ABSTRACT: The specific impulse (Isp) is an important performance parameter that describes energy efficiency of propellant combustion and is intimately related to the rocket engine thrust. In this study, it was possible by using only two variables, i.e., the heat of reaction (Q) and the number of moles of gaseous reaction products per gram of propellant (Ng) calculated according to [H2O-CO2] arbitrary decomposition assumption and constants derived from the ISPBKW code to predict the specific impulse of more than 165 compositions belonging to virtually all classes of propellants such as monopropellants, single-base, double-base, triple-base, and cast modified double-base (CMDB) propellants, pseudo-propellants, composite propellants, liquid mono- and bipropellants, and finally hybrid propellants. Further analysis reveals that for C-H-N-O containing propellants, the specific impulse values estimated using the new method should not deviate more than 5% from the output of the ISPBKW thermochemical code.
KEYWORDS: Specific impulse prediction,Solid propellants,Liquid propellants,Hybrid propellants,ISPBKW code.
ORIGINAL PAPER
A Reliable Method for Predicting the Specific Impulse of Chemical Propellants
Received: 25 May 2017
Accepted: 29 October 2017
The first propellant ever created was black powder or gunpowder, which consists of a physical mixture of saltpeter, charcoal and sulfur. For many centuries, this low energy composite mixture served as the sole energetic material for both military and civilian applications. The need for cleaner, more energetic propellants led to the invention of smokeless powder, a homogenous mixture of two well-known explosives substances: nitrocellulose (NC) and nitroglycerine (NG) (Klapötke 2011). For instance, a solid explosive like 2,4,6-Trinitrotoluene (TNT) will detonate when subject to a powerful shock wave but it will only burn or deflagrate when brought into contact with a flame, and hence it is not surprising that most of today's propellants formulations contain large quantities of explosive materials since the thermochemistry of both explosives and propellants is essentially the same (Kubota 2015) but they do differ in their rate of energy release which, to a large extent, depends on the nature and amplitude of the external stimuli that cause them to react in one way or another. Given the relatively long burning time from several seconds to several minutes and coupled with the generation of substantial amounts of hot gaseous products, chemical propellants, whether in solid or liquid state are the principle source of the propulsive force that accelerates rockets, guided missiles and artillery shells. The thrust that a rocket motor develops is directly linked to the specific impulse (Isp) defined as the thrust delivered per unit flow weight of propellants consumed (Bourasseau 1990). Two commonly used units for (Isp) are seconds (s) and Ns g-1 (or Ns kg-1). Achieving high (Isp) values is always desirable especially in the case of long range missiles. It can be shown, that almost 45% gain in range can be realized by increasing only 5% the specific impulse of a typical intercontinental ballistic missile having an initial range of 5000 nautical miles (Thompson Jr. 1960).
More specifically, the rocket combustion chamber temperature (Tc) and the average molecular weight (M) of the exhaust gas are the primary dominant factors determining the specific impulse (Kinney 1960). Thus, the relationship relating (Isp) to (Tc) and (M) is shown by:
As Eq. 1 states, this highest (Isp) value can be reached by finding a propellant formulation capable of generating the highest (Tc) and the lowest possible (M). However, during rocket operation, the combustion chamber walls may be weakened due to the large heat transfer from the burning propellant, which can ultimately lead to a catastrophic structural failure; therefore combustion temperature should be kept at an acceptable level without sacrificing overall performance. Another problem facing propellant formulators is the difficulty encountered in the experimental evaluation of the specific impulse that requires hundreds of kilograms of potentially dangerous and explosives energetic materials (Bhat et al. 1988; Lempert et al. 2011). Consequently, researchers rely heavily on thermochemical code such as NASA CEA (Gordon and McBride 1996) or TERRA code (Trusov 2002) to accurately compute propellant performance at a determined pressure ratio defined as (Pc: Pa) where (Pc) and (Pa) are the combustion chamber pressure and the ambient pressure at the nozzle exit, respectively. Similarly to thermochemical codes, relationships derived from empirical data present a viable alternative to compute the performance of explosives and propellants. Recent studies show that condensed explosive detonation velocity (Keshavarz 2012), pressure (Keshavarz et al. 2014), temperature (Keshavarz and Nazari 2006) and other performance parameters (Frem 2017), as well as the specific impulse of monopropellants (Frem 2016), can be precisely calculated using only few experimental data (e.g. crystal density, heat of formation, etc.) and no more than a hand-held calculator. Accordingly, the intent of the next section is to present a detailed derivation of a new and simple method for predicting the specific impulse of C-H-N-O chemical propellants that do not contain any metal additives such as aluminum.
