Química
Effect of pomegranate seed oil on fatty acids composition of Oreochromis niloticus trough supplemented diet
Effect of pomegranate seed oil on fatty acids composition of Oreochromis niloticus trough supplemented diet
Acta Scientiarum. Technology, vol. 41, 2019
Universidade Estadual de Maringá
Received: 06 July 2017
Accepted: 07 December 2017
Abstract: Several health benefits have been attributed to conjugated linolenic acid isomers (CLnA). In this study, the diet for Nile tilapia was supplied with pomegranate seed oil (at 2.1%) as source of CLnA. The diet was provided to the fish for 15 and 30 days. The punic acid (40.58-53.77 mg g-1 of total lipids) was incorporated into Nile tilapia fillet after 15 and 30 days of experiment, respectively. When compared to the control diet, the level of total saturated fatty acids in fish were slightly increased. No change in the total amount of monounsaturated fatty acid was observed. The pomegranate seed oil diet was effective in the transference of CLnA to Nile tilapia.
Keywords: CLnA, Punica granatum, tilapia.
Introduction
The pomegranate (Punica granatum) is cultivated around the world, especially in the Mediterranean area. In Brazil, pomegranate is widely distributed and commonly found in parks and gardens, being used for ornamental purposes and for its medicinal properties (Takata, Silva, Corsato, & Ferreira, 2014). The pomegranate was commonly used in folk medicine for eliminating parasites, to treat and cure aphtae, ulcers, hemorrhage, microbial infections, and respiratory pathologies (Viuda-Martos, Fernández-López, & Pérez-Álvarez, 2010). According to Aviram and Rosenblat (2013), 50% of the fruit is edible. The edible parts of pomegranate fruits are consumed fresh or used for the preparation of fresh juice, canned beverages, jelly, jam, and paste, and it is also used for flavoring and coloring beverage products (Viuda-Martos et al., 2010). 20% of the edible pomegranate part (seeds) is considered a byproduct, which is obtained during the processing of pomegranate juice (Aviram & Rosenblat, 2013).
Seeds of pomegranate are a source of conjugated linolenic acid (CLnA). The CLnA is a mixture of positional and geometric isomers of octadecatrienoic acid, and its chain has three conjugated (-C = C-C = C-C = C-) double bonds. In nature, the CLnA isomers are also found in plant seeds, such as bitter gourd (Momordica charantia), catalpa (Catalpa ovata), Chinese cucumber (Trichosanthes kirilowii), jacaranda (Jacaranda mimosifolia), marigold (Calendula officinalis), and tung (Aleurites fordii) (Koba, Belury, & Sugano, 2007).
The main CLnA found in pomegranate seed oil is the punic or punicic acid (PuA, 18:3 9c, 11t, 13c). About 70-80% of PuA is present in the total fatty acids of pomegranate seed oil (Koba et al., 2007). Other constituents of pomegranate seed oil are the conjugated linoleic acid (CLA), linoleic acid (18:2n-6), oleic acid (18:1n-9), stearic acid (18:0), eleostearic acid (18:3 9c, 11t, 13t) and catalpic acid (18:3 9t, 11t, 13c) (Viuda-Martos et al., 2010).
Studies suggest the use of CLnA from edible plant seeds as a functional food ingredient, and several health benefits including lipid metabolism regulation, anti-carcinogenic, anti-inflammatory and antioxidant activities have been attributed to CLnA isomers (Yuan, Chen, & Li, 2014). Besides the strong antioxidant activity, pomegranate seed oil has inhibitory effect on breast and skin cancers (Shaygannia, Bahmani, Zamanzad, & Rafieian-Kopaei, 2015).
Tilapia (including all species) is the second most important group of farmed fish after carps, and the most widely grown among farmed fish (Food and Agriculture Organization of the United Nations). The quality of human food must be sought, and the investigation of the lipid composition of fish is important because it will discriminate the lipid profile of the food and the investigation of the lipid composition of fish is important because it will discriminate the lipid profile of this food, which is frequently consumed by the population. Besides, several studies have been conducted aiming the incorporation of specific fatty acids (as conjugated linolenic acid and gamma-linolenic acid) in fish through supplemented diet (Bonafé et al., 2013; Schneider et al., 2015), in order to provide these fatty acids to the human diet.
