Science, Food Technology and Food Engineering
Improvement of lipid quality on nile tilapia fillet composition with low protein feeding treatment
Improvement of lipid quality on nile tilapia fillet composition with low protein feeding treatment
Acta Scientiarum. Technology, vol. 42, e45271, 2020
Universidade Estadual de Maringá

Esta obra está bajo una Licencia Creative Commons Atribución 4.0 Internacional.
Recepción: 11 Agosto 2018
Aprobación: 25 Julio 2019
Abstract: The lipid composition is directly related to nutritious significance of fish meat. In this paper we evaluate the possibility of tuning lipid composition of Nile Tilapia fillet changing the feeding treatments with different levels of energy and protein using lipidomic approach. Five feeding treatments were used varying protein and energy content with soy protein, corn carbohydrates and soy oil. Easy Ambient Sonic-Spray Ionization mass spectrometry in negative mode was used for lipidomic characterization of polar lipids in Nile Tilapia fillet, that were extracted using the Bligh & Dyer method and evaluated by principal component analyzes revealing differences over the treatments. High Resolution Mass Spectrometry data obtained were imported into Lipid Maps (www.lipidmaps.org, accessed on 10/19/2018) for molecular identification. The major influence to separate the treatments was protein content, energy does not appear to differentiate the groups. Thirteen different ions were observed as the major differentiators of the groups and nine compounds were identified comparing the mass spectrometric results to Lipidomic library, and they are related to high metabolic activity, lipids with anti-diabetic effect, and fat levels reducers.
Keywords: functional food, lipid composition, Oreochromis niloticus, EASI-HRMS.
Introduction
Mass spectrometry (MS) is a centenary analytical tool with many kinds of applications and has become fundamental in applied research providing key information on the exploration of a great variety of materials in virtually all-experimental fields. Among MS techniques, ambient ionization such as Desorption Electrospray Ionization (DESI) has been widely applied for analyses of surfaces (Takáts, Wiseman, Gologan, & Cooks, 2004) such as, environmental analysis (Gerbig, Brunn, Spengler, & Schulz, 2015), microbiology (Watrous & Dorrestein, 2011; Rodrigues, Prova, Moraes, & Ifa, 2018), medicine (Eberlin et al., 2012; D'Hue et al., 2018), and food chemistry (Yang et al., 2009; Gerbig, Neese, Penner, Spengler, & Schulz, 2017). A gentler ambient ionization technique is called Easy Ambient Sonic-Spray Ionization (EASI) had been developed and has been used in many areas, such as, perfume fingerprint (Haddad, Milagre, Catharino, & Eberlin, 2008).
EASI source produce charged droplets at atmospheric pressure that result from statistical (unbalanced) charge distribution during droplet formation in the supersonic pneumatic spray. These charged droplets collide to the sample surface and the molecules on the surface are both ionized and desorbed, and ions subsequently enter the high vacuum part of the mass spectrometer. EASI produce neither degradation nor lateral reaction and mass spectrum in this technique represents the true lipid profile. Lipids are easily ionized using EASI and can be analyzed directly from vegetable and animal fat (Riccio et al., 2010). Muscle lipids are present at a low concentration in this kind of tissue and the direct analysis using EASI is therefore not suitable. To access the information provided by these compounds in muscle tissue, it is necessary to extract them using organic solvent. It was showed (Chramow, Hamid, Eberlin, Girod, & Ifa, 2014) that also with a DESI source, the major signals are obtained from organs containing high lipid concentrations and not from muscle tissue.
Nile Tilapia fillet (Oreochromis niloticus) is the fourth most produced fish around the world (Food and Agriculture Organization [FAO], 2018) and the research to enhancement of the quality of the lipids from this source of food might interest both consumers and producers. The success of the Tilapia production may be attributed to its adaptability to extensive cultivation with low to none artificial feeding, and intensive cultivation that include rich and complete feeding (Watanabe, Losordo, Fitzsimmons, & Hanley, 2002). Corporeal growing of fish demands nutrients and energy, among which proteins are the most important, being the precursor of amino acids. Energy may come from different resources, such as, oxidation of carbohydrates, lipids, and proteins. The study of protein and energy variation in food may directly affect the corporal composition of Nilo Tilapia, and especially its consumed part, the Filet.