The driving force behind the current study was the work of Kamlet & Jacobs (K-J) published in 1968 (Kamlet and Jacobs 1968), where the authors have showed that the detonation velocity (D) and pressure (P) for C-H-N-O containing explosives can be predicted following Eqs. 2 and 3:
where: ρ0 (g·cm-3) is the explosive initial density; A, B, K are constants and equal to 1.01, 1.30, and 15.58, respectively, while; (Ng) is the number of moles of gaseous detonation products per gram of explosive; (Mg) is the average molecular weight of these gases; and (Q) is the heat of detonation in (cal·g-1). The (K-J) method presumes that for C-H-N-O explosives at an initial ρ0 = 1.7 - 1.9 g·cm-3, the major decomposition products are water (H2O), carbon dioxide (CO2) and nitrogen (N2), and often termed the [H2O-CO2] arbitrary decomposition assumption. Despite its simplicity, the detonation velocity and pressure calculated using the [H2O-CO2] arbitrary are in good agreement with the value obtained using complex thermochemical codes. The (Φ) parameter was later used to obtain the Gurney velocity (√2EG) (Hardesty and Kennedy 1977; Kamlet and Finger 1979), an important performance parameter that represents the ability of a given explosive to push and accelerate a surrounding metal shell. Given the fact that explosives and propellants possess comparable energy content and, in many cases similar chemical compositions, it was felt that the [H2O-CO2] arbitrary could, in principle, be used to estimate the specific impulse. To test this hypothesis, a thorough study was made in order to uncover the potential influence of (Ng), (Mg), and (Q) on specific impulse. The results show, that only (Ng) and (Q) were significant in obtaining a relationship capable of accurately predicting propellant (Isp) (Eq. 4):
where the intercept X1 and the coefficient C1 - C2 were derived using a multiple linear regression analysis (MLRA) and the computed (Isp) values for thirty-seven C-H-N-O monopropellants shown in Table 1 (Eq. 5):
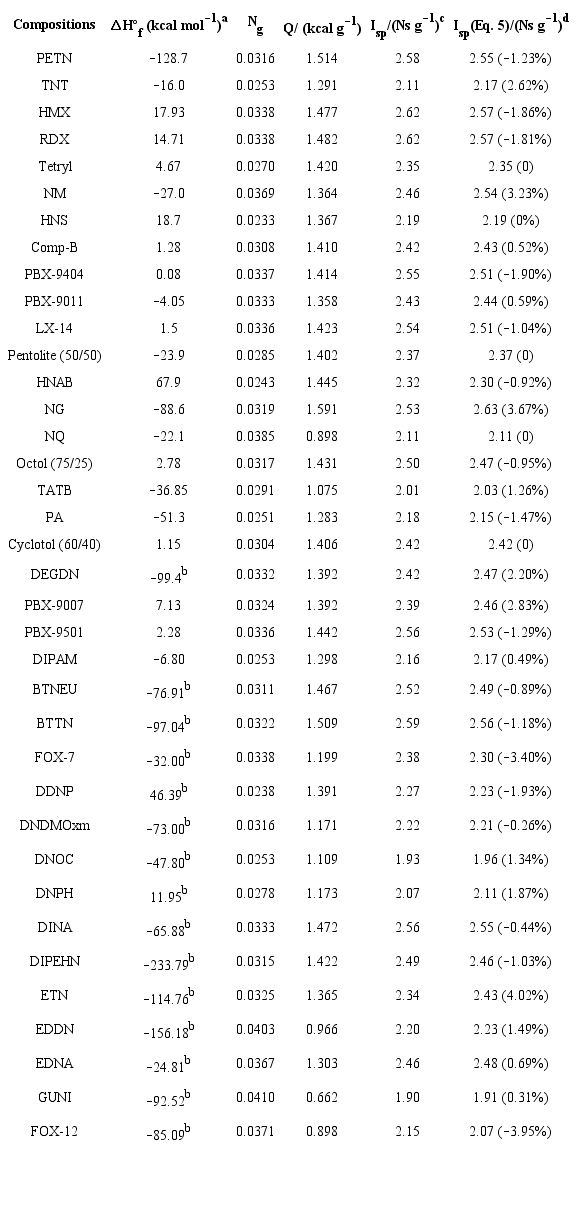
where (Q) and (Ng), now termed the heat of reaction in (kcal g-1) and the number of moles of gaseous reaction products per gram of propellant, respectively, were calculated according to Eqs. 6 and 7:
where: a, b, c and d are the number of carbon (C), hydrogen (H), nitrogen (N) and oxygen (O) atoms in the propellant composition, (ΔHºf kcal mol-1) represents the condensed phase heat of formation (HOF), and Mw is the composition's molecular weight. The calculated specific impulse of the training set was obtained using the ISPBKW thermochemical code (Mader 2008) at a predetermined chamber and nozzle exit pressure of 68.9 and 1 bar (Pc: Pa = 68.9:1), respectively. The coefficient of determination (R2) of the regression equation is equal to 0.948. Relevant statistical results (t-stat, P-values, regression coefficients C1 - C2, etc.) obtained from the (MLRA) are summarized in Table 2. The P-values < 0.05 and the t-values clearly indicate that the suggested independent variables are significant to estimate specific impulse. Moreover, the very small significance F value (1.45810-22) confirms the validity of the regression output.
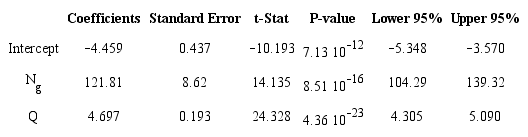
It can be seen from Table 1 that there is a good agreement between the specific impulses calculated using the ISPBKW code and the values obtained by applying Eq. 5 and in all of the studied cases the deviation did not exceed ± 3 - 4%.
The intention of the next sections is to evaluate the predictive ability of the obtained model by using a test set comprising not only solid single component monopropellants, but also multicomponent solid, liquid, and hybrid propellants.
