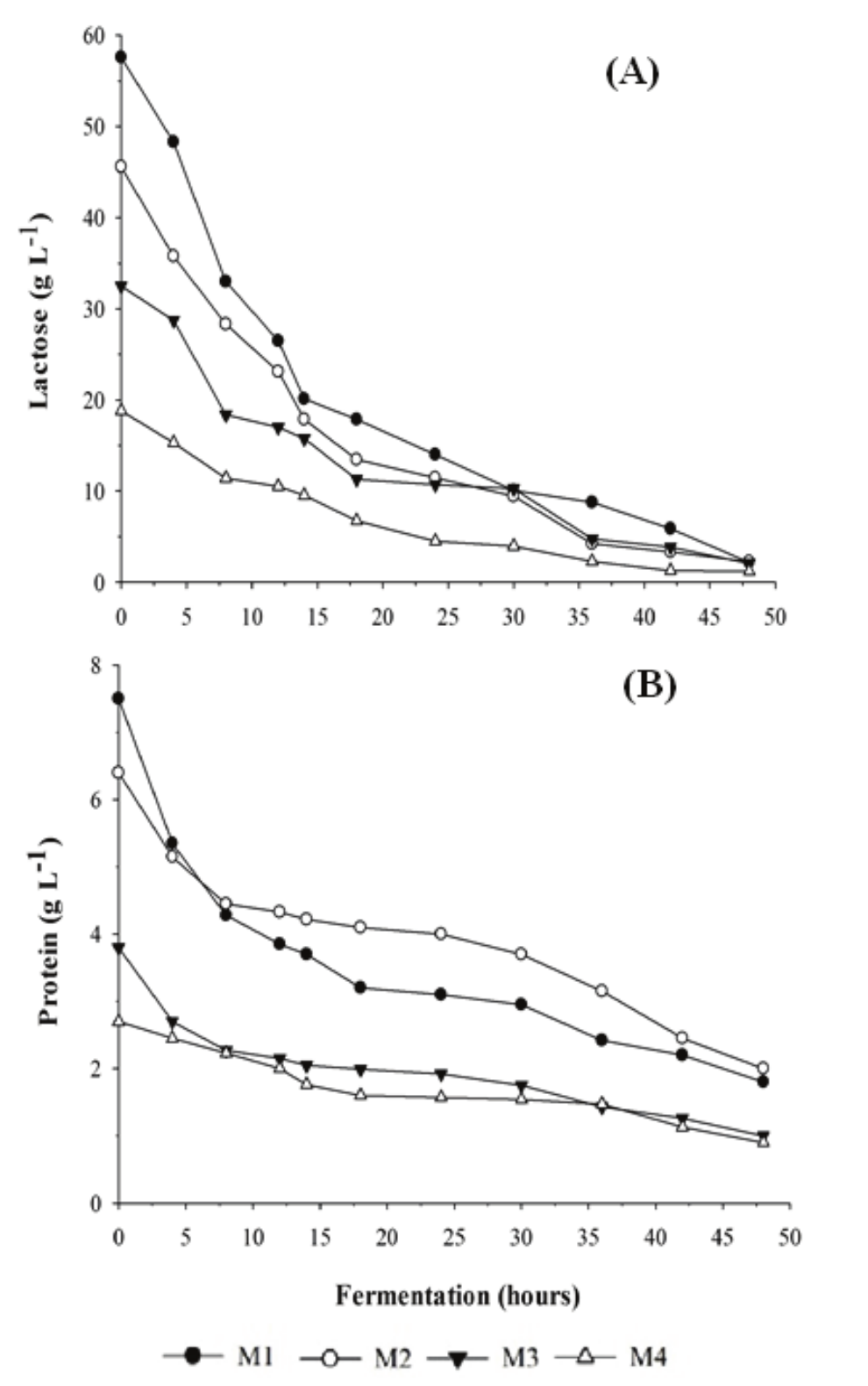
Figure 1.
Consumption of lactose (A) and protein (B) by K. marxianus in different whey concentrations after 48 hours of fermentation.
Biomass and ethanol production
Figure 2A and Table 2 show that after 48 hours of fermentation the media showing higher ethanol production (P) were those with higher initial substrate concentration. The M1 medium showed an ethanol production of 16.90 g L-1, M2 medium of 13.28 g L-1, followed by M3 at 9.17 and M4 at 5.10 g L-1, with significant differences (p < 0.05) amongst the four media tested.
Maximal production of ethanol was achieved after 20 hours, as shown in Figure 2A. It can be seen that high lactose substrate loads were required for the production of ethanol, since part of the substrate was used for cell growth (Figure 2B) and other vital activities of the microorganism.
