Engenharia de Pesca
Integration between bioflocs and periphyton in Nile tilapia culture tanks
Integração entre bioflocos e perifíton em tanques de cultivo da tilápia do Nilo
Integration between bioflocs and periphyton in Nile tilapia culture tanks
Acta Scientiarum. Technology, vol. 39, 05, pp. 601-607, 2017
Universidade Estadual de Maringá

This work is licensed under Creative Commons Attribution 4.0 International.
Received: 14 December 2015
Accepted: 29 November 2016
Abstract: The present study aimed to assess the possible beneficial effects of the integration between bioflocs and periphyton to the Nile tilapia’s water quality and growth performance. There were four treatments with five replicates each: (1) Control: green waters, (2) Periphyton: substrate-based system, (3) BFT: bioflocs technology for aquaculture, and (4) Biophyton: integration between bioflocs and periphyton. Fish (1.63 ± 0.07 g) were reared for 10 weeks in twenty 250 L outdoor tanks. Two polyethylene boards were vertically set out in the Periphyton and Biophyton tanks as underwater substrates. The C: N ratios of water in the BFT and Biophyton tanks were adjusted to 15:1 with dry molasses applications. The concentrations of total ammonia nitrogen were higher in the Control and Periphyton tanks than in the BFT and Biophyton ones. On the other hand, the concentrations of reactive phosphorus were higher in the BFT and Biophyton tanks than in the Control and Periphyton ones. The fish final body weight, specific growth rate and fish yield have not differed between the tanks. The integration between bioflocs and periphyton has not brought clear benefits to tilapia culture on water quality and growth performance.
Keywords: aquaculture, Oreochromis niloticus, water quality.
Resumo: O presente trabalho teve por objetivo determinar os possíveis efeitos benéficos da integração entre bioflocos e perifíton na qualidade de água e no desempenho zootécnico da tilápia do Nilo. Trabalhou-se com quatro tratamentos com cinco repetições cada: (1) Controle: águas verdes, (2) Perifíton: sistema de cultivo baseado em substrato, (3) BFT: tecnologia bioflocos para aquicultura e (4) Biofíton: bioflocos + perifíton. Os peixes (1,63 ± 0,07 g) foram mantidos por dez semanas, em 20 tanques externos de 250 L. Duas pranchas de polietileno foram posicionadas verticalmente, nos tanques Perifíton e Biofíton, como substratos submersos. A relação C: N da água dos tanques BFT e Biofíton foi ajustada para 15:1 pela aplicação de melaço à água. As concentrações de nitrogênio amoniacal total foram maiores nos tanques Controle e Perifíton do que nos tanques BFT e Biofíton. Por outro lado, as concentrações de fósforo reativo foram maiores nos tanques BFT e Biofíton que nos tanques Controle e Perifíton. O peso corporal final, a taxa de crescimento específico e a produtividade de peixe não diferiram entre os tanques. A integração entre bioflocos e perifíton não apresentou claros benefícios para a criação da tilápia, em relação à qualidade da água e ao desempenho produtivo.
Palavras-chave: aquicultura, Oreochromis niloticus, qualidade de água.
Introduction
The use of inadequate feeding strategies, such as the excessive delivery of artificial diet, is not a rare event in intensive fish culture. As a consequence, a great amount of organic residues accumulates in the productive units when there is excessive feeding (Cyrino, Bicudo, Sado, Borghesi, & Dairiki, 2010). Guo, Li, Xie, and Ni (2009) have estimated that not more than 30% of all phosphorus and nitrogen present in the artificial diets are retrieved in the harvested fish biomass. At the end of the culture cycle, the release of the tank effluents into the receiving water bodies can cause eutrophication with harmful consequences to the environment (Green, Brandon, & Hardy, 2002). One sustainable solution to attain a greater productivity in aquaculture is to increase the use of the natural foods available in the system, such as bioflocs and periphyton. Beyond their nutritional value, bioflocs and periphyton are also capable to remove nitrogen, phosphorus and toxic compounds from the water (Azim & Little, 2008).
The bioflocs technology (BFT) smartly change the microbial community present in the rearing tanks by adjusting upward the C: N ratio of water (Ray, Dillon, & Lotz, 2011). The BFT system allows the sustainable culture of aquatic animals in high stocking densities. The immobilization of ammonia carried out by the heterotrophic bacteria happens faster than the removal done by the nitrifying bacteria (Crab, Defoirdt, Bossier, & Verstraete, 2012). The heterotrophic bacteria’s growth rate can be up to ten times higher than that for the nitrifying bacteria (Schveitzer et al., 2013a).
