Ciência, Tecnologia Alimentar e Engenharia de Alimentos
Macronutrients, vitamins, minerals and bioactive compounds in fresh and dehydrated basil (Ocimum basilicum) and its hot and cold infusions
Macronutrients, vitamins, minerals and bioactive compounds in fresh and dehydrated basil (Ocimum basilicum) and its hot and cold infusions
Acta Scientiarum. Technology, vol. 43, e55423, 2021
Universidade Estadual de Maringá
Recepción: 25 Agosto 2020
Aprobación: 14 Octubre 2020
Abstract: Basil (Ocimum basilicum L.) is one of the most popular plants cultivated worldwide. It is a good source of natural antioxidants and contains significant amounts of important phytochemicals. Thus, this study aimed to evaluate the macronutrients, minerals, vitamins and bioactive compounds of fresh and dehydrated basil and its hot and cold infusions. The basil presented high moisture (94.12%) and low lipid (0.20%), carbohydrates (5.25%) and dietary fibers (2.33%) content. There was no difference in vitamin C concentrations, total phenolics and antioxidant capacity between fresh and dehydrated basil and between the hot and cold infusions (p > 0.05). Potassium and calcium were the most prevalent minerals in dehydrated basil. The consumption of basil, especially in dehydrated form, can contribute significantly to the daily intake of minerals, vitamins C and E, and infusions are not a relevant choice of consumption to achieve nutritional recommendations.
Keywords: Aromatic herb, basil tea, antioxidant capacity, micronutrients, HPLC.
Introduction
Basil (Ocimum basilicum L.), belonging to the family Lamiaceae, is part of a group of medicinal and aromatic plants of great economic value. It is one of the most popular plants cultivated worldwide (Dumbrava, Moldovan, Raba, & Popa, 2012), including Brazil, where the climate is favourable (Carvalho et al., 2012).
It is widely used for cooking, but also in commercial fragrances, flavourings and medicines (Dumbrava et al., 2012). Its use is recommended as a digestive tonic, with antimicrobial, antibacterial, anticonvulsant and anticarcinogenic properties (Ch, Naz, Sharif, Akram, & Saeed, 2015; Kwee & Niemeyer, 2011). It is also used for curing ailments such as warts, inflammations, colds and headaches (Ch et al., 2015). Basil is a good source of natural antioxidants, and contains significant amounts of important phytochemicals, such as phenolic compounds and ascorbic acid contents (Aburigal et al., 2017; Kwee & Niemeyer, 2011; Muráriková & Neugebauerová, 2018). However, the nutritional and bioactive characterization, especially vitamins and carotenoids, of this herb is scarce in the literature, with a greater focus on its antioxidant capacity (Prinsi, Morgutti, Negrini, Faoro, & Espen, 2020; Silva, 2011) and phenolic compounds (Prinsi et al., 2020). To date we found no studies that evaluated the bioactive compounds of basil tea prepared by hot and cold infusion.
Tea is the second most consumed beverage aside from water and has gained much attention due to its health-promoting benefits associated in part to the antioxidant activity of chemical compounds present in teas, especially flavonoids and phenolic acids (Zielinski et al., 2014). The temperature plays an important role on the extraction of bioactive compounds in tea. The use of hot water in the preparation of tea infusion is more popular, and the cold extraction is an alternative to the consumers, resulting in a beverage with higher concentrations of bioactive compounds (Rodrigues, Silva, Santos, Zielinski, & Haminiuk, 2015). Thus, the evaluation of nutrients and bioactive compounds in hot and cold infusions is essential to determine their health benefits.
This study aimed to analyze the occurrence and concentration of macronutrients, dietary fibers, carotenoids, vitamins, minerals, phenolic compounds and antioxidant capacity of fresh and dehydrated basil and its hot and cold infusions.
Material and methods
Chemicals
Analytical grade reagents (VETEC, São Paulo, Brazil) were used for vitamins, carotenoids and flavonoids extraction. For analysis, we used HPLC grade reagents (methanol, acetonitrile, formic acid, ethyl acetate, acetone, hexane, isopropanol, glacial acetic acid) obtained from Tedia (São Paulo, Brazil).
The standards of carotenoids (β-carotene and lutein) and flavonoids (flavones: luteolin and apigenin; and flavanones: naringenin and eriodictyol) were purchased from Sigma-Aldrich. The vitamin C standard (L-ascorbic acid) was obtained from Proquímios and vitamin E standards (α, β, γ and δ-tocopherols and tocotrienols) from Calbiochem. The purity of the standards was calculated by spectrophotometry. Standards purity was above 98%.
Samples
About 200-400 g of Ocimum basilicum L. were purchased from a local market or were collected at various branches and flowerbeds at two locations in the rural area of the municipality of Viçosa, Minas Gerais, Brazil. The leaves were washed in running water, dried with paper towels and stored at 4 °C ± 1 °C, protected from light, until analysis of carotenoids, vitamins and flavonoids.
For the analysis of macronutrients, dietary fibers and minerals, the herbs were dehydrated in an oven (Quimis, model Q320M, Brazil) at 80 °C for 3h, protected from light and stored at -18 ± 1 °C until the analyses.
Preparation of infusions
Hot extraction was performed according to Zielinski et al. (2014). Two grams of dehydrated sample were added with 100 mL of distilled water at 80°C, leaving under magnetic stirring for 7.5 min.
The extraction in cold water was performed with 2.0 g of dehydrated sample in 100 mL of distilled water, at a temperature of 7 to 10°C, immersed for about 15 min. under magnetic stirring.
All extracts were filtered on 125 mm quantitative filter paper and stored at -18 ± 1°C until analysis. An aliquot of the extract was withdrawn for analysis.
Determination of the centesimal composition
Moisture, ashes, proteins and lipids were determined in triplicate, and dietary fiber in duplicate, all according to the Association of Official Analytical Chemists (AOAC, 2012).
Moisture was determined using a forced air circulation oven (Nova Ética, model 400/6ND, São Paulo, Brazil) at 105°C and the ashes were quantified using a muffle furnace (Quimis, model Q320 M, Brazil) at 550°C. Carbohydrates were calculated by difference, using the following equation: [100 - (% moisture + % lipids + % protein + % total dietary fiber + % ash)]. The total energetic value of basil was estimated considering the conversion factors of 4 kcal g-1 for protein or carbohydrates and 9 kcal g-1 for lipids.
Determination of minerals and inorganic contaminants
Mineral analysis was performed only on the dehydrated herb, since it resembles the herb marketed in sachets in markets, with easy access to the population.
