Farmácia
Evaluation of nutraceutical product labelings which contain omega-3 fatty acid in drugstores networks located in Cascavel-PR
Avaliação da rotulagem de produtos nutracêuticos contendo ômega-3 em redes de farmácias de Cascavel-PR
Evaluation of nutraceutical product labelings which contain omega-3 fatty acid in drugstores networks located in Cascavel-PR
Acta Scientiarum. Health Sciences, vol. 40, 2018
Universidade Estadual de Maringá
Received: 02 May 2017
Accepted: 25 October 2017
Abstract: Omega-3 is a polyunsaturated fat considered essential for the body. This compound is SOLD as a nutraceutical in Brazil, being widely consumed by the population due to its functional properties. According to its health benefits, this paper aimed to investigate the labeling of nutraceutical products sold in drugstores in the city of Cascavel, Paraná state - Brazil and verify its compliance with current legislation. The research was carried out in drugstore networks located in Cascavel, Paraná state, in 09 different establishments, from May to June 2016. Nutraceutical products containing omega-3 in their formulations were identified at the sites visited. Their images were captured and proceeded with the research analyzing these labels according to the Legislation in force for products of functional and or health claim. We analyzed 13 labels, only 38.46% (five labels) presented no irregularities. Therefore, it was concluded that the laws in Brazil are not being followed by the companies responsible for manufacturing these products and the lack of mandatory information on their labels can put the consumer´s health at risk, being that it must be guaranteed the consumption safety of adequate and quality products.
Keywords: polyunsaturated fatty acid, legislation, Anvisa, labels.
Resumo: O Ômega-3 é uma gordura poli-insaturada considerada essencial ao organismo. No Brasil este composto é comercializado como nutracêutico, sendo amplamente consumido pela população em virtude das suas propriedades funcionais. Tendo em vista o aumento deste consumo, em função dos seus benefícios à saúde, este trabalho teve como objetivo pesquisar a rotulagem de produtos nutracêuticos comercializados em farmácias da cidade de Cascavel/PR - Brasil e verificar a sua conformidade com a Legislação vigente. A pesquisa foi realizada em redes de Farmácias de Cascavel/PR, sendo 09 estabelecimentos diferentes, no período de maio a junho de 2016. Identificaram-se, nos locais visitados, produtos nutracêuticos que continham Ômega-3 em suas formulações. Realizou-se captura de imagens dos mesmos e prosseguiu-se com a pesquisa analisando estes rótulos de acordo com a Legislação vigente para produtos de alegação funcional e ou de saúde. Foram analisados 13 rótulos, apenas 38,46% (cinco rótulos) não apresentaram nenhuma irregularidade. Portanto, concluiu-se que as legislações vigentes no Brasil não vêm sendo seguidas pelas empresas responsáveis pela fabricação desses produtos e a falta de informações obrigatórias em seus rótulos pode colocar a saúde do consumidor em risco, sendo que a ele deve ser garantida a segurança do consumo de produtos adequados e de qualidade.
Palavras-chave: ácido graxo poli-insaturado, legislação sanitária, Anvisa, rótulos.
Introduction
Omega-3 is a polyunsaturated fat considered essential for the organism, has a long carbon chain with two or more bonds, the last double bond at the third carbon from the end of the molecule, that is the methyl end carbon. The family representatives are alpha linolenic acid (ALA) (18: 3n-3) of plant origin, eicosapentaenoic acid (EPA) (22: 6n-3) and docosahexaenoic acid (DHA) (22: 6n-3). They are considered essential fatty acids, since they are essential for the organism, and cannot be synthesized by it, therefore, they must be offered in the diet (Martin et al., 2006; Carmo & Correia, 2009; Kayser, Kresky, Oliveira, Liberali, & Coutinho, 2010).
Lipid components, especially fatty acids, play important roles in the structure of cell membranes and in metabolic processes (Martin et al., 2006). Several studies have shown that the consumption of dietary fiber and monounsaturated and polyunsaturated fats such as omega-3 in adequate amounts contributes to the prevention of hypertension because it is related to the decrease in total cholesterol, triglycerides and, consequently, increased levels of high density lipoprotein (HDL) (Kayser et al., 2010; Pioltine, Paternez, & Morimoto, 2013).
