Artículos
Preliminary report of the toxicity of Lippia junelliana (Lamiales: Verbenaceae) and Elionorus muticus (Poales: Poaceae) on Apis mellifera (Hymenoptera: Apidae)
Informes preliminares de la toxicidad de aceites esenciales de Lippia junelliana (Lamiales: Verbenaceae) y Elionorus muticus (Poales:Poaceae) en Apis mellifera (Hymenoptera: Apidae).
Preliminary report of the toxicity of Lippia junelliana (Lamiales: Verbenaceae) and Elionorus muticus (Poales: Poaceae) on Apis mellifera (Hymenoptera: Apidae)
Revista de la Sociedad Entomológica Argentina, vol. 81, núm. 1, 2022
Sociedad Entomológica Argentina
Recepción: 05 Agosto 2021
Aprobación: 25 Enero 2022
Publicación: 31 Marzo 2022
Abstract: Several phytochemicals, such as extracts and essential oils, are among the ecological alternatives that have been studied for the control of pathogens that affect humans, animals, and plants. Essential oils are aromatic oily liquids obtained from plant material and their use to control pathogens is widespread. However, they can have adverse effects when taken in high concentrations. The aim of this study was to evaluate the survival and palatability of a balanced-energy protein diet with the addition of two essential oils, Lippia junelliana (Mold.) Tronc. and Elionorus muticus (Spreng.) Kuntze, in newly hatched worker bees (Apis mellifera L.) during a period of 15 days. Consumption and number of live bees were measured daily for each treatment. For L. junelliana, our results showed a non-toxic trend in long-term survival trials (15 days) at low concentrations. In the case of E. muticus, a toxic effect with low survival was observed at all concentrations. Both essential oils showed good palatability, even at toxic levels. Our results allow us to propose the use of L. junelliana essential oil in field experiments of L. junelliana hives.
Keywords: Consumption, Honey bee, Phytochemical, Survival.
Resumen: Varios fitoquímicos, como extractos y aceites esenciales, se encuentran entre las alternativas ecológicas que se han estudiado para el control de patógenos que afectan a humanos, animales y plantas. Los aceites esenciales son líquidos oleosos aromáticos obtenidos de materiales vegetales. Sin embargo, las plantas aromáticas pueden tener efectos adversos cuando se suministran en dosis elevadas. El objetivo de este estudio fue evaluar la supervivencia y palatabilidad de una dieta proteica energética balanceada con la adición de dos aceites esenciales, Lippia junelliana (Mold.) Tronc. y Elionorus muticus (Spreng.) Kuntze, en abejas (Apis mellifera L.) obreras recién nacidas durante un período de 15 días. El consumo y el número de abejas vivas se midieron diariamente para cada tratamiento. Para L. junelliana, nuestros resultados mostraron ausencia de toxicidad en los ensayos de supervivencia a largo plazo (15 días) a concentraciones bajas. Por otro lado, E. muticus fue tóxico, con bajos valores de supervivencia en todas las dosis. Ambos aceites esenciales mostraron una buena palatabilidad, incluso a niveles tóxicos. Estos resultados permitirían el uso del aceite esencial de L. junelliana en colmenas de A. mellifera a campo.
Palabras clave: Abeja melífera, Consumo, Fitoquímico, Supervivencia.
INTRODUCTION
Agroecosystems are directly linked to environmental change, which could affect their ability to maintain long-term agricultural functions (Tylianakis et al., 2008). In flower-insect networks, it has been observed that reducing the number of botanical families also reduces the diversity of floral visiting insects and the interactions recorded in the networks of an ecosystem (Moreira et al., 2020). Changes in the ecosystem generate critical impacts on human food security, because 90% of the world's flowering plant species (angiosperms) are pollinated by insects (Ollerton et al., 2011), including 70% of the world food-producing crops (Hinkel et al., 2014).
