Ciência, Tecnologia de Alimentos e Engenharia de Alimentos
Centesimal composition, fatty acids profile and the nutritional quality index of four seafood species from the southern region of Brazil
Composição centesimal, perfil de ácidos graxos e índices de qualidade nutricional de quatro espécies de frutos do mar da região sul do Brasil
Centesimal composition, fatty acids profile and the nutritional quality index of four seafood species from the southern region of Brazil
Acta Scientiarum. Technology, vol. 40, 2018
Universidade Estadual de Maringá

This work is licensed under Creative Commons Attribution 4.0 International.
Received: 06 September 2017
Accepted: 21 December 2017
Abstract: The objective of this study was to evaluate the centesimal composition, fatty acid profile and the nutritional quality index of four seafood species: pink shrimp (Penaeus brasiliensis), Pacific white shrimp (Litopenaeus vannamei), mussel (Perna perna) and octopus (Octopus vulgaris), that were collected in Santa Catarina, Brazil. The nutritional quality of the lipid fraction was evaluated by the index of thrombogenicity (IT), index of atherogenicity (IA) and the hypocholesterolaemic fatty acids-to-hypercholesterolaemic fatty acids ratio (h/H). All the studied species presented a high protein content (12.88-21.61%) and a low amount of total lipid (0.26-4.07%). The pink shrimp had the most protein and octopus contained the least total lipid. Eicosapentaenoic acid and docosahexaenoic acid were detected in all samples, presenting the highest values in mussel (204.62 and 123.57 mg 100 g-1, respectively). The analysed species presented low IT and IA values and an h/H ratio around 2 or higher, and therefore, they are considered potentially healthy foods. Moreover, the shrimp species can be classified as a source of omega-3s, while the mussels can be categorised as having a high content in these polyunsaturated fatty acids.
Keywords: polyunsaturated fatty acids, omega-3, omega-6, eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA).
Resumo: Este estudo teve como objetivo avaliar a composição centesimal, perfil de ácidos graxos e índices de qualidade nutricional de quatro espécies: camarão rosa (Penaeus brasiliensis), camarão branco do pacífico (Litopenaeus vannamei), mexilhão (Perna perna) e polvo (Octopus vulgaris), coletados em Santa Catarina, Brasil. A qualidade nutricional da fração lipídica foi determinada pelos índices de trombogenicidade (IT), aterogenicidade (IA) e a relação entre ácidos graxos hipocolesterolêmicos (h): hipercolesterolêmico (H) h/H. Os resultados demonstraram que as espécies estudadas apresentam alto teor de proteína (12,88 a 21,61%) e baixo teor de lipídios (0,26 a 4,07%), sendo o camarão rosa o alimento mais proteico e o polvo com menor teor de lipídios totais. Em relação aos ácidos graxos, foram detectados os ácidos eicosapentaenoico (EPA) e docosahexaenóico (DHA), em todas as amostras, sendo o mexilhão Perna perna, a espécie que apresentou maiores valores (204,62 e 123,57 mg 100 g-1, respectivamente). Desta forma, as espécies analisadas demostraram-se como alimentos potencialmente saudáveis, pelos baixos valores de IA e IT e valores próximos ou superior a 2 do hH. As espécies de camarão podem ser classificadas como fonte de ômega-3, enquanto que o mexilhão pode ser classificado como alto teor desse constituinte.
Palavras-chave: poli-insaturados, ômega-3, ômega-6, ácidos, eicosapentaenoico (EPA), docosahexaenóico (DHA).
Introduction
In 2013, Brazil's fishery production increased by 66% over the year 2012, from 1.5 Mt to 2.5 Mt. However, the Food and Agriculture Organisation of the United Nations (Food and Agriculture Organisation [FAO], 2004) still regards Brazil as a small global producer, considering the country’s hydrological potential and diversity, with more than 8000 km of coastline.
According to Me´dale, Lefèvre, and Corraze (2003), fish, shrimp and molluscs occupy an important place in human nutrition. Particularly, these marine foods are a valuable source of high quality protein, minerals, vitamin D, polyunsaturated fatty acids (PUFAs) and essential fatty acids, particularly, omega-3s (n3s), such as α-linolenic acid (18:3n3), eicosapentaenoic acid (EPA, 20:5n3) and docosahexaenoic acid (DHA, 22:6n3). The well-documented health benefits of omega-3s include a reduction of risk factors associated with cardiovascular diseases, hypertension, general inflammation, depression, asthma, psoriasis and others (Simopoulos, 2008; Calder & Yagoob, 2009; Hoffmire, Block, Thevenet-Morrison, & Wijngaarden, 2012; Mohamad, Baharuldin, Moklas, Fakurazi, & Muhammad, 2014).
The nutritional quality of lipids can be verified through the omega-6-to-omega-3 (n6/n3) fatty acids ratio and the polyunsaturated-to-saturated fatty acids ratio (PUFAs/SFAs) (Souza, Bentes, Simões, & Fontenelle, 2008). However, Santos-Silva, Bessa, and Santos-Silva (2002) recommend evaluating the nutritional quality of the fatty acid profile based on the functional effects of the fatty acids, such as the hypocholesterolaemic-to-hypercholesterolaemic ratio (h/H), index of atherogenicity (IA) and index of thrombogenicity (IT).
The h/H ratio considers the functional activity of fatty acids in the metabolism of lipoproteins that transport plasma cholesterol, whose type and quantity is related to the higher or lower risk of cardiovascular disease incidence (Santos-Silva et al., 2002).
