Biotecnologia
Bioactive compounds in the leaves of Baccharis dracunculifolia: extraction process and characterization
Bioactive compounds in the leaves of Baccharis dracunculifolia: extraction process and characterization
Acta Scientiarum. Technology, vol. 43, e49826, 2021
Universidade Estadual de Maringá
Recepción: 03 Septiembre 2019
Aprobación: 22 Abril 2020
Abstract: Baccharis dracunculifolia has been used in popular medicine because of its bioactive properties, which include anti-inflammatory, antiulcerative and hepatoprotective potentials. A 2³ central composite rotational design was used to define the best conditions for extracting the bioactive compounds from the leaves of Baccharis dracunculifolia. The parameters studied included the solvent concentration, time and temperature of extraction and the influence of these on the total phenolics content and antioxidant activity. The best conditions were 95% methanol (v/v), 20 min extraction time, and 70°C temperature. Under the optimized conditions, high contents of total phenolic compounds (72.5 mg GAE g-1) and high DPPH (865.6 µmol TE g-1) and ABTS (331.11 μmol TE g-1) free radical scavenging capacity and a high ferric reducing antioxidant power (1558.00 μmol FeSO..7H.O g-1), were obtained. The extract also showed promising anti-microbial activity, demonstrating fungistatic action against C. tropicalis and C. albicans (5 mg mL-1 and 7 mg mL-1, respectively) and bacteriostatic effect against Escherichia coli (60 e 70 mg mL-1), Salmonella enterica typhimurium (40; 60 and 70 mg mL-1) and Staphylococcus aureus (4 mg mL-1). HPLC-DAD analysis showed an extract rich in the phenolic compounds catechin (0.659 mg g-1), caffeic acid (1.135 mg g-1), .-coumaric acid (0.772 mg g-1), ferulic acid (6.24 mg g-1), pinocembrina (0.155 mg g-1) and rutin (0.037 mg g-1).
Keywords: biological activity, experimental design, leaf extracts, natural products.
Introduction
Baccharis dracunculifolia is a shrub of the plant family Asteraceae, native to South America, where it is popularly known as vassourinha do campo, or alecrim do campo (field rosemary). This plant species has been used as an alternative and complementary medicine to treat human disease conditions. B. dracunculifolia is also the main raw material of green propolis, a high-valued honeybee (Apis mellifera) bioproduct on the international market (Cestari, Bastos, & Di Stasi, 2011; Figueiredo-Rinhel et al., 2013; Santos et al., 2010).
In the scientific literature, different biological functions have been reported for B. dracunculifolia, including anti-inflammatory activity (Santos et al., 2010), antioxidant capacity, protective activity against oxidative mitochondrial damage (Guimarães et al., 2012), and bactericidal (Pereira, Costa, Liporini, Rego, & Jorge, 2016) and antidiabetic properties (Hocayen et al., 2016). The medicinal properties of this plant are related to several phytochemical compounds present in its extracts, as well as its essential oils. In this context, fourteen volatile compounds have been identified in the essential oils fraction from this plant (Sforcin et al., 2012). Similarly, thirteen flavonoids, and especially 3,5-diprenyl-p-coumaric acid (artepillin C) were identified in ethanolic extracts of leaf buds from B. dracunculifolia (Maróstica Junior et al., 2008).
Many biological properties of the phytochemicals of B. dracunculifolia are associated with the antioxidant capacity of the phenolic compounds present. Phenolic compounds are a class of chemicals that have free hydroxyl groups attached to an aromatic (benzyl) ring, and are specialized plant metabolites that act as biochemical defense agents against pathogens, and UV radiation (Manach, Scalbert, Morand, Rémésy, & Jiménez, 2004). The antioxidant ability of phenolic compounds is directly linked to their oxidation-reduction potential, in addition to their ability to neutralize and absorb free radicals, decompose peroxides, and chelate triplet and singlet oxygen species (Boutennoun et al., 2017). Antioxidant substances may contribute to improve hyperglycemia in type 1 diabetes (mellitus), considering that this dysfunction leads to decreased endogenous antioxidant defenses, and increased reactive oxygen species (ROS) and free radicals (Matough, Budin, Hamid, Alwahaibi, & Mohamed, 2012).
Free radicals are chemical species that have unpaired electrons, are usually quite reactive, and therefore, may cause damage to cells. Cellular damage caused by free radicals, however, can be prevented by antioxidants, which will quickly eliminate them before their destructive action (Fatehi-Hassanabad, Chan, & Furman, 2010; Ullah, Khan Abad, & Khan, 2016). In this context, many studies have been described on B. dracunculifolia as a medicinal plant with antioxidant potential (Santos et al., 2010; Figueiredo-Rinhel et al., 2013).
Regarding the extraction of bioactive and antioxidant compounds, some studies have emphasized the importance of the type of solvent used to obtain the extractive ingredients, and among them methanol has been described as one solvent for this purpose.
In the study reported herein, a methanolic extract of B. dracunculifolia leaves was obtained and characterized. A 2³ central composite rotational design (CCRD) and the response surface methodology were used to define the best conditions for extracting the bioactive compounds. In addition, the antioxidant potential, antimicrobial activity, and the chemical nature of the phytochemicals present, and their concentrations in the methanolic extract obtained under the conditions optimized, were assessed.
Material and methods
Botanical material and reagents
Baccharis dracunculifolia leaves were collected on the campus of Universidade Estadual do Centro Oeste Paraná, Guarapuava-Paraná, Brazil; coordinates (25°32´7.24”) South Latitude, (50°39’42.39”) West Longitude, and altitude, (1,120 m). The voucher specimen was identified by Dr. Paulo Vitor Farago and deposited in the Herbarium of the Universidade Estadual de Ponta Grossa, Ponta Grossa-Paraná (Brazil) and registered under the N°. 18173. Samples of fifty plants were collected from native shrubs in November of 2016 (spring in Brazil), which corresponded to the vegetative period (June to November) of the plant.
Methanol, gallic acid, 6–hydroxyl-2,5,7,8-tetramethylchroman-2-carboxylic acid (Trolox), Folin-Ciocalteu reagent, 1,1-diphenyl-2-picrylhydrazyl (DPPH), 2,2′-Azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS), potassium persulfate, Brain Heart Infusion broth and Sabouraud agar, resazurin, fluconazole and tetracycline were obtained from Sigma-Aldrich (St Louis, MO, USA). All reagents used in this study were of analytical grade.
Extraction of bioactive compounds from the leaves of Baccharis dracunculifolia
Leaves from B. dracunculifolia were dehydrated at room temperature for (48h) and in a drying oven at (40ºC) until reaching a constant mass (@ 50 h). The dried material was ground, sieved (mesh 1.19 mm), and stored in polyethylene bottles at (-18ºC) until required. Methanolic extraction of the bioactive compounds was performed using a 1:4 ratio (mass of ground material to volume of extraction solvent, methanol in water according to the concentrations described in Table 1) in an orbital incubator. Extraction parameters were optimized using a central composite rotational design (2³ CCRD), and analysis by response surface methodology.
The parameters examined (independent variables) were the concentration of the extracting solution, extraction time, and temperature, whereas the response variables (dependent variables) were the total phenolic content and the antioxidant activity.
Table 1 describes the matrix of the experimental design, with real and coded values of the variables studied. The solvent in the extract obtained under the optimized conditions was removed under vacuum by rotary evaporation at (45°C), and the material obtained through the optimized extraction procedure was frozen at (-50°C) and freeze-dried for further characterization.

Independent variables: A (methanol concentration), B (extraction time), C (temperature). *DPPH scavenging activity expressed as µmol Trolox Equivalent / g of extracted sample (TE g-1). aValues are presented as mean ± SE (n = 3).
