Biotechnology
Effect of agro-industrial residues mixtures on the production of endoglucanase by Aspergillus niger in solid state fermentation
Effect of agro-industrial residues mixtures on the production of endoglucanase by Aspergillus niger in solid state fermentation
Acta Scientiarum. Technology, vol. 42, 2020
Universidade Estadual de Maringá

This work is licensed under Creative Commons Attribution 4.0 International.
Received: 19 January 2018
Accepted: 06 April 2018
Abstract: The low-cost production of cellulolytic complexes that present high action at mild conditions is one of the major bottlenecks for the economic viability of the production of cellulosic ethanol. The influence of agro-industrial residues was assessed to enhance endoglucanase production by Aspergillus niger 426 grown in solid state fermentation. The highest percentage of lignin degradation was found on soybean hulls (56%) followed by sugarcane bagasse (36%) and rice straw (8.5%). The cellulose degradation, around 90%, was observed on soybean hulls and sugarcane bagasse, but only 50% on rice straw, and maximum production of endoglucanase (112.34 ± 0.984 U mL-1) was observed for soybean hulls. The best Experimental Mixture Design condition was under cultivation of 2.5 g of sugarcane bagasse, 2.3 g of rice straw and 5.2 g of soybean hulls, leading to a maximum activity of 138.92 ± 0.02 U mL-1. The statistical methodology enabled an increase of over 20% in the production of endoglucanase using agro-industrial waste. These data demonstrate that A. niger 426 is a potential source of cellulases which can be obtained by solid state fermentation using agro-industrial waste.
Keywords: solid state fermentation, endoglucanase, experimental mixture design, Aspergillus niger..
Introduction
Agro-industrial residues contain lignocellulosic material available for exploitation as sources of chemical feedstocks, fuels, foods and feeds. Lignocellulose consists of three types of polymers, i.e., cellulose, hemicellulose, and lignin. The major component of lignocellulose is cellulose, which is the most abundant renewable organic resource, and comprises approximately 30–50% of the dry weight of lignocellulose (Harris & DeBolt, 2010). Cellulose is also the major component of agro-industrial waste.
Agro-industrial residues can be used as a carbon source to produce enzymes, especially holocellulases. Within this group of enzymes, endoglucanases have important roles in the hydrolysis of holocellulose (Segato, Damásio, de Lucas, Squina, & Prade, 2014). Cellulases are relatively costly enzymes, and a significant reduction in cost will be important for their commercial use in biorefineries. Cellulase-based strategies that will make the biorefinery processing more economical include: increasing commercial enzyme volumetric productivity, producing enzymes using cheaper substrates and producing cellulases with higher specific activity on solid substrates (Ekwe, Morgenstern, Tsang, Storms, & Powlowski, 2013;Hingsamer & Jungmeier, 2019).
The production of cellulases with solid state fermentation (SSF) is gaining interest as a cost-effective technology due to its lower capital investment, lower operating expenses, simpler equipment, and higher productivity per reactor volume as compared to submerged fermentation (Hafiz, Ishtiaq, Muhammad, & Muhammad, 2011; Anwar, Gulfraz, & Irshad, 2014). The nature of solid substrate is the most crucial factor in SSF for cellulase production as it not only supplies nutrients to the culture but also serves as an anchorage for the microbial cells. However, due to the complex nature of residual lignocellulosic materials, the physiological responses of fungi, in terms of type of secreted enzyme, enzyme titers, rate of biosynthesis, and so on, are quite variable. Appropriate studies of this behavior, using complex lignocellulosic materials must be undertaken for the different filamentous fungi strains used for endoglucanase biosynthesis (Delabona, Farinas, Lima, Azzoni, & Pradella, 2013).
Experimental mixture design (EMD) is a convenient statistical technique for bioprocess development such as studies of culture media selection and optimization of enzyme mixture for biomass enzymatic hydrolysis (Suwannarangsee et al., 2012). The methodology is attractive because it can be molded to study the exact proportion of the components in a given culture medium and can aid identification of the relationships amongst these components during endoglucanase production, which could not be done with factorial experiments, for example. This paper aimed to optimize, through EMD, the SSF production of endoglucanase from Aspergillus niger 426 using the combination of sugarcane bagasse, rice straw and soybean hulls.