The monopropellants shown in Fig. 1 were carefully chosen so as to cover the most important class of energetic molecules such as nitroaromatics, aliphatic nitrate esters and salts of high-nitrogen content heterocycles. Moreover, the specific impulses for the twenty studied structures were predicted using both Eqs. 5 and 8:
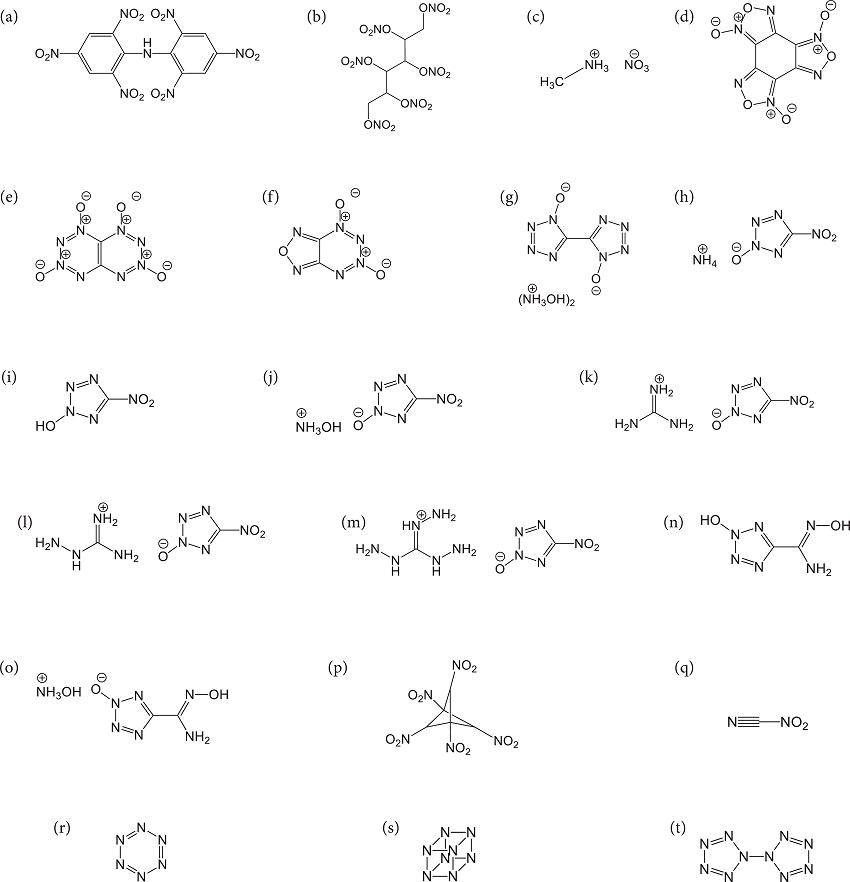
where: a, b, c and d are the number of carbon (C), hydrogen (H), nitrogen (N) and oxygen (O) atoms while is the number of -NH and -NH2 groups, and (nAr) is the number of aromatic rings that might be present in the propellant compositions (Keshavarz 2008). Since both Eqs. 5 and 8 were derived with the help of the ISPBKW code, it will be useful to compare the predictive power of each of them with the code output. The results reported in Table 3 show that the specific impulses calculated using Eq. 5 are in good agreement (3 - 4% deviation) with the actual values obtained from the ISPBKW code. On the other hand, the application of Eq. 8 can, in certain instances, result in a large deviation (> 7%) in the calculated (Isp) such as in the cases of structures showed in Figs. 1b, 1c, 1f, 1l, 1m, and 1q to 1t.

A graphical plot of the predicted (Isp) using Eq. 5 and Eq. 8 versus ISPBKW code results is depicted in Fig. 2.
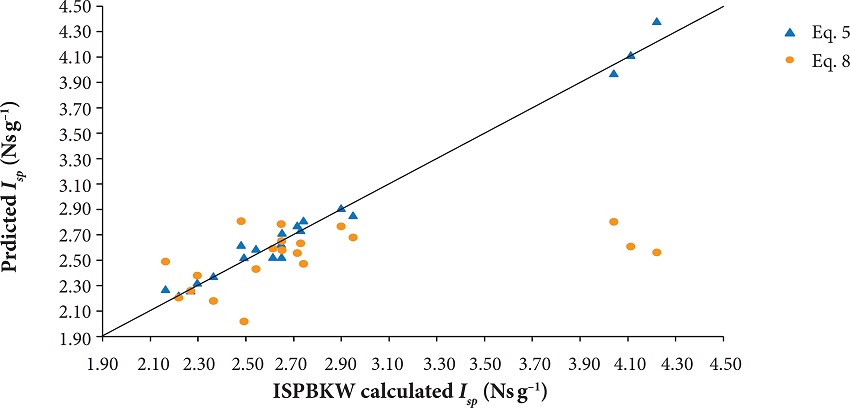
It can be shown that the data points obtained using the Keshavarz's method are more scattered around the diagonal line bisecting the graph (i.e. the line of perfect agreement between predicted and code results) compared to the ones obtained using the new method. Obviously, the major advantage of using Eq. 5 over Eq. 8 is the capability of the first to accurately predict the specific impulse of the yet hypothetical homoleptic polynitrogen compounds possessing the general formula Nx (x = 6, 8, 10, etc.).
Typical single-base (SB), double-base (DB), composite modified double-base (CMDB) and triple-base (TB) propellant formulations are listed in Table 5. Single-base propellants contain nitrocellulose, which has been gelatinized in acetone or in alcohol-ether solvent mixture and to which has been added various additives in order to improve the quality of the propellant powder. Likewise, double-base (DB) propellants are mixtures of nitrocellulose and nitroglycerine along with other additives, such as plasticizers, stabilizers and burn rate controllers, all of which alter the mechanical and thermal properties of the composition. The incorporation of solid oxidizers (e.g. ammonium perchlorate, ammonium dinitramide, etc.) and in some cases metallic fuel (e.g. aluminum) into the (DB) formulation, results in the formation of composite modified double-base (CMDB) propellants characterized by high specific impulse figures (Agrawal 2010).