Determination of total phenolic content and antioxidant activity
The content of total phenolic compounds extracted was determined by the Folin-Ciocalteu method using gallic acid as the standard, as described by Singleton and Rossi (1965). Antioxidant potential was determined by 1,1-Diphenyl-2-picrylhydrazyl (DPPH) scavenging activity was assessed according to Brand-Willliams, Cuvelier and Berset (1995), 2,2'-azino-bis 3-ethylbenzothiazoline-6-sulphonic acid (ABTS) free radical scavenging capacity was measured according to Re et al. (1999) and ferric reducing antioxidant power performed according Benzie and Strain (1996).
Antimicrobial activity
Antimicrobial activity was evaluated qualitatively by the broth-microdilution method and determining the minimum inhibitory concentrations (MIC) as described by Krichen et al. (2015). An aqueous solution of the optimized extract of B. dracunculifolia (concentrations: 0.36; 0.5; 1.0; 2.0; 3.0; 4.0; 5.0; 6.0; 7.0; 8.0; 9.0; 10.0; 20.0; 40.0; 60.0; 70.0 mg mL-1) was evaluated against Staphylococcus aureus ATCC 25923, Salmonella enterica Typhimurium ATCC 0028, Escherichia coli ATCC 25922 bacterial strains. The antimicrobial activity against the yeasts, Candida albicans ATCC 10231 and Candida tropicalis ATCC 13803, was also assessed. As positive controls, the antimicrobials fluconazole (1 mg mL-1) and tetracycline (1 mg mL-1) were used for tests with yeasts and bacteria, respectively. The negative control consisted of peptone water (0.1%). All experiments were performed in triplicate.
HPLC analysis of phenolic compounds
Identification and quantification of the phenolic compounds in the extract of B. dracunculifolia leaves was performed using a Varian 920 LC HPLC system (Varian Inc., Walnut Creek, CA, USA) coupled to photodiode array detector (DAD). A reverse-phase column C-18 RP (250 x 4.6 mm x 5 μm) (Microsorb MV-100, Agilent Technologies, Wilmington, DE, USA) was used. The column was kept at 30 °C and injected with 10 μL extract samples at a concentration of (0.05 g mL-1).
Two methods of analysis were used to identify a higher number of bioactive compounds. Protocol 1: The elution of the HPLC column was run in a gradient mode and the solvents used were acidified water with acetic acid (2%), v/v (solvent A); and acetonitrile:water:acetic acid at (58:40:2) v/v (solvent B) at a flow rate of 1 mL min-1. The solvent gradient mixtures used for eluting the column were: mobile phase A (95%) and B (5%) for 2 min; (80%) A and (20%) B for 13 min; A (75%) and B (25%) for 10 min; A (15%) and B (85%) for 7 min; A (5%) and B (95%) for 4 min; A (95%) and B (5%) for 9 min. Protocol 2: the mobile phase consisted of a mixture of solvents A (water) and B (acidified methanol with ortho-phosphoric acid), and the flow rate was 1 mL min-1.
The solvent gradient mixtures used for eluting the column were: mobile phase A (70%) and B (30%) for 15 min; 36% A and 64% B for 26 min; 25% A and 75% B for 28 min; 5% A and 95% B for 32 min, and returning to 70% B and 30% A.
Peak areas were determined at 280 nm for gallic acid, vanillic acid, catechin, and epicatechin; 300 nm for p-coumaric acid, salicylic acid, and resveratrol; 320 nm for caffeic and ferulic acid; and 360 nm for the flavonoids rutin, quercetin, myricetin, and pinocembrin.
Data analysis
Data obtained from experimental tests corresponding to the experimental design were analyzed by ANOVA, after the homogeneity of variance was checked by Levene's test and normal distribution of results checked by the Shapiro-Wilk test with at 5% significance level, using Statistica 8.0 software for data analysis.
Results and discussion
Evaluation of data of the experimental design
The multivariate approach (2³ CCRD experimental design) is a statistical tool that allows obtaining the maximum amount of information with the least number of experiments, generating savings of materials and optimizing the time of experimental execution (Santos et al., 2014). Results of the effects of the studied variables on the responses were plotted in Pareto charts, as described in Figure 1 (a, b, and c).
As shown in Figure 1 (a), the temperature used in the extraction process had a significant positive effect (quadratic effect) on the content of phenolic compounds extracted considering a 95% confidence level (p < 0.05). Greater extraction capacity of phenolic compounds occurred when higher temperatures were used in the process. This result suggested that these compounds did not suffer degradation at the higher temperatures assessed (@ 70°C). In fact, some studies in the scientific literature mentioned that higher temperatures could have contributed to the solubilization of some of these compounds, thus, increasing the coefficient of extraction (Tan, Stathopoulos, Parks, & Roach, 2014; Thoo, Ho, Liang, Ho, & Tan, 2010). Thoo et al. (2010) found that temperatures around 65°C promoted greater extraction of total phenolics in an ethanolic extract of Morinda citrifolia. Similarly Tan et al. (2014), verified that high temperatures (80°C) in combination with a short time interval of extraction with solvent (5 min) proved to be the ideal combination for the extraction of phenolic compounds from Momordica charantia L.
In relation to DPPH free radical scavenging activity (Figure 1b), the only variable that contributed significantly at a 95% confidence level (p < 0.05), and had a positive linear effect, was the concentration of methanol. Higher concentrations of methanol in the extracting solution (lower polarity) promoted a higher extractability of compounds with antioxidant activity against the DPPH radical in the extract of B. dracunculifolia (Table 5).
Hocayen et al. (2016) reported that higher total phenolic contents were found in extracts of B. dracunculifolia (70.5 mg GAE L-1) prepared with methanol. Other solvents such as ethanol (46.2 mg GAE L-1) and acetone (30.3 mg GAE L-1) also showed high extraction potential, but with lower performance compared Lto methanol. The toxicity of the crude methanolic extract of Baccharis dracunculifolia was evaluated by bioassay using Artemia salina, and very high concentrations of the extract were necessary for it to be considered lethal; mean lethal concentration (LC50) of (1008.51 ug mL-1 and 921.32 ug mL-1) at 24 and 48h, respectively (Hocayen, Campos, Pochapski, & Malfatti, 2012).
On the other hand, when analyzing the effect of time on the response to DPPH scavenging activity (Figure 1c) at a 90% confidence level (p < 0.1), a negative linear effect was found. In this context, longer times promoted the obtainment of an extract with lower DPPH scavenging potential. According to Salar, Purewal and Bhatti (2016) prolonged extraction times may promote oxidation or degradation of the phenolic compounds, which are the main responsible for antioxidant activity. According to Veiga et al. (2017) samples with high antioxidant activity in extracts of B. dracunculifolia were richest in flavonoid compounds, similarly Rezende et al. (2014) reported that leaf extracts of B. dracunculifolia showed high amounts of phenolic compounds and flavonoids related to antioxidant activity.