The average lactose consumption speeds showed that the media with higher substrate concentrations (M1 and M2) provided increased consumption, so as to sustain a sufficiently high glycolytic flux to produce energy, as confirmed by the increased ethanol production in these media (Figure 2A).
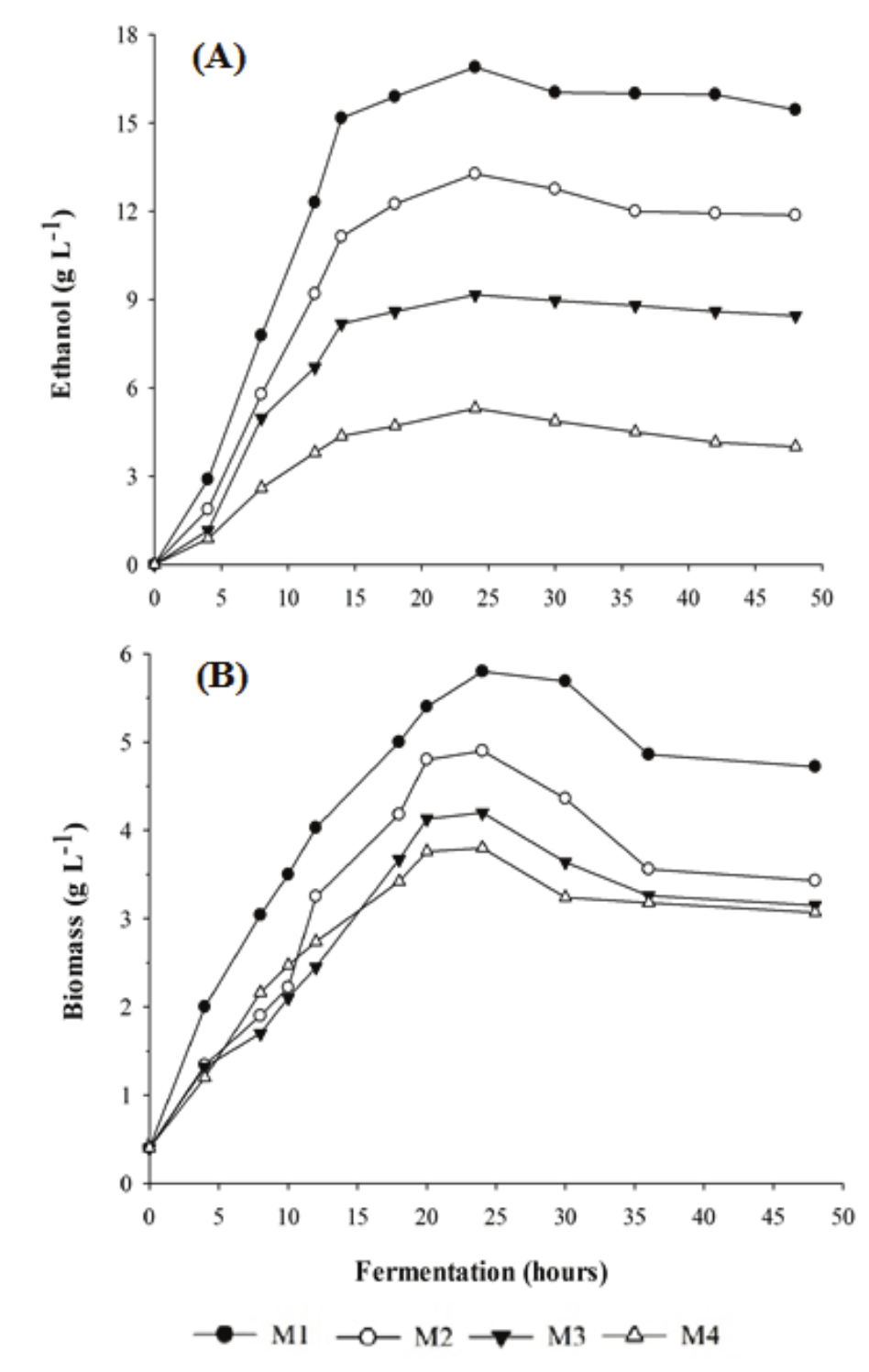
Figure 2.
Ethanol production (A) and biomass production (B) by the yeast K. marxianus in different whey concentrations after 48 hours fermentation.