For analysis and determination of Ca, Mg, Fe, Cu, Zn, Mn, Ni, Cd, Al and Pb, it was used nitroperchloric digestion (Sarruge & Haag, 1974) and the analyses were performed by optical emission spectrometry with inductively coupled plasma (ICP-OES). The determination of P was made by colorimetry using the ascorbic acid method (Braga & Defelipo, 1974); K determination was performed by flame photometry; the determination of S was performed by turbidimetry according to the method of Alvarez, Dias, Ribeiro Jr., and Fonseca (2001).
Extraction and analysis of carotenoids
Carotenoids (lutein and β-carotene) were extracted according to the Rodriguez-Amaya (2001) method, with adaptations. 2.5 g of dehydrated basil, 5 g of fresh basil, or 20 mL of the infusions were homogenized in 20 mL of cooled acetone with a microgrinder (IKA® T18 basic Ultra Turrax®) for 5 min. The suspension obtained was vacuum filtered on Büchner funnel with filter paper, with the residue being maintained in the extraction tube. The extraction procedure was repeated two more times. The filtrate was then transferred, in three fractions, to a separatory funnel containing 50 mL of cooled petroleum ether. Each fraction was washed with distilled water 3 times to remove all acetone. Subsequently, the extract was concentrated in a rotary evaporator (Tecnal TE-211) at 35 ± 2°C, transferred to a 25 mL volumetric flask, the volume filled with petroleum ether, and stored in an amber glass at -18 ± 2°C. An aliquot of 5 mL of the sample was evaporated under nitrogen gas flow, and then recovered in 2 mL of HPLC grade acetone. The extract was filtered in filter units (0.45 μm, Millipore, Brazil).
The analyses were according to Sant’Ana, Stringheta, Brandão, Páez, and Queiroz (1998), using a high performance liquid chromatography system (HPLC) (Shimadzu, SCL 10AT VP model, Japan) comprised of a high-pressure pump (Shimadzu, LC-10AT VP model, Japan), an autosampler with a loop of 500 µL (Shimadzu, SIL-10AF model, Japan) and a diode array detector (DAD) (Shimadzu, SPD-M10A model, Japan). The following chromatographic conditions were used: chromatographic column (Phenomenex Gemini, 250 x 4.6 mm, 5 µm), fitted with a guard column (C18) (Phenomenex ODS 4 mm x 3 mm); mobile phase composed of methanol: ethyl acetate: acetonitrile (80:10:10), with flow rate of 2.0 mL min-1, run time of 12 min. and injection volume of 30 µL. The chromatograms were obtained at 450 nm.
The identification of the carotenoids was performed by comparing the retention times and the absorption spectra of authentic standards and of the peaks of interest in the samples, analyzed under the same conditions. Quantification was performed by external standardization using analytical curves constructed from the injection of six different concentrations of standard solutions. The carotenoids were expressed in mg.100g-1 of sample, as isolated components and as a sum of carotenoids. Vitamin A concentration was calculated according to the recommendations of the Institute of Medicine (2001), wherein 1 Retinol Activity Equivalent (RAE) corresponds to 1 μg of retinol; 12 μg β-carotene; 24 μg of other pro-vitamin carotenoids.
Extraction and analysis of vitamin C
The conditions used for extraction and analysis of vitamin C as ascorbic acid (AA) were that optimized by Campos, Ribeiro, Della Lucia, Sant’Ana, and Stringheta (2009).
2.5 g of dehydrated basil and 5 g of fresh basil were added with 15 mL of extractive solution (3% metaphosphoric acid, 8% acetic acid, 0.3 N sulfuric acid and 1 mM EDTA) and homogenized with microgrinder (IKA® T18 basic Ultra Turrax®) for 5 min., followed by centrifugation (Nuve® NF 1200R Bench-Top Centrifuge) at 2349 g for 15 min. The supernatant was collected, and vacuum filtered on Büchner funnel with filter paper.
For the extraction of AA from the infusions, an aliquot of 20 mL was added with 15 mL of the extractive solution, following the same extraction steps proposed by Campos et al. (2009).
For the analysis of AA, the chromatographic conditions used was: HPLC-DAD system (Shimadzu, SPD-M10A, Japan); Chromatographic column Synergy Hydro 100 RP 250, 250 x 4.6 mm, 5 μm, equipped with guard column (C 18, 4 mm x 3 mm); mobile phase composed of 1 mM NaH2PO4, 1 mM EDTA, pH adjusted to 3.0; flow rate of 1.0 mL min-1. The running time was approximately 7 min.
The identification of AA was performed by comparing the retention times and by the absorption spectra of the authentic standard and the peak of interest in the samples, analyzed under the same conditions. The quantification was performed by external standardization, using an analytical curve constructed by injection, in duplicate, of a standard solution.
Extraction and analysis of vitamin E
The extraction and analysis of vitamin E components (α, β, γ and δ-tocopherols and tocotrienols) were performed only on fresh and dehydrated basil. Vitamin E analysis was not performed on infusions, since the protocol used is not validated for analysis in aqueous medium and, furthermore, because they are apolar, the reagents used are not capable of extracting vitamin E from the water.
The method was based on Sant'ana et al. (2011), with some modifications. Ten grams of fresh basil and 5 grams of dehydrated basil were added to 4 mL of warm ultrapure water (80 ± 1°C), 10 mL of isopropanol, 1 mL of hexane containing 0.05% BHT, 5 g of anhydrous sodium sulfate and 25 mL of the extraction solvent mixture (hexane: ethyl acetate, 85:15, v v-1). Subsequently, the suspension was homogenized using microgrinder (IKA T 18 Ultra Turbax) for 1 min. followed by vacuum filtration on Büchner funnel using filter paper. The extraction step was repeated, adding to the residue 5 mL of isopropanol and 30 mL of the extraction solvent mixture, with subsequent homogenization for 1 min. The extract was then concentrated in a rotary evaporator (Quimis, model 344.1) at 70 ± 1°C (2 min.), transferred to volumetric flask and the volume completed to 25 mL with extraction solvent mixture.
After extraction, aliquots of 5 mL of the extract were evaporated under nitrogen gas flow, recovered in 2 mL of HPLC grade hexane and filtered using 0.45 μm porosity filter units (Millipore, Brazil).
Vitamin E analyses were performed by injecting 1 and 10 μL of the extracts. Chromatographic conditions used included: HPLC system (Shimadzu, SCL 10AD VP) with fluorescence detector (excitation at 290 nm and emission 330 nm); chromatographic column Phenomenex Luna Si100 (250 x 4.6 mm, 5 μm) coupled to the Phenomenex Si100 guard column (4 x 3 mm); mobile phase composed of hexane: isopropanol: glacial acetic acid (98.9: 0.6: 0.5, v v v-1); flow rate of 1.0 mL min.-1 and run time of 21 min.
