Ciência, Tecnologia de Alimentos e Engenharia de Alimentos
Chemical changes in Prata-Anã banana stored under reduced O2 and increased CO2 levels
Transformações químicas na banana Prata-Anã armazenada em baixas concentrações de O2 e níveis crescentes de CO2
Chemical changes in Prata-Anã banana stored under reduced O2 and increased CO2 levels
Acta Scientiarum. Technology, vol. 40, 2018
Universidade Estadual de Maringá

This work is licensed under Creative Commons Attribution 4.0 International.
Received: 02 October 2017
Accepted: 11 December 2017
Abstract: Banana has high respiratory rate, which reduces its storage time. This study evaluated the chemical characteristics of Prata-Anã banana stored under reduced O2 and increased CO2 levels, in order to slow down the ripening. The atmospheres were settled at 1, 3 and 5% O2 combined with 2, 6 and 10% CO2, with ethylene adsorption, performing nine treatments. The controlled atmosphere was also settled at high oxygen level in ambient atmosphere (20.6 O2 and 0.1% CO2) with ethylene adsorption, and it was compared to the control treatment in normal atmosphere without ethylene adsorption. The chemical evaluations were performed after 14 day (days) under controlled atmosphere and after 3 days in ambient atmosphere. It was measured the peel color, mass loss, total acidity, pH, starch content, soluble sugars, total soluble solids and total phenolic compounds. Regardless of the CO2 level, the ripening was impaired under suppressed O2 levels (1, 3, and 5%), even after 3 days in normal atmosphere, with the fruits remaining unripe, with the smallest acidity and total soluble sugars, but with the highest starch content. The fruits reached total ripening after 14 days in normal atmosphere, presenting the smallest starch content. The content of phenolic compounds was increased in fruits stored under suppressed O2 levels.
Keywords: Musa spp, controlled atmosphere, phenolic compounds, starch content, soluble sugars.
Resumo: A banana tem alta taxa respiratória que reduz seu tempo de armazenamento. Este trabalho investigou alterações químicas de bananas Prata-Anã estocadas em níveis reduzidos de O2 e níveis crescentes de CO2 para minimizar o amadurecimento. As atmosferas foram mantidas em 1, 3 e 5% de O2, combinadas com 2, 6 e 10% de CO2, com adsorção de etileno, totalizando nove tratamentos. Aplicou-se também atmosfera com alto teor de oxigênio (20,6 de O2 e 0,1% de CO2) e adsorção de etileno, comparando com a atmosfera do ambiente normal sem adsorção de etileno. Análises foram realizadas após 14 dias em atmosfera controlada e após 3 dias em atmosfera ambiente. Foram realizadas medidas de coloração da casca, perda de massa, acidez total, pH, conteúdo de amido, açúcares solúveis, sólidos solúveis e compostos fenólicos totais. Independentemente do nível de CO2, o amadurecimento foi paralisado em níveis limitantes de O2 (1, 3 e 5%), mesmo após 3 dias em atmosfera normal, em que os frutos permaneceram verdes e com menores conteúdos de acidez e de açúcares solúveis totais, mas com elevado conteúdo de amido. Os frutos estavam maduros após 14 dias em atmosfera normal, quando apresentaram baixo teor de amido. O conteúdo de compostos fenólicos aumentou nos frutos estocados em níveis limitantes de O2.
Palavras-chave: Musa spp, atmosfera controlada, compostos fenólicos, amido, açúcares solúveis.
Introduction
Banana cv. Prata (Musa sp., subgroup Prata) and cv. Nanicão (Musa sp., subgroup Cavendish) are the most produced banana cultivars in Brazil. The Prata cultivar has the major acceptability in the country, and the Nanicão in the world market (Nascimento Junior, Ozorio, Rezende, Soares, & Fonseca, 2008). Brazil is the third major world producer of bananas with 6,892,622 t, of which only 97,976 t were exported in 2013, mainly to the South American countries (Food and Agriculture Organizations of the United Nations [Faostat], 2013).
As a climacteric fruit, bananas are commercially picked from the tree at the mature green stage, and then shipped to distant markets, usually by air or sea. To protect mature green fruit from fast ripening, a common practice is to pre-cool fruit to 13ºC and maintain the low temperature during the storage period. However, refrigeration is costly, and an alternative low-cost method that delays ripening would be the application of controlled atmospheres (CA) and modified atmospheres (MA) (Imahori, Yamamoto, Tanaka, & Bai, 2013).
CA storage promotes decreasing metabolism and suppressing postharvest decay. The effectiveness of low oxygen CA in delaying fruit ripening increases with decreasing oxygen level, as long as the oxygen partial pressure does not drop below the level that engenders anaerobic fermentation, which may affect aromatic quality (Palomer, Roig-Villanova, Grima-Calvo, & Vendrell, 2005). Accumulated ethanol in fruit drives the biosynthesis of esters to ethyl esters, thus, it decreases the production of typical fruit aromas, such as butyl acetate, isobutyl acetate and isoamyl acetate, and also it causes off-flavors (Imahori et al., 2013).
The climacteric respiration of banana is suppressed under high CO2 probably due to decrease of phosphofructokinase. With the CO2 treatment, the climacteric respiration is suppressed along with decreasing contents of pyruvate, 2-oxoglutarate, and malate (Liu, Yang, Murayama, & Taira, 2004).
According to Kader, Sommer, and Arpaia (2002), Cavendish bananas cannot be stored in atmosphere with more than 5% CO2 and less than 2 to 5% O2, but the gas tolerance level is related to different cultivars. However, the tolerance of Prata banana has not yet been evaluated with respect to suppressing levels of oxygen concentrations, in association to a broad range of CO2 levels. The objective of the present study is to investigate the changes in physical and chemical composition of banana stored at ambient temperature under decreasing concentrations of O2 and increasing level of CO2, aiming to extend the storage time and quality maintenance of the fruits. The content of phenolics compounds is evaluated as indicative parameter of atmospheric stress.
Material and methods
Raw material
Bananas cv Prata-Anã [Musa spp. (AAB)] were selected at the packing house of Bananas EP Company, located in Cariacica – state Espírito Santo – Brazil (-20° 15' 59,1270'' S, -40° 25' 18,9422'' W). The fruits were harvested with 18 weeks after anthesis, presenting clear green peel without scratches and spots, with middle length pattern. Hands with ten to twelve fingers (1 kg) of uniform size were selected in the second line of the banana bunches. They were washed with 1% aluminum solution and then with water to remove latex. After draining the water, banana hands were carefully packed in plastic boxes covered with silk paper and transported to the laboratory, where the experiments were conducted 4 hours after harvest.
