Engenharia Mecânica
Biodiesel as fuel for boilers
Biodiesel como combustível para caldeiras
Biodiesel as fuel for boilers
Acta Scientiarum. Technology, vol. 39, 05, pp. 609-614, 2017
Universidade Estadual de Maringá

This work is licensed under Creative Commons Attribution 4.0 International.
Received: 16 October 2015
Accepted: 05 August 2016
Abstract: Frying oil is one of the major environmental problems in urban centers, causing blockage of sewer pipes and pollution of rivers. Producing biodiesel with frying oil is an alternative to reduce the environmental impacts generated in the undue discarding of the same. Because it has high calorific power, great availability, low emission of pollutants when burned, besides being renewable, which guarantees its uninterrupted supply. This study evaluated the use of diesel blends with biodiesel from frying oil, in proportions varied by 10% for each treatment, ranging from B0 with 0% to B100 with 100% biodiesel, at room temperature and after heating at an average temperature of 50.63°C. The blends were evaluated through measurements of density, viscosity, calorific power and fuel consumption, which were useful for determining the power supplied. Without preheating, there was a gradual increase in fuel consumption, when the percentage of biodiesel in blends increased. However, preheating resulted in a decrease in consumption. Thus, without preheating, there was a low decrease of the power supplied by biodiesel compared to diesel oil. With preheating, this reduction was significantly higher.
Keywords: fuel blends, consumption, diesel fuel, heating, frying oil.
Resumo: O óleo de fritura é um dos maiores problemas ambientais nos centros urbanos, causando obstrução nas tubulações de esgoto e poluição dos rios. Produzir biodiesel com óleo de fritura é uma alternativa para reduzir os impactos ambientais gerados no descarte indevido do mesmo. Pois possui elevado poder calorífico, grande disponibilidade, baixa emissão de gases poluentes ao ser queimado, além de ser renovável o que garante seu suprimento ininterrupto. Este estudo avaliou o uso de misturas de óleo diesel com biodiesel de óleo de fritura, em proporções variadas em 10% para cada tratamento, sendo B0, 0% e B100, 100% de biodiesel, à temperatura ambiente e após aquecimento a uma temperatura média de 50,63ºC. Realizou-se nas blendas medições de: densidade, viscosidade, poder calorífico e consumo de combustível, que foram úteis para determinar a potência fornecida. Sem pré-aquecimento, obteve-se um aumento gradual no consumo de combustível, quando o percentual de biodiesel nas misturas aumentou. No entanto, pré-aquecendo houve uma diminuição no consumo. Assim, sem pré-aquecer, obteve-se uma baixa redução na potência fornecida pelo biodiesel em comparação com diesel de petróleo. Pré-aquecendo, esta redução foi significativamente maior.
Palavras-chave: mistura de combustíveis, consumo, óleo diesel, aquecimento, óleo de fritura.
Introduction
According to Brasil (2013), petroleum and its derivatives account for 39.3% of internal energy supply, and diesel fuel is the main fuel sold in Brazil, representing 42.25% of the byproducts of petroleum, followed by gasoline, which has 21.17% of supply.
For Agência Nacional do Petróleo, Gás Natural e Biocombustíveis (Brasil, 2012), fossil fuels are great polluters of the environment and considering that they tend to exhaustion, biofuels have gained notable participation in the world energy market.
Also according to Brasil (2012), the two main liquid biofuels used in Brazil are ethanol extracted from sugarcane, and, in increasing scale, biodiesel, which is produced from vegetable or animal fats and added to petroleum-based diesel in varying proportions, being able to replace, partially or totally, petroleum derivatives and natural gas fuels in internal combustion engines or other type of energy generation. In 2013, biodiesel production reached 2917 (103 toe), an increase of 6.85% over the previous year (Soares & Pinto, 2014). Yet Still according to the authors, the main raw materials used in biodiesel production are: soybean oil (78.08%), cattle fat (17.74%), and chicken fat (2.07%), while frying oil accounts for 0.56%.