Identification of vitamin E components was performed by comparing the retention time of the authentic standards, and the components of interest in the samples, analyzed under the same conditions. The quantification was performed by external standardization, comparing the peak areas with those obtained in the analytical curves constructed from the injection, in duplicate, of six different concentrations of solutions of the commercial standards. Vitamin E concentrations were expressed in mg.100g-1 as isolated components and as total vitamin E (sum of the vitamin E components found in the samples).
Extraction and analysis of flavonoids
Flavones (luteolin and apigenin) and flavanones (naringenin and eriodictyol) were investigated in the herb. 2.5 grams of dehydrated and fresh basil were added to 10 mL of 1% methanol/HCl solution (v:v), followed by stirring (2h, 180 rpm) (Marconi, MA231, Brazil) and centrifugation (Nuve® NF 1200R Bench Top Centrifuge) at 2349 g for 5 min. (Dykes, Peterson, Rooney, & Rooney, 2011). The concentration of flavonoids was analyzed simultaneously by HPLC. The method proposed by Yang, Allred, Geera, Allred, and Awika (2012) and modified by Cardoso et al. (2014) was used to identify and quantify flavonoids in basil.
The analyses were performed in a HPLC-DAD system (Shimadzu, SPD-M10A, Japan), with quaternary high pressure pump (Shimadzu, LC-10AT VP, Japan), autosampler with a loop of 500 μL (Shimadzu, SIL- 10AF, Japan) and helium gas mobile phase degassing system (Shimadzu, DGU-2A, Japan). Chromatographic conditions used included Kinetex C-18 column (150 mm x 4.6 mm, 5 μm) equipped with C-18 guard column (4 mm x 3 mm) (Phenomenex, Torrance, CA), column temperature at 35°C, injection volume of 15 and 30 μL, spectrum scan of 200 to 700 nm, with detection at 360 nm for flavones and 280 nm for flavanones. The mobile phase was composed of 2% formic acid in ultrapure water (line A) and 2% formic acid in acetonitrile (line B), and it was degassed with helium gas (50 kPa) during the runs.
The identification of the flavonoids was performed by comparing the retention times and the absorption spectra of the authentic standards and the peaks of interest in the samples, analyzed under the same conditions. Quantification was made by external standardization, using analytical curves constructed by injection, in duplicate, of five different concentrations of standard solutions. The compounds were expressed in mg.100g-1 of sample, as isolated components and as a sum of flavonoids.
Determination of total phenolic compounds
For extraction of phenolic compounds, 1 gram of fresh and dehydrated basil was added to 20 mL of 60% methanolic solution. For the infusions, a 10 mL sample of the hot and cold infusions was added with 15 mL of 60% methanolic solution. The suspension was then stirred at 180 rpm (2h) and centrifuged at 2790 g (15 min.). The supernatant was added to a beaker and the volume was completed to 20 mL with the methanolic solution, transferred to an amber flask and stored at -18 ± 1°C until analysis.
Total phenolic compounds were determined using the Folin-Ciocalteau reagent (Singleton, Orthofer, & Ranvenson, 1999). Aliquots of 0.5 mL of extract were added to 0.5 mL of Folin-Ciocalteu reagent (20%). After homogenization, 0.5 mL of sodium carbonate (7.5%) was added. The reaction mixture was homogenized by vortex (2865 g, 10 s) and incubated at room temperature (30 min.) in the dark. Absorbance reading was performed on a spectrophotometer (ThermoScientific, Evolution 60S, USA) at 765 nm. Quantification was performed using an analytical curve constructed from reading the absorbance at 765 nm of five solutions with different concentrations of gallic acid, in duplicate. The results were expressed in milligrams of gallic acid equivalents per 100 grams of sample (mg GAE 100 g-1 sample).
Determination of antioxidant capacity
The radical removal activity (RRA) of the samples was performed from the extract prepared for the analysis of total phenolic compounds using the DPPH (2,2-diphenyl-2-picryl-hydrazyl) radical method (Bloor, 2001). For this purpose, a 2.5 mL aliquot of extract was added with 1.5 mL of 0.1 mM DPPH methanolic solution (Sigma-Aldrich, Germany) (1.0 mL). Vortex agitation was performed; then the extract was incubated at room temperature (30 min.) in the dark. The absorbance was read on a spectrophotometer (ThermoScientific, ELISA Multiskan GO) at 517 nm.
Experimental design and statistical analysis of data
The concentrations of nutrients and bioactive compounds of basil were evaluated in the completely randomized design, with three replicates. Data were assessed for normality using the Shapiro-Wilk test. Then, the data were analyzed by Student's t-test or Mann Whitney test. SPSS software version 23.0 was used, adopting a level of significance (α) of 5%.
Results and discussion
Centesimal composition of basil
The basil moisture (94.12%) was higher than that observed by Ferreira and Silveira Junior (1999) (72.39%) and Reis, Devilla, Ascheri, Servulo, and Souza (2012) (82.7%) in basil samples. The ash concentration (7.9 g.100g-1) was similar to that found by Almeidai et al. (2010) in dried basil samples (5.93 to 8.46%). The lipids content (0.20%) was close to that reported by Pedro et al. (2016) for barbatimão, carqueja, and brave mangava. The carbohydrate concentration of basil was close to that reported in the Brazilian Table of Food Composition (3.6%) (Núcleo de Estudos e Pesquisas em Alimentação [NEPA], 2011)
The different values of the centesimal composition found among the studies may be related to the origin and the varieties of the herbs and the different plant structures analyzed (Bertol, Almeida, & Almeida, 2015).
Carotenoids, vitamin C and vitamin E in basil
Figure 1 shows the chromatograms of carotenoids, ascorbic acid and vitamin E in fresh and dehydrated basil.
β-carotene was found in a lower concentration than lutein in fresh and dehydrated basil (Table 1). The retention of β-carotene was around 100% in dehydrated basil, showing that this compound was preserved after heating or that the temperature could have promoted further extraction of this component and, consequently, increased its concentration.
