Artículos
Alfa and beta diversity of thrips (Thysanoptera) and biological control agents in chrysanthemum flowers (Chrysanthemum sp.) of two ecoregions in Jujuy province, Argentina
Diversidad alfa y beta de trips (Thysanoptera) y agentes de control biológico en flores de crisantemo (Chrysanthemum sp.) de dos ecorregiones de la provincia de Jujuy, Argentina
Alfa and beta diversity of thrips (Thysanoptera) and biological control agents in chrysanthemum flowers (Chrysanthemum sp.) of two ecoregions in Jujuy province, Argentina
Revista de la Sociedad Entomológica Argentina, vol. 80, núm. 4, pp. 1-13, 2021
Sociedad Entomológica Argentina
Recepción: 29 Septiembre 2020
Aprobación: 30 Agosto 2021
Publicación: 30 Diciembre 2021
Abstract: The objectives of this study are to identify thrips and their biological controllers, to analyze their abundance in three flower opening stages stages of chrysanthemum crops, and to determine alpha and beta diversity in two ecoregions of Jujuy province (Argentina). The study was carried out in the Chaco ecoregion (ECH) in October and November 2016, and in the Prepuna ecoregion (EP) in February and March 2018. On each date, six replicates of five flowers were excised in the stages: flower bud , semi-open flower and open flower. Diversity profiles and rank-abundance curves were built, and Jaccard's coefficient of similarity was applied. The diversity profiles showed significant differences in thrips communities. In the ECH, 15 thrips species were identified; the dominant and permanent species were Microcephalothrips abdominalis (Crawford) and Frankliniella occidentalis (Pergande). Five species were recorded in the EP, although Frankliniella gemina Bagnall and Thrips tabaci Lindeman on their own represented 78% of the total abundance. In the ECH, 20 entomophagous species/morphospecies were recognized, and 19 in the EP; antocorids were dominant in both ecoregions. The thrips and the entomophagous communities of chrysanthemum flowers were different (34%) between the ecoregions studied.
Keywords: Agroecology, Biological control, Floriculture, Frankliniella, Microcephalothrips abdominalis.
Resumen: Los objetivos de este trabajo son identificar las especies de trips y los controladores biológicos, analizar la abundancia de los mismos en tres estados de apertura de las flores del cultivo de crisantemo, y determinar su diversidad alfa y beta en dos ecorregiones de la provincia de Jujuy (Argentina). El estudio se realizó en octubre y noviembre de 2016 en la ecorregión Chaco (ECH), y en febrero y marzo de 2018 en la ecorregión Prepuna (EP). En cada fecha se cortaron cinco flores en los estados de botón floral, flor semiabierta y flor abierta, en seis repeticiones. Se realizaron perfiles de diversidad, curvas de rango-abundancia y se aplicó el coeficiente de similitud de Jaccard. Los perfiles de diversidad mostraron diferencias significativas en las comunidades de thrips. En la ECH se identificaron 15 especies de trips; las dominantes y permanentes fueron Microcephalothrips abdominalis (D. L. Crawford) y Frankliniella occidentalis (Pergande). En la EP se registraron cinco especies, aunque solo Frankliniella gemina Bagnall y Thrips tabaci Lindeman alcanzaron el 78% de la abundancia total. En la ECH se reconocieron 20 especies/morfoespecies de entomófagos y 19 en la EP; los antocóridos fueron dominantes en ambas ecorregiones. Las comunidades de trips y entomófagos de las flores de crisantemos fueron disimiles (34%) entre las ecorregiones estudiadas.
Palabras clave: Agroecología, Control biológico, Floricultura, Frankliniella, Microcephalothrips abdominalis.
INTRODUCTION
Most studies addressing floricultural production in Argentina, especially those carried out at the regional level, point out that it is an important activity with high growth and development potential. Such potential is associated with employment creation and diversification possibilities for producers in rural and periurban areas surrounding the main consumption centers of the country (Villanova et al., 2013). In Argentina, the main production areas are located in the metropolitan area of Buenos Aires and San Pedro (which concentrate 50% of the production), followed by Misiones, Santa Fe, Jujuy, Corrientes, Entre Ríos and Mendoza provinces (Herrero et al., 2017).
In Jujuy province, floriculture is based on the production of cut flowers such as the chrysanthemum (Chrysantemum L.), carnation (Dianthus L.), gladiolus (Gladiolus L.), statice (Limonium sinuatum L. Mill.), lisiantus (Eustoma grandiflorum (Raf.) Shinners), marigold (Calendula L.), daisies (Leucanthemum Mill.), roses (Rosa L.), and ornamental plants. The production area is of approximately 135 ha, distributed in contrasting agroclimatic zones. In the temperate valleys of the Chaco, where Las Pampitas and El Pongo localities are settled, the production of cut flowers takes place in winter and spring; while in Prepuna, especially in Maimará and Uquía, it takes place in spring and summer. In 2020, this activity received an important boost in order to improve crop quality, introduce technology, promote the cultivation of other species, and improve commercialization. In this process of floriculture expansion, pests represent one of the main limitations due to the expense of their control, and secondarily, due to the impacts of chemical products on the environment and on the health of the farmers.
In ornamental plant-producing countries, Thysanoptera or thrips, especially Frankliniella occidentalis (Pergande), are considered key pests because, in addition to the direct and indirect damage they cause on crops, they can be carried on cut flowers. This generates losses or phytosanitary sanctions that impact on their commercial performance (Arévalo et al., 2003; Reitz & Funderburk, 2012; Held & Pickens, 2018).
In Argentina, references to thrips in ornamental plants can be found in systematic lists of host plants by De Santis et al. (1980), de Borbón (2005, 2013), de Borbón et al. (1999), Carrizo et al. (2008), Curis et al. (2015), and de Borbón & Zamar (2018). There are also studies about other damaging species on these crops (Carrizo & Klasman, 2001; Castresana et al., 2008). In Jujuy province, Zamar et al. (2009, 2014) provided knowledge about thrips associated to ornamental flowers, while Alejo & Zamar (2018) and Alejo et al. (2019) analyzed the arthropods present on chrysanthemus.
Regarding thrips biological controllers in chrysanthemum flowers, the existing literature only reports species lists (García Baca, 1983; Castro et al., 1999; Mendes Carvalho et al., 2006) and ecological interactions with pest thrips, mainly F. occidentalis (Van Lenteren et al., 1995; Lacasa et al., 1996; Miranda-Salcedo & Loera-Alvarado, 2019; Farkas et al., 2016). In Argentina, the only studies that analyze thrips diversity and abundance within the framework of arthropod fauna in chrysanthemum flowers, both in the field and in greenhouses, are those by Alejo & Zamar (2018) and Alejo et al. (2019), respectively.
In general, pest management of these crops is based on chemical control. In the case of Argentine floriculture, there are no specific programs of integrated pest management, and application of the BPA (Good Farming Practices) regulation is lacking (Fernández & Fernández, 2013; Fernández Acevedo, 2016). Therefore, as pointed out by Salazar Villarreal et al. (2019), agrobiodiversity inventories are necessary to understand the functional structures of agricultural ecosystems, while the application of indices becomes a useful tool for the design of sustainable systems based on the use and management of local biodiversity.
The objectives of this study are to identify thrips and biological controller species, analyze their abundance in three flower opening states of chrysanthemum crops, and determine alpha and beta diversity in two ecoregions of Jujuy province (Argentina).
MATERIAL AND METHODS
This study was carried out in chrysanthemum smallholder farms located in the Chaco Serrano (ECH) and Prepuna (EP) ecoregions of Jujuy province (Argentina). In each ecoregion, three farms were selected, whose characteristics are indicated in Fig. 1.
