Ciência, Tecnologia Alimentar e Engenharia de Alimentos
Effect of pH on the flaxseed (Linum usitatissimum L. seed) mucilage extraction process
Effect of pH on the flaxseed (Linum usitatissimum L. seed) mucilage extraction process
Acta Scientiarum. Technology, vol. 43, e50457, 2021
Universidade Estadual de Maringá
Recepción: 17 Octubre 2019
Aprobación: 04 Abril 2020
Abstract: Water-soluble mucilage extracted from flaxseed has excellent hydrocolloid potential in gel formation; moreover, it can become a product of high technological value in the food, pharmaceutical and cosmetic industries because it has high viscosity in aqueous solution. The aim of the current study is to investigate the effect of extraction medium pH on extraction yield and soluble fiber content. In addition, the purification step effect was evaluated through Fourier transform infrared spectroscopy (FTIR). Extractions were carried out at 3 different pH values (3.81, 6.75 and 9.69) for 13.25 h at 45 .C. The highest yield was recorded at pH 6.75; the highest soluble fiber content was found at pH 3.81. It was possible seeing that the extraction of flaxseed macromolecules in basic medium (pH 9.69) presented the best quality in comparison to other extractions. The purification step based on precipitation with ethanol did not fully remove impurities from the samples.
Keywords: purification, fourier transform infrared, macromolecules, polysaccharide, polymers, linseed.
Introduction
Natural polymers play important roles in plants’ cell wall, as well as in several plant tissues. For example, seed coat tissues undergo strategic changes to activate embryo protection and make dispersion and future germination easier. This modification is associated with mucilage (hydrophilic polymers) deposition in the apoplast of epidermal coating cells, which becomes available after seed hydration in order to wrap it with a gel. The term mucilage refers to polysaccharides (mainly pectic or hemicellulose) released by plants during their development. Gums, which have the same content as mucilage, are secreted by plants affected by wounds and pathogenicity capable of causing cell wall degradation (Western, 2012). According to Western (2012), the specific role played by these biological macromolecules appears to depend on plant species and on environmental conditions plants are subjected to.
Mucilages and gums are interesting biological macromolecules that can be used in several pharmaceutical preparations due physicochemical properties such as their high swelling capacity, low cost, non-toxicity-related safety, biocompatibility due to their biodegradability, and their abundance in nature (Prajapati, Jani, Moradiya, & Randeria, 2013). It is worth highlighting studies available in the literature about the effective use of these natural polymers in pharmaceutical formulations, in different ways, namely: controlled release system matrix (Adeleye, Femi-Oyewo, &Odeniyi, 2014), coating agents and films (Patel, Patel, &Upadhyay, 2012), binding agents (Odeku & Patani, 2005), mucoadhesive agents (Nerkar &Gattani, 2013), disintegrants (Khinchi, Gupta, Bhandari, Agarwal, & Sharma, 2011), emulsifying agents (Verma &Razdan, 2003) and suspending agents (Deore & Khadabadi, 2008). Linseed biopolymer particles can be used in the food industry to modulate the functional properties of food products (Nasrabadi, Goli, Doost, & Van der Meeren, 2020).
Linum usitatissimum L., popularly known as flax, is one of the first plants grownby man and an important agricultural commodity worldwide (Dzuvor, Taylor, Acquah, Pan, & Agyei, 2018). The whole plant can be commercially used, from clothing, paper and oil manufacturing to by-products applied to animal feed. Thus, similar to other natural gums, flaxseed mucilage is a potential excipient of modified release systems, as observed by Nerkar and Gattani (2011, 2013), Hasseb, Hussain, Yuk, Bashir and Nauman (2016) and Hasseb, Hussain, Bashir, Ashraf and Ahmad (2017). However, the use of flaxseed mucilage as pharmaceutical excipient remains poorly explored in comparison to studies about other mucilages and gums (Prajapati et al., 2013).
Water-soluble mucilage extracted from flaxseed is composed of acidic and neutral polysaccharides. It is an excellent hydrocolloid that can become a product of high technological value in the food, pharmaceutical and cosmetic industries due its high viscosity in aqueous solution (Chen, Xu, &Wang, 2006).
The quality of the mucilage extracted from Linum usitatisimum L. seeds (flaxseed) can change depending on the extraction and cultivation processes, since it is a natural product. Cui and Mazza (1996) have observed different features in flaxseed mucilages extracted from different cultures. According to Kaewmanee et al. (2013), different culture conditions lead to different mucilage properties. The purification step in the aforementioned studies was based on ethanol addition to enable fiber precipitation. They used ethanol at volume 3 times greater than that of mucilage; subsequent procedures comprised filtration, oven drying, fiber solubilization in water and lyophilization.