The highest substrate to ethanol conversions (YP/S), productivities (QP) and ethanol yields occurred in the four media tested (M1, M2, M3 and M4) between 10 and 12 hours of fermentation, as shown in Figure 2A and Table 2 and 3.
The highest ethanol productivity (QP) occurred in M1 medium (1.26 g L-1 hour-1), followed by M2 (0.93 g L-1 hour-1), M3 (0.68 g L-1 hour-1) and M4 (0.38 g L-1 hour-1). There was a significant difference between the media tested (p < 0.05).
The highest substrate to ethanol conversion (YP/S) occurred in M3 medium (0.48 g g-1), followed by M4 (0.47 g g-1) (p > 0.05); the M1 and M2 media showed conversions of 0.40 g g-1 (p > 0.05).
For the ethanol yield (), the medium presenting the most relevant result was also M3, with 82.30% in 12 hours of fermentation and an initial substrate concentration of 32.50 g L-1. This means a value 43.57% lower than that of the medium with the highest substrate concentration (M1) (Table 1), followed by M4 medium with (η) 80.30%, also in 12 hours fermentation. There was no significant difference between M3 and M4 (p > 0.05) and it should be noted that these media were also those showing the highest ethanol substrate conversions (YP/S).
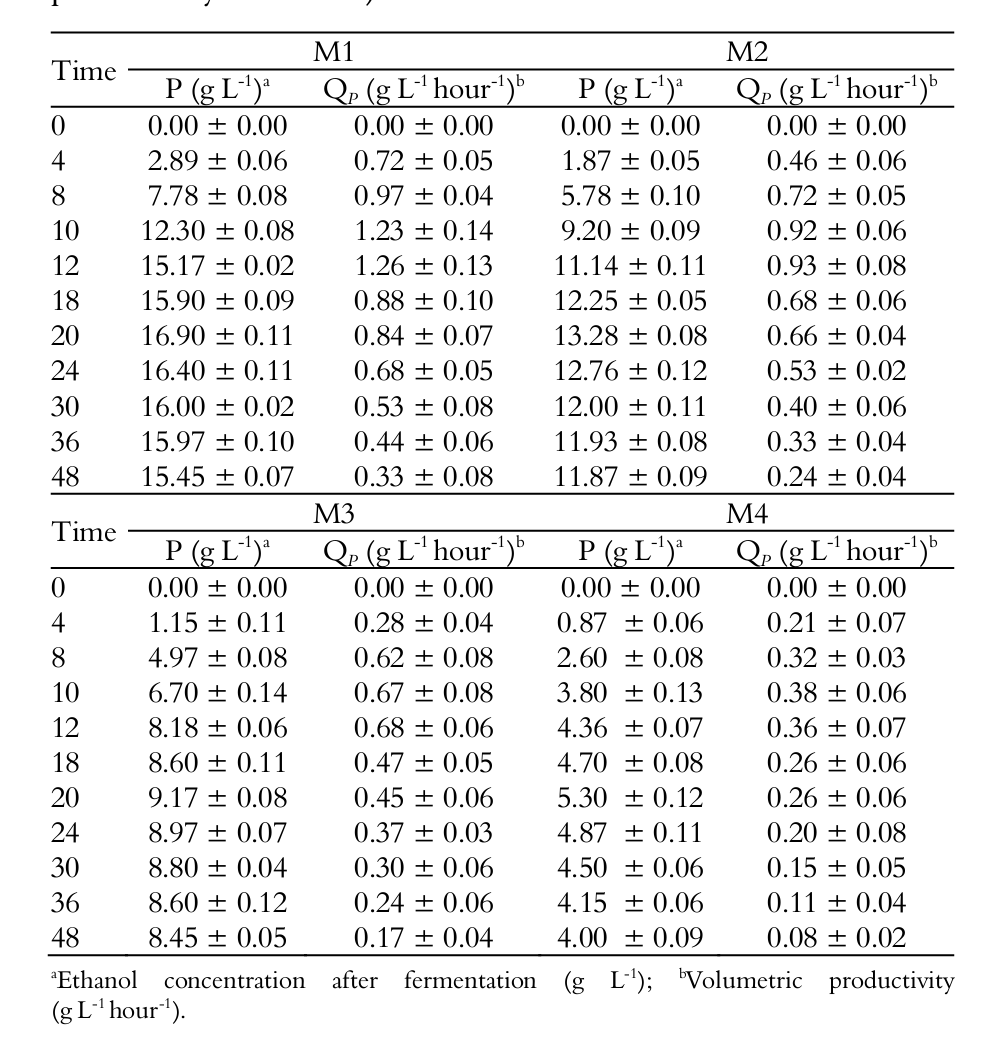
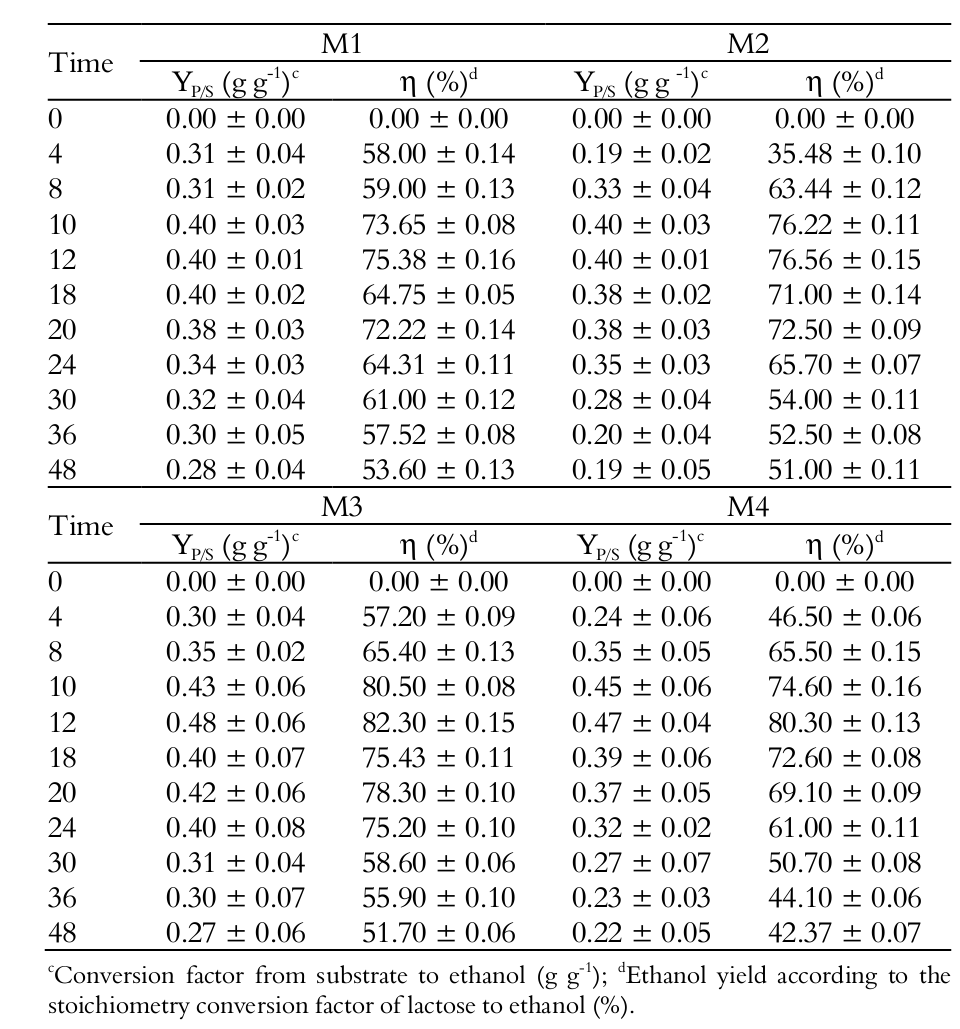