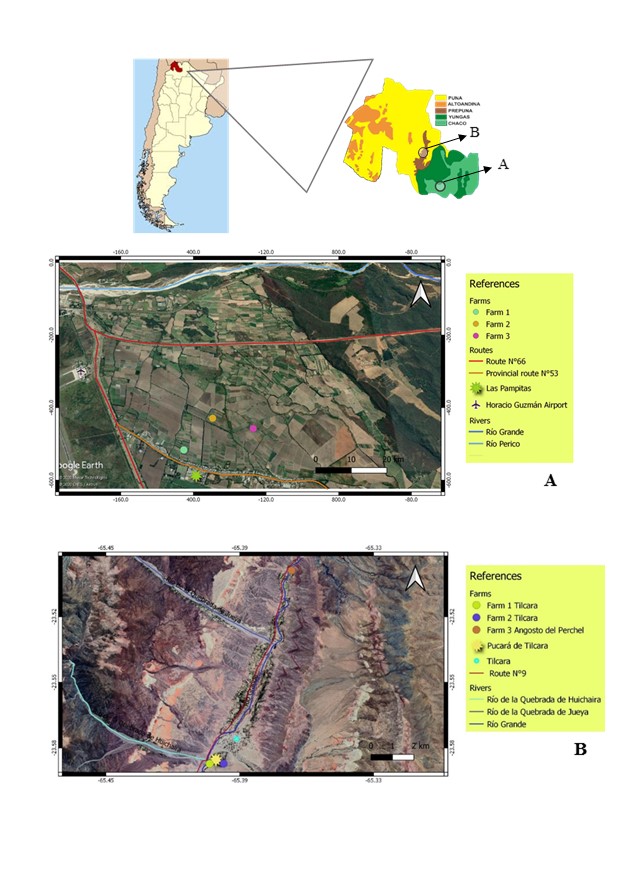
Fig 1. Location of the farms sampled in the Chaco (A) and Prepuna (B) ecoregions of Jujuy province, Argentina.
Chaco ecoregion: it is located between the eastern slopes of the Puna and the sub-Andean mountain ranges, forming a wide ecotone between the Monte and Yungas phytogeographical provinces, whose mean altitude is 974 m.a.s.l. in Los Pericos area. The climate is temperate, with a mean annual rainfall between 500 and 1,000 mm. concentrated in summer. Mean annual temperature is slightly above 20° C in the warmest month, and 11°C in the coldest month. Native vegetation corresponds to that of xerophilous, sub-humid forests, and species from the Chaco and ecotone are found (Piccolo et al., 2008). The main crops of the region are cut flowers, tobacco, vegetables, beans, strawberries and peaches.
Prepuna ecoregion: It is an extensive region with subparallel steep mountains, with a north-northeast orientation separated by deep valleys, with a mean altitude of 2,500 m.a.s.l. in Tilcara. The climate is dry and temperate-cold, with well-defined annual seasons. Mean annual temperature is between 10-14° C; day temperatures are high, but daily frosts take place all through winter, and are frequent in spring but absent from January to March. Rainfall is scarce (less than 180 mm annually) and occurs almost exclusively in summer. Winds are soft during the morning and increase between midday and 3 p.m. Vegetation is shrubby, low and scattered; dominated by steppe and xerophyte shrubby bushland physiognomies. The main crops of the region are vegetables, corn, garlic, onions, cut flower and peaches.
This study was carried out during the crop flowering stage, in October and November 2016 in the Chaco ecoregion, and in February and March 2018 in the Prepuna ecoregion, since both ecoregions have contrasting agroclimatic characteristics. Sampling consisted of the random collection of five chrysanthemum flowers undergoing different opening stages: flower bud (FB), semi-open flower (SF), open flower (OF).
Six replicates were carried out at each state of opening of the flowers in each farm (30 flowers), totaling 90 flowers per sampling date. Flowers of each replicate were placed in polyethylene bags, labeled, and kept in a fridge until their examination.
Extraction of larvae and adult thrips and of entomophagous species was performed under a Leica EZ4 stereoscopic microscope with a thin brush dipped in 70% ethanol. The material of each sample was kept in labeled bottles for their subsequent taxonomic identification.
The identification of thrips species was performed on semipermanent microscopical preparations based on the Mound & Marullo (1996) technique, using specific keys for adults. Thrips larvae were counted and classified as Thripidae larvae. Entomophagous species were identified to the species/morphospecies or family levels using available keys (Haramboure et al., 2014). All the collected specimens are kept at the entomological collection of the Instituto de Biología de la Altura, Universidad Nacional de Jujuy, Jujuy province.
To find out the total number of thrips and entomophagous species during the flowering stage of chrysanthemums, absolute and relative abundance tables (indicating the number of orders, families and species/morphospecies) were performed for each ecoregion.
To evaluate whether the data adjusted to a normal distribution, Shapiro and Levene tests were carried out. Since these assumptions were not met, a Kruskal-Wallis test was used. These analyses allowed determining statistical differences between the response variable (thrips abundance) and ecoregion, and among the farms sampled at each ecoregion. The same analysis was applied for the opening stages of chrysanthemum flowers with a 95% confidence level. For the cases in which statistical differences were found, pairwise comparisons using Wilcoxon tests were carried out. These analyses were also performed in R statistical software (R CoreTeam, 2018).
To perform effective comparisons between the two ecoregions, the true diversity was measured (Jost, 2006). This analysis facilitates the ordination and comparison of communities. The units of true diversity measurement correspond to the effective number of species, thus, the three diversity orders were calculated:
- Order 0D diversity (species richness) is not sensitive to species abundance. Thus, the obtained value simply represents species richness.
- 1D diversity (Shannon index exponential) includes both common and rare species (such as dominant species).
- 2D diversity (inverse of Simpson index), considers dominant species and excludes rare species, according to Jost (2006) (Moreno et al., 2011).
Diversity profiles were built using the curves of the different orders (q) of thrips communities and entomophagous species in the two regions. These analyses were performed using the package “SpadeR” version 3.2.5. in R statistical software (R Core Team 2018).
To evaluate and compare arthropod community structure, rank-abundance curves were built for each sampling date at each ecoregion. These curves consist of a graphical method to rank species in decreasing order of abundance. They allow to compare biologically important aspects of species diversity between samples, and provide information about dominance and about the presence of rare species in the communities (Whittaker, 1972).
To evaluate beta diversity of thrips and biological controller communities in chrysanthemum crops, both among farms and between ecoregions, the Jaccard similarity index (JI) was used. This index associates the number of common species/families found in the compared samples, and it is calculated as follows:
JI = c / (a + b – c)
where a = number of species/families present in sample A; b = number of species/families present in sample B; c = number of species/families present in both samples. The values of the index range between 0 (when there are no shared species/families between samples), and 1 (when the two samples have the same species composition).
RESULTS
Abundance and diversity of thrips species
In both ecoregions, 7,407 adult thrips (35.3%) and 13,442 larvae (64.5%) were found in chrysanthemum flowers. Based on the number of species, the represented families were Thripidae (10), Aeolothripidae (1) and Phlaeothripidae (4). The latter two families were registered only in the Chaco ecoregion (ECH). The five species shared among ecoregions belonged to Thripidae (Frankliniella australis Morgan, Frankliniella gemina Bagnall, Frankliniella schultzei Trybom, Frankliniella occidentalis (Pergande) and Thrips tabaci Lindeman).
In ECH 6,082 (55%) adult thrips were found, corresponding to Thripidae (99.67%), Aeolothripidae (0.016%), Phlaeothripidae (0.31%). The highest abundances were found at farm 3 (7,107 thrips), followed by farm 1 (2,526 thrips) and 2 (1,435 thrips). Fourteen species with different representativeness percentages were identified: Microcephalothrips abdominalis D.L. Crawford (58%), F. occidentalis (23%), F. schultzei (8%), F. gemina (7%), Frankliniella oxyura Bagnall (1%), F. australis (1%), Frankliniella sp. (1%), Haplothrips sp.2 (0.13%), Haplothrips sp.4 (0.12%), Haplothrips sp. 1 (0.05%), Neohydatothrips burungae Hood, Caliothrips phaseoli Hood, T. tabaci (Thripidae), Stomatothrips angustipennis Hood (Aeolothripidae) and Haplothrips sp.3 (Phlaeothripidae) (0.02%) (Table I). Farm 1 reached the highest richness (11), followed by farms 3 (nine) and 2 (eight).
In the Prepuna, 1,325 adults (13.5%) belonging to Thripidae were collected, while the larvae reached 86.4%. The highest abundances were registered in farm 2 (4,356 thrips), followed by farm 3 (2,950) and farm 1 (2,800 thrips). The identified species and their representativeness percentages were: F. gemina (49%), T. tabaci (29%), F. occidentalis (21%), F. australis (1%) and F. schultzei (0.15%). Each farm reached the same number of species, except for farm 1, where F. schultzei was also found, although with minimum abundances (Table I).