The aim of the current study was to investigate the influence of the extractive aqueous medium pH on the quality and quantity of soluble fibers extracted from flaxseed, based on the evaluation of process yield, soluble fiber content and infra-red spectra before and after the ethanol-based purification step.
Material and methods
Materials
The botanical material, known as Linum usitatissimum L. (Golden Linseed), was purchased at a market in Niterói City, Rio de Janeiro State, Brazil, in 2017 (Figure 1). The herein used buffers - acetate at pH 3.81, acetate at pH 6.75 and borate at pH 9.69, were prepared based on the methodology described in the Brazilian Pharmacopoeia 5th edition (Anvisa & Fundação Oswaldo Cruz, 2010). All reagents used in the current study were of the analytical grade type. Different pHs, starting from a pH value close to neutrality, were used in the acidic and basic regions in order to investigate their influence on the extracted mucilage. Thus, equidistant values were used within the range of natural polymer extraction found in the literature (Cui, Mazza, Oomah, & Biliaderis, 1994; Koocheki et al., 2010; Koocheki, Taherin, Razavi, & Bostan, 2009; Lima Junior et al., 2013)

Figure 1. Botanical material: golden linseed.
Extraction process
Mucilage was extracted from Linum usitatissimum L. seeds (Flaxseed) under three different pH values. Whole flaxseed (10 g) was placed in a 250 mL flat bottom flask. Next, 130 mL of pH buffer was added to the flask and preheated at the experimental temperature of 45°C. Containers were kept in an orbital shaker incubator (Marconi - Model: MA-830), at controlled temperature of 45°C, under stirring at 180 rpm, for 13.25h. After the time set for extraction was over, samples were filtered and the supernatant was frozen and lyophilized in Freeze-dryer (Liotop – Model: L108). All extractions were performed in triplicate. Extraction yield (Y) was calculated based on the ratio between weight of all lyophilized soluble substances and total seed mass (10 g). Extraction yield (Y) was calculated through equation 1.

wherein: PL is the total weight of the lyophilized extraction product; S is the weight of the whole flaxseed.
Figure 2 shows the steps adopted in the extraction process, which were presented in the current section.
First, 0.1 g of each sample was weighed after yield calculation. Next, samples were solubilized in 20 mL of water and added with 60 mL of ethanol to enable fiber precipitate formation, based on the methodology described by Cui et al. (1994). The precipitated material was transferred to a Petri dish and placed in oven at 40 oC to enable ethanol evaporation. The dried material was solubilized in water, frozen and lyophilized for further infrared analysis.

Figure 2. Summary of the extraction process
Purification
Quantification of total soluble fibers
The official non-enzymatic method by Horwitz (2000) n. 993.21, recommended for low starch content samples (≤2 %), was the method used to enable total fiber quantification. Approximately 0.5 g of lyophilized sample was weighed and added with 25 mL of distilled water; the mixture was stirred in reciprocating water bath shaker at 37°C for 90 min. Next, 100 mL of 95% ethanol was added to the mixture at room temperature (25 ± 2°C) and left to rest for 60 min. The generated precipitate was vacuum filtered in a fiber-specific porous crucible and washed twice in 20 mL of 78% ethanol, twice in 10 mL of 95% ethanol and, finally, once in 10 mL of acetone. The fiber was dried for 2h in oven at 105oC and weighed.
The precipitate was placed in the muffle at 525oC for 5h to allow quantifying the mineral material found in the sample; then, the mineral retained in the fiber was weighed. This value was subtracted from the fiber weight in order to find the fiber mass (FS) in 0.5 g of lyophilized sample (PL).
Total fiber mass (FT), in grams (g), in all lyophilized materials can be calculated through equation 2.

wherein: PL is the total weight of the lyophilized extraction product; FS is the fiber mass in 0.5 g of lyophilized sample.
The fiber content (F) in the whole flaxseed was calculated through equation 3.

wherein: FT is the total fiber mass in PL; S is the whole flaxseed weight.
Figure 3 shows the steps adopted in the total soluble fiber quantification process, which were presented in the current section.
Fourier transform infrared spectroscopy (FTIR)
The transmittance spectra of the mucilage sample was generated in the infrared (IR) spectrophotometer (Nicolet-Model: IS50 FT-IR) at wavelength range from 650 to 4000 cm-1 by using approximately 5.0 mg of each analyzed sample. The product extracted from linseed was analyzed with, and without, ethanol-based purification.
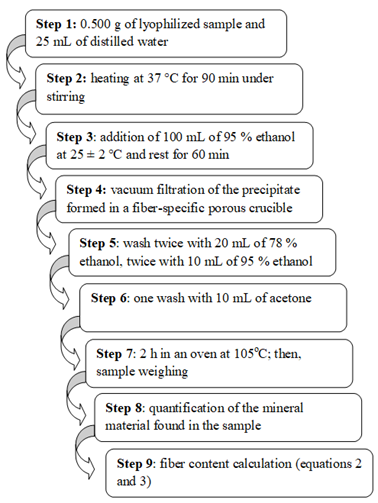
Figure 3. Summary of total soluble fiber quantification.