Material and methods
Material
All chemical compounds were purchased from Sigma Chemical Co. (St. Louis, MO, USA). The residues, such as soybean hulls, sugarcane bagasse and rice straw were provided by an agro-industry located in the southeast region of Parana state, then dried to 2% humidity in an oven at 70°C for 24 hours and ground in a mill Wiley type in the particle size of approximately 2 mm (Association of Official Analytical Chemists [AOAC], 1995) and stored in plastic bags at 4°C for later use as substrates in this study. Aspergillus niger strain 426 was isolated from dried prunes and identified by the Institute of Food Technology, Unicamp, Brazil. Stock cultures were stored at 4°C on potato dextrose agar (PDA) slants. Microorganism activation was carried out in PDA plates at 28°C for 3 days.
Compositional analysis of agro-industrial residues
Cellulose and lignin were determined by the detergent system (Van Soest, 1963). The standard acid detergent lignin (ADL) was determined as part of a sequential extraction comprising three refluxing and filtering steps. Firstly, with neutral detergent (NDF), followed by acid detergent (ADF) and lastly 12M H2SO4 digestion to leave lignin and ash residues. The NDF comprises hemicellulose, cellulose, lignin and ash. Digestion of NDF in an acid detergent removes hemicellulose leaving the acid detergent fiber (ADF) fraction (cellulose), and a further digestion in 12M sulfuric acid removes cellulose leaving lignin and ash. Lignin was calculated from 12M sulfuric acid residue less the values for ash. Ash and Dry matter contents were determined as described by AOAC (1995).
The extraction of soluble sugars from defatted substrates was performed according to the methodology proposed by Saravitz, Pharr, and Carter (1987) and filtrated through 0.45 μm filter (Millipore). For the analysis of sugars, aliquots of 20 μL extracted samples were injected to HPLC (Model: Hitachi L-5000) using a Aminex HPX-87P column (300 x 7.8 mm) and eluted in a mobile phase of acetonitrile: water (75:25, v v-1) in the following chromatographic conditions: 35°C injection temperature, flow rate of 1.0 mL min.-1 in a HPLC system equipped with a RID detector (Shimadzu model RID - 10A). Each analysis was performed in triplicate.
Fermentation conditions
Experiments were conducted in 250 mL flasks containing 10 g (dry weight) of powdered solid substrates. The solid substrates were moistened with MW culture media adapted from Mandels and Weber (1969). The MW culture media had the following composition (g L-1 in deionized water): 1.4 (NH4)2 SO4, 2.0 KH2PO4, 0.3 CaCl2, 0.3 MgSO4.7H2O, 0.1% (v v-1) Tween-80, 1.0 Celufloc microcrystalline cellulose (Celuflok Ind. Com., Brazil), 0.005 FeSO4·7H2O, 0.0016 MnSO4·7H2O, 0.0014 ZnSO4·7H2O and 0.002 CoCl2·6H2O. The initial pH was adjusted to pH 5.0.
Inoculum was prepared by harvesting the spores from 3-day-old cultures by adding 0.1% (v v-1) Tween-80 and 8-10 undrilled glass beads. The spores were dislodged by gentle shaking and diluted to the desired spore concentration (1 × 108 spores/g substrate). The pour plate technique was used to calculate the number of spores. A volume equal to 10% (v v-1) of inoculum was transferred to previously sterilized Erlenmeyer flasks containing 10 mL of MW culture media with 10 g of powdered solid substrates and incubated for 5 days at 28°C with 97% (v v-1) controlled atmospheric humidity. The moisture content was adjusted to 70% (v v-1) by the addition of distilled water into the 250 mL Erlenmeyer flasks. The remaining MW culture medium components were always the same. Erlenmeyer flasks were sterilized at 121°C for 20 min. and, after cooling, inoculated and incubated at 28°C.
Statistical design
Assessment was made of the effects of the presence of the three substrates selected for endoglucanase production and their concentrations in the culture media were established using experimental mixture design (EMD). A mixture experiment is a special type of response surface experiment in which the factors are the ingredients or components of a mixture, and the response is a function of the proportions of each ingredient. Because the mixture space is a simplex, all design points must be at the vertices, on the edges or faces, or in the interior of a simplex (Cornell, 2002).
The EMD was set up as a simplex centroid design for three components at a fixed total amount of 10 g. The design comprised 9 experiments, including 3 center point experiments to minimize the error of ANOVA. The randomized design table for these experiments is presented in Table 1. A special cubic model was used to represent the mean of the response variable as a function of the factors described in Equation 1. In this example x1 + x2 + x3 = 1 or 100%; and because of this constraint, the levels of the factors can not be chosen independently (Cornell, 2002).