The molecules of vegetable oils contain glycerin and its use in engines without modification may cause carbonization problems and deposits in the nozzles injectors, in intake valves and exhaust, premature wear of pistons, rings segment and cylinder lube oil dilution, difficulty in starting in the cold, uneven burning, reduced thermal efficiency and unpleasant odor of exhaust fumes. To reduce these problems, mixtures of vegetable oils may be used with oil diesel up to 30%, using fatty acid esters obtained by chemical transformation of vegetable oil in biodiesel (Poulton, 1994; Acioli, 1994; Ribeiro, 2002).
Biodiesel is obtained through the transesterification reaction of vegetable or animal oil, with ethanol or methanol and in the presence of a catalyst, with better ignition properties, pour point and flow properties (viscosity and specified density). Besides the transesterification, which is the best know method and used in large-scale for production of biodiesel, there are three other: dilution, micro-emulsification and pyrolysis (Ferrari, Oliveira, & Scabio, 2005; Knothe, Gerpen, Krahl, & Ramos, 2006).
Biodiesel has a higher viscosity than diesel fuel, which according Lôbo, Ferreira, and Cruz (2009) impairs the atomization in the nozzle of motors. As a result, it changes the ratio air/fuel admitted in the motor, causing waste deposition on its internal parts. The authors also stated that the pour point, cold filter plugging, fog and solidification occur at higher temperatures than diesel oil, requiring adjustments of the air inlet, the pressure and the distance from the nozzle of the burner in the combustion chamber.
Serfaty (2007) claims that the burner is a device that places a flame in a certain position, by releasing air and fuel with a given mixture, which depends on the air pressure available or the drawing along the burner and the fuel discharge energy, which can ensure continuous ignition and complete combustion. Its function is to subdivide the fuel into droplets with the smallest possible diameter, and introduce the fog of the small particles properly within the combustion chamber.
When increasing the nebulization pressure, the droplets obtained have smaller diameter, which allows reaching the ignition condition nearest to the nebulizer nozzle. The vaporization rate of a droplet increases as the thermal conductivity and the enthalpy of reaction increase, and decrease with the reduction of specific heat at constant pressure, enthalpy of vaporization and boiling temperature of the liquid (Serfaty, 2007).
The principle of burning of fuels (liquid, solid or gaseous) has similar characteristics, but each flame has certain properties, which influences the power and the pollutant formation. For each fuel type, the burner used is different. Small temperature variations of the fuel quickly change the viscosity, which affects their atomization during injection into the combustion chamber (Faulkner Jr., 1987; Canciam, 2010).
Peterson, Reece, Thompson, Beck, and Chase (1996) claim that, to improve the combustion of biofuels produced from biomass, in addition to preheating, some changes are required in engines such as the adjustment of the distance from the nozzle, pressure adjustment and air input, among others.
Biodiesel has a lower transfer of radiation heat in the furnace, but can substitute diesel fuel in fire tube boilers. To improve the burning of biofuel, it is recommend monitoring the temperature in the chimney inlet to control fuel injection and ensure better income to the boiler (heat transfer in the furnace and tube banks). The diesel flame is formed in a region closer to the nozzle and the air inlet has a larger angle. It can also be observed through a sensitive analysis that in biodiesel burning, combustion products showed reduced darkening, aggressiveness and unpleasant smell than diesel fuel (Souza, Santos, Martins, & Santos, 2008).
Analyzing the performance of an agricultural tractor blends with fossil diesel and castor biodiesel, Tabile et al. (2009) found that at a load of 13 KN, the specific consume was 0.456 kg hour-1 KW, and increasing this load to 23 KN, the specific consume decreased to 0.358 kg hour-1 KW, according to Rojas-González, Chaparro-Anaya, and Andrés-Ospina (2011).
The burning of biodiesel from residual oil, palm and canola, with different loads coupled to a motor-generator is a directly proportional relation, KW 0.131 kg hour-1 with a load of 0.54 kW and kWh 0.174 kg hour-1 with a load of 2.41 KW.
Studies have shown a good efficiency in mixtures of diesel and biodiesel for use in boilers, especially when considering the combustion efficiency and the composition of the combustion gases (Ghorbani et al. 2011).