Omega-3 can be obtained from fish, especially those from deep and cold waters. It is important to note that not all fish contain the same amounts of Omega-3 due to the feeding diversity of each species. The fish that contains the largest quantity of it are mackerel, sardines, salmon and trout, as well as phytoplankton, which form the basis of the ocean food chain (Kris-Etherton, Harris, & Appel, 2002; Andrade & Carmo, 2006; Scherr, Gagliardi, Miname, & Santos, 2015).
Although vegetables have small amounts of alpha-linolenic acid because of their low lipid content, vegetable consumption can contribute to increase the intake of linoleic and alpha-linolenic acids, especially in vegetarian diets, including watercress, cabbage, lettuce, spinach and broccoli (Martin et al., 2006).
The interest in studying omega-3 began in the 1960s from the low occurrence of cardiovascular diseases in Eskimos, probably related to the fish-rich diet (Kayser et al., 2010). According to the study conducted by Scherr et al. (2015), fat concentrations of some fish frequently consumed in Brazil were analyzed. The analyzes were performed after cooking and showed important variations among the species. The results obtained by the researchers were that all the fish that were part of the study had low levels of saturated fat, however, the majority contained little amount of omega-3 fatty acids. Among the fish that were studied, merluchons had the highest Omega-3 tissue concentrations, being a good source, especially if consumed in the amount of at least 222 g day-1, followed by salmon, with a consumption of 253 g day-1 to reach the recommendations of 2,000 mg day-1 of Omega-3 for prevention of cardiovascular disease. The study concluded that all studied fish of Brazilian species had low concentrations of Omega-3. This fact may be related to the type of food consumed by fish, which is incorporated into their adipose tissue.
National and international institutions such as the American Heart Association (AHA), the Brazilian Society of Cardiology (SBC) and the Ministry of Fisheries and Aquaculture (MPA) recommend ingestion of portions ranging from 0.075 to 0.112 kg of fish rich in these fatty acids at least two per week, but estimates indicate a consumption of 8.3 to 10.0 kg / person per year in Brazil, less than the recommended amounts, which is 12 kg / person per year. These estimates regarding consumption are due to numerous factors such as high prices and poor quality of fish triggered by problems such as: deficiencies in handling fish in natura commercialization, conservation and storage. Because it is a highly perishable food, special care is needed, from capture to consumption by humans (Oliveira, 2013).
The product legislation of functional or health claims highlights some of the requirements for labels with criteria for food rich in Omega-3. These claims are established with a view to stimulating the reformulation and development of more nutrient-friendly industrialized products and the use of clarifications and warnings related to their use in a visible and readable form on packaging (Brasil, 2016).
In view of the increased omega-3 intake because of its benefits to human health, this study aimed to investigate the labeling of nutraceutical products sold in drugstores located in the city of Cascavel, Paraná state - Brazil and verify compliance with the current legislation.
Material and methods
This research was carried out in drugstore networks located in the city of Cascavel, Paraná state, where the choice was made randomly by the representativeness of these networks and which are pole of the others, in 09 establishments, from May to June of 2016. The places of business received a letter of presentation used as an authorization document to be applied to the survey.
During the visitation of the place of businesses, the nutraceutical products containing omega-3s in their formulations were identified and, in order to enable the analysis of the labels, images of them were captured by means of photographs.
Subsequently, the study was directed to the evaluation of a questionnaire answered and interpreted by the authors of this study. The product brand and its pharmaceutical form were evaluated and from the answers the information was correlated with the required standards, which were obtained with the online search on the ANVISA website about the resolutions for labeling products containing Omega-3. Due to the lack of specific standards for nutraceuticals and the fact that it presents an approved claim regarding its functional property, other nonspecific standards related to food became necessary.