Current conventional production systems use synthetic pesticide as the main method to control insects (Gontijo et al., 2013) despite the high risks of human and environmental contamination and adverse effect on non-target organisms (Sánchez-Bayo & Goka, 2014), such as the western honey bees Apis mellifera L (Hymenoptera: Apidae). The association of pesticides with pollinator decline has been reported around the world (Kulhanek et al., 2017; Requier et al., 2018). Moreover, recent studies have reported losses of 20-30% of honey bee colonies in several Latin American countries (Requier et al., 2018) due to the presence of different parasites and pathogens such as Varroa destructor (Acari) (Giacobino et al., 2018); Vairimorpha (=Nosema.ceranae (Microsporidia) (Genchi García et al., 2019; Maggi et al., 2020); Paenibacillus larvae subsp larvae (Bacteria) (Maggi et al., 2016); Ascosphaera apis (Ascomycota) (Reynaldi et al., 2003; Maggi et al., 2020) and viruses (Salina et al., 2021). To make matters worse, the decrease in semi-natural habitats and the alteration of the availability of floral resources have led to a decrease in pollinator populations, including honey bees, in agricultural areas (Potts et al., 2010; Requier & Leonhardt, 2020).
Concomitantly, the demand for healthy natural food has grown worldwide, and Latin America is positioning itself as an important region for food production following agroecological guidelines (Altieri et al., 2011; Giraldo et al., 2019). As a result, there is extensive research on the consequences of synthetic chemicals on the environment in general and insect communities in particular. An alternative to synthetic chemicals, which is consistent with the agroecological approach, is the use of plant-derived products (phytochemicals) such as extracts and essential oils, which can contribute to the sustainability of production (Brzozowski & Mazourek, 2018). Chouhan et al. (2017) described aromatic oily liquids called essential oils (EOs) obtained from plant materials (leaves, buds, fruits, flowers, herbs, twigs, bark, wood, roots and seeds). Steaming or hydro-distillation is the most common method for obtaining EOs at a commercial scale. Having a density generally lower than that of water, EOs are volatile, liquid, limpid, lipid-soluble, rarely colored, and soluble in organic solvents. Being natural mixtures of very complex nature, EOs may consist of about 20-60 major components and others are trace elements. The amount of the components in EOs varies amongst different species and plant tissue. They are chemically derived from terpenes and their oxygenated derivatives (terpenoids), which are aromatic and aliphatic acid esters and phenolic compounds.
Aromatic plants and EOs have also been reported to contain bioactive compounds that act as a potential multifunctional food supplement (Zeng et al., 2015; Simitzis, 2017). EOs are also used for their antimicrobial properties against bacteria, fungi and viruses in humans and animals (Chouhan et al., 2017; Wińska et al., 2019; Aljaafari et al., 2021). Several phytochemicals, such as EOs, plant extracts and other nutraceuticals have been evaluated in vitro for the control of honey bee pathogens and parasites such as V. destructor (Lin et al., 2020); P. larvae (Pellegrini et al., 2017); A. apis (Nardoni et al., 2018; Tutun et al., 2018) and V. ceranae (Yilmaz et al., 2020). However, very few in vivo studies have been conducted on honey bee colonies to control P. larvae (Albo et al., 2003; Gende et al., 2009), V. destructor (Gregorc et al., 2019), and V. ceranae (Bravo et al., 2017; Porrini et al., 2017; Borges et al., 2020). Given that it is known that high concentrations of medicinal and aromatic plants can have adverse effects (Albo et al., 2010; Borges et al., 2020), the effectiveness and safety of any EO, plant extract or nutraceutical must be demonstrated to ensure that it can be used safely in honey bees (Kammon, 2017) before testing its efficacy to control honey bee pathogens’.
The aim of this study was to evaluate the survival and palatability of a balanced-energy protein diet supplemented with one of two essential oils, one obtained from Lippia junelliana (Mold.) Tronc. (Lamiales: Verbenaceae) and the other from Elionorus muticus (Spreng.) Kunth. (Poales: Poaceae) on worker honey bees from two to three days after emergence and during a 15-day period. Several EOs concentrations were evaluated with the purpose of determining which concentration/s of EOs do not cause deleterious effects in honey bees. This information is crucial to test EOs as potential control agents of honey bee pathogens.