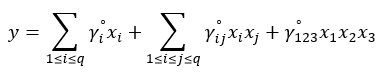 (1)
(1)where:
Y is the endoglucanase activity (U mL-1), x1 the sugarcane bagasse, x2 the rice straw, x3 the soybean hulls and γ the estimated parameters for q = 3 components.
The ‘Statistica’ Version 8.0 (Statsoft, Tulsa, OK, USA) software was used for regression and graphical analysis of data. The statistical significance of the regression coefficients was determined by Fischer’s test for analysis of variance (ANOVA) at a significance level p ≤ 0.05, and the extent of variance explained by each model was given by the determination coefficient R2. Experimental and predicted values were compared in order to determine the validity of the models.
Preparation of the crude extracts
To extract the enzyme, fermented substrates was mixed with distilled water at a solid/liquid ratio of 1 g initial dry substrate/10 mL water (Mrudula & Murugammal, 2011). The mixture was stirred at 30°C and 150 rpm for 60 min. Subsequently, solids were separated by vacuum filtration through cheese cloth. After centrifugation (9000 g, 20 min. at 4°C), the supernatant was submitted to analyses for determination of enzyme activity.
Determination of endoglucanase activity
The method chosen to determine the activity of endoglucanase is based on the dose of reducing sugars (Ghose, 1987) produced by the degradation of carboxymethylcellulose (CMC) at 2% (w v-1), previously diluted in a sodium citrate solution with pH of 4.8 at 50 mM. The dinitrosalicylic acid (DNS) method has been used for quantification (Miller, 1959). Reaction assays were conducted by adding 0.5 mL of sodium citrate buffer solution with a pH of 4.8 at 50 mM, 0.5 mL of enzyme extract, and 0.5 mL of CMC (2% w v-1) to an assay tube. The samples were incubated at 50°C for 30 min. The reaction was interrupted by the addition of 3 mL of DNS. After that, the tubes were submerged into boiling water, for 5 min., and shortly after, 20 mL of distilled water were added for a subsequent measurement of absorbance – in the 540 nm range – carried out using a spectrophotometer.
| Run | Coded mixture (x1; x2; x3)** | Original mixture* (x1; x2; x3)** | Endoglucanase activity (U mL-1) |
| 1 | (1; 0; 0) | (10.0; 0; 0) | 82.361 |
| 2 | (0; 1; 0) | (0; 10.0; 0) | 67.551 |
| 3 | (0; 0; 1) | (0; 0; 10.0) | 111.46 |
| 4 | (½; ½; 0) | (5.0; 5.0; 0) | 72.488 |
| 5 | (½; 0; ½) | (5.0; 0; 5.0) | 129.457 |
| 6 | (0; ½; ½) | (0; 5.0; 5.0) | 105.744 |
| 7 | (⅓; ⅓; ⅓) | (3.3; 3.3; 3.3) | 136.142 |
| 8 | (⅓; ⅓; ⅓) | (3.3; 3.3; 3.3) | 135.742 |
| 9 | (⅓; ⅓; ⅓) | (3.3; 3.3; 3.3) | 135.867 |
Results and discussion
Fermentation kinetics for endoglucanase production
Before substrate selection, the fermentation kinetics for the central point of the EMD was carried out to determine the optimal time for enzyme production. The central point was composed of a 33.33% ternary mixture of each supporting substrate (sugarcane bagasse, rice straw and soybean hulls). The experiments were performed in triplicate, and differences smaller than 10% were obtained for endoglucanase, as well as for residual reducing sugars concentrations. The evaluation of the time course is of prime importance for cellulase biosynthesis by fungi. As observed in Figure 1, endoglucanase production in the standing conditions increased substantially until the 3rd day of culture (136.021 ± 0.87 U mL-1), drastically decreasing in the subsequent days. The yields of reducing sugars (3.892 ± 1.32 mg mL-1), obtained from saccharification of the substrates, reached the maximum at 72 hours and decreased along with the endoglucanase expression.
It is important to note that cellulases and most of the plant cell-wall hydrolyzing enzymes are inducible and are also influenced by catabolite repression in many fungi (Adnan et al., 2017). The decline of endoglucanase activity after the maximum peak of enzyme secretion could be attributed to cumulative release of cellobiose at the start of the hydrolysis. The increase of initial reducing sugar level also strongly inhibits subsequent hydrolysis (Kobakhidze, Asatiani, Kachlishvili, & Elisashvili, 2016). Furthermore, the shape of the hydrolysis curves could be from enzyme inactivation by some other process in addition to product inhibition, since enzyme productivity did not increase again after the decline of reducing sugars, like irreversible adsorption (ability of cellulases to stay on a substrate) of the enzyme. The binding of cellulolytic enzymes is complex due to substrate heterogeneity with areas of differing crystallinity and chemical composition (Park, Baker, Himmel, Parilla, & Johnson, 2010).
