Educação Física
Influence of β-Hydroxy-β-methylbutyrate on predominantly white and red muscles of rats not submitted to physical exercise
Influência do β-hidroxi-β-metilbutirato em músculos predominantemente branco e vermelho de ratos não submetidos ao exercício físico
Influence of β-Hydroxy-β-methylbutyrate on predominantly white and red muscles of rats not submitted to physical exercise
Acta Scientiarum. Health Sciences, vol. 40, 2018
Universidade Estadual de Maringá
Received: 18 July 2017
Accepted: 28 April 2018
Abstract: This study evaluated the influence of β-hydroxy-β-methylbutyrate (HMB) on the body weight and morphometry of white and red muscle fibers of sedentary rats. 29 male rats were divided in: initial control group (ICG ) (n = 5), euthanized at study onset, and three treated groups (n = 8 each): placebo control group (PCG) received saline for 4 weeks; experimental group (EG) received 0.3 g kg-1 of HMB daily and controlled diet; and experimental group ad libitum, (EGA) received 0.3g kg-1 of HMB daily, without food restriction. There was no difference in food intake and body weight between treated groups. For the EDL muscle, treated groups were different from ICG. Concerning the soleus muscle, no treated group was different from each other and only the EGA presented difference compared to the ICG. Data also did not reveal correlation between the variables food intake and body weight. HMB did not change the body weight or morphometry of EDL muscle fibers. However, the HMB combined with ad libitum feeding was able to change the morphometry of soleus muscle fibers.
Keywords: leucine, supplementation, skeletal muscle, extensor digitorum longus, soleus.
Resumo: Este estudo avaliou a influência do β-hidroxi-β-metilbutirato (HMB) no peso corporal e na morfometria de fibras brancas e vermelhas dos músculos extensor digital longo e sóleo, respectivamente de ratos sedentários. 29 ratos machos divididos em: grupo de controle inicial (ICG) (n = 5), sem tratamento algum, eutanasiados no início do estudo; e grupos tratados (n = 8 cada): o grupo controle placebo (PCG) recebeu solução salina por 4 semanas sem restrição alimentar; o grupo experimental (EG) recebeu 0,3 g kg-1 de HMB diariamente e dieta controlada; e grupo experimental ad libitum, (EGA) recebeu 0,3 g kg-1 de HMB diariamente, sem restrição alimentar. Não houve diferença na ingestão alimentar e peso corporal entre grupos tratados. No músculo extensor digital longo, os grupos tratados foram diferentes do ICG. No músculo sóleo, nenhum dos grupos tratados foi diferente entre si e somente EGA apresentou diferença em relação ao ICG. Também não houve correlação entre as variáveis: ingestão alimentar e peso corporal. O HMB não alterou o peso corporal ou morfometria das fibras do músculo EDL. Contudo, HMB combinado com alimentação ad libitum foi capaz de alterar a morfometria das fibras brancas e vermelhas do músculo sóleo.
Palavras-chave: leucina, suplementação, músculo esquelético, extensor digitorum longus, soleus.
Introduction
Leucine is an important branched-chain amino acid (BCAA) that plays a fundamental role in muscle metabolism, performing a regulatory function in both the synthesis and inhibition of protein degradation (Baxter, Carlos, Thurmond, Rehani, Bultman, & Frost, 2005). Some of these effects are partially assigned to its metabolite β-hydroxy-β-methylbutyrate (HMB) (Zanchi et al., 2010; Gerlinger-Romero, Guimarães-Ferreira, Giannocco, & Nunes, 2011).
The anti-catabolic effects of HMB on the skeletal muscle have been reported, reducing the protein degradation or increasing the rates of protein synthesis (Zanchi et al. 2010; Gerlinger-Romero et al., 2011; Pinheiro et al., 2012; Hasselgreen, 2014). These beneficial effects have been used in different manners, including muscle mass gain, when associated with physical exercise (Di Luigi, 2008; Portal, 2011); or reduction of muscle loss, in cases of sepsis (Kovarik, Muthny, Sispera, & Holecek, 2010; Supinski & Callahan, 2014); cancer-induced cachexia (Smith, Wyke, & Tisdale, 2004; Smith, Mukerji, & Tisdale, 2005; Eley, Russell, Baxter, Mukerji, & Tisdale, 2007; Eley, Russell & Tisdale, 2008a, 2008b, Sonni et al., 2008; Aversa et al., 2011); sarcopenia (Flakoll et al., 2004; Fitschen, Wilson, Wilson, & Wilund 2013; Alway, Pereira, Edens, Hao, & Bennetti, 2013); and AIDS (Fitschen et al., 2013), in which the effects of HMB were positive. However, not all studies have found beneficial effects of HMB supplementation (Wu et al., 2015).