The substrate-based aquaculture systems aim to develop plenty of algae and bacteria biofilms (periphyton) on any submersible surfaces present in the rearing water (Schrader, Green, & Perschbacher, 2011). Periphyton can be efficiently used by, for example, tilapias as a food source (Asaduzzaman et al., 2009). Moreover, nitrifying bacteria can act as biological filters, by removing ammonia and nitrite from the water, and colonize those substrates (Schveitzer et al., 2013b).
Both systems, BFT and substrate-based (periphyton), however, have some setbacks. In the BFT system, despite its lower levels of ammonia (Luo, Avnimelech, Pan, & Tan, 2013), there is an accumulation of nitrite in the water (Luo et al., 2014). In the substrate-based system, the removal rates of ammonia are generally low when compared to those found in the BFT system (Schveitzer et al., 2013b). The integration between bioflocs and periphyton (biophyton system) in a same rearing unit might combine the positive effects of those both systems in a synergic process. Hence, the biophyton system might remove more ammonia and nitrite from the water than what would be possible by the isolated use of those systems. To improve our knowledge on this subject, the present study had the objective to assess the beneficial effects of the integration between periphyton and bioflocs to the water quality and Nile tilapia growth performance.
Material and methods
One thousand masculinized Nile tilapia, Oreochromis niloticus, juveniles (body weight = 1.23 ± 0.07 g) were obtained from a regional fish farmer and hauled up to the laboratory facilities. Initially, the fish were maintained for four days in one circular 1,000 L tank for acclimatization to the lab’s conditions. In this phase, fish were fed on four times daily at 8 and 11 a.m.; 2 and 5 p.m. with a commercial diet for omnivorous tropical fish, which contained 49.4% of crude protein, and it was delivered at 10% of the live weight daily.
At the onset of the experiment, one hundred and eighty Nile tilapia juveniles with a body weight of 1.63 ± 0.07 g were set out in twenty 250 L polyethylene outdoor tanks at 9 fish per tank. Over 10 weeks, fish were fed with three commercial diets delivered daily at 8 and 11 a.m.; 2 and 5 p.m. Initially, the diet was allowed daily at 8.9% of the fish live weight. The feeding rates were then adjusted downward periodically according to the fish weightings. The final feeding rate adopted was 3.1% of the fish live weight. Three artificial diets were used to feed the fish over the experiment. The first one was a powdered diet with 49.4% crude protein; the second one was an extruded diet with 0.8-1.2 mm particle size and 43.4% crude protein; the third diet was an extruded one with 2-3 mm particle size and 35.9% crude protein.
The following treatments were tested with five replications each:
(1) Control_ conventional fish culture system in green waters, without underwater substrates for periphyton or adjustment of the C: N ratio of water for bioflocs;
(2) Periphyton_ substrate-based fish culture system with underwater substrates for periphyton, but no adjustment of the C: N ratio of water for bioflocs;
(3) Bioflocs_ BFT fish culture system, without underwater substrates for periphyton, but with adjustment of the C: N ratio of water for bioflocs;
(4) Biophyton_ bioflocs + periphyton fish culture system, with underwater substrates for periphyton and adjustment of the C: N ratio of water for bioflocs.
Two transversally interconnected polyethylene boards were vertically set out in the water column of the Periphyton and Biophyton tanks as underwater substrates. Those boards had a useful area of 0.90 m2, which corresponded to 135% of the total surface water area. In the Control and Periphyton tanks, nocturnal aeration was provided from 6 p.m. to 6 a.m., starting at the third experimental week. A 2.5 HP radial air compressor (blower) was used with that purpose. In the Bioflocs and Biophyton tanks, non-stop aeration was provided throughout the experimental period.
The experimental design adopted was a 2 x 2 factorial, where the treatments were arranged in randomized blocks. The blocking design was necessary to avoid the unforeseen influence of non-controlled environmental factors on the response variables, mainly insolation and winds. The experimental factors under evaluation were bioflocs and periphyton, in two levels each one, absence and presence. That way, the four treatments were formed, i.e., Control, Periphyton, Bioflocs and Biophyton. The amount of dry molasses applied daily into the water to raise the C: N ratio of water to 15:1 followed the guidelines of Avnimelech (1999). For that aim, it was used the information on the feeding management, i.e., the percentage of crude protein in the diet and daily allowances of diet. No water exchange was performed over the entire experimental period in any of the culture tanks, just the replenishing to maintain the initial water level.