The IT considers myristic (14:0), palmitic (16:0) and stearic (18:0) as thrombogenic, and PUFAs (omega-6 and omega-3) and monounsaturated fatty acids (MUFAs) as antithrombogenic, with n3 fatty acids providing the greatest effect (Ulbricth & Southgate, 1991). The IA relates pro- and antiatherogenic acids. IT and IA indicate the potential for stimulating platelet aggregation, i.e., the lower the IT and IA values, the greater the antiatherogenic fatty acid content in a given oil/fat and, consequently, the greater the potential of preventing coronary diseases (Turan, Sönmez, & Kaya, 2007).
This study aimed to evaluate the centesimal composition, fatty acid profile and lipid nutritional quality indices of mussel, octopus and two shrimp species collected in Santa Catarina (Brazil).
Material and methods
Sampling
About 3 kg of each of the following species were randomly collected in the city of Itajaí, Santa Catarina State, Brazil: pink shrimp (Penaeus brasiliensis), Pacific white shrimp (Litopenaeus vannamei), octopus (Octopus vulgaris) and brown mussel (Perna perna). The species were sanitised, homogenised in a processor and frozen at -18.0°C, until analysis.
Centesimal composition
The centesimal composition was determined according to the official methods of the Association of Official Analytical Chemists (AOAC, 2016). The moisture content was evaluated by oven drying at 105ºC for 4h, and the ash content by incineration in a muffle at 550°C, until constant weight. The protein content was determined by the semi-micro Kjeldahl method, and the total lipid content was performed according to the method described by Bligh and Dyer (1959).
Esterification and transesterification of total lipids
The total fatty acids were methylated as described by Hartman and Lago (1973). Approximately 0.050 g of total lipids were added to a esterification tube, then 500 μL of a standard solution of 1,000 mg mL-1 methyl tricosanoate (23:0) was added as an internal standard in isooctane. The solvent was removed with a flow of gaseous N2. Next, 4.00 mL of 0.5 mol L-1 methanolic sodium hydroxide was added to the tube, heated at 100°C for 5 min. and, then, cooled to room temperature. Next, 5.0 mL of an esterifying reagent solution (ammonium chloride and sulfuric acid solution in methanol) was added to the tube, which was heated at 100°C for an additional 5 min. After cooling, 5.0 mL of a saturated sodium chloride solution was added with stirring for 30 s. Finally, 2.0 mL of isooctane was added and the mixture stirred for 30 s. The esterified sample was refrigerated until separation of the phases. The supernatant was collected and transferred to a vial for further chromatographic analysis.
Chromatographic conditions
Fatty acid methyl esters (FAME) were separated on a Thermo Trace Ultra gas chromatograph, equipped with a flame ionisation detector and a fused silica 7420 capillary column CP-Select for FAME (100 m × 0.25 mm i.d., 0.25 μm cyanopropyl). The gas flow rates were 1.2 mL min.-1 for the carrier gas (H2), 30 mL min.-1 for the auxiliary gas (N2) and 35 and 350 mL min.-1 for H2 and synthetic air, respectively, to the detector flame. The volume injected was 2.0 μL, using a split of 1:80. The injector and detector temperatures were 240°C. The column temperature was held at 165°C for 7 min., then increased to 185 at 4°C min.-1 and held for 3 min. and, finally, heated to 235 at 6°C min.-1 and held for 1.67 min., totalling 25 min. of analysis. Retention times and peak areas of the FAME were determined using the ChromQuest™ 5.0 software. Fatty acids were identified by comparing their retention times with Sigma 189-19 standard PUFA mix (USA) of known composition. The absolute quantification of FAME was performed through internal standardisation, and the calculations were done as described by Joseph and Ackman (1992). Theoretical correction factors were used to determine the concentration values (Visentainer, 2012). The concentration of fatty acids in the samples was calculated in mg 100 g-1sample, using Equation 1.
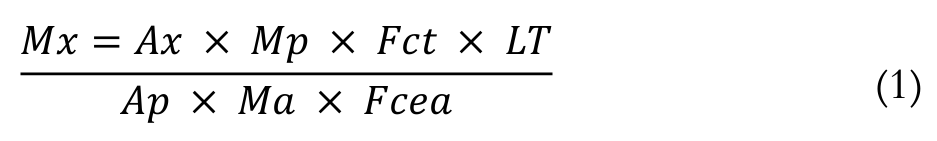 (1)
(1)where: Mx is the mass of the fatty acid X (mg 100 g-1 of sample), Ax is the peak area of the fatty acid X, Ap is the peak area of the internal standard (23:0), Mp is the mass of the internal standard added to the sample (mg), Ma is the mass of the sample (g), Fct is the theoretical correction factor of the flame ionisation detector, Fcea is the conversion factor of methyl ester to fatty acid, and LT is the total lipid in the sample.
Nutritional quality indices of lipids
The IA was calculated by the following Equation 2 (Ulbricth & Southgate, 1991):
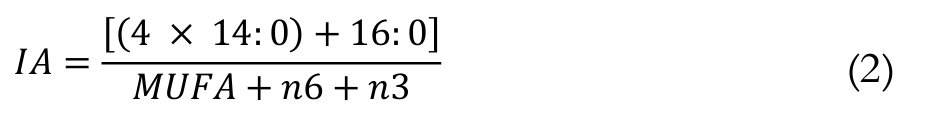 (2)
(2)The IT was calculated, as shown in Equation 3 (Ulbricht & Southgate, 1991).