For the interpretation and analysis of the labeling, the following resolutions were used as basis: RDC n. 2 (Brasil, 2002a), RDC n. 259 (Brasil, 2002b), RDC n. 360 (Brasil, 2003a), RDC n. 278 (Brasil, 2005), RDC n. 26 (Brasil, 2015), and Law n. 10,674 (Brasil, 2003b) and the IX List based the claim of functional property (Brasil, 2008) of Omega-3, as well as the specific requirements related to its presence.
The data obtained in the research were qualitatively evaluated and expressed in frequency charts.
Results and Discussion
A total of 22 gelatin capsule formulations containing Omega-3 were found in the 9 establishments surveyed. It was verified that some brands were repeated, resulting on the exclusion of 09 formulations, remaining 13 for analysis.
Figure 1 shows formulations with regularities and irregularities found in the products that were analyzed.
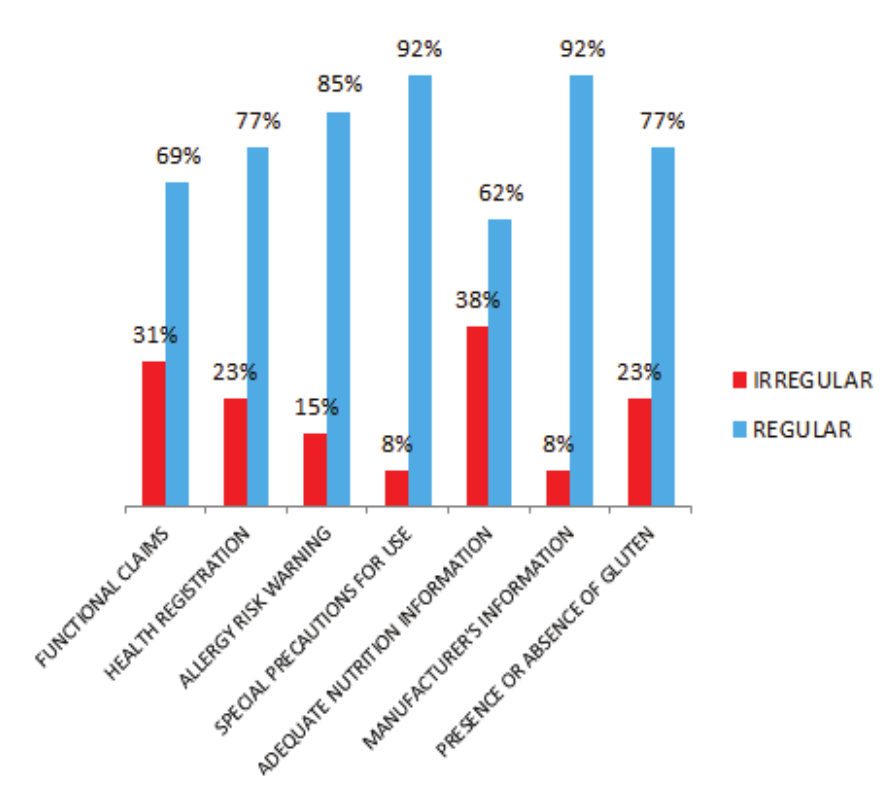
Figure 1.
Regularities and irregularities in relation to the evaluated items in 13 products containing Omega-3 in their formulations, marketed in drugstores located in the city of Cascavel, Paraná.
In relation to the above-mentioned aspects, an important obligatory characteristic of Omega-3-containing products in relation to functional claims would be the presence, on the label, of the phrase: ‘The consumption of fatty acids assists in the healthy levels of triglycerides, combined with a balanced diet and healthy lifestyle habits’. It was verified that 30.76% (four) of the products did not have this information on their labels. Considering that such products are on the market, it is very important to inform the consumer that their functional activity in the isolated form will not have the same effects as when used in association with a balanced diet and healthy lifestyle.