MATERIAL AND METHODS
Essential oils
The EOs used in this study were derived from L. junelliana and E. muticus, two widespread native species, the former in mountainous regions of central and northwestern Argentina (Juarez et al., 2015) and the latter in the Pampa biome, which includes Brazil, Argentina and Uruguay (Fuller et al., 2014). The EO of L. junelliana was distilled from flowering materials collected from a population of La Higuera, Córdoba, by the Aromatics group of the Biological Resources Institute (BRI), INTA Hurlingham, Buenos Aires, Argentina. This EO was obtained following Juarez et al. (2015) technique with modifications. Briefly, aerial parts were hydrodistilled using a Clevenger device, and its composition was determined by GC/FID with a KNK device 3006 and by GC/MS with an HP 6890 machine. Finally, the compounds were identified by their retention indices and mass spectra (unpublished results). The aerial material had a yield of 0.92-2.29% and was classified as chemotype I. For E. muticus, the material was extracted from flowering plants in the experimental plots of BRI, Argentina. Elionorus muticus components were determined in the same way as for L. junelliana. The E. muticus aerial material had a yield of 0.39%, which was similar to the results reported by Padula et al. (1977). Table I shows the main components of both EOs.
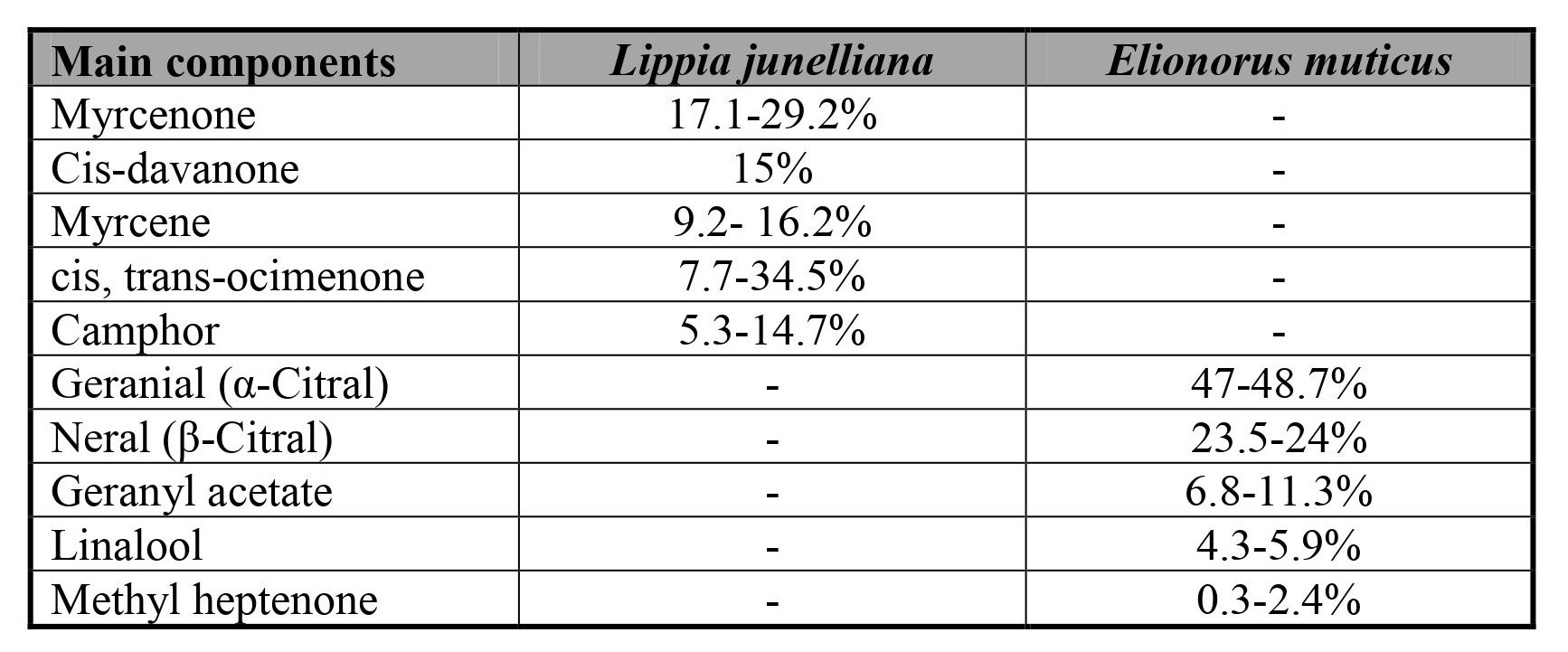
Table I. Composition of the essential oils tested in this study.
