Papers
Received: 17 June 2024
Accepted: 18 December 2024
Published: 21 January 2025
DOI: https://doi.org/10.14393/SN-v37-2025-74055
Abstract: Plastic pollution has emerged as one of our most pervasive and pressing environmental issues, impacting ecosystems, wildlife, and even human health globally. Microplastic research has primarily focused on oceans, whether in water, sediments, or organisms, generating a significant gap in understanding their presence and impact on other environments like rivers, which is a concern worldwide, and of paramount importance for us in Latin America and the Caribbean. To address this situation, we examined the current research on microplastics in South American rivers by conducting a Google Scholar search with keywords and Boolean operators, which allowed us to recover a series of articles related to this topic. We reviewed 49 articles published in 2023 to know methods for collecting and analyzing river samples. Our findings revealed limited information on microplastics in South America, with data only from Argentina, Brazil, Colombia, Ecuador, Paraguay, and Peru. Additionally, we found considerable variations in sample collection and analysis methods, hindering study comparisons. Bridging this knowledge gap is crucial for comprehending the extent of plastic pollution in the region. Since rivers are major microplastic contributors to oceans, this research will significantly aid in environmental protection efforts, emphasizing the global relevance of addressing riverine plastic pollution.
Keywords: Microplastics, Latin American rivers, Sampling methods, Laboratory methods.
INTRODUCTION
Plastic pollution is a critical global issue affecting ecosystems across terrestrial, freshwater, and marine environments (Salgado-Hernanz et al., 2021). The proliferation of plastic pollution is driven by its high production levels, durability, and persistence in the environment (Plastics Europe, 2022), compounded by the generation of microplastics (GESAMP, 2019). Over the past decade, global plastic consumption has surged, fueled by industrial sectors such as electronics, construction, and automotive (USEPA, 2021). Plastic waste accounts for 12% of municipal solid waste, with projections indicating a significant increase by 2050, particularly in low-income countries, where the situation is dire, with inadequate waste management infrastructure and low recycling rates (Kaza et al., 2018).
Addressing plastic pollution aligns with Sustainable Development Goals (SDGs: 6,11,12,13,14,15), emphasizing water quality, effective waste management, sustainable production and tourism, and ecosystem conservation. SDG14 targets plastic debris density to assess and mitigate marine pollution, underscoring the importance of effective plastic waste management. Plastic pollution poses significant hazards in aquatic ecosystems due to slow degradation, release of toxic compounds, and capacity to carry additional pollutants and pathogens, exacerbating environmental impacts (Liu et al., 2020). Plastics undergo degradation influenced by environmental factors like heat, sunlight, wind, and waves, leading to fragmentation, photochemical degradation, biological fouling, and aggregation, resulting in progressively smaller particles, including microplastics and nanoplastics (Corcoran, 2022).
Microplastics (MPs), defined as plastic particles smaller than 5 mm, and prevalent in the environment, originate from various sources, including plastic pellet industries, macroplastic fragmentation, and synthetic fibers from textiles, the latter entering aquatic ecosystems through laundering and wastewater treatment plants, these being ineffective in removing them (de Jesus Silva et al., 2024). Nanoplastics ranging from 1 to 1000 nm, resulting from the fragmentation of both macro and microplastics (Gigault et al., 2018), raise concerns due to their colloidal properties and potential for greater harm relative to microplastics, as they can infiltrate biological organisms and accumulate within marine food chains (Cai et al., 2021).
MPs are classified into primary and secondary categories based on their origin, with land-based sources contributing significantly to plastic pollution in nearshore environments (GESAMP, 2019). River basin urban areas are primary sources of MPs, and major vectors of microplastics into marine environments (Lebreton et al., 2017), with proximity to the source, water flow speed, and particle size influencing particle spread (de Faria et al., 2021). Since California’s 2011 initial report (Moore et al., 2011) to the recent studies in various locations including Tibet (Jiang et al., 2019), Taiwan (Kunz et al., 2023), China (Wu et al., 2023; Liu et al., 2023), and Bangladesh (Parvin et al., 2022), microplastic concentrations have been reported worldwide.
China and the United States have the highest number of published microplastic studies (Davtalab et al. 2023). A brief Google Scholar review of the literature on microplastics in various environmental compartments (Marine, Freshwater, Wastewater, and Soils) reveals an interesting trend. Before 2010, there were few studies on this topic. However, starting in 2011, there was a noticeable surge in interest in microplastics, especially in marine environments. From 2021 to 2023, the number of documents across all these compartments reached similar figures, with approximately 17,000 to 18,000 articles (until October 2023). This highlights the growing interest and concern within the scientific community regarding plastic pollution.
Despite emerging research on MPs in South American marine environments, with studies indicating a higher prevalence of plastic particles in coastal systems of Ecuador, Peru, Brazil, Uruguay, and Argentina (Truchet et al., 2022), significant gaps exist in understanding the distribution and abundance of microplastics in Latin American and Caribbean rivers. This review aims to summarize recent studies in these regions, assessing regional research efforts and exploring global trends in microplastic analysis techniques and sampling methodologies in 2023.
METHODOLOGY
Latin American and Caribbean Research Review
A Google Scholar (GS) literature review was conducted using Boolean operators. Quotation marks were employed to refine the search and results. The specific query was "microplastics in rivers." The Boolean operator "AND" was used, followed by the target country in quotation marks ("microplastics in rivers" AND "country"). Since the study focuses on Latin America and the Caribbean, we reviewed research from 33 countries: 12 in South America, eight in Central America (including Mexico), and 13 in the Caribbean (Table 1), searching in English, Spanish, and Portuguese to assess the regional research landscape, with a focus on South America.