The determinations of water quality carried out were the followings:
(1) Daily: pH (pH meter mPA210 - MS Tecnopon®), specific conductance (conductivity meter CD-850) and temperature, at 8 and 3 p.m.;
(2) Weekly: dissolved oxygen (8 a.m.; DO2; Winkler method with azide modification) and total ammonia nitrogen (TAN; indophenol method);
(3) Fortnightly: nitrite (sulfanilamide method), nitrate (method of reducing Cd column), reactive phosphorus (molybdenum blue method), total alkalinity (titration with H2SO4 standard solution), total hardness (titration with EDTA standard solution), organic matter (method of the consumed KMnO4), primary phytoplankton productivity (light and dark bottles method) and total dissolved sulfides (titration with Na2S2O3 standard solution).
The concentrations of NH3 in the water were calculated by applying the TAN, pH and water temperature results into the Emerson’s Formula (El-Shafai, El-Gohary, Nasr, Van der Steen, & Gijzen, 2004). The H2S concentrations were calculated out of the total dissolved sulfide results, according to Boyd (2000). All water quality determinations were performed following the American Public Health Association (Apha, 1999)’s standard methods. A diel monitoring of dissolved oxygen, pH and temperature of water was carried out in the ninth experimental week. For that, water samples were taken from the tanks at 8 a.m., noon, 4 p.m., 8 p.m., midnight, 4 a.m. and 8 a.m., in the following day.
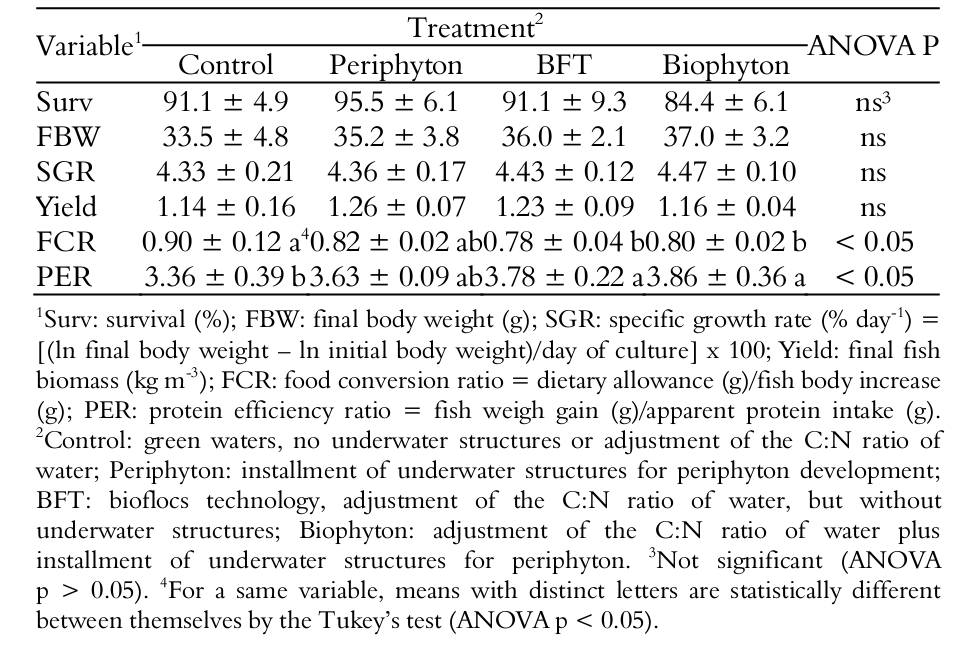
The growth performance variables analyzed were the followings: fish’s final body weight (g), survival (%), weekly body gain, food conversion ratio (FCR = feed consumed/body weight gain), specific growth rate (SGR = [Ln (final weight) - Ln (weight initial)]/days of culture) x 100), fish yield and protein efficiency ratio (PER = weight gain/protein consumed).
Initially, the variables of water quality and growth performance were evaluated by the two-way ANOVA for randomized blocks experiments. Since there were no significant interactions between the bioflocs and the periphyton for any of the variables, the water quality and growth performance results were analyzed by the one-way ANOVA. When a significant difference was detected between the treatments, the means were compared two by two with the Tukey’s test. The assumptions of normal distribution (Shapiro-Wilk’s test) and homogeneity of variance (Levene’s test) were checked before the analyses. The SPSS v.15.0 and Windows Excel 2010 software were used in the statistical analyses. The minimum level of significance was set out at p < 0.05.