On the other hand, standard triple-base (TB) propellants contain nitroguanidine (picrite), which has been added to the (NC-NG) matrix in order to reduce muzzle flash and gun barrel erosion (U.S. Army Material Command 1965). The condensed heats of formation and atomic compositions of (CMDB) and (TB) propellants (except M15, M17 and T34) were calculated from their individual ingredients shown in Table 4. The estimated specific impulses using Eq. 5 were in close agreement with thermochemical code results. Upon combustion, ammonium perchlorate containing compositions produces hydrogen chloride which is not taken into account in calculating the value of (Ng) and (Q), which explains the relatively high deviation of about ~4% in the predicted specific impulse for the CMBD formulations containing 29.5% ammonium perchlorate. It should be noted that many of the investigated (SB), (DB) and (TB) formulations contain small amount of mineral additives (e.g. KNO3, K2SO4) used as flash reducers; however in this study, these salts were excluded in computing the (Isp) values.
Pseudo-propellants are homogenous mixtures formed by physically mixing two or more ingredients possessing particle sizes on the order of 10 µm or less (Beckstead 2006). Numerical and experimental studies have been performed in order to study the combustion behavior and flame structure of pseudo-propellants especially nitramine/energetic binder binary systems (Kim et al. 2002; Lee et al. 1999). The use of energetic binders in propellant formulations has a distinctive advantage over traditional inert binders (e.g. HTPB) because they offer substantial additional energy during burning, which ultimately increases the overall specific impulse (Talawar et al. 2009). Some of the most studied nitramine-based pseudo-propellants are listed in Table 6 along with the calculated and estimated (Isp) values, where one can clearly see that there is a very good agreement between the specific impulse values predicted using Eq. 5 and thermochemical code output.

Ammonium perchlorate (AP)-based composite propellants (CPs) are by far the most important class of solid rocket propellants. Typical compositions are heterogeneous mixtures of an oxidizer mainly AP (60 - 80%) dispersed in a polymeric binder (e.g. HTPB, 10 - 15%) to which a metallic fuel such as aluminum (15 - 20%) may be added (Jain et al. 2009). Moreover, AP-based (CPs) offers high performance and excellent mechanical properties (Davenas 1993); however, they have some drawbacks, including the generation of large amounts of pollutants like toxic hydrogen chloride gas, which contributes to the depletion of the ozone layer (Lempert et al. 2006). Furthermore, the interaction of hydrogen chloride with the ambient atmosphere stimulates moisture condensation resulting in the formation of white secondary smoke, thereby making the firing position highly vulnerable to hostile action (Chaturvedi and Dave 2015). The need for powerful propellants with low environmental impact led to the development of chlorine-free oxidizers such as ammonium dinitramide (ADN) and hydrazinium nitroformate (HNF) which, in combination with suitable binders, can yield highly performant (CPs). The specific impulses for a number of (CP) formulations based on (ADN), (HNF) ammonium nitrate (AN) and high-enthalpy C-N-O organic oxidizers (see Fig. 3) have been calculated and are shown in Table 7.

The good agreement (± 3 - 4% deviation) seen between ISPBKW code and Eq. 5 calculations indicates that the new model is capable of accurately predict the specific impulse of composite propellants having diverse chemical compositions and a wide range of performance capabilities.
From an engineering point of view, a solid rocket motor (SRM) has far fewer components than a liquid rocket engine (LRE). While a (SRM) is basically a propellant charge (grain) fitted inside a metallic case to which is attached a supersonic exhaust nozzle, a (LRE) consists of a myriad of parts including thrust chambers, propellant tanks connected to a piping network, power sources, etc., all of which should work together in order to deliver the required thrust (Sutton and Biblarz 2001). Moreover, and in contrast to solid propellants, the fuel and the oxidizer that make up the liquid bipropellant are kept in separate tanks and are only mixed when injected into the engine's combustion chamber.
It is important to note, that an optimal mixing ratio (mass of oxidizer to mass of fuel, O/F) is carefully chosen not only to obtain high specific impulse, but also to keep the combustion temperature at an acceptable level.
The two major groups of bipropellant systems used today in (LRE) are cryogenic and storable propellants. The selection of one of these is influenced by many factors such as performance, cost, handling, toxicity, supply, and storage considerations. Other missile or space shuttle components like the auxiliary power drives and the roll-control thrusters (Huzel and Huang 1992) employ monopropellant systems that, unlike bipropellants, do not require an external oxidizer source in order to undergo an exothermic reaction. Only monopropellants formed by an oxidizer and a fuel dissolved in a homogenous liquid phase will be treated here; however other classes of liquid monopropellants also exist. For example nitromethane and isopropyl nitrate are those monopropellants where both the oxidizer and the fuel are held together through covalent bonds in the same molecule. The final class of liquid monopropellants includes those materials formed by an unstable arrangement of atoms such as hydrogen peroxide and hydrazine which, when brought into contact with a suitable catalyst, will decompose with the generation of thermal energy and gaseous products (U.S. Army Material Command 1969). Some representative liquid monopropellant and bipropellant compositions, along with their calculated performance characteristics, are shown in Tables 8 and 9. As it is evident from the two tables and the percentage deviation values, the specific impulse estimated using Eq. 5 compare favorably with the output of ISPBKW thermochemical code.

The concept of a hybrid rocket, which employs a solid fuel and a liquid oxidizer (or vice-versa) as a propellant, is not new and dates back to the early 1930s (Krishnan 2002). In a typical hybrid rocket a highly pressurized gas is used to inject liquid oxidizer into the combustion chamber containing the solid grain fuel which causes the later to erode and vaporize leading to ignition and subsequent combustion. It is also well known that hybrid propellants burn as a macroscopic turbulent diffusion flame where the mixing ratio differs along the length of the grain, unlike solid and liquid propellants where the (O/F) value is uniform throughout the combustion chamber (Altman and Holzman 2007). Some features, like safety, the possibility to restart, and low cost are only few of the advantages that a hybrid rocket has over (SRM) and (LRE). A number of hybrid propellant formulations are shown in Table 10 along with their mixture ratios, from which the specific impulse was computed using the ISPBKW code and Eq. 5.