Lignocellulosic biomass (i.e. soybean hulls, sugarcane bagasse, rice straw etc.) are heterogenous, mainly composed of cellulose, lignin and hemicellulose, which make the structure compact. Lignin limits the enzyme access to cellulose by forming a physical barrier. It also nonproductively adsorbs enzymes thereby reducing the amount of active enzymes (Ko, Ximenes, Kim, & Ladisch, 2014; Nakagame, Chandra, & Saddler, 2011). The endoglucanases act randomly to hydrolyze amorphous cellulose and soluble derivatives of cellulose. Typically, amorphous cellulose has been reported to be rapidly degraded to cellobiose, while the hydrolysis of crystalline cellulose is slower, and the rate depends on the degree of polymerization and the crystallinity of the cellulose (Park, Baker, Himmel, Parilla, & Johnson, 2010; Hosseini, & Shah, 2011). The initial rapid rate of hydrolysis followed by decay of hydrolysis (Figure 1) could be due to the rapid hydrolysis of the amorphous constituents of the cellulosic substrates. The recalcitrance of the residual material was thought to be due to a higher inherent degree of crystallinity. However, particularly with the saccharification of naturally occurring lignocellulosic, there is a tendency for a substantial amount of the residual cellulosic material to remain in a recalcitrant, insoluble state after hydrolysis is stalled. Based on the above, it was decided to study the lignin removal and cellulose degradation by the microorganism on the different substrates.
Cellulosic composition of substrates for the production of endoglucanase
In this study, Aspergillus niger 426 was grown in solid state cultures of sugarcane bagasse, rice straw and soybean hulls supplemented with MW culture added 1 g L-1 CMC. The agro-industrial residues are cheap carbon sources for the growth of microorganisms, including filamentous fungi, and for holocellulases production. The differences in the complexity of the carbon sources could account for the disparity in cellulase production. In this context, crude supernatants from A. niger cultures were prepared to investigate their ability to degrade lignocellulosic substrates (Table 2). Cellulose and lignin contents were assessed before and after 72 hours of fermentation and are presented in terms of % dry matter (DM) of the starting material. Sugarcane bagasse was found to be particularly rich in cellulose, followed by soybean hulls and rice straw. Notably higher cellulosic composition is ideal for good growth of fungal cultures and cellulase production. Compositional analysis also revealed that the highest lignin content was detected in rice straw and soybean hulls. The highest percentage of lignin degradation by A. niger was found on soybean hulls (56%) followed by sugarcane bagasse (36%) against 8.5% on rice straw. The cellulose degradation, around 90%, was observed on soybean hulls and sugarcane bagasse, but only 50% on rice straw. Thus, cellulose of sugarcane bagasse, rice straw and soybean hulls residue should induce synthesis of the enzyme involved in its breakdown and 3 days fermentation seems to be appropriate to achieve significant reduction of cellulose by A. niger grown in soybean hulls and sugarcane bagasse. Furthermore, soybean hulls and sugarcane bagasse supported the highest endoglucanase productivity (112.34 and 80.56 U mL-1, respectively). This may be due to increased access to cellulose by efficient removal of lignin content and its very high percentage of cellulose.
Substrate selection for endoglucanase production
Table 1 summarizes the different combinations of sugarcane bagasse, rice straw and soybean hulls concentrations used for the A. niger cultivations, and the maximum endoglucanase activities that resulted from the use of EMD. According to this method, the measured response is assumed to depend only on the proportions of the ingredients present in the mixture. Maximum endoglucanase values obtained in the experiments ranged from 67.551 (run 2) to 136.142 U mL-1 (run 7). Among single substrates, soybean hulls supported maximum endoglucanase activity (111.46 U mL-1), which was 18 times higher than that reported by Boggione, Allasia, Bassani, and Farruggia (2016), who used the same substrate and A. niger cultivated under 96 hours of solid-state fermentation. A preliminary analysis of this data revealed the significance of the culture medium components as substrates for endoglucanase biosynthesis, since the least favorable condition was that where rice straw alone was used as carbon source (run 2). Sugarcane bagasse alone produced 17.98% more than rice straw. Better inducing capability of sugarcane bagasse could be attributed to the high content of cellulose of this substrate (Table 1). In contrast, Kogo et al. (2017) reported that rice straw was a viable carbon source for fungal production of cellulases.