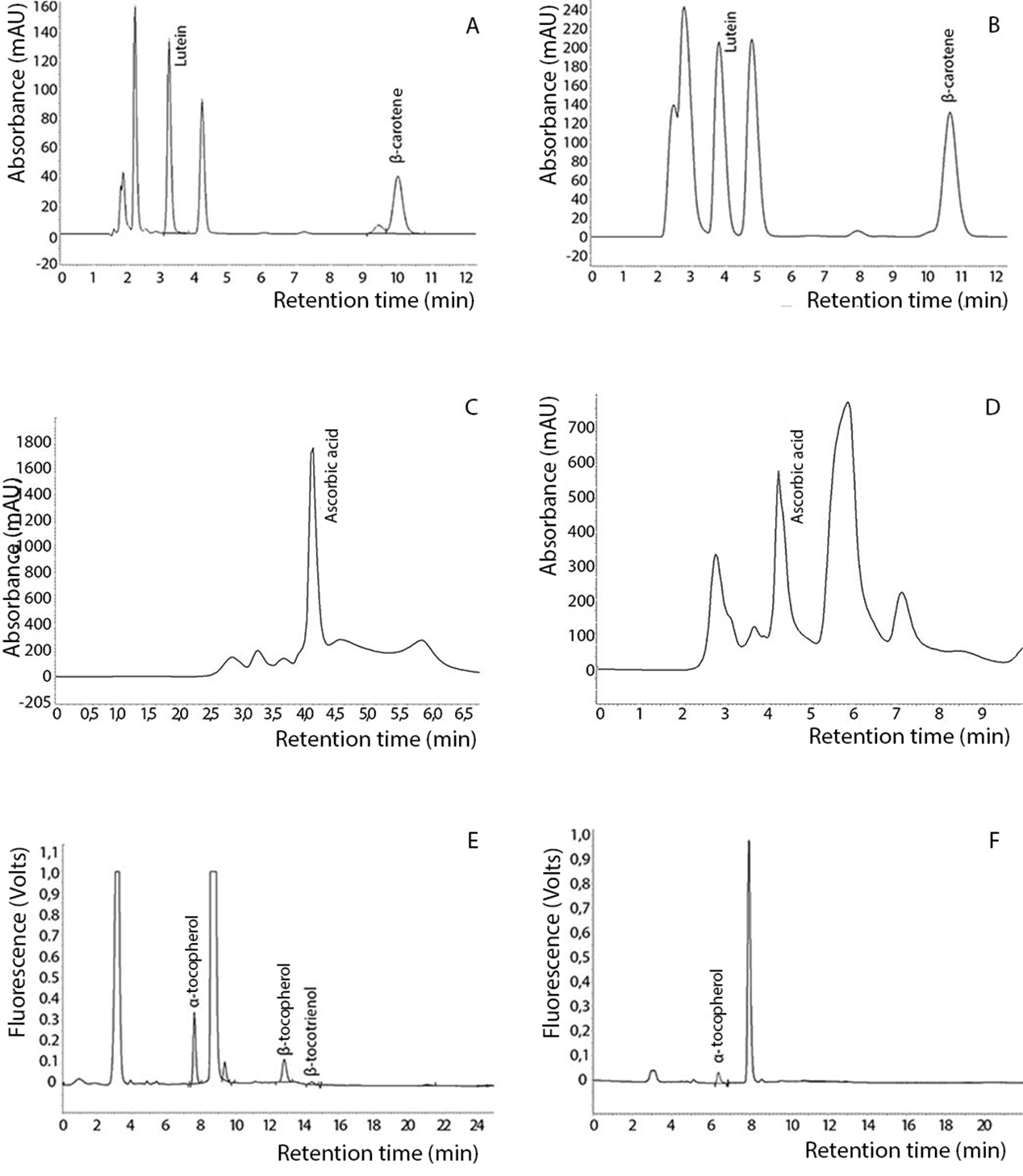
Figure 1. Analysis by HPLC of carotenoids in fresh basil (A) and dehydrated basil (B); ascorbic acid in fresh basil (C) and dehydrated basil (D); vitamin E in fresh basil (E) and dehydrated basil (F).
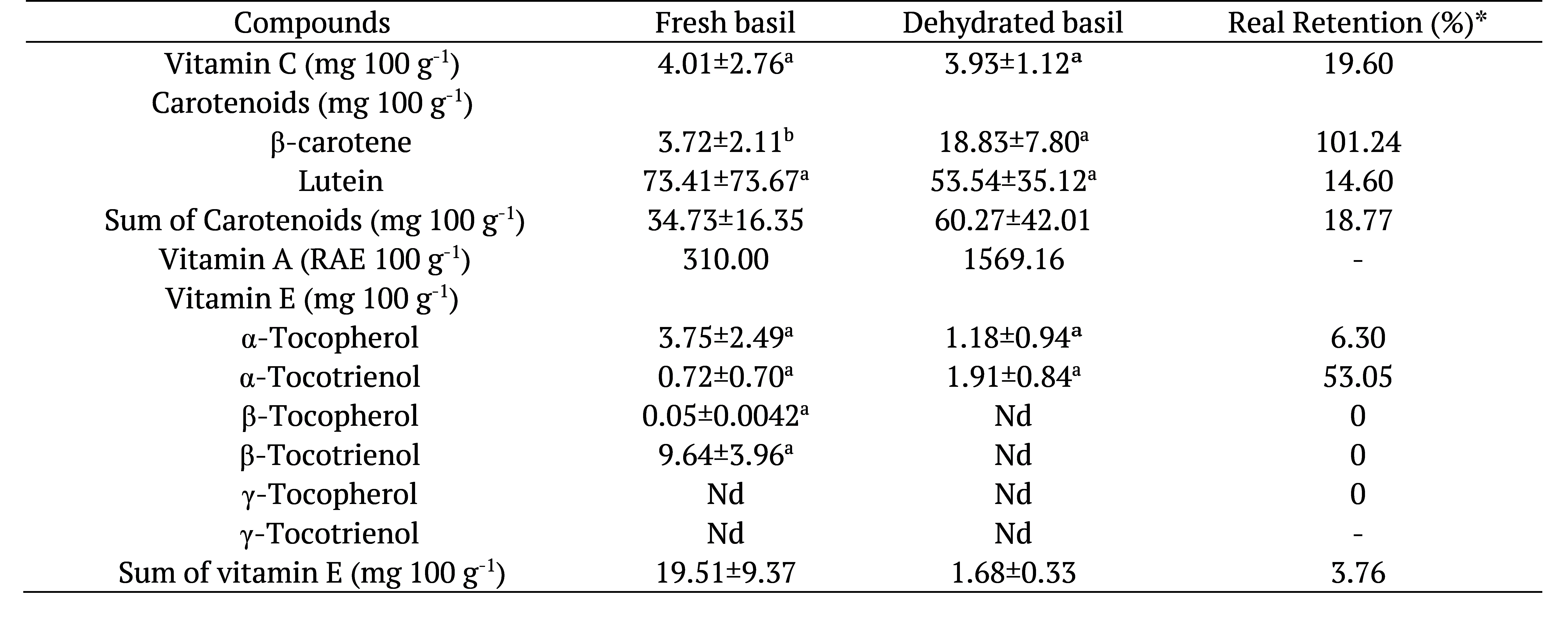
Data expressed as dry matter as mean of 3 replicates ± standard deviation. Means followed by the same letter in the rows, for each compound, do not differ statistically at 5% probability by Test t. nd: not determined; *Real Retention (% RR): Calculated according to the formula of Murphy, Criner and Gray (1975) –
The concentration of β-carotene in fresh basil was similar to that reported by Sousa and Pereira (2013) in fresh parsley (2.78 mg 100 g-1) and sage (4.56 mg 100 g-1). The concentration of lutein in fresh basil was higher when compared to fresh sage (6.35 mg 100 g-1) and fresh parsley (8.30 mg 100 g-1), respectively (Sousa & Pereira, 2013).