Results and discussion
Water quality
Due to the non-stop aeration in the BFT and Biophyton tanks, their concentrations of dissolved oxygen in water (DO2) were greater than those found in the Control and Periphyton tanks (p < 0.05; Table 1). On the other hand, the underwater structures to periphyton have not significantly affected the concentrations of DO2 in water. The total alkalinity of water was higher (p < 0.05) in the non-bioflocs tanks (Control and Periphyton) than in the bioflocs ones (BFT and Biophyton). The input of CO2 in bioflocs tanks is generally high due to their elevated rates of respiration and decomposition of the organic matter (Azim, Little, & Bron, 2008). Since there is a minimal or even no water renewal in bioflocs systems, their water alkalinity tends to decrease over time (Furtado, Poersh, & Wasielesky, 2011). The total hardness of water was greater in the BFT and Biophyton tanks than in the Control and Periphyton ones (p < 0.05; Table 1). There can be a significant input of calcium in bioflocs culture media through the applications of molasses in the water because dry molasses is a calcium rich source (Rostagno et al., 2011).
The pH of water was higher in the non-bioflocs tanks (Control and Periphyton) than in the bioflocs tanks (BFT and Biophyton; Table 1). The probable greater amount of CO2 released in the bioflocs tanks has promoted water acidification in those tanks (Furtado et al., 2011). Despite that, the pH of water has remained inside the proper range for fish growth in the BFT and Biophyton tanks (pH = 6-9). The specific conductance of water was greater (p < 0.05) in the bioflocs tanks than in the non-bioflocs ones, indicating the higher concentration of organic matter in the former (Boyd & Tucker, 1998).
The concentrations of TAN and NH3 were higher in the Control and Periphyton tanks than in the BFT and Biophyton ones (p < 0.05; Table 1). These results confirm those observed by Avnimelech and Kochba (2009), and Luo et al. (2013), demonstrating that a higher ammonia removal rate can be obtained by the adjustment of the C: N ratio of water. The concentrations of TAN and NH3 in water have not differed significantly between the BFT and Biophyton tanks (p > 0.05). Therefore, the underwater structures added in the BFT tanks for the periphyton growth were not capable to increase the rate of ammonia removal from the water.
The concentrations of nitrite in the water have not differed significantly between the treatments (Table 1). This result is in agreement with the observations made by Liu, Hu, Dai, and Avnimelech (2014) and Luo et al. (2014). Those authors have also witnessed the incapacity of the bioflocs to remove great amounts of nitrite from the culture water. The concentrations of nitrate in the BFT and Biophyton tanks were greater than in the Control and Periphyton ones (p < 0.05). Krummenauer et al. (2012) have also observed the accumulation of nitrite and nitrate in the water of BFT shrimp tanks. The periphyton present in the underwater structures has not significantly affected the concentrations of nitrate in the present work’s water. Azim et al. (2004) have demonstrated that the periphyton can remove nitrogenous compounds from the water, such as ammonia, nitrite and nitrate. However, the removal rates of nitrogenous compounds by periphyton could be insignificant when their concentrations are very high, a fact that is generally present in intensive aquaculture tanks. This suggests that the substrate-based system for aquaculture are unfit for intensive farming due to their lack of response.
The concentrations of reactive phosphorus were higher in the BFT and Biophyton tanks than in the Control and Periphyton ones (p < 0.05; Table 1). One active sink of waterborne phosphorus is the absorption by the phytoplankton. However, the phosphorus absorption by phytoplankton can be significantly hampered in bioflocs tanks due to water turbidity (Xu, Morris, & Samocha, 2016). Additionally, more phosphorus is generally released in the bioflocs tanks due to their higher rates of organic matter mineralization.
Furtado et al. (2011) have found that the bioflocs are not very efficient in removing phosphorus from the water when the phosphorus concentrations are high. In the present research, the lowest concentration of reactive phosphorus in water was observed in the Periphyton tanks, yet without statistical significance. That suggests that the periphyton has some ability for removing phosphorus from green waters.
The concentrations of organic matter in water were higher in the BFT and Biophyton tanks than in the Control and Periphyton ones (p < 0.05; Table 1). The adjustment of the C: N ratio of water is capable to promote an exponential increase in the organic matter levels of water (Asaduzzaman et al., 2008, Crab et al. 2012). Neither the underwater structures for periphyton, nor the adjustment of the C: N ratio of water have affected the concentrations of hydrogen sulfide in the water of the present study.