Due to the high availability of pomegranate fruits in Brazil, this study aimed to evaluate the modifications in fatty acid composition and the incorporation of CLnA into tilapia fish fillet, after the fish being fed with diets supplemented with pomegranate seed oil.
Material and methods
Punica granatum seed oil extraction and diet preparation
Pomegranate fruits were harvested in Apucarana, state Paraná. Pomegranate seed oil was extracted in the laboratory. The pulp was separated from the pomegranate rind and squeezed to remove the juice. The seeds were dried in an oven with air circulation at 60ºC for 10 hour. The dried seeds were ground in the mill. Pomegranate seed powder was obtained after sieving the crushed seeds (16-mesh). Then, hexane and pomegranate seed powder (4:1, v w-1) were stirred for 3 hour. The mixture was centrifuged for 10 min (18,000 g) and then the solvent was vacuum-evaporated at 36-38 ºC to obtain the pomegranate seed oil.
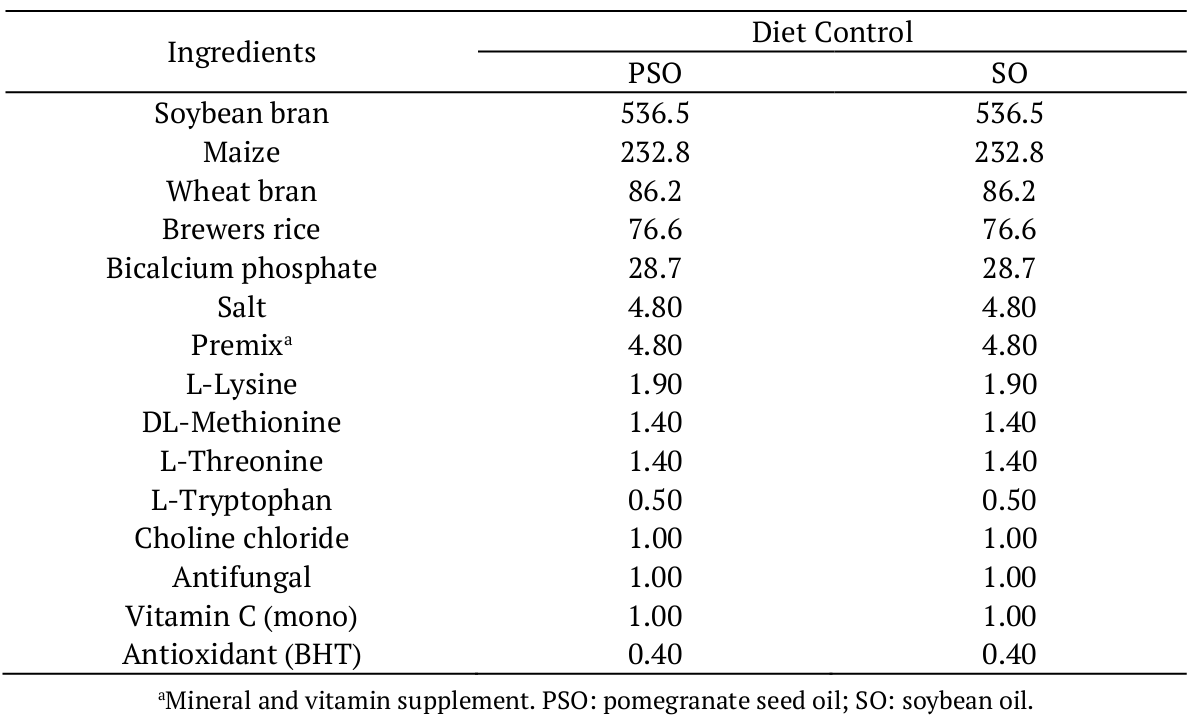
As shown in Table 1, the supplemented diet was prepared containing pomegranate seed oil. Soybean oil was used as control. The ingredients were milled, sieved, mixed with water to obtain the pellets (3 mm of diameter), and dried in an oven with air circulation at 55ºC for 10 hour, according to Schneider et al. (2015). The pellets were vacuum-packed, protected from light and kept at -18 ºC until use in fish feeding.