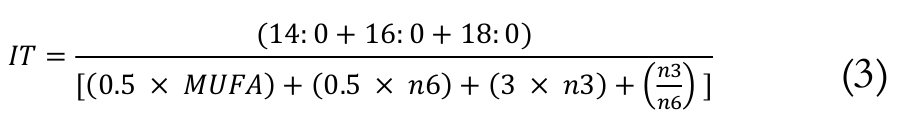 (3)
(3)The h/H ratio was determined by the following Equation 4 (Santos-Silva et al., 2002):
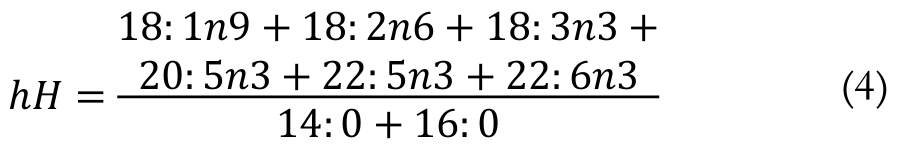 (4)
(4)Statistical analysis
The data were evaluated according to descriptive statistics methods, calculating the mean values and standard deviation for all variables. All analyses were performed in triplicate. The data were submitted to unidirectional analysis of variance (ANOVA), and the means were compared by Tukey’s test. The level of significance was 5% (p < 0.05). Data were processed using the Statistica software, version 10.0.
Results and discussion
Centesimal composition
As shown in Table 1, the centesimal composition differed between the species (Table 1). In general, moisture was the major component, followed by crude protein.
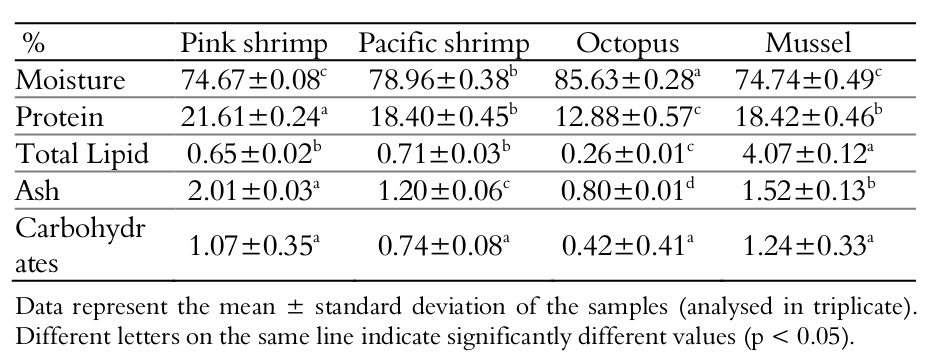
The Pink shrimp presented the highest protein content of all studied species. The pacific white shrimp and mussel also presented protein content higher than 18%. Seafood is a source of proteins of high biological value (Germano & Germano, 2008), has all the essential amino acids and has high content of lysine, an amino acid initiator of the digestive process. The digestibility is high, above 95%, due to the minimal amount of connective tissue (Oetterer, Regitano-D´arce, & Spoto, 2006). Regarding the total lipids content, mussel showed the highest value (4.07%), being categorised as semi-fat according to Ackman (1989) (4-8% of fat). The other species can be considered as lean, since they showed total lipids content lesser than 2% (Ackman, 1989). The consumption of these seafood species in a regular diet, especially when replacing others animal protein sources, shows advantages given the lower relative content of lipids, allowing them to be used in diets with restriction of lipids and cholesterol (Rosa, Pereira, & Nunes, 2004).
According to Table 1, the total lipids of the Penaeus brasiliensis and Litopenaeus vannamei shrimps were 0.65 and 0.71%, respectively. These values are close to those found by other researchers, such as Bragagnolo and Rodriguez-Amaya (1997), who reported 0.8-1.1% of total lipid in pink shrimp (P. brasiliensis) in natura. Similarly, Moura and Tenuta-Filho (2002) found 1.0-1.4% total lipids in native Brazilian shrimp. The moisture contents for L. vannamei (78.96%) and P. brasiliensis (74.67%) are comparable to those verified by Sriket, Benjakul, Visessanguan, and Kijroongrojana (2007), who observed average values of 77.21% for L. vannamei and Moura, Torres, and Mancini-Fillho (2002), who documented 78%. The P. brasiliensis and L. vannamei presented significantly different protein contents (p < 0.05), however, the values were consistent with that found in L. vannamei by Sriket et al. (2007), of 18.8%.
The moisture (74.74%) and ash (1.52%) contents detected in P. perna mussel species were lower than those found by Furlan, Galvão, Salan, and Oetterer (2007) and Cordeiro, Lopes, Oetterer, Porto, and Galvão (2007), of 83.8 and 85% for moisture and 1.8 and 3.8% for ash, respectively. Conversely, the crude protein (18.42%) and total lipids (4.07%) were higher than those verified by these same previous studies, which reported a corresponding 9.1 and 7.2% for protein and 1.1 and 1.1% for lipids, respectively. Such difference may be associated with removal of the shell, a common pre-baking process, or opening of the shells by cutting the adductor muscle. Both procedures decrease the moisture content and increase the crude protein and total lipid values.
The O. vulgaris showed different values of centesimal than those reported by Vaz-Pires, Seixas, and Barbosa (2004) and Mendes, Silva, Anacleto, and Cardoso (2011). Vaz-Pires et al. (2004), when working with octopus stored on ice, found moisture contents (79.80%), crude protein (16.30%), ash (1.86%) and total lipids (0.56%) lower than those found in the current study. Conversely, Mendes et al. (2011), analysing the samples under the same conditions, found results higher than those reported in Table 1.
Fatty acids profile
Table 2 shows the fatty acids profile of the studied species. The results are expressed in percentage of the relative area for the comparison to other authors and as mg of fatty acids per 100 g of sample, to inform about the nutritional properties of the four types of seafood evaluated.
Studies suggest that diets rich in saturated fatty acids are not healthy, the recommendation of fat consumption for the maintenance of good health is the reduction in the consumption of saturated fatty acids and consumption of mono and polyunsaturated fatty acids (Costa & Borém, 2003). Typically, fish species have a low content of saturated fatty acids in comparison to unsaturated fatty acids (Simopoulos, 1997). Similar results were observed in the fatty acids profile of the four species studied (Table 2). The saturated fatty acids (SFA) content ranged from 21.16-36.98%, whereas monounsaturated fatty acids (MUFA) varied between 21.90 and 22.92% and polyunsaturated fatty acids (PUFA) ranged from 23.26 – 44.02%.