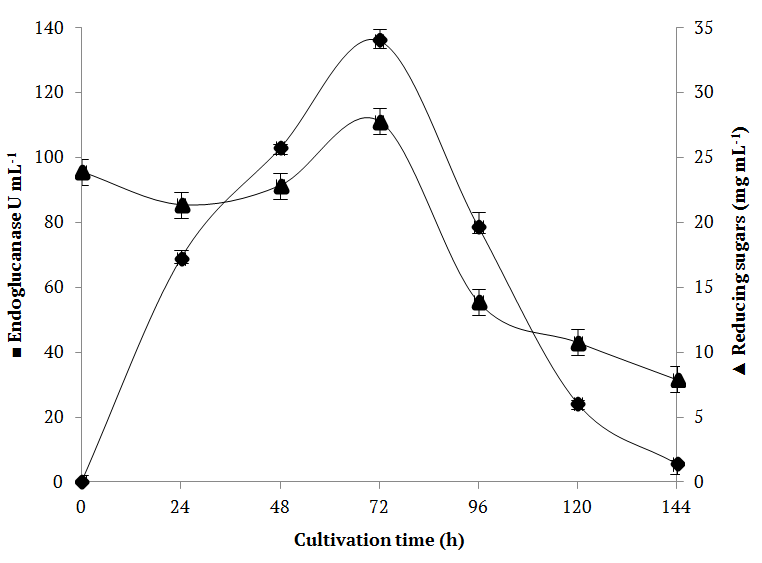
Figure 1.
Endoglucanase production and reducing residual sugar concentration as a function of time, for SSF at 28°C, 70% a.w. and initial pH 5.0 using ternary mixture of EMD. (▲) Reducing sugars (mg mL-1); (■) endoglucanase activity (U mL-1). Results are presented as means ± standard deviation.
| Substrate (10 g of dry weight) | Time (h) | Endoglucanase (U mL-1) | Lignin (%DM) | Cellulose (%DM) |
| Sugarcane bagasse | 0 | 0 | 24.67 ± 0.543 | 44.8 ± 0.14 |
| Rice straw | 0 | 0 | 44.05 ± 0.034 | 32.17 ± 0.962 |
| Soybean hulls | 0 | 0 | 44.59 ± 0.321 | 33.49 ± 1.458 |
| Sugarcane bagasse | 72 | 80.56 ± 0.396 | 15.73 ± 1.112 | 3.66 ± 0.467 |
| Rice straw | 72 | 68.67 ± 0.623 | 40.29 ± 0.012 | 15.89 ± 0.076 |
| Soybean hulls | 72 | 112.34 ± 0.984 | 19.62 ± 1.376 | 2.08 ± 0.209 |
Studies on other applications of lignocellulosic mixtures have demonstrated that combining substrates usually has higher yields than single substrates. Silva et al. (2016) produced endoglucanase and 𝛽-glucosidases from the thermophilic fungus Myceliophthora heterothallica under SSF using a mixture of cardboard or wheat bran in mixture with sugarcane bagasse at 1:1. In medium with cardboard, the endoglucanase production was higher than with wheat bran. This result is explained by the higher percentage of cellulose and lower lignin content found in cardboard. Oke, Annuar, and Simarani (2016) studied bacterial endoglucanase production from single and mixed lignocellulosic substrates by submerged fermentation. The bacterium showed better growth on mixed lignocellulosic than on single substrates. These results were possibly due to the combination of favorable characteristics (e.g., nutrients, cellulose accessibility, etc.) for each individual lignocellulosic material in the mixture.
Some authors reported that enhancement of cellulases production requires a little amount of free sugars in initial fermentation culture. Adeoyo, Pletschke, & Dames (2017) indicated that endoglucanase was induced by glucose and maltose. Bone and Munoz (1984) showed that addition of glucose at levels 5 and 10% of straw weight stimulated lignin degradation and cellulose hydrolysis when Polyporus sp A-336 was cultivated on oat straw under SSF conditions.