The utilization of HMB presents further advantages as the lack of reports of adverse effects when up to 5% of the daily diet is taken (Baxter et al. 2005), as well as its classification as legal supplement by the Olympic Committee (Alvares & Meirelles, 2008).
Many athletes are currently searching for dietary supplements to achieve better outcomes in their sports activities, yet other people search for such substances aiming at fat loss or body esthetics. However, little is known about their effects in individuals who do not exercise, and morphometric data on this aspect are still scarce.
Considering these aspects, this study evaluated the influence of HMB on the body weight and morphometry of fibers on white (Extensor Digitorum Longus - EDL) and red (Soleus) muscles in rats not submitted to physical exercise.
Material and methods
Division of groups and animal care
The study was conducted on 29 young Wistar rats at 60 days post birth. The animals were divided in the following groups: initial control group (ICG) n = 5; and treated groups – placebo control group (PCG) n = 8, experimental group (EG) n = 8, experimental group ad libitum (EGA) n = 8. The present study was approved by the local Institutional Review Board on Animal Research of University of São Paulo under the protocol of registry (CEEPA Proc. No. 009/2011 - University of São Paulo, Bauru, São Paulo, Brazil).
The animals were initially weighed and grouped by similar weights. Thereafter, randomization was performed to identify in which study groups the animals would be included, so as all study groups had animals with similar weights.
This methodology was followed because there is a known relationship between the weight of animals and the area of muscle fibers. This aimed to prevent the achievement of groups with significantly greater body weight at study onset, which might impair or confound the outcomes at study completion, concerning the possible change that might occur in some type of muscle fiber [22]. For effective dietary control, the animals were kept in individual cages with individual food and drink dispensers. The animals were also maintained in a room at temperature of 22°, respecting a light/dark cycle of 12 hours controlled by a timer.
Animals in the ICG were not submitted to any treatment and were killed at study onset. All other groups were treated for 4 weeks.
Animal feeding
The groups PCG and EGA received water ad libitum and a daily portion of 30 g of food (above the average daily ingestion of a normal rat). The food was served daily, so as the remaining food could be weighed to evaluate the quantity of food ingested by each animal on the previous day. Following, the mean food ingestion was calculated for each group. Animals in the EG individually received the mean quantity of food ingested by the PCG in the previous day. This method was maintained, so as animals in the EG would ingest only the same quantity of food ingested by animals in the PCG, allowing comparison of the actual effect of the drug, controlling both the diet (based on the PCG) and the combined effect of supplement and diet (EGA). Food was controlled and weighed daily until completion of the study period. Finally, the mean food intake was calculated for the four-week period.
Supplementation with HMB
Treatment with HMB was performed for the EG and EGA by gavage at a dose of 0.3 g kg-1 of body weight of HMB diluted in saline solution, for four weeks. Animals in the PCG received only saline in identical conditions as animals in the EG and EGA, following the method described by Gerlinger-Romero et al. (2011), also used by our group in Daré et al. (2015).
Weighing of animals
The animals were weighed weekly during the study period. For statistical analysis, the weight gain of rats was calculated in percentage comparing the initial and final weight of the animals.
Euthanasia and achievement of muscle specimens
The animals were euthanized by anesthetic overdose, by intramuscular injection of xylazine hydrochloride (Anasedan®) associated with ketamine hydrochloride (Dopalen®). Following, the Soleus and EDL muscles were dissected from the right posterior limb of each animal.
Histological processing
After dissection, the muscles were frozen in liquid nitrogen and stored in a freezer at -80ºC. Subsequently, cross-sections of muscle fragments were performed with 10µm thickness in a cryostat. The histological sections obtained from the middle portion of each muscle belly were placed on histological slides and stained with Hematoxylin-Eosin (HE).
Morphometric analysis of Soleus and EDL muscle fibers
Images of central field of HE-stained sections were obtained using a trifocal microscope Olympus BX 50 connected to a camera Olympus DP 71. The morphometry of sections was manually performed on the software Image ProPlus 6.5, measuring the transverse section area of 220 fibers per field of muscle.
Statistical analysis
Feeding - The mean food intake was calculated for the four-week period. The means were statistically compared by one-way Anova and the Tukey test.
Body Weight Gain - The percent weight gain was considered for comparison of animal weight. These data were submitted to the Kruskal-Wallis test, because there was no homogeneity of variances between data when submitted to the Bartlett test.