In this research we propose a tuning of lipid content of Nile Tilapia fillet using energy protein feeding treatments variables evaluated by an EASI High Resolution Mass Spectrometry (EASI-HRMS) screening method. The data treatment was performed using principal component analysis (PCA) to evaluate the data and HRMS to obtain the identification of polar lipids with lipidomic library, with qualitative proposes. The methodology shown herein was able to determine 13 ions that directly were related to samples differentiation and nine identified molecules. The samples were discriminated using PCA and demonstrated the aquiculture applicability of the screening of polar lipids to evaluate the result of different feeding treatments in lipid composition of Nile Tilapia fillet.
Material and methods
Nile Tilapia treatments
120 Nile Tilapias, with initial mean weights of 486.1 g were distributed randomly in 5 treatments, with three repetitions, and each experimental unit constituted of 8 fishes in 1,000 L inert tanks, with physical and biological filtration to remove residues and feces.
Fishes were submitted to five feeding treatments with different digestible protein (DP) and energy (DE) levels (14% DP and 3,200 DE; 19% DP and 3,200 DE; 24% DP and 2,800 DE; 34% DP and 2,800 DE; 34% DP and 3,000 DE). The feeding composition contains soybean meal, corn, soy oil, bicalcium phosphate, limestone, vitamin-mineral premix and salt. For the manufacture of animal food ingredients were milled in a knife mill with 0.5 mm sieve, mixed according to the formulation (Table 1) and processed by extrusion.
At the end of the experiment, the fish were fasted for 24 hours, anesthetized in ice water, slaughtered, filleted and stored frozen for subsequent homogenization and sample withdrawal. The fillet samples were analyzed for total lipid content and molecular lipid composition using mass spectrometry. For total lipid content analyses it was conducted ethylic ether extraction.
Mass spectrometry
Data was acquired using an EASI source coupled to a Thermo Scientific Q Exactive Hybrid Quadrupole-Orbitrap Mass Spectrometer operating in the positive and negative mode at a resolution of 70,000. Data was acquired in the range of 100-1200 m/z. The EASI source consisted of an optimized prototype as described earlier (Haddad et al., 2006). Primary conditions were as follows: methanol flow rate set at 10 μL min.-1, nebulizing gas backpressure at 100 psi and a capillary temperature at 320ºC. Data was processed using Thermo Xcaliber Qual Browser version 2.4.0.1824. High Resolution Mass Spectrometry (HR-MS) was used to access exact molecular masses that were inserted into the Lipid Maps (www.lipidmaps.org, accessed on 10/19/2018) to compare to the molecular formula database from Lipids, using [M-H]- species with tolerance of +/- 0.05 of mass.
Sample preparation
The Bligh & Dyer method (1959), with modifications, was used for lipid extraction. 1.0 g of sample was weighed in a 10 mL screw-cap extraction tube, to which 2.0 methanol and 1.0 mL chloroform were added. The mixture was agitated in a tube agitator for 5 min. 1.0 mL of chloroform was then added and the mixture was subsequently agitated for another 2 min. The mixture was then centrifuged at 14,000 rpm for 5 min. After phase separation, the lower phase was transferred to a 2 mL Eppendorf tube and subsequently stored at -18°C until analysis.