The initial results were refined by excluding citations, patents, reviews, theses, reports, documents lacking microplastic content or distribution results, marine studies, duplicates, articles from unlisted countries or other continents, and those in different languages. Despite using specific keywords, some irrelevant results appeared. Articles meeting the criteria were reviewed in depth, focusing on methodology, especially sampling and laboratory tests.
Countries included in this search.

Source: The authors (2024).
The search yielded 314 articles: 19 from Central America, 29 from the Caribbean and The Bahamas, and 266 from South America. Occasionally, there were no results for the three languages. Several articles did not contain keywords, and some were non-research articles, or merely mentioned in the grey literature, necessitating a more meticulous review. A more thorough examination allowed for a selection of the information obtained, reducing the number of articles developed in South America to nine (Figure 1). Further verification ensured that they involved collecting and processing water or river sediment samples.
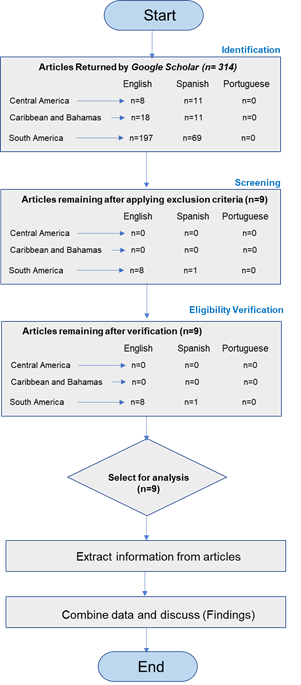
Figure 1
Review workflow and results of the first step.
Source: The authors (2024).
This selection was thoroughly reviewed, considering: the year of publication, country/locality, sample unit, sample collection, laboratory analysis, reported abundance, and polymer determination.
MPs Detection Methods in Rivers (2023)
To research global trends in river water sampling and microplastics analysis, a GS search was conducted using the phrases "microplastics abundance" AND "rivers" excluding citations and patents and selecting only works published in 2023 (from September to December 2023), yielding 224 pre-selected articles. A subsequent review eliminated those lacking the keywords in the title or abstract provided by the search engine, as well as books, reports, repositories, and reviews. Articles exclusively analyzing microplastics in marine environments or determining their presence in organisms were also discarded.
RESULTS AND DISCUSSION
The distribution of articles by country of Latin America and the Caribbean is shown in Figure 2a, highlighting the scarcity of information in Central America and the Caribbean, with emerging interest in South America, particularly Ecuador. The articles for the region began appearing in 2020 with increased production in 2021 and 2023 (Figure 2b), emphasizing water sample analysis (34%) and studies analyzing both water and sediments (33%) (Figure 2C).
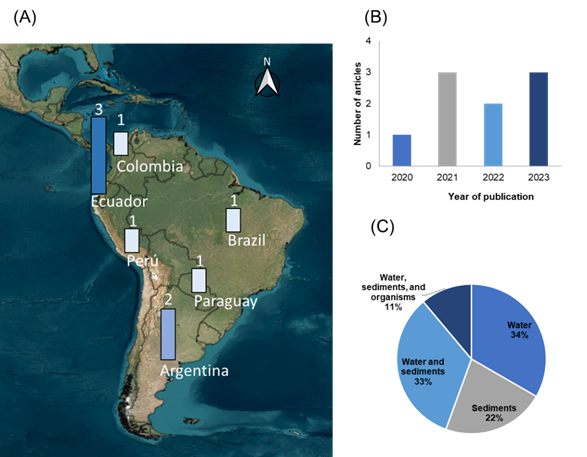
Figure 2
Distribution of articles according to (A) country, (B) year of publication, (C) sample unit.
Source: The authors (2024).
Water sample collection
The review focused on water sample studies, resulting in a detailed analysis of seven articles. Manual collection using glass bottles of varying capacities was the most common method, though plankton and Nansen nets were also reported. Only two studies reported preserving samples via refrigeration during transport and before laboratory testing (Table 2).
Kinds of water sampling and preservation conditions.
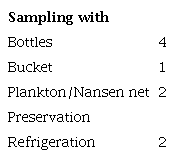
Source: The authors (2024).
Laboratory analysis
These seven articles show notable differences in laboratory procedures for microplastic extraction, identification, and classification, ranging from four to eight steps (Table 3). A key disparity is the absence of chemical analyses in three studies. The primary technique for polymer identification was Fourier transform infrared spectroscopy (FTIR), often supplemented by scanning electron microscopy (SEM) with energy-dispersive X-ray spectroscopy (EDS) and X-ray diffraction (XRD). Some studies used multiple techniques to gather chemical information on plastic particles. Discrepancies in chemical analysis choices likely stem from financial constraints or the need to outsource analyses. Though particle classification methods varied, all papers used stereo microscopy of varying magnification levels to characterize properties like color, size, and shape.
Further disparities are evident in the sequencing and execution of certain steps, including the method of filtration, the treatment of organic matter that involved reagents, digestion temperature, and the application of density separation, not universally implemented across all investigations. Additionally, the material obtained from filtration underwent drying or sieving at varying temperatures, although room temperature was the prevailing choice. It is important to underscore that the laboratory phase is intricately linked to the resources available to researchers, equipment, materials, reagents, and their familiarity with assays adaptable to microplastic study.
Laboratory steps for analysis of microplastics in river waters.