Results and discussion
Fiber content and extraction yield
The flaxseed mucilage extraction at different pH values enabled evaluating its effect on extraction yield, fiber content and quality of the seed-extracted product. Figure 4 depicts the post-lyophilization extraction products.

Figure 4. Samples 1 (pH 3.81), 2 (pH 6.75) and 3 (pH 9.69) after lyophilization.
Fiber content and extraction yield under each experimental condition are shown in Table 1.

SD = standard deviation.-Values followed by different letters in the same column are significantly different from each other (p < 0.05).
According to the Tukey's test, fiber and yield results recorded for acidic and basic pH extractions did not show statistically significant difference between them, i.e., extraction values recorded for both acidic and basic pH values were very close to each other. However, extraction carried out at almost neutral pH has shown statistically significant difference in fiber and yield results between acidic and basic pH values, as seen in Figure 5.
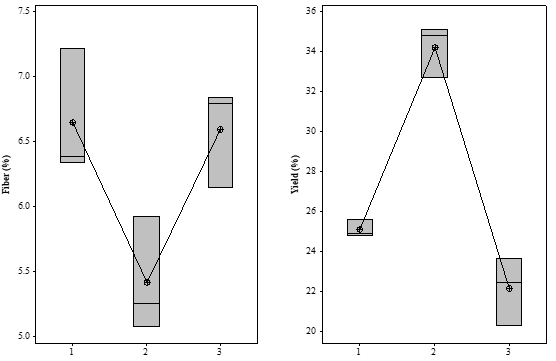
Figure 5. Boxplot graph generated for Fiber (%) and Yield (%) – Samples 1, 2 and 3.
The highest yield was recorded at almost neutral pH of 6.75. Similar results were reported by Cui et al. (1994), whose study about the optimization of flaxseed mucilage extraction showed that pH between 6.5 and 7.0 was the best extraction condition.
It was possible observing that the maximum yield led to the lowest fiber rate. This outcome suggests that the extraction of other substances took place at pH 6.75, as well as that the other extraction conditions- such as acidic and basic pH, which led to higher fiber values- enabled the extraction of smaller quantities of non-fiber substances.
According to Hasseb et al. (2016), who assessed the swelling and retraction ability of flaxseed polymers at pH 1.2 and 7.4, these polymers are sensitive to pH. The aforementioned authors have observed that polymers transferred from pH 7.4 to pH 1.2 have suffered shrinkage. This response may be associated with the presence of COOH groups in the polysaccharide structural network, which makes secondary bonds such as hydrogen and intramolecular bonding, and bonding between polysaccharide chains.
Swelling, on the other hand, results from an inverse process, according to which hydrogen bonds are broken due to electrostatic repulsion generated by COO-NA+ and COO- groups, rather than by COOH groups (Hasseb et al., 2016). The formation of these ionic bonds increases molecule polarity, which can likely account for its high extraction in basic aqueous media, since water is a polar solvent (Moreira et al., 2010).
Several hydroxyl groups found in polysaccharides suffer protonation in acidic medium, because the pair of electrons in the oxygen atom turns it into basic medium. Thus, hydroxyl acts as base and accepts positively-charged protons (Solomons & Fryhle, 2001). This process increases molecule polarity, as well as its affinity to the polar solvent (water). This is a likely explanation for the higher fiber rates recorded for acidic and basic pH extractions than for the extraction performed at almost neutral pH.
Purification stage
Water-soluble polymers extracted from linseed form a true solution in water, where the molecular polymer-solvent interaction is greater than the polymer-polymer interaction, a fact that separates polymer chains from each other. Polymeric conformation stability in the solution is linked to the hydrodynamic volume taken by the polymer, which increases and, consequently, gets more stable in the presence of a good solvent.
The opposite happens in the precipitation process, since the addition of a non-solvent (ethanol) to the real solution formed by the polymers and a good solvent leads to decreased hydrodynamic polymer volume. Furthermore, the polymer-polymer interaction increases to the point of being greater than the polymer-solvent interaction, a fact that leads to increased approximation and interactions between chains. This process accounts for the collapse of the conformational structure of polymers and triggers the immediate precipitation of macromolecule chains (Canevarolo Junior., 2006).