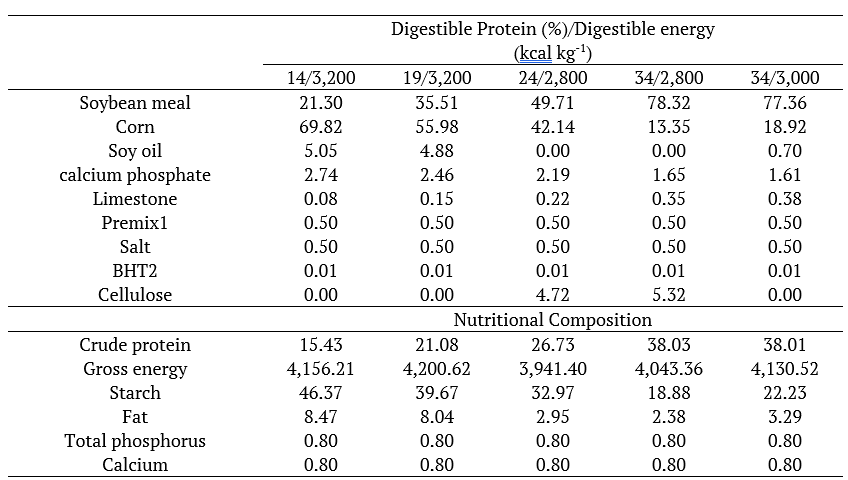
1vitamin-mineral premix; 2Butyl hydroxyl toluene (antioxidant).
Sample analysis
A volume of 2 μL lipid extract was placed on paper and allowed to dry. The surface of blank paper and subsequently the dried lipid extract was then analyzed using EASI-MS, for the duration of approximately 5 s each. A background subtraction was performed of the paper with and without dried lipid extract to obtain a clean spectrum of the pure lipids. The total analysis time from sample deposition until and including background subtraction was approximately 30 s.
PCA analysis
For the data evaluation it was conducted Principal Component Analysis (PCA) (Petenuci et al., 2018). The purpose of PCA is to reduce the dimensionality of a data set formed by interrelated variables, conserving the variation of the data as much as possible. This is achieved through a transformation to a new set of non-correlated variables known as principal components, ordered from those that retain the most to those that retain the least variation present in all the original variables. The calculation of the PCs is the solution to the problem of eigenvalues and eigenvectors in a positive definite symmetric matrix. Usually, a subset of principal components is selected (in order) in a way that best represents the original set of data, but with fewer dimensions. The technique is particularly useful if the number of principal components selected is considerably lower than the number of original values (highly reduced dimensionality). PCA avoid problems linked to multicollinearity (variables that are highly correlated with one another) among the prediction attributes. The present paper applies PCA for visualization and comparative analysis between the Nile Tilapia treatments groups obtained from the MS data
Results and discussion
Total fat composition were 2.76% for 14/3200 (Protein%/Energy in kcal) treatment, 0.69 for 19/3200 treatment, 0.47 for 24/3200 treatment, 0.48 for 34/2800 treatment, and 0.45% for 34/3000 treatment. It seems that the high percentage of accumulated fat is related to higher level of energy and lower levels of proteins, interestingly increasing protein in feeding reflect to the amount of fat decrease, as observed. The total lipid analysis is very useful in aquaculture research but this bulk analysis of lipid content cannot access the molecular composition of polar lipids that might be helpful on the quality analysis of NTF. To access molecular composition EASI-MS was used to verify if energy/protein feeding treatments would be able to tune different lipid profiles from Nile Tilapia Fillet and if there is any change, what would that be.
Molecular content was accessed using EASI-HRMS experiments, which were carried out obtaining high-resolution spectra in positive and negative modes. The purpose was to obtain molecular speciation information of the samples to evaluate the polar lipid classes in the ‘filet’ fat fraction. Positive mode spectra were poorly reproducible despite the high intensity signals. Negative mode spectra were therefore used since these were highly reproducible despite their significantly lower intensity. Negative mode mass spectra of all treatments presented free fatty acids with no major changes. Figure 1 represents a clear example of an EASI-HRMS profile in a range with the observed changes for the treatments analyzed from extracted lipids from fish that were treated with high energy and high protein containing food.
The mass spectrum is clearly divided into two groups of ions. These are: i) fatty acid esters of hydroxy Fatty Acids and diacylglycerols, present between m/z 400 and m/z 700, and ii) polar substituted acyl glycerol, present above m/z 700.