The aim of this study was to evaluate mixtures in different proportions of diesel and biodiesel from cooking oil, with and without preheating, applied on a burner for heating swimming pools.
Material and methods
This study was conducted in a covered pool in an enclosed space with 64 m3 capacity, in the city of Cascavel, Paraná, Brazil, at the campus of the State University of Werstern Paraná (Unioeste), were the average annual rainfall is 1.640 mm and the average temperature is 19°C according to Instituto Agronômico do Paraná (Iapar, 2011). The site has two heating systems of the pool water: the electric, which is used in the summer months due to lower required heat transfer, and powered by diesel oil, used in winter, though only one of them can be operating at the same time.
The heating of pool water is made by a water-tube boiler, which has a reservoir of 180 L, and the circulation is made by a motor pump of 1.5 CV, with average flow of 4,716 m3 hour-1. The input and output water pipes in the boiler are metallic and have 40 mm diameter, in the rest of the system the tubing are in PVC with 50 mm diameter.
The physicochemical parameters measured in the experiment were: Density at 20°C ( ρ ), according to César, Paoli, and Andrade (2004) (Equation 1) is made by pycnometer method, which is performed by measuring the weight of the pycnometer with the fuel mixture, knowing its volume.
 (1)
(1)where:
ρ = absolute density (kg m-3);
mlíquid = kg;
Vlíquid = m3.
Lower heating value (LHV), which was determined by a bomb calorimeter model E2K which has a precision of 10-3 g. Mixtures of fuels are inserted in an adiabatic container and ignition occurs through a wire, which passes electrical current. The bomb calorimeter was pressurized to 3 MPa and inserted into the calorimeter, which measures the values of the High heating value (HHV) of mixtures in MJ kg-1 and as Volpato, Conde, Barbosa, and Salvador (2009) (Equation 2) describe, the values of the lower heating value are obtained.
 (2)
(2)where:
LHV = Lower Heating Value (MJ kg-1);
HHV = Higher Heating Value (MJ kg-1).
Volumetric consumption corrected temp of fuel (Vcct), adapted by Yanai, Silveira, Lanças, Corrêa, and Maziero (1999) and corrected by Official Tables (Brasil, 1970) (Equation 3).This data were collected by the Data logger to each second. With this data, we obtained the power supplied, adapted from Nogueira, Rocha, Nogueira, and Monteiro (2005) (Equation 4).
 (3)
(3)where:
Vcct = Volumetric consumption corrected temp of fuel (kg hour-1);
=correction factor (tabulated);
Vct = Volumetric consumption time (kg hour-1).
 (4)
(4)where:
P = Power supplied by the burner (KWh).
To perform the experiment, it was adapted to the existing system: a reservoir of 20 L used for insertion of the fuel blends; pipes in polyurethane of 6 and 8 mm, pressure connectors and fuel filters. In a Data logger, the following sensors were coupled: flow meter Sparling Oval M-III – model LSF41L, with an accuracy of ± 1%, to measuring consumption; temperature sensor PT 100 - rod type, with an accuracy of ± 0.2ºC, for measuring the instantaneous fuel temperature; 2 thermocouple sensors - type j, with an accuracy of ± 2.2ºC, for monitoring inlet and outlet water temperatures in the boiler. The burner of the boiler has air inlet (damper) and pressure adjustment, which should be set at 9 bar. According to the manufacturer's manual of the burner Hof a mat 125 used in the experiment, the equipment is specific to diesel fuel; however, biodiesel can be used. It has a minimum power of 55 KWh and maximum power of 125 kWh at oil rate of 4.7 to 10.3 kg hour-1.
To heat the fuels, a 350 W resistance was inserted in a recipient with water and a temperature controller, which is positioned externally to the fuel flow. On the controller, which turns on and off the resistance, a thermocouple temperature sensor - type j was inserted, maintaining the temperature at 85°C.