Diel monitoring and primary productivity
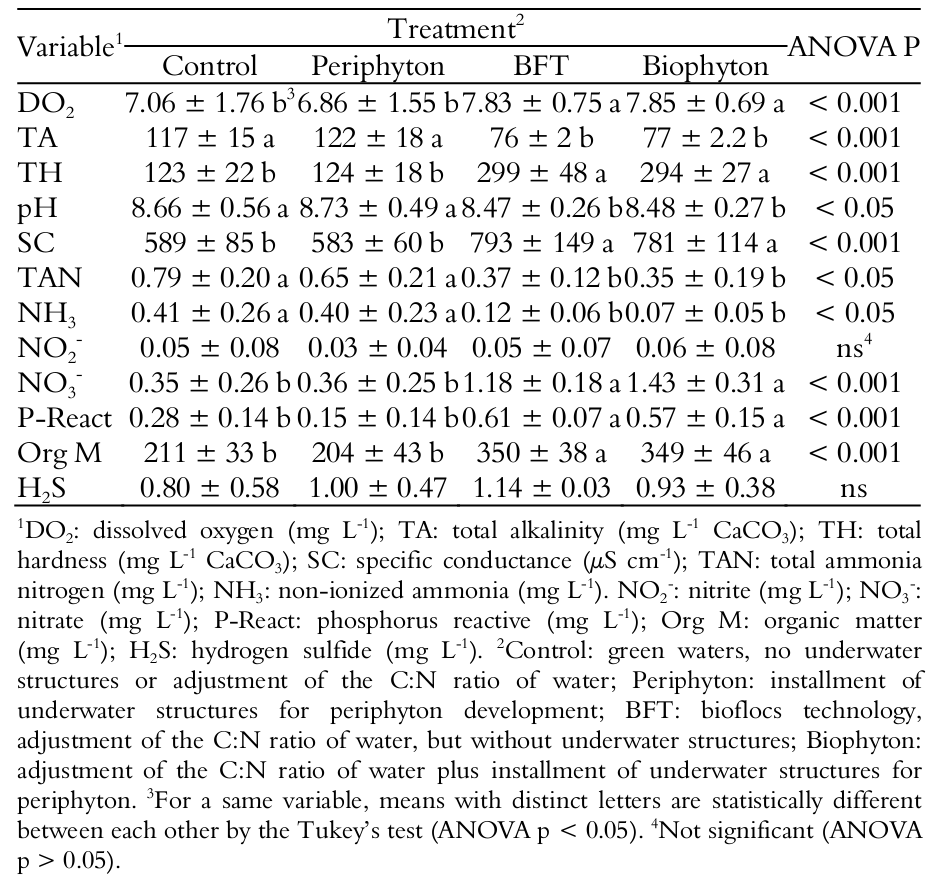
The pH of water in the BFT and Biophyton tanks has remained relatively steady during the diel monitoring (Figure 1). The pH range for those two treatments was equal to 0.5 (minimum and maximum pH = 8.0 and 8.5, respectively). On the other hand, the pH range for the Control and Periphyton tanks was equal to 1.4, that is, almost three times higher (minimum and maximum pH = 7.7 and 9.1, respectively). As the pH of water can significantly affect several of the water quality indicators (Boyd, Tucker, & Viriyatum, 2011), the bioflocs tanks can be considered a more stable fish culture media than the conventional green water tanks.
The DO2 concentrations in water have varied more in the Control and Periphyton tanks than in the BFT and Biophyton ones (Figure 2). In the Control and Periphyton tanks, the DO2 concentrations have varied from below 6 mg L-1, at 11 p.m., to more than 16 mg L-1, at 3 p.m. In the BFT and Biophyton tanks, however, the DO2 concentrations remained close to 8 mg L-1 over the entire observational period. Therefore, the mechanical aeration that was provided continuously to the BFT and Biophyton tanks have promoted a less stressful culture medium for fish culture than the nocturnal aeration.
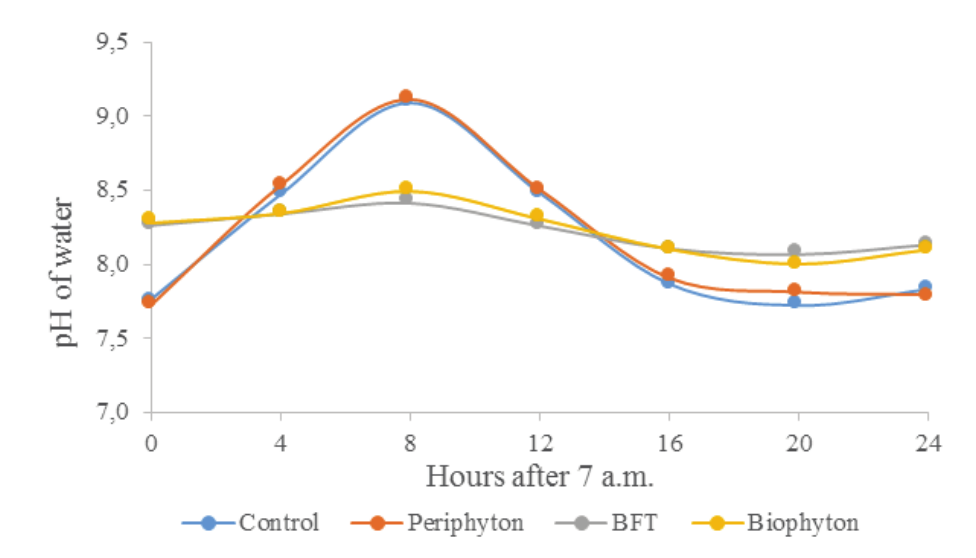
Figure 1.