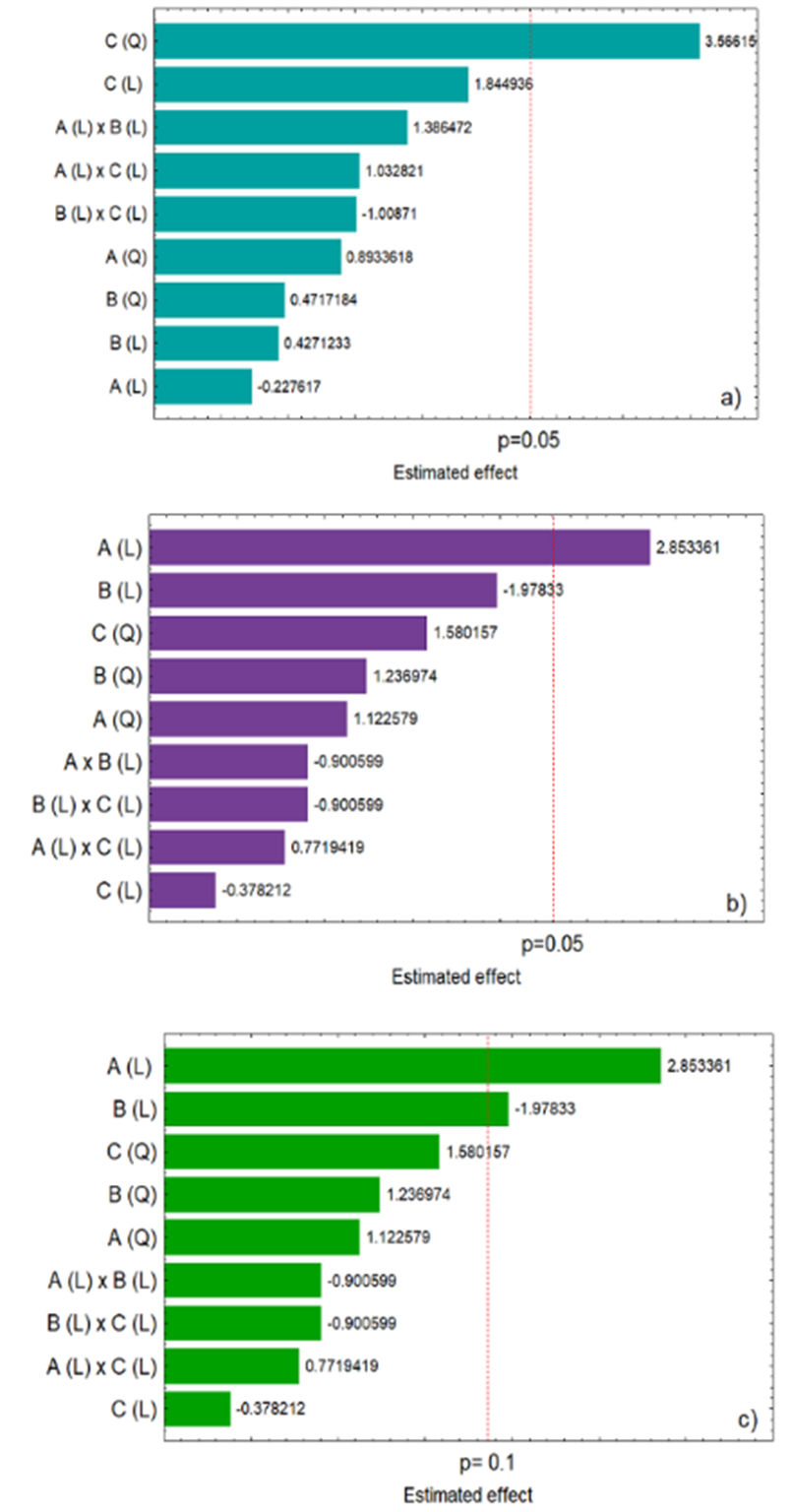
Figure 1. Estimated effects of methanol concentration (A), extraction time (B), and temperature (C) on the total phenolics content (a) and DPPH radical scavenging activity (b) at p = 0.05 and at p = 0.1 (c). Linear effects (L); Quadratic effects (Q).
Response surfaces were generated for the graphical observation of the effects of the studied variables (solvent concentration, time and extraction temperature) on the responses (Figure 2).
In Figure 2a, it is possible to observe the tendency of greater extraction of phenolic compounds when using higher temperatures when associated with the use of an extractant containing higher methanol concentrations. Similarly, a greater tendency of extraction of phenolic compounds (Figure 2b) was also observed when a higher temperature of the extractant was associated with shorter extraction times.
Similar to extracting total phenolic content, the response surfaces for DPPH scavenging activity (Figure 2c, d and e) showed that extracts with higher antioxidant activity were obtained when the extraction process combined higher methanol concentration in the extracting solution and shorter extraction times (Figure 2c). The combination of a higher concentration of methanol in the extracting solution with the use of higher temperatures also promoted obtaining extracts with higher antioxidant activity (Figure 2d). The response surface shown in Figure 2e indicated that a higher DPPH scavenging activity in the extracts was also found when using higher temperatures and shorter extraction times.
From the results achieved in the experimental tests, we considered that the best conditions for obtaining extracts of B. dracunculifolia leaves were: 70°C temperature, 20 min of extraction time, and 95% methanol concentration.
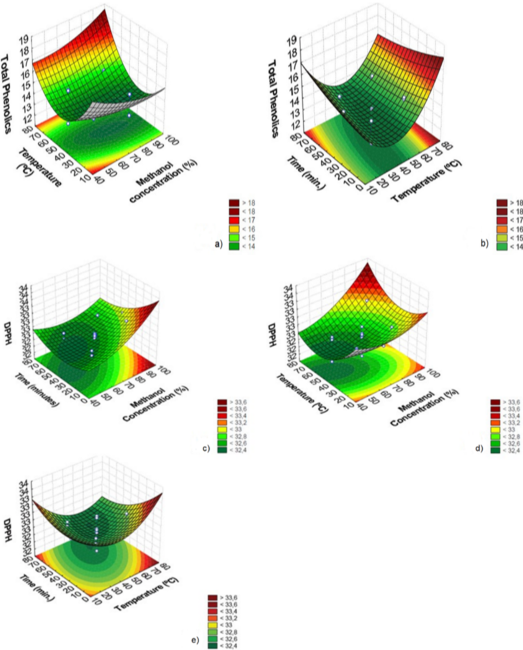
Figure 2. Response surface for extracting total phenolics. Temperature versus methanol concentration (a); Time versus Temperature (b). Response surface for DPPH scavenging activity: Time versus methanol concentration (c); Temperature versus Methanol concentration (d); Time versus Temperature (e). (n = 3).
Coefficient of determination of (R²) of the mathematical models of the response surfaces, which describe the influence of the studied variables on the total phenolic content (y = 12.52644 + 0.55676 C2) and DPPH scavenging activity (y = 32.45183 + 0.15242 A), were obtained through linear regression. A R² of 0.72 (R²adj: 0.40, Confidence level = 99.3%) was obtained for the model regarding the total phenolic content, and in relation to the DPPH scavenging capacity, a R² of 0.69 (R²adj: 0.35, Confidence level = 97.8%) was verified (Table 2).