The HPLC analysis showed that only soybean hulls have soluble sugars such as glucose in a concentration of 36.969 ± 0.098 mg g-1 of dry substrate and traces of raffinose and stachyose (Table 3), which were possibly used for microorganism growth. This could explain why even though soybean hulls have the highest lignin content (Table 1), which limits the accessibility of the enzyme, it induced greater activity. The lack of soluble sugars in rice straw may be related to the worse degradation of lignin and cellulose. When compared to rice straw better degradation of lignocellulose content occurred on sugarcane bagasse, this can be explained by the content of soluble sucrose (0.110 mg g-1 of dry substrate). Based on these results, it appears that the enzyme induction pattern depends on the nature of the substrate.
The outcome of ANOVA can be visualized in a Pareto chart (Figure 2), in which the absolute value of the magnitude of the standardized estimated effect of each factor is plotted in decreasing order and compared to the minimum magnitude of a statistically significant factor with 95% of confidence (p = 0.05), represented by the vertical dashed line. From this figure it can be observed that all variables were significant for endoglucanase production. The results obtained in this work demonstrated that the order of significance was soybean hulls > sugarcane bagasse > rice straw. For binary interactions, a robust synergism between sugarcane bagasse and soybean hulls can be observed. Endoglucanase production was more significantly affected by the ternary mixture of each substrate followed by the binary mixture of sugarcane bagasse and soybean hulls (p < 0.05).
Finally, the regression equation obtained after the analysis of variance (ANOVA) provides an estimation of the level of endoglucanase production as a function of sugarcane bagasse, rice straw and soybean hulls concentration. The production of endoglucanase may be best predicted by the following Equation 2:
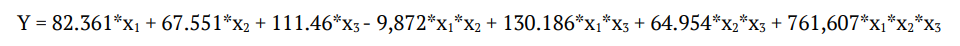 (2)
(2)The ternary combination region between the independent variables x1, x2 and x3 can be observed through the contour lines shown in Figure 3. The Figure 3 shows the boundary regions of the surface response for the endoglucanase production that were obtained using Equation 2. With an R2 adjusted value of 0.9999, these data indicate that the model explains 99.9% of the variation in the experimental data. These results revealed a broad region of elevated endoglucanase production. The analysis also demonstrated that the regions with increased proportions of both sugarcane bagasse and rice straw exhibited decreased endoglucanase activity and soybean hulls had a greater influence on enzyme production; its presence in the cultures was essential for increased activity.
Using our data, a predictive analysis estimated the maximum endoglucanase activity of 140.04 U mL-1 in cultures that contained 25 sugarcane bagasse, 23 rice straw and 52% soybean hulls. An additional culture was performed to validate the proposed model and yielded an endoglucanase activity level of 138.92 ± 0.02 U mL-1. This result corresponded to 99% of the expected value, validating the effectiveness of the predictive model and confirming the substrates proportions.
| Substrate | Glucose | Sucrose | Raffinose | Stachyose |
| Sugarcane bagasse | * | 0.110 ± 0.007 | ND | ND |
| Soybean hulls | 36.969 ± 0.208 | 0.098 ± 0.010 | * | * |
| Rice straw | * | ND | ND | ND |
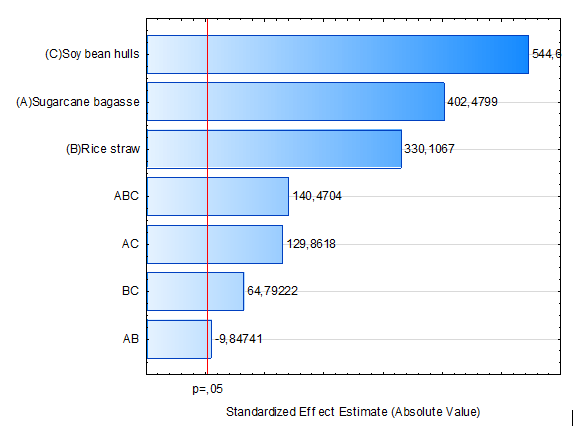
Figure 2.
Pareto chart showing the order of significant substrates variables.
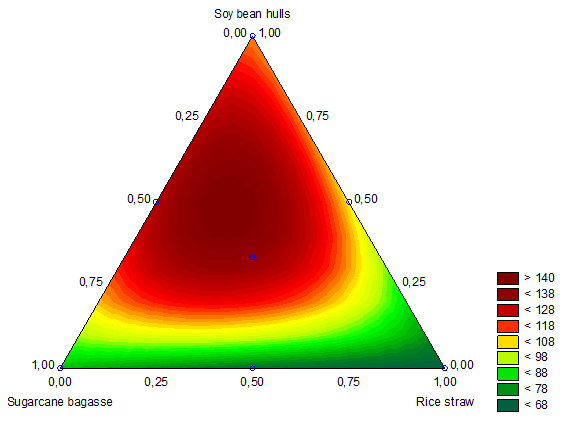
Figure 3.