Diel pH of water in Nile tilapia rearing tanks. Control: green waters, no underwater structures or adjustment of the C:N ratio of water; Periphyton: installment of underwater structures for periphyton development; BFT: bioflocs technology, adjustment of the C:N ratio of water to 15: 1; Biophyton: adjustment of the C:N ratio of water to 15:1 plus installment of underwater structures for periphyton.
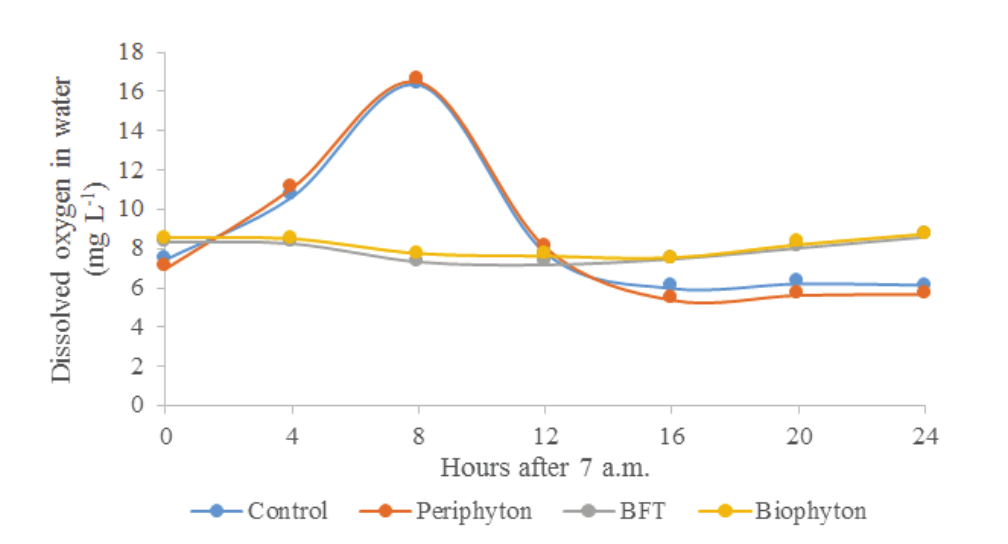
Figure 2.
Diel concentrations of dissolved oxygen in Nile tilapia rearing tanks. Control: green waters, no underwater structures or adjustment of the C: N ratio of water; Periphyton: installment of underwater structures for periphyton development; BFT: bioflocs technology, adjustment of the C:N ratio of water to 15:1; Biophyton: adjustment of the C:N ratio of water to 15:1 plus installment of underwater structures for periphyton.
The net primary productivity (NPP) was higher in the Control and Periphyton tanks than in the BFT and Biophyton ones (Figure 3). The NPP became negative in the latter tanks in the fourth culture week and beyond. Therefore, the bioflocs systems (BFT and Biophyton) have spent circa of one mouth to become steady or ripe. According with Hari, Kurup, Varghese, Schrama, and Verdegem (2004), the culture medium becomes a DO2 consumer when the NPP is negative.
Figure 3.
Net primary productivity of Nile tilapia rearing tanks. Control: green waters, no underwater structures or adjustment of the C: N ratio of water; Periphyton: installment of underwater structures for periphyton development; BFT: bioflocs technology, adjustment of the C:N ratio of water to 15:1; Biophyton: adjustment of the C:N ratio of water to 15:1 plus installment of underwater structures for periphyton.