According to Oohma and Mazza (1993), proteins found in the water-soluble product extracted from flaxseedare natural polymers that present low solubility in ethanol. This finding suggests that proteins also suffer a precipitation process due to alcohol precipitation along with polysaccharides. However, the amount of protein found in the flaxseed mucilage is lower than the amount of polysaccharides in it, since polysaccharides are found in the outer layer of the seed husk. Based on the methodology adopted to investigate proteins found in flaxseed, the husk is removed before the extraction process, which indicates that the protein is found in the inner seed layer (Krause, Schultz, & Dudek, 2002). According to the Brazilian Food Composition Table (Table 2), flaxseed has the following composition:
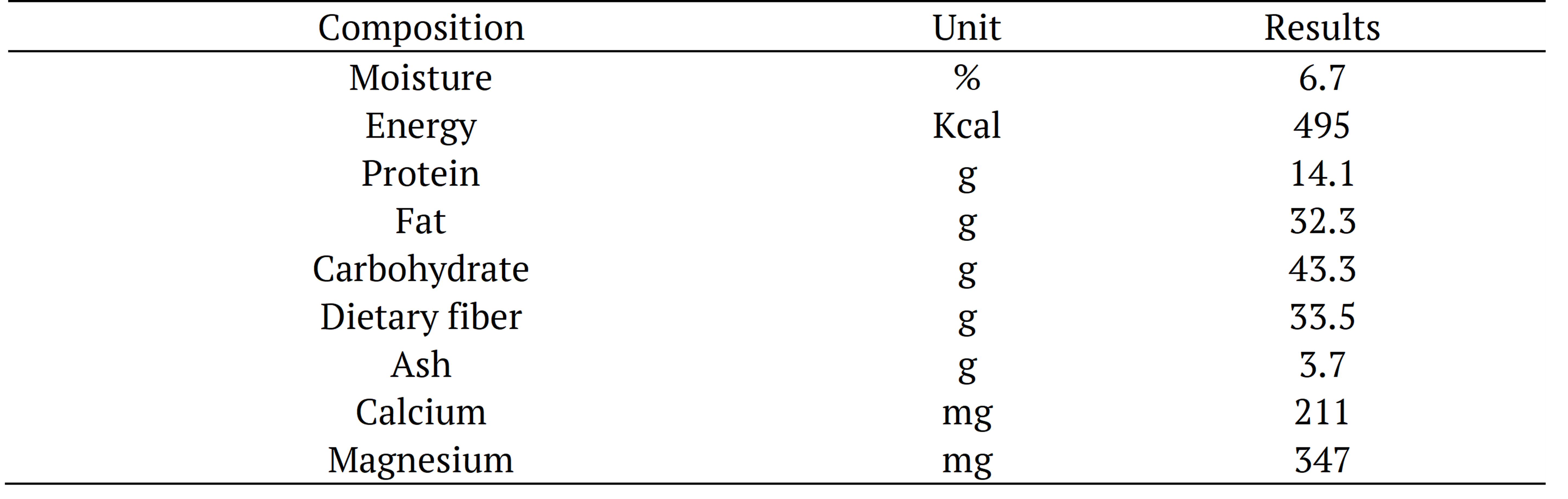
Source: Núcleo de Estudos e Pesquisas em Alimentação-Unicamp (2006).
According to Wannerberger, Nylander, and Nyman (1991), the flaxseed mucilage composition has shown low protein amounts in different flaxseed crops. More recently, Kaushik, Dowling, Adhikari, Barrow, and Adhikari (2017) have found protein and polysaccharidevalues in linseed mucilages extracted under different conditions remarkably close to that found by Wannerberger et al. (1991). Kaushik et al. (2017) recorded 7.9% of protein and 87% of polysaccharides, on average.
The association between protein solubility profile and medium pH was analyzed by Krause et al. (2002), who recorded the highest protein solubility at acid pH, which was followed by basic pH, whereas minimal protein solubilization took place at almost neutral pH.
Infrared spectroscopy is widely used to feature polymeric materials. It allows gathering information about structural aspects, such as chemical composition, based on the vibrational analysis of polymers, since it indicates the type of chemical bond taking place in them and the intensity of the absorbed infrared radiation, which is associated with the bond rate recorded for the polymer.
FTIR was carried out in the present study in order to identify the main functional groups of samples, to investigate whether extraction carried out at different pH values would influence the quality of the extracted product, as well as to check the effect of ethanol-based precipitation, as recommended by the literature in the field (Cui et al., 1994).