All the fruits were sanitized in chlorinated solution 0.01% for 15 min and air dried. Each sample was identified, weighted, evaluated by color measurements and stored in controlled atmosphere chamber (100 L).
Experimental design
The experimental arrangement was a completely randomized design (CRD) with eight replicates of composed samples (two fingers) for physical analysis, and three replicates of composed samples (five fingers) for chemical analysis. The bananas were evaluated after 14 day under controlled atmospheres, and after 3 day under normal ambient atmosphere in a chamber maintained at 23oC and 90% relative humidity (RH) (Climasul, Brazil). The atmospheres were settled at 1, 3 and 5% O2 combined with 2, 6 and 10% CO2, with ethylene adsorption, performing nine treatments. Furthermore, the controlled atmosphere was also settled at high oxygen level in ambient atmosphere (20.6 O2 and 0.1% CO2) with ethylene adsorption, and it was compared to the control treatment in normal atmosphere without ethylene adsorption.
The oxygen and CO2 concentrations were controlled by flushing with nitrogen gas and adding CO2. The chosen levels were kept constant by scrubbing excess CO2 from the respiration and adding air for the O2. Gas concentrations were monitored daily using computerized analyzers with paramagnetic (O2) and infrared (CO2) detection. Ethylene adsorption was performed by pumping the air in a permanganate column, and this technique is applied to minimize the self-catalytic effect on maturation.
The samples of the pulp were removed from the equatorial portion of the fruits using a stainless steel knife, and the collected material was involved in aluminium foil prior to immersion in liquid N2. The samples were packed in polyethylene bags and stored in freezer at – 80ºC. Prior to chemical analysis in each sampling time, the frozen pulp tissue of the banana was firstly ground to fine powder in liquid nitrogen.
Measurements of peel color and mass loss of fruits
The peel color was measured at two equidistant points on opposite sides of the middle ends of banana, with 16 replicates. The measurements were recorded with a spectrophotometer (Hunterlab MiniScan XE Plus, USA) calibrated to a standard white and black reflective plate, employing a D65 illuminant and a 10o standard observer, and the results were expressed by the average of Hunter L, a, b parameters.
The mass loss was measured after 14 day of storage in controlled atmosphere, and after 3 day under normal ambient atmosphere, with eight replicates each. It was used an electronic balance (Gehaka, model BC 2000, Brazil), with measurements normalized to 0.1 kg of fruit.
Carbohydrate determination
Starch from the frozen samples (300 mg) was dissolved in 4 mL of 0.5 N NaOH. After neutralization with 4 mL of 0.5 N acetic acid, an aliquot was precipitated with 4 mL of absolute ethanol. The precipitated starch was hydrolyzed with amyloglucosidase (EC 3.2.1.3), and the resultant glucose was determined by the glucose oxidase (EC 1.1.3.4) glucose peroxidase (EC 1.11.1.7) glucose ABTS (2,20-azinobis[3-ethylbenzthiazoline] sulfonate) system, as described by Cordenunsi and Lajolo (1995). Soluble sugars from 100 g of sample were extracted three times with 80% ethanol at 80ºC. After centrifugation, the supernatants were combined, and the ethanol was evaporated under vacuum using a speed-vac system. The residues were reconstituted with water, followed by filtration through 0.22 μm membrane filters and analyzed by high performance anion exchange chromatography coupled with pulsed amperometric detection (HPAEC-PAD-Dionex, Sunnyvale, CA, USA). The analytical column employed was a Carbopac PA1 (250×4 mm, 5 μm particle size). The mobile phase was 20 mmol NaOH with constant flow rate 1.0 mL min-1 at 30°C (Cordenunsi, Shiga, & Lajolo, 2008). Glucose, fructose and sucrose were used as commercially available standard.
Measuring amounts of soluble solids (TSS), titratable acidity and pH
The pulp (25 g) was homogenized using polytron mixer (PT 10/35, Brinkmann) and then, it was filtered through nylon fabric. The filtrate was collected to determine the total soluble solids by a digital refractometer (Krüss DR 201-95) and the results were expressed as Brix; the titratable acidity was determined by titration with standardized sodium hydroxide (NaOH), as established by the AOAC Official Method 942.15 (Association of Official Analytical Chemistry [AOAC], 1995) and it was expressed as % malic acid, and the pH was evaluated using a digital potentiometer (Metrohm 827), as established by the AOAC Official Method 981.12 (AOAC, 1995).
Quantification of total phenolics
Total phenolics were determined according to Swain and Hills (1959), using the Folin-Ciocalteu reagent. Briefly, during extraction, 0.6 g of green banana or 1 g of ripe banana were homogenized with 25 mL of 70% methanol using polytron mixer (PT 10/35, Brinkmann) and the obtained solution was maintained over a plate shaker (Variomag) in the dark at 300 rpm for 30 min at 4°C. Then, the solution was filtered to quantification. For this, into 250 uL of the filtrate, it was sequentially added 2 mL of ultrapure water, 250 uL of Folin-Ciocalteu reagent (Sigma-Aldrich) and 250 uL of a saturated solution of sodium carbonate. The tubes were heated in a water bath at 37°C for 30 min. The absorbance of each sample was read using UV-visible spectrophotometer (Hewlett Packard Agilent 8453) at 765 nm. The concentration of phenolic in the samples was obtained from a standard curve with different concentrations of gallic acid. The results were expressed as milligrams of gallic acid equivalents (GAE) per 100 g of fresh weight (FW).
Statistical analysis
Peel color and mass loss of fruits were interpreted by descriptive statistics, according to the simple random sampling method (SRS), considering an infinite population of fruits with 95% probability. The data of chemical analysis were evaluated by factorial arrangements. The means were compared using ‘t’ test of Student for confidence intervals. The results were processed by the software Sistema para Análises Estatísticas e Genéticas, version 9.0 (Sistema para Análises Estatísticas e Genéticas [SAEG], 2007).
Results and discussion
Characterization of fruit peel color
The fruits of the control treatment (under normal atmosphere) were evaluated as totally mature after 14 day of storage at 23oC, when the bananas presented the fully yellow peel. After the following 3 day of storage, the color peel indexes remained constant (Figure 1).