Many hybrid compositions typically employ HTPB as fuel due to its ease of processing, low cost of production and availability (Sutton and Biblarz 2001). The results of specific impulse calculations show that, as in the case of solid and liquid propellants, Eq. 5 is also valid to estimate the specific impulse of hybrid formulations and an expected deviation of ± 3 - 5% from ISPBKW code is obtained for all tested samples.
The principal aim of the present study is to prove that, for a large body of C-H-N-O containing propellants, the specific impulse (Isp) can be accurately obtained by applying Eq. 5 that employs only two variables, namely, the heat of reaction (Q) and the number of moles of gaseous reaction products per gram of propellant (Ng) calculated according to the well-known [H2O-CO2] arbitrary decomposition assumption, which is a method proposed and used by chemist Mortimer J. Kamlet and physicist Sigmund J. Jacobs to predict condensed high explosives performance in the late 1960s. Throughout the present study, Eq. 5 was used to estimate the (Isp) of solid, liquid and hybrid propellants. The results were compared to the output of the ISPBKW code, which shows that a deviation of no more than ± 3 - 4% was obtained in most cases. Finally, future work should be directed toward finding a modified form of Eq. 5 capable of predicting the specific impulse of propellant formulations containing chlorine-based oxidizers and metals such as aluminum and boron.
AB: Active binder, poly(methylvinyltetrazole)/NG/2,4-dinitro-2,4-diazapentane (C19H34.5N19O29.5)
ADN: Ammonium dinitramide (H4N4O4)
Aerozine-50: 50/50 hydrazine/UDMH (C1.667H12.909N4.788)
AN: Ammonium nitrate (NH4NO3)
AP: Ammonium perchlorate (NH4ClO4)
BAMO: 3,3-bis(azidomethyl)oxetane (C4H6N6O)n
BDNPA-F: bis(2,2-dinitropropyl) acetal/bis(2,2-dinitropropyl) formal, 50/50 (C2.347H4.068N1.254O3.134)
BTNEU: 1,3-bis(2,2,2-trinitroethyl)urea (C5H6N8O13)
BTTN: 1,2,4-Butanetriol trinitrate (C4H7N3O9)
Comp-B: 63/36/1 RDX/TNT /Wax (C2.03H2.64N2.18O2.67)
CL-20: 2,4,6,8,10,12-Hexanitro-2,4,6,8,10,12-hexaazaisowurtzitane (C6H6N12O12)
Cyclotol 60/40: RDX/TNT (C 2.04H2.50N2.15O2.68)
DCPD: Dicyclopentadiene (C10H12)
DEGDN: Diethyleneglycol Dinitrate (C4H8N2O7)
DEP: Diethyl Phthalate (C12H14O4)
DETA: Diethylenetriamine (C4H13N3)
DDNP: Diazodinitrophenol (C6H2N4O5)
DINA: Dioxyethylnitramine Dinitrate (C4H8N4O8)
DIPAM: 3,3'-diamino-2,2',4,4',6,6'-hexanitrobiphenyl (C12H6N8O12)
DIPEHN: Dipentaerythritol Hexanitrate (C10H16N6O19)
DNDMOxm: Dinitrodimethyloxamide (C4H6N4O6)
DNOC: 4,6-Dinitro-o-cresol (C7H6N2O5)
DNPH: (2,4-Dinitrophenyl)hydrazine (C6H6N4O4)
EDDN: Ethylenediamine Dinitrate (C2H10N4O6)
EDNA: Ethylenedinitramine (C2H6N4O4)
ETN: Ethriol Trinitrate (C6H11N3O9)
FOX-7: 1,1-Diamino-2,2-dinitroethene (C2H4N4O4)
FOX-12: N-guanylurea-dinitramide (C2H7N7O5)
GAP: Glycidyl azide polymer (C3H5N3O)n
GUNI: Guanidine Nitrate (CH6N4O3)
HAN: Hydroxylammonium nitrate (H4N2O4)
HEH: Hydroxyethylhydrazine (C2H8N2O)
HMX: 1,3,5,7-Tetranitro-1,3,5,7-tetraazacyclooctane (C4H8N8O8)
HNAB: 2,2',4,4',6,6'-Hexanitroazobenzene (C12H4N8O12)
HNF: Hydrazinium nitroformate (CH5N5O6)
HNS: 2,2',4,4',6,6'-Hexanitrostilbene (C14H6N6O12)
HTPB: Hydroxyl-terminated polybutadiene (C7.075H10.65N0.063O0.223)