Source: The authors (2024)
Quality and contamination control
Quality assurance and contamination management are crucial in microplastic studies due to their ubiquity in environments like tap water. However, three of the articles reviewed lack quality control measures. Before sample collection, sampling materials and instruments were washed and prepared, including rinsing with local water and washing glassware and metalware with filtered deionized water or 70% methanol. Reagents were prepared using distilled/deionized water and filtered, with materials and samples covered in aluminum foil to prevent contamination. Sampling bottles were rinsed three times before use, and cotton clothing, lab coats, and face masks were used to avoid textile contamination.
Findings summary of selected papers
Table 4 summarizes some of the findings reported in this review. The contents vary depending on the characteristics of each body of water evaluated and its features (dimensions, morphology, others), the proximity and characteristics of population centers (urban or rural areas), potential sources of contamination, specifically the influx of untreated wastewater (Forero-López et al., 2021), and even the sampling and analysis conditions in terms of the size limits of the particles to be evaluated.
These methods did not follow the same protocols due to the lack of a unified convention as in the case of marine environments. Although such protocols do not exist, and this complicates comparisons between studies conducted around the world, making them can be helpful to have a reference of the pollution status in different locations.
The highest abundance of microplastics was reported in Argentina with an average of 6162 items/m3 (Forero-López et al., 2021), followed by Colombia, 3704.64 items/m3 (Chavés-Velasco et al., 2023). Fibers and fragments are the most frequently reported shapes in South American rivers, with transparent plastic particles prevailing. The polymers found included cellulose (CE), polyethylene (PE), polypropylene (PP), polyethylene terephthalate (PET), polyamide (PA), polyacrylonitrile (PAN), polyvinyl chloride (PVC), and alkyd resins (AR).
Some reported findings in South America.

Source: The authors (2024).
As observed, there is a growing interest within the Latin American scientific community in tackling this research topic, with notable findings reported in Ecuador, which has the highest number of publications.
However, while the reviewed articles demonstrate a rigorous approach in sample collection, pretreatments, laboratory assays including chemical characterization or identification, and quality control measures, there are differences in the methods employed to report microplastic abundance in river water samples. These discrepancies pose challenges that must be addressed to advance research on plastic pollution and contribute to potential solutions.
2023 River MPs detection methods
This review on GS was carried out between September and December 2023, allowing the final selection of 49 articles after the exclusion criteria. Figure 3 illustrates the sifting process for the articles subsequently reread to know the methodological details applied. Figure 4 illustrates the geographical distribution of the articles returned by the search, observing that most of these were developed in Asia (80%), followed in equal percentage by the American, European, and African continents (6%), and the lowest, Oceania (2%).
Eighty percent of the studies analyzed river waters, either alone (20 articles) or with sediment samples (19 articles), while 20% focused solely on sediments (10 articles). Furthermore, all primarily emphasized the spatial factor, with only 60% examining the temporal or seasonal influence on riverine microplastic content. Subsequently, a detailed review highlighted discrepancies in sample collection and laboratory analyses.
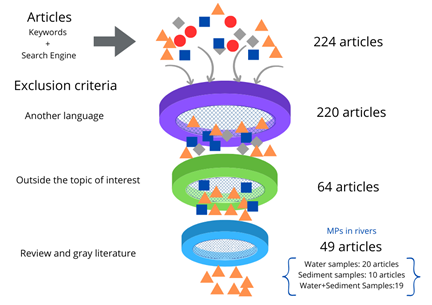
Figure 3
The sifting process applied to the results from Google Scholar.
Source: The authors (2024).
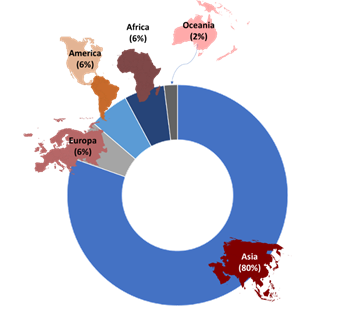
Figure 4
Geographical distribution of articles selected.
Source: The authors (2024).
Details of sampling methods
Regarding sampling methods, an examination of the procedures for collecting river water samples revealed various techniques, both manual and automatic methods (pumping and ultrafiltration). Manual water collection is the most common, including the use of buckets (20%), glass bottles (15%), or stainless-steel water samplers (12%). Interestingly, some articles reference nets traditionally used for sampling marine organisms (27%; Figure 5). However, the amount of water collected is inconsistent across studies, leading to high variability in volume.
Some authors report collecting 500 mL (Alam et al., 2023), while others mention volumes of 1L (Qaiser et al., 2023), 2L (Ragoobur et al., 2023), or even 5L (Wu et al., 2023; Karing et al., 2023), with one study even referring to the collection of 5L to complete a total of 25L (Shen et al., 2023). Additionally, it is important to note that all samples were collected at a surface level between 0.0-0.3m, as indicated in studies reporting sampling depth.
Details of Laboratory Assays of Water Samples
Twenty-six of 39 articles reported sieving or filtering (67%) either during water sample collection or laboratory analysis, using stainless steel sieves with different mesh sizes and filter paper (e.g., glass microfiber, pretreatment), regardless of the collection method. Both techniques are referenced in the European JPI Oceans protocol (Gago et al., 2018) and the NOAA manual (Masura et al., 2015), subsequently, the samples underwent drying. Of the 26 studies that applied initial water sample filtration, only 14 mentioned the drying process lasting between 30 minutes and 72 hours at an ambient temperature of 105°C.