Vitamin C concentration did not differ (p > 0.05) between fresh and dehydrated basil, indicating that the heat processing has not been able to generate significant changes of gain or loss for the extraction. However, in dehydrated basil, there was lower concentration (19.60%) and low retention (3.76%), evidencing losses after dehydration, even without statistical significance, we can infer that the use of this plant in fresh form is a good option to take advantage of the vitamin C content.
The concentration of carotenoids and vitamin C was low and did not differ (p > 0.05) between hot and cold infusions (Table 2). The content of vitamin C and E in foods is influenced by several factors, such as crop growing conditions, soil characteristics, weather conditions, storage, and processing of food (Eitenmiller, 1997).
Leafy are good sources of vitamin E, with predominance of α-tocopherol, especially in dark green vegetables. However, β-tocotrienol predominated in the fresh basil and α-tocotrienol in the dehydrated herb (Table 1). Retention of α-tocotrienol was greater than 50%. It is important to emphasize that the information on the concentration of vitamin E in leafy vegetables, especially in herbs and herbal products is very scarce (Eitenmiller, 1997), being a differential of our study.
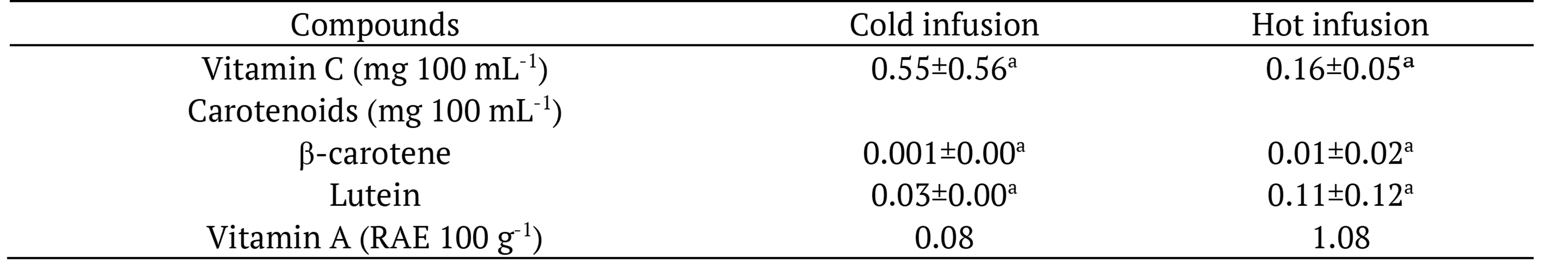
Data expressed as fresh matter as mean of 3 replicates ± standard deviation. Means followed by the same letter in the rows, for each compound, do not differ statistically at 5% probability by Test t.
Concentration of minerals and inorganic contaminants in basil
The basil stood out in the concentrations of calcium, phosphorus, and copper (Table 3) when compared to yerba mate analyzed by Heinrichs and Malavolta (2001). The concentration of minerals in the plants is variable and depends on several factors, such as variety, species, soil type, climatic conditions, fertilizer use and soil fertility (Sarruge & Haag, 1974).
No contamination was observed in basil with nickel (Ni) and cadmium (Cd), which is important since these are also considered toxic. In relation to the aluminum (Al), in 100 g of dehydrated basil we found 864.11 mg. According to Soliman and Zikovsky (1999) dietary intake of Al should not exceed 6 mg day-1. Thus, daily consumption of basil, even in small amounts (1 g), exceeds the recommendation (8.64 mg 100 g-1) and may have effects on growth, reproduction, bone, and brain tissue (Natesan & Ranganathan, 1990).
The use of fertilizers and agrochemicals, urban-industrial activities, type of irrigation and fertilization can interfere in the accumulation of trace elements in the soil, which can cause contamination of foods with toxic elements, besides increasing the bioavailability of these minerals to chemical alterations (He et al., 2015).
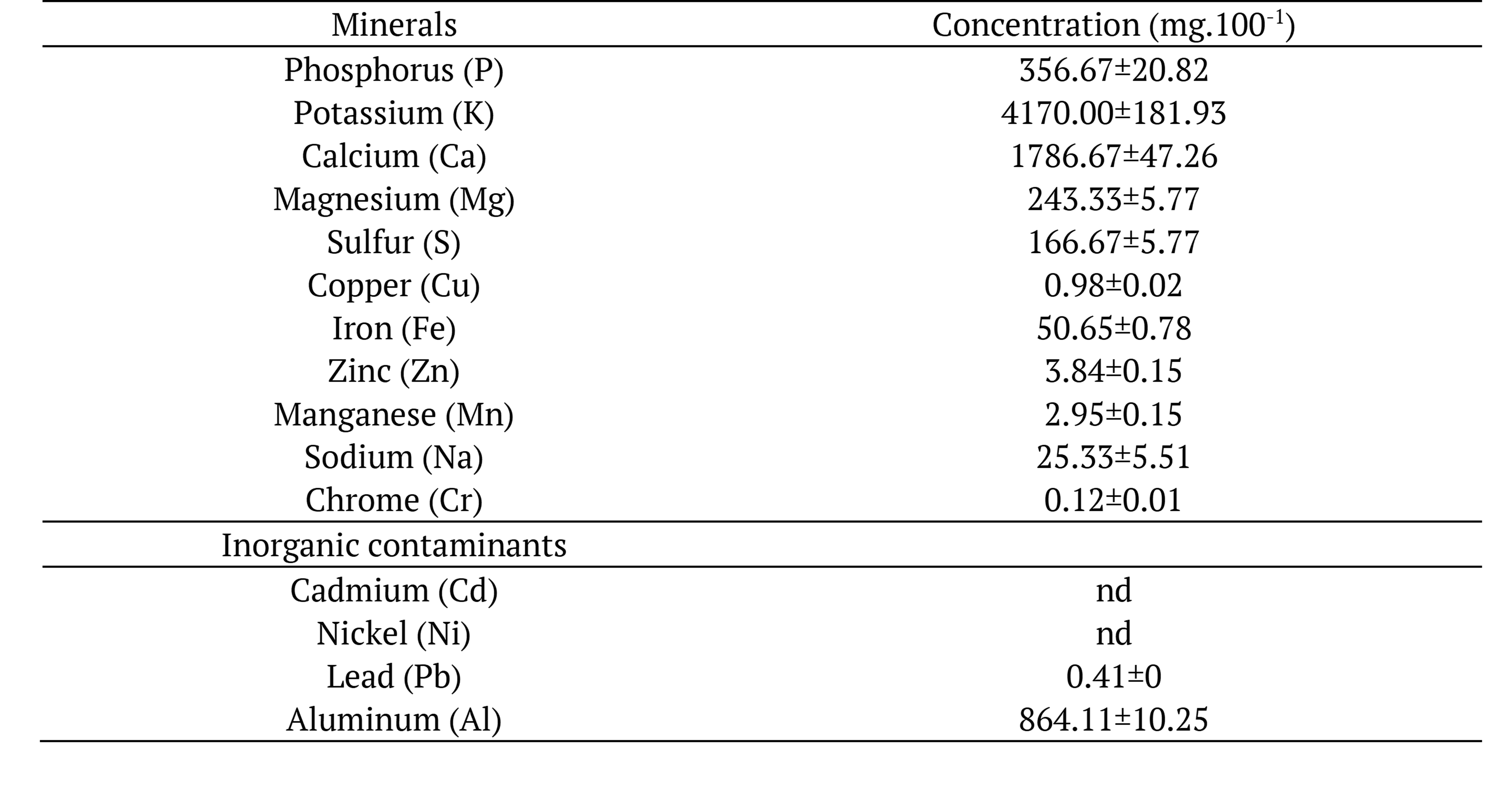
*Data expressed as dry matter; mean of 3 replicates ± standard deviation. nd = not detected