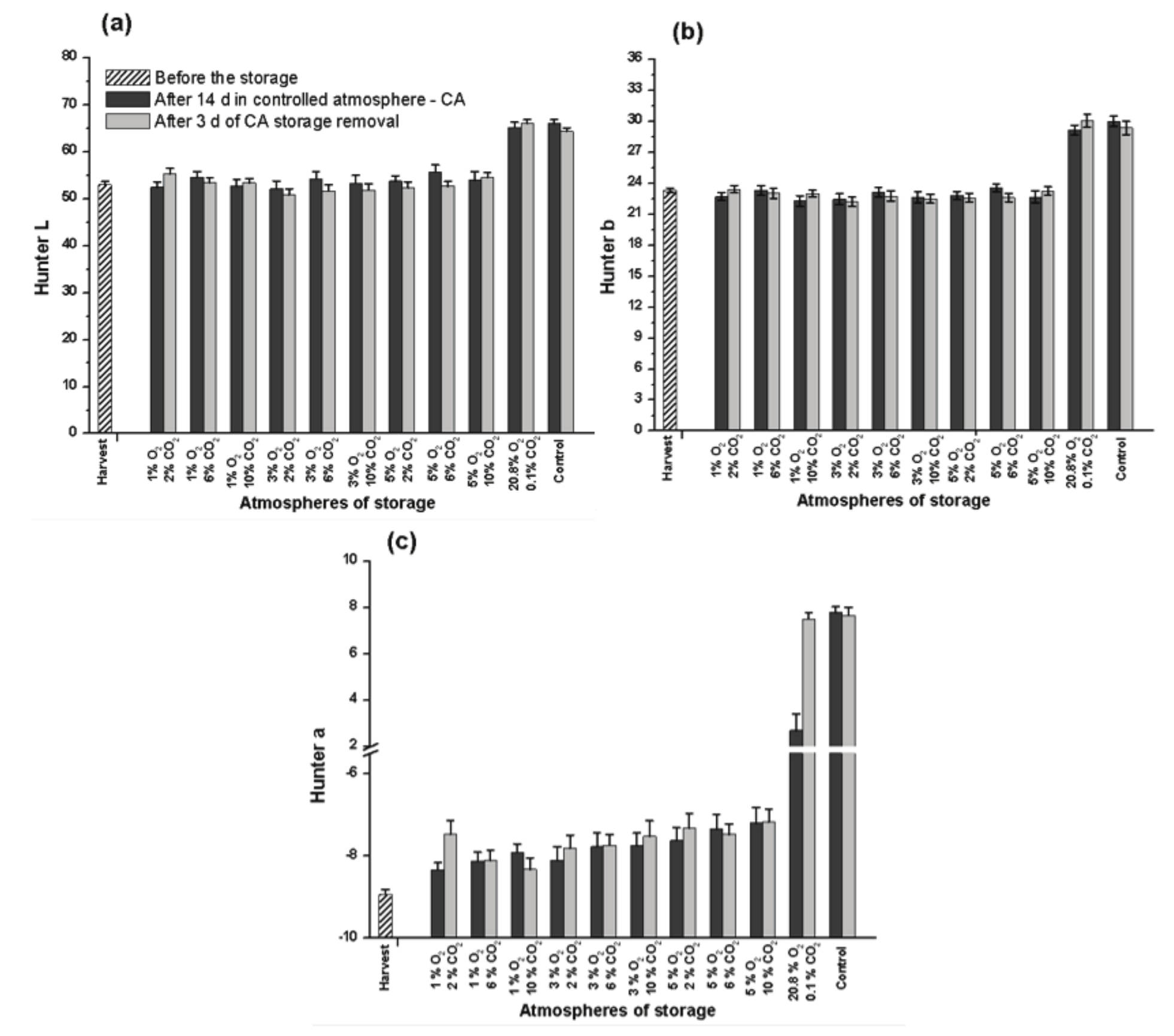
Figure 1.
Measurements of Hunter L (a), Hunter b (b) and Hunter a (c) parameters of the peel of Prata-Anã banana stored for 14 day at 23°C and 90 % RH under conditions of CA with ethylene adsorption, and after 3 day under normal atmosphere. Control treatment at ambient atmosphere. Vertical bars are indicated with confidence intervals at 5% by Student t-Test.
Regarding the bananas stored under ambient conditions of controlled atmosphere (20.8 O2 and 0.1% CO2) with ethylene adsorption, the ripening process slowly, as indicated by the smaller value of Hunter a parameter (Figure 1c). However, after 3 day under normal atmosphere, the fruits reached total ripening. Lobo, Gonzales, Pena, and Marrero (2005) also noted a direct correlation between ethylene content and ripening rate of bananas. According to Yang et al. (2009), the chlorophyll and carotenoids contents in banana peel are correlated by parameters of Hunter a and Hunter b during the ripening process.
After 14 day storage under lower O2 concentrations (1, 3 and 5%), the bananas presented color indexes like those evaluated before the storage (Figure 1a and b), with only a minimal change in green color index (Hunter a), mainly in those maintained in higher O2 level (Figure 1c). After 3 day at normal ambient atmosphere, the fruits remained green and with the same color parameters. These results are in accordance with those described by Santos, Vilas Boas, Botrel, and Pinheiro (2006), that verified the complete ripening of bananas stored under ambient atmosphere at 12.5oC for 16 day, while the fruits stored under atmosphere with 2, 3 and 4 kPa remained green.
The CO2 concentration did not change the fruit ripening in the treatments with limiting O2 levels (Figure 1), however, in the low O2 levels the ripening process may have been impaired.
Mass loss of the fruit
The fruits of the control treatment presented the higher mass loss after 14 day at 23°C. Furthermore, the mass loss was increased after 3 day of storage, reaching the final average of 6% (Figure 2).
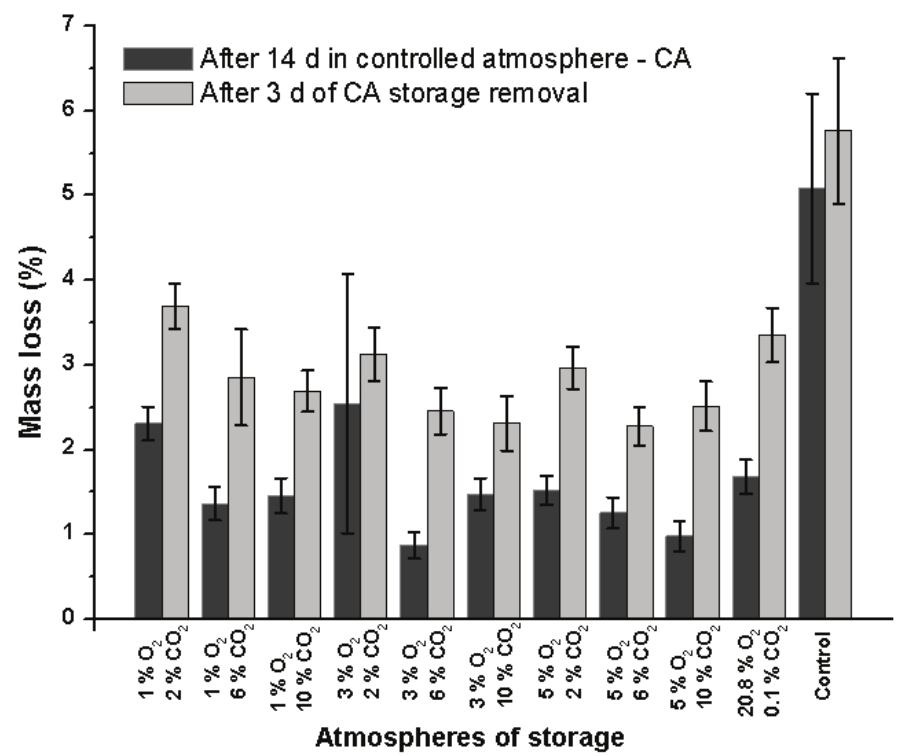
Figure 2.