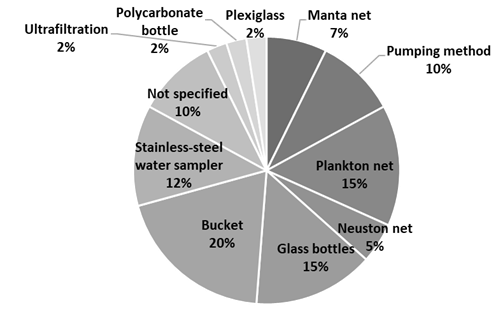
Figure 5
Distribution of the sampling methods used.
Source: The authors (2024).
Organic matter (OM) removal was documented in 28 of the 39 articles (72%). Among these, 30% hydrogen peroxide (H2O2) emerged as the predominant reagent for OM degradation, utilized in 18 articles (64.29%), with reaction times from 12 hours to 7 days and digestion temperature between 25 °C and 70 °C. Some studies used Fenton reagent (six articles, 21.43%) or KOH (14.29%).
Following OM degradation, filtration was employed in 11 articles with pore sizes from 1μm to 200μm, the most common being 0.45mm. Filter materials included glass microfiber, steel, Whatman paper, and nitrocellulose. Density separation was employed in 19 articles to isolate plastics, with sodium chloride or zinc chloride as the most frequently used reagents (Table 5).
Filtration post-density separation was performed in 13 articles (33.4%) and drying was reported after OM removal or post-filtration in eight articles (20.5%). After obtaining the solid residue, plastic particles were visually classified regardless of the preceding steps, with 12 articles using stereomicroscopes or microscopes. Notably, OM was removed in one of the studies after density separation (Xu et al., 2023), optimizing reagent use and waste generation.
Reagents used for density separation of MPs

1 Health hazard retrieved from NFPA/HMIS forms and toxicity values retrieved from MSDS forms. 2 Reference prices consulted online. These may vary according to brand, country, etc. Source: The authors (2024).
Plastic particles characterization
To observe, separate, and characterize the plastic particles found in the samples, 31 articles used microscopes/stereoscopes with magnifications from 5x to 200x, and only one used direct visualization. The choice of devices and magnification levels depended on equipment availability and particle size under study. In some cases, a microscope with a digital camera (Priyanka et al., 2023) or an electron microscope (Shi et al. 2022) was used. Particle classification primarily considered shape (26 articles), color (24 articles), and size (17 articles).
Shapes and colors
The criteria for classifying particles by shape and color varied, with categories ranging from 3 to 7 (Figure 6a) and different names for particle shapes. Common shape descriptors, summarized in Figure 6b, include fragments, fibers, films, and foams, while less frequently used terms include lumps, frags, cylinders, and flakes.
MPs are heterogeneous and exhibit a range of morphologies that can provide some indication of potential sources and their environmental behavior (GESAMP, 2019). Until now there has been no standardized scheme for their morphological characterization, Table 6 provides an overview of the shapes employed by various authors to describe MPs in the environment. There are discernible differences in the approaches used, which coincides with the findings of this review of determinations made in rivers. In this sense, establishing a unified classification criterion is imperative to facilitate meaningful comparisons across studies.
Summary of shapes used to characterize microplastic in the environment.

Source: The authors (2024).
Guidelines like those from GESAMP (2019) classify marine plastic litter. For instance, fragments are irregular particles broken from larger plastics, while the foam is near-spherical or granular, deformable under pressure, and partially elastic. These definitions help outline plastic particle properties. Color also characterizes microplastics, aiding in their separation from debris-filled matrices. Distinctive hues facilitate isolation and photodegradation, sea surface residence time, and the extent of tarring or weathering (Zhao et al., 2022).
Simple categories such as colored and noncolored have been used to refer to the appearance of MPs (Mohsen et al., 2023). However, most studies report the specific colors of recovered MPs. Our review indicates that a minimum of two and a maximum of fourteen colors have been used, including transparent, white, black, yellow, green, blue, red, orange, violet, and others, highlighting that in most cases the authors used between six and ten colors to describe the properties of MPs (Figure 7). Five reviewed articles have the term "other" to simplify the description.
The colors of MPs in the environment depend on the larger plastics they stem from, the time of exposure to the environment, and consumption preferences, among other factors. It is advisable to report the hues associated with the specific systems being analyzed and consider the larger plastic litter present in the surroundings, which could indicate potential sources of MPs.
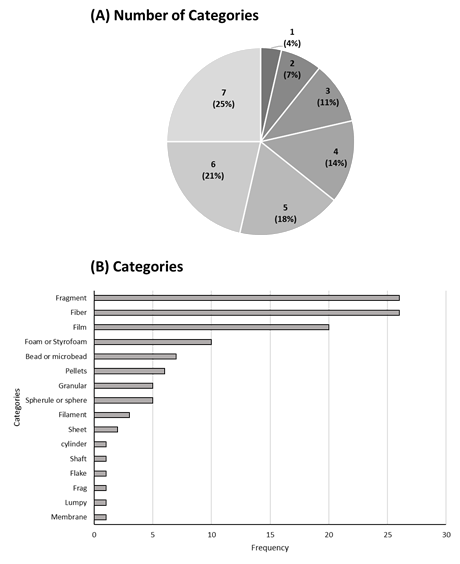
Figure 6
Categories of microplastic in rivers: (A) Number of categories and (B) Frequency of use.
Source: The authors (2024).