Trial
All Ethical Principles, Protocols and Regulations on Experimentation with Laboratory Animals were used according to the standards established internationally and by the approved project by the Institutional Ethics Committee of State University of Maringá (UEM), the Ethics Committee on Animal Use in Experimentation (Ceae)/UEM. 120 fish (Nile tilapia) were obtained from Fish Experimental Station UEM/Codapar, located in Floriano District of Maringá, state Paraná, with initial average weight of 12.00 ± 1.00 g. Trial and fish sampling were similar to the one reported by Schneider et al. (2015) and each treatment was conducted in duplicate. Fish were fed with the same control diet containing soybean oil at 2.1% for adaptation to the new conditions for 7 days. On the 8th day, the initial treatment was established. Then, tanks were divided randomly (two tanks remained with control diet whole two tanks were submitted to pomegranate seed oil diet). Fish were fed twice a day for 30 days. After 15 days, a sample (composed of 12 fish) was removed from each tank. Sequentially, the experiment was carried out for more 15 days, when the period of 30 days was completed. Samples were also performed by collecting 12 fish per tank. Fish were euthanized with a lidocaine overdose (10 g L-1). The samples were disemboweled, washed, filleted, vacuum packed in polyethylene bags and stored at -18°C for later analysis (Schneider et al., 2015).
Analyses
Analyses of proximate composition of the feed diets were carried out in triplicate. Total lipids were extracted by the Bligh and Dyer method and moisture, ash, and protein contents were determined in accordance with the Association of Official Analytical Chemists (AOAC, 1995).
Total lipids were extracted from the tilapia fillet according to Bligh and Dyer (1959). Fatty acid methyl esters were prepared by methylation (Hartman & Lago, 1973) and modified by Maia and Rodriguez-Amaya (1993). Methyl esters were separated by gas chromatography in a Thermo model Trace Ultra 3300 equipped with a flame ionization detector and a cyanopropyl capillary column (100 m x 0.25 i.d., 0.25 μm film thickness, CP-7420) (Martin, Oliveira, Visentainer, Matsushita, & Souza, 2008). The gas flow rates used were 1.2 mL min-1 carrier gas (H2); 30 mL min-1 makeup gas (N2); 35 and 350 mL min-1 flame gases (H2 and synthetic air, respectively). The sample (2 μL) splitting ratio was 1:80. Operating parameters were as follows: detector temperature, 240ºC; injection port temperature, 230ºC. Initially, the column temperature was maintained at 165ºC for 7 min. It was then raised to 185ºC, at a rate of 4ºC min-1, and kept at this temperature for 4.67 min. After this period, it was once again raised to 235ºC at a rate of 6ºC min-1 and maintained for 5 min, totaling 30 min of chromatographic run. Analyses were carried out in triplicate. Peak areas were determined by the Software Chromquest 5.0. Fatty acids were quantified against tricosanoic acid methyl ester (Sigma, St. Louis, MO, USA) as an internal standard, as described by Joseph and Ackman (1992). Theoretical FID (flame ionization detector) correction factor values were used to calculate fatty acid concentration values in mg g-1 of total lipids with Equation 1, according to Visentainer (2012):
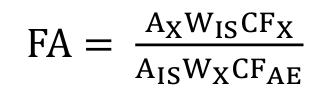 (1)
(1)where:
FA is mg of fatty acids per g of total lipids, AX is the peak area (fatty acids), AIS is the peak area of internal standard methyl ester of tricosanoic acid (23:0), WIS is the internal standard weight (mg) added to the sample, WX is the sample weight (g), CFX is the theoretical correction factor, and CFAE is the conversion factor necessary to express results as mg of fatty acids rather than as methyl esters.
For identification of fatty acids, the retention times of the common fatty acids (not conjugated) were compared to those of standard methyl esters (Sigma, St. Louis, MO, USA). The observed area (size) and retention time of the main fatty acid of pomegranate seed oil was defined as PuA. The oil chromatogram and respective diet/fish chromatogram were compared. As a result, PuA was identified. Smaller peaks close to the main conjugated fatty acid were identified as ‘possible conjugated isomers of 18:3’.