Contour plots of endoglucanase response for the A. niger Experimental mixture design, using the culture medium components sugarcane bagasse, rice straw and soybean hulls.
The carbon source has been estimated as a major cost factor in enzyme production. Industrial application of endoglucanase, however, would only be feasible if the carbon sources were available in significant quantities at competitive price. A reduction of the production cost can be achieved by the usage of inexpensive crude raw materials, which are cheap, easily available and often abundant alternative for large-scale fermentation.
Conclusion
An Experimental Mixture Design (EMD) approach was used to improve endoglucanase production by A. niger 426 in solid state fermentation. The results demonstrated that soybean hulls were the most significant component of the culture medium for maximization of endoglucanase activity. It was also observed that soluble sugars played a key role in endoglucanase biosynthesis. In addition, A. niger was able to remove 56% of lignin from soybean hulls and hence increase the accessibility of cellulose to cellulolytic enzymes. Overall, it has been shown that A. niger 426 is a potentially valuable microorganism for cellulolytic enzymes production to be used for biomass deconstruction.
Acknowledgements
The authors are grateful to Capes for financial support of this work and scholarships.
References
Adeoyo, O. R., Pletschke, B. I., & Dames, J. F. (2017). Improved endoglucanase production and mycelial biomass of some ericoid fungi. AMB Express, .(1).doi:10.1186/s13568-016-0312-y
Adnan, M., Zheng, W., Islam, W., Arif, M., Abubakar, Y., Wang, Z., & Lu, G. (2017). Carbon catabolite repression in filamentous fungi. International Journal of Molecular Sciences, 19(1), 48. doi:10.3390/ijms19010048
Anwar, Z., Gulfraz, M., & Irshad, M. (2014). Agro-industrial lignocellulosic biomass a key to unlock the future bio-energy: A brief review. Journal of Radiation Research and Applied Sciences, 7(2), 163-173. doi:10.1016/j.jrras.2014.02.003
Association of Official Analytical Chemists [AOAC]. (1995). Official methods of analysis of the association of the analytical chemists(6th ed.). Washington, DC: AOAC.
Boggione, M. J., Allasia, M. B., Bassani, G., & Farruggia, B. (2016). Potential use of soybean hulls and waste paper as supports in SSF for cellulase production by Aspergillus niger. Biocatalysisand Agricultural Biotechnology, 6, 1-8. doi: 10.1016/j.bcab.2016.02.003
Bone, D. H., & Munoz, E. (1984). Solid state fermentation of oat straw by polyporus SP A-336 and the effect of added sugars. Biotechnology Letters, 6(10), 657-662. doi: 10.1007/BF00133832
Cornell, J. A. (2002). Experiments with mixtures: designs, models and the analysis of mixture data (3rd ed.). New York, NY: John Wiley and Sons.
Delabona, P. S., Farinas, C. S., Lima, D. J. S., Azzoni, S. F., & Pradella, J. G. C. (2013). Experimental mixture design as a tool to enhance glycosyl hydrolases production by a new Trichoderma harzianum P49P11 strain cultivated under controlled bioreactor submerged fermentation. Bioresource Technology, 132, 401-415. doi: 10.1016/j.biortech.2012.11.087
Ekwe, E., Morgenstern, I., Tsang, A., Storms, R., & Powlowski, J. (2013). non-hydrolytic cellulose active proteins: research progress and potential application in biorefineries. Industrial Biotechnology, 9(3), 123–131. doi:10.1089/ind.2013.0010
Ghose, T. K. (1987). Measurement of cellulase activities. Pure and Applied Chemistry, 59(2), 257-268. doi: 10.1351/pac198759020257
Hafiz, M. N., I., Ishtiaq, A., Muhammad, A. Z., & Muhammad, I. (2011). Purification and characterization of the kinetic parameters of cellulase produced from wheat straw by Trichoderma viride under SSF and its detergent compatibility. Advances in Bioscience and Biotechnology, 2(3), 149-156. doi: 10.4236/abb.2011.23024