Mass loss of Prata-Anã banana stored for 14 day at 23°C and 90% RH under different conditions of CA with ethylene adsorption, and after 3 day of storage in normal ambient atmosphere. Control treatment at ambient atmosphere. Vertical bars are indicated with confidence intervals at 5% by Student t-Test.
The mass loss of fruits stored for 14 day under different CAs were lower than that of the control treatment, even in fruits maintained under ambient CA (20.8 O2 and 0.1% CO2) with ethylene adsorption (Figure 2). This is probably due to the closed chamber that minimizes the water loss.
After 3 day under normal ambient atmosphere, the mass loss of the fruits increased in all the treatments; however, the final averages in these treatments were lower than that of the control treatment (Figure 2). The same behavior was noted by Santana, Benedetti, Sigrist, Sato, and Anjos (2011) in peaches stored under CA, which presented a faster rate of mass loss after 4 day of removal from controlled atmosphere.
Determination of pH and pulp acidity
The acidity of fruits in the control treatment was increased to 0.68% after 14 day under normal ambient atmosphere (Figure 3a). Nevertheless, the acidity decreased to 0.61% after the following 3 day of storage. According to Kader et al. (2002), the lowering in acidity is indicative of over ripening, that promotes consumption of organic acids as energy source. According to Zhang et al. (2011), the acidity of strawberry is increased during the ripening, however, it is declined in advanced stage of senescence.
Ramos, Leonel, and Mischan (2009) evaluated the pulp acidity and pH of Prata-Anã banana and found values similar to those found in the present study at the harvest point, with average of 0.13% acidity and pH 5.92.
The ethylene adsorption was effective in minimize the acidity change in banana along 14 day under ambient CA (20.8 O2 and 0.1% CO2), reaching 0.54% acidity (Figure 3a). Nevertheless, after 3 day under normal ambient atmosphere, its value was similar to that of the control treatment evaluated after 14 day under normal ambient atmosphere (0.67%). The same behaviour was noted for pH measurements in the pulp (Figure 3b).
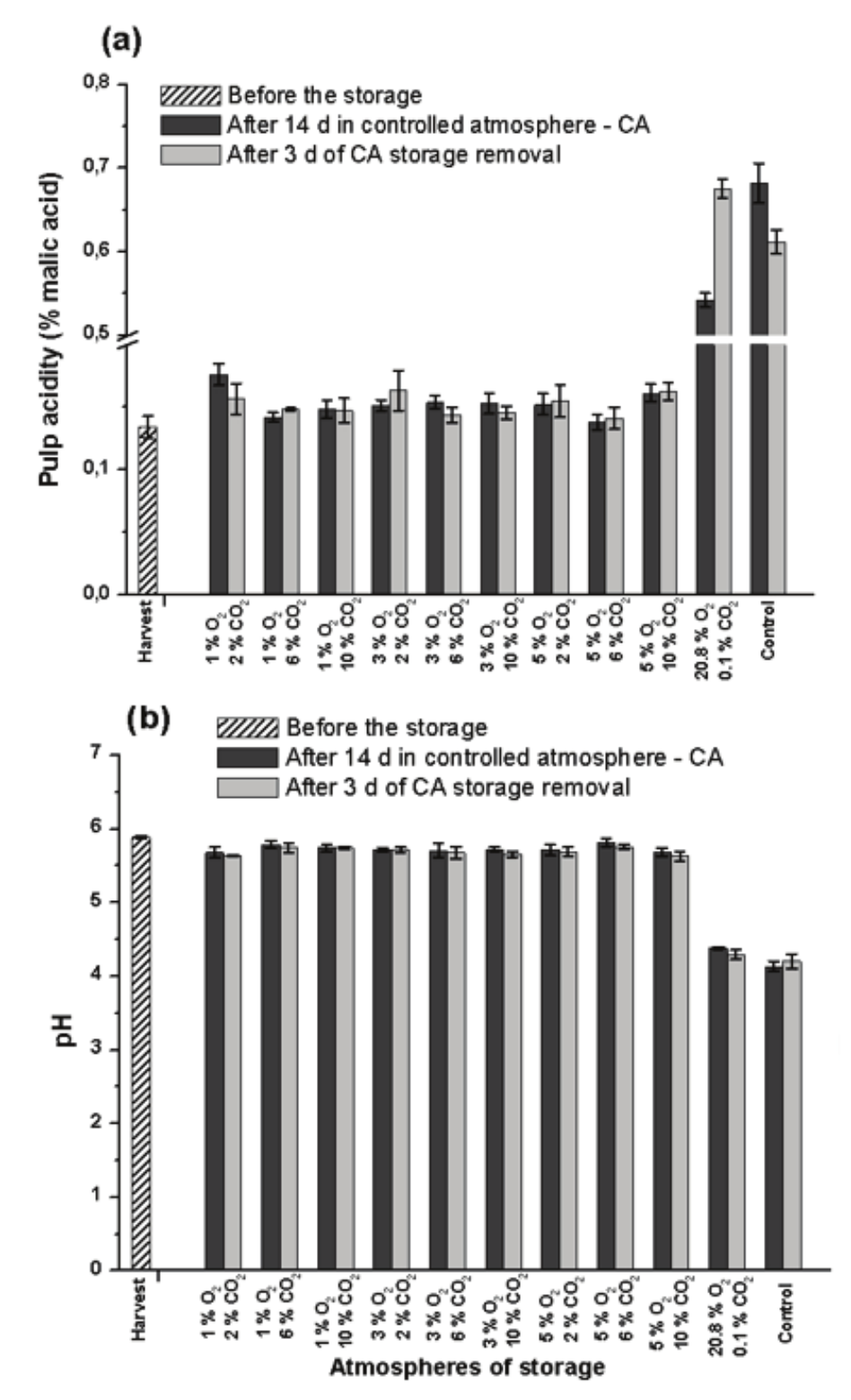
Figure 3.