Regarding the Sanitary Register, the RDC n. 278 (Brasil, 2005) establishes that bioactive substances with a claim of functional and / or health properties must have it. The IX List of functional property claims (Brasil, 2008) mentions that such a registry may be granted by the National Health Surveillance Agency (ANVISA) or by the Ministry of Agriculture, Livestock and Food Supply (MAPA). In the labeling of the products analyzed it was observed that 23.08% (three products) did not have any information or registration number of any of the aforementioned agencies. This irregularity is worrying, since the products are being freely commercialized and consumed and without the consent of the regulating bodies, therefore, without any control of quality and suitability of their manufacturers, resulting in products without the necessary quality guarantee. The lack of inspection in the manufacturing process can cause health problems to population due to the origin uncertainty of the materials used and the manufacturing process, as well as the use of questionable raw materials and possible contaminations such as: microbiological, cross contamination, among others.
The presence of the Nutritional Table is mandatory according to the RDC n. 360 (Brasil, 2003), which indicates the energy value related to the composition, amount of lipids and other nutrients, when appropriate. Regarding the type and quantity of fats, fatty acids or cholesterol that are present, the amount of all lipid fractions should be indicated. Based on this, it was observed that 38.46% (five products) present incomplete nutritional information as shown in the following Figure 2.
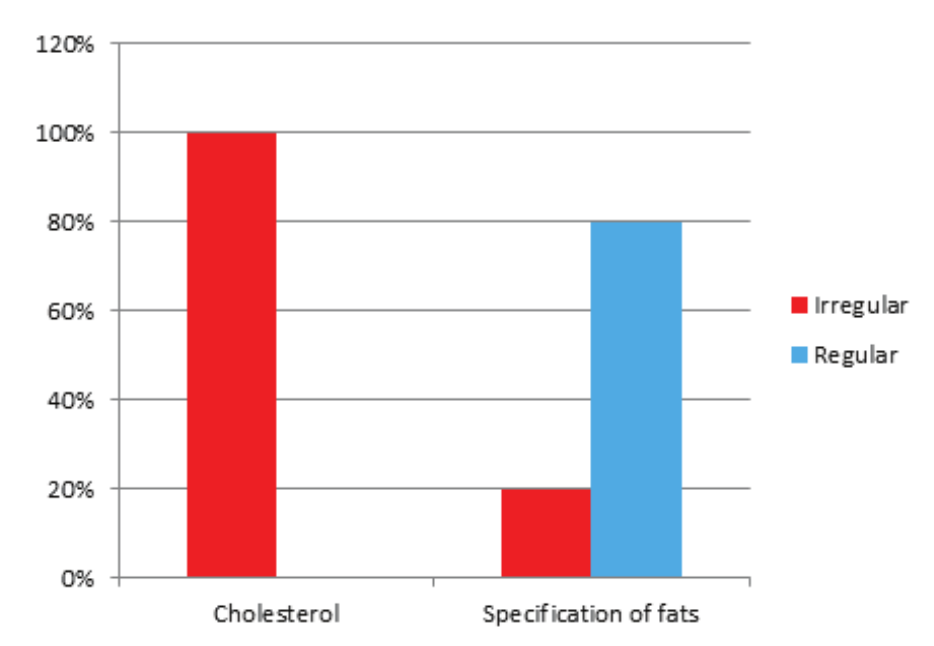
Figure 2.
Regularities and irregularities in relation to the nutritional table evaluated in 13 products containing Omega-3 in their formulations, marketed in drugstores located in the city of Cascavel, Paraná.
It was observed that the absence of the specification in relation to the amount of cholesterol occurred in all the irregular products and 20% of them did not specify the four types of fat, according to legislation mentioned previously, this implies in the uncertainty of the presence of Omega-3, noting that this is a polyunsaturated fatty acid and its amount must be expressed in the Nutritional Table guaranteeing to the consumer its presence in the content of the capsules, since the reason for the consumption of this nutraceutical is the supplementation of Omega-3 due to the low consumption of fish.
In relation to the minimum amounts of EPA and DHA required for the functional claim, as described in Brazil, 2008, it was observed that all products have presented themselves accordingly. However, there was a recent update referred to the addition of the following information: “ANVISA reevaluated scientific evidence on the functional properties of fatty acids, EPA and DHA type, from food and supplement intake. It was concluded that the minimum amounts of 100 mg requiered of EPA and DHA are not sufficient to produce the beneficial effects related to triglyceride levels” (Brasil, 2016). Thus, since then, companies must perform studies to prove the effectiveness of fatty acids at the time of registration of the product, making it unnecessary to inform the amount of EPA and DHA on their labels. Therefore, after this note, it is expected that all companies will adjust, making a compliance assessment unfeasible at this moment.