Statistica
The results (triplicate) were submitted to variance analysis (ANOVA) at 5% significance level with Statistica software version 8.0 (Statsoft, USA) and means were compared by t test and Tukey’s test.
Results and discussion
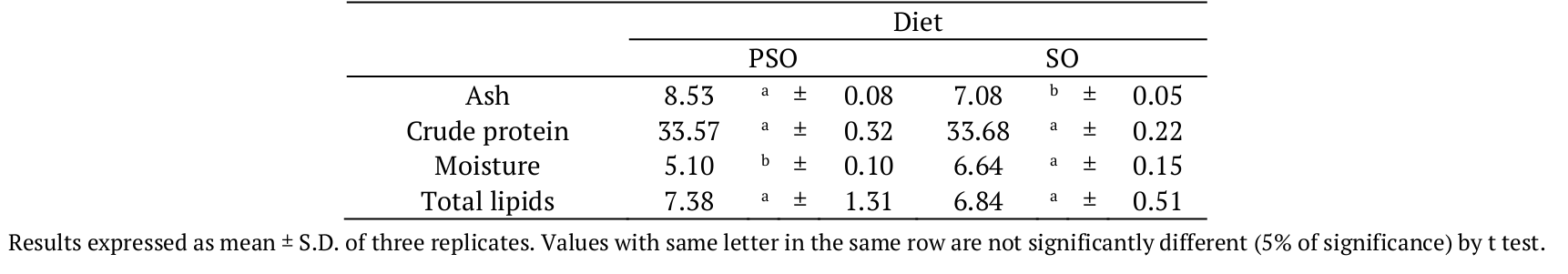
Table 2 shows the proximate composition of the diets supplemented with pomegranate seed and soybean oils. Statistically (at 5% of significance), the same values of crude protein and total lipids were determined in the both diets, which is desirable in conducting an experiment.
As expected, the main difference between PSO and SO diets was the CLnA composition (Table 3). The PuA, from pomegranate seed oil, was the most found among the conjugated fatty acids in PSO diet (172.75 mg g-1 of total lipids). The SO diet did not present CLnA. Data reported by Bonafé et al. (2013) indicated a total of 121.44 mg α-ESA per g of total lipids in diet supplemented with a mixture of conjugated linoleic acid (CLA) and tung oil (totalizing 4.2%). The same treatment presented 202.47 mg CLnA g-1 of total lipids, including the β-eleosteric acid and three more unidentified CLnA isomers.