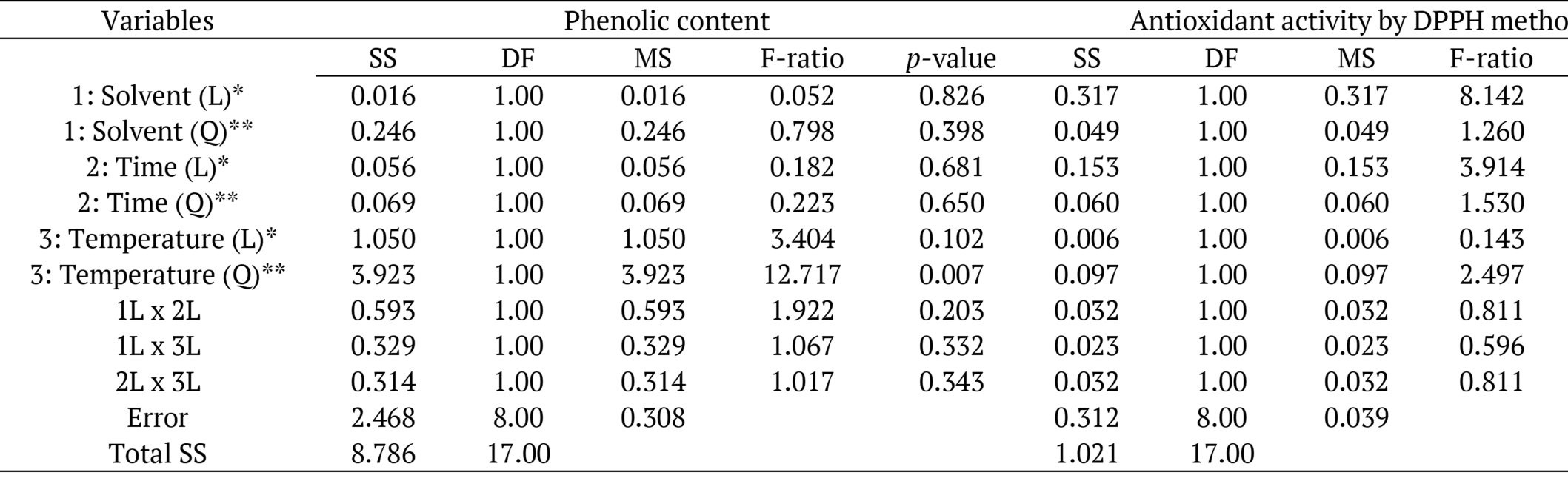
*Linear and **quadratic effects, SS: sum of squares, DF: degrees of freedom, MS: mean square (variance estimate).
Phenolic compounds identified in the methanolic extract of leaves from Baccharis dracunculifolia
Analysis by high-performance liquid chromatography (HPLC-DAD) comparing retention times and UV-VIS absorption profiles of the chemicals in the methanolic extract with authentic reference standards of phenolic compounds, allowed the identification and quantification of six phenolic compounds: catechin, caffeic acid, p-coumaric acid, rutin, ferulic acid, and pinocembrin (Table 3).
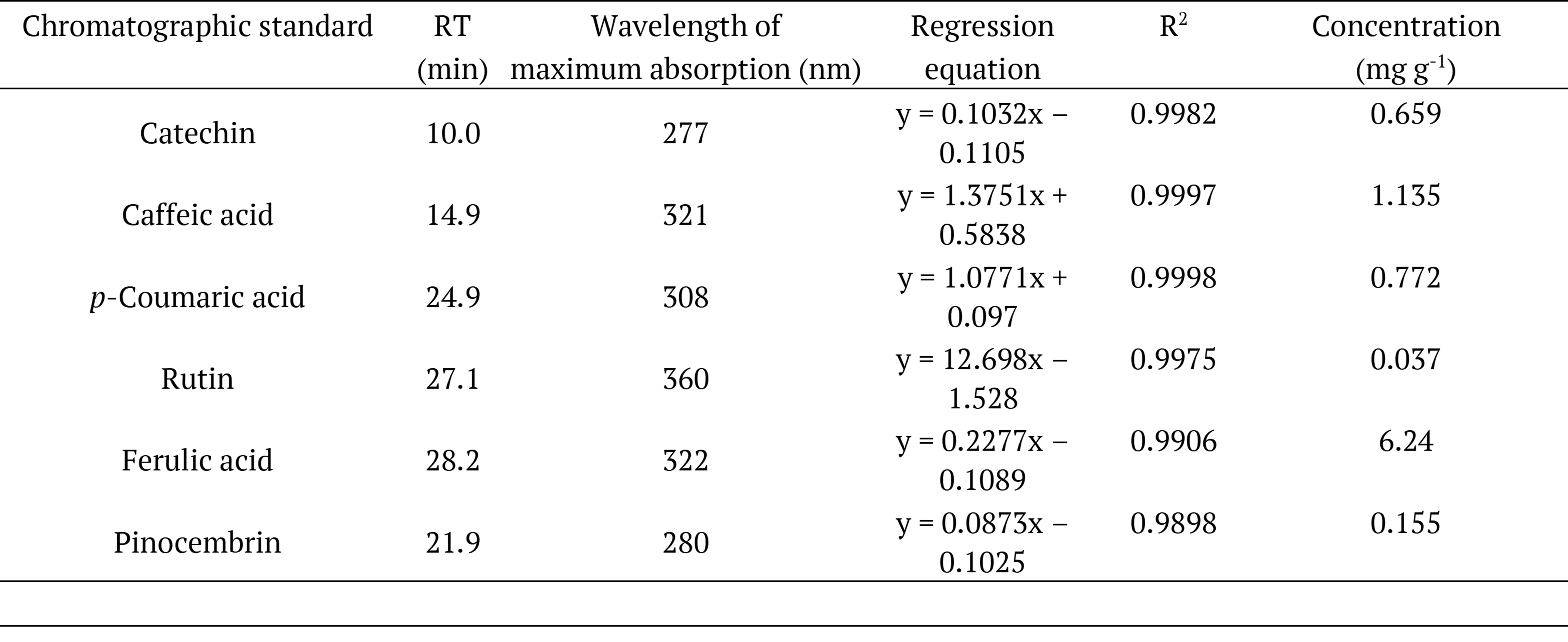
RT: retention time, R²: coefficient of determination (R-Squared).
The methanolic extract of B. dracunculifolia, obtained under the optimized conditions of extraction, was injected into the chromatograph using two distinct protocols; protocol 1 was developed for the detection and analysis of phenolic acids, and protocol 2 for the detection of flavonoids (Figure 3a and b, respectively).
The compounds found in higher concentration were ferulic (6.24 mg g-1) and caffeic (1.13 mg g-1) acids. Lower values of ferulic acid (4.80 mg g-1) were reported by Park, Paredes-Guzman, Aguiar, Alencar and Fujiwara (2004) in the ethanolic extract of B. dracunculifolia, as well as lower values of caffeic acid (0.692 mg g-1) were reported by Guimarães et al. (2012) with a glycolic extract of B. dracunculifolia. The specialized metabolite p-coumaric acid (0.772 mg g-1) and the flavonoid rutin (0.037 mg g-1) were also identified in the studies reported by Guimarães et al. (2012) and Roberto, Matsumoto, Jamal, Malaspina and Marin-Morales (2016) respectively.
Catechin is most commonly found in other species of the Baccharis genus, such as B. dentata, B. uncinella and B. anômala (Dias, Nozari, & Santarém, 2017). Pinocembrin, a flavonoid, is usually found in the green propolis produced from plants of the genus Baccharis, as highlighted in the study of Zhang et al. (2016). The identification and quantification of these two phenolic compounds in low amounts in methanolic extracts from B. dracunculifolia may be related to the geographical location of collection of the plant, as well as soil and climatic features at this location. In addition, it is also important to emphasize the effectiveness of the extraction method of the bioactive compounds, including catechin and pinocembrin. In fact, the results regarding the antioxidant capacity of the methanolic extract of B. dracunculifolia proved to be rich in bioactive compounds, with scavenging activity against DPHH and ABTS radicals, and ferric ion reduction activity, which corroborates the results of the identification and quantification of the bio-compounds in the methanolic extract.