Thinking about the safety use of the products, it is of paramount importance the presence of usage information, clarifying its restrictions and related warnings. ANVISA recommends the presence of the following warning highlighted in bold: "People suffering from illness or physiological changes, pregnant or lactating women should consult their physician before using the product" (Brasil, 2016). In the evaluation performed, 7.69% (one product) did not show the warning, which shows a risk for these groups of consumers, because without the information on the product label, they may use them and trigger complications for their health.
In order to ensure the origin of the products, the Resolution RDC n. 259 (Brasil, 2002b) establishes that the origin identification has mandatory presentation, where it must be included: manufacturer's name, full address, country and municipality origin, as well as registration number or identification code of the establishment. However, in the labels analyzed, 7.69% (one product) did not show this information, a relatively serious fact, since a product available on the market for consumption without any information of its origin generates at least doubt as to its quality, origin, manufacturing process and several other important points.
In one of the products, the type of packaging made it impossible to verify the information of the manufacturer and the line manager, due to the way in which the product was exposed for sale.
In recent years, the increase in allergy rates in the population has attracted considerable attention and has been widely discussed. Great efforts were made so that the population had greater access to information on the packaging or food labels present on the market. With this purpose, the Law n. 10.674 (Brasil, 2013) was created, which obliges all industrialized food products to report the presence or absence of gluten. 23.08% (three of them) of the products analyzed did not show in their labels information about the presence or absence of gluten. Considering the importance of this information, it can be emphasized that the absence of information about gluten will influence the sale of the product, since the consumer who cannot ingest gluten will not buy a product that does not provide information on the presence or absence of the substance, and its consumption is risky.
In the same context, the RDC n. 26 (Brasil, 2015) specifies that all products containing ingredients, food additives and food supplements which cause allergies should contain statements warning their presence. Considering that Omega-3 comes from fish and that some fish allergens can be linked together with their extracts, it is necessary to state this risk on the labels of the products. The research has shown that 15.38% (two) of the products analyzed did not have this warning. In relation to risk coming from fish, the consumption of these products by allergic individuals can trigger an anaphylactic reaction, and may even lead to death depending on the degree of reaction.
The following Figure 3 shows the frequency of irregularities observed in the survey.
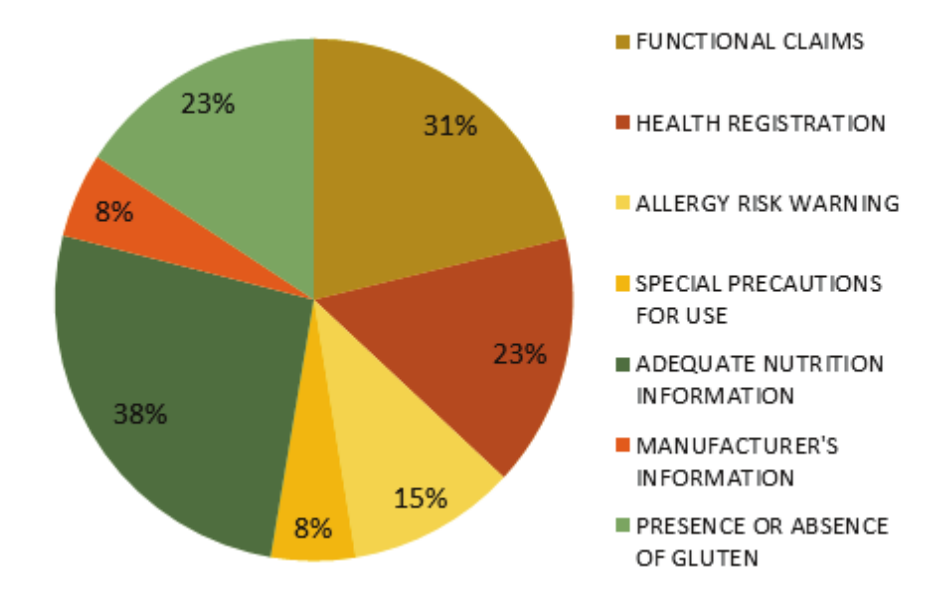
Figure 3.