Growth performance
The four culture systems (green waters, Periphyton, BFT and Biophhyton) have not significantly affected the fish survival in the present work (p > 0.05; Table 2). Likewise, the fish final body weight, specific growth rate and fish yield have also not differed between the tanks (p > 0.05). Those results disagree with Schveitzer et al. (2013b) and Azim and Little (2008) who have observed positive growth performance results by the adjustment of the C: N ratio of water and the installation of underwater structures for periphyton. In the present study, it is supposed that the adoption of a non-restrictive feeding management of fish and the use of nutritionally complete artificial diets have made irrelevant the presence of periphyton in the tanks. Besides, the acknowledged rusticity of the Nile tilapia has possibly interfered in the growth performance results, not allowing the observation of a better efficiency in the bioflocs tanks, despite their superior water quality. It is suggested that the advantages of the bioflocs technology for aquaculture are clearer when there is some level of restriction in the artificial diets allowances and/or a very high fish or shrimp’s stocking density is used.
The food conversion ratio (FCR) was significantly better in the BFT and Bioflocs tanks than in the Control ones (p < 0.05; Table 2). Therefore, the tilapias have ingested more bioflocs as a food source than phytoplankton. The protein efficiency ratio (PER) was greater in the BFT and Biophyton tanks than in the Control ones (p < 0.05; Table 2). Higher PER results mean that a lower content of protein is present in fish feces and, consequently, the TAN concentrations of water will be moved downwards. Therefore, the bioflocs technology for aquaculture helps to make fish and shrimp farming a more environmentally sustainable activity (Xu, Pan, Zhao, & Huang, 2012).
Conclusion
The present study has showed that bioflocs technology removes more ammonia from water than green water or substrate-based systems (periphyton). Moreover, the integration between bioflocs and periphyton (biophyton system) has not brought clear benefits to tilapia culture on water quality and growth performance.
References
American Public Health Association. (1999). Standard methods for the examination of water and waste water (20th ed.). New York City, NY: American Public Health Association.
Asaduzzaman, M., Wahab, M. A., Verdegem, M. C. J., Benerjee, S., Akter, T., Hasan, M. M., & Azim, M. E. (2009). Effects of addition of tilapia Oreochromis niloticus and substrates for periphyton developments on pond ecology and production in C/N-controlled freshwater prawn Macrobrachium rosenbergii farming systems. Aquaculture, 287(3-4), 371-380.
Asaduzzaman, M., Wahab, M. A., Verdegem, M. C. T., Huque, S., Salam, M. A., & Azim, M. E. (2008).C/N ratio control and substrate addition for periphyton development jointly enhance freshwater prawn Macrobrachium rosenbergii production in ponds. Aquaculture, 280(1-4), 117-123.
Avnimelech, Y. (1999). Carbon and nitrogen ratio as a control element in Aquaculture systems. Aquaculture, 176(3-4), 227-235.
Avnimelech, Y., & Kochba, M. (2009). Evaluation of nitrogen uptake and excretion by tilapia in biofloc tanks, using 15N tracing. Aquaculture, 287(1-2), 163-168.
Azim, M. E., & Little, D. C. (2008). The biofloc technology (BFT) in indoor tanks: water quality, biofloc composition, and growth and welfare of Nile tilapia (Oreochromis niloticus). Aquaculture, 283(1-4), 29-35.
Azim, M. E., Little, D. C., & Bron, J. E. (2008). Microbial protein production in activated suspension tanks manipulating C:N ratio in feed and the implications for fish culture. Bioresource Technology, 99(9), 3590-3599.
Azim, M. E., Wahab, M. A., Biswas, P. K., Asaeda, T., Fujino, T., & Verdegem, M. C. J. (2004). The effect of periphyton substrate density on production in freshwater polyculture ponds. Aquaculture, 232(1-4), 441-453.
Boyd, C. E. (2000). Water quality. An introduction. Dordrecht, NL: Kluwer Academic Publishers.
Boyd, C. E., & Tucker, C. S. (1998). Pond aquaculture water quality management. Dordrecht, NL: Kluwer Academic Publishers.
Boyd, C. E., Tucker, C. S., & Viriyatum, R. (2011). Interpretation of pH, acidity, and alkalinity in aquaculture and fisheries. North American Journal of Aquaculture, 73(4), 403-408.
Crab, R., Defoirdt, T., Bossier, P., & Verstraete, W. (2012). Biofloc technology in aquaculture: Beneficial effects and future challenges. Aquaculture, 356-357(1), 351-356.
Cyrino, J. E. P., Bicudo, A. J. A., Sado, R. Y., Borghesi, R., & Dairiki, J. K. (2010). A piscicultura e o ambiente: o uso de alimentos ambientalmente corretos em piscicultura. Revista Brasileira de Zootecnia, 39(supl.), 68-87.
El-Shafai, S. A., El-Gohary, F. A., Nasr, F. A., Van der Steen, N. P., & Gijzen, H. J. (2004). Chronic ammonia toxicity to duckweed-fed tilapia (Oreochromis niloticus). Aquaculture, 232(1-4), 117-127.