Hydine: 60/40 UDMH/DETA (C3.551H13.040N3.163)
LX-14: 95/5 HMX/Estane (C1.52H2.92N2.59O2.66)
MA: Methylamine (CH5N)
NG: Nitroglycerine (C3H5N3O9)
NQ: Nitroguanidine (CH4N4O2)
NM: Nitromethane (CH3NO2)
Octol 75/25: 75/25 HMX/TNT (C1.78H2.58N2.3602.69)
PA: Picric Acid (C6H3N3O7)
Paraffin wax: (C50H102)
PBX-9007: 90/9.1/0.5/0.4 RDX/PS/DOP/Rosin (C1.97H3.22N2.43O2.44)
PBX-9011: 90/10 HMX/Estane (C1.73H3.18N2.45O2.61)
PBX-9404: 94/3/3 HMX/Nitrocellulose/ Tris-β-Chloroethyl phosphate (C1.40H2.75N2.57O2.69Cl0.03P0.01)
PBX-9501: 95/2.5/2.25 HMX/Estane/BDNPF (C1.47H2.86N2.60O2.69)
PE: Polyethylene (C2H4)n
Pentolite (50/50): 50/50 TNT/PETN (C2.33H2.37N1.29O3.22)
PETN: Pentaerythritol tetranitrate (C5H8N4O12)
PGN: poly(glycidyl nitrate) (C3H5N1O4)n
PLN: Polynitromethyloxetane (C3H9N1O4)n
PMVT: Poly(methylvinyltetrazole) (C4H6N4)n
PVMDO: Poly(vinylmethoxydiazen-N-oxide) (C3H6N2O2)n
RDX: 1,3,5-Trinitro-1,3,5-triazacyclohexane (C3H6N6O6)
RFNA: Red fuming nitric acid 84/14/2 HNO3/NO2/H2O (H0.931N0.981O2.826)
RP-1: Mixture of naphthenes/paraffins/C12 olefins (CH1.95)n
TAGAZ: Triaminoguanidinium azotetrazolate (C4H8N22)
TATB: 1,3,5-Triamino-2,4,6-trinitrobenzene (C6H6N6O6)
Tetryl: 2,4,6-Trinitrophenyl-N-methylnitramine (C7H5N5O8)
TMETN: Trimethylolethane trinitrate (C5H9N3O9)
TNM: Tetranitromethane (CN4O8)
TNT: 2,4,6-Trinitrotoluene (C7H5N3O6)
NC (12%N): Nitrocellulose (C6H7.74N2.26O9.52)
NC (13.35%N): Nitrocellulose (C6H7.29N2.71O10.41)
UDMH: Unsymmetrical dimethylhydrazine (C2H8N2)
2-NDPA: 2-Nitrodiphenylamine (C12H10N2O2)
1a: 2,4,6,2',4',6'-Hexanitrodiphenylamine (C12H5N7O12)
1b: Mannitol Hexanitrate (C6H8N6O18)
1c: MAN, Methylamine Nitrate (CH6N2O3)
1d: BTF, Benzotris[1,2,5]oxadiazole-1,4,7-trioxide (C6N6O6)
1e: DTTO, di-1,2,3,4-tetrazine tetraoxide (C2N8O4)
1f: FTDO, [1,2,5]Oxadiazolo[3,4-e][1,2,3,4]-Tetrazine-4,6- di-N-Oxide (C2N6O3)
1g: TKX-50, dihydroxylammonium 5,5'-bistetrazole-1,1'-diolate (C2H8N10O4)
1h: ANTX, Ammonium 5-Nitrotetrazolate-2N-oxide (CH4N6O3)
1i: 5-Nitrotetrazole-2N-oxide (CHN5O3)
1j: HxNTX, Hydroxylammonium 5-Nitrotetrazolate-2N-oxide (CH4N6O4)
1k: GNTX, Guanidinium 5-Nitrotetrazolate-2N-oxide (C2H6N8O3)
1l: AGNTX, Aminoguanidinium 5-Nitrotetrazolate-2N-oxide (C2H7N9O3)
1m: TAGNTX, Triaminoguanidinium 5-Nitrotetrazolate-2N-oxide (C2H9N11O3)
1n: 5-Aminohydroximoyl-2-hydroxytetrazole (C2H4N6O2)
1o: Hydroxylammonium 5-aminohydroxyimoyl-tetrazole-2-oxide (C2H7N7O3)
1p: 1,2,3,4,5-pentanitrobicyclo[1.1.1]pentane (C5H3N5O10)
1q: Nitryl cyanide (CN2O2)
1r: Hexaazabenzene (N6)
1s: Octaazacubane (N8)
1t: Bipentazole (N10)
3u: 5-Trinitromethyl-bistetrazolo[1,5-a:1',5'-c][1,3,5]triazine (C4N12O6)
3v: Di(1H-triazirin-1-yl)-6-trinitromethyl-1,3,5-triazine (C4N12O6)
M1: 83.13/9.78/4.89/0.98/0.73/0.49 NC (13.15%N)/Dinitrotoluene/Dibutylphthalate/Diphenylamine/Ethyl alcohol/Water (C2.535H3.102N0.894O3.370)
M1A1: 83.17/9.84/4.44/0.98/0.98/0.59 NC (12.60%N)/Dinitrotoluene/Dibutylphthalate/Diphenylamine/Ethyl alcohol/Water (C2.577 H3.237N0.862O3.357)
M6: 84.96/9.77/2.93/0.98/0.88/0.49 NC (13.15%N)/Dinitrotoluene/Dibutylphthalate/Diphenylamine/Ethyl alcohol/Water (C2.467H3.015N0.911O3.412)
M10: 96.93/0.99/0.10/1.48/0.49 NC (13.15%N)/Diphenylamine/Graphite/ Ethyl alcohol/Water (C2.214H2.854N0.916O3.606)