Morphometry - The mean transverse section areas of fibers of the Soleus and EDL muscles were compared using one-way ANOVA, followed by the Tukey test.
Statistical correlations - Morphometric data of fiber area of the Soleus and EDL were also submitted to the Spearman correlation test, to analyze if there was correlation between the transverse section area of the fiber and food intake, and between body weight and the transverse section area of the fiber.
Results
Animal feeding
Data did not reveal statistically significant difference between treated groups (Graph 1). Animals in the ICG were not included in this comparison, since they were killed before the treatment period.
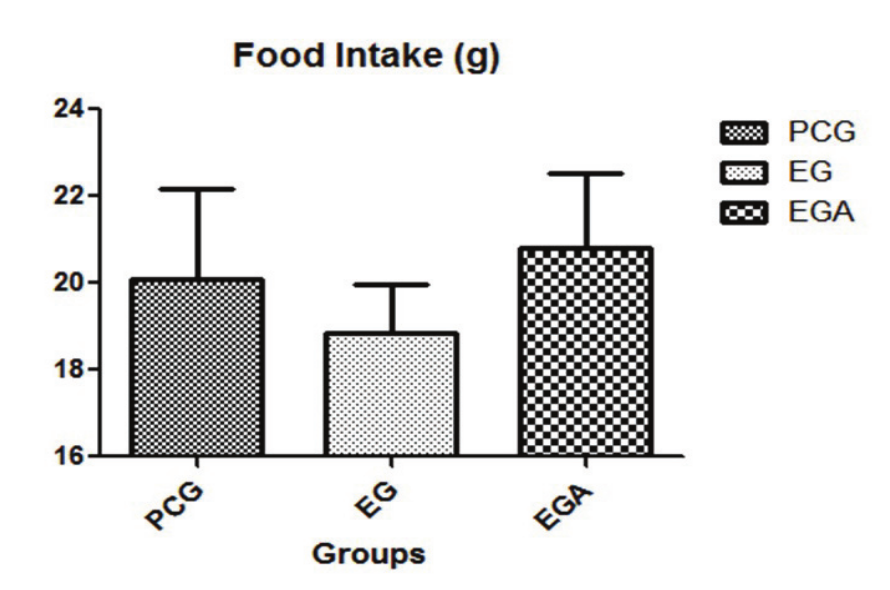
Graph 1.
Graph of mean food intake. PCG, placebo control group; EG, experimental group; EGA experimental group ad libitum. Values are means ± SD, (p < 0.05, by analysis of variance (ANOVA), followed by Tukey test).
Weight gain of animals
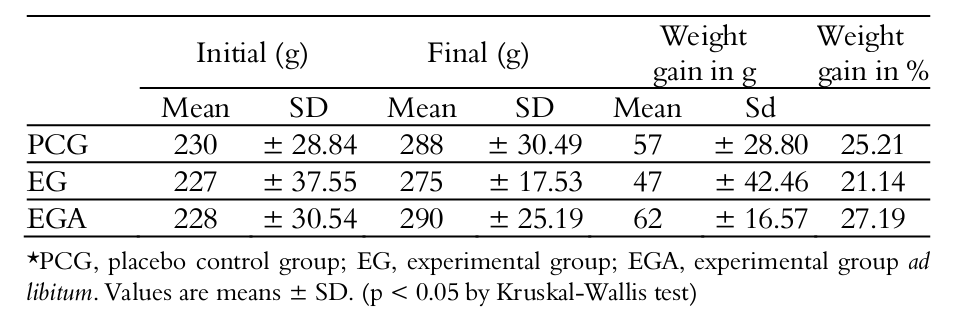
There was no statistically significant difference in weight gain between animals in the treated groups (Table 1).