Flavonoids, total phenolic compounds, and antioxidant capacity of basil
Eriodictyol was higher in dehydrated herb when compared to fresh basil (Table 4). However, after dehydration, there was a loss of total flavonoids (20.70% retention), possibly caused by the heating process. According to Oliveira, Muzitano, Coutinho, Melo, and Costa (2011), temperature and storage time are important factors related to the preservation of flavonoids and functional properties during the shelf life of the food. Thus, these factors can lead to loss of flavones and flavanones due to the lower stability of these compounds and consequently low retention when subjected to heating process, which can explain the results found in our study.
Luteolin and naringenin were the most significant flavonoids found in fresh and dehydrated basil. The concentration of total flavonoids in fresh and dehydrated basil was much lower than that reported by Silva (2011) in hydroalcoholic extracts of fresh basil (Ocimum basilicum L.) (1,487.00 mg.100g-1) collected in Pato Branco, Brazil, and higher than that found by Alezandro, Lui, Lajolo, and Genovese (2011) in dried basil collected in São Paulo, Brazil (86.50 mg.100g-1). However, the comparison between different studies of total flavonoid concentration, as well as that of total carotenoids and total vitamin E is difficult because, in most cases, research does not investigate the presence of all compounds.

Data expressed in dry matter as mean of 3 replicates ± standard deviation. Means followed by the same letter in the rows, for each compound, do not differ statistically at 5% probability by Test t. RRA: radical removal activity. *Real Retention (% RR): Calculated according to the formula of Murphy, Criner, and Gray (1975) –
In hot and cold infusions, it was not possible to quantify flavonoids (Table 5), since only traces of them were found, making it impossible to identify and quantify these compounds. Thus, two factors may have occurred: drastic reduction in the concentration of flavonoids in infusions or extremely reduced transfer of flavonoids from fresh basil to infusions.
Flavonoids are commonly found in herbs and spices, acting as potent antioxidants, metal chelators and free radical scavengers (Jayasinghe, et al., 2003). They are beneficial compounds to health, highlighting their ability to reduce proliferation, differentiation and increase the apoptosis of cancer cells.
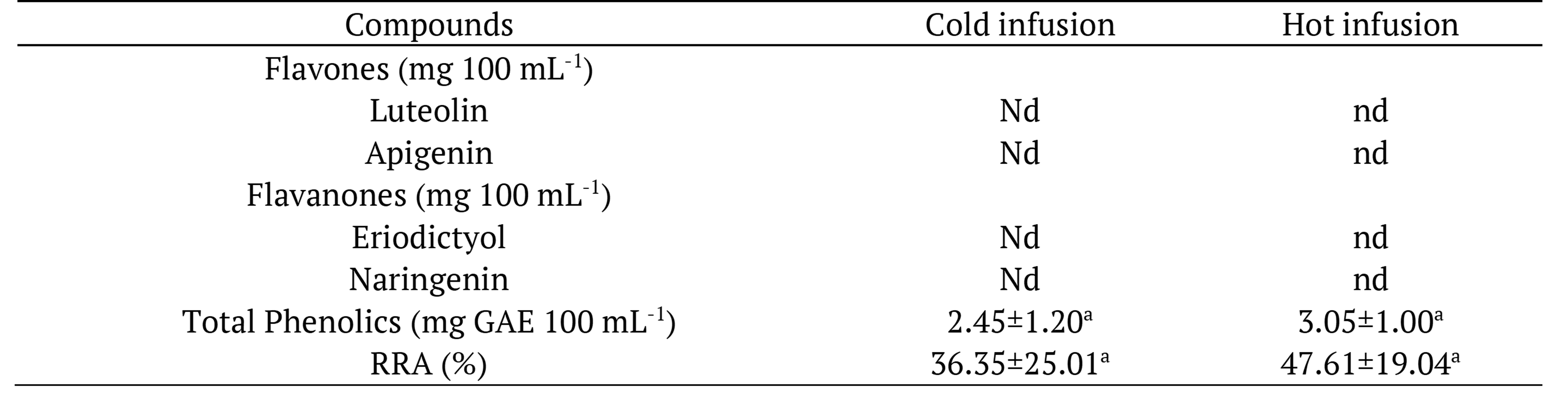
Data expressed in fresh matter as mean of 3 replicates ± standard deviation. Means followed by the same letter in the rows, for each compound, do not differ statistically at 5% probability by Test t. RRA: radical removal activity; nd: not determined.
Total phenolic concentrations in basil did not differ between treatments (p > 0.05). It was lower than those found by Prinsi et al. (2020) in basil (557 mg GAE 100 g-1) and similar to Alezandro et al. (2011) (52.50 mg GAE 100 g-1).