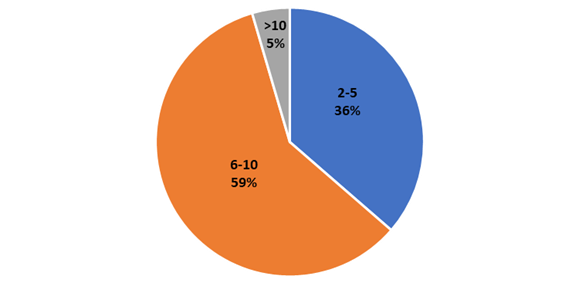
Figure 7
Number of colors used in microplastics description.
Source: The authors (2024).
Size
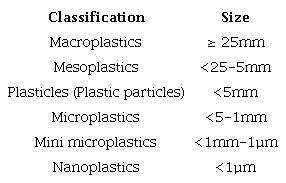
Size is a crucial criterion in the articles reviewed, with most addressing various size fractions. These fractions range from three (<0.50mm, 0.50-1.00mm, 1.00-5.00mm or, <0.25 mm, 0.25-0.50mm, 0.50-1.00mm) to six fractions (<0.50mm, 0.50-1mm, 1-2mm, 2-3mm, 3-4mm, 4-5mm). While MPs are commonly defined as particles smaller than 5mm, no internationally agreed definition exists.
GESAMP (2019) proposed a robust classification system to harmonize monitoring approaches across varying applications, depending on the research focus. However, other size ranges can also be used. Table 7 presents a classification encompassing particles ranging from macroplastics (>25mm) to nanoplastics (<1µm). The reviewed articles typically used size ranges from <0.3mm to >5mm.
Polymer Identification
Thirty articles employed chemical identification techniques to characterize MPs (Table 8). Overall, the polymer identification methods used include FTIR and Raman spectroscopy. These techniques offer insights into plastic weathering and rely on molecular vibration resulting from the incidence of a light source, typically either laser or IR radiation (Rathore et al., 2023).
Raman is an advanced method of chemical analysis used for material characterization and µRaman, a variant, utilizes an optical microscope system to focus the laser beam on a specific area of the sample, enabling the analysis of samples at a minute scale, even in the micrometer range, which is particularly advantageous to detect and characterize MPs in the environment (Medina Faull et al., 2021). However, due to the high analysis expense, some authors selected only a representative portion (Manbohi et al., 2023).
Polymer identification methods used in the selected database.
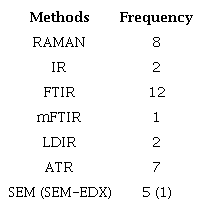
Source: The authors (2024).
IR spectra such as FTIR and mFTIR were the tools most widely used to characterize MPs in river waters (15 articles). Additionally, Attenuated Total Reflectance Analysis (ATR) was used in seven articles, enhancing microplastic identification without special sample preparation and allowing spectra comparison with equipment libraries. Two articles reported using a laser-based direct infrared chemical imaging system (LDIR), an advanced technique combining microscopy and infrared spectroscopy to map chemical composition. Besides polymer identification, studies also used scanning electron microscopy (SEM) to examine MPs surfaces, revealing cracks, polymer aging, and mechanical and oxidative weathering information.
Table 9 depicts the polymers most identified in the 2023 river water studies. Those most frequently mentioned were PP, PE, polystyrene (PS), PA, PET, and PVC, which align with findings in other studies, according to Geyer et al. (2017), who indicated that common polymers worldwide follow the trend: PE>PP>PVC>PET>PS.
Main polymers reported in river waters.
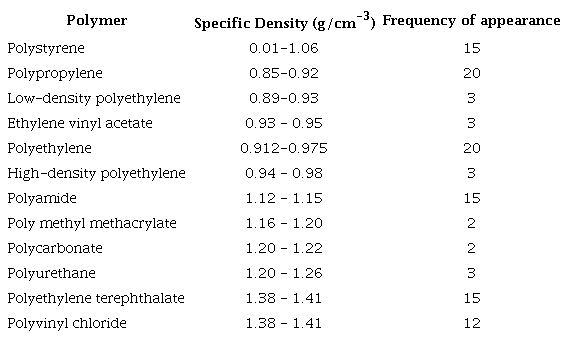
Source: The authors (2024).
The findings presented in this section contribute to our understanding of the current research on microplastics in river ecosystems. They provide valuable insights into the diverse sampling methodologies, laboratory analyses, and characterization techniques employed across studies worldwide. The variations observed in sampling methods and procedures underscore the complexity of the field.
Moreover, they emphasize the pressing need for standardization or the adoption of generic methods to ensure the reliability and comparability of data. Additionally, the diverse approaches to classifying MPs by shape, color, and size underscore the intricate nature of this research. Establishing unified criteria in these aspects will undoubtedly be pivotal for guiding future research endeavors to comprehensively understand and effectively mitigate the impact of microplastic pollution on river ecosystems.
FINAL CONSIDERATIONS
Microplastics research has predominantly focused on oceans, leaving a significant gap in understanding their presence in rivers, especially in Latin America and the Caribbean, which have limited data from six countries with an emerging interest in South America, particularly Ecuador.
Detailed analysis provided insights into geographical distribution, temporal trends, sample collection methodologies, laboratory techniques, and findings. There are disparities in sample collection, pretreatment, and lab procedures, highlighting the need for standardization to ensure data reliability, despite adaptability to geographical conditions or research groups.
The laboratory process generally in river water included filtration, organic matter removal, and density separation, and the classification criteria often focused on size, color, and shape, with polymer identification limited by equipment availability and costs.
Our synthesis provides valuable insights into microplastic pollution in South American river ecosystems, emphasizing the need for further research and coordinated efforts. Enhancing understanding of this pollution in freshwater environments will help develop effective management and rehabilitation strategies to protect aquatic ecosystems and human health.