Content of acidity (a) and pH (b) in pulp of Prata-Anã banana stored for 14 day at 23°C and 90% UR in different conditions of CA with ethylene adsorption, and after 3 day of storage in normal ambient atmosphere. Control treatment at ambient atmosphere. Vertical bars are indicated with confidence intervals at 5% by Student t-Test.
The fruits stored in lower O2 levels (1, 3 and 5%), in a broad range range of CO2, remained with the smaller acidity (Figure 3a), indicantaing that the ripening was sensible impaired along 14 day in CA followed by 3 day of storage in the normal atmosphere.
Content of total soluble solids, starch and soluble sugars in pulp of banana
The total soluble solids (TSS) in the pulp of banana changed from 0.5 to 22.7 oBrix in the fruits stored for 14 day under normal ambient atmosphere, however, it tends to decline to 21.8 oBrix after the following 3 day of storage (Figure 4a). Sañudo, Cepeda, Rangel and Heredita (2009) verified that TSS content in ripe banana (22 oBrix) decreased to 20 oBrix in the phase of senescence.
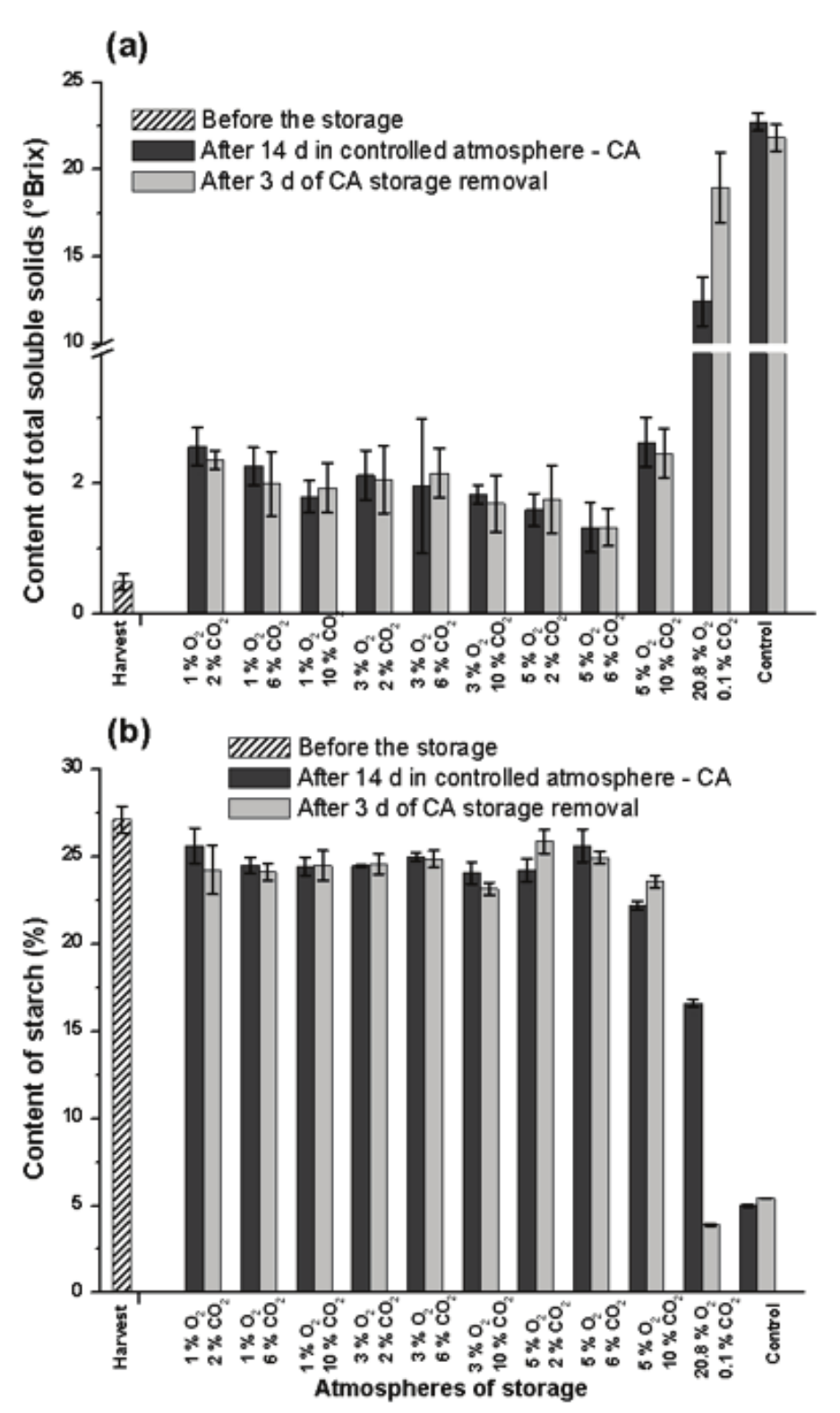
Figure 4.
Content of total soluble solids (a) and starch (b) in pulp of Prata-Anã banana stored for 14 day at 23°C and 90% UR in different conditions of CA with ethylene adsorption, and after 3 day of storage in normal ambient atmosphere. Control treatment at ambient atmosphere. Vertical bars are indicated with confidence intervals at 5% by Student t-Test.
The initial content of starch (27.13%) was metabolized along 14 day of storage under normal atmosphere and reached the smallest value of 5.01% (Figure 4b), remaining constant in further 3 day of storage, so revealing that the starch content did not change in this phase of senescence. Peroni-Okita et al. (2010) found 22 % of starch in green Nanicão banana and 5% in ripe fruits. Santos et al. (2006) also found 5.4% of starch in ripe Prata-Anã banana.
In CA ambient conditions (20.8 O2 and 0.1% CO2) with ethylene adsorption, the TSS content increases to 12.4 oBrix in 14 day of storage, and it reaches 18.9 oBrix after 3 day under normal atmosphere, with an average value lower than the control treatment. This result indicates that the fruit did not reach the highest TSS content that is typical of ripe fruits (Figure 4a). The drop in the starch content was less accentuated in fruits stored for 14 day under the same condition (Figure 4b), when compared to the control treatment. However, after 3 day under normal atmosphere, the drop in starch content was deeply and reached the lowest value (3.9%).
In CA with low O2 (1, 3 and 5%) the change in TSS content was minimized after 14 day of storage, and it remained constant after 3 day under normal atmosphere, endorsing the correlation with the minimal change in starch content (Figure 4b). Ortiz, Graell, López, Echeverría, and Lara (2010) noted a minimal change in TSS content of peaches stored in 3 kPa O2 and 10 kPa CO2 during 21 day at 0oC, and after 7 day storage in normal atmosphere at 20oC.