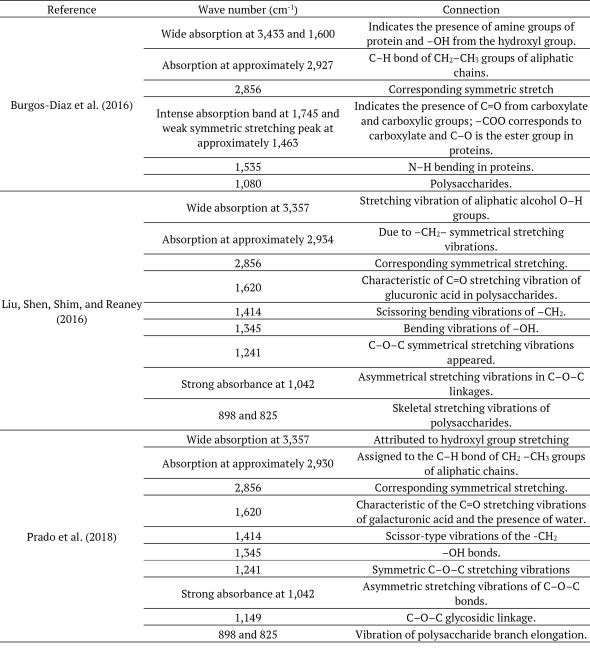
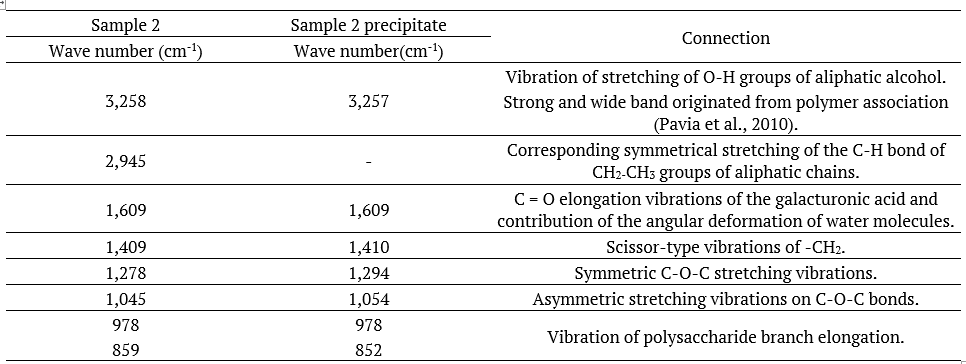
Spectra generated from different samples were compared to the description of peaks already reported in the literature (Table 3).
Based on the analysis applied to the spectrum of Sample 1extracted at acid pH (Figure 6), it was possible observing peaks associated with polymeric structure andprotein bonds (Table 4).
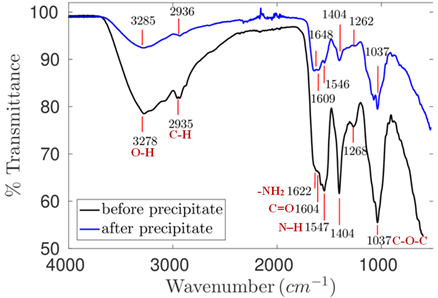
Figure 6. Infrared spectrum of the product extracted from flaxseed at pH 3.81, 45°C and 13.25h of extraction.

Based on the comparison between pre- and post-precipitation spectra, the ethanol-based polymer purification step did not remove all impurities from mucilage because the protein structure-related peaks remained in the sample, as seen in Figure 6 and Table 4.
According to Cui et al. (1994), the highest protein extraction rate was recorded at acid pH (pH 2); this result was justified by protein solubility. However, there were low intensity polymer and protein peaks, and it indicates likely reduced concentration of these bonds. This reduction may be linked to material loss since, according to Asp, Johansson, Hallmer, & Siljestrom (1983), there may be acidic hydrolysis of the polymer that, once broken, does not undergo full precipitation process. It happens because part of the arabinose side chains gets free and presents low molecular weight, which leads to loss of these in the solution during purification stage.
The spectrum of sample 2 - pH 6.75 (Figure 7), extracted at almost neutral pH, presented the peaks of bonds observed in the flaxseed polymer structure, as described in the literature (Table 5).
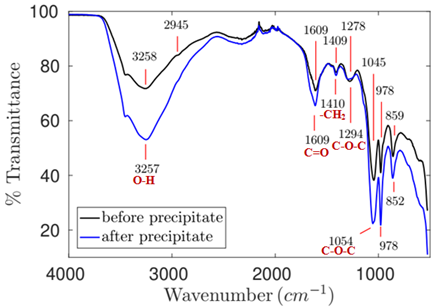
Figure 7. Infrared spectrum of the product extracted from flaxseed at pH 6.75, 45 °C and 13.25h of extraction.
The spectrum of sample 2 after ethanol-based purification has virtually shown the same flaxseed polymer bonding peaks, although some bonds presented greater absorption intensity, such as the asymmetrical elongation vibrations in the C-O-C bonds and the elongation vibration of the O-H groups of aliphatic alcohol.
The higher bond peak intensity indicates that the concentration of these bonds has increased after the sample purification step. It may have happened due to increase in the corresponding groups, which may have failed to link to some impurity that has been removed.
The presence of protein in the sample was not observed due to lack of peaks associated with protein structure. The fact that the extraction took place at almost neutral pH has influenced its extraction, since according to Krause et al. (2002), there is minimal protein solubility at this pH range.