REFERENCES
ALAM, M. J.; SHAMMI, M.; TAREQ, S. M. Distribution of microplastics in shoreline water and sediment of the Ganges River Basin to Meghna Estuary in Bangladesh. Ecotoxicology and Environmental Safety, v. 266, p. 115537, 2023. https://doi.org/10.1016/j.ecoenv.2023.115537
BLAIR-CRAWFORD, C.; QUINN, B. Microplastic pollutants. Elsevier, 2016.
CAI, H.; XU, E. G.; DU, F.; LI, R.; LIU, J.; SHI, H. Analysis of environmental nanoplastics: Progress and challenges. Chemical Engineering Journal, v. 410, p. 128208, 2021. https://doi.org/10.1016/j.cej.2020.128208
CAPPARELLI, M. V.; MOLINERO, J.; MOULATLET, G. M.; BARRADO, M.; PRADO-ALCÍVAR, S.; CABRERA, M.; GIMILIANI, G.; ÑACATO, C.; PINOS-VELEZ, V.; CIPRIANI-AVILA, I. Microplastics in rivers and coastal waters of the province of Esmeraldas, Ecuador. Marine Pollution Bulletin, v. 173, p. 113067, 2021. https://doi.org/10.1016/j.marpolbul.2021.113067
CHÁVES VELASCO, J. J.; ARIAS HOYOS, A.; MUÑOZ SOLARTE, D. M.; ROMERO PUENTES, R. B. Estudio de microplásticos en muestras de agua y sedimentos de un río urbano del suroccidente de Colombia. Investigación e Innovación en Ingenierías, v. 11, n.2, p. 120-130, 2023. https://doi.org/10.17081/invinno.11.2.6637
CORCORAN, P. L. Degradation of microplastics in the environment. Handbook of Microplastics in the Environment Cham: Springer International Publishing, 2022. https://doi.org/10.1007/978-3-030-39041-9_10
DAVTALAB, M.; BYČENKIENĖ, S.; UOGINTĖ, I. Global research hotspots and trends on microplastics: a bibliometric analysis. Environmental Science and Pollution Research, v. 30, p. 107403-107418, 2023. https://doi.org/10.1007/s11356-023-27647-1
DE FARIA, E.; GIRARD, P.; NARDES, C. S.; MORESCHI, A.; CHRISTO, S. W.; JUNIOR, A. L. F.; COSTA, M. F. Microplastics pollution in the south American Pantanal. Case Studies in Chemical and Environmental Engineering, v. 3, p. 100088, 2021. https://doi.org/10.1016/j.cscee.2021.100088
DE JESUS SILVA, A. J.; DA SILVA, K. C. B.; TANANTA, T. M. M.; DOS SANTOS, E. O.; DE OLIVEIRA, E. S. Ocorrência de microplásticos na orla do rio Javari, em Benjamin Constant, interior do Amazonas, Brasil. Journal of Education Science and Health, v. 4, n. 1, p. 1-20, 2024. https://doi.org/10.52832/jesh.v4i1.237
DONOSO, J. M.; RIOS-TOUMA, B. Microplastics in tropical Andean rivers: A perspective from a highly populated Ecuadorian basin without wastewater treatment. Heliyon, v. 6, n.7, p. e04302, 2020. https://doi.org/10.1016/j.heliyon.2020.e04302
FORERO-LÓPEZ, A. D.; RIMONDINO, G. N.; TRUCHET, D. M.; COLOMBO, C. V.; BUZZI, N. S.; MALANCA, F. E.; SPETTER, CV; FERNÁNDEZ-SEVERINI, M. D. Occurrence, distribution, and characterization of suspended microplastics in a highly impacted estuarine wetland in Argentina. Science of the Total Environment, v. 785, p. 147141, 2021. https://doi.org/10.1016/j.scitotenv.2021.147141
FRIAS, J.; FILGUEIRAS, A.; GAGO, J.; PEDROTTI, M. L.; SUARIA, G.; TIRELLI, V.; GERDTS, G. Standardised protocol for monitoring microplastics in seawater. Deliverable 4.2, 2019.
GAGO, J.; FILGUEIRAS, A.; PEDROTTI, M. L.; CAETANO, M.; FRIAS, J. Standardised protocol for monitoring microplastics in seawater. Deliverable 4.1, 2018.
GESAMP. Guidelines or the monitoring and assessment of plastic litter and microplastics in the ocean. Rep. GESAMP, n. 99, p. 130, 2019. Available: http://www.gesamp.org/publications/guidelines-for-the-monitoring-and-assessment-of-plastic-litter-in-the-ocean. Accessed on: may. 08, 2024.