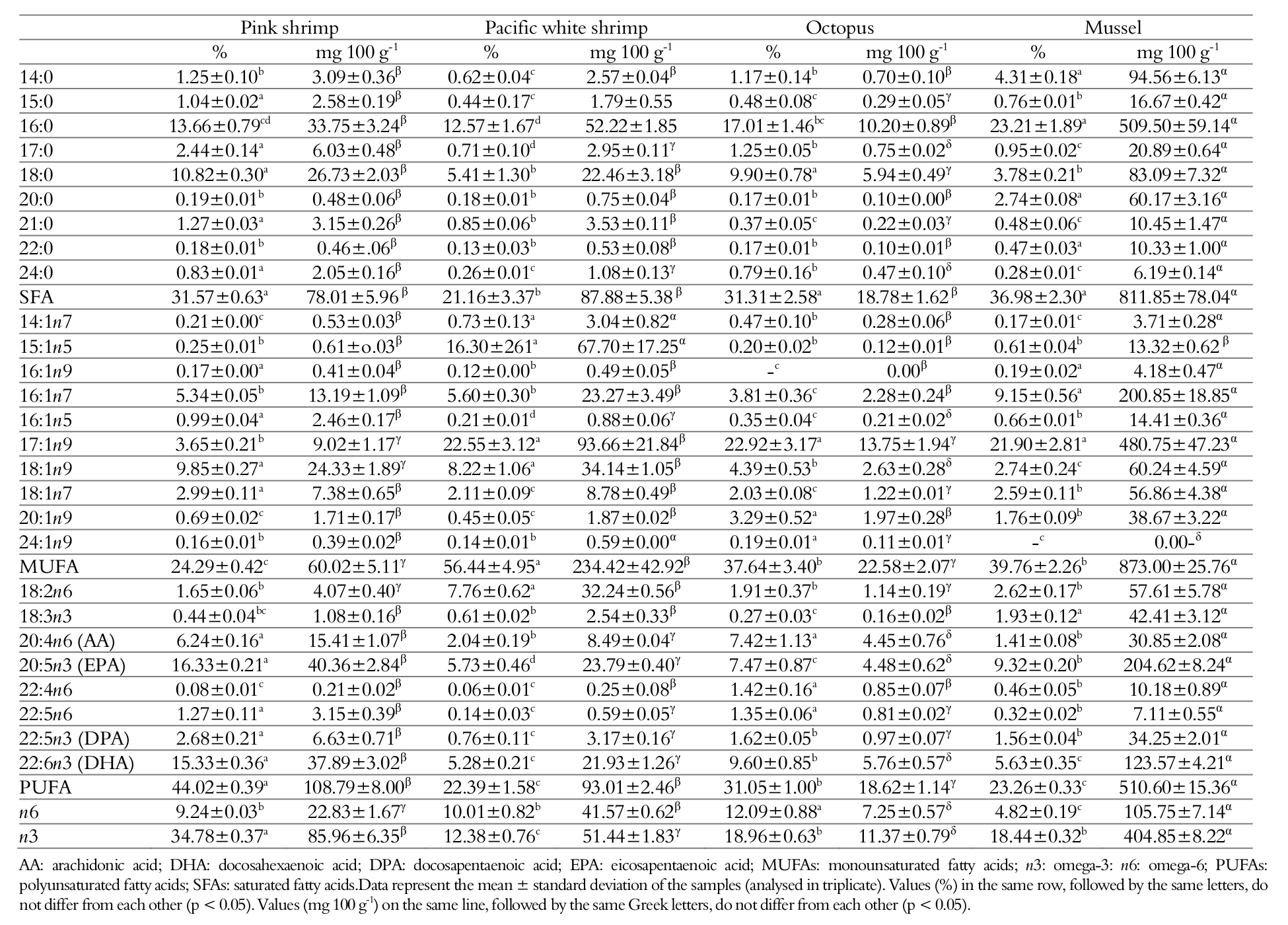
The lower content of SFA compared to unsaturated fatty (MUFA + PUFA) acids is beneficial, since the SFAs are directly related to increase in coronary disease as reported by Hu et al. (1999), and the high intake of myristic (14:0), palmitic (16: 0), and lauric acid (12:0), increase cholesterol synthesis and support the retainment of low density lipoproteins - LDL (Mensink, Zock, Kester, & Katan 2003).
Heptadecenoic acid (17:1n9) was the main MUFAs among three of the four species studied, presenting a variation between 21.90 and 22.92%, In general, high concentrations of MUFA are important because they have antithrombogenic and antiatherogenic functions, helping to prevent cardiovascular diseases (Ulbricth & Southgate, 1991). The oleic acid (18:1n-9) was the most plentiful MUFA in Pink Shrimp and the second in the other species. This fatty acid is used as primary source of metabolizable energy during fast growing (Lira et al., 2005).
The most abundant PUFAs were mainly of the n3 type. Marine animals accumulate these compounds, mainly EPA and DHA, through the trophic chain, with algae and phytoplankton being the primary producers (Nunes, Bandarra, & Batista, 2003; Turan et al., 2007). In relation to the percentage, EPA was the most abundant omega-3s in Pink Shrimp, Pacific white shrimp and mussel followed by DHA, while octopus had a higher DHA content than EPA. Normally, the DHA composition is related to the predatory activity (crustaceans, molluscs and larvae for example), indicating that the octopus has a predominantly carnivorous feeding, and the EPA composition is related more to the consumption of plankton (Bandarra, Batista, Nunes, & Empis, 2001). Literature studies show that omega-3 fatty acids are capable of improve cardiac hemodynamic factors, such as blood pressure, left ventricular diastolic filling, heart rate and endothelial function (Adkins & Kelley, 2010).