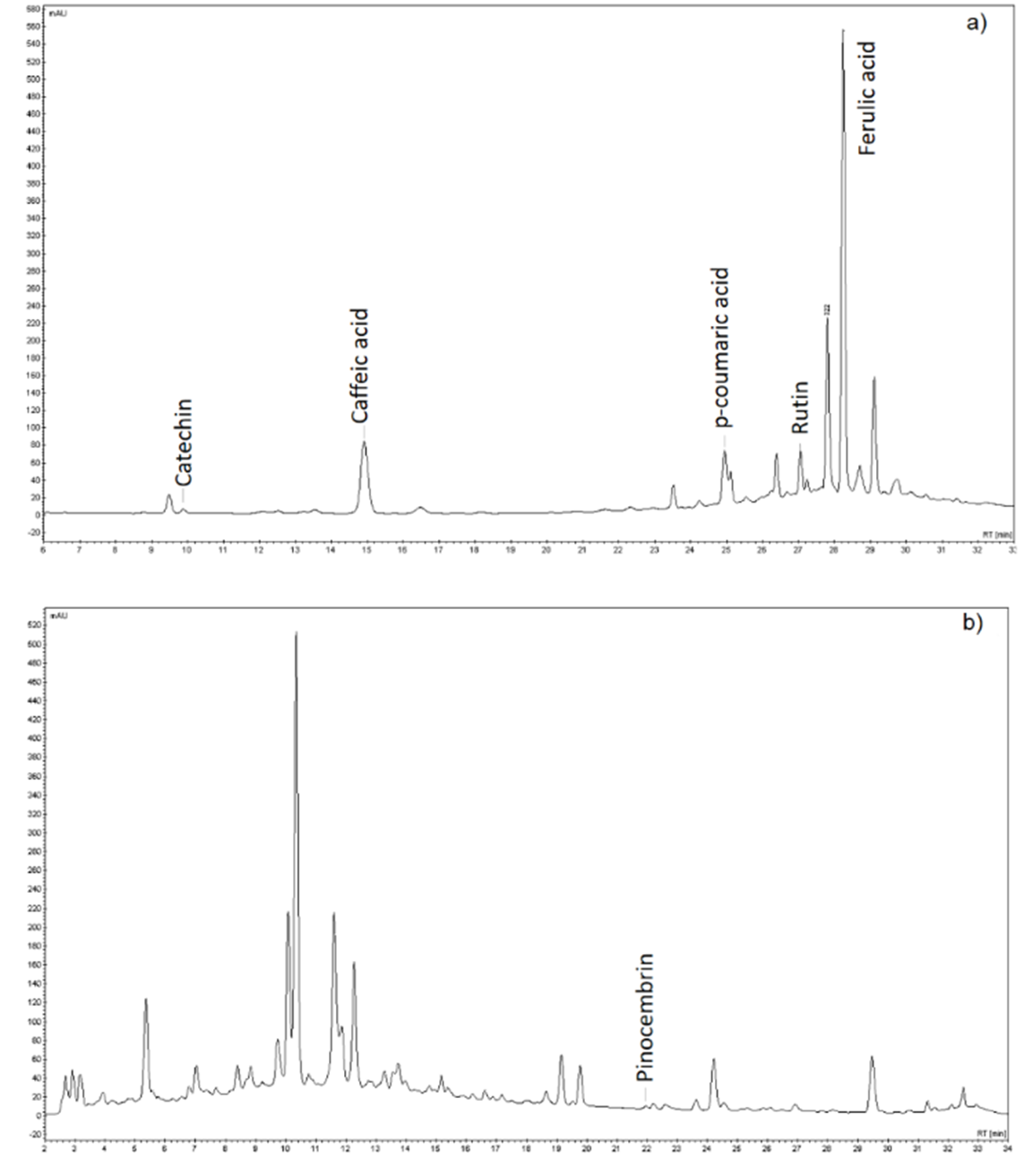
Figure 3. HPLC-DAD Chromatograms showing phenolic compounds identified using protocol 1 (a), and protocol 2 (b). (n = 3).
Total phenolics content and antioxidant activity in the methanolic extract obtained under optimized conditions
In natural products, the phenolic and polyphenolic compounds are the main substances involved in the processes of scavenging free radicals, and cellular protection against oxidative damage (Rezende et al., 2014). It is important to note that there may be synergistic, additive or antagonistic relationships between the different phenolic compounds and polyphenols present in plant extracts (Sonam & Guleria, 2017). In this sense, the analytical protocols commonly used for the evaluation of antioxidant activity express the results as total antioxidant activity, where the interactions (joint action) of bioactivity between the compounds present are considered. Likewise, there is also a correlation between the phenolic compounds and polyphenols present in the extract and their antimicrobial potential.
The extract from B. dracunculifolia leaves obtained under the optimized conditions presented high total phenolic content (72.51 mg GAE g-1). Similar values to those found in this study were reported by Martinez-Correa et al. (2012) in the extract of leaves of a B. dracunculifolia cultivated in a different area of Brazil (Campinas-SP). These authors employed a combination of extracting processes including the use of supercritical carbon dioxide, ethanol and water. They found values of between 48.4 and 197 mg GAE g-1 in the extract from a mixture of young and older, developed leaves. In the extract from only young leaves, the concentrations varied from 69 to 180 mg GAE g-1. It is important to emphasize that the different phenolic content between samples of natural extracts can be explained by the variables involved, such as season of growth, plant age, type of soil, and extraction conditions employed (Sartor et al., 2013).
The phenological stage, as well as the place of cultivation of the plant has a major influence on the concentration of flavonoids and phenolic compounds produced, and consequently on antioxidant and microbial activities. Sartor et al. (2013) studied the methanolic extract of the aerial parts of Baccharis dentata, analyzing total phenolic compounds and flavonoids over a one-year period, found that highest antioxidant activities were present in samples collected in summer and winter. Sforcin et al. (2012) studied the role of season on the phenolics content of Baccharis dracunculifolia, and suggested that higher contents of phenolic compounds could be obtained if the plant was harvested during the period between December to April. The B. dracunculifolia studied in our work was collected in spring, when the plant was in its vegetative stage, and resulted in high total phenolic contents and antioxidant activity, which suggests that in addition to the collection period, other factors influenced the concentrations of bioactive compounds in the plant. Pavarini, Pavarini, Niehues and Lopes (2012) described the synthesis of special metabolites was influenced by several biotic and abiotic factors, such as longer light exposure, temperature variations, enriched carbon dioxide atmosphere, altitude, drought stress, continuous solar radiation, exposure to red light radiation, high availability of nutrients, which can all induce variations in the accumulation or biogenesis of special metabolites.
The methanolic extract obtained under the optimized conditions also showed a significant antioxidant potential (Table 4). Concerning the DPPH scavenging potential, an activity of 865.6 ± 11.64 µmol TE g-1. was found. Similarly, the extract showed high ABTS radical scavenging capacity (331.11 ± 13.93 µmol TE g-1), presenting values higher than those reported by Zampini, Ordoñez and Isla (2010) in an ethanolic extract of Baccharis boliviensis (150 μmol TE g-1). Antioxidant activity expressed as ferric reducing capacity was also found in the extract of B. dracunculifolia and was exceptionally high (1558 ± 4.33 µmol FeSO4.7H2O g-1). Lower values of ferric reducing capacity (629.5 µmol FeSO4.7H2O g-1) were reported by Borneo, Aguirre and Cantero (2008) in an ethanolic extracts of Baccharis sessiflora Vahl.

*Gallic acid equivalent per gram; **Trolox equivalent per gram; #Values are presented as mean ± SE (n = 3).