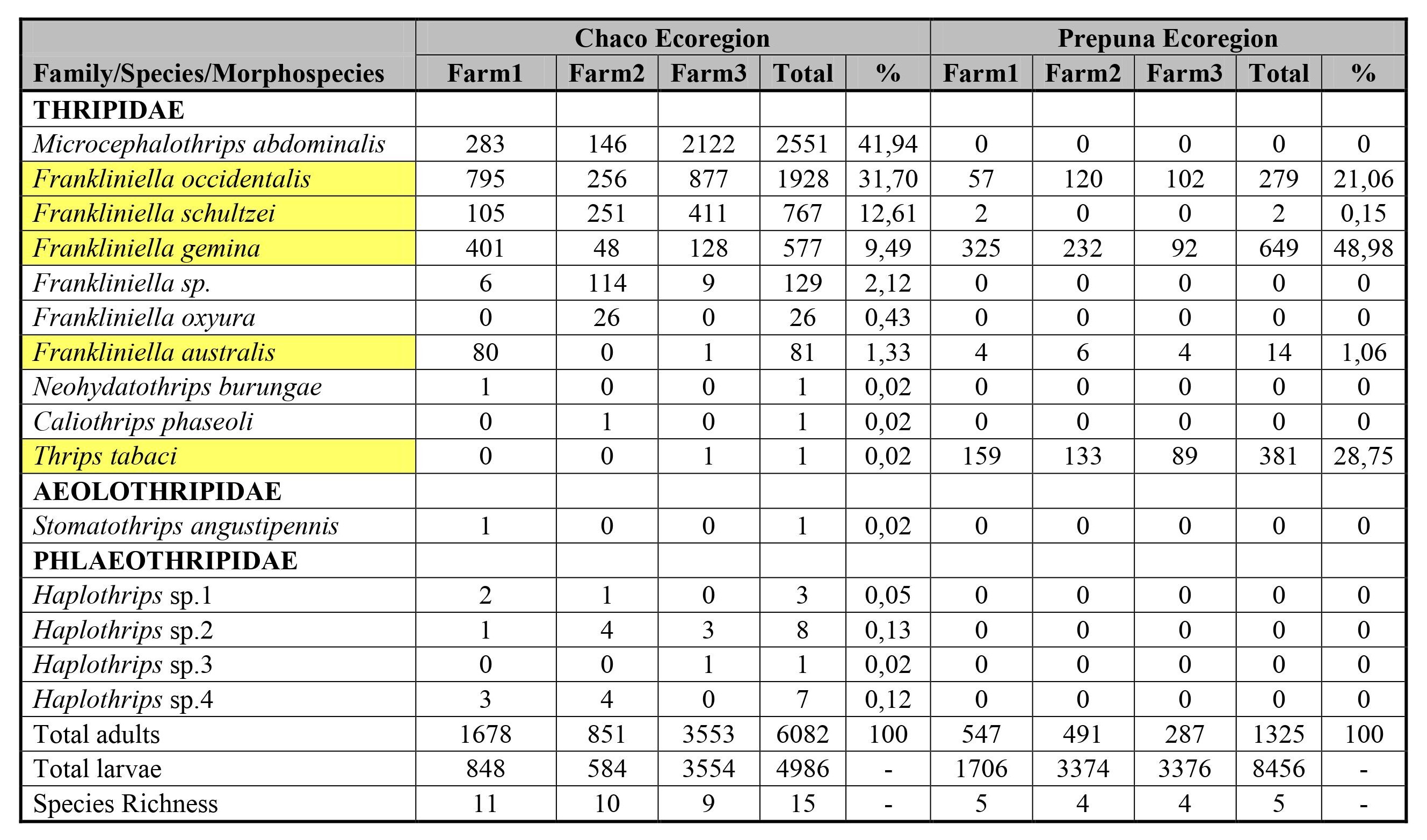
Table I
Table I. Distribution of richness and abundance of species of Thysanoptera found in chrysanthemum flowers of the Chaco and Prepuna ecoregion (Jujuy, Argentina) between October and November, 2016; and February and March, 2018, respectively.
Shared species are highlighted in yellow.
Abundance and diversity of entomophagous species
A total of 402 specimens of entomophagous were collected, of which 172 corresponded to ECH and 230 to EP, distributed in the classes Insecta (86.6%) and Arachnida (13.4%). In Table II, the systematic list with data on the presence and abundance of each species/morphospecies at each ecoregion is presented.
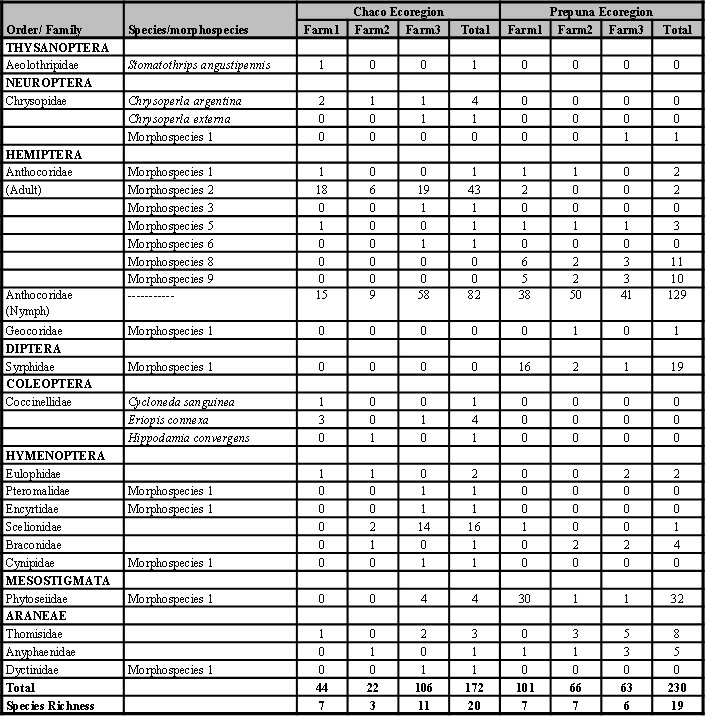
Table II
Table II. Distribution of richness and abundance of entomophagous species/morphospecies registered in chrysanthemum flowers of the Chaco and Prepuna ecoregions (Jujuy province, Argentina) between October and November, 2016; and February and March, 2018, respectively.
Abundance and diversity of thrips in the opening stages of chrysanthemum flowers
In both ecoregions, the open flower stage had higher quantity of adult thrips and larvae, followed by the semi open flower, and the flower bud stages (Fig. 2). Regarding thrips diversity and abundance in flowers of each ecoregion, in the ECH, M. abdominalis was dominant at the three opening stages (FB: 756; SF: 1947; OF: 2833), followed by F. occidentalis (FB: 287; SF: 561; OF: 1288), F. schultzei (FB: 91; SF: 185; OF: 515) and F. gemina (FB: 125 and SF: 184; OF: 366). N. burungae, S. angustipennis, Haplothrips sp. 1, Haplothrips sp. 2 and Haplothrips sp. 3 were present with low number of individuals (Fig. 2). In PE, the dominant species was F. gemina (FB: 35; SF: 157; OF: 457), followed by T. tabaci (FB: 42; SF: 114; OF: 225) and F. occidentalis (FB: 10; SF: 73; OF: 196). In this ecoregion, only two individuals of F. schultzei were found in open and semi open flowers (Fig. 2).