GEYER, R.; JAMBECK, J. R.; LAW, K. L. Production, use, and fate of all plastics ever made. Science advances, v. 3, n. 7, p. e1700782, 2017. https://doi.org/10.1126/sciadv.1700782
GIGAULT, J.; TER HALLE, A.; BAUDRIMONT, M.; PASCAL, P. Y.; GAUFFRE, F.; PHI, T. L.; EL HADRI, H.; GRASSL, B.; REYNAUD, S. Current opinion: what is a nanoplastic? Environmental pollution, v. 235, p. 1030-1034, 2018. https://doi.org/10.1016/j.envpol.2018.01.024
HIDALGO-RUZ, V.; GUTOW, L.; THOMPSON, R.; THIEL, M. Microplastics in the Marine Environment: A Review of the Methods Used for Identification and Quantification, Environment Science and Technology, v. 46. n.6, p. 3060-3075, 2012. https://doi.org/10.1021/es2031505
JIANG, C.; YIN, L.; LI, Z.; WEN, X.; LUO, X.; HU, S.; YANG, H.; LONG, Y.; DENG, B.; HUANG, L.; LIU, Y. Microplastic pollution in the rivers of the Tibet Plateau. Environmental Pollution, v. 249, p. 91-98, 2019. https://doi.org/10.1016/j.envpol.2019.03.022
KARING, D. J.; ANGGIANI, M.; CAO, L. T.; EL-SHAAMMARI, M. Occurrence of Microplastics in Kemena River and Niah River of Sarawak, Malaysia. Tropical Environment, Biology, and Technology, v. 1, n. 1, p. 1-13, 2023. https://doi.org/10.53623/tebt.v1i1.220
KAZA, S.; YAO, L.; BHADA-TATA, P.; VAN WOERDEN, F. What a waste 2.0: a global snapshot of solid waste management to 2050. World Bank Publications, 2018. https://doi.org/10.1596/978-1-4648-1329-0
KUNZ, A.; SCHNEIDER, F.; ANTHONY, N.; LIN, H. T. Microplastics in rivers along an urban-rural gradient in an urban agglomeration: Correlation with land use, potential sources and pathways. Environmental Pollution, v. 321, p. 121096, 2023. https://doi.org/10.1016/j.envpol.2023.121096
LEBRETON, L.; VAN DER ZWET, J.; DAMSTEEG, J.; SLAT, B.; ANDRADY, A.; REISSER, J. River plastic emissions to the world’s oceans. Nature Communication, v. 8, p. 15611, 2017. https://doi.org/ 10.1038/ncomms15611
LEÓN-MUEZ, D.; PEÑALVER DUQUE, P.; FRANCO FUENTES, E.; BENFATTI, E.; COMES AGUILAR, L.; CIUDAD TRILLA, C.; MUÑOZ, M.; GÜEMES, S.; FERNANDO DE FUENTES, A. Protocolo para la planificación, muestreo, análisis e identificación de microplásticos en ríos. ProyectoLibera, p. 1-66, 2020. [Online]. Available: https://proyectolibera.org/storage/recursos/protocolo-muestreo-analisis-microplasticos-rios-proyecto-libera-hyt-web.pdf. Accessed on: may. 15, 2024.
LIU, P.; ZHAN, X.; WU, X.; LI, J.; WANG, H.; GAO, S. Effect of weathering on environmental behavior of microplastics: Properties, sorption and potential risks. Chemosphere, v. 242, p. 125193, 2020. https://doi.org/10.1016/j.chemosphere.2019.125193
LIU, Q.; XIONG, X.; WANG, K.; WANG, H.; LING, Y.; LI, Q.; XU, F.; WU, C. Homogenization of microplastics in alpine rivers: Analysis of microplastic abundance and characteristics in rivers of Qilian Mountain, China. Journal of Environmental Management, v. 340, p. 118011, 2023. https://doi.org/10.1016/j.jenvman.2023.118011.
MANBOHI, A.; MEHDINIA, A.; RAHNAMA, R.; DEHBANDI, R. Microplastic pollution in inshore and offshore surface waters of the southern Caspian Sea. Chemosphere, V. 281, p. 130896, 2021. https://doi.org/10.1016/j.chemosphere.2021.130896
MASURA, J.; BAKER, J.; FOSTER, G.; COURTNEY, A. Laboratory methods for the analysis of microplastics in the marine environment: recommendations for quantifying synthetic particles in waters and sediments. NOAA Technical Memorandum, 2015. http://dx.doi.org/10.25607/OBP-604
MEDINA FAULL, L. E.; ZALIZNYAK, T.; TAYLOR, G. T. Assessing diversity, abundance, and mass of microplastics (~1-300μm) in aquatic systems. Limnology and Oceanography: Methods, v. 19, n. 6, p. 369-384, 2021. https://doi.org/10.3389/fmars.2020.00360
MOHSEN, A.; BALLA, A.; KISS, T. High spatiotemporal resolution analysis on suspended sediment and microplastic transport of a lowland river. Science of The Total Environment, v. 902, p. 166188, 2023. https://doi.org/10.1016/j.scitotenv.2023.166188.
MOORE, C. J.; LATTIN, G. L.; ZELLERS, A. F. Quantity and type of plastic debris flowing from two urban rivers to coastal waters and beaches of Southern California. Revista de Gestão Costeira Integrada, v. 11, n. 1, p. 65-73, 2011. https://doi.org/10.5894/rgci194
PANIZZON, J.; DE LIMA, A. C.; DE OLIVEIRA, D. D. L.; FÜRH, K. T.; GEHLEN, G.; SOBRAL, P.; JAHNO, V. D. Microplastics in the Sinos River, Southern Brazil. SSRN, 2022. http://dx.doi.org/10.2139/ssrn.4061624
PARVIN, F.; HASSAN, M. A.; TAREQ, S. M. Risk assessment of microplastic pollution in urban lakes and peripheral Rivers of Dhaka, Bangladesh. Journal of Hazardous Materials Advances, v. 8, p. 100187, 2022. https://doi.org/10.1016/j.hazadv.2022.100187
PLASTICS EUROPE. Plastics-The Facts 2022. Belgium: Plastics Europe, 2022. Available: https://plasticseurope.org/wp-content/uploads/2022/10/PE-PLASTICS-THE-FACTS_V7-Tue_19-10-1.pdf. Accessed on: jun. 16, 2024.