Frequency of irregularities observed in the 13 products containing Omega-3 in its formulations, marketed in drugstores located in the city of Cascavel, Paraná state.
Figure 3 shows the frequency that irregularities appeared, where the items with the highest number of nonconformities were the nutritional information and the functional claim of the products, respectively. Since they are nutraceuticals consumed with the objective of helping to maintain health, due to their functional properties, it is of the utmost importance that these products are of good quality and that they possess all the information regarding their composition and use, guaranteeing thus a safe and conscious consumption, sin0ce they are over-the-counter products, and often the population makes use of them without indication or guidance of a physician or pharmacist.
The frequency of conformities and nonconformities were observed considering the total number of occurrences in the survey, evidencing that most of the products had at least one irregularity in the samples analyzed and only 38.46% (five products) were in accordance with the specifications required by current legislation. Considering this result, there is evidence of a lack of inspection before regulatory agencies, both in the approval of the product sale and the after-sale, allowing companies to market their products in a ‘free’ way without any guarantee to the consumer regarding the provenance and quality. On the other hand, the consumer relies on products on the market and consumes these products in order to help maintain health and ends up being ‘cheated’ because the desired effect does not appear.
Conclusion
Research in the current legislation indicates a lack of specific standards for nutraceuticals, making it necessary to use existing laws and resolutions for packaged foods.
The assessment of the labeling of nutraceuticals marketed in the pharmacies surveyed in the city of Cascavel / PR - Brazil showed a large number of irregularities that generally were greater than regularities, considering the set of evaluated items.
The lack of mandatory information on product labels can put the health of the consumer at risk. This way, it’s necessary for the inspection and registration agencies to act together requiring the manufacturers to correct them.
References
Andrade P. M. M., & Carmo M. G. T. (2006). Ácidos graxos n-3: um link entre eicosanoides, inflamação e imunidade. MN-Metabólica, 8(3), 135-143.
Brasil. Ministério da Saúde. Agencia Nacional de Vigilância Sanitária. (1999). Resolução n. 18, de 30 de abril de 1999. Aprova o Regulamento Técnico que estabelece as diretrizes básicas para análise e comprovação de propriedades funcionais e ou de saúde alegadas em rotulagem de alimentos, constante do anexo desta portaria. Retrieved from http://www.saude.rj. gov.br/comum/code/MostrarArquivo.php?C=MjI0OQ%2C%2C.
Brasil. Ministério da Saúde. Agencia Nacional de Vigilância Sanitária. (2002a). Resolução RDC n. 2, de 7 de janeiro de 2002. Aprova o regulamento técnico de substâncias bioativas e probióticos isolados com alegação de propriedades funcional e ou de saúde. Retrieved from http://www.ibama.gov.br/sophia/cnia/legislacao/AGENCIAS/ANVISA/RS0002-070102. PDF.
Brasil. Ministério da Saúde. Agencia Nacional de Vigilância Sanitária. (2002b). Resolução RDC n. 259, de 20 de setembro de 2002. Aprova o regulamento técnico sobre rotulagem de alimentos embalados. Retrieved from https://lcqa.farmacia.ufg.br/up/912/o/resoluo_ rdc_n_259_2002_-_rotulagem_em_geral.pdf.
Brasil. Ministério da Saúde. Agencia Nacional de Vigilância Sanitária. (2003a). Resolução RDC n. 360 de 23 de dezembro de 2003. Regulamento técnico sobre rotulagem nutricional de alimentos embalados, tornando obrigatória a rotulagem nutricional. Retrieved from http://www.economia-snci.gob.mx/ politicacomercial/Archivos/Brasil%20resoluci%C3%B3n%20360-2013.pdf.