At the harvest point, the sucrose content of banana was 0.27%, however, after 14 day under normal ambient atmosphere it reached the higher level (11.4%) and it remained constant after 3 day in normal atmosphere (Figure 5a). The contents of glucose (Figure 5b) and fructose (Figure 5c) also increased after 14 day of storage, reaching 3.2 and 3.8%, respectively. Furthermore, it continued to increase in the following 3 day of storage and it reached 3.6 and 4.3%, respectively, serving as a direct source of sugar to sustain the metabolism of the fruit.
The content of starch in bananas drops deeply during the ripening with simultaneous increase in soluble sugars (Cordenunsi & Lajolo, 1995). The degrading of starch releases the carbon to synthesis of sucrose and volatile compounds, which are responsible for the flavor in ripe fruits (Shiga et al., 2011). Saraiva et al. (2013) also detected 20% of starch and 0.5% of soluble sugars in green Nanica banana, but reaching 5 and 13%, respectively, in ripe fruits. In the present study, the content of total soluble sugars in Prata-Anã banana was 0.3% in green fruits, and its final content in ripe fruits was 18.5%.
Ethylene adsorption was effective in delaying the changes in soluble sugars after 14 day under CA ambient (20.8 O2 and 0.1% CO2), when the fruits reached 5.6 of sucrose, 0.99 of glucose and 1.19% of fructose. However, after 3 day under normal ambient atmosphere, the pulp reached similar values as the ones found in the ripe fruits of the control treatment (Figure 5).
In CA with low O2 (1, 3 and 5%), the content of soluble sugars presented minimal increase after 14 day of storage, also remaining in minimal levels after 3 day storage under normal ambient atmosphere (Figure 5). These results confirm that in atmosphere with at least 5% O2 the metabolic rate of the fruit was quite impaired.
In the fruits stored under normal ambient atmosphere, the starch content dropped from 27.1 to 5.0% (Figure 4b), with a change of 22.1%, which is considered like the increase in TSS content, which was from 0.5 to 22.7 oBrix during 14 day of storage (Figure 4a). However, the sum of the content of sucrose (Figure 5a), glucose (Figure 5b) and fructose (Figure 5c) in this period reaches 18.2%, so indicating that part of the starch was metabolized to yield energy or to synthesize enzymes or another chemical compound, as for example the organic acids (Figure 3a).
Total phenolic content in banana pulp
The content of phenolics reduced 49.3% after 14 day under normal ambient atmosphere and lost 71.2% of its initial content after 3 day storage under the same condition (Figure 6). Bennett, Shiga, Hassimotto, Rosa, and Lajolo (2010) also reported a decrease in total phenolics compounds during the ripening of different cultivars of bananas.
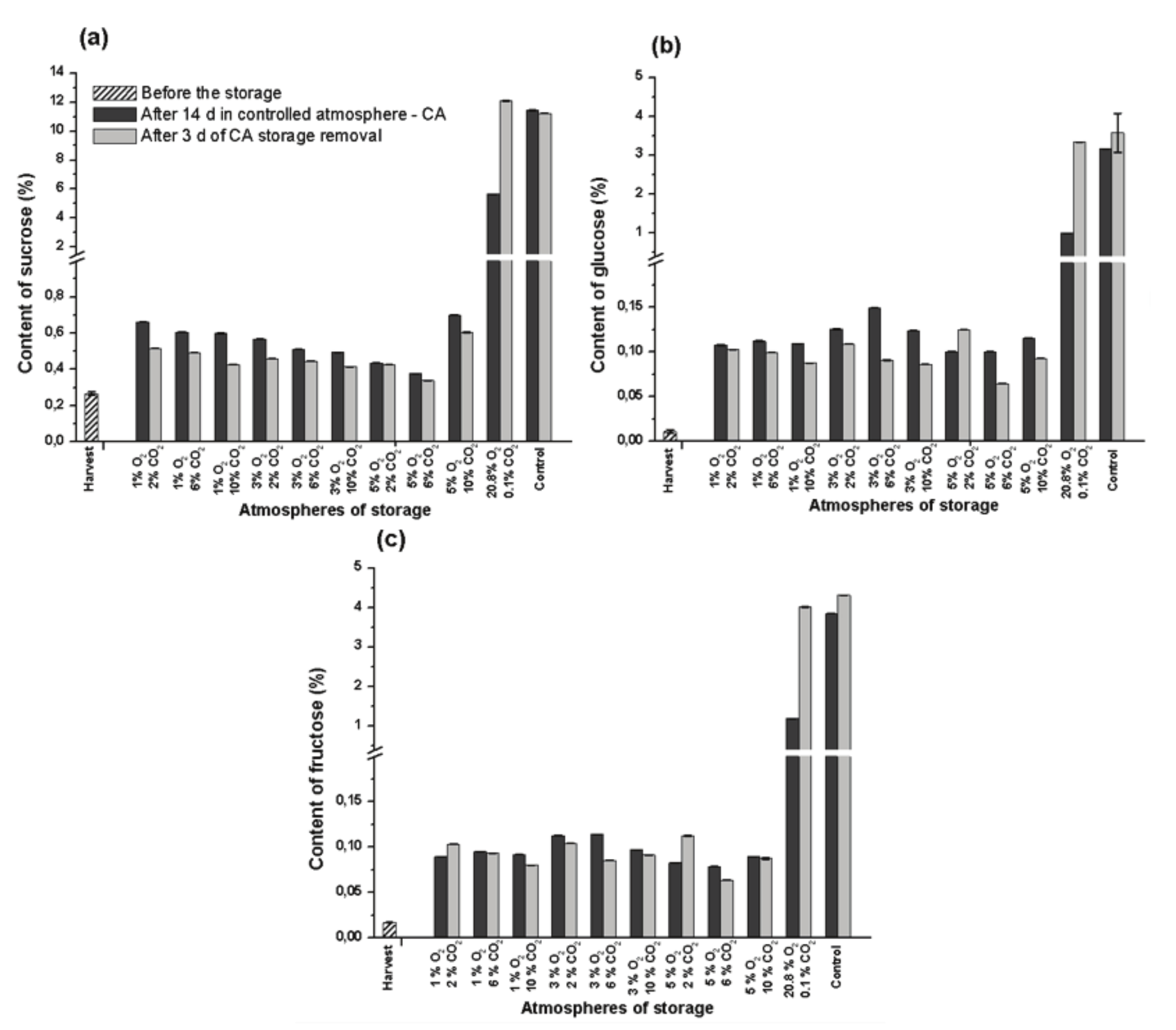
Figure 5.