The M1 and M2 media showed a maximum ethanol yield (η) of 75.38 and 76.56% respectively after 12 hours of fermentation, with no significant difference between them (p > 0.05). These results were also observed by Gonzáles and Fernández (2006), who obtained the highest ethanol efficiency in media with lower substrate concentrations (45 g L-1 lactose). The same authors obtained a fermentation process efficiency of 89% in relation to theoretical transformation coefficient of lactose into ethanol, using a fermentation temperature of 30ºC for 24 hours.
The fermentation time for ethanol production varies with the substrate concentration: at 20 hours of fermentation (Figure 2A), the highest ethanol production (P) occurred in the media with higher initial substrate concentrations (M1 and M2) and, for 10 to 12 hours of fermentation, the highest ethanol production was obtained from the media with lower initial substrate concentrations (M3 and M4).
Other authors also observed that the higher the initial substrate concentration, the lower the final ethanol yield (Ozmihci & Kargi, 2007, Koushki et al., 2012, Zoppellari & Bardi, 2013). Sansonetti, Curcio, Calabrò, and Iorio (2010), for instance, obtained an ethanol yield of 79% after 18 hours of fermentation at 34ºC with a lactose concentration of 49.27 g L-1. The same authors, when using a lactose concentration of 47.06 g L-1, obtained an ethanol yield of 84% at the same fermentation time. In the present study, similar results were obtained for ethanol yield as compared to these authors, presenting yields of 82.30 and 80.30% (M3 and M4, respectively), but with lower initial lactose concentrations (32.50 and 18.88 g L-1 respectively) and lower fermentation time (12 hours).