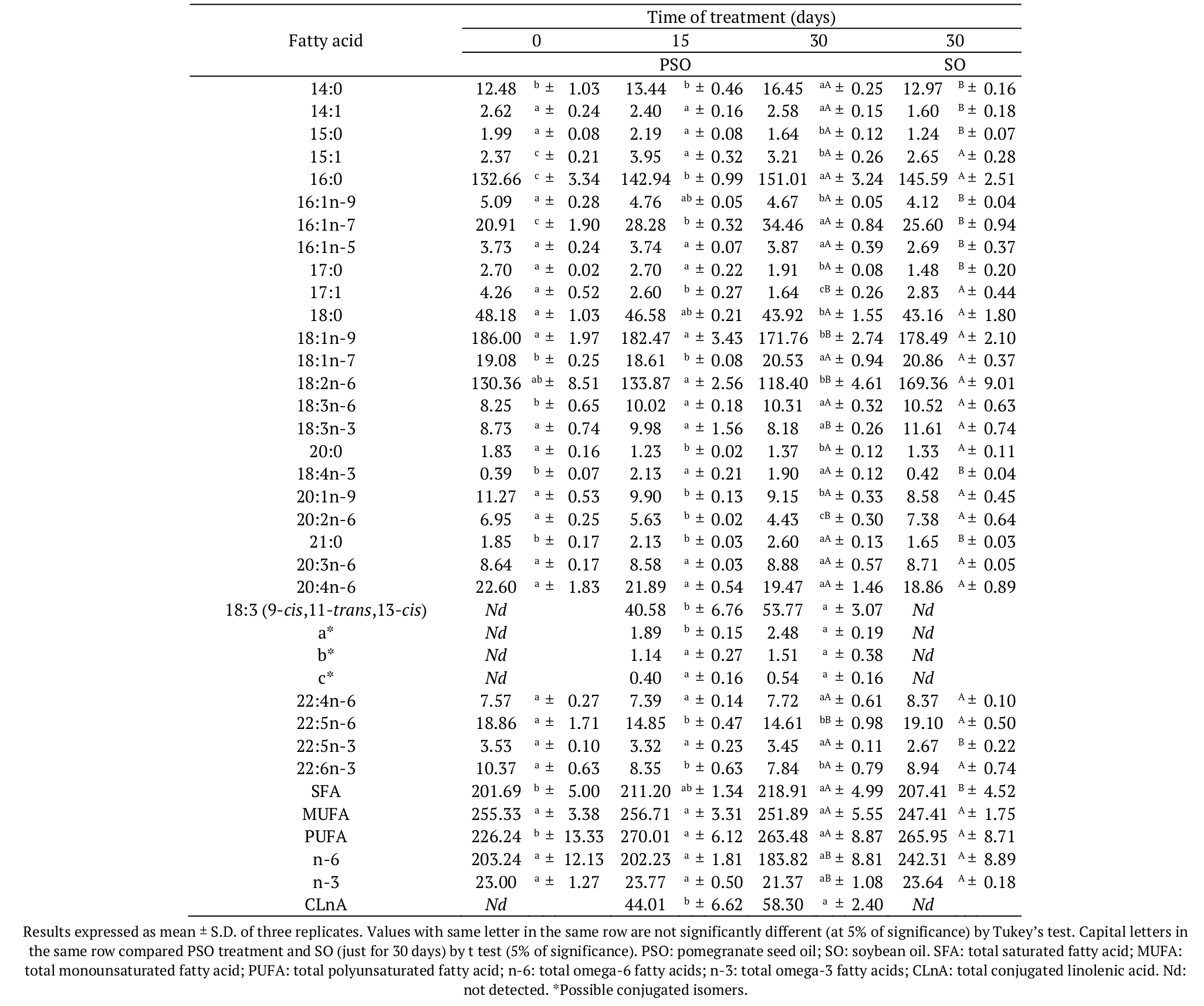
Total saturated fatty acids (SFA), total monounsaturated fatty acids (MUFA), total omega-6 (n-6) and total omega-3 (n-3) were higher in the control diet that used soybean oil. However, the total polyunsaturated fatty acids (PUFA) was higher in the PSO diet. Table 4 shows the fatty acid composition of Nile tilapia submitted to the treatment with pomegranate seed oil. Some studies aim to increase the levels of essential fatty acids (as n-3 fatty acids) in fish through supplemented diet (Carbonera et al., 2014; Silva et al., 2014). However, the diet added with pomegranate seed oil presented lower levels of essential fatty acids, when compared to the soybean oil diet.
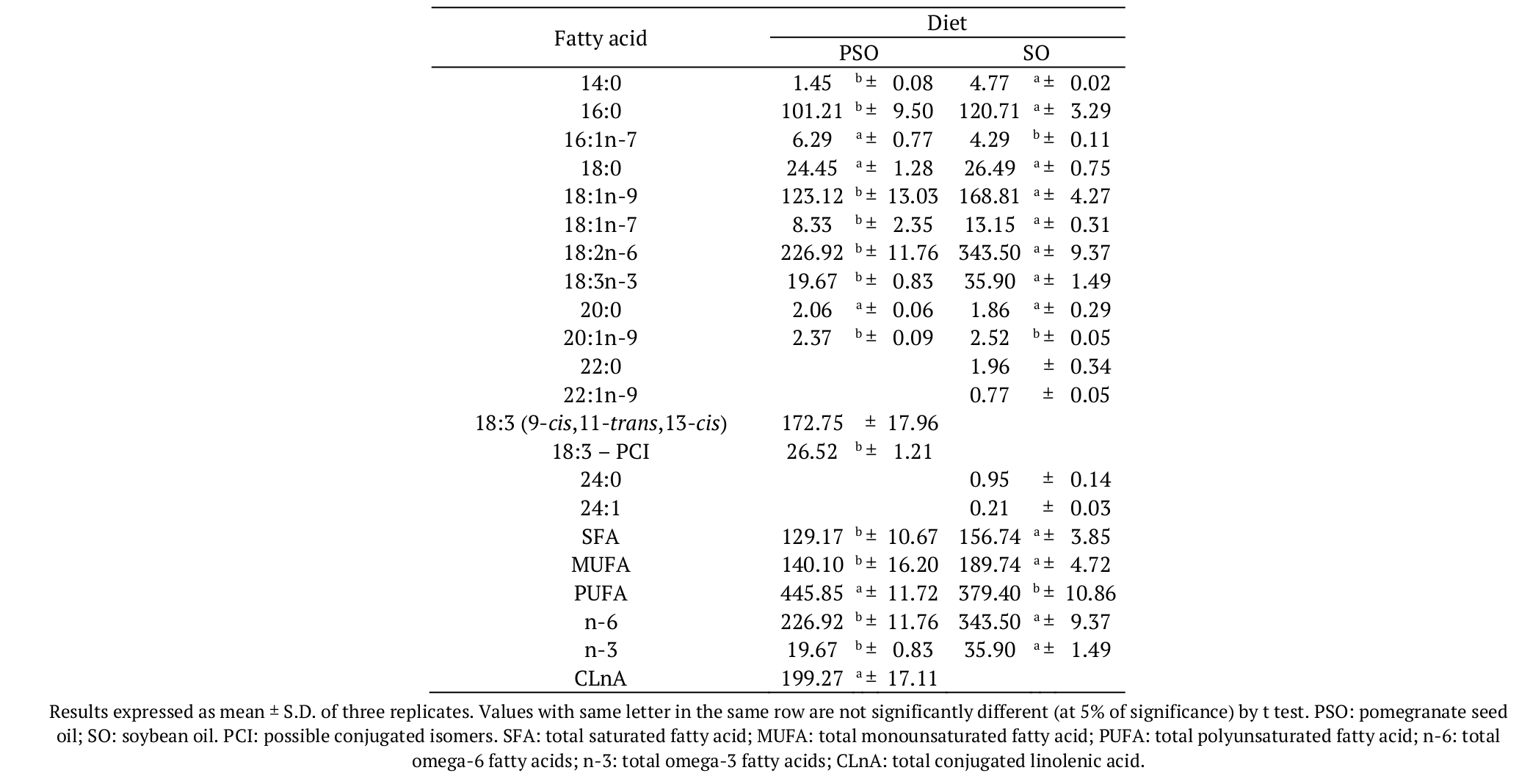
It should be highlighted that 15 days of treatment were effective in incorporating significant amounts of PuA into fish lipids. The first 15 days of treatment with pomegranate seed oil incorporated 40.58 mg PuA g-1 of total lipids, while 30 days was responsible for the transference of 53.77 mg PuA g-1 of total lipids to the Nile tilapia fillet. These values are higher than those reported by Bonafé et al. (2013), who obtained from 5.37 to 7.44 mg alpha-eleostearic acid g-1 of total lipids in fish fillet, after 30 days of treatment.
Regarding the total CLnA (including isomers), the amount varied from 44.01 to 58.30 mg g-1 of total lipids, i.e., The PuA was the more expressive CLnA transferred to the fish, and it represented 92.2% of total CLnA incorporated into fish lipids after 15 or 30 days of treatment. Researchers quantified around 16-24 mg CLnA g-1 of total lipids in tilapia fed with a diet containing CLA + tung oil for 10, 20 and 30 days, included in the total CLnA, the α and β-eleostearic acid and three more unidentified isomers (Bonafé et al., 2013). In a recent experiment, Bonafé et al. (2017) improved the nutritional quality of Nile tilapia meat through enriched diets with CLnA isomers from tung oil and, after 30 days of treatment, fish fillet incorporated 24.00 g CLnA kg-1 of total lipids.
No significant differences were observed in the levels of MUFA at 5% of significance. Total saturated fatty acids were slightly higher in the fish submitted to PSO diet.