Morphometry
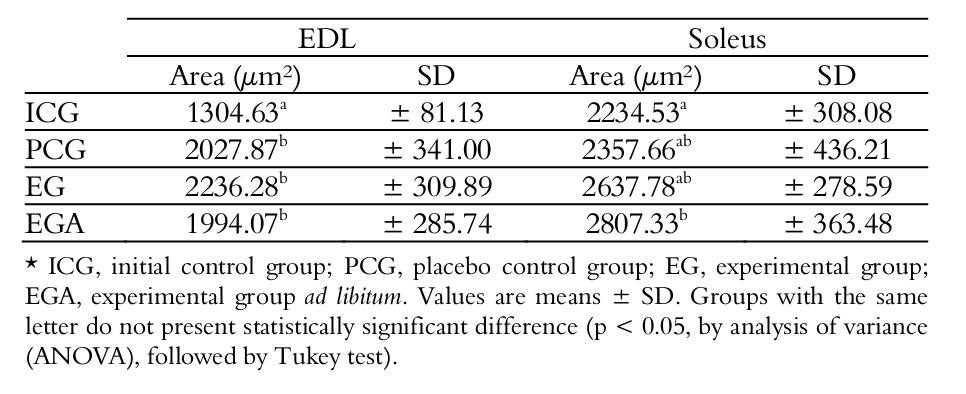
Concerning the EDL muscle, the groups PCG, EG and EGA did not present statistically significant difference from each other; however, they were statistically different from ICG. With regard to the Soleus muscle, the only statistically significant difference was observed between ICG and EGA (Table 2).
Statistical correlations
Analysis of data by the Spearman correlation test did not indicate correlation between the fiber area and the quantity of food intake, nor between the body weight and the fiber area, for both muscles analyzed.
Discussion
Food intake
According to Ward (2011), food has direct influence on body development and the maintenance of muscular mass. Conversely, Millward (2012) claims that food restriction or caloric surplus can determine loss or gain of muscular mass, respectively. Therefore, in this study, one group received food ad libitum (EGA) and the other received controlled food surplus (EG).
In the present study, diet was controlled only for the EG, and data did not reveal significant difference between groups concerning this factor. This may explain the lack of change and/or variation in the weight of animals, during this treatment period.
Effect of HMB on body weight
The difference between the mean final weights of groups did not reveal statistically significant difference, demonstrating that administration of 0.3 g kg-1 HMB per day for four weeks, in rats not submitted to physical exercise, was unable to cause changes in the body weight of rats. These data corroborate the findings of Baxter et al. (Baxter et al. 2005), in which the rats were supplemented for 90 days with doses of 1, 3 and 5% of HMB in their daily diet, and no significant difference was found in the weight of animals between supplemented and non-supplemented groups. Both data reinforce the assumption that HMB does not increase the body mass when not associated with physical exercise. In their review, Szcześniak, Ostaszewski, Fuller, Ciecierska and Sadkowski (2015) reported three manuscripts, namely Tatara, Sliwa, & Krupski (2007), Tatara, Sliwa, Krupski, & Worzakowska (2008) and Tatara, Krupski, Tymczyna, & Studziński (2012), in which the offspring of pigs and sheep were supplemented with HMB, and similar final body weight was observed between the control and supplemented groups. However, it was observed that supplemented animals were born larger and reached the slaughter weight more quickly than the control groups.
Effect of HMB on the skeletal muscle tissue
Investigations relating the HMB and its hypertrophic gains when associated with resistance exercises in untrained individuals are well established, yet little is known on its effects in sedentary individuals. Some papers have reported its effects on the muscle fibers in the absence of exercise, as reported by Kornasio, Riederer, Butler-Browne, Mouly, Uni, & Halevy (2009), who analyzed the effect of HMB on myoblasts in cell culture and observed positive effect on the proliferation and myogenic differentiation in cell culture.
Some authors in the literature, such as Gerlinger-Romero et al. (2011), Pimentel et al. (2011), Pinheiro et al. (2012), Yonamine et al. (2014) and Daré et al. (2015), presented results using the same study period and approximately the same dose of HMB. The three former papers did not report muscle atrophy when analyzing the muscle weight, corroborating the present results. Conversely, the study of Yonamine et al. (2014) revealed muscle atrophy based on the morphometry of the soleus muscle fibers, different from the present results. However, data obtained by Daré et al. (2015) are similar to the present findings, while Sciote, Horton, Rowlerson and Link (2003) stated that there are some differences between branchiomeric muscles (mastication) and somitic muscles (limbs and trunk).
Gerlinger-Romero et al. (2011) analyzed the administration of HMB for 4 weeks at a dose of 320 mg kg-1 of body weight per day, for four weeks in rats not submitted to physical exercise. At treatment completion, expression of the growth hormone (GH) in the pituitary gland and the serum level of insulin-like growth factor 1 (IGF-1) were increased. However, analysis of the muscle weight of the soleus and extensor Digitorum longus muscles of the control and supplemented groups did not reveal statistically significant difference, corroborating the present data.
Following the same study design, Pinheiro et al. (2012) evaluated the white and red portions of the gastrocnemius muscle, assessing the glycogen and ATP content in both parts, and observed that HMB significantly increased both the glycogen and ATP levels in both portions. However, no difference was observed in muscle weight between the supplemented and placebo groups.