Figure 2 shows the results of the PCA (plot of the first two columns of scores, i.e. the ratings data projected onto the first two principal components, PC1 and PC2) for the 14 samples of Nile Tilapia feed with five distinct treatments. It would be 15 samples (3 samples by treatment), unfortunately, the fish feed with medium protein and high energy was lost during the experiments.
Figure 2 shows a scatter plot of PC2 versus PC1 from the data matrix obtained from EASI-HRMS data. The replicates of each type of feeding treatment of Nile Tilapia showed an expected biological variation even then PCA clearly grouped the samples in two distinct groups: highest feed protein content (34), and other protein (24,19,14), there was just one sample (34-2800) with the highest feed protein content that appears dislocated, probably due to biological or analyses error. The two groups suggested might be observed in the two PC1 quadrants. Different energy contents did not group the samples in any quadrant. Figure 3 complements the PCA analyses presenting the ions that represent best each group.
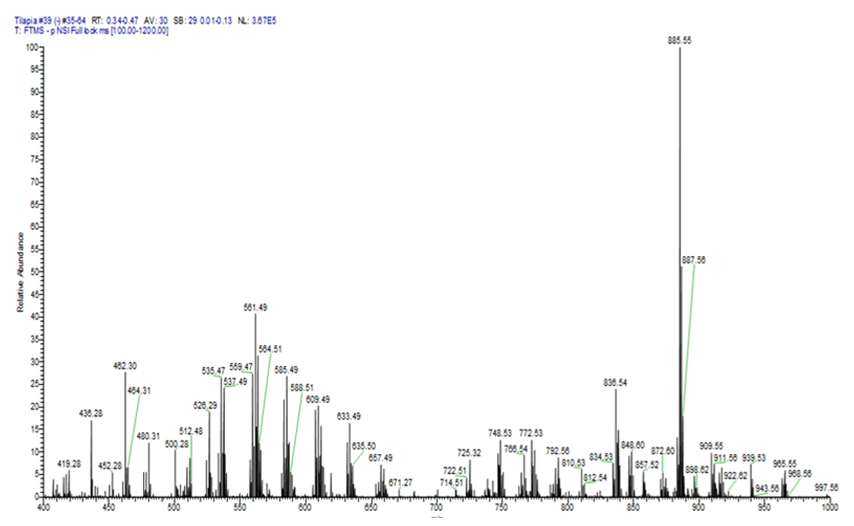
Figure 1.
EASI -HRMS of lipid extract from high energy and high protein treatment.
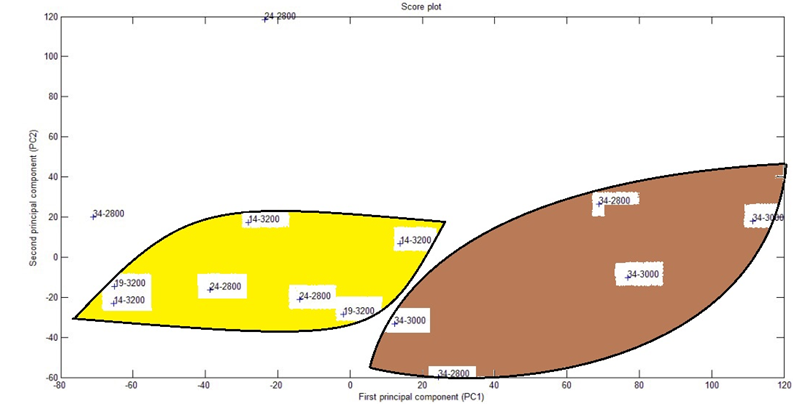
Figure 2.
Principal Component Analysis for the data of the 14 samples of Nile Tilapia feed with five distinct blends of protein and energy.