The level of PUFA in fish fillet was higher after the treatment with PSO (Table 4), however, it did not differ from the PUFA value of fish submitted to the control diet after 30 days of experiment (at 5% of significance). Conversely, n-6 and n-3 values in PSO fish were smaller when compared to the SO fish. Table 3 shows the differences in the values of the essential fatty acids linoleic (18:2n-6) and linolenic (18:3n-3) in the formulated diets. These molecules were the unique source of n-6 and n-3 fatty acids for the fish. Bonafé et al. (2017) observed an increase of n-3 fatty acids and a decrease of n-6 fatty acids in fish submitted to tung oil (CLnA) diet after 30 days of treatment.
Yasmin, Takeuchi, Hirota, and Ishida (2004), who evaluated the effect of PuA on the lipid composition of Oncorhynchus mykiss, observed an increase in the oleic acid level and a decrease in the linoleic acid level, but the level of SFA was unaffected by using of 2% PuA in a diet for fish. The results found in the present study demonstrated that PSO diet increased the SFA level and did not alter the MUFA level in fish, as showed in the Table 4.
Other researchers studied the incorporation of CLA in Piaractus mesopotamicus and Pimedolus maculatus. Their results reported the incorporation of 20.18 mg CLA g-1 (of total lipids) in Piaractus mesopotamicus after 49 days of treatment and 12.65 mg CLA g-1 (of total lipids) in Pimedolus maculatus after 50 days of treatment, by using as a feed ingredient a commercial CLA supplement (Zanqui et al., 2013; Barilli et al., 2014). Compared to these studies, the use of pomegranate seed oil, in a short period of time, as the source of CLnA in fish feeding, provided higher incorporation of CLnA in tilapia.
Conclusion
This research showed satisfactory results in the transference of CLnA to Nile tilapia by using pomegranate seed oil as a diet ingredient. Furthermore, the pomegranate seed oil, as a lipid source in fish feeding, allowed a considerable incorporation of PuA into the fish fillet, consequently improving the fish meat quality. Besides incorporating CLnA, the treatments slightly increased the levels of SFA in the tilapia fillet and did not alter the MUFA levels.
Acknowledgements
The authors are grateful for the support of the Universidade Tecnológica Federal do Paraná and the Universidade Estadual de Maringá in conducting this research.
The authors have declared no conflict of interest.
References
Association of Official Analytical Chemists [AOAC]. (1995). Official methods of analysis of AOAC (16th ed.). Arlington, VA: AOAC.
Aviram, M., & Rosenblat, M. (2013). Pomegranate for your cardiovascular health. Rambam Maimonides Medical Journal, 4(2), 1-12. doi: 10.5041/RMMJ.10113
Barilli, D. J., Santarosa, M., Zanqui, A. B., Boscolo, W. R., Feiden, A., Furuya, W. M., ... Matsushita, M. (2014). Incorporation of conjugated linoleic acid (CLA) and α-linolenic acid (LNA) in pacu fillets. Food Science and Technology, 34(1), 74-81. doi: 10.1590/S0101-20612014005000010
Bligh, E. G., & Dyer, W. J. (1959). A rapid method of total lipid extraction and purification. Canadian Journal of Biochemistry and Physiology, 37(8), 911-917. doi: 10.1139/o59-099
Bonafé, E. G., Boeing, J. S., Matsushita, M., Claus, T., Santos, O. O., Oliveira, C. C., ... Visentainer, J. V. (2013). Evaluation of conjugated fatty acids incorporation in tilapia through GC-FID and EASI-MS. European Journal of Lipid Science and Technology, 115(10), 1139-1145. doi: 10.1002/ejlt.201300032
Bonafé, E. G., Figueiredo, L. C., Martins, A. F., Monteiro, J. P., Junior, O. O. S., Canesin, E. A., ... Visentainer, J. J. (2017). Incorporation of conjugated fatty acids into Nile tilapia (Oreochromis niloticus). Journal of the Science of Food and Agriculture, 97(10), 3469-3475. doi: 10.1002/jsfa.8149
Carbonera, F., Bonafé, E. G., Martin, C. A., Montanher, P. F., Ribeiro, R. P., Figueiredo, L. C., ... Visentainer, V. V. (2014). Effect of dietary replacement of sunflower oil with perilla oil on the absolute fatty acid composition in Nile tilapia (GIFT). Food Chemistry, 148(1), 230-234. doi: 10.1016/j.foodchem.2013.10.038
Hartman, L., & Lago, B. C. N. (1973). A rapid preparation of fatty methyl esters from lipids. Laboratory Practice, 22(6), 475-476.