Furtado, P. S., Poersch, L. H., & Wasielesky, W. (2011). Effect of calcium hydroxide, carbonate and sodium bicarbonate on water quality and zootechnical performance of shrimp Litopenaeus vannamei reared in bio-flocs technology (BFT) systems. Aquaculture, 321(1-2), 130-135.
Green, J. A., Brandon, E. L., & Hardy, R. H. (2002). Effects of dietary phosphorus and lipid levels on utilization and excretion of phosphorus and nitrogen by rainbow trout (Oncorhynchus mykiss). 2. Production-scale study. Aquaculture Nutrition, 8(4), 291-298.
Guo, L., Li, Z., Xie, P., & Ni, L. (2009). Assessment effects of cage culture on nitrogen and phosphorus dynamics in relation to fallowing in a shallow lake in China. Aquaculture International, 17(3), 229-241.
Hari, B., Kurup, B. M., Varghese, J. T., Schrama, J. W., & Verdegem, M. C. J. (2004). Effects of carbohydrate addition on production in extensive shrimp culture systems. Aquaculture, 241(1-4), 179-194.
Krummenauer, D., Seifert Jr., C. A., Poersch, L. H., Foes, G. K., Lara, G. R., & Wasielesky Jr., W. (2012). Cultivo de camarões marinhos em sistema de bioflocos: análise da reutilização da água. Atlântica, 34(2), 103-111.
Liu, L., Hu, Z., Dai, X., & Avnimelech, Y. (2014). Effects of addition of maize starch on the yield, water quality and formation of bioflocs in an integrated shrimp culture system. Aquaculture, 418-419(1), 79-86.
Luo, G., Gao, Q., Wang, C., Liu, W., Sun, D., Li, L., & Tan, H. (2014). Growth, digestive activity, welfare, and partial cost-effectiveness of genetically improved farmed tilapia (Oreochromis niloticus) cultured in a recirculating aquaculture system and an indoor biofloc system. Aquaculture, 422-423(20), 1-7.
Luo, G.-Z., Avnimelech, Y., Pan, Y.-F., & Tan, H.-X. (2013). Inorganic nitrogen dynamics in sequencing batch reactors using biofloc technology to treat aquaculture sludge. Aquacultural Engineering, 52, 73-79.
Ray, A. J., Dillon, K. S., & Lotz, J. M. (2011). Water quality dynamics and shrimp (Litopenaeus vannamei) production in intensive, mesohaline culture systems with two levels of biofloc management. Aquacultural Engineering, 45(3), 127-136.
Rostagno, H. S., Albino, L. F. T., Donzele, J. L., Gomes, P. C., Oliveira, R. F., Lopes, D. C., ... Barreto, S. L. T. (2011). Composição de alimentos e exigências nutricionais de aves e suínos: composição de alimentos e exigências nutricionais (3a ed.). Viçosa, MG: Imprensa Universitária.
Schrader, K. K., Green, B. W., & Perschbacher, P. W. (2011). Development of phytoplankton communities and common off-flavors in a biofloc technology system used for the culture of channel catfish (Ictalurus punctatus). Aquacultural Engineering, 45(3), 118-126.
Schveitzer, R., Arantes, R., Baloi, M. F., Costódio, P. F. S., Arana, L. V., Seiffert, W. Q., & Andreatta, E. R. (2013b). Use of artificial substrates in the culture of Litopenaeus vannamei (Biofloc System) at different stocking densities: Effects on microbial activity, water quality and production rates. Aquacultural Engineering, 54, 93-103.
Schveitzer, R., Arantes, R., Costódio, P. F. S., Espirito-Santo, C. M., Arana, L. V., Seiffert, W. Q., & Andreatta, E. R. (2013a). Effect of different biofloc levels on microbial activity, water quality and performance of Litopenaeus vannamei in a tank system operated with no water exchange. Aquacultural Engineering, 56, 59-70.
Xu, W. J., Pan, L. Q., Zhao, D. H., & Huang, J. (2012). Preliminary investigation into the contribution of bioflocs on protein nutrition of Litopenaeus vannamei fed with different dietary protein levels in zero-water exchange culture tanks. Aquaculture, 350-353(20), 147-153.
Xu, W.-J., Morris, T. C., & Samocha, T. M. (2016). Effects of C/N ratio on biofloc development, water quality, and performance of Litopenaeus vannamei juveniles in a biofloc-based, high-density, zero-exchange, outdoor tank system. Aquaculture, 453, 169-175
Notes