Figure 3 showed the loadings of the analyzed samples and the ion correspondent to PI (38:4) was the one that best represent the highest feed protein content samples, the lower feed protein content samples were better represented by DG (32:3), FAHFA (36:1), Mayolene-18, FAHFA (34:1), the ion of m/z 562.49, DG (34:5), DG (35:6), FAHFA (34:0), PC (20:0), the ion of m/z 434.05, the ion of m/z 794.26, the ion of m/z 848.32. Figure 4 showed the VIP ions that differentiate the groups.
Thirteen ions were used to distinguish between NTF samples from different feed treatments (Table 1). Nine ions were identified (Figure 4) and some were not. Overall behavior shows more intense ions for high energy feed treatment, as expected. The species were labelled using the high resolution mass obtained for each ion observed.
The results showed a variety of compounds that may affect the quality of the lipid content of the NTF that might be involved to the nutraceutical properties of the final product, and the results suggest that the feeding treatment affect efficiently the final lipid composition. For the samples coming from the highest feeding protein treatment content, phosphatidylinositol (38:4) was found as the molecule that most differentiate the high protein treatment with higher intensity when compared to low protein treatment, this lipid is synthesized by phosphatitilinusitol 3 kinase that might be more activated, phosphatitilinusitol 3 kinase is a family of enzymes involved in cellular functions such as cell growth, proliferation, differentiation, motility, survival and intracellular trafficking, which in turn are involved in cancer (Craene, Bertazzi, Bär, & Friant, 2017).
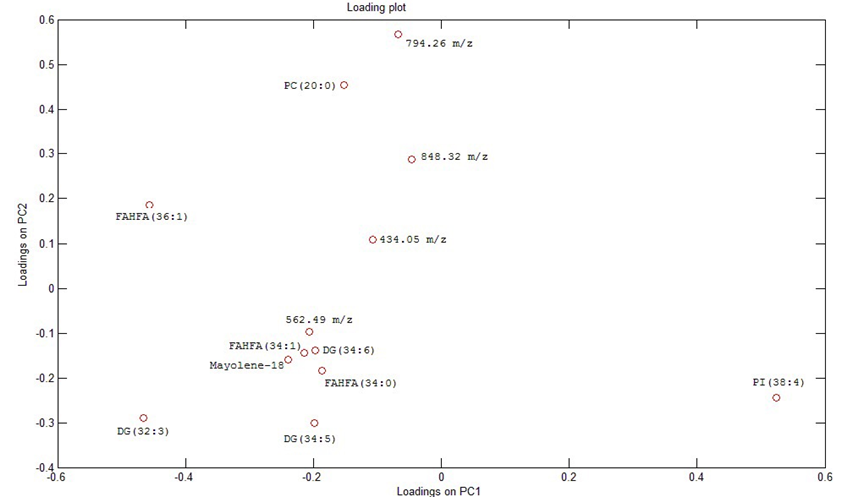
Figure 3.
Loadings of PCA analysis with 14 samples of Nile Tilapia feed with five distinct blends of protein and energy.
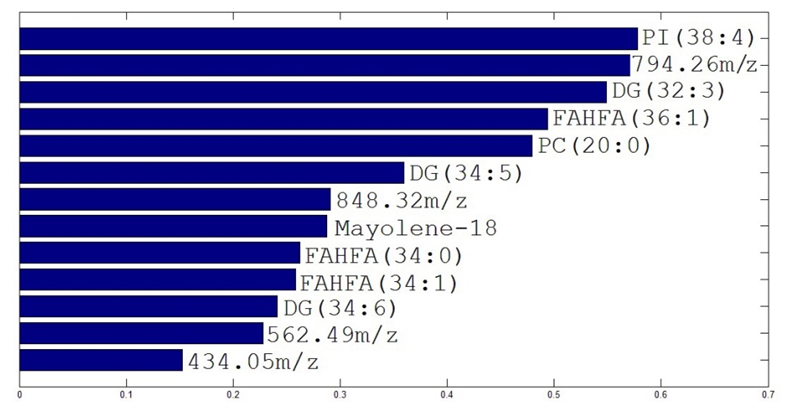
Figure 4.