Joseph, J. D., & Ackman, R. G. (1992). Capillary column gas chromatographic method for analysis of encapsulated fish oils and fish oil ethyl esters: collaborative study. Journal of AOAC International, 75(3), 488-506.
Koba, K., Belury, M. A., & Sugano, M. (2007). Potential health benefits of conjugated trienoic acids. Lipid Technology, 19(9), 200-203. doi: 10.1002/lite.200700067
Maia, E. L., & Rodriguez-Amaya, D. B. (1993). Avaliação de um método simples e econômico para a metilação de ácidos graxos com lipídios de diversas espécies de peixes. Revista do Instituto Adolfo Lutz, 53(1-2), 27-35.
Martin, C. A., Oliveira, C. C., Visentainer, J. V., Matsushita, M., & Souza, N. E. (2008). Optimization of the selectivity of a cyanopropyl stationary phase for the gas chromatographic analysis of trans fatty acids. Journal of Chromatographic A, 1194(1), 111-117. doi: 10.1016/j.chroma.2008.04.033
Schneider, V. V. A., Carbonera, F., Lopes, A. P., Santos, O. O., Oliveira, C. C., Souza, N. E., & Visentainer, J. V. (2015). Effect of dietary replacement of soybean oil with different sources of gamma-linolenic acid on fatty acid composition of Nile tilapia. Journal of the American Oil Chemists' Society, 92(2), 225-231. doi: 10.1007/s11746-014-2586-z
Shaygannia, E., Bahmani, M., Zamanzad, B., & Rafieian-Kopaei, M. (2015). A review study on Punica granatum L. Journal of Evidence-Based Complementary & Alternative Medicine, 21(3), 221-227. doi: 10.1177/2156587215598039
Silva, B. C., Santos, H. M. C., Montanher, P. F., Boeing, J. S., Almeida, V. C., & Visentainer, J. V. (2014). Incorporation of omega-3 fatty acids in nile tilapia (Oreochromis niloticus) fed chia (Salvia hispanica L.) bran. Journal of the American Oil Chemists' Society, 91(3), 429-437. doi: 10.1007/s11746-013-2391-0
Takata, W., Silva, E. G., Corsato, J. M., & Ferreira, G. (2014). Germinação de sementes de romãzeiras (Punica granatum L.) de acordo com a concentração de giberelina. Revista Brasileira de Fruticultura, 36(1), 254-260. doi: 10.1590/0100-2945-269/13
Visentainer, J. V. (2012). Aspectos analíticos da resposta do detector de ionização em chama para ésteres de ácidos graxos em biodiesel e alimentos. Quimica Nova, 35(2), 274-279. doi: 10.1590/S0100-40422012000200008
Viuda-Martos, M., Fernández-López, J., & Pérez-Álvarez, J. A. (2010). Pomegranate and its many functional components as related to human health: a review. Comprehensive Reviews in Food Science and Food Safety, 9(6), 635-654. doi: 10.1111/j.1541-4337.2010.00131.x
Yasmin, A., Takeuchi, T., Hirota, T., & Ishida, S. (2004). Effect of conjugated linolenic acid (cis-9, trans-11, cis-13 - 18:3) on growth performance and lipid composition of fingerling rainbow trout Oncorhynchus mykiss. Fisheries Science, 70(6), 1009-1018. doi: 10.1111/j.1444-2906.2004.00901.x
Yuan, G. F., Chen, X. E., & Li, D. (2014). Conjugated linolenic acids and their bioactivities: a review. Food & Function, 5(7), 1360-1368. doi: 10.1039/c4fo00037d
Zanqui, A. B., Maruyama, S. A., Barilli, D. J., Ribeiro, S. A. O., Gomes, S. T. M., Visentainer, J. V., ... Matsushita, M. (2013). Incorporation of conjugated linoleic and alpha linolenic fatty acids into Pimedolus maculatus fillets. Food Science and Technology, 33(3), 532-540. doi: 10.1590/S0101-20612013005000066
Author notes
vanessavivian@utfpr.edu.br