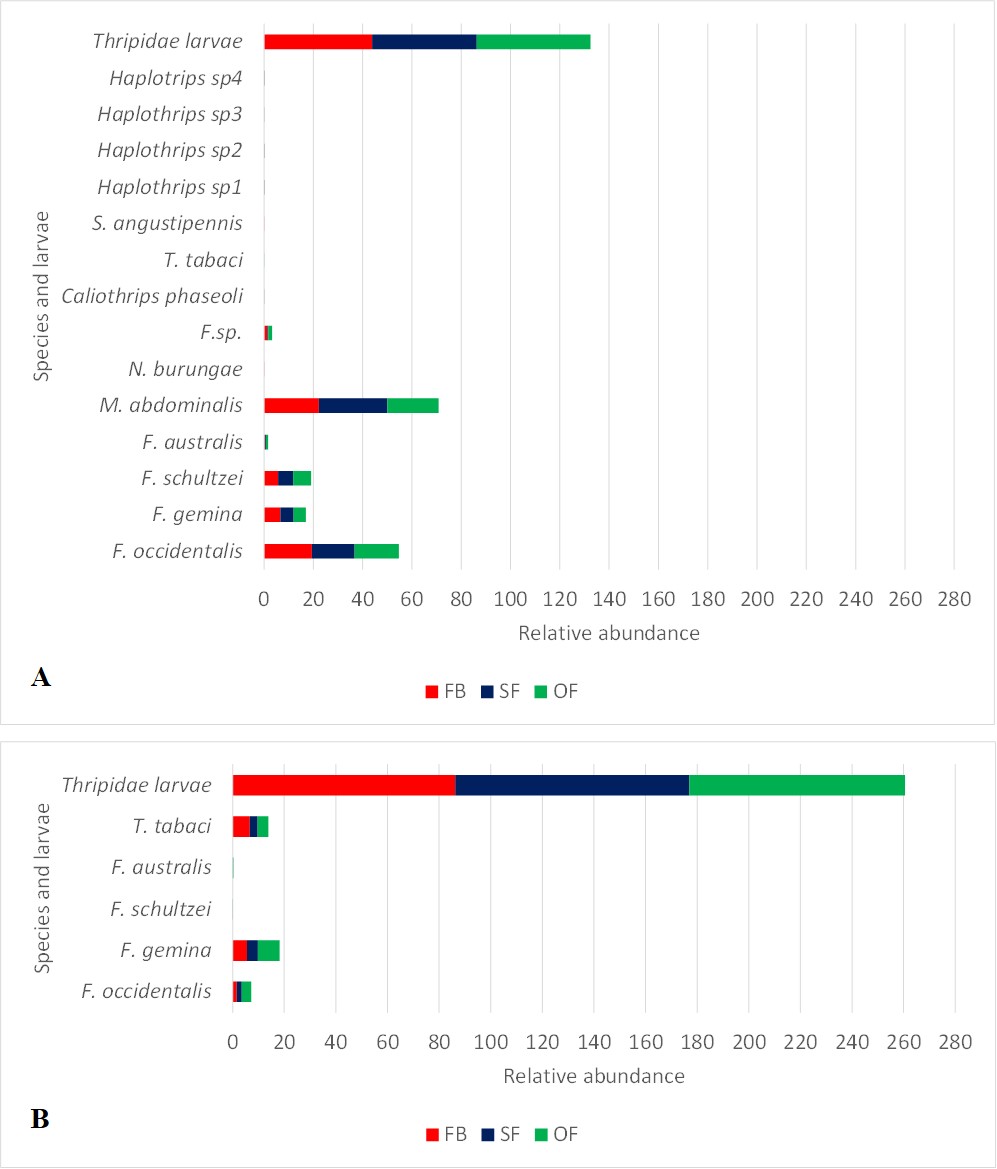
Figure 2
Fig. 2. Abundance of larvae and adult thrips in the different flower opening stages of the Chaco (A) and Prepuna (B) ecoregions of Jujuy province (Argentina)
The Kruskal Wallis test indicated significant differences between the medians of thrips abundances in the different opening stages (H ECH: 13.5; p-value ECH: 0.0012; H EP: 13.08; p-value EP: 0.0014). For the ECH, the median comparison test determined that FB and SF were different to OF. The opposite pattern was found in the Prepuna, where the medians of SF and OF differed from FB.
Statistical analysis of thrips abundance in chrysanthemum flowers among the studied farms of each ecoregion
In the ECH, significant differences in thrips abundance were found between the studied farms (H: 13.74; df: 2; p-value: 0.0010). The median test determined that farms 1 and 2 differed from farm 3. In the farms of EP, no significant differences were found (H: 0.98; df: 2; p-value: 0.6232).
Number of thrips per chrysanthemum flower
In Table III, the mean values of thrips (larvae and adults) in 90 revised flowers considering all the opening stages are observed. In the ECH, larvae were present in the flower bud, with values from two to 19 larvae/flower, and five to 22 adults/flower. In the EP, the occurrence of larvae was higher, with three-36 per flower, but the number of adults remained less than six per flower.
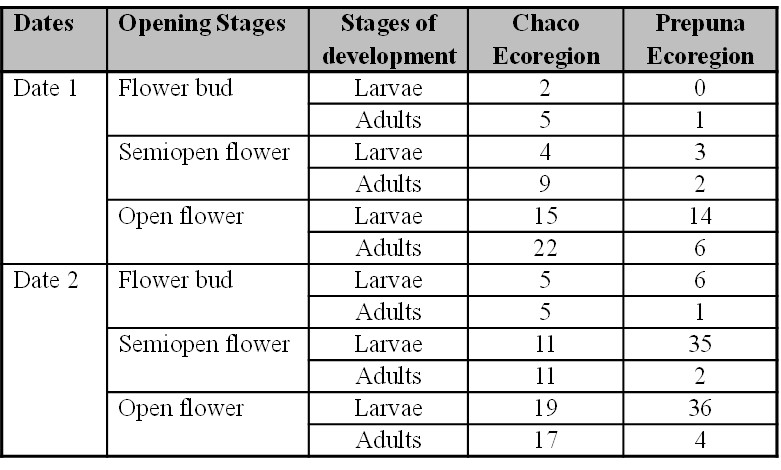
Table III
Table III. Mean number of thrips larvae and adults by flower on each sampling date and Chaco and Prepuna ecoregions
Date 1 ECH: 18/10/2016; Date 2 ECH: 8/11/2016; Date 1 EP: 19/2/2018; Date 2 EP: 12/3/2018.
Diversity profiles
The alpha diversity analysis showed that the confidence intervals of the EP and ECH thrips communities’ profile curves did not overlap, indicating significant differences between them. In the ECH, the order 0D indicated the presence of 14 effective species, while in the EP there were five. In the order 1D, the ECH was more diverse, with 4.038 effective species, while the order 2D exhibited 3.304 effective species (Fig. 3A).
Regarding the entomophagous species of both ecoregions, the confidence intervals of the curves overlapped, reflecting no significant differences between them. In the ECH, the order 0D showed the existence of 16 effective species. In the order 1D, the PR was more diverse, with 5.206 effective species, while the order 2D showed 4.038 effective species (Fig. 3B).
The Kruskal Wallis analysis did not show significant differences between the medians of thrips abundance in the two ecoregions (H: 0.7817; p-value: 0.3766).
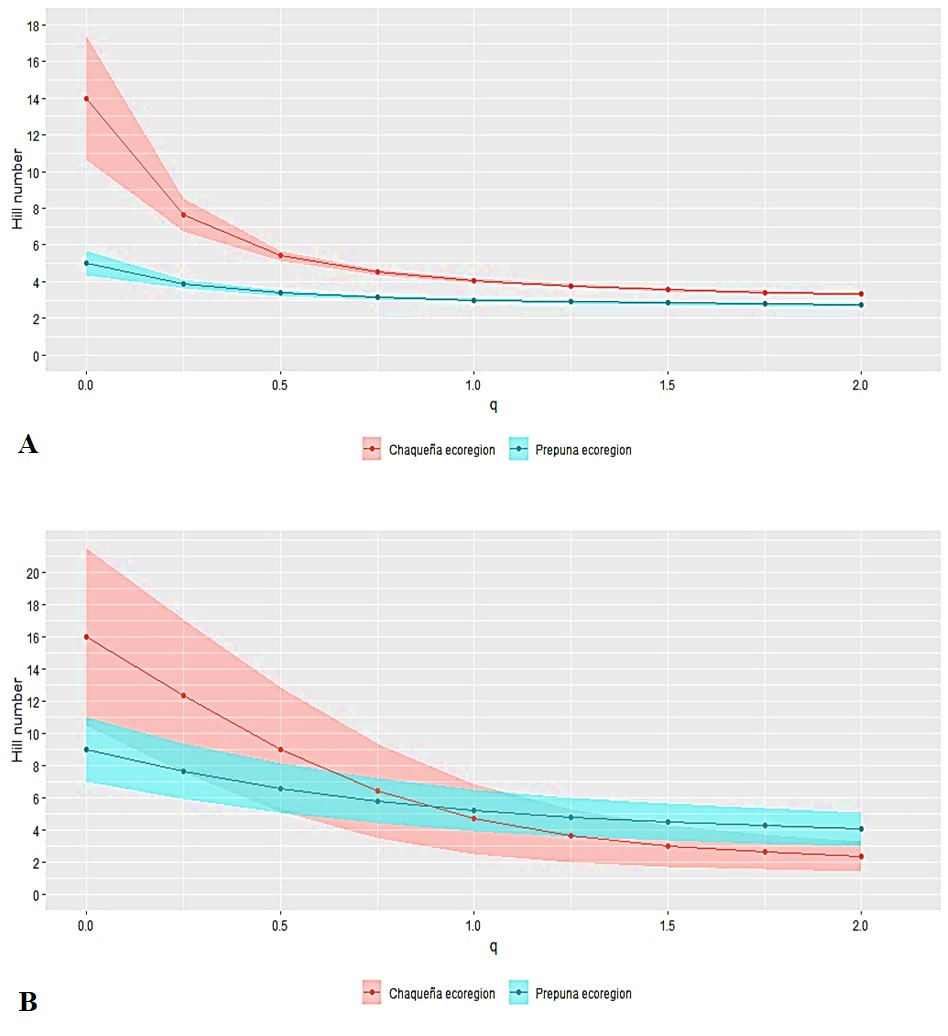
Figure 3
Fig. 3. Alfa diversity profiles of A: thrips and B: entomophagous species in the Chaco and Prepuna ecoregions. Species richness (q0), Shannon exponential (q1) and abundant species (inverse Simpson) (q2) are indicated.