Brasil. Casa Civil. (2003b). Lei n. 10.674, de 16 de Maio de 2003. Obriga que os produtos alimentícios comercializados informem sobre a presença de glúten como medida preventiva e de controle da doença celíaca. Retrieved from https://natal.rn.gov.br/sms/ covisa/nucleos/alimentos/440852.pdf.
Brasil. Ministério da Saúde. Agencia Nacional de Vigilância Sanitária. (2005). Resolução RDC n. 278, de 22 de setembro 2005. Estabelece as categorias de alimentos e embalagens dispensados e com obrigatoriedade de registro. Retrieved from http://www.saude.rj.gov.br/comum/code/Mostrar Arquivo.php?C=MjI1Mg%2C%2C.
Brasil. Ministério da Saúde. Agência Nacional de Vigilância Sanitária. (2008). Alimentos com alegações de funcionais e ou de saúde, novos alimentos/ingredientes, substâncias bioativas e probióticos -IX - Lista de alegações de propriedade funcional aprovadas, em julho de 2008. Retrieved from http://www.anvisa.gov.br/alimentos/ comissoes/tecno_lista_alega.htm.
Brasil. Ministério da Saúde. Agencia Nacional de Vigilância Sanitária. (2015). Resolução RDC n. 26, de 02 de julho de 2015. Dispõe os requisitos para rotulagem obrigatória dos principais alimentos que causam alergias alimentares. Retrieved from http://adcon.rn.gov.br/ACERVO/ Suvisa/doc/DOC000000000083199.PDF.
Brasil. Ministério da Saúde. Agência Nacional de Vigilância Sanitária. (2016). Alimentos com alegações de propriedades funcionais e ou de saúde, novos alimentos/ingredientes, substâncias bioativas e probióticos -IX - Lista de alegações de propriedade funcional aprovadas, em 22 dezembro de 2016. Retrieved from http://portal. anvisa.gov.br/alimentos/alegacoes.
Carmo, M. C. N. S., & Correia, M. I. T. D. (2009). A importância dos ácidos graxos ômega-3 no câncer. Revista Brasileira de Cancerologia, 55(3), 279-287.
Kayser, C. G. R., Kresky, H. L., Oliveira, R. M., Liberali, R. & Coutinho, V. (2010). Benefícios da ingestão de ômega 3 e a prevenção de doença crônica degenerativas - Revisão Sistêmica. Revista Brasileira de obesidade, Nutrição e Emagrecimento, 4(21), 137-146.
Martin, C. A., Almeida, V. V., Ruiz, M. R., Visentainer, J. E. L., Matshushita, M., Souza, N. E., & Visentainer, J. V. (2006). Ácidos graxos poliinsaturados ômega-3 e ômega-6: importância e ocorrência em alimentos. Revista de Nutrição, 19(6), 761-770. Doi: 10.1590/S1415-52732006000600011.
Oliveira, M. J. (2013). O peixe é saúde: das recomendações para o consumo às possibilidades ambientais de atende-lo. Segurança Alimentar e Nutricional, 20(1), 141-146. Doi: 10.20396/san. v20i1supl.8634592.
Kris-Etherton, P. M. E., Harris, W. S., & Appel, L. J. (2002). Fish consumption, fish oil, ômega-3 fatty acids and cardiovascular disease. American Heart Association, Nutrition Committee, 106(21), 2747-2757. PMID: 12438303.
Pioltine, B. M., Paternez, C. A. A. C., & Marimoto, M. J. (2013). Perfil dietético e risco cardiovascular em adolescentes de uma escola privada em São Paulo. Revista Cientifica & Saúde, 6(3), 149-156. Doi: 10.15448/1983-652X.2013.3.
Scherr, C., Gagliardi, A. C. M., Miname, M. H., & Santos, R. D. (2015). Fatty acid and cholesterol concentrations in usually consumed fish in Brazil. Arquivos Brasileiros de Cardiologia, 104(2), 152-158. Doi: 10.5935/ abc.20140176.
Notes
Author notes
luciana.farina@unioeste.br