To obtain the fuel for the experiment, commercial diesel fuel was acquired, S500 and B5, and biodiesel was produced at Unioeste experimental plant, using as raw material 200 L of frying oil, filtered and dehumidified, 30 L of methanol and 8 L of sodium methylate in solution at 30% (catalyst) through the triglycerides trans esterification. The reagents were left in the reactor for 40 min and in decanters for 2 hours to separate the glycerin from the biodiesel.
The experimental design used was entirely randomized in triplicate, and the treatments used were diesel oil (ODxx), waste frying oil biodiesel (Bxx), with and without preheating of the fuel in mixtures of 0 (B0) to 100% (B100), varied by 10% (B0 refers to the commercial diesel oil, that has 5% biodiesel). The time of each sample was predetermined in three min to burning fuel, checking the fuel consumption and making the conversion to L hour-1.
The density at 20ºC and the lower heating value were collected in triplicate, the results were submitted to analysis of variance (ANOVA) and the means were compared by Tukey test, adopting the significance level of 1-5% using the statistical package version Assistat® 7.7 beta (Silva, 2014). One second-order polynomial of the values obtained from volumetric consumption corrected time and power supplied was applied.
Results and discussion
The average density at 20°C () and lower heating value (LHV) of 3 samples are presented in Table 1.
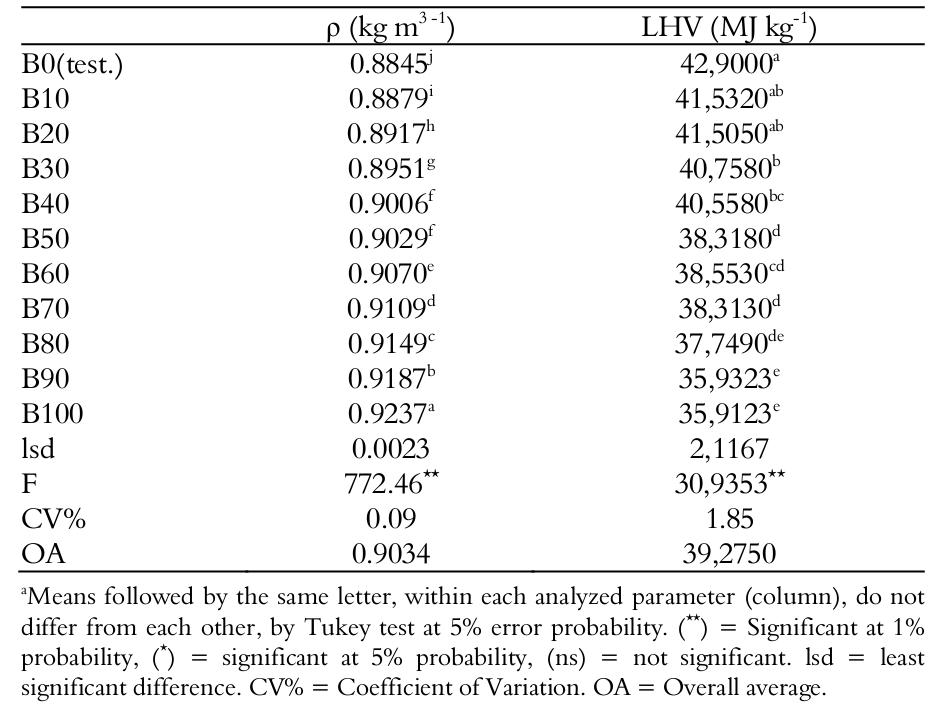
Note that with increasing percentage of biodiesel present in the mixtures, the density tends to increase. According to the Resolution N°14 (Brasil, 2012), which regulates fuel in Brazil, the density should be between 0.85 and 0.90 kg m3.-1. The density is considered the relationship between mass and a determined volume of liquid, according to César et al. (2004). With the process of frying, the oil properties are altered and the trend is that its density increases. However, when producing biodiesel, the density is corrected. Nevertheless, the density obtained for B100 was 0.924 kg m3.-1, higher value than the stipulated limit.