Pimentel et al. (2011) followed the same methodology and, among studies using this design, this was the only reporting increase in muscle weight (EDL muscle). The authors also observed increase in the expression of mTOR and in phosphorylation of p70S6k, both regulators of protein synthesis in skeletal muscles.
The literature evidences that supplementation with HMB creates a favorable environment for muscle growth, concerning the hormonal levels (Gerlinger-Romero et al., 2011), energetic substrate (Pinheiro et al., 2012) and protein expression (Pimentel et al., 2011). Otherwise, from a practical point of view, it is observed that the absence of hypertrophic stimulus under exercise results in difficult muscle mass gain.
Effect of HMB on the Soleus and Extensor Digitorum longus muscles
Experimental studies on the skeletal muscles, in both animals and humans, greatly focus on the size and type of muscle fibers, including investigations on sports and neuromuscular diseases.
The present study investigated the soleus muscle (also called red, gravitational, ankle plantar flexor with predominance of slow contraction fibers) (Eng et al., 2008), with frequency of 80.7 to 98.4% of type I fibers (Narusawa, 1985; Soukup, Smerdu, & Zachařová, 2009; respectively); and the EDL (called white, anti-gravitational, ankle dorsiflexor with predominance of fast contraction fibers) (Eng et al., 2008), with 94.5 to 98% of type II fibers (Soukup, Zachařová, & Smerdu, 2002; Armstrong & Phelps, 1984; respectively).
In the present study, in the EDL muscle, the muscle fiber morphometry did not present statistically significant difference between treated groups, demonstrating that HMB had no interference, despite the higher values compared to animals in the ICG. Comparison between the treated groups revealed that neither feeding nor supplementation induced statistically significant atrophy or hypertrophy in the transverse section area of fibers.
Utilization of the HMB produced different effect in the soleus muscle compared to the EDL muscle. Only the transverse section area of EGA was significantly greater compared to ICG, demonstrating that administration of HMB associated with ad libitum feeding promoted significant growth, without physical activity, during this study period. Probably, since this is a postural muscle, it may have been under greater demand than the EDL during the treatment period. This difference might be assigned to stimulation due to gravity load, reminding that the Soleus muscle has postural and antigravity action, while the EDL presents phasic and gravity action (Eng et al., 2008). Kovarik et al. (2010) working on septic rats, also observed more pronounced effect of HMB treatment on the soleus muscle than on EDL.
This fact is highlighted in the study of Hao, Jackson, Wang, Edens, Pereira, & Always (2011), who analyzed the effect of HMB on the limb muscle of suspended rats and observed that this substance was unable to prevent the atrophy in both red and white muscles, produced by reducing the body weight over the limbs of these animals. These data suggest that administration of HMB associated with ad libitum feeding would be able to promote significant growth in the transverse section area of the fiber in the soleus muscle, when compared to the ICG during the study period, which was not observed for the other groups.
Daré et al. (2015) analyzed the masticatory muscles of rats and observed a tendency of greater areas of fibers in the masseter of animals in experimental groups receiving treatment with HMB and ad libitum feeding. In the same study, this was not observed for the fibers of the anterior belly of the digastric muscle. It should be considered that the masseter is the most powerful muscle for mandible elevation and may also be considered an antigravitational muscle.
Within this context, the present results partly corroborate the current thinking and collaborate for the scientific basis, providing morphometric data on this aspect. However, further studies are necessary to elucidate other possible potentialities of HMB and its effects in other situations, such as reduction of fiber areas of mastication muscles submitted to soft diet (Vreeke, Langenbach, Korfage, Zentner, & Grünheid, 2011) with consequent change in craniofacial development (Kiliaridis, Engström, & Thilander, 1988). This concern is bases on several studies that demonstrated a tendency of increase in the number of children and adolescents who ingest easily chewable foods (Varrela, 1992).
Conclusion
In conclusion, in sedentary rats, the HMB did not promote change in body weight or in the morphometry of the EDL muscle fibers. However, when associated with ad libitum feeding and depending on the daily activity of the muscle (postural), it may provide positive changes, as observed for the soleus muscle.
Acknowledgements
All authors thank CAPES and University of São Paulo for the financial and material support, respectively.