PRIYANKA, R.; GOVINDARAJULU, B. Transportation of microplastics from the rural area into the urban area of the Kosasthalaiyar River in the metropolitan city of Chennai. Regional Studies in Marine Science, v. 66, p. 103151, 2023. https://doi.org/10.1016/j.rsma.2023.103151
QAISER, N.; SIDRA, S.; JAVID, A.; IQBAL, A.; AMJAD, M.; AZMAT, H.; AROOJ, F.; FAROOQ, K.; NIMRA, A.; ALI, Z. Microplastics abundance in abiotic and biotic components along aquatic food chain in two freshwater ecosystems of Pakistan. Chemosphere, v.313, p.137177, 2023. https://doi.org/10.1016/j.chemosphere.2022.137177
RAGOOBUR, D.; AMODE, N. S.; SOMAROO, G. D.; NAZURALLY, N. Microplastics in estuarine water and sediment in Mauritius. Regional Studies in Marine Science, v. 57, p. 102766, 2023. https://doi.org/10.1016/j.rsma.2022.102766
RATHORE, C.; SAHA, M.; GUPTA, P.; KUMAR, M.; NAIK, A.; DE BOER, J. Standardization of micro-FTIR methods and applicability for the detection and identification of microplastics in environmental matrices. Science of the Total Environment, v. 888, p. 164157, 2023. https://doi.org/10.1016/j.scitotenv.2023.164157
SALGADO-HERNANZ, P. M.; BAUZÀ, J.; ALOMAR, C.; COMPA, M.; ROMERO, L.; DEUDERO, S. Assessment of marine litter through remote sensing: recent approaches and future goals. Marine Pollution Bulletin, v. 168, p. 112347, 2021. https://doi.org/10.1016/j.marpolbul.2021.112347
SHEN, J.; GU, X.; LIU, R.; FENG, H.; LI, D.; LIU, Y.; JIANG, X.; QIN, G.; AN, S.; LI, N.; LENG, X. Damming has changed the migration process of microplastics and increased the pollution risk in the reservoirs in the Shaying River Basin. Journal of Hazardous Materials, v. 443, p. 130067, 2023. https://doi.org/10.1016/j.jhazmat.2022.130067
SHI, B.; PATEL, M.; YU, D.; YAN, J.; LI, Z.; PETRIW, D.; PRUYN, T.; SMYTH, K.; PASSEPORT, E.; DWAYNE MILLER, R. J.; HOWE, J. Y. Automatic quantification and classification of microplastics in scanning electron micrographs via deep learning. Science of The Total Environment, v. 825, p. 153903, 2022.
DE JESUS SILVA, A. J.; DA SILVA, K. C. B.; TANANTA, T. M. M.; DOS SANTOS, E. O.; DE OLIVEIRA, E. S. Ocorrência de microplásticos na orla do rio Javari, em Benjamin Constant, interior do Amazonas, Brasil. Journal of Education Science and Health, v. 4, n. 1, p. 1-20, 2024. https://doi.org/10.52832/jesh.v4i1.237
SOOKSAWAT, T.; WATTANAKORNSIRI, A.; KOHKAEW, R.; PAGE, L. M.; TONGNUNUI, S. Microplastic accumulation in local dominant shellfish from the Khwae Noi Basin in Western Thailand and its environmental factors. ScienceAsia, v. 49, n. 3, p. 445-453, 2023. https://doi.org/10.2306/scienceasia1513-1874.2023.030
TRUCHET, D. M.; ARDUSSO, M. G.; FORERO-LÓPEZ, A. D.; RIMONDINO, G. N.; BUZZI, N. S.; MALANCA, F.; SPETTER, C. V.; FERNÁNDEZ-SEVERINI, M. D. Tracking synthetic microdebris contamination in a highly urbanized estuary through crabs as sentinel species: An ecological trait-based approach. Science of The Total Environment, v. 837, p. 155631, 2022. https://doi.org/10.1016/j.scitotenv.2022.155631
USEPA. The national recycling strategy is part one of a series on building a circular economy for all, 2021 Available: https://www.epa.gov/system/files/documents/2021-11/final-national-recycling-strategy.pdf. Accessed on: dec. 20, 2024.
WU, J.; YE, Q.; SUN, L.; LIU, J.; HUANG, M.; WANG, T.; WU, P.; ZHU, N. Impact of persistent rain on microplastics distribution and plastisphere community: A field study in the Pearl River, China. Science of The Total Environment, v. 879, p. 163066, 2023. https://doi.org/10.1016/j.scitotenv.2023.163066
XU, N.; JIANG, L.; ZHANG, Y.; SHEN, Y.; WANG, Y.; WANG, S.; YUAN, Q. Microplastic pollution in the offshore sea, rivers and wastewater treatment plants in Jiangsu coastal area in China. Marine Environmental Research, v. 188, p. 105992, 2023. https://doi.org/10.1016/j.marenvres.2023.105992
ZHANG, C.; WANG, S.; SUN, D.; PAN, Z.; ZOU, J. Investigation of Microplastics in Surface Water and Estuarine Mullet Mugil cephalus from 23 Estuary Areas, South China. Sustainability, v. 15 n. 5, p. 4193, 2023. https://doi.org/10.3390/su15054193
ZHAO, X.; WANG, J.; YEE LEUNG, K. M.; WU, F. Color: an important but overlooked factor for plastic photoaging and microplastic formation. Environmental Science & Technology, v. 56, n. 13, p. 9161-9163, 2022. https://doi.org/10.1021/acs.est.2c02402