Alptekin and Canakci (2009) report that the density of biodiesel produced from soy oil in natura is 0.88 kg m3.-1, while Reolon, Rosa, Dolci, Secco, and Santos (2012) found 0.8835 kg m-3. For Moecke, Werner, Gelsleichter, Avila, and Silveira (2012), the density at 20°C of the biodiesel from residual oil is 0.894 kg m-3, differing from the value obtained at 3.25%.
As cited by Ramey, Lumley, Prieto, Porter, and Cath (2015), the higher heat value (HHV) is the value where the water vapors present in the fuel gases are condensed and the lower heat value (LHV) is the value at which these vapors do not condense. Mittelbach and Tritthart (1988) also claim that the biodiesel of residual frying oil provides a LHV of 37.50 MJ kg-1, which differs in 4.23% from the average value of 35.912 MJ kg-1 found. This difference represents a smaller amount of energy released in combustion, which requires more fuel to raise the same temperature.
The volumetric consumption corrected time (Vcct) of fuel was determined by measuring the fuel mass volume consumed during the sampling and corrected to the temperature of 20°C, using the Official Tables (Brasil, 1970), represented in Figure 1. The average temperature of the fuel during all the experiment was 30.72°C, and with preheating was 50.63ºC.
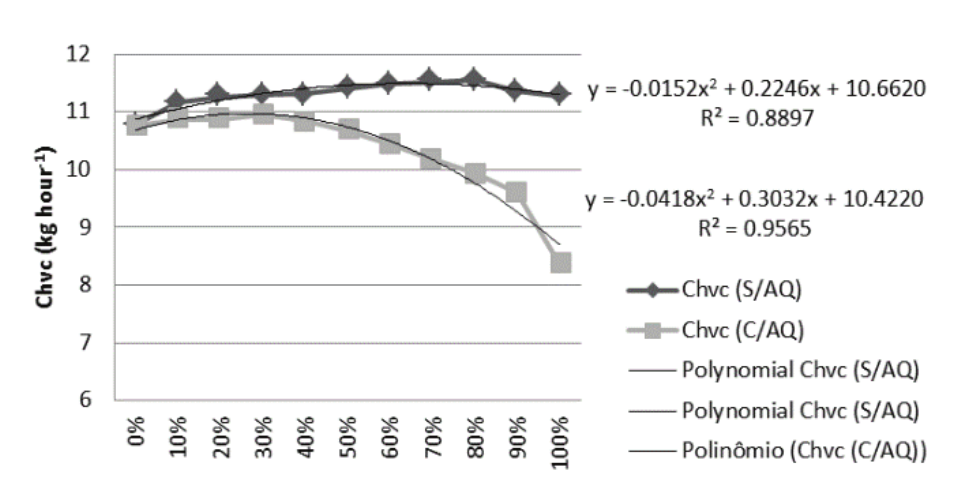
Figure 1.
Volumetric consumption time fixed in fuel blends.
A burner 111.6 KWh net maximum power of the diesel consumes 12.1 L hour-1, according to the manufacturer of the boiler CA – OGS (Broilo, 2016) in a boiler has an efficiency of 92% using a 3.92 bar pressure, however the burner used in the experiment consumed 10.91% less fuel, using B0 fuel. Tabile et al. (2009) obtained an increase in volumetric consumption of biodiesel from mammon 14.6% higher compared to diesel in an agricultural tractor, while Soranso et al. (2008) found a difference of 12.9 for diesel and 14.9 L hour-1 for biodiesel from frying oil. The increase of biodiesel in the blend leads to a higher fuel consumption with the maximum value for the B80 mixing without heating, 11.54 L hour-1. The value for B100 was 11.28 L hour-1 being higher than 4.05% commercial diesel oil (5%).
Regarding the functioning of the burner, in percentages up to 80% (B80) of biodiesel, without preheating, there were no combustion failures. The pressure had to be reduced to 8.0 bar to B80 and 7.0 Bar to B90 and B100. The inlet air was maintained in all the mixtures. With preheating, there were no failures at any percentage, though a 9 to 10 bar adjustment was required for that the burner combustion do not display failure.
With increasing temperature, the viscosity is reduced, which improves fuel flow in the pipes and the atomization nozzle within the combustion chamber. This ensures better burning, lower fuel consumption and better efficiency.