The caffeic (3-(3,4-Dihydroxyhenyl)-2-propenoic acid), p-coumaric (4-hydroxycinnamic acid) and ferulic (4-hydroxy-3-methoxycinnamic acid) acids present in the extract of B. dracunculifolia, are hydroxycinnamates, and according to Nimse and Pal (2015) the antioxidant activity of these substances is related to patterns of methylation and hydroxylation of the aromatic ring. The antioxidant activity of catechins is involved in ultra-rapid electron transfer from catechins to reactive oxygen species (ROS)-induced radical sites on DNA, and can also chelate metal ions (Cu2+, Fe3+) forming inactive complexes that prevent the generation of potentially damaging free radicals. Another mechanism by which catechins scavenge free radicals is by forming stable semiquinone free radicals, thus, preventing the deaminating-ability of free radicals. In addition, the oxidation of catechins in reactions with free radicals promote the formation of a dimerized product, which was demonstrated to increase superoxide scavenging ability and iron-chelating potential (Sutherland, Rahman, & Appleton, 2006). Pinocembrin (5,7-dihydroxy-2-phenyl flavanone) has been described as a natural flavonoid with the ability to reduce ROS activity, however, the specific mechanism of antioxidant action of this molecule remains unclear (Jin, Liu, Jia, & Wang, 2015). The antioxidant activity of rutin (quercetin 3-O-rhamnosyl-rhamnosyl-glucoside) is associated with its ability to bind free radicals and metal ions; for example, chelating Fe2+ and Fe3+ ions, which can initiate oxygen free-radical formation. Rutin’s capacity to bind free radicals plays an important role in protecting DNA against oxidative damage, which is important because ROS’s (oxygen radicals, superoxide anion, hydroxides, peroxyls, alkoxyls, etc) can contribute to the onset of oncological diseases (Koval’skii et al., 2014).
Antimicrobial activity
Natural extracts containing phenolic acids commonly present antimicrobial activity. Such activity may vary considerably depending upon the amounts and types of phenolic acids present, which may act synergistically and increase the antimicrobial potential (Teodoro, Ellepola, Senevirante, & Koga-Ito, 2015). Caffeic acid may interfere in the synthesis of (1®3)-β-D-glucan (a constitutive macromolecule of the cell wall), and also in the permeability of the cytoplasmic membrane (Borges, Ferreira, Saavedra, & Simões, 2013). On the other hand, ferulic acid and the catechins act by modifying the charge and hydrophobicity of the cell surface of bacteria (G+ and G-) causing cell death by extravasation of the cytoplasmic contents (Hirasawa & Takada, 2004; Ma, Wang, Zhang, Zhang, & Ding, 2010). p-Coumaric acid act as a antimicrobial by disrupting bacterial cell membranes and through binding to bacterial genomic DNA to inhibit cellular functions (Lou et al., 2012). The antimicrobial activity of pinocembrin is related to interference in energy homeostasis, and inducing damage to the plasma membrane of the pathogen (Rasul et al., 2013). Rutin has antibacterial activity by inhibiting the enzyme, DNA isomerase IV (Ganeshpurkar & Saluja, 2017).
Table 5 and Figure 4 shows the antimicrobial potential of the methanolic extract of B. dracunculifolia leaves against different yeasts and bacteria. The extract presented fungistatic activity against the yeasts, Candida tropicalis ATCC 13803 and C. albicans ATCC 10231, at concentrations 5 mg mL-1 and 7 mg mL-1, respectively. With respect to antibacterial activity, the methanolic extractive material inhibited the growth of E. coli ATCC 25922 at concentrations of 60 and 70 mg mL-1, and S. aureus ATCC 25923 at concentrations of 4 mg mL-1. The inhibitory effect against Salmonella enterica Typhimurium ATCC 0028 was also demonstrated at concentrations of 40, 60, and 70 mg mL-1. Bactericidal effect was found against Staphylococcus aureus ATCC 25923 at concentrations 8 mg L-1 or higher.
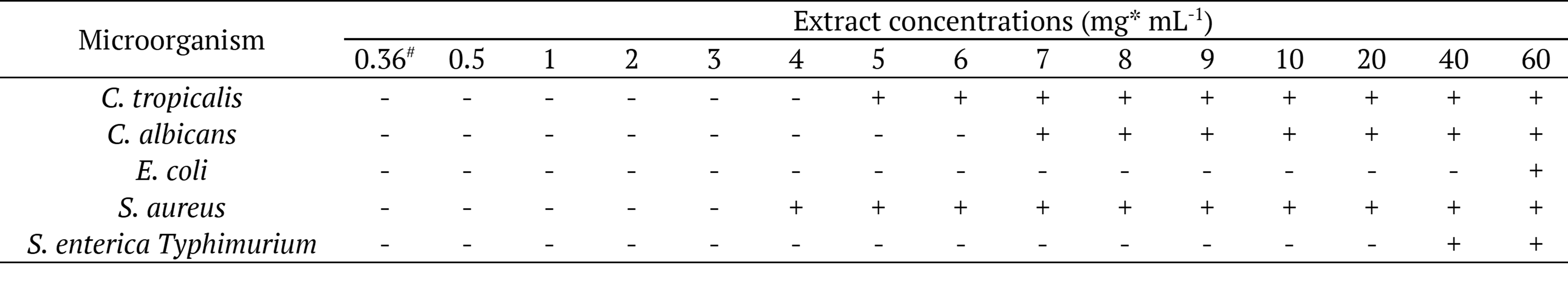
*mg of total extracted material (dry weight) mL-1. #Scoring: a positive sign (+) indicates inhibition of microbial growth, while a negative sign (-) indicates microbial growth. (n = 3).

Figure 4. 1: Culture medium, microorganisms and B. dracunculifolia extract; 2: culture medium, microorganisms and commercial antimicrobial; 3: culture medium and microorganisms; 4: Culture medium, microorganisms and B. dracunculifolia extract. The concentrations of B. dracunculifolia extract are distributed in decreasing order. a) C. albicans b) C. tropicalis c) E. coli d) S. aureus e) S. enterica Typhimurium.
Aleixo, Herrera, Ribeiro, Lima, and Ferreira (2013) and Morales, Paredes, Sierra, and Loyola (2008) studied the antimicrobial activity of extracts from four species of Baccharis (B. trimera and B. microphylla; B. petiolata; B. santelicis) and reported that Gram-positive bacteria were more sensitive. In our study, antimicrobial effectiveness against Gram-positive bacteria was also observed, and was highest with Staphylococcus aureus. Oliveira et al. (2005) evaluated the antimicrobial activity (MIC) of extracts of other species of Baccharis from southern Brazil (B. spicata, B. trimera, B. usterii) and reported MIC values of 12.5 mg mL-1 in the extract of B. usterii against S. aureus. In our study, the extract obtained presented antimicrobial activity 3 times greater (MIC: 4 mg mL-1) than the activity reported by Oliveira et al. (2005) against S. aureus. In addition, the extract of B. dracunculifolia showed fungistatic activity against C. tropicalis ATCC 13803 (MIC: 5 mg mL-1) and C. albicans ATCC 10231 (MIC: 7 mg mL-1), and bacteriostatic activity against E. coli and S. enterica Typhimurium ATCC 0028 (MIC: 60 and 40 mg mL-1, respectively), which shows the antimicrobial potential of the methanolic extract from B. dracunculifolia.
Conclusion
Methanolic solution (95% v/v) associated with 70°C temperature and 20 min extraction time promoted the extraction of phytochemicals from the leaves of B. dracunculifolia with high phenolic compounds content and appreciable antioxidant potential. HPLC-DAD analysis showed an extract rich in catechin, caffeic acid, p-coumaric acid, rutin, ferulic acid, and pinocembrin. The extract of B. dracunculifolia showed promising anti-microbial activity with fungicidal activity against the yeasts C. tropicalis and C. albicans, and show bacteriostatic effect against E. coli and S. entericaTyphimurium. In addition, bactericidal effect was also observed against Staphylococcus aureus.
Acknowledgements
The authors thank the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), and the Fundação Araucária (M. A. A. Cunha) for financial support.
References
Aleixo, Á. A., Herrera, K. M. S., Ribeiro, R. I. M. A., Lima, L. A. R. S., & Ferreira, J. M. S. (2013). Antibacterial activity of Baccharis trimera (Less.) DC. (carqueja) against bacteria of medical interest. Revista Ceres, 60(5), 731-734. doi: 10.1590/S0034-737X2013000500019
Benzie, I. F. F., & Strain, J. J. (1996). The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Analytical Biochemistry, 239(1), 70-6. doi: 10.1006/abio.1996.0292.