The shading of each curve represents the 95% confidence interval.
Rank abundance curves by sampling date
Thrips larvae were dominant in the sampling dates of the two ecoregions (Fig. 4), followed by M. abdominalis adults in the ECH, and by T. tabaci and F. gemina in the EP. In both ecoregions, F. occidentalis was dominant in the third and fourth order, respectively.
Regarding entomophagous species, nymphs of Anthocoridae (Hemiptera) were dominant in the curves of both ecoregions. Other entomophagous species with high abundance in the ECH were morphospecies 2 of Anthocoridae in October, and Scelionidae (Hymenoptera) in November (Fig. 4A). In the EP, the morphospecies 8 and 9 of Anthocoridae and larvae of Syrphidae (Diptera) predominated on both sampling dates and Phytoseiidae (Acari: Mesostigmata) in February (Fig. 4B).
Likewise, differences regarding the diversity of rare species were found in the two ecoregions. The EP was limited to morphospecies 2 of Anthocoridae and Braconidae (Hymenoptera) in both sampling dates. In contrast, in the ECH there was a higher number of rare species with only one individual, especially in October (Fig. 4A). This group of rare species was constituted by thrips and entomophagous (N. burungae, C. phaseoli, T. tabaci, S. angustipennis, Haplothrips sp. 4, sp.1, sp.2, morphospecies 1, 3 and 5 of Anthocoridae, and the Coleoptera Cycloneda sanguinea (L.) and Hippodamia convergens Guérin-Méneville, one morphospecies of Pteromalidae, and the families Braconidae (Hymenoptera), Thomisidae and Anyphaenidae (Araneae). On the other hand, in November, Haplothrips sp.1 (Thysanoptera), Ch. argentina, Ch. externa (Neuroptera) and one morphospecies of Encyrtidae, Cynipidae, Dyctinidae (Hymenoptera) and Phytoseiidae (Mesostigmata) were registered.
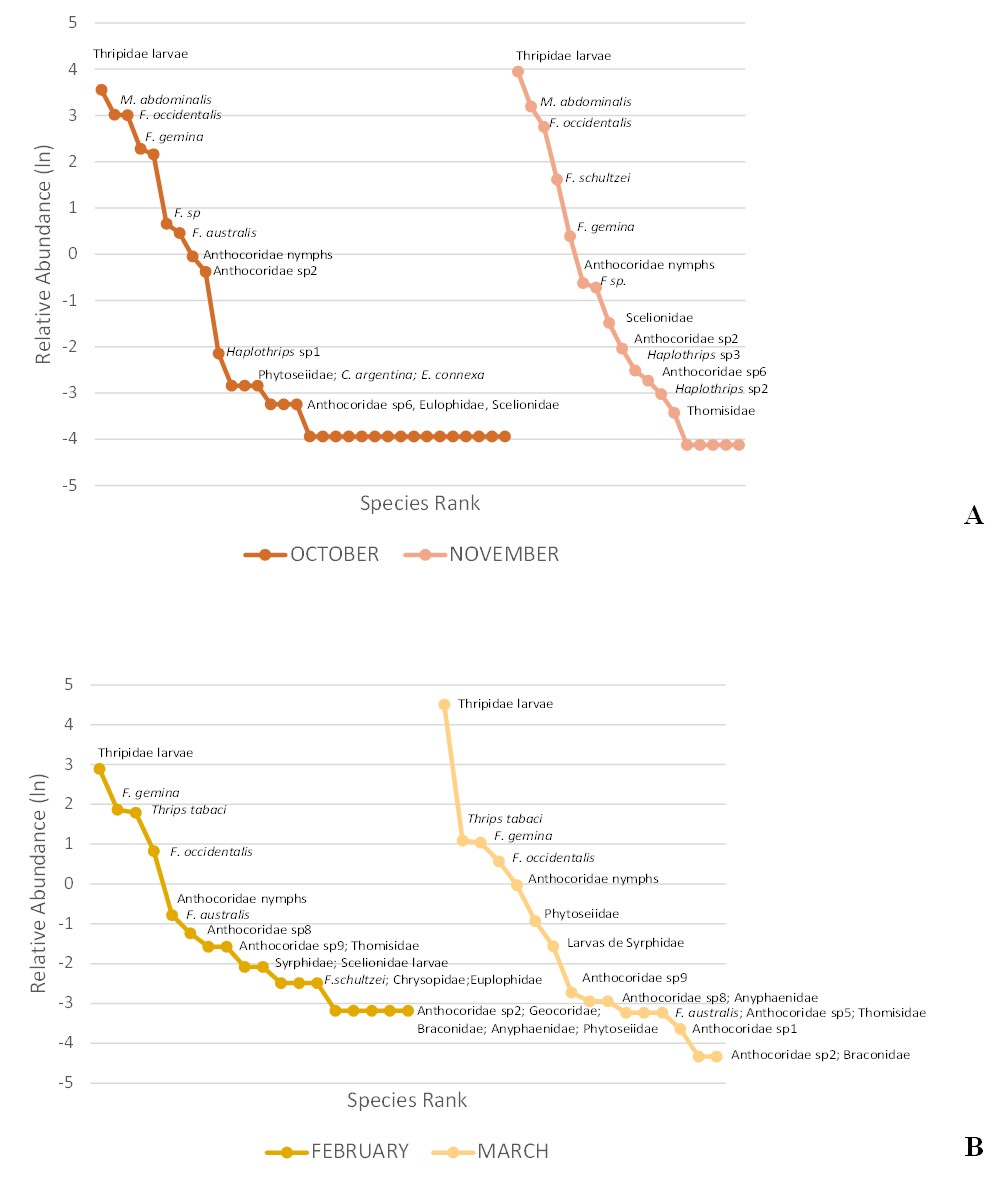
Figure 4
Fig. 4. Rank abundance curves of thrips and entomophagous species by sampling date in A: Chaco ecoregion; B: Prepuna ecoregion.
Beta Diversity
The Jaccard index in the ecoregions was 34%, indicating few shared species between ECH and EP. Additionally, it should be highlighted that in the former, farms were not very similar, ranging from 30% to 42%. However, in the EP the values of the comparisons between farms were higher than 55% (Table IV).
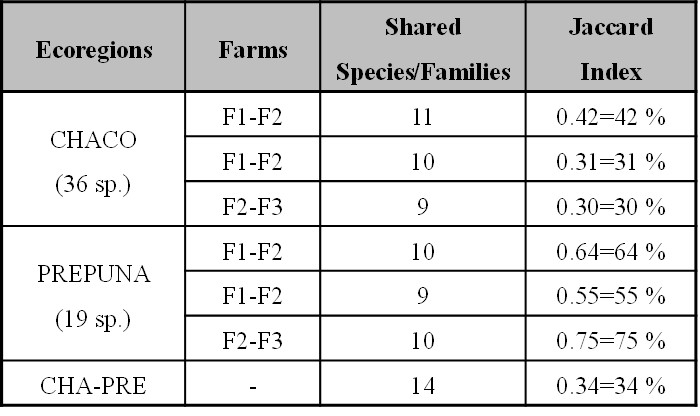
Table IV
Table IV. Shared species and Jaccard similarity index calculated for thrips and entomophagous communities of the studied farms of each ecoregion.