Based on the spectrum analysis of sample 3 – pH 9.69 (Figure 8), extracted at basic pH, it was possible observing peaks associated with bonds in the linseed polymer structure (Table 6).
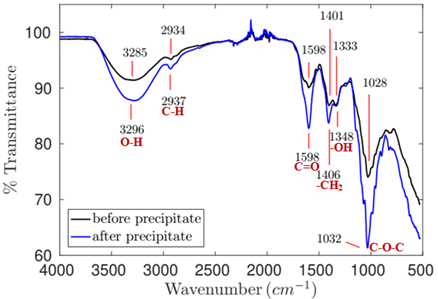
Figure 8. Infrared spectrum of the product extracted from flaxseed at pH 9.69, 45°C and 13.25h of extraction.
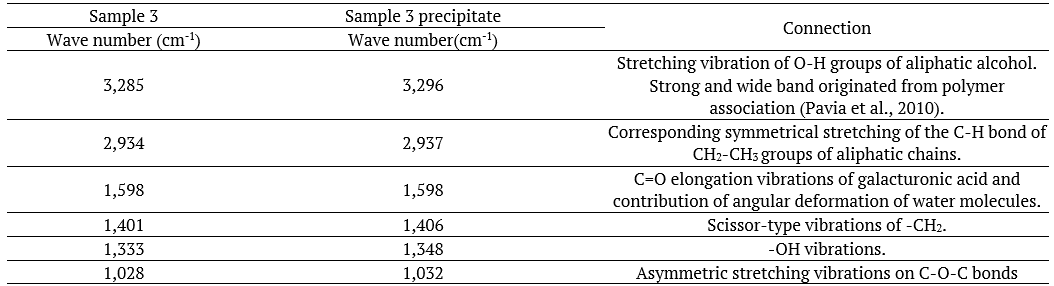
The spectrum of sample 3, after the ethanol-based purification step, has shown all peaks referring to the linseed polymer structure bonds. However, unlike sample 2, almost all peaks recorded for the IR spectrum of sample 3 (after purification) became more intense. In this case, impurity removal based on precipitation was more efficient, since there was likely increase in the concentration of the main polymeric bonds.
Based on the comparison between spectra of samples 1 (pH 3.81) and 3 (pH 9.69) (Figures 6 and 8, respectively), which were generated at pH values capable of enabling the highest fiber content (Table 1), it was possible seeing that the sample extracted in basic medium appeared to be purer than the sample extracted in the acidic medium, since it presented transmittance peaks of connections associated with the structure of flaxseed polymers; the presence of protein in it was not clear.
According to Wannerberger et al. (1991), there was not significant difference between the relative percentage of constituents in the total product extracted from linseed and the precipitated product, in comparison to constituents in polymeric units. Results reported by Wannerberger et al. (1991) have reinforced the lack of need for an ethanol-based purification stage in the case of flaxseed mucilage obtained through aqueous extraction. Based on the analysis applied to the spectra of the three experimental conditions (acidic, neutral and basic pH) and of the three precipitates, it was possible observing that pH has influenced the quality of the product, that ethanol-based precipitation did not remove the proteins and that other impurities did not affect the type of bond between polymers, although it affected the intensity of these bonds. The importance of the precipitation stage must be analyzed in each case, since the cost and the work with an additional purification stage would not be advantageous. Results in the current study have evidenced small changes in the quality of the product after precipitation.
Conclusion
It was possible determining that the aqueous extraction of Linum usitatissimum L. seed at pH 9.69 enabled the best quality extraction product. Based on the physical characterization of samples through infrared spectroscopy, the product extracted in basic medium at pH 9.69 appeared to present better purity degree than the product extracted in acidic medium. Apparently, the protein concentration in the basic medium was relatively small, to the point that its bonds did not appear in the infrared spectrum.
Extraction conducted in aqueous medium at acidic pH has also resulted in good percentage of biological macromolecules. However, the infrared spectra enabled determining the presence of protein in the mucilage. On the other hand, the aqueous extraction at almost neutral pH resulted in the lowest amount of hydrophilic polymers, although the highest yield was recorded for the extraction of all water-soluble substances found in flaxseed.
The polymer purification step carried out through ethanol-based precipitation was not an essential process, since it was not capable of fully remove impurities from the samples. Suppressing this step enabled shortening the time and decreasing the amount of material to be used in the process, a fact that increased process safety and improved the scale-up.
Acknowledgements
The authors would like to thank the Brazilian Government agency CAPES for their support.
References
Adeleye, O. A., Femi-Oyewo, M. N., & Odeniyi, M. A. (2014). The effect of processing variables on the mechanical and release properties of tramadol matrix tablets incorporating Cissus populnea gum as controlled release excipient. Polimery W Medycynie, 44(4), 209-220. Retrieved from https://bit.ly/36vuRg7
Anvisa, & Fundação Oswaldo Cruz. (2010). Farmacopeia Brasileira (5a ed., Vol. 1). Brasília, DF: Anvisa.