References
Alvares, T. & Meirelles, C. M. (2008). Efeitos da suplementação de β-hidroxi-β-metilbutirato sobre a força e a hipertrofia. Revista de Nutrição, 21(1), 49-61. Doi: 10.1590/S1415-52732008000100006.S.,
Always, S. E., Pereira, S. L., Edens, N. K., Hao, Y., & Bennetti, B. T. (2013). β-Hydroxy-β-methylbutyrate (HMB) enhances the proliferation of satellite cells in fast muscles of aged rats during recovery from disuse atrophy. Experimental Gerontology, 48(9), 973-984. Doi: 10.1016/j.exger.2013.06.005.
Armstrong, R. B., & Phelps, R. O. (1984). Muscle fiber type composition of the rat hindlimb. American Journal Anatomy, 171(3), 259-272. Doi: 10.1002/aja.1001710 303.
Aversa, Z., Bonetto, A., Costelli, P., Minero, V. G., Penna, F., Baccino, F. M., ... Muscaritoli, M. (2011). Beta-hydroxy-beta-methylbutyrate (HMB) attenuates muscle and body weight loss in experimental cancer cachexia. International Journal of Oncology, 38(3), 713-720. Doi: 10.3892/ijo.2010.885.
Baxter, J. H., Carlos, J. L., Thurmond, J., Rehani, R. N., Bultman, J., & Frost, D. (2005). Dietary toxicity of calcium b-hydroxy-b-methylbutyrate (CaHMB). Food and Chemical Toxicology, 43(12), 1731-1741. Doi: 10.1016/j.fct.2005.05.016.
Daré, L. R., Dias, D. V., Rosa Junior, G. M., Bueno, C. R., Buchaim, R. L., Rodrigues. A. C., & Andreo, J. C. (2015). Effect of β-hydroxy-β-methylbutyrate in masticatory muscles of rats. Journal of Anatomy, 226(1), 40-46. Doi: 10.1111/joa.12256.
Di Luigi, L. (2008). Supplements and the endocrine system in athletes. Clinics in Sports Medicine, 27(1), 131-151. Doi: 10.1016/j.csm.2007.09.003.
Eley, H. L., Russell, S. T., Baxter, J. H., Mukerji, P,. & Tisdale, M. J. (2007). Signaling pathways initiated by β-hydroxy-β-methylbutyrate to attenuate the depression of protein synthesis in skeletal muscle in response to cachectic stimuli. American Journal of Physiology Endocrinology and Metabolism, 293(4), E923–E931. Doi: 10.1152/ajpendo.00314.2007.
Eley, H. L., Russell, S. T., & Tisdale, M. J. (2008a). Attenuation of depression of muscle protein synthesis induced by lipopolysaccharide, tumor necrosis factor, and angiotensin II by β-hydroxy-β-methylbutyrate. American Journal of Physiology Endocrinology and Metabolism, 295(6), E1409-E1416. Doi: 10.1152/ ajpendo.90530.2008.
Eley, H. L., Russell, S. T., & Tisdale, M. J. (2008b). Mechanism of attenuation of muscle protein degradation induced by tumor necrosis factor-α and angiotensin II by β-hydroxy-β-methylbutyrate. American Journal of Physiology Endocrinology and Metabolism, 295(6), E1417-E1426. Doi: 10.1152/ ajpendo.90567.2008.
Eng, C. M., Smallwood, L. H., Rainiero, M. P., Lahey, M., Samuel, R., Ward, S. R., & Lieber, R. L. (2008). Scaling of muscle architecture and fiber types in the rat hindlimb. The Journal of Experimental Biology, 211(14), 2336-2345. Doi: 10.1242/jeb.017640.
Fitschen, P. J., Wilson, G. J., Wilson, J. M., & Wilund, K. R. (2013). Efficacy of b-hydroxy-b-methylbutyrate supplementation in elderly and clinical populations. Nutrition, 29(1), 29-36. Doi: 10.1016/j.nut.2012.05.005.
Flakoll, P., Sharp, R., Baier, S., Levenhagen, D., Carr, C., & Nissen, S. (2004). Effect of beta-hydroxy-beta-methylbutyrate, arginine, and lysine supplementation on strength, functionality, body composition, and protein metabolism in elderly women. Nutrition, 20(5), 445-451. Doi: 10.1016/j.nut.2004.01.009.
Gerlinger-Romero, F., Guimarães-Ferreira, L., Giannocco, G., & Nunes, M. T. (2011). Chronic supplementation of beta-hydroxy-beta methylbutyrate (HMβ) increases the activity of the GH/IGF-I axis and induces hyperinsulinemia in rats. Growth Hormone & IGF Research, 21(2), 57-62. Doi: 10.1016/ j.ghir.2010.12.006.