DISCUSSION
During the flowering stage of chrysanthemum crops in the Chaco and Prepuna ecoregions of Jujuy, differences in both species composition and abundance were detected, which were clearly reflected on the diversity profiles (Fig. 3A, B). In ECH, 15 representative species of Thripidae, Aeolothripidae and Phlaeothripidae were identified, although the dominant and permanent species were M. abdominalis and F. occidentalis, which reached 74% of the total abundance. In EP, five species of Thripidae were registered, of which F. gemina and T. tabaci reached 78% of the total abundance, although F. occidentalis was also relevant, reaching 21% (Table II, Fig. 2).
Unlike the research carried out on the diversity of thrips in chrysanthemum crops in Mexico (Loera-Alvarado et al., 2017) and Colombia (Arévalo et al., 2003), this study analyzes this information based on the stages of flower opening in two contrasting agricultural ecoregions.
The taxonomic composition and incidence of thrips in a same crop can vary among countries. Thus, in Mexico and Colombia, 15 and seven thrips species were found, respectively (op. cit.); while in Jujuy (Argentina) 15 species were registered, but the complex was different to that reported in the aforementioned countries. However, the only shared species was F. occidentalis, considered as a key pest in countries that produce chrysanthemums (Arévalo et al., 2003; Reitz & Funderburk, 2012; Held & Pickens, 2018). The results obtained show the influence of biogeography in insect diversity, taking the association with a given crop as reference. In this way, thrips diversity in chrysanthemums from higher altitudes (EP) within a xerophytic context was scarce compared to that from lower altitudes, in temperate environmental conditions (ECH). The responses of hexapod diversity to an altitudinal gradient have been reported by several authors, especially in the Americas. However, studies have focused on natural ecosystems (Janzen et al., 1976; García-Gómez et al., 2011; Guerrero & Sarmiento, 2010; Ramos, 2018). Furthermore, to this date, no studies have analyzed changes in arthropod taxonomic composition in agricultural systems of different altitudes. The only reference for Jujuy province regarding this issue is the study by Zamar (2011), who analyzed thrips diversity in crops, and spontaneous and natural vegetation in the Prepuna and Puna ecoregions. The list of thrips included species commonly found in crops of the EP: F. australis, F. gemina, F. occidentalis, F. schultzei and T. tabaci (Zamar & Arce de Hamity, 1999; Zamar et al., 2006, 2007), which were also collected from chrysanthemum flowers (Zamar et al., 2009). In agricultural systems of the ECH, F. occidentalis, F. gemina, F. schultzei, F. australis, M. abdominalis, C. phaseoli, and N. burungae were cited in chrysanthemums (Alejo & Zamar, 2018; Alejo et al., 2019), tobacco, strawberries and beans (Rodríguez et al., 2014; Vivanco & Zamar, 2014; Zamar et al., 2020).
The differences in species richness between the two ecoregions may have implications on the type of thrips management in the crop, which could be more focused on the PE where fewer species were found. Regarding this issue, it is important to highlight that thrips behavior hinders their control, especially in cut flowers, because the larvae remain hidden in the flower organs; prepupae and pupae remain in the soil, and adults have great mobility.
Another difference with the studies by Arévalo et al. (2003) and Loera-Alvarado et al., (2017) is that in the present study, the abundances of thrips adults and larvae were analyzed. In that sense, another contrast among ecoregions was observed: in ECH, the total number of adults and larvae was high (6,082 and 4,985 individuals, respectively). In EP, the number of larvae (8,456) was much higher than that of adults (1,325), especially on the second sampling date (Fig. 4B). These evidences show the need to review the type of management applied to control thrips in chrysanthemums in both ecoregions in order to optimize the marketing of this cut flower.
When relating the flower opening stages with thrips abundance in each ecoregion, contrasting situations were also found. In the ECH, abundance medians of the initial flower opening stages differed from those of the open flower, while in the PE, differences between the flower bud and other opening stages were found. These results might be associated to the type of agricultural management rather than to environmental conditions of each ecoregion. However, this possibility should be further explored. Either way, it can be clearly distinguished that in ECH, larvae abundance ranged between 44 and 46% at each flower stage, while in EP larvae reached values higher than 80% in all stages (Fig. 2). When expressing these values per flower unit of each flowering stage (Table IV), semi-open and open flowers were found to have a higher number of larvae: 4-19 larvae/flower in the ECH and 3-36 larvae/flower in the EP.
In ornamental plants, or in TSWV-susceptible plants, the threshold of thrips density should be very low, due to the aesthetic damage caused by the feeding of these insects (AgriSover, 2019) or by the effect of droppings (Zamar et al., 2009). The information generated marks a baseline to establish the thresholds of economic damage of thrips in the areas of chrysanthemum production in the province of Jujuy, and for the implementation of integrated pest management of the crop.
Regarding thrips diversity at each flower opening stage, in the ECH the proportion of dominant species and larvae was kept, while in the EP, an increase in the abundance of F. gemina and F. occidentalis, and a decrease in T. tabaci as flowers open were observed (Fig. 2). The group of rare thrips species fluctuated at very low abundances, such as Haplothrips in ECH. This genus breeds in flowers, particularly Asteraceae and Poaceae (Mound & Matsunaga, 2017). The appearance of C. phaseoli and N. burungae can be considered accidental, since they are folivores (Contreras & Zamar, 2010; Sosa et al., 2017). Thus, insect monitoring should be performed from the flower bud; as flower opening progresses, they offer higher pollen, nectar and shelter resources. This aspect is in agreement with the results of Carrizo & Klasman (2001) about thrips in carnations, who indicated that abundance of both adults and larvae was higher in the completely open flower.
With respect to entomophagous species present in chrysanthemum flowers, 21 species/morphospecies distributed in 14 families of Insecta were found (Aeolothripidae, Anthocoridae, Geocoridae, Coccinellidae, Chrysopidae, Syrphidae, Eulophidae, Pteromalidae, Encyrtidae, Scelionidae, Braconidae and Cynipidae), along with four families belonging to Arachnida (Phytoseiidae, Thomisidae, Anyphaenidae and Dyctinidae). Stomatothrips angustipennis is reported for the first time in chrysanthemum flowers as a probable predator, and its distribution in northwestern Argentina is also updated. This Aeolothripidae had been registered only in the provinces of Tucumán, Buenos Aires and Entre Ríos (De Santis et al., 1980). Mound & Marullo (1996), indicating that knowledge on the biology of this genus is scarce. They also note that some species such as S. angustipennis are mimetic of ants, and are associated with gramineous vegetation, where they apparently feed on small arthropods.
Most of the families were reported as thrips predators or parasitoids by other authors, especially of T. tabaci and F. occidentalis (Loomans & Van Lenteren, 1990; Riudavets, 1995), or recorded in chrysanthemum crops (García Baca, 1983; Castro et al. 1999; Mendes Carvalho et al., 2006; Alejo & Zamar, 2018; Alejo et al., 2019). Among the most efficient entomophagous groups for thrips control, seven morphospecies of Anthocoridae, one of Phytoseiidae and one of Syrphidae were found in both ecoregions. The remaining predators had low abundances (< 8). Parasitoids were well-represented (six families), although the only family present in both ecoregions (and which may include thrips parasitoid species) was Eulophidae (Lommans & Van Lenteren, 1995). Among the most numerous generalist endoparasitoid species, only Scelionidae was found (Margaria, 2012), with 16 individuals in the ECH.
In summary, thrips and entomophagous species communities in chrysanthemum flowers of the Chaco and Prepuna ecoregions were dissimilar (34%). This implies that thrips biological control programs should be adjusted to the ecological conditions of each region. At the same time, when analyzing the composition of thrips and entomophagous species between farms, similarities and differences were found in EP and ECH, respectively, which might be related to the type of agricultural management carried out by producers on their farms.