The ANVISA Resolution n° 54 of 12 November 2012 regulates the supplementary nutritional information on food labels. This law indicates that to be considered as a source of omega-3, the food must contain at least 40 mg of EPA + DHA in 100 g. Furthermore, to be considered high in omega-3, the food must contain at least 80 mg of EPA + DHA in 100 g. Thus, through this study, it was verified that Pink Shrimp and Pacific White Shrimp, can be considered sources of omega-3 (78.25 and 45.72 mg 100 g-1, respectively), whereas the mussel can be regarded as a food with high omega-3 content (328.19 mg 100 g-1).
The arachidonic acid (AA, 20:4n-6) was predominant in the n-6 family in all studied species. The mussel showed the highest concentration (30.85 mg 100 g-1), followed by pink shrimp (15.41 mg 100 g-1). This fatty acid is derived from the desaturation/elongation of linoleic acid (18:2n-6) and is the major component of membrane lipids (phospholipids), acting as an eicosanoid precursor (Wang et al., 2006).
SFAs (31.31%) and PUFAs (31.05%) found in O. vulgaris were similar to those presented by Vasconcelos and Zapata (2010), ranging from 25.13-30.76 and 23-58-31.43%, respectively. However, the level of MUFAs (37.64%) was higher than that reported by the same authors (7.50-14.99%). In the current study, O. vulgaris presented a significant amount (7.42%) of AA (20:4n6). Likewise, Miliou, Fintikaki, Tzitzinakis, Triantaphyllos, and Verriopoulos (2006) and Navarro and Villanueva (2003) found octopus containning 2.4-9.6 and 2.8-5.6% AA, respectively.
The results obtained for P. perna mussel, indicated a predominance of palmitic (16:0; 23.21%), heptadecenoic (17:1n9; 21.90%), EPA (20:5n3; 9.32%), palmitoleic (16:1n7; 9.15%) and DHA (22:6n3; 5.63%) fatty acids. The high n3 values are favorable, considering that these fatty acids are important in the prevention of cardiovascular diseases. Moreover, according to the Agência Nacional de Vigilância Sanitária (ANVISA, 2012), the values of EPA + DHA obtained (328.19 mg 100 g-1) indicate that this species has a high omega-3 content (> 80 mg 100 g-1). Lira et al. (2004) studied various mollusc species from the Northeast coast of Maceió in Alagoas, Brazil. These authors reported that the predominant fatty acids in Mytella falcata mussel and stout razor clam (Tagelus plebeius) were palmitic (16:0), palmitolenic (16:2n7), oleic (18:1n9), DHA (22:6n3) and EPA (20:5n3). The values are similar to those found in this study for P. perna mussel.
Evaluating the results of table 2, in both shrimp species, the predominant SFA was the palmitic, followed by stearic. The high percentages of palmitic acid (16:0; 14.9%) found in this study were also observed by Bragagnolo and Rodriguez-Amaya (1997) in pink shrimp and Pacific white shrimp.
PUFA values were 44.02% for pink shrimp and 22.39% for Pacific white shrimp. Moura et al. (2002) observed 41% PUFAs in refrigerated commercial samples of pink shrimp (P. brasiliensis and P. paulensis), while Bragagnolo and Rodriguez-Amaya (1997) reported 45%, in average. Pink shrimp presented high levels of EPA (20:5n3; 16.39%) and DHA (22:6n3; 15.33%), summing 31.72%. The values of EPA (5.73%) and DHA (5.28%) were lower for Pacific white shrimp. In shrimp, the ability to synthesize DHA and EPA is limited, therefore, the contribution of these constituents comes from diet (Ying, Yang, & Zhang, 2006). The studies carried out in Brazil have shown that the meat of fish from captivity has lower levels of omega-3 in relation to the fish obtained by extractivism (Carbonera et al., 2014). Thus, the differences in omega 3 content in the studied shrimp species may indicate that the Pacific white shrimp was probably bred in captivity, whereas the pink shrimp are derived from extractivism.
Nutritional quality of the lipid fraction
The nutritional quality of the lipid fraction was evaluated by different indexes and the results are shown in Table 3.
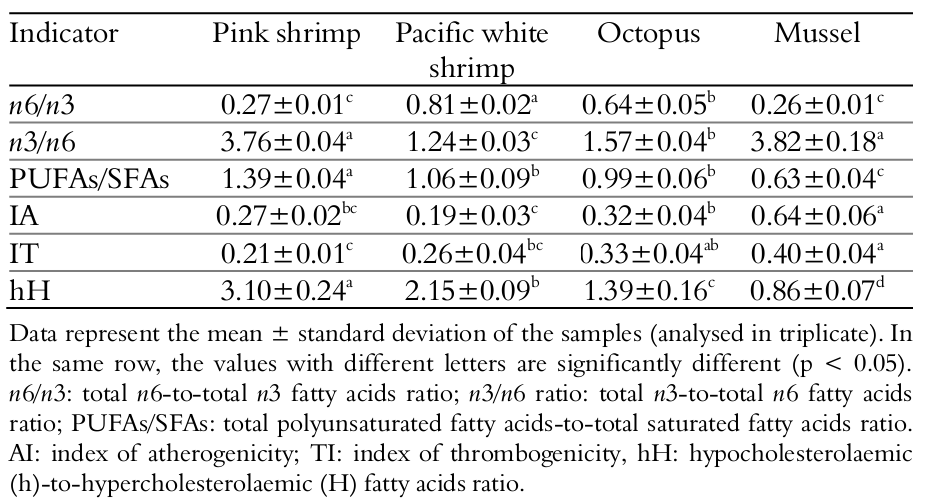
Simopoulos (2008) reported that a diet with an n6/n3 ratio of up to 4.0 is associated with a 70% reduction in death from coronary heart disease. Furthermore, an n6/n3 ratio of 2.5 is associated with a reduction of rectal cell proliferation in colorectal cancer patients, and a ratio between 2.0 to 3.0 is linked to suppression of inflammation in patients with rheumatoid arthritis (Simopoulos, 2008). According to Pigott and Tucker (1990), the n3/n6 ratio is a useful indicator for comparing the nutritional value of different species. Goodstine et al. (2003) confirmed that an increased n3/n6 ratio in the diet might reduce the risk of breast cancer, particularly in premenopausal women. According to Simopoulos (2006), the ideal n3/n6 ratio present in the diet should be equal to or greater than 1. All species studied herein had an n3/n6ratio (1.24-3.82) and the n6/n3ratio (0.26-0.81) was less than 4.0. Therefore, these species can be considered sources of good quality lipids.