The antioxidant capacity did not differ between fresh and dehydrated basil (Table 4), neither between infusions (p > 0.05) (Table 5). Silva (2011) reported that basil presented 88.74%, a result higher than that found in our study, being able to be related to harvest season, soil characteristics and higher extraction during the process. Many herbs and spices stand out because of their antioxidant properties, such as basil, marjoram, rosemary, anise, chamomile, cinnamon, cardamom, cumin, clove, lemon balm (Hinneburg, Dorma, & Hiltunen, 2006; Jayasinghe, Gotoh, Aoki, & Wada, 2003).
There was no difference (p > 0.05) in relation to the concentration of phenolic compounds in hot and cold infusions (Table 5). According to Venditti et al. (2010), the contents of phenolic compounds are always higher in hot infusions. However, Rodrigues et al. (2015) assessed hot and cold infusions of different types of teas, and verified highest concentration in the cold infusions, probably associated with the thermosensitivity of phenolic compounds.
Conclusion
In synthesis, basil presented high moisture, low concentration of proteins, lipids and dietary fibers. There were no significant differences regarding the concentrations of vitamin C, vitamin E, phenolic compounds and antioxidant capacity between fresh and dehydrated basil and in hot and cold infusions. There was a higher concentration and retention of β-carotene and eriodictyol in the dehydrated herb. The concentrations of vitamins C and A in the infusions were very low. Thus, the consumption of basil, especially in dehydrated form, can contribute significantly to the daily intake of minerals, vitamins C and E, and infusions are not a relevant form of consumption to achieve nutritional recommendations.
Acknowledgements
The authors thank the Coordenação de Aperfeiçoamento de Pessoal de Ensino Superior (CAPES, Brazil), for financial support for conducting the study.
References
Aburigal, Y. A. A., Mirghani, M. E. S., Elmogtaba, E. Y., Sirible, A. A. M., Hamza, N. B., & Hussein, I. H. (2017). Total phenolic content and antioxidant capacity of basil (Ocimum basilicum L.) leaves from different locations. International Food Research Journal, 24, 378-381.
Alezandro, M. R., Lui, M. C. Y., Lajolo, F. M., & Genovese, M. I. (2011). Commercial spices and industrial ingredients: evaluation of antioxidant capacity and flavonoids content for functional foods development. Food Science and Technology, 31(2), 527-533. DOI: http://doi.org/10.1590/S0101-20612011000200038
Almeidai, D. M. d., Chaves, L. d. L., Del-Vechio-Vieira, G., Pinho, J. d. J. R. G. d., Yamamoto, C. H., & Sousa, O. V. d. (2010). Teores de óleo essencial e flavonóides totais em amostras de Rosmarinus officinalis L. Revista Brasileira de Farmácia, 91(4), 170-175.
Alvarez, V. V. H., Dias, L. E., Ribeiro Jr., E. S., & Fonseca, C. A. (2001). Métodos de análises de enxofre em solos e plantas. Viçosa, MG: UFV.
Association of Official Analytical Chemist [AOAC]. (2012). Official methods of analysis of AOAC international: Agricultural chemicals, contaminants, drugs. (16th ed.). Gaithersburg, MD: AOAC International.
Bertol, A., Almeida, S. M. Z., & Almeida, L. P. d. (2015). Determinação de minerais em algumas plantas medicinais utilizadas em Xanxerê – oeste catarinense. Unoesc & Ciência – ACBS, 6(1), 37-44.
Bloor, S. J. (2001). Overview of methods for analysis and identification of flavonoids. In P. Lester (Ed.), Methods in Enzymology (p. 3-14). Academic Press.
Braga, J. M., & Defelipo, B. V. (1974). Determinação espectrofotométrica do fósforo em extrato de solo e material vegetal. Revista Ceres, 21(113), 73-85.
Campos, F. M., Ribeiro, S. M. R., Della Lucia, C. M., Sant’Ana, H. M. P., & Stringheta, P. C. (2009). Optimization of methodology to analyze ascorbic and dehydroascorbic acid in vegetables. Química Nova, 32(1), 87-91. DOI: http://doi.org/10.1590/S0100-40422009000100017
Cardoso, L. d. M., Montini, T. A., Pinheiro, S. S., Queiroz, V. A. V., Sant'Ana, H. M. P., Martino, H. S. D., & Moreira, A. V. B. (2014). Effects of processing with dry heat and wet heat on the antioxidant profile of sorghum. Food Chemistry, 152, 210-217. DOI: http://doi.org/10.1016/j.foodchem.2013.11.106
Carvalho, A. C. B., Branco, P., Fernandes, L. A., Marques, R. F. d. O., Cunha, S. C., & Perfeito, J. P. S. (2012). Regulação brasileira em plantas medicinais e fitoterápicos. Revista Fitos, 7(1), 5-16.
Ch, M. A., Naz, S. B., Sharif, A., Akram, M., & Saeed, M. A. (2015). Biological and pharmacological properties of the sweet basil (Ocimum basilicum). British Journal of Pharmaceutical Research International, 7(5), 330-339. DOI: http://doi.org/10.9734/BJPR/2015/16505
Dumbrava, D.-G., Moldovan, C., Raba, D.-N., & Popa, M.-V. (2012). Vitamin C, chlorophylls, carotenoids and xanthophylls content in some basil (Ocimum basilicum L.) and rosemary (Rosmarinus officinalis L.) leaves extracts. Journal of Agroalimentary Processes and Technologies, 18(3), 253-258.
Dykes, L., Peterson, G. C., Rooney, W. L., & Rooney, L. W. (2011). Flavonoid composition of lemon-yellow sorghum genotypes. Food Chemistry, 128(1), 173-179. DOI: http://doi.org/10.1016/j.foodchem.2011.03.020
Eitenmiller, R. R. (1997). Vitamin E content of fats and oils – nutritional implication. Food Technology, 51(5), 78-81.
Ferreira, F. C., & Silveira Junior, V. (1999). Secagem a vácuo a diferentes pressões: avaliações físicas e sensoriais de manjericão. Revista Brasileira de Produtos Agroindustriais, 1(1), 59-65. DOI: http://doi.org/10.15871/1517-8595/rbpa.v1n1p59-65
He, X., Li, J., Zhao, W., Liu, R., Zhang, L., & Kong, X. (2015). Chemical fingerprint analysis for quality control and identification of Ziyang green tea by HPLC. Food Chemistry, 171, 405-411. DOI: http://doi.org/10.1016/j.foodchem.2014.09.026
Heinrichs, R., & Malavolta, E. (2001). Composição mineral do produto comercial da erva-mate (Ilex paraguariensis St. Hil.). Ciência Rural, 31(5), 781-785. DOI: http://doi.org/10.1590/S0103-84782001000500007
Hinneburg, I., Dorma, H. J. D., & Hiltunen, R. (2006). Antioxidant activities of extracts from selected culinary herbs and spices. Food Chemistry, 97(1), 122-129. DOI: http://doi.org/10.1016/j.foodchem.2005.03.028
Institute of Medicine. (2001). Dietary reference intakes for vitamin a, vitamin k, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium and zinc. Washington, D.C.: National Academy Press.