Hao, Y., Jackson, J. R, Wang, Y., Edens, N., Pereira, S. L., & Always, S. E. (2011). β-Hydroxy-β-methylbutyrate reduces myonuclear apoptosis during recovery from hind limb suspension-induced muscle fiber atrophy in aged rats. American Journal of Physiology - Regulatory, Integrative and Comparative Physiology, 1(3), R701-R715. Doi: 10.1152/ajpregu.00840.2010.
Hasselgreen, P. (2014). β-hydroxy-β-methylbutyrate (HMB) and prevention of muscle wasting. Metabolism Clinical and Experimental, 63(1), 5-8. Doi: 10.1016/j.metabol.2013.09.015.
Kiliaridis, S., Engström, C., & Thilander, B. (1988). Histochemical analysis of masticatory muscle in the growing rat after prolonged alteration in the consistency of the diet. Archives of Oral Biology, 33(3), 187-193. Doi: 10.1016/0003-9969(88)90044-1.
Kornasio, R., Riederer, I., Butler-Browne, G., Mouly, V., Uni, Z., & Halevy, O. (2009). Beta-hydroxy-beta-methylbutyrate (HMB) stimulates myogenic cell proliferation, differentiation and survival via the MAPK/ERK and PI3K/Akt pathways. Biochimica et Biophysica Acta, 1793(5), 755-763. Doi: 10.1016/ j.bbamcr.2008.12.017.
Kovarik, M., Muthny, T., Sispera, L., & Holecek, M. (2010). Effects of beta-hydroxy-beta-methylbutyrate treatment in different types of skeletal muscle of intact and septic rats. Journal of Physiology and Biochemistry, 66(4), 311-319. Doi: 10.1007/s13105-010-0037-3.
Millward, D. J. (2012). Nutrition and sarcopenia: evidence for an interaction. Proceedings of the Nutrition Society, 71(1), 566-575. doi: 10.1017/S0029665112000201.
Narusawa, M. (1985). Change in fiber type in partially-denervated soleus muscle of the rat. The Tokai Journal of Experimental and Clinical Medicine, 10(5), 499-507. PMID:3837400.
Pimentel, G. D., Rosa, J. C., Lira, F. S., Zanchi, N. E., Ropelle, E. R., Oyama, L. M., … Santos, R. V.,T. (2011). ß-hydroxy-ß-methylbutyrate (HMB) supplementation stimulates skeletal muscle hypertrophy in rats via the mTOR pathway. Nutrition & Metabolism, 8(1), 1-7. Doi: 10.1186/1743-7075-8-11.
Pinheiro, C. H. J., Gerlinger-Romero, F., Guimarães-Ferreira, L., Souza-Jr, A. L., Vitzel, K. F., Nachbar, R. T., ... Curi, R. (2012). Metabolic and functional effects of beta-hydroxy-beta-methylbutyrate (HMB) supplementation in skeletal muscle. European Journal of Applied Physiology, 112(7), 2531-2537. Doi: 10.1007/s00421-011-2224-5.
Portal, S., Zadik, Z., Rabinowitz, J., Pilz-Burstein, R., Adler-Portal, D., Meckel, Y., … Nemet, D. (2011) The effect of HMB supplementation on body composition, fitness, hormonal and inflammatory mediators in elite adolescent volleyball players: a prospective randomized, double-blind, placebo-controlled study. European Journal of Applied Physiology, 111(9), 2261-2269. Doi: 10.1007/s00421-011-1855-x.
Sciote, J. J., Horton, M. J., Rowlerson, A. M., & Link, J. (2003). Specialized cranial muscles: how different are they from limb and abdominal muscles? Cells Tissues Organs;174(1-2), 73-86. Doi: 10.1159/000070576.
Smith, H .J., Mukerji, P., & Tisdale, M. J. (2005). Attenuation of proteasome-induced proteolysis in skeletal muscle by ß-hydroxy-ß-methylbutyrate in cancer-induced muscle loss. Cancer Research, 65(1), 277-283. PMID: 15665304.
Smith, H. J., Wyke, S. M., & Tisdale, M. J. (2004). Mechanism of the attenuation of proteolysis-inducing factor stimulated protein degradation in muscle by ß-hydroxy-ß-methylbutyrate. Cancer Research, 64(23), 8731-8735. Doi: 10.1158/0008-5472.CAN-04-1760.