Harris, D., & DeBolt, S. (2010). Synthesis, regulation and utilization of lignocellulosic biomass. Plant Biotechnology Journal, 8(3), 244–262. doi:10.1111/j.1467-7652.2009.00481.x
Hingsamer, M., & Jungmeier, G. (2019). Biorefineries. The Role of Bioenergy in the Bioeconomy, 179–222. doi:10.1016/b978-0-12-813056-8.00005-4
Hosseini, S. A., & Shah, N. (2011). Modelling enzymatic hydrolysis of cellulose part I: Population balance modelling of hydrolysis by endoglucanase. Biomass and Bioenergy, 35(9), 3841-3848. doi:10.1016/j.biombioe.2011.04.026
Ko, J. K., Ximenes, E., Kim, Y., & Ladisch, M. R. (2014). Adsorption of enzyme onto lignins of liquid hot water pretreated hardwoods. Biotechnology and Bioengineering, 112(3), 447-456. doi:10.1002/bit.25359
Kobakhidze, A., Asatiani, M., Kachlishvili, E., & Elisashvili, V. (2016). Induction and catabolite repression of cellulase and xylanase synthesis in the selected white-rot basidiomycetes. Annals of Agrarian Science, 14(3), 169–176. doi:10.1016/j.aasci.2016.07.001
Kogo, T., Yoshida, Y., Koganei, K., Matsumoto, H., Watanabe, T., Ogihara, J., & Kasumi, T. (2017). Production of rice straw hydrolysis enzymes by the fungi Trichoderma reeseiand Humicola insolensusing rice straw as a carbon source. Bioresource Technology, 233, 67–73.doi: 10.1016/j.biortech.2017.01.075
Mandels, M., & Weber, J. (1969). The production of cellulases. Advances in Chemistry Series, 95, 391-414. doi: 10.1021/ba-1969-0095.ch023
Miller, G. L. (1959). Use of dinitrosalicylic acid reagent for determination of reducing sugar. Analytical Chemistry, 31(3), 426-428. doi: 10.1021/ac60147a030
Mrudula, S., & Murugammal, R. (2011). Production of cellulase by Aspergillus niger under submerged and solid state fermentation using coir waste as a substrate. Brazilian Journal of Microbiology, 42(3), 1119-1127. doi: 10.1590/S1517-83822011000300033
Nakagame, S., Chandra, R. P., & Saddler, J. N. (2011). The Influence of Lignin on the Enzymatic Hydrolysis of Pretreated Biomass Substrates. Sustainable Production of Fuels, Chemicals, and Fibers from Forest Biomass, 145–167.doi:10.1021/bk-2011-1067.ch006
Oke, M. A., Annuar, M. S. M., & Simarani, K. (2016). Enhanced endoglucanase production by Bacillus aeriuson mixed lignocellulosic substrates. Biores, 11(3), 5854-5869. doi: 10.15376/biores.11.3.5854-5869
Park, S., Baker, J. O., Himmel, M. E., Parilla, P. A., & Johnson, D. K. (2010). Cellulose crystallinity index: measurement techniques and their impact on interpreting cellulase performance. Biotechnology for Biofuels, 3(1). 10. doi:10.1186/1754-6834-3-10
Saravitz, D. M., Pharr, D. M., & Carter, T. E. (1987). Galactinol synthase activity and soluble sugars in developing seeds of four soybean genotypes. Plant Physiology, 83(1), 185-189. doi: 10.1104/pp.83.1.185
Segato, F., Damásio, A. R. L., de Lucas, R. C., Squina, F. M., & Prade, R. A. (2014). Genomics Review of Holocellulose Deconstruction by Aspergilli. Microbiology and Molecular Biology Reviews, 78(4), 588–613. doi:10.1128/mmbr.00019-14
Silva, V. C. T., Coto, A. L. S., Souza, R. C., Neves, M. B. S., Gomes, E., & Bonilla-Rodriguez, G. O. (2016). Effect of pH, temperature, and chemicals on the endoglucanases and 𝛽-Glucosidases from the thermophilic fungus Myceliophthora heterothallicaF.2.1.4. Obtained by solid-state and submerged cultivation. Biochemistry Research International, 2016, 1-9. doi: 10.1155/2016/9781216
Suwannarangsee, S., Bunterngsook, B., Arnthong, J., Paemanee, A., Thamchaipenet, A., Eurwilaichitr, L., & Champreda, V. (2012). Optimisation of synergistic biomass-degrading enzyme systems for efficient rice straw hydrolysis using an experimental mixture design. Bioresource Technology, 119, 252-261. doi: 10.1016/j.biortech.2012.05.098
Van Soest, P. J. (1963). Use of detergents in the analyses of fibrous feeds II: A rapid method for determination of fiber and lignin. Journal Association of Analytical Chemistry, 46(5), 829-835.