Content of sucrose (a), glucose (b) and fructose (c) in the pulp of Prata-Anã banana stored for 14 day at 23°C and 90% UR in different conditions of CA with ethylene adsorption, and after 3 day of storage in normal ambient atmosphere. Control treatment at ambient atmosphere. Vertical bars are indicated with confidence intervals at 5% by Student t-Test.
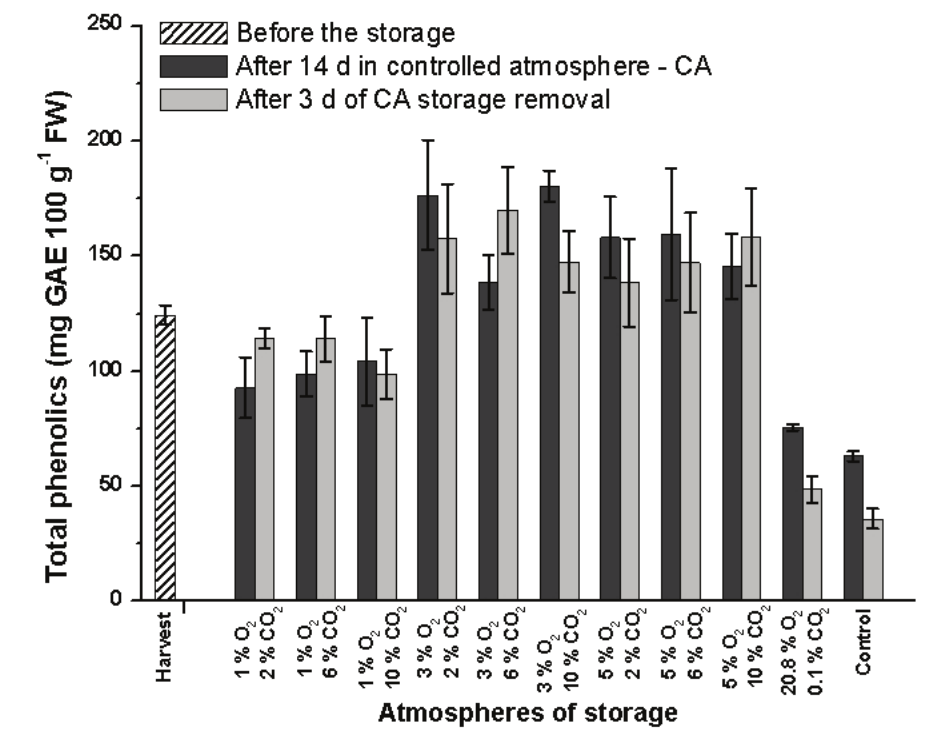
Figure 6.
Total phenolics content in pulp of Prata-Anã banana stored for 14 day at 23°C and 90% UR in different conditions of CA with ethylene adsorption, and after 3 day of storage in normal ambient atmosphere. Control treatment at ambient atmosphere. Vertical bars are indicated with confidence intervals at 5% by Student t-Test.
In different cultivars of bananas harvested in commercial maturation condition, Sulaiman et al. (2011) reported values ranging from 21 to 263 mg gallic acid (GAE) per 100 g of fresh sample. In the present study, it was found 124 mg GAE 100 g-1 of fresh pulp of mature green bananas. The authors Lim, Lim, and Tee (2007), when studying ripe bananas (Musa sapientum), found 51±7 mg GAE 100 g-1 of fresh pulp. In the present study, the banana cv Prata evaluated after 14 day under normal ambient atmosphere reached 63 mg GAE 100g-1 of fresh pulp, which dropped to 35 mg GAE 100g-1 after 3 day under the same condition.
The changing in phenolic compounds was less pronounced in the banana stored in CA ambient conditions with ethylene adsorption, reaching a drop of 60.8% of its initial content in the end of 14 day plus 3 day of storage (Figure 6), therefore, highlighting the effect of ethylene adsorption on the reduction of the breakdown of phenolics compounds.
In contrast, fruits stored for 14 day in CA with low O2 presented higher levels of phenolic compounds in relation to the initial value, also remaining in higher levels after 3 day at the ambient atmosphere (Figure 6). Bananas have the capability to develop defense mechanism against incidence of sunlight or high temperature, increasing its level of phenolic compounds as antioxidative components (Kanazawa & Sakakibara, 2000; Sulaiman et al., 2011).
The fruits stored in CA with 1% O2 did not increase its content of phenolics, probably because its metabolic synthesis was impaired. After 3 day under normal atmosphere, it is noted a slight stimulation of the synthesis of phenolics content, as observed in Figure 6.
Conclusion
Chemical changes in Prata-Anã banana stored under normal atmosphere were characterized by increasing contents of acidity, total soluble solids, sucrose, glucose and fructose, along with a decrease in phenolic compounds.
The ethylene adsorption was effective in delaying the ripening under normal atmosphere. The ripening was impaired during 14 day under CA with low O2 (1, 3 and 5%), and also during the following 3 day under normal atmosphere.
An atypical increase in phenolic compounds was noted in atmospheres with at least 5% O2. Nevertheless, the ethylene treatment aiming to force the ripening and a further sensorial analysis should be conducted to evaluate any eventual quality disturbance in bananas.
Acknowledgements
The authors gratefully acknowledge Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (Capes) for the research grants, and also Bananas EP Company by the availability of raw material and use of its facilities.
References
Association of Official Analytical Chemistry [AOAC]. (1995). Methods of analysis of the association of official analytical chemistry (16th ed.). Washington, DC: AOAC.
Bennett, R. N., Shiga, T. M., Hassimotto, N. M., Rosa, E. A., & Lajolo, F. M. (2010). Phenolics and antioxidant properties of fruit pulp and cell wall fractions of postharvest banana (Musa acuminata Juss.) cultivars. Journal of Agricultural and Food Chemistry, 58(13), 7991-8003. doi: 10.1021/jf1008692
Cordenunsi, B. R., & Lajolo, F. M. (1995). Starch breakdown during banana ripening: Sucrose synthase and sucrose phosphate synthase. Journal of Agricultural and Food Chemistry, 43(2), 347-351. doi: 10.1021/jf00050a016
Cordenunsi, B. R., Shiga, M. S., & Lajolo, F. (2008). Non-starch polysaccharide composition of two cultivars of banana (Musa acuminata L.: cvs Mysore and Nanicão). Carbohydrate Polymers, 71(1), 26-31. doi: 10.1016/j.carbpol.2007.05.009
Food and Agriculture Organizations of the United Nations [FAOSTAT]. (2013). Statistics division production/Crops 2013. Retrivied from http://faostat3.fao.org/browse/Q/QC/E
Imahori, Y., Yamamoto, K., Tanaka, H., & Bai, J. (2013). Residual effects of low oxygen storage of mature green fruit on ripening processes and ester biosynthesis during ripening in bananas. Postharvest Biology and Technology, 77, 19-27. doi: 10.1016/j.postharvbio.2012.11.004
Kader, A. A., Sommer, N. F., & Arpaia, M. L. (2002). Postharvest handling systems: tropical fruits. In A. A. Kader (Ed.), Postharvest Technology of Horticultural Crops (p. 385-398). Oakland, CA: University of California.