Foods with a PUFAs/SFAs ratio below 0.45 have been considered undesirable in the diet because of their potential in inducing blood cholesterol increase (Departament of Health and Social Security, 1994). In all species studied, this ratio was higher than 0.45. In the mussel, the PUFAs/SFAs ratio was 0.63, which was close to the values described in three mollusc species (0.61 on average) studied by Lira et al. (2004). The pink shrimp and Pacific white shrimp presented a PUFAs/SFAs ratio of 1.39 and 1.06, respectively, in line with the advice provided by the Departament of Health and Social Security (1994). In several other shrimp species, the PUFA/SFA ratio ranged from 1.2-1.5 (Moura et al., 2002; Bragagnolo & Rodriguez-Amaya, 1997).
The indexes of atherogenicity and thrombogenicity and the relation between hypocholesterolemic and hypercholesterolemic fatty acids are used to evaluate the food nutritional value according to each fatty acid. There is no recommended value considered more favorable to human health (Sousa Bentes, Souza, Simões, & Mendoça, 2009). According to Table 3 both shrimp species showed the least values of IA and IT, respectively (0.27 and 0.21 Pink Shrimp) and (0.19 and 0.26 Pacific White Shrimp), whereas octopus (0.32 and 0.33) and mussel (0.64 and 0.40) showed higher values. All studied species are considered to potentially prevent diseases, since its IA and IT values were lower than one (<1), indicating that the influence of monounsaturated fatty acids and polyunsaturated fatty acids are higher than the influence of saturated fatty acids.
Hypocholesterolemic fatty acids act to reduce LDL (low density lipoprotein), thereby, preventing cardiovascular disease. Saturated fatty acids, which are part of the hypercholesterolemic group, increase blood cholesterol levels by reducing the activity of LDL cholesterol receptor, thus reducing LDL clearance in the bloodstream. There is no recommended value in literature for the index h/H for fish products, the value 2.0 assigned to meat products is considered as a reference (Santos-Silva et al., 2002). From a nutritional perspective, h/H values >2.0 correspond to products with a desirable fatty acid composition, considering they are composed, for the most part, of hypocholesterolaemic fatty acids and, consequently, reduce the risk of cardiovascular disease.
Hence, the shrimp offered the best consumption options. Moreover, pink shrimp and Pacific white shrimp presented h/H > 2.0, showing a better nutritional quality of the composition of fatty acids.
Conclusion
All the studied species presented a high protein content and low total lipid. Moreover, all the species contained MUFA + PUFA > SFA, with DHA and EPA, as the main PUFAs. The mussel can be considered as a high omega-3 content, while both shrimp species can be classified as a source of omega-3. The indices of nutritional quality indicated that all studied species have the potential to improve health, highlighting both shrimp species, Penaeus brasiliensis and Litopenaeus vannamei.
Acknowledgements
The authors thank the support of the State University of Maringá, in enabling the accomplishment of this research.
References
Ackman, R. G. (1989). Nutritional composition of fats in seafoods. Progress in Food and Nutrition Science, Oxford, 13(1), 161-241.
Adkins, Y., & Kelley, D. S. (2010). Mechanisms underlying the cardioprotective effects of omega-3 polyunsaturated fatty acids. The Journal Nutritional Biochemistry, 21(9), 781-792. doi: 10.1016/j.jnutbio. 2009.12.004
Agencia Nacional de Vigilância Sanitária [ANVISA]. (2012). Ministério da Saúde. Resolução n° 54. Dispõe sobre o Regulamento Técnico sobre Informação Nutricional Complementar. Diário Oficial da União, Brasília, DF.
Association of Official Analytical Chemistry [AOAC]. (2016). Official methods of analysis of the Association of Official Analytical Chemistry (20th ed.). Washington, DC: AOAC.
Bligh, E. G., & Dyer, W. J. (1959). A rapid method of total lipid extraction and purification. Canadian Journal of Biochemistry and Physiology, 37(8), 911-917. doi:10.1139/o59-099
Bandarra, N., Batista, I., Nunes, M., & Empis, J. E. (2001).Seasonal variation in the chemical composition of horse-mackerel (Trachurus trachurus), European Food Research and Technology, 212(5), 535-539. doi: 10.1007/s002170100299
Bragagnolo, N., & Rodriguez-Amaya, D. (1997). Otimização da determinação de colesterol por CLAE e teores de colesterol, lipídio total e ácidos graxos em camarão rosa (Penaeus brasiliensis). Ciência e Tecnologia de Alimentos, 17(3), 275-280. doi: 10.1590/S0101-20611997000300016
Calder, P. C., & Yagoob, P. (2009). Ômega – 3 polyunsaturated fatty acids and human health outcome. Biofactors, 35(3), 266-272. doi: 10.1002/ biof.42
Carbonera, F., Bonafe, E. G., Martin, C. A., Montanher, P. F., Ribeiro, R. P., Figueiredo, L. C., ... Visentainer, J. V. (2014). Effect of dietary replacement of sunflower oil with perilla oil on the absolute fatty acid composition in Nile tilapia (GIFT). Food Chemistry, 148(1), 230-234. doi: 10.1016/j.foodchem.2013.10.038
Cordeiro, D., Lopes, T. G. G., Oetterer, M., Porto, E., & Galvão, J. A. (2007). Qualidade do mexilhão Perna perna submetido ao processo de cocção, congelamento e armazenamento. Boletim CEPPA, 25(1), 165-179. doi: 10.5380/cep.v25i1.8404
Costa, N. M. B., & Borém, A. (2003). Biotecnologia e Nutrição (214p.). São Paulo, SP: Editora Nobel.