ACKNOWLEDGMENTS
We thank the producers of Las Pampitas, Tilcara and Angosto del Perchel who kindly lent us their farms to carry out the study. We are also grateful to Ivan Escalier and Luis Choque. This study was funded by the project F/0026 of Secretaría de Ciencia y Técnica y Estudios Regionales-Universidad Nacional de Jujuy and INECOA-CONICET-UNJu. Finally, we thank the editor and anonymous reviewers for their valuable suggestions and comments.
REFERENCES
Alejo, G.B., & Zamar, M.I. (2018) Diversidad y roles tróficos de artrópodos en flores de Chrysanthemum (Asteraceae) cultivados a escala de pequeños productores de Jujuy (Argentina). Fitosanidad, 22, 65-79.
Alejo, G.B., Zamar, M.I., & Contreras, E.F. (2019) Diversidad y grupos funcionales de artrópodos en el cultivo de Chrysanthemum morifolium Ramat. (Asterales: Asteraceae) en invernadero en Jujuy, Argentina. Revista de la Sociedad Entomológica Argentina, 78(1), 22-35.
AgriSolver. (2019) Manejo Integrado de trips (Frankliniella occidentalis) en invernadero. Available at: https://www.agrisolver.com/blog/manejo-integrado-de-trips-frankliniella-occidentalis-en-invernadero.
Arévalo, P.E., Omaira, X., Quintero, F., & Correa, G. (2003) Reconocimiento de trips (Insecta: Thysanoptera) en floricultivos de tres corregimientos del municipio de Medellín, Antioquia (Colombia). Revista Colombiana de Entomología,29, 169-175.
Cáceres, S., Miño, V.S., & Aguirre, M.R.A. (2011) Guía práctica para la identificación y el manejo de las plagas de pimiento. Ediciones INTA, Estación experimental Agropecuaria Bella Vista, Corrientes, Argentina.
Calvente, M. (2015) Guía: diagnóstico y control de enfermedades y plagas animales para cultivos florícolas. Ediciones INTA, Argentina.
Cambero-Campos, J., Johansen-Naime, R., García-Martínez, O., Cantu-Sifuentes, M., Cerna-Chavez, E., & Retana-Salazar, A. (2011) Especies depredadoras de trips (Thysanoptera) asociadas a huertas de aguacate en Nayarit, México. Acta Zoológica Mexicana, 27, 115-121.
Carrizo, P., Gastelú, C., Longoni, P., & Klasman, R. (2008) Especies de trips (Insecta: Thysanoptera: Thripidae) en las flores de ornamentales. IDESIA, 26, 83-86.
Carrizo, P., & Klasman, R. (2001) Infestación por Frankliniella occidentalis durante el proceso de apertura floral en dos variedades de clavel para corte (Dianthus cariophyllus). Boletín de Sanidad Vegetal-Plagas, 27, 185-191.
Castresana, J., Gagliano, E., Puhl, L., Bado, S., Vianna, L., & Castresana, M. (2008) Atracción del trips Frankliniella occidentalis (Pergande) (Thysanoptera: Thripidae) con trampas de luz en un cultivo de Gerbera jamesonii (G.). IDESIA,26, 51-561.
Castro, U., Mesa, N.C., & Prieto, A. (1999) Búsqueda de enemigos naturales nativos de Frankliniella occidentalis (Pergande) (Thysanoptera: Thripidae), sobre Dendranthema grandiflorum en el municipio de Piendamo, Cauca. Revista Facultad Nacional de Agronomía Medellín, 52, 527-537.
Contreras, E.F., & Zamar, M.I. (2010) Descripción morfológica de los estados adultos e inmaduros de Neohydatothrips denigratus (De Santis) (Thysanoptera: Thripidae). Neotropical Entomology, 39, 384-390.
Curis, M.C., Zamar, M.I., Bertolaccini, M., & Kurtz, M.E. (2015) Primer registro de Gynaikothrips ficorum (Marchal) sobre Ficus microcarpa L. de la provincia de Santa Fe y de Gynaikothrips uzeli (Zimmermann) (Thysanoptera, Phlaeothripidae) sobre Ficus benjamina L. (Urticales: Moraceae) de la provincia de Jujuy, Argentina. Revista de la Sociedad Entomológica Argentina, 74(1-2), 85-88.
de Borbón, C.M. (2005) Los trips del suborden Terebrantia de la provincia de Mendoza. Ed. INTA, Buenos Aires.
de Borbón, C.M. (2013) Especies del género Frankliniella (Thysanoptera: Thripidae) registradas en la Argentina, una actualización. Revista de la Facultad de Ciencias Agrarias, 45, 259-284.
de Borbón, C.M., & Zamar, M.I. (2018) Two new species of Frankliniella (Thysanoptera: Thripidae) from Argentina with a key to species from Argentina and Chile. Zootaxa, 4369, 419-431.
de Borbón, C.M., Gracia, O., & De Santis, L. (1999) Survey of Thysanoptera occurring on vegetable crops as potential Tospovirus vectors in Mendoza, Argentina. Revista de la Sociedad Entomológica Argentina, 58(3-4), 59-66.
De Santis, L., Gallego de Sureda, A.E., & Merlo, E.Z. (1980) Estudio sinóptico de los tisanópteros argentinos (Insecta). Obra del Centenario del Museo de La Plata, ., 91-166.
Economía y Viveros (2016) Jujuy: destinan un proyecto de $70.000 para la producción de Lisianthus. Last access: 28-07-2020. Available at: http://www.economiayviveros.com.ar/febrero2016/actualidad_floricola_2.html
Farkas, P., Bagi, N., Szabó, Á., Ladányi, M., Kis, K., Sojnóczki, A., Reiter, D., Pénzes, B., & Fail, J. (2016) Biological control of thrips pests (Thysanoptera: Thripidae) in a commercial greenhouse in Hungary. Polish Journal of Entomology, 85, 437-451.
Fernández Acevedo, V. (2016) Las prácticas del cultivo de flores en el cinturón verde de La Plata y su compatibilidad con las Buenas Prácticas Agrícolas: el caso del Lilium. Tesis de grado. Facultad de Ciencias Agrarias y Forestales. Universidad Nacional de La Plata.
Fernández, R., & Fernández, H. (2013) Principales problemas sanitarios en la producción de flores de corte en el AMBA. Prácticas de manejo que predisponen a su aparición. Curso Sanidad en Cultivos Intensivos 2013. Módulo 4. Flores y ornamentales: el difícil arte de la belleza (ed. Mitidieri, M.S., & Francescangeli, N.), pp. 8-10. Ed. INTA, San Pedro, Buenos Aires.
García Baca, U. (1983) Ocurrencia, abundancia relativa e importancia de insectos y otros artrópodos en el crisantemo. Revista Peruana de Entomología,26, 31-39.
García-Gómez, A., Castaño-Meneses, G., & Palacios-Vargas, J.G. (2011) Distribución y diversidad de colémbolos (Hexapoda: Collembola) en el gradiente altitudinal de un bosque templado en México. Revista de Biología Tropical, 59, 315-327.
Grondona, M.M., & Morisigue, D. (2008) Caracterización microclimática de la región de los Valles en Jujuy. In: 4. Congreso Argentino de Floricultura y Plantas Ornamentales, 10as Jornadas Nacionales de Floricultura, 2008, pp. 685-691. Corrientes. Argentina.
Guerrero, R.J., & Sarmiento, C.E. (2010) Distribución altitudinal de hormigas (Hymenoptera, Formicidae) en la vertiente noroccidental de la Sierra Nevada de Santa Marta (Colombia). Acta Zoológica Mexicana (n.s.), 26, 279-302.
Haramboure, M., Reguilón, C., Alzogaray, R.A., & Schneider, M.I. (2014) First record of Chrysoperla asoralis and C. argentina (Neuroptera: Chrysopidae) in horticultural fields of La Plata associated with the sweet pepper (Capsicum annuum L.). Revista de la Sociedad Entomológica Argentina, 73(3-4), 187-190.
Held, D., & Pickens, J. (2018) Thrips: Pests of Ornamental Plants. Technical Report. Extension Alabama Cooperative Extension System. Alabama AyM University and Auburn University, USA.