There was a reduction of only 0.13% in fuel consumption when preheating pure diesel oil (B0). However, by increasing the percentage of biodiesel in the mixtures, this difference becomes more expressive. Whereas the consumption of pure biodiesel (B100) using the pre-heating, the reduction in consumption was 25.75% in relation to no preheating it. This represents a reduction of 22.32% in the volumetric consumption time of diesel oil (B0) and without preheating (existing system), in relation to the volumetric consumption time of biodiesel (B100) with preheating (implanted system).
For Nwafor (2003), engines powered by diesel from canola oil in natura has a higher specific consumption than diesel; however, when heated to 80°C, the consumption becomes similar to that of diesel. A burner 111.6 KWh net maximum power of the diesel consumes 12.1 L per hour, according to the manufacturer of the boiler CA – OGS (Broilo, 2016) a boiler has an efficiency of 92% and using a 3.92 bar pressure, which agrees with the values obtained.
Souza, Santos, Ferreira, Martins and Módolo (2008) found in a furnace with 180000 kcal hour-1 using a monobloc burner for oil and gas, while maintaining the same consumption of frying oil biodiesel and diesel fuel, the flame temperature of the biodiesel (1040-1090°C) is lower when compared to diesel oil (1150-1200°C) due to its lower calorific value, which results in a lower efficiency.
The power supplied by the burner, preheating the pure diesel, was 113.498 KWh, 0.13% lower than the not-preheated, indicating that the installation of a system similar to the experiment, using diesel oil, does not result in significant reduction in consumption and power supplied. The maximum power provided by the equipment occurred in mixtures of 20% biodiesel (115.708 KWh without preheating, 111.802 KWh with preheating). When pure biodiesel from frying oil (B100) was used, without heating, a power of 103.936 KWh was obtained, different from the installed heating system, which provided only 77.175 kWh, resulting in a reduction of 25.75%. Figure 2 demonstrates a comparison between the power supplied by burning mixtures with and without pre-heating.
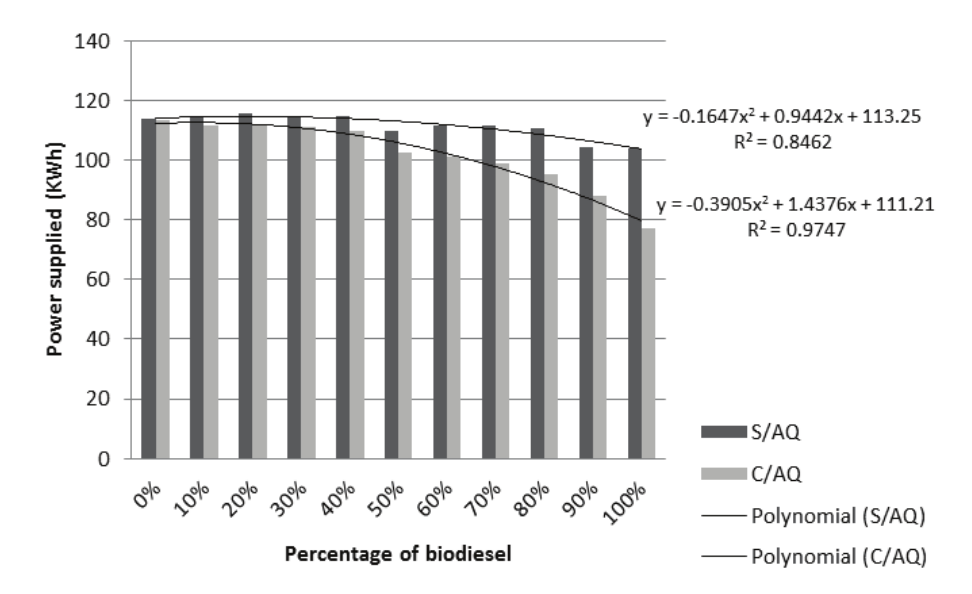
Figure 2.
Power supplied by the burner in the blends.