Borges, A., Ferreira, C., Saavedra, M. J., & Simões, M. (2013). Antibacterial activity and mode of action of ferulic and gallic acids against pathogenic bacteria. Microbial Drug Resistance, 19(4), 256-265. doi: 10.1089/mdr.2012.0244
Borneo, R., Aguirre, A., & Cantero, J. J. (2008). In vitro antioxidant activity of species collected in Córdoba (Argentina). Electronic Journal of Environmental, Agricultural and Food Chemistry, 7(8), 3168-3170. Retrieved from https://bit.ly/2Tu7OfB
Boutennoun, H., Boussouf, L., Rawashdeh, A., Al-Qaoud, K., Abdelhafez, S., Kebieche, M., & Madani, K. (2017). In vitro cytotoxic and antioxidant activities of phenolic components of Algerian Achillea odorata leaves. Arabian Journal of Chemistry, 10(3), 403-409. doi: 10.1016/j.arabjc.2014.05.013
Brand-Willliams, W., Cuvelier, M. E., & Berset, C. (1995). Use of a free radical method to evaluate antioxidant activity. LWT – Food Science and Technology, 28(1), 25-30. doi: 10.1016/S0023-6438(95)80008-5
Cestari, S. H., Bastos K., J., & Di Stasi, L. C. (2011). Intestinal anti-inflammatory activity of Baccharis dracunculifolia in the trinitrobenzenesulphonic acid model of rat colitis. Evidence-Based Complementary and Alternative Medicine, 2011, 1-9. doi: 10.1093/ecam/nep081
Dias, M. P., Nozari, R. M., & Santarém, E. R. (2017). Herbicidal activity of natural compounds from Baccharis spp. on the germination and seedlings growth of Lactuca sativa and Bidens pilosa. Allelopath Journal, 42(1), 21-36. doi: 10.26651/2017-42-1-1103
Fatehi-Hassanabad, Z., Chan, C. B., & Furman, B. L. (2010). Reactive oxygen species and endothelial function in diabetes. European Journal of Pharmacology, 636(1-3), 8-17. doi: 10.1016/j.ejphar.2010.03.048
Figueiredo-Rinhel, A. S. G., Kabeya, L. M., Bueno, P. C. P., Jorge-Tiossi, R. F., Azzolini, A. E. C. S., Bastos, J. K., & Lucisano-Valim, Y. M. (2013). Inhibition of the human neutrophil oxidative metabolism by Baccharis dracunculifolia DC (Asteraceae) is influenced by seasonality and the ratio of caffeic acid to other phenolic compounds. Journal of Ethnopharmacology, 150(2), 655-664. doi: 10.1016/j.jep.2013.09.019
Ganeshpurkar, A., & Saluja, A. K. (2017). The pharmacological potential of rutin. Saudi Pharmaceutical Journal, 25(2), 149-164. doi: 10.1016/j.jsps.2016.04.025
Guimarães, N. S. S., Mello, J. C., Paiva, J. S., Bueno, P. C. P., Berretta, A. A., Torquato, R. J., ... Rodrigues, T. (2012). Baccharis dracunculifolia, the main source of green propolis, exhibits potent antioxidant activity and prevents oxidative mitochondrial damage. Food and Chemical Toxicology, 50(3-4), 1091-1097. doi: 10.1016/j.fct.2011.11.014
Hirasawa, M., & Takada, K. (2004) Multiple effects of green tea catechin on the antifungal activity of antimycotics against Candida albicans. The Journal of Antimicrobial Chemotherapy, 53(2), 225-229. doi: 10.1093/jac/dkh046
Hocayen, P. A. S., Campos, L. A., Pochapski, M. T., & Malfatti, C. R. M. (2012). Avaliação da toxicidade do extrato bruto metanólico de Baccharis dracunculifolia por meio do bioensaio com Artemia salina. Insula, 41(1), 23-31. doi: 10.5007/2178-4574.2012n41p23
Hocayen, P. A. S., Grassiolli, S., Leite, N. C., Pochapski, M. T., Pereira, R. A., Silva, ... Malfatti, C. R. M. (2016). Baccharis dracunculifolia methanol extract enhances glucose-stimulated insulin secretion in pancreatic islets of monosodium glutamate induced-obesity model rats. Pharmaceutical Biology, 54(7), 1263-1271. doi: 10.3109/13880209.2015.1067232
Jin, X., Liu, Q., Jia, L., Li, M., & Wang, X. (2015). Pinocembrin attenuates 6-OHDA-induced neuronal cell death through Nrf2/ARE pathway in SH-SY5Y cells. Cellular and Molecular Neurobiology, 35(3), 323-333. doi: 10.1007/s10571-014-0128-8
Koval’skii, I. V., Krasnyuk, I. I., Krasnyuk, I. I., Jr., Nikulina, O. I., Belyatskaya, A. V., Kharitonov, Y. Y., ... Lutsenko, S. V. (2014). Mechanisms of rutin pharmacological action (review). Pharmaceutical Chemistry Journal, 48(2), 73-76. doi: 10.1007/s11094-014-1050-6
Krichen, F., Karoud, W., Sila, A., Abdelmalek, B. E., Ghorbel R., Ellouz-Chaabouni, S., & Bougatef, A. (2015). Extraction, characterization and antimicrobial activity of sulfated polysaccharides from fish skins. International Journal of Biological Macromolecules, 75, 283-289. doi: 10.1016/j.ijbiomac.2015.01.044
Lou, Z., Wang, H., Rao, S., Sun, J., Ma, C., & Li, J. (2012). .-Coumaric acid kills bacteria through dual damage mechanisms. Food Control, 25(2), 550-554. doi: 10.1016/j.foodcont.2011.11.022
Ma, Z., Wang, J., Zhang, L., Zhang, Y., & Ding, K. (2010). Evaluation of water soluble β-D-glucan from Auricularia auricular-judae as potential anti-tumor agent. Carbohydrate Polymers, 80(3), 977-983. doi: 10.1016/j.carbpol.2010.01.015
Manach, C., Scalbert, A., Morand, C., Rémésy, C., & Jiménez, L. (2004). Polyphenols: Food sources and bioavailability. The American Journal of Clinical Nutrition, 79(5), 727-747. doi: 10.1093/ajcn/79.5.727
Maróstica Junior, M. R., Daugsch, A., Moraes, C. S., Queiroga, C. L., Pastore, G. M., & Park, Y. K. (2008). Comparison of volatile and polyphenolic compounds in Brazilian green propolis and its botanical origin Baccharis dracunculifolia. Ciência e Tecnologia de Alimentos, 28(1), 178-181. doi: 10.1590/S0101-20612008000100026
Martinez-Correa, H. A., Cabral, F. A., Magalhães, P. M., Queiroga, C. L., Godoy, A. T., Sánchez-Camargo, A. P., & Paviani, L. C. (2012). Extracts from the leaves of Baccharis dracunculifolia obtained by a combination of extraction processes with supercritical CO., ethanol and water. The Journal of Supercritical Fluids, 63, 31-39. doi: 10.1016/j.supflu.2011.12.016
Matough, F. A., Budin, S. B., Hamid, Z. A., Alwahaibi, N., & Mohamed, J. (2012). The role of oxidative stress and antioxidants in diabetic complications. Sultan Qaboos University Medical Journal, 12(1), 5-18. doi: 10.12816/0003082
Morales, G., Paredes, A., Sierra, P., & Loyola, L. A. (2008). Antimicrobial activity of three Baccharis species used in the traditional medicine of northern Chile. Molecules, 13(4), 790-794. doi: 10.3390/molecules13040790
Nimse, S. B., & Pal, D. (2015). Free radicals, natural antioxidants, and their reaction mechanisms. RSC Advances, 5(35), 27986-28006. doi: 10.1039/C4RA13315C