Asp, N. G., Johansson, C. G., Hallmer, H., & Siljestrom, M. (1983). Rapid enzymatic assay of insoluble and soluble dietary fibers. Journal of Agricultural and Food Chemistry, 31(3), 476-482. doi: 10.1021/jf00117a003
Burgos-Díaz, C., Rubilar, M., Morales, E., Medina, C., Acevedo, F., Marqués, A. M., & Shene, C. (2016). Naturally occurring protein–polysaccharide complexes from linseed (Linum usitatissimum) as bioemulsifiers. European Journal of Lipid Science and Technology, 118(2), 165-174. doi: 10.1002/ejlt.201500069
Canevarolo Junior., S.V. (2006). Ciência dos polímeros: Um texto básico para tecnólogos e engenheiros (2a ed.). São Paulo, SP: Artliber.
Chen, H. H., Xu, S. -Y., & Wang, Z. (2006). Gelation properties of flaxseed gum. Journal of Food Engineering, 77(2), 295-303. doi: 10.1016/j.jfoodeng.2005.06.033
Cui, W., & Mazza, G. (1996). Physicochemical characteristics of flaxseed gum. Food Research International, 29(3-4), 397-402. doi: 10.1016/0963-9969(96)00005-1
Cui, W., Mazza, G., Oomah, B. D., & Biliaderis, C.G. (1994). Optimization of an aqueous extraction process for flaxseed gum by response surface methodology. LWT – Food Science and Technology, 27(4), 363-369. doi: 10.1006/fstl.1994.1074
Deore, S. L., & Khadabadi, S. S. (2008). Standardisation and pharmaceutical evaluation of Chlorophytum borivilianum mucilage. Rasayan Journal Chemistry, 1(4), 887-92. Retrieved from https://bit.ly/3kIbarf
Dzuvor, C. K. O., Taylor, J. T., Acquah, C., Pan, S., & Agyei, D. (2018). Bioprocessing of functional ingredients from flaxseed. Molecules, 23(10), 1-18. doi: 10.3390/molecules23102444
Haseeb, M. T., Hussain, M. A., Bashir, S., Ashraf, M. U., & Ahmad, N. (2017). Evaluation of superabsorbent linseed-polysaccharides as a novel stimuli-responsive oral sustained release drug delivery system. Drug Development and Industrial Pharmacy, 43(3), 409-420. doi: 10.1080/03639045.2016.1257017
Haseeb, M. T., Hussain, M. A., Yuk, S.H., Bashir, S., & Nauman, M. (2016). Polysaccharides based superabsorbent hydrogel from linseed: Dynamic swelling, stimuli responsive on-off switching and drug release. Carbohydrate Polymers, 136, 750-756. doi: 10.1016/j.carbpol.2015.09.092
Horwitz, W. (2000). Official methods of analysis of AOAC Internacional (17th ed.). Gaithersburg, MD: AOAC.
Kaewmanee, T., Bagnasco, L., Benjakul, S., Lanteri, S., Morelli, C. F., Speranza, G., & Cosulich, M. E. (2013). Characterisation of mucilages extracted from seven Italian cultivars of flax. Food Chemistry, 148, 60-69. doi: 10.1016/j.foodchem.2013.10.022
Kaushik, P., Dowling, K., Adhikari, R., Barrow, C. J., & Adhikari, B. (2017). Effect of extraction temperature on composition, structure and functional properties of flaxseed gum. Food Chemistry, 215, 333-340. doi: 10.1016/j.foodchem.2016.07.137
Khinchi, M. P., Gupta, M. K., Bhandari, A., Agarwal, D., & Sharma, N. (2011). Studies on the disintegrant properties of seed powder, husk powder and mucilage of Plantago ovata by formulation of orally disintegrating tablet. International Journal of Pharmaceutical Sciences and Research, 2(1), 145-152. doi: 10.13040/IJPSR.0975-8232.2(1).145-52
Koocheki, A., Mortazavi, S. A., Shahidi, F., Razavi, S. M. A., Kadkhodaee, R., & Milani, J. M. (2010). Optimization of mucilage extraction from qodume shirazi seed (Alyssum homolocarpum) using response surface methodology. Journal of Food Process Engineering, 33(5), 861-882. doi: 10.1111/j.1745-4530.2008.00312.x
Koocheki, A., Taherian, A. R., Razavi, S. M., & Bostan, A. (2009). Response surface methodology for optimization of extraction yield, viscosity, hue and emulsion stability of mucilage extracted from Lepidium perfoliatum seeds. Food Hydrocolloids, 23(8), 2369-2379. doi: 10.1016/j.foodhyd.2009.06.014
Krause, J. P., Schultz, M., & Dudek, S. (2002). Effect of extraction conditions on composition, surface activity and rheological properties of protein isolates from flaxseed (Linum usitativissimum L). Journal of the Science of Food and Agriculture, 82(9), 970-976. doi: 10.1002/jsfa.1140