Herrero, E., Camescasse, I., & Babbit, S. (2017) San Pedro: La producción de plantas ornamentales en Argentina y San Pedro. Revista INASE, 3, 35-36.
Janzen, D.H., Ataroff, M., Farinas, M., Reyes, S., Rinc, N.N., Soler, A., Soriano, P., & Vera, M. (1976) Changes in the arthropod community along an elevational transect in the Venezuelan Andes. Biotropica, 3, 193-203.
Jost, L. (2006) Entropy and diversity. Oikos, 113, 363-375.
Lacasa, A., Contreras, J., Sanchez, J.A., Lorca, M., & García, F. (1996) Ecology and natural enemies of Frankliniella occidentalis (Pergande, 1895) in south-east Spain. Folia Entomologica Hungarica, 62 (Suppl.), 67-74.
Lefebvre, M.G., Reguilón, C., & Kirschbaum, D.S. (2013) Evaluación del efecto de la liberación de Orius insidiosus (Hemiptera: Anthocoridae), como agente de control biológico de trips en el cultivo de frutilla. Revista de Investigaciones Agropecuarias, 39, 273-280.
Loera-Alvarado, E., Ortega-Arenas, L.D., Johansen-Naime, R.M., González-Hernández, H., Lomelí-Flores, R., Santillán-Galicia, M., & Ochoa-Martínez, D.L. (2017) Diversidad de tisanópteros en crisantemo [Dendranthema grandiflorum (Ramat.) Kitamura] var. Harman en Texcoco, estado de México. Acta Zoológica Mexicana, 33, 1-8.
Loomans, A.J.M., & Van Lenteren, J.C. (1990) Hymenopterous parasites as biological control agents of Frankliniella occidentalis (Perg.). IOBC, WPRS Bulletin, 13, 109-114.
Loomans, A.J.M., & Van Lenteren, J.C. (1995) Biological control of thrips pests. A review on thrips parasitoids. Wageningen Agricultural University Papers, 95, 88-201.
Margaría, C. (2012) Enemigos Naturales de Arañas y de Insectos Plaga: avispas esceliónidas (Hymenoptera: Platygastroidea), su importancia agronómica como potenciales agentes de control [Last access: 26-08-2020] Available at: https://www.agro.unlp.edu.ar/sites/default/files/paginas/margaria_2012_enemigos_naturales.pdf
Mejía Baena, C.M.; Ospina, L., & Palacio, M.M. (2018) Relación entre método directo e indirecto de monitoreo de trips (Insecta: Thysanoptera) en un cultivo comercial de crisantemo Dendranthema (Dc.) Des Moul (Asterácea) del Oriente Antioqueño, Colombia. Metroflor, 84, 25-32.
Mendes Carvalho, L., Paes Bueno, V.H., & Mendes, S.M. (2006) Ocorrência e flutuação populacional de tripes, pulgões e inimigos naturais em crisântemo de corte em casa de vegetação. Bragantia, 65, 139-146.
Miranda-Salcedo, M.A., & Loera-Alvarado, E. (2019) Fluctuación poblacional de enemigos naturales de trips (Thysanoptera: Thripidae) asociados a limón mexicano (Citrus aurantifolia Swingley) en Michoacán. Entomología Mexicana, ., 151-155.
Moreno, C.E., Barragán, F., Pineda, E., & Pavón, N.P. (2011) Reanálisis de la diversidad alfa: alternativas para interpretar y comparar información sobre comunidades ecológicas. Revista Mexicana de Biodiversidad, 82, 1249-1261.
Morisigue, D., Mata, D., Facciuto, G., & Bullrich, L. (2012) Floricultura. Pasado y presente de la floricultura argentina. Ed. INTA, Argentina.
Mound, L.A., & Marullo, R. (1996) The thrips of Central and South America: an introduction (Insecta, Thysanoptera). Memoirs on Entomology, International, .6 1-488.
Mound, L.A., & Matsunaga, J.N. (2017) The species of Haplothrips (Thysanoptera, Phlaeothripinae) and related genera recorded from the Hawaiian Islands. Zookeys, 662, 79-92.
Núñez Naranjo, D. (2016) Control biológico de trips en el cultivo de frutilla del noreste de la provincia de Buenos Aires: tasa de depredación de Orius insidiosus (Hemiptera: Anthocoridae) y su interacción con Neoseiulus californicus (Acari: Phytoseiidae). Tesis de Maestría. Facultad de Agronomía, Universidad de Buenos Aires.
Parrella, M.P., & Jones, V.P. (1987) Development of integrated pest management strategies of floricultural crops. Bulletin of the Entomological Society of America, 33, 28-34.
Píccolo, A., Giorgetti, M., & Chavez, D. (2008) Zonas Agro Económicas Homogéneas Salta-Jujuy. Estudios socioeconómicos de la sustentabilidad de los sistemas de producción y recursos naturales. Ed. INTA, Buenos Aires.
Ramos, C.S. (2018) Interacciones biológicas y patrones espaciales de diversidad: plantas, artrópodos herbívoros y herbivoría en gradientes altitudinales. Tesis Doctoral Facultad de Ciencias Exactas y Naturales. Universidad de Buenos Aires.
R Core Team (2018) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. Available at: https://www.R-project.org/
Reitz, S.R., & Funderburk, J. (2012) Management strategies for western flower thrips and the role of insecticides. Insecticides - Pest Engineering (ed. Perveen, F.), pp. 355-384. InTech, Rijeka, Croatia.
Riudavets, J. (1995) Predators of Frankliniella occidentalis and Thrips tabaci: A review. Wageningen Agricultural University Papers,95, 43-87.
Saini, E., & Polack, A. (2002) Enemigos naturales de los trips sobre flores de malezas. Revista de Investigaciones Agropecuarias, 29, 117.
Salazar Villareal, M. del C., Vallejo Cabrera F.A., & Salazar Villarreal, F.A. (2019) Inventarios e índices de diversidad agrícola en fincas campesinas de dos municipios del Valle del Cauca, Colombia. Entramado, 15, 264-274.
Sosa, M.R., Zamar, M.I., & Torrejon, S.E. (2017) Ciclo de vida y reproducción de Caliothrips phaseoli (Hood) Thysanoptera: Thripidae) sobre plantas fabáceas y solanáceas. Revista de la Sociedad Entomológica Argentina, 76(3-4), 1-6.
Van Lenteren, J.C., Loomans, A.J.M., Tommasini, M.G., Maini, S., Riudavets, J., Loomans, J.M., & Van Lenteren, J.C. (1995) Biological control of thrips pests. Wageningen Agricultural University Papers, 95, 1-87.
Villanova, I.; Brieva, S.S., & Ceverio, R. (2013) Producción y comercialización de flores de corte en el AMBA. Estudios socioeconómicos de los sistemas agroalimentarios y agroindustriales, 13, 1-45.
Vittar, M.C., Mansilla, C., & Rodríguez, M. (2013) Situación actual y perspectivas de la floricultura en el NOA. Investigación y Desarrollo Tecnológico para la Agricultura Familiar Región NOA, INTA Hornillos, Jujuy. Available at: https://inta.gob.ar/noticias/situacion-actual-y-perspectivas-de-la-floricultura-en-el-noa
Whittaker, R.H. (1972) Evolution and measurement of species diversity. Taxon, 21, 213-251.
Zamar, M.I. (2011). La diversidad de thrips del Cono Sur. El caso de las zonas áridas en Jujuy, Argentina. Métodos en Ecología y Sistemática, 6, 71-88.
Zamar, M.I., Neder de Román, L.E., Montero, T.E., Linares, M.A., Hamity, V.C., & Contreras, E.F. (2009) Incidencia de Thysanoptera en cultivos ornamentales y frutales de la Quebrada de Humahuaca (Jujuy, Argentina). Agraria, 66, 13-16.
Zamar, M.I., Neder, L.E., Linares, M.A., Hamity, V.C., Contreras, E.F., & Gomez, G.C. (2014) Tisanópteros (Insecta) asociados a plantas ornamentales de Jujuy (Argentina). Revista agronómica del Noroeste Argentino, 34, 261-262.
References
Zamar, M. I., & de Hamity, M. A. (1999). Interacción tisanóptero-planta en el modelo de manejo agroecológico. Idesia, 17, 101-110.
Notas de autor
gabhyalejo@gmail.com