Food and Agriculture Organization [FAO]. (2004). Organização Das Nações Unidas Para Agricultura E Alimentação. Consumo per capita de peixe cresce no Brasil. Retrieved on Sep 15, 2014 from www.onu.org.br/consumo-per-capita-de-peixes-cresce-no-brasil-diz-fao
Goodstine, S. L., Zheng, T., Holford, T. R., Ward, B. A., Carter, D., Owens, P. H., & Mayne, S. T. (2003). Dietary (n-3)/(n-6) fatty acid ratio: Possible relationship to premenopausal but not post menopausal breast cancer risk in U.S. women. Journal of Nutrition, 133(5), 1409-1414. doi:10.1093/jn/133. 5.1409
Furlan, E. F., Galvão, J. A., Salan, E. O., & Oetterer, M. (2007). Estabilidade físico-química e mercado do mexilhão (Perna perna) cultivado em Ubatuba – SP. Ciência e Tecnologia de Alimentos, 27(3), 516-523. doi: 10.1590/S0101-20612007000300015
Germano, P. M. L., & Germano, M. I. S. Higiene e vigilância sanitária de alimentos. (3a ed.). São Paulo, SP: Manole; 2008.
Hartman, L., & Lago, R. C. (1973). Rapid preparation of fatty acid methyl esters from lipids. Laboratory Practice, 22(8), 475-476.
Departament of Health and Social Security [DHSS] (1994). Nutritional aspects of cardiovascular disease. Reporton on Health and Social Subjects, London: HMSO, 46(1), 178.
Hoffmire, C. A., Block, R. C., Thevenet-Morrison, K., & Wijngaarden, E. V. (2012). Associations between omega-3 poly-unsaturated fatty acids from fish consumption and severity of depressive symptons: An analysis of the 2005-2008 National Health and Nutrition Examination Survey. Prostaglandins, Leukotrienes & Essential Fatty Acids, 86(4-5), 155-160. doi: 10.1016/j.plefa.2012.03.003
Hu, F. B., Stampfer, M. J., Manson, J. E., Ascherio, A., Colditz, G. A., Speizer, F. E., ... Willett, W. C. (1999). Dietary saturated fats and their food sources in relation to the risk of coronary heart disease in women. The American Journal of Clinical Nutrition, 70(6), 1001-1008. doi: 10.1093/ajcn/70.6.1001
Joseph, J. D., & Ackman, R. G. (1992). Capillary column gas-chromatographic method for analysis of encapsulated fish oils and fish oil ethyl-esters Collaborative study. Journal of AOAC International, 75(3), 488-506.
Lira, G. M., Mancini Filho, J., Tores, R. P., Oliveira, A. C., Vasconcelos, A. M. A., Omena, C. M. B., & Almeida, M. C. S. (2005). Composição centesimal, valor calórico, teor de colesterol e perfil de ácidos graxos da carne de búfalo (Bubalis bubalis) da cidade de São Luiz do Quitunde-AL. Instituto Adolfo Lutz, 64(1), 31-38.
Lira, G. M., Mancini Filho, J., Sant’ana, L. A., Torres, R. P., Oliveira, A. C., Omena, C. M. B., & Silva Neta, M. L. (2004). Perfil de ácidos graxos, composição centesimal e valor calórico de moluscos crus e cozidos com leite de coco da cidade de Maceió, AL. Revista Brasileira de Ciências Farmacêuticas, 40(4), 1-11. doi: 10.1590/S1516-93322004000400010
Me´Dale, F., Lefe`Vre, F., & Corraze, G. (2003). Qualite´ nutritionnelle et die´te´tiquedespoissons, constituants de lachair et facteurs de variations. Cahiers de Nutrition et de Die´ te´tique, 38(1), 37-44. doi: CND-02-2003-38-1-0007-9960-101019-ART4
Mendes, R., Silva, H. A., Anacleto, P., & Cardoso, C. (2011). Effect of CO2 dissolution on the shelf life of ready to eat Octopus vulgaris. Innovative Food Science and Emerging Technologies, 12(4), 551-561. doi: 10.1016 /j.ifset.2011.07.003
Mensink, R. P, Zock, P. L, Kester, A. D., & Katan, M. B. (2003). Effects of dietary fatty acids andcarbohydrates on the ratio of serum total to HDL cholesterol and on serum lipids andapolipoproteins: a meta-analysis of 60 controlled trials. American Journal of Clinical Nutrition, 77(5), 1146-1155. doi: 10.1093/ajcn/77.5.1146
Miliou, H., Fintikaki, M., Tzitzinakis, M., Triantaphyllos, K., & Verriopoulos, G.(2006). Fatty acids composition of the common Octopus vulgaris, in relation to rearing temperature and body weight. Aquaculture, 256(1-4), 311-322. doi: 10.1016/j.aquaculture.2006.02.050
Mohamad, L. A., Baharuldin, T. H., Moklas, M. A. M., Fakurazi, S., & Muhammad, S. I. (2014). Antidepressant-like effects of omega-3 fatty acid in postpartum model of depression in rats. Behavioral Brain Research, 271(1), 65-71. doi: 10.1016/j.bbr. 2014.05.036
Moura, A. F. P., Torres, R. P., & Mancini-Filho, J. (2002). Caracterização da fração lipídica de amostras comerciais de camarão-rosa. Archivos Latinoamericanos de Nutrición, 52(2), 207-211.