The preheated mixture B100 consumed 77.17 kWh. Adding the power used by the resistance to preheating of 0.35 kWh, the reduction in fuel consumption in the implanted system is great. Considering that the frying oil collected has no cost (transport, cost methanol, catalyst and operation of the experimental plant are not included), the production of biodiesel of the frying oil and the implantation of the developed system are alternatives that reduce fuel consumption and the costs involved in pool heating systems.
Conclusion
The values of density at 20°C obtained in the blends increase with the percentage of biodiesel present, while the lower heating value (LHV) decreases.
The preheating of fuel decreases Vcct, especially when using biodiesel, which represents an improvement of energetic efficiency, and the higher the power supplied by the burner, the higher the fuel consumption.
In addition to the preheating of the fuels, the use of thermal cover in the pool, appropriate adjustment of the pressure and air inlet and the utilization of waste heat in the chimney are, among many others, measures adopted that improve system efficiency.
The high cost of diesel oil, the reduction in consumption and emission of polluting gases are incentives to the implementation of this type of system, in addition to the low cost.
References
Acioli, J. D. L. (1994). Fontes de energia. Brasília, DF: UnB.
Alptekin, E., & Canakci, M. (2009). Characterization of the key fuel properties of methyl ester–diesel fuel blends. Fuel, 88(1), 75-80.
Brasil. (1970, 13 de Junho). Resolução CNP nº 6, de 25 de junho de 1970 – 1521ª Sessão Ordinária. Diário Oficial [da] República Federativa do Brasil, Brasília, DF.
Brasil. Agência Nacional do Petróleo, Gás Natural e Biocombustíveis. (2012). Resolução n. 14, de 11 de maio de 2012. Diário Oficial da União, Brasília, DF.
Brasil. Empresa de Pesquisa Energética. (2013). Balanço energético nacional 2013: ano base 2012. Rio de Janeiro, RJ: EPE.
Broilo, S. A. (2016). Boilers diesel and gas CA-OGS. Retrieved from http://broilo.com.br/wp-content/uploads/laminas/caldeira_ca-ogs.pdf
Canciam, C. A. (2010). Efeito da temperatura na viscosidade de óleos vegetais refinados. Ciências Exatas e da Terra, Agrárias e Engenharias, 16(1), 7-12.
César, J., Paoli, M. A., & Andrade, J. C. A. (2004). Determinação da densidade de sólidos e líquidos. CHEMKEY, Liberdade para Aprender, 4(16), 16-22.
Faulkner Jr., E. A. (1987). Guide to efficient burner operation: gas, oil, and dual fuel (2nd ed.). Lilburn, GA: Fairmont Press.
Ferrari, R. A., Oliveira, V. S., & Scabio, A. (2005). Biodiesel de soja: taxa de conversão em ésteres etílicos, caracterização físico-química e consumo em gerador de energia. Química Nova, 28(1), 19-23.
Ghorbani, A., Bazooyar, B., Shariati, A., Jokar, S. M., Ajami, H., & Naderi, A. (2011). A comparative study of combustion performance and emission of biodiesel blends and diesel in an experimental boiler. Applied Energy, 88(12), 4725-4732.
Instituto Agronômico do Paraná [IAPAR]. (2011). Médias históricas das estações do IAPAR. Recuperado de http://www.iapar.br/modules/conteudo/conteudo.php?conteudo=1070>
Knothe, G., Gerpen, J. V., Krahl, J., & Ramos, L. P. (2006). Manual de biodiesel Edgard Blücher. São Paulo, SP: Blucher.
Lôbo, I. P., Ferreira, S. L. C., & Cruz, R. S. (2009). Biodiesel: parâmetros de qualidade e métodos analíticos. Química Nova, 32(6), 1596-1608.
Mittelbach, M., & Tritthart, P. (1988). Diesel fuel derived from vegetable oils, III. Emission tests using methyl esters of used frying oil. Journal of the American Oil Chemists' Society, 65(7), 1185-1187.