Oliveira, S. Q., Trentin, V. H., Kappel, V. D., Barelli, C., Gosmann, G., & Reginatto, F. H. (2005). Screening of antibacterial activity of south Brazilian Baccharis. species. Pharmaceutical Biology, 43(5), 434-438. doi: 10.1080/13880200590963754
Park, Y. K., Paredes-Guzman, J. F., Aguiar, C. L., Alencar, S. M., & Fujiwara, F. Y. (2004). Chemical constituents in Baccharis dracunculifolia as the main botanical origin of southeastern Brazilian propolis. Journal of Agriculture and food chemistry, 52(5), 1100-1103. doi: 10.1021/jf021060m
Pavarini, D. P., Pavarini, S. P., Niehues, M., & Lopes, N. P. (2012). Exogenous influences on plant secondary metabolite levels. Animal Feed Science Technology, 176(1-4), 5-16. doi: 10.1016/j.anifeedsci.2012.07.002
Pereira, C. A., Costa, A. C. B. P., Liporoni, P. C. S., Rego, M. A., & Jorge, A. O. C. (2016). Antibacterial activity of Baccharis dracunculifolia in planktonic cultures and biofilms of Streptococcus mutans. Journal of Infection and Public Health, 9(3), 324-330. doi: 10.1016/j.jiph.2015.10.012
Rasul, A., Millimouno, F. M., Ali Eltayb, W., Ali, M., Li, J., & Li, X. (2013). Pinocembrin: A novel natural compound with versatile pharmacological and biological activities. Biomed Research International, 2013(Special Issue), 1-9. doi: 10.1155/2013/379850
Re, R., Pellegrini, N., Proteggente, A., Pannala, A., Yang, M., & Rice-Evans, C. (1999). Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radical Biology and Medicine, 26(9-10), 1231-1237. doi: 10.1016/s0891-5849(98)00315-3
Rezende, T. P., Corrêa, J. O. A., Aarestrup, B. J. V., Aarestrup, F. M., Sousa, O. V., & Silva Filho, A. A. (2014). Protective effects of Baccharis dracunculifolia leaves extract against carbon tetrachloride- and acetaminophen-induced hepatotoxicity in experimental animals. Molecules, 19(7), 9257-9272. doi: 10.3390/molecules19079257
Roberto, M. M., Matsumoto, S. T., Jamal, C. M., Malaspina, O., & Marin-Morales, M. A. (2016). Evaluation of the genotoxicity/mutagenicity and antigenotoxicity/antimutagenicity induced by propolis and Baccharis dracunculifolia, by in vitro study with HTC cells. Toxicology in Vitro, 33, 9-15. doi: 10.1016/j.tiv.2016.02.005
Salar, R. K., Purewal, S. S., & Bhatti, M. (2016). Optimization of extraction conditions and enhancement of phenolic content and antioxidant activity of pearl millet fermented with Aspergillus awamori MTCC-548. Resource-Efficient Technologies, 2(3), 148-157. doi: 10.1016/j.reffit.2016.08.002
Santos, D. K. F., Brandão, Y. B., Rufino, R. D., Luna, J. M., Salgueiro, A. A., Santos, V. A., & Sarubbo, L. A. (2014). Optimization of cultural conditions for biosurfactant production from Candida lipolytica. Biocatalysis and Agricultural Biotechnology, 3(3), 48-57. doi: 10.1016/j.bcab.2014.02.004
Santos, D. A., Fukui, M. J., Dhammika, N. N. P., Khan, S. I., Sousa, J. P. B., Bastos, J, K., ... Quintão, N. L. M. (2010). Anti-inflammatory and antinociceptive effects of Baccharis dracunculifolia DC (Asteraceae) in different experimental models. Journal of Ethnopharmacology, 127(2), 543-550. doi: 10.1016/j.jep.2009.09.061
Sartor, T., Xavier, V. B., Falcão, M. A., Mondin, C. A., Santos, M. A., Cassel, E., ... Santarém, E. R. (2013). Seasonal changes in phenolic compounds and in the biological activities of Baccharis dentata (Vell.) G.M. Barroso. Industrial Crops and Products, 51, 355-359. doi: 10.1016/j.indcrop.2013.09.018
Sforcin, J. M., Souza, J. P. B., Silva Filho, A. A., Bastos, K. J., Búfalo, M. C., & Tonuci, L. R. S. (2012). Baccharis dracunculifolia: Uma das principais fontes vegetais da própolis brasileira. São Paulo, SP: Editora Unesp.
Singleton, V. L., & Rossi, J. A. (1965). Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. American Journal of Enology and Viticulture, 16(3), 144-158. Retrieved from https://bit.ly/3kAFy73
Sonam, K. S., & Guleria, S. (2017). Synergistic antioxidant activity of natural products. Annals of Pharmacology Pharmaceutics, 2(8), 1-6. Retrieved from https://bit.ly/2HGXUVa
Sutherland, B. A., Rahman, R. M. A., & Appleton, I. (2006). Mechanisms of action of green tea catechins, with a focus on ischemia-induced neurodegeneration. The Journal of Nutritional Biochemistry, 17(5), 291-306. doi: 10.1016/j.jnutbio.2005.10.005
Tan, S. P., Stathopoulos, C., Parks, S., & Roach, P. (2014). An optimised aqueous extract of phenolic compounds from bitter melon with high antioxidant capacity. Antioxidants, 3(4), 814-829. doi: 10.3390/antiox3040814
Teodoro, G. R., Ellepola, K., Seneviratne, C. J., & Koga-Ito, C. Y. (2015). Potential use of phenolic acids as anti-Candida agents: A review. Frontiers in Microbiology, 6, 1-11. doi: 10.3389/fmicb.2015.01420
Thoo, Y. Y., Ho, S .K., Liang, J. Y., Ho, C. W., & Tan, C. P. (2010). Effects of binary solvent extraction system, extraction time and extraction temperature on phenolic antioxidants and antioxidant capacity from mengkudu (Morinda citrifolia). Food Chemistry, 120(1), 290-295. doi: 10.1016/j.foodchem.2009.09.064
Ullah, A., Khan, A., & Khan, I. (2016). Diabetes mellitus and oxidative stress – A concise review. Saudi Pharmaceutical Journal, 24(5), 547-553. doi: 10.1016/j.jsps.2015.03.013
Veiga, R. S., Mendonça, S., Mendes, P. B., Paulino, N., Mimica, M. J., Lagareiro Netto, ... Marcucci, M. C. (2017). Artepillin C and phenolic compounds responsible for antimicrobial and antioxidant activity of green propolis and Baccharis dracunculifolia DC. Journal of Applied Microbiology, 122(4), 911-920. doi: 10.1111/jam.13400
Zampini, I. C., Ordoñez, R. M., & Isla, M. I. (2010). Autographic assay for the rapid detection of antioxidant capacity of liquid and semi-solid pharmaceutical formulations using ABTS•+ immobilized by gel entrapment. AAPS PharmSciTech, 11(3), 1159-1163. doi: 10.1208/s12249-010-9484-y
Zhang, J., Shen, X., Wang, K., Cao, X., Zhang, C., Zheng, H., & Hu, F. (2016). Antioxidant activities and molecular mechanisms of the ethanol extracts of Baccharis propolis and Eucalyptus propolis in RAW64.7 cells. Pharmaceutical Biology, 54(10), 2220-2235. doi: 10.3109/13880209.2016.1151444
Notas de autor
genice.iur@gmail.com