Lima Junior, F. A., Conceição, M. C., Resende, J. V., Junqueira, L. A., Pereira, C. G., & Prado, M. E. T. (2013). Response surface methodology for optimization of the mucilage extraction process from Pereskia aculeata Miller. Food Hydrocolloids, 33(1), 38-47. doi: 10.1016/j.foodhyd.2013.02.012
Liu, J., Shen, J., Shim, Y. Y., & Reaney, M. J. T. (2016). Carboxymethyl derivatives of flaxseed (Linum usitatissimum L.) gum: Characterisation and solution rheology. International Journal of Food Science & Technology, 51(2), 530-541. doi: 10.1111/ijfs.12985
Moreira, L. M., Rodrigues, M. R., Oliveira, H. P. M., Lima, A., Soares, R. R. S., Batistela, V. R., ... Machado, A. E. D. H. (2010). Influência de diferentes sistemas de solvente água-etanol sobre as propriedades físico-químicas e espectroscópicas dos compostos macrocíclicos feofitina e clorofila Α. Química Nova, 33(2), 258-262. doi: 10.1590/S0100-40422010000200005
Nasrabadi, M. N., Goli, S. A. H., Doost, A. S., & Van der Meeren, P. (2020). Characterization and enhanced functionality of nanoparticles based on linseed protein and linseed gum biocomplexes. International Journal of Biological Macromolecules, 151, 116-123. doi: 10.1016/j.ijbiomac.2020.02.149
Nerkar, P. P., &Gattani, S. G. (2011). In vivo, in vitro evaluation of linseed mucilage based buccal mucoadhesive microspheres of venlafaxine. Drug Delivery, 18, 111-121. doi: 10.3109/10717544.2010.520351
Nerkar, P. P., & Gattani, S. G. (2013). Oromucosal delivery of venlafaxine by linseed mucilage based gel: in vitro and in vivo evaluation in rabbits. Archives of Pharmacal Research, 36(7), 846-853. doi: 10.1007/s12272-013-0097-3
Núcleo de Estudos e Pesquisas em Alimentação-Unicamp. (2006). Tabela brasileira de composição de alimentos (2a. ed.). Campinas, SP: Nepa-Unicamp.
Odeku, O. A., & Patani, B. O. (2005). Evaluation of dika nut mucilage (Irvingia gabonensis) as binding agent in metronidazole tablet formulations. Pharmaceutical Development and Technology, 10(3), 439-446. doi: 10.1081/PDT-54477
Oomah, B. D., & Mazza, G. (1993). Flaxseed proteins: A review. Food chemistry, 48(2), 109-114. doi: 10.1016/0308-8146(93)90043-F
Patel, M. T., Patel, J. K., &Upadhyay, M. U. (2012). Assessment of various pharmaceutical excipient properties of natural Moringa oleifera gum. International Journal of Pharmacy & Life Sciences, 3(7) 1833-1847. Retrieved from https://bit.ly/363Z45Z
Pavia, D. L., Lampman, G. M., & Kriz, G. S. (2010). Introdução à espectroscopia. São Paulo, SP: Cengage Learning.
Prado, N. S., Silva, I. S. V., Silva, T. A. L., Oliveira, W. J., Motta, L. A. C., Pasquini, D., & Otaguro, H. (2018). Nano composite films based on flaxseed gum and cellulose nanocrystals. Materials Research, 21(6). doi: 10.1590/1980-5373-mr-2018-0134
Prajapati, V. D., Jani, G. K., Moradiya, N. G., & Randeria, N.P. (2013). Pharmaceutical applications of various natural gums, mucilages and their modified forms. Carbohydrate Polymers, 92(2), 1685-1699. doi: 10.1016/j.carbpol.2012.11.021
Solomons, G., & Fryhle, C. (2001). Química orgânica (7a ed., Vol. 1). Rio de Janeiro, RJ: Editora LTC.
Verma, P. R. P., &Razdan, B. (2003). Studies on Leucaena leucocephala seed gum: Emulsifying properties. Journal of Scientific & Industrial Research, 62, 198-206. Retrieved from https://bit.ly/3jLKDIi
Wannerberger, K., Nylander, T., & Nyman M. (1991). Rheological and chemical properties of mucilage in different varieties from linseed (Linum usitatissimum). Acta Agriculturae Scandinavica, 41(3), 311-319. doi: 10.1080/00015129109439914
Western, T. L. (2012). The sticky tale of seed coat mucilages: Production, genetics, and role in seed germination and dispersal. Seed Science Research, 22(1), 1-25. doi: 10.1017/S0960258511000249
Notas de autor
souzarocha18@gmail.com