Moecke, E. H. S., Werner, S. M., Gelsleichter, Y. A., Avila, A. F. A., & Silveira, T. C. (2012). Produção de biodiesel a partir do óleo de fritura usado e o empoderamento da comunidade. Revista Gestão & Sustentabilidade Ambiental, 1(1), 33-40.
Nogueira, L. A. H., Rocha, C. R., Nogueira, F. J. H., & Monteiro, M. A. G. (2005). Eficiência energética no uso de vapor: manual prático. Rio de Janeiro, RJ: Eletrobrás.
Nwafor, O. M. I. (2003). The effect of elevated fuel inlet temperature on performance of diesel engine running on neat vegetable oil at constant speed conditions. Renewable energy, 28(2), 171-181.
Peterson, C. L., Reece, D. L., Thompson, J. C., Beck, S. M., & Chase, C. (1996). Ethyl ester of rapeseed used as a biodiesel fuel-a case study. Biomass and Bioenergy, 10(5-6), 331-336.
Poulton, M. L. (1994). Alternative fuels for road vehicles. Billerica, MA: Computational Mechanics Publications.
Ramey, D. F., Lumley, N. P., Prieto, A. L., Porter, J. M., & Cath, T. Y. (2015). Evaluating air-blown gasification for energy recovery from wastewater solids: Impact of biological treatment and point of generation on energy recovery. Sustainable Energy Technologies and Assessments, 9, 22-29.
Reolon, C. B., Rosa, H. A., Dolci, B. D., Secco, D., & Santos, R. F. (2012). Rentabilidade e propriedades físico-químicas do biodiesel de cinco espécies vegetais. Cultivando o Saber, 5(3), 22-32.
Ribeiro, S. K. (2002). Barreiras na implantação de alternativas energéticas para o transporte rodoviário no Brasil. Rio de Janeiro, RJ: Coppe/UFRJ.
Rojas-González, A., Chaparro-Anaya, Ó., & Andrés-Ospina, C. (2011). Evaluación de mezclas biodiésel-diésel en la generación de energía eléctrica. Ingeniería y Universidad, 15(2), 319-336.
Serfaty, R. (2007). Combustão e queimadores. Rio de Janeiro, RJ: Cenpes.
Silva, F. A. (2014). Versão 7.7 beta. Campina Grande, PB: Deag-CTRN-UFCG.
Soares, R. W., & Pinto, F. V. (2014). Boletim mensal de biodiesel. Rio de Janeiro, RJ: ANP.
Soranso, A. M., Gabriel Filho, A., Lopes, A., Souza, E. G., Dabdoub, M. J., Furlani, C. E. A., & Camara, F. T. (2008). Desempenho dinâmico de um trator agrícola utilizando biodiesel destilado de óleo residual. Revista Brasileira de Engenharia Agrícola e Ambiental, 12(5), 553-559.
Souza, G. R., Santos, A. M., Ferreira, S. L., Martins, K. C. R., Módolo, D. L. (2008). Evaluation of the performance of biodiesel from waste vegetable oil in a flame tube furnace. Applied Thermal Engineering, 29, 2562-2566.
Souza, G. R., Santos, A. M., Martins, K. R., & Santos, R. F. E. (2008). Utilização de biodiesel de óleo de fritura em fornalha calorimétrica flamotubular. Minerva, 2(1), 75-78.
Tabile, R. A., Lopes, A., Dabdoub, M. J., Camara, F. T., Furlani, C. E. A., & Silva, R. P. (2009). Biodiesel de mamona no diesel interior e metropolitano em trator agrícola. Engenharia Agrícola, 29(3), 412-423.
Volpato, C. E. S., Conde, A. P., Barbosa, J. A., & Salvador, N. (2009). Desempenho de motor diesel quatro tempos alimentado com biodiesel de óleo de soja (B 100). Ciência e Agrotecnologia, 33(4), 1125-1130.
Yanai, K., Silveira, G. M., Lanças, K. P., Corrêa, I. M., & Maziero, J. V. G. (1999). Desempenho operacional de trator com e sem o acionamento da tração dianteira auxiliar. Pesquisa Agropecuária Brasileira, 34(8), 1427-1434.
Notes