This relationship can be explained from the fact that higher concentrations of sugar and other dissolved solids can lead to an increased osmotic pressure in the fermentation medium, which can result in a considerable loss of sugar transport activity in the yeast cells under anaerobic conditions, resulting in lower yields (Ozmihci & Kargi, 2008, Koushki et al., 2012).
As in case of ethanol production, higher cell biomass production occurred in the media with higher initial substrate concentrations after 24 hours of fermentation (Table 4 and Figure 2B), i.e. M1 showed a biomass of 5.80; M2 4.90; M3 4.20 and M4 3.80 g L-1, with no significant difference for M3 and M4 (p > 0.05). Cell production in M1 was 34.5% higher than in M4 (p < 0.05).
This difference can be explained by the lower substrate concentration in M4, that is, lower carbon and nitrogen concentrations for biomass conversion. Gonzáles and Fernández (2006), testing different lactose concentrations in whey fermentation by Kluyveromyces fragilis, also achieved the highest cell production (5.96 g L-1) in the medium with the highest substrate concentration. Sansonetti et al. (2010) also obtained the highest results (7.36 g L-1) in more concentrated media.
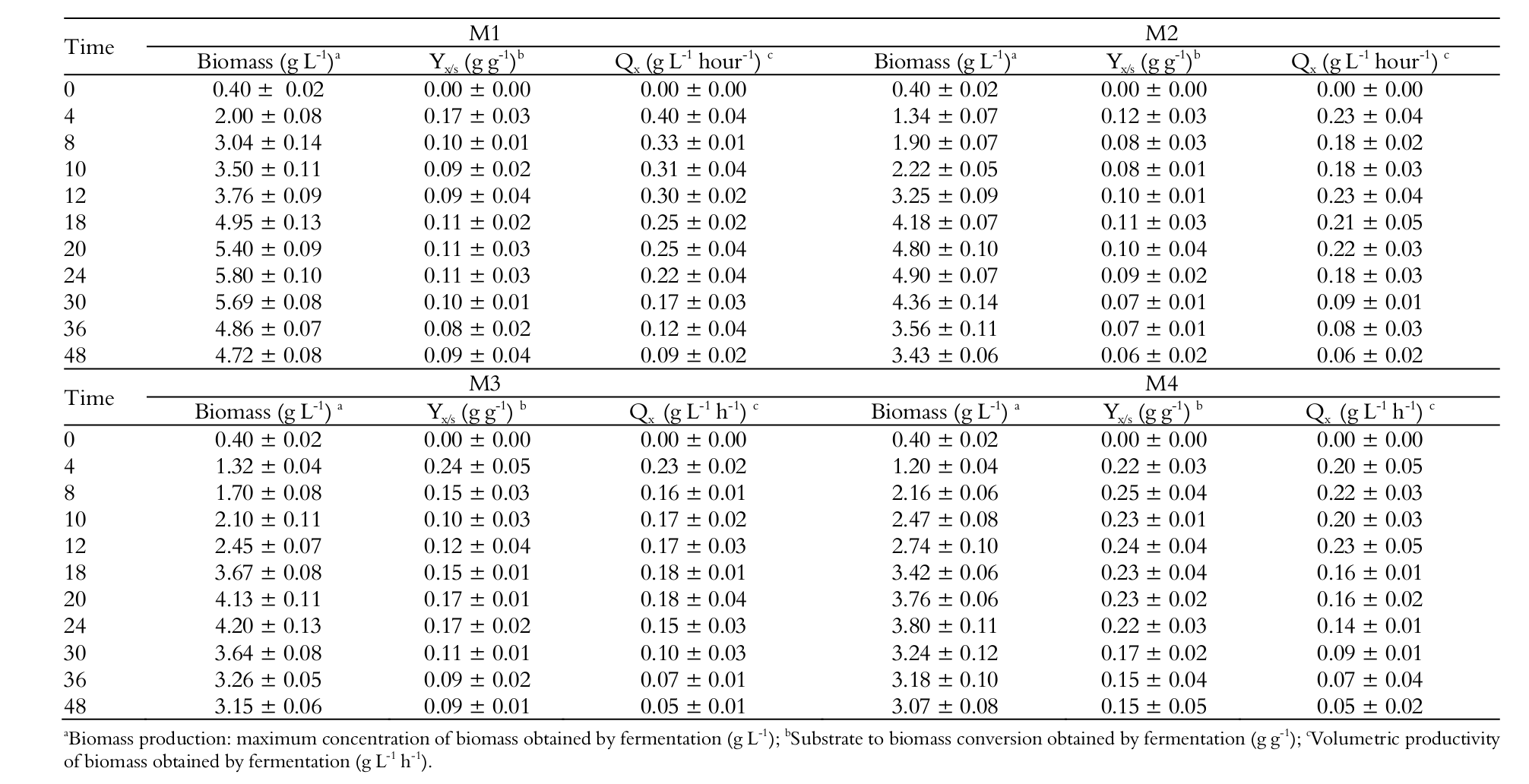
The highest biomass productivity (Qx) also occurred in M1 (0.40 g L-1 hour-1), followed by M2 and M3 (0.23 g L-1 hour-1) and M4 (0.22 g L-1 hour-1), with no significant difference between M2, M3 and M4 (p > 0.05) (Table 4). Except for M1 medium, similar results were found by Zumbado-Rivera, Esquivel-Rodrigues, and Wong-Gonzáles (2006), who obtained a biomass productivity of 0.22 g L-1 hour-1 by Kluyveromyces marxianus.