Jayasinghe, C., Gotoh, N., Aoki, T., & Wada, S. (2003). Phenolics composition and antioxidant activity of sweet basil (Ocimum basilicum L.). Journal of Agricultural and Food Chemistry, 51(15), 4442-4449. DOI: http://doi.org/10.1021/jf034269o
Kwee, E. M., & Niemeyer, D. E. (2011). Variations in phenolic composition and antioxidant properties among 15 basil (Ocimum basilicum L.) cultivars. Food Chemistry, 128(4), 1044-1050. DOI: http://doi.org/10.1016/j.foodchem.2011.04.011
Muráriková, A., & Neugebauerová, J. (2018). Seasonal variation of ascorbic acid and nitrate levels in selected basil (Ocimum basilicum L.) varieties. Horticultural Science, 45(1), 47-52. DOI: http://doi.org/10.17221/216/2016-HORTSCI
Natesan, S., & Ranganathan, V. (1990). Content of various elements in different parts of the tea plant and in infusions of black tea from southern India. Journal of the Science of Food and Agriculture, 51, 125-139. DOI: http://doi.org/10.1002/jsfa.2740510112
Oliveira, L. S. d., Muzitano, M. F., Coutinho, M. A. S., Melo, G. d., & Costa, S. S. (2011). Plantas medicinais como recurso terapêutico em comunidade do entorno da reserva biológica do tinguá, RJ, Brasil–metabólitos secundários e aspectos farmacológicos. InterScience Place, 1(17).
Pedro, F. G. G., Arruda, G. L., Oliveira, J. C., Santos, A. D., Sigarini, K. S., Hernandes, T., ... Oliveira, A. P. (2016). Composição centesimal e mineral de plantas medicinais comercializadas no mercado do Porto de Cuiabá, Mato Grosso, Brasil. Revista Brasileira de Plantas Medicinais, 18(1), 297-306. DOI: http://doi.org/10.1590/1983-084X/15_144
Sant'ana, H. M. P., Guinazi, M., Oliveira, D. d. S., Della Lucia, C. M., Reis, B. d. L., & Brandão, S. C. C. (2011). Method for simultaneous analysis of eight vitamin E isomers in various foods by high performance liquid chromatography and fluorescence detection. Journal of Chromatography A, 1218(47), 8496-8502. DOI: http://doi.org/10.1016/j.chroma.2011.09.067
Sant’Ana, H. M. P., Stringheta, P. C., Brandão, S. C. C., Páez, H. H., & Queiroz, V. M. V. d. (1998). Evaluation of total carotenoids, alpha- and beta-carotene in carrots (Daucus carota L.) during home processing. Food Science and Technology, 18(1), 39-44. DOI: http://doi.org/10.1590/S0101-20611998000100009
Prinsi, B., Morgutti, S., Negrini, N., Faoro, F., & Espen, L. (2020). Insight into composition of bioactive phenolic compounds in leaves and flowers of green and purple Basil. Plants, 9(1), 22. DOI: http://doi.org/10.3390/plants9010022
Reis, R. C. d., Devilla, I. A., Ascheri, D. P. R., Servulo, A. C. O., & Souza, A. B. M. (2012). Cinética de secagem de folhas de manjericão (Ocimum basilicum L.) via infravermelho. Revista Brasileira de Engenharia Agrícola Ambiental, 16(12), 1346-1352. DOI: http://doi.org/10.1590/S1415-43662012001200012
Rodrigues, V. d. C., Silva, M. V. d., Santos, A. R. d., Zielinski, A. A. F., & Haminiuk, C. W. I. (2015). Evaluation of hot and cold extraction of bioactive compounds in teas. International Journal of Food Science and Technology, 50(9), 2038–2045. DOI: http://doi.org/10.1111/ijfs.12858
Rodriguez-Amaya, D. B. (2001). A guide to carotenoid analysis in foods. Washington, DC: ILSI.
Sarruge, J. R., & Haag, H. P. (1974). Análise química em plantas. Piracicaba, SP: Esalq.
Sousa, M. S., & Pereira, M. C. d. A. (2013). Mecanismos moleculares de ação anti-inflamatória e antioxidante de polifenóis de uvas e vinho tinto na aterosclerose. Revista Brasileira de Plantas Medicinais, 15(4), 617-626. DOI: http://doi.org/10.1590/S1516-05722013000400020
Silva, M. G. F. d. (2011). Atividade antioxidante e antimicrobiana in vitro de óleos essenciais e extratos hidroalcóolicos de manjerona (Origanum majorana L.) e manjericão (Ocimum basilicum L.). (Graduate, Universidade Tecnológica Federal do Paraná, Pato Branco). Recovered from http://repositorio.roca.utfpr.edu.br/jspui/handle/1/440
Singleton, V. L., Orthofer, R., & Ranventós, R. M. L. (1999). Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteau reagent. Methods of Enzymology, 299, 152-178. DOI: http://doi.org/10.1016/S0076-6879(99)99017-1
Soliman, K., & Zikovsky, L. (1999). Concentrations of al in food sold in Montreal, Canada, and its daily dietary intake. Journal of Radioanalytical and Nuclear Chemistry, 242(3), 807-809. DOI: http://doi.org/10.1007/BF02347400
Núcleo de Estudos e Pesquisas em Alimentação [NEPA]. (2011). Tabela Brasileira de Composição de Alimentos [TACO]. Campinas, SP: NEPA.
Venditti, E., Bacchetti, T., Tiano, L., Carloni, P., Greci, L. & Damiani, E. (2010). Hot vs. cold water steeping of different teas: do they affect antioxidant activity? Food Chemistry, 119(4), 1597–1604. DOI: http://doi.org/10.1016/j.foodchem.2009.09.049
Yang, L., Allred, K. F., Geera, B., Allred, C. D., & Awika, J. M. (2012). Sorghum phenolics demonstrate estrogenic action and induce apoptosis in nonmalignant colonocytes. Nutrition and Cancer, 64(3), 419-427. DOI: http://doi.org/10.1080/01635581.2012.657333
Zielinski, A. A. F., Haminiuk, C. W. I., Alberti, A., Nogueira, A., Demiate, I. M., & Granato, D. (2014). A comparative study of the phenolic compounds and the in vitro antioxidant activity of different Brazilian teas using multivariate statistical techniques. Food Research International, 60, 246-254. DOI: http://doi.org/10.1016/j.foodres.2013.09.010
Notas de autor
nutripamella@gmail.com