M12: 89.63/7.34/0.73/1.38/0.98 NC (13.15%N)/Dinitrotoluene/Diphenylamine/Ethyl alcohol/Water (C2.309H2.922N0.927O3.522)
M14: 88.02/7.82/1.96/0.98/0.98/0.25 NC (13.15%N)/Dinitrotoluene/Dibutylphthalate Diphenylamine/Ethyl alcohol/Water (C2.406H2.940N0.918O3.456)
IMR: 89.92/7.19/0.63/1.35/0.90 NC (13.15%N)/Dinitrotoluene/Diphenylamine/Ethyl alcohol/Water (C2.301H2.912N0.927O3.527)
M2: 76.80/19.34/0.59/0.30/2.28/0.69 NC (13.25%N)/NG/Ethyl centralite/Graphite/Ethyl alcohol/Water (C2.049H2.837N0.986O3.669)
M5: 81.26/14.87/0.59/0.30/2.28/0.69 NC (13.25%N)/NG/Ethyl centralite/Graphite/Ethyl alcohol/Water (C2.085H2.855N0.970O3.655)
M7: 58.71/38.17/0.97/0.86/1.29 NC (13.15%N)/NG/Ethyl centralite/ Ethyl alcohol/Carbon black (C1.965H2.565N1.065O3.683)
M8:52.60/43.47/0.61/0.40/3.03 NC (13.25%N)/NG/Ethyl centralite/ Ethyl alcohol/Diethylphthalate (C1.911H2.609N1.075O3.710)
M9: 58.33/40.40/0.76/0.51 NC (13.25%N)/NG/Ethyl centralite/ Ethyl alcohol (C1.844H2.527N1.091O3.751)
M18: 79.60/9.95/8.96/1.00/0.50 NC (13.15%N)/NG/Dibutylphthalate/Diphenylamine/Ethyl alcohol (C2.440H3.145N0.885O3.446)
M26: 67.22/24.99/6.00/0.30/1.20/0.30 NC (13.15%N)/NG/Ethyl centralite/Graphite/Ethyl alcohol/ Water (C2.224H2.951N1.006O3.515)
T25: 73.21/19.99/5.00/0.30/1.20/0.30 NC (13.15%N)/NG/Ethyl centralite/Graphite/Ethyl alcohol/ Water (C2.223H2.890N0.989O3.532)
M15: 20.00/19.00/54.70/6.00/0.30 NC (13.15%N)/NG/NQ/Ethyl centralite/Ethyl alcohol (C1.597H3.532N2.586O2.565)
M17: 21.98/21.48/54.65/1.50/0.10/0.30/0.10 NC (13.15%N)/NG/NQ/Ethyl centralite/ Graphite/Ethyl alcohol/Water (C1.395H3.301N2.602O2.718)
T34: 20.00/19.00/54.70/4.00/2.00/0.30 NC (12.60%N)/NG/NQ/Dibutylphthalate/2-Nitrodiphenylamine/Ethyl alcohol (C1.572H3.524N2.552O2.614)
28/22.5/1.5/48 NC(12%N)/NG/Carbamite/Picrite: C1.490H3.273N2.393O2.830
28/22.5/1.5/48 NC(13.35%N)/NG/Carbamite/Picrite: C1.445H3.171N2.420O2.846
20.8/20.6/3.6/55 NC(13.35%N)/NG/Carbamite/Picrite: C1.468H3.369N2.611O2.649
28/22.5/1.5/38/10 NC(12%N)/NG/Carbamite/Picrite/RDX: C1.529H3.159N2.278O2.908
28/22.5/1.5/33/15 NC(12%N)/NG/Carbamite/Picrite/RDX: C1.548H3.102N2.221O2.947
28/22.5/1.5/28/20 NC(12%N)/NG/Carbamite/Picrite/RDX: C1.568H3.045N2.164O2.986
29.5/32/8/1/29.5 DNC/NG/DEP/2-NDPA/AP: C1.598H3.150N0.944O3.469Cl0.251
29.5/32/2/1/29.5/6 DNC/NG/DEP/2-NDPA/AP/BDNPA-F: C1.415H3.016N1.020O3.549Cl0.251
29.5/32/8/1/29.5 DNC/NG/DEP/2-NDPA/RDX: C1.996H2.942N1.490O3.261
29.5/32/2/1/29.5/6 DNC/NG/DEP/2-NDPA/RDX/BDNPA-F: C1.813H2.808N1.565O3.341
29.5/32/8/1/29.5 DNC/NG/DEP/2-NDPA/ADN: C1.598H3.096N1.644O3.416
29.5/32/8/1/29.5 DNC/NG/DEP/2-NDPA/HNF: C1.759H2.951N1.499O3.431
30/40/30 DNC/CL/TAGAZ: C1.908H3.576N2.448O2.448
80/20 RDX/GAP: C1.687H3.171N2.767O2.363
71/9/20 RDX/GAP/BTTN: C1.564H2.953N2.440O2.755
70/30 HMX/GAP: C1.854H3.406N2.800O2.194
80/20 RDX/BAMO: C1.600H2.940N2.940O2.291
70/30 HMX/BAMO: C1.725H3.059N3.059O2.085
70/30 CL-20/GAP: C1.868H2.474N2.826O2.220
80/20 CL-20/BAMO: C1.615H1.875N2.970O2.321
80/20 ADN/GAP: C0.606H3.589N3.185O2.781
75/25 ADN/GAP: C0.758H3.681N3.176O2.671
70/30 ADN/GAP: C0.909H3.772N3.166O2.560
65/35 ADN/GAP: C1.061H3.863N3.156O2.449
60/40 ADN/GAP: C1.212H3.955N3.147O2.339
50/50 ADN/GAP: C1.515H4.137N3.127O2.117
74/26 ADN/AB: C0.494H3.282N2.880O3.153
85/15 ADN/PMVT: C0.545H3.559N3.286O2.741
80/20 ADN/PVMDO: C0.588H3.756N2.972O2.972