The higher biomass yields (YX/S) were obtained in M4 with 0.25 and in M3 with 0.24 g g-1 (p < 0.05) when compared to the other media studied. M1 showed a yield of 0.17 and M2 0.12 g g-1 (p > 0.05) (Table 4), with a significant difference between them and between the media with lower substrate concentrations (p < 0.05). This indicates that lower substrate concentrations in the media result in better substrate to cell mass conversion efficiency. All the media gave their highest yields (YX/S) and biomass productivities (Qx) after 4 hours of fermentation, except for M4, which was after 8 hours of fermentation (Table 4).
M1 was the medium with the highest substrate consumption and highest average speed for substrate consumption, also obtaining the highest cell mass production and substrate to cell conversion. M1 also showed the highest protein consumption (Figure 1B), reinforcing the idea that it was used in cell growth.
Another aspect of the fermentation process is the capacity of the yeast to support the toxic effects caused by the ethanol produced during fermentation (Figure 2A). Even when reaching its maximum ethanol production after 20 hours of fermentation, this yeast strain was still able to grow for a while, reaching its biomass production peak after 24 hours of fermentation (Figure 2B).
The results showed that with low substrate concentrations the fermentation process did not reflect in process efficiency with respect to the ethanol yield, and production of cell biomass, considering that these treatments avoided being inhibited by the higher substrate concentrations, which can lead to osmotic stress of the yeast, as discussed above. On the other hand, this suggests the possibility of using separation techniques to eliminate inhibition by the metabolic product, and maintain high ethanol yields and cell productivity, as also suggested by Bai, Anderson, and Moo-Young (2008).
Chemical oxygen demand (COD)
The chemical oxygen demand (COD) was determined simultaneously with the carbohydrate analysis during the course of the experiments.
The M4 medium showed the highest efficiency in reducing the organic matter (82.28%), followed by M3 (70.57%) (Table 5 and Figure 3). These media presented lower initial substrate concentrations (Table 1) and consequently started from lower COD contents, 22.680 and 38.880 mg L-1, respectively, with significant differences between them (p > 0.05). Similar work carried out by Schultz et al. (2006) showed an efficiency of 83.00% in COD reduction after whey fermentation.

Reduction of COD by K. marxianus after 48 hours fermentation at different substrate concentrations.