Kanazawa, K., & Sakakibara, H. (2000). High content of dopamine, a strong antioxidant in Cavendish banana. Journal of Agricultural and Food Chemistry, 48(3), 844-848. doi: 10.1021/jf9909860
Lim, Y. Y., Lim, T. T., & Tee, J. J. (2007). Antioxidant properties of several tropical fruits: a comparative study. Food Chemistry, 103(3), 1003-1008. doi: 10.1016/j.foodchem.2006.08.038
Liu, S., Yang, Y., Murayama, H., & Taira, S. (2004). Effects of CO2 on respiratory metabolism in ripening banana fruit. Postharvest Biology and Technology, 33(1), 27-34. doi: 10.1016/j.postharvbio.2004.01.006
Lobo, M. G., Gonzales, M., Pena, A., & Marrero, A. (2005). Effects of ethylene exposure temperature on shelf life, composition and quality of artificially ripened bananas (Musa acuminata AAA, cv. Dwarf Cavendish). Food Science and Technology International, 11(2), 99-105. doi: 10.1177/1082013205052568
Nascimento Junior, B. B., Ozorio, L. P., Rezende, C. M., Soares, A. G., & Fonseca, M. J. A. (2008). Differences between Prata and Nanicão banana cultivars during ripening: physicochemical characteristics and volatile compounds. Ciência e Tecnologia de Alimentos, 28(3), 649-658. doi: 10.1590/S0101-20612008000300022
Ortiz, A. A., Graell, J., López, M. L., Echeverría, G., & Lara, I. (2010). Volatile ester-synthesising capacity in ‘Tardibelle’ peach fruit in response to controlled atmosphere and 1-MCP treatment. Food Chemistry, 123(3), 698-704. doi: 10.1016/j.foodchem.2010.05.037
Palomer, X., Roig-Villanova, I., Grima-Calvo, D., & Vendrell, M. (2005). Effects of nitrous oxide (N2O) treatment on the postharvest ripening of banana fruit. Postharvest Biology and Technology, 36(2), 167-175. doi: 10.1016/j.postharvbio.2004.12.008
Peroni-Okita, F. H. G., Simão, R. A., Cardoso, M. B., Soares, C. A., Lajolo, F. M., & Cordenunsi, B. R. (2010). In vivo degradation of banana starch: Structural characterization of the degradation process. Carbohydrate Polymers, 81(2), 291-299. doi: 10.1016/j.carbpol.2010.02.022
Ramos, D. P., Leonel, S., & Mischan, M. M. (2009). Caracterização físico-química dos frutos de genótipos de bananeira produzidos em botucatu-SP. Ciência e Agrotecnologia, 33, 1765-1770. doi: 10.1590/S1413-70542009000700011
Santana, L. R. R., Benedetti, B. C., Sigrist, J. M. M., Sato, H. H., & Anjos, V. D. A. (2011). Effect of controlled atmosphere on postharvest quality of ‘Douradão’ peaches. Ciência e Tecnologia de Alimentos, 31(1), 231-237. doi: 10.1590/S0101-20612011000100036
Santos, C. M. S., Vilas Boas, E. V. B., Botrel, N., & Pinheiro, A. C. M. (2006). Influência da atmosfera controlada sob a vida pós-colheita e qualidade de banana ‘Prata Anã’. Ciência e Agrotecnologia, 30(2), 317-322. doi: 10.1590/S1413-70542006000200018.
Sañudo, M. B., Cepeda, J. S., Rangel, D. M., & Heredita, B. J. (2009). Extending the shelf life of bananas with 1-methylcyclopropene and a chitosan-based edible coating. Journal of the Science of Food and Agriculture, 89(14), 2343-2349. doi: 10.1002/jsfa.3715
Saraiva, L. A., Castelan, F. P., Shitakubo, R., Hassimotto, N. M. A., Purgatto, E., Chillet, M., & Cordenunsi, B. R. (2013). Black leaf streak disease affects starch metabolism in banana fruit. Journal of Agricultural and Food Chemistry, 61(23), 5582-5589. doi: 10.1021/jf400481c
Shiga, T. M., Soares, C. A., Nascimento, J. R., Purgatto, E., Lajolo, F. M., & Cordenunsi, B. R. (2011). Ripening-associated changes in the amounts of starch and non-starch polysaccharides and their contributions to fruit softening in three banana cultivars. Journal of the Science of Food and Agriculture, 91(8), 1511-1516. doi: 10.1002/jsfa.4342
Sistema para Análises Estatísticas e Genéticas [SAEG]. (2007). Sistema para análises estatísticas. Versão 9.1. Viçosa, MG: Fundação Arthur Bernardes/UFV.
Sulaiman, S. F., Yusoff, N. A. M., Eldeen, I. M., Seow, E. M., Sajak, A. A. B., & Ooi, K. L. (2011). Correlation between total phenolic and mineral contents with antioxidant activity of eight Malaysian bananas (Musa sp.). Journal of Food Compostion and Analysis, 24(1), 1-10. doi: 10.1016/j.jfca.2010.04.005
Swain, T., & Hills, W. E. (1959). The phenolic constituents of Prunus domestica: The quantitative analysis of phenolic constituents. Journal of the Science of Food and Agriculture, 10(1), 63-68. doi: 10.1002/jsfa.2740100110
Yang, X., Zhang, Z., Joyce, D., Huang, X., Xu, L., & Pang, X. (2009). Characterization of chlorophyll degradation in banana and plantain during ripening at high temperature. Food Chemistry, 114(2), 383-390. doi: 10.1016/j.foodchem.2008.06.006
Zhang, J., Wang, X., Yu, O., Tang, J., Gu, X., Wan, X., & Fang, C. (2011). Metabolic profiling of strawberry (Fragaria x ananassa Duch.) during fruit development and maturation. Journal of Experimental Botany, 62(3), 1103-1118. doi: 10.1093/jxb/erq343
Notes
Author notes
eresende@uenf.br