VIP ions extracted from the loadings of PCA analysis.
For the samples coming from the lower feeding protein treatment content, the molecules DAG, PC, FAHFA have higher intensities and were found as indicators from low protein treatments. Diacyl glycerols are know to be related to beneficial effects on obesity that reduce the accumulation of body fat in animals and humans (Lo, Tan, Long, Yusoff, & Lai, 2008). Phosphatidyl choline found enhanced in low protein treatment NTF was tested as a lipid based therapy for ulcerative colitis toward the modulation of intestinal mucus membrane due to the anti-Inflammatory signaling by PC (Schneider, Braun, Füllekrug, Stremmel, & Ehehalt, 2010). Fatty acid esters of hydroxy fatty acids that appears to be enhanced on low protein treatment NTF was found with potential to treat type 2 diabetes (Yore et al., 2014). The ions not identified need further analysis using MS/MS approach that might help the identification and confirmation if there were isomers present. Since we used high resolution for identification, isomers with the same molecular formula may have been identified as the same molecule.
Conclusion
In this study, the tuning of lipid content of the Nile Tilapia Filet was achieved; the use of EASI-HRMS to investigate Nile Tilapia lipid extract was effective. The simple adjusting of the feeding treatment turns the approach easy to improve the quality of the lipids of the fish meat. It is verified that the lipid profile changes from low protein to high protein feed treatment, and energy feed treatment did not differentiate the acyl glycerol profile. The low protein feeding treatment for Nile Tilapia increased nutraceutical important lipids, such as, DAG, PC, and FAHFA. The use of EASI as a soft ambient ionization technique greatly facilitates the study of biological tissues by reducing time needed for sample analysis to a mere 30 seconds without standards. The ability to analyze whole organisms could allow for future research on distribution of the lipids in different growing phases of Nile Tilapias to verify nutritional and signaling matters for this very important kind of fish.
Acknowledgements
The authors are grateful to the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (Capes), Araucaria Foundation, Fapesp, and UFPR for the financial support.
References
Bligh, E. G., & Dyer, W. J. (1959). A rapid method of total lipid extraction and purification. Canadian Journal of Biochemistry and Physiology, 37(8), 911-917. doi: 10.1139/y59-099
Chramow, A., Hamid, T. S., Eberlin, L. S., Girod, M., & Ifa, D. R. (2014). Imaging of whole zebra fish (Danio rerio) by desorption electrospray ionization mass spectrometry. Rapid Communication in Mass Spectrometry, 28(19), 2084-2088. doi: 10.1002/rcm.6993
Craene, J.-O., Bertazzi, D. L., Bär, S., & Friant, S. (2017). Phosphoinositides, major actors in membrane trafficking and lipid signaling pathways. International Journal of Molecular Science, 18(3), 634. doi: 10.3390/ijms18030634
D'Hue, C., Moore, M., Summerlin, D. J., Jarmusch, A., Alfaro, C., Mantravadi, A., ... Cooks, R. G. (2018). Feasibility of desorption electrospray ionization mass spectrometry for diagnosis of oral tongue squamous cell carcinoma. Rapid Communication in Mass Spectrometry, 32(2), 133-141. doi: 10.1002/rcm.8019
Eberlin, L. S., Norton, I., Dill, A. L., Golby, A. J., Ligon, K. L., Santagata, S., ... Agar, N. Y. (2012). Classifying human brain tumors by lipid imaging with mass spectrometry. Cancer Research, 72(3), 645-654. doi: 10.1158/0008-5472.CAN-11-2465