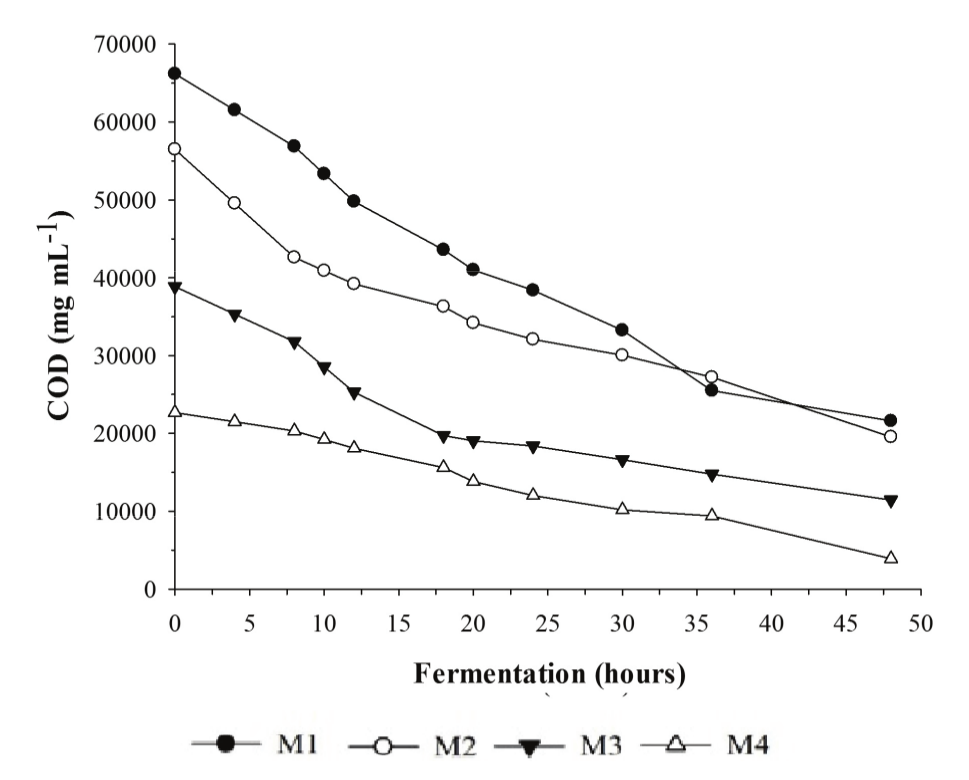
Figure 3.
Reduction of the organic matter content by K. marxianus after 48 hours of fermentation in different whey concentrations.
According to the results, even after the microorganism reached maximum growth (Figure 2B), the COD continued decreasing until reaching a plateau after 48 hours of fermentation. During this period, the cells remained in the stationary phase. The cells possibly consumed the carbon source dissolved in the medium (mainly lactose) during the growth phase, resulting in the formation of organic acids. After the start of the stationary phase, it is presumed that the microorganisms used these acids to maintain cell metabolism, thus contributing to the reduction in COD (Reinbold & Takemoto, 1998, Zafar & Owais, 2006).
Conclusion
The results obtained for whey fermentation by K. marxianus CCT 4086 indicated that the ethanol production (g L-1), volumetric ethanol productivity (g L-1 hour-1), biomass production (g L-1) and biomass productivity (g L-1 hour-1) were favored by the medium with higher nutrient concentrations (M1).
This study provided evidence of the potential use of whey in bioprocessing, and that the initial nutrient concentrations present in the medium were an important factor for process efficiency.
The generation of bioethanol from whey is an effective alternative for obtaining a renewable and environmentally friendly biofuel, adding value to a waste that has a high polluter potential and limited possible destinations.
Acknowledgements
The authors are grateful to Capes - Coordenação de Aperfeiçoamento de Pessoal de Nível Superior for the financial support of this research.
References
American Public Health Association [APHA]. (1995). Standard methods for the examination of water and wastewater (19th ed.). Washington, DC: Apha.
Association of Official Analytical Chemists [AOAC]. (1995). Official methods of analysis (15th ed). Washington, DC: AOAC.
Bai, F. W., Anderson, W. A., & Moo-Young, M. (2008). Ethanol fermentation technologies from sugar and starch feedstocks. Biotechnology Advances, 26(2), 89-105.
Banaszewska, A., Cruijssen, F., Claassen, G. D. H., & Van der Vorst, J. G. A. J. (2014). Effect and key factors of byproducts valorization: The case of dairy industry. Journal Dairy Science, 97(4), 1893-1908.
Carvalho, F., Prazeres, A. R., & Rivas, J. (2013). Cheese whey wastewater: Characterization and treatment. Science Total Environment, 445-446(15), 385-396.
Fonseca, G. G., Heinzle, E., Wittmann, C., & Gombert, A. K. (2008). The yeast Kluyveromyces marxianus and its biotechnological potential. Applied Microbiology, 79(3), 339-354.
González, C. P., & Fernández, M. D. (2006). Effect of the initial concentration of whey on the alcoholic fermentation by Kluyveromyces fragilis. Revista de la Sociedad Venezolana de Microbiología, 26(1), 35-41.
Grubb, C. F., & Mawson, A. J. (1993). Effects of elevated solute concentrations on the fermentation of lactose by Kluyveromyces marxianus Y-113. Biotechnology Letters, 15(6), 621-626.