75/25 AN/AB: C0.475H4.610N2.349O3.548
85/15 AN/PMVT: C0.545H5.066N2.669O3.186
85/15 AN/PVMDO: C0.441H5.130N2.418O3.480
60/20/20 AN/GAP/TMETN: C0.998H4.714N2.340O3.156
70/15/15 AN/GAP/TMETN: C0.749H4.785N2.380O3.304
60/15/15/10 AN/GAP/TMETN/NC(12%N): C0.976H4.578N2.216O3.290
40/15/15/30 AN/GAP/TMETN/NC(12%N): C1.431H4.165N1.887O3.262
40/15/15/30 AN/GAP/TMETN/HMX: C1.154H4.096N2.441O2.990
80/20 HNF/GAP: C1.043H3.195N2.791O2.824
80/20 HNF/PGN: C0.941H3.025N2.353O3.294
80/20 HNF/PLN: C0.925H3.648N2.347O3.272
80/20 HNF/BAMO: C0.956H2.964N2.964O2.752
80/20 HNF/HTPB: C1.852H4.315N2.197O2.666
85/15 1f/AB: C1.374H0.520N3.557O2.074
85/15 3u/AB: C1.374H0.520N3.557O2.074
85/15 3v/AB: C1.374H0.520N3.557O2.074
85/15 1e/AB: C1.134H0.520N3.687O2.140
69.70/0.6/14.79/14.91 HAN/AN/MeOH/H2O: C0.462H6.435N1.466O4.215
77.25/0.67/17.19/4.89 HAN/AN/MeOH/H2O: C0.537H5.940N1.625O4.051
72.30/0.62/11.62/15.47 HAN/AN/EtOH/H2O: C0.505H6.274N1.521O4.146
73.41/0.63/10.26/15.70 HAN/AN/1-PrOH/H2O: C0.512H6.198N1.544O4.124
63.63/0.54/22.22/13.61 HAN/AN/Glycine/H2O: C0.592H5.669N1.635O4.018
60/30/10 ADN/MAN/Urea: C0.485H4.514N2.905O3.058
40/40/20 ADN/MAN/Urea: C0.758H5.172N2.806O2.898
30/40/30 ADN/MAN/Urea: C0.925H5.516N2.817O2.742
59.86/25/15.14 H2O2(70%)/AN/EtOH: C0.657H7.680N0.625O4.727
80/8/12 H2O2(70%)/H2O/EtOH: C0.521H8.410O5.330
36.67/51.20/12.13 H2O2(70%)/ADN/EtOH: C0.527H5.962N1.651O4.034
N2O4/HEH (O/F= 1.94): C0.895H3.579N2.330O3.317
N2O4-UDMH/HEH (80/20) (O/F= 2.45): C0.927H3.713N2.471O3.161
N2O4-UDMH/HEH (90/10) (O/F= 2.55): C0.922H3.695N2.482O3.156
N2O4/UDMH (O/F=2.60): C0.927H3.707N2.496O3.139
N2O4-UDMH/HEH (60/40) (O/F= 2.32): C0.920H3.679N2.439O3.196
RFNA/UDMH (O/F=2.92): C0.850H4.559N2.070O3.516
RFNA-UDMH/HEH (90/10) (O/F= 2.85): C0.850H4.557N2.061O3.523
RFNA/HEH (O/F= 2.14): C0.837H4.408N1.954O3.637
O2/RP-1 (O/F= 2.60): C1.989H3.878O4.513
O2/N2H4 (O/F= 0.91): H6.529N3.265O2.981
O2/Toluene (O/F= 1.87): C2.644H3.021O4.075
O2/Methylcyclohexane (O/F= 2.04): C2.345H4.691O4.194
O2/n-heptane (O/F= 2.05): C2.291H5.238O4.200
O2/Ethylene oxide (O/F= 1.10): C2.157H4.313O4.360
O2/Nitroethane (O/F= 0.65): C1.615H4.037N0.807O4.077
O2/EtOH-75% (O/F= 1.30): C1.413H5.445O4.847
TNM/N2H4 (O/F= 1.40): C0.298H5.181N3.784O2.388
H2O2 (90%)/N2H4 (O/F= 1.50): H8.836N2.497O3.509
RFNA-DETA/MA (80/20) (O/F= 3.00): C0.936H4.491N1.971O3.539
RFNA/Hydine (O/F= 3.17): C0.852H4.312N2.004O3.587
N2O4/N2H4 (O/F= 1.30): H5.418N3.940O2.461
N2O4/Aerozine-50 (O/F= 2.00): C0.555H4.299N3.044O2.900
N2O4/NO (70/30)-MeOH (O/F= 2.10): C1.005H4.020N1.710O3.746
N2O4/NO (70/30)-NH3 (O/F= 2.10): H5.672N3.601O2.741
O2/HTPB (O/F=2.30): C2.144H3.227N0.019O4.424
H2O2 (90%)/PE (O/F=7.80): C0.814H7.302O5.181
H2O2 (98%)/PE (O/F=7.00): C0.893H7.023O5.140
H2O2 (98%)/DCPD (O/F=6.20): C1.051H6.415O5.058
H2O2 (86%)/HTPB (O/F= 7.50): C0.842H7.093N0.007O5.167
H2O2 (92%)/HTPB (O/F= 6.50): C0.941H6.877N0.008O5.105
RFNA/HTPB (O/F=4.90): C1.196H3.092N1.371O3.959
RFNA-HTPB/AP (90/10) (O/F=3.80): C1.324H3.296N1.326O3.850Cl0.018
N2O/Paraffin wax (O/F= 7.00): C0.890H1.816N3.976O1.988
N2O/HTPB (O/F= 7.40): C0.842H1.267N4.011O2.028
HAN(95%)/HTPB (O/F= 9.60): C0.665H5.089N1.798O3.857
Correspondence author: Dany Frem | FREM Co. | Beirut - Lebanon | E-mail: frem.dany@gmail.com