Food and Agriculture Organization [FAO]. (2018). The state of world fisheries and aquaculture. Roma, IT: FAO. Retrieved from http://www.fao.org/3/i9540en/I9540EN.pdf
Gerbig, S., Brunn, H. E., Spengler, B., & Schulz, S. (2015). Spatially resolved investigation of systemic and contact pesticides in plant material by desorption electrospray ionization mass spectrometry imaging (DESI-MSI). Analytical and Bioanalytical Chemistry, 407(24), 7379-7389. doi: 10.1007/s00216-015-8900-2
Gerbig, S., Neese, S., Penner, A., Spengler, B., & Schulz, S. (2017). Real-time food authentication using a miniature mass spectrometer. Analytical Chemistry, 89(20), 10717-10725. doi: 10.1021/acs.analchem.7b01689
Haddad, R., Milagre, H. M. S., Catharino, R. R., & Eberlin, M. N. (2008). Easy ambient sonic-spray ionization mass spectrometry combined with thin-layer chromatography. Analytical Chemistry, 80(8), 2744-2750. doi: 10.1021/ac702216q
Lo, S.-K., Tan, C. P., Long, K., Yusoff, M. S. A., & Lai, O. M. (2008). Diacylglycerol oil-properties, processes and products: a review. Food Bioprocess Technology, 1(3), 223-233. doi: 10.1007/s11947-007-0049-3
Petenuci, M. E., Schneider, V. V. A., Lopes, A. P., Gonçalves, R. M., Santos, V. J., Matsushita, M., & Visentainer, J. V. (2018). Effect of alpha-linolenic acid sources in diets for nile Tilapia on fatty acid composition of fish fillet using principal component analysis. Journal of Aquatic Food Product Technology, 27(4), 464-476. doi: 10.1080/10498850.2018.1448917
Riccio, M. F., Saraiva, S. A., Marques, L. A., Alberici, R., Haddad, R., Moller, J. C., ... Catharino, R. R. (2010). Easy mass spectrometry for metabolomics and quality control of vegetable and animal fats. European Journal of Lipid Science and Technology, 112(4), 434-438. doi: 10.1002/ejlt.200900090
Rodrigues, J. P., Prova, S. S., Moraes, L. A. B., & Ifa, D. R. (2018). Characterization and mapping of secondary metabolites of Streptomyces sp. from caatinga by desorption electrospray ionization mass spectrometry (DESI-MS). Analytical and Bioanalytical Chemistry, 410(27), 7135-7144. doi: 10.1007/s00216-018-1315-0
Schneider, H., Braun, A., Füllekrug, J., Stremmel, W., & Ehehalt, R. (2010). Lipid based therapy for ulcerative colitis-modulation of intestinal mucus membrane phospholipids as a tool to influence inflammation. International Journal of Molecular Science, 11(10), 4149-4164. doi: 10.3390/ijms11104149
Takáts, Z., Wiseman, J. M., Gologan, B., & Cooks, R. G. (2004). Mass spectrometry sampling under ambient conditions with desorption electrospray ionization. Science, 306(5695), 471-473. doi: 10.1126/science.1104404
Watanabe, W. O., Losordo, T. M., Fitzsimmons, K., & Hanley, F. (2002). Tilapia production systems in the Americas: technological advances, trends, and challenges. Reviews in Fisheries Science, 10(3), 465-498. doi: 10.1080/20026491051758
Watrous, J. D., & Dorrestein, P. C. (2011). Imaging mass spectrometry in microbiology. Nature Reviews in Microbiology, 9(9), 683-694. doi: 10.1038/nrmicro2634
Yang, S., Ding, J., Zheng, J., Hu, B., Li, J., Chen, H., ... Qiao, X. (2009). Detection of melamine in milk products by surface desorption atmospheric pressure chemical ionization mass spectrometry. Analytical Chemistry, 81(7), 2426-2436. doi: 10.1021/ac900063u
Yore, M. M., Syed, I., Moraes-Vieira, P. M., Zhang, T., Herman, M. A., Homan, E. A., ... Kahn, B. B. (2014). Discovery of a class of endogenous mammalian lipids with anti-diabetic and anti-inflammatory effects. Cells, 159(2), 318-332. doi: 10.1016/j.cell.2014.09.035
Notas de autor
eduardo.meurer@gmail.com