Guimarães, P. M. R., Teixeira, J. A., & Domingues, L. (2010). Fermentation of lactose to bio-ethanol by yeasts as part of integrated solutions for the valorisation of cheese whey. Biotechnology Advances, 28(3), 375-384.
Gupta, R. B., & Demirbas, A. (2010). Gasoline, diesel, and ethanol biofuels from grasses and plants. New York City, NY: Cambridge University Press.
Harish, B. S., Ramaiah, M. J., & Uppuluri, K. B. (2015). Bioengineering strategies on catalysis for the effective production of renewable and sustainable energy. Renewable and Sustainable Energy Reviews, 51(1), 533-547.
Hartree, E. E. (1972). Determination of protein: A modification of the Lowry Method that gives a linear photometric response. Analytical Biochemistry, 48(2), 422-427.
Koushki, M., Jafari, M., & Azizi, M. (2012). Comparison of ethanol production from cheese whey permeate by two yeast strains. Journal Food Science Technology, 49(5), 14-619.
Miller, G. L. (1959). Use of dinitrosalycilic acid reagent for determination of reducing sugar. Analytical Biochemistry, 31(3), 426-428.
Murari, C. S., Moraes, D. C., Bueno, G. F., & Del Bianchi, V. L. (2013). Evaluation of the reduction in pollution of dairy products from whey fermentation in ethanol by yeast Kluyveromyces marxianus 229. Revista do Instituto de Laticínios Cândido Tostes, 68(393), 42-50.
Ozmihci, S., & Kargi, F. (2007). Kinetics of batch ethanol fermentation of cheese-whey powder (CWP) solution as function of substrate and yeast concentrations. Bioresource Technology, 98(16), 2978-2984.
Ozmihci, S., & Kargi, F. (2008). Ethanol production from cheese whey powder solution in a packed column bioreactor at different hydraulic residence times. Biochemical Engineering Journal, 42(2), 80-185.
Reinbold, R. S., & Takemoto, J. (1998). Use of Swiss cheese whey permeate by Kluyveromyces fragilis and mixed culture of Rhodopseudomonas sphaeroides and Bacillus megaterium. Journal Dairy Science, 71(7), 1799-1802.
Rocha, J., & Buckeridge, M. (2009). Biofuelling the future. New Science, 202(2709), 26.
Sansonetti, S., Curcio, S., Calabrò, V., & Iorio, G. (2010). Optimization of ricotta cheese whey (RCW) fermentation by response surface methodology. Bioresource Technology, 101(23), 9156-9162.
Schultz, N., Chang, L., Hauck, M. R., & Syldatk, C. (2006). Microbial production of single-cell protein from deproteinized whey concentrates. Applied Microbiology and Biotechnology, 69(5), 515-520.
Silveira, W. B., Passos, F. J. V., Mantovani, H. C., & Passos, F. M. L. (2005). Ethanol production from cheese whey permeate by Kluyveromyces marxianus UFV-3: A flux analysis of oxido-reductive metabolism as a function of lactose concentration and oxygen levels. Enzyme and Microbial Technology, 36(7), 930-936.
Todhanakasem, T., Sangsutthiseree, A., Areerat, K., Young, G. M., & Thanonkeo, P. (2014). Biofilm production by Zymomonas mobilis enhances ethanol production and tolerance to toxic inhibitors from rice bran hydrolysate. New Biotechnology, 31(5), 451-459.
Van Ooyen, A. J., Dekker, P., Huang, M., Olsthoorn, M. M., Jacobs, D. I., & Colussi, P. A. (2006). Heterologous protein production in the yeast Kluyveromyces lactis. FEMS Yeast Research, 6(3), 381-392.
Vincenzi, A., Maciel, M. J., Burlani, E. L., Oliveira, E. C., Volpato, G., Lehn, D., & Souza, C. F. V. (2014). Ethanol bio-production from ricotta cheese whey by several strains of the yeasts Kluyveromyces. American Journal of Food Technology, 9(6), 281-291.
Zafar, S., & Owais, M. (2006). Ethanol production from crude whey by Kluyveromyces marxianus. Biochemical Engineering Journal, 27(3), 295-298.
Zoppellari, F., & Bardi, L. (2013). Production of bioethanol from effluents of the dairy industry by Kluyveromyces marxianus. New Biotechnology, 30(6), 607-613.
Zumbado-Rivera, W., Esquivel-Rodrigues, P., & Wong-Gonzáles, E. (2006). Selección de una levadura para la producción de biomasa: crecimiento en suero de queso. Agronomía Mesoamericana, 17(2), 151-160.
Author notes
samaramurari@gmail.com