Sonni, P. V., Iannuzzi, S., Aversa, Z., Tommasi, V., Ramaccini C., Pinto, G. … Muscaritoli, M. (2008). Beta-hydroxy-beta-methylbutyrate: a new therapeutic tool to counteract weight and muscle loss in cancer cachexia? Oral Communications / European Journal of Internal Medicine, 19(Suppl. 1), S1-S59. Doi: 10.1016/S0953-6205(08) 60047-3.
Soukup,T., Zachařová, G., & Smerdu, V. (2002). Fibre type composition of soleus and extensor digitorum longus muscles in normal female inbred Lewis rats. Acta Histochemica, 104(4), 399-405. Doi: 10.1078/0065-1281-00660.
Soukup, T., Smerdu, V., & Zachařová, G. (2009). Fiber type composition of unoperated rat soleus and extensor digitorum longus muscles after unilateral isotransplantation of a foreign muscle in long-term experiments. Physiological Research, 58(2), 253-262.
Supinski, G. S., & Callahan, L. A. (2014). β-hydroxy-β-methylbutyrate (HMB) prevents sepsis-induced diaphragm dysfunction in mice. Respiratory Physiology & Neurobiology, 196(1), 63-68. Doi: 10.1016/j.resp.2014. 02.015.
Szcześniak, K. A., Ostaszewski, P., Fuller, J. C., Ciecierska, A., & Sadkowski, T. (2015). Dietary supplementation of β-hydroxy-β-methylbutyrate in animals - a review. Journal of Animal Physiology and Animal Nutrition, 99(3), 405-417. Doi: 10.1111/jpn.12234. Epub 2014 Aug 7.
Tatara, M. R., Krupski, W., Tymczyna, B., & Studziński, T. (2012). Effects of combined maternal administration with alpha-ketoglutarate (AKG) and β-hydroxy-β-methylbutyrate (HMB) on prenatal programming of skeletal properties in the offspring. Nutrition & Metabolism, 9(1), 1-12. doi: 10.1186/1743-7075-9-39.
Tatara, M. R., Sliwa, E., & Krupski, W. (2007). Prenatal programming of skeletal development in the offspring: Effects of maternal treatment with β-hydroxy-β-methylbutyrate (HMB) on femur properties in pigs at slaughter age. Bone; 40(6), 1615–1622. Doi: 10.1016/j. bone.2007.02.018.
Tatara, M. R, Sliwa, E., Krupski, W., & Worzakowska, M. (2008). 3-Hydroxy-3-methylbutyrate administration diminishes fundectomy-induced osteopenia of the lumbar spine in pigs. Nutrition, 24(7-8), 753-760. Doi: 10.1016/j.nut.2008.03.009.
Varrela, J. (1992). Dimensional variation of craniofacial structures in relation to changing masticatory-functional demands. European Journal of Orthodontics, 14(1), 31-36. Doi: 10.1093/ejo/14.1.31.
Vreeke, M., Langenbach, G. E., Korfage, J. A., Zentner, A., & Grünheid, T. (2011). The masticatory system under varying functional load. Part 1: Structural adaptation of rabbit jaw muscles to reduced masticatory load. European Journal of Orthodontics, 33(4), 359-364. Doi: 10.1093/ejo/cjq083.
Ward, K. (2011). Symposium 2: vitamins in muscular and skeletal function musculoskeletal phenotype through the life course: the role of nutrition. Proceedings of the Nutrition Society, 71(1), 27-37. Doi: 10.1017/S0029 665111003375.
Wu, H., Xia, Y., Jiang, J., Du, H., Guo, X., Liu, X., … , K. (2015). Effect of beta-hydroxy-beta-methylbutyrate supplementation on muscle loss in older adults: a systematic review and meta-analysis. Archives of Gerontology and Geriatrics, 61(2), 168-175. Doi: 10.1016/j.archger.2015.06.020.
Yonamine, C. Y., Teixeira, S. S., Campello, R. S., Gerlinger-Romero, F., Rodrigues, C.F., Guimarães-Ferreira. L., … Nunes, M. T. (2014). Beta hydroxy beta methylbutyrate supplementation impairs peripheral insulin sensitivity in healthy sedentaryWistar rats. Acta Physiologica, 212 (1), 62-74. Doi: 10.1111/apha.12336.
Zanchi, N. E., Gerlinger-Romero, F., Guimaraes-Ferreira, L., Siqueira-Filho, M. A., Felitti, V., Lira, F.S., … Lancha-Jr, A. H. (2010). HMB supplementation: clinical and athletic performance-related effects and mechanisms of action. Amino Acids, 40(4). 1015-1025. Doi: 10.1007/s00726-010-0678-0.
Notes
Author notes
mizael.pereira@usp.br