Navarro, J. C., & Villanueva, R. (2003). The fatty acid composition of Octopus vulgaris paralarvae reared with live and inert food: deviation from their natural fatty acid profile. Aquaculture, 219(1-4), 613-631. doi: 10.10 16/S0044-8486(02)00311-3
Nunes, M., Bandarra, N., & Batista, I. (2003). Fish products: contribution for a healthy food. Electronic Journal of Environmental Agriculture Food Chemistry, 2(4), 453-457.
Oetterer, M., Regitano-D´arce, M. A. B., & Spoto, M. H. F. (2006). Fundamentos de ciência e tecnologia de alimentos. Barueri, SP: Manole.
Pigott, G. M., & Tucker, B. W. (1990). Effects of technology on nutrition. New York, NY: Marcel Decker.
Rosa, R., Pereira, J., & Nunes, M. L. (2004). Nutritional quality os red shrimp, Aristeus antennatus (Risso), Pink shrimp, Parapenaeus longirostris (Lucas) and Norway lobster, Nephrops norvegicus (Linnaeus). Journal of the Science of Food and Agriculture, 84(1), 89-94. doi: 10.10 02/jsfa.1619
Santos-Silva, J., Bessa, R. J. B., & Santos-Silva, F. (2002). Effect of genotype, feeding system and slaughter weight on the quality of light lambs: Fatty and composition of meat. Livestock Production Science, 77(2), 187-194. doi: 10.1016/S0301-6226(02)00059-3
Simopoulos, A. (1997). Nutritional aspects of fish. In J. B. Luten, T. Borresen, J. Oehlenschläger (Eds.), Seafood from producer to consumer (p. 589-607). Amsterdam, Elsevier Science
Simopoulos, A. (2006). Evolutionary aspects of diet, the omega-6/omega-3 ratio and genetic variation: nutritional implications for chronic diseases. Biomedicine & Pharmacotherapy, 60(9), 502-507. doi: 10.1016/j.biopha.2006.07.080
Simopoulos, A. (2008). The importance of the omega-6/omega-3 fatty acid ratio in cardiovascular disease and other chronic diseases. Experimental Biology and Medicine, 233(6), 674-688. doi: 10.3181/0711-MR-311
Sousa Bentes, A., Souza, H. A. L, Simões, M. G., & Mendonça, X. M. F. (2009). Caracterização física e química e perfil lipídico de três espécies de peixes amazônicos. Revista Brasileira de Tecnologia Agroindustrial, 3(2), 97-108. doi: 10.3895/S1981-36862009000200011
Souza, H. A. L., Bentes, A. S., Simões, M. G., & Fontenelle, M. J. P. (2008). Caracterização física e nutricional de três espécies de peixes amazônicos. Revista Brasileira de Tecnologia Agroindustrial, 4(2), 141-152. doi: 10.385/S1981-36862010000200003.
Sriket, P., Benjakul, S., Visessanguan, W., & Kijroongrojana, K. (2007). Comparative studies on chemical composition and termal properties of black tiger shrimp (Penaeus monodon) and White shrimp (Penaeus vannamei) meats. Food Chemistry, 103(4), 1199-1207. doi: 10.1016/j.foodchem.2006.10.039
Turan, H., Sönmez, G., & Kaya, Y. (2007). Fatty acid profile and proximate composition of the thornback ray (Raja clavata, L. 1758) from the Sinop coast in the Black Sea. Journal of Fisheries Sciences, 1(2), 97-103. doi: 10.3153/jfscom.2007012
Ulbricht, T. L. V., & Southgate, D. A. T (1991). Coronary heart disease: Seven dietary factors. Lancet, 338(8773), 985-992. doi: 10.1016/0140-6736(91)91846-M
Vasconcelos, M. M., & Zapata, J. F. F. (2010). Efeito sazonal sobre os ácidos graxos e colesterol do polvo Octopus vulgaris, Cuvier 1797. Ciências e Tecnologia de Alimentos, 30(2), 325-329. doi: 10.1590/S0101-20612010000200006
Vaz-Pires, P., Seixas, P., & Barbosa, A. (2004). Aquaculture potential of the common Octopus (Octopus vulgaris Cuvier, 1797): a review. Aquaculture, 238(1-4), 221-238. doi: 10.1016/j.aquaculture.2004. 05.018
Visentainer, J. V. (2012). Aspectos analíticos da resposta do detector de ionização em chama para ésteres de ácidos graxos em biodiesel e alimentos. Química Nova, 35(2), 274-279. doi: 10.1590/S0100-40422012000200008
Wang, C., Harris, W. S., Chung, M., Lichtenstein, A. H., Balk, A. M., Kupelnick, B., ... Jordan, L. (2006). n-3 Fatty acids from fish or fish-oil supplements, but not α-linolenic acid, benefit cardiovascular disease outcomes in primary- and secondary-prevention studies: a systematic review. The American Journal of Clinical Nutrition, 84(1), 5-17. doi: 10.1093/ajcn/84.1.5
Ying, X. P., Yang, W. X., & Zhang, Y. P. (2006). Comparative studies on fatty acid composition of the ovaries and hepatopancreas at different physiological stages of the Chinese mitten crab. Aquaculture, 256(1-4), 617-623. doi: 10.1016/j.aquaculture.2006.02.045
Notes
Author notes
isabellagualda@gmail.com