Experimental procedure
The experimental procedure was similar to the one used by Porrini et al. (2017), with modifications. A healthy colony with a new queen was selected from the experimental apiary of the Animal Production Course, School of Agricultural and Forest Sciences, National University of La Plata, Argentina (34°54'38.4"S; 57°55'37.6"W). In this colony, a technical basket was placed with the queen with an empty brood frame. After checking the presence of red-eyed pupae, we transferred the brood frame to the laboratory to allow incubation under controlled conditions (32 °C and 60% RH).
Upon emergence, bees were fed with a sucrose solution and a pollen substitute (Nutribee®), free of bee pathogens. At the age of 2-3 days, worker honey bees were anesthetized following Albo et al. (2010), with modifications, to confine them in the test flasks (experimental unit, see below). Thirty honey bees were included in each flask and allowed to recover spontaneously; those that did not show vital signs were replaced in the flasks.
The experimental unit consisted of 500 cm3 transparent plastic flasks with pressure lids. Holes of 3 mm in diameter were made on the walls of the flask and lid to allow air circulation. Two 1.5 ml conical Eppendorf-type tubes (total 3 ml) were used as feeders. Both feeders were placed on the side walls, and perforated on the outer side to enable the administration of treatments.
The diet to provide the honey bees was prepared daily to avoid evaporation or crystallization, and administered every day for 15 days. The daily consumption of each diet was calculated (in grams) by subtracting the final and initial weight of the feeder. This value was transformed into µl/l according to the density of the solution. Feeders were replaced every day and the number of live honey bees/flask was recorded daily.
Treatments
Following Albo et al. (2017), the diets with the EOs were prepared using 2.5% v/v ethanol 70° as diluent and then formulated in a sucrose solution at 50% w/v in sterile distilled water with pollen substitute (Nutribee®) at 1% w/v. A total of 9 different treatments were prepared as follows: L. junelliana and E. muticus OEs at concentrations of 500, 1000, 2000 and 4000 μl/l each and a control treatment consisting of 50% w/v sucrose solution without essential oil. Each treatment had four repetitions.
Statistical analysis
The survival rates were calculated, plotted and compared by means of Kaplan-Meier survival analysis and Gehan-Breslow tests (P < 0.05) which estimates also the median survival time (GraphPad, 2019). Average daily food consumption data were analyzed by means of a Krüskal-Wallis test (P < 0.05) and compared to the control group using Dunn's method (P < 0.05). The Kruskal Wallis test was carried out with the statistical software INFOSTAT, version 2019 (Di Rienzo, 2019). For Gehan-Breslow test and Dunn's method, the GraphPad Software, version 8.0.0 was used (GraphPad, 2019).
RESULTS
Survival analysis
The survival of the honey bees to systemic feeding with the EOs of E. muticus and L. junelliana showed statistically significant differences (P < 0.05). In the analysis of the curves, three different defined groups were observed. The first group included the bees fed L. junelliana with 4000 µl/l, which differed significantly from the control group (P = 0.0368) with an average survival time (50% of the population) of 9.34 days. The second group included bees fed L. junelliana at concentrations 500, 1000 and 2000 µl/l which showed non-statistically significant differences from the control group (P > 0.05), with an estimated survival time (50% of the population) that ranged from 14.14 to 15.94 days (Fig. 1.a). Lastly, the third group included the four concentrations of E. muticus, which showed the highest toxicity and differed significantly from the control group (P < 0.0001), with estimated survival time (50% of the population) ranging from 5.10 to 6.54 days (Fig. 1.b).
![Fig.1. Survival curves of honey bees. (a) Survival curves for treatments of Lippia junelliana. (b) Survival curves for treatments of Elionorus muticus. Estimated survival time (50% of the population) given by Gehan-Breslow Test shown between brackets. Statistical differences between treatments denoted with asterisks: [*] P < 0.05]; [**] P < 0.0001.](../322070162007_gf3.png)
Fig.1. Survival curves of honey bees. (a) Survival curves for treatments of Lippia junelliana. (b) Survival curves for treatments of Elionorus muticus. Estimated survival time (50% of the population) given by Gehan-Breslow Test shown between brackets. Statistical differences between treatments denoted with asterisks: [*] P < 0.05]; [**] P < 0.0001.
Consumption
The consumption showed significant differences in EO between treatments (Krüskal-Wallis H =75.67, P < 0.0001). The post-hoc Dunn’s Test revealed significant differences only for the concentrations of 500 µ/l (48.11 ± 1.66 µl/bee/day) (P = 0.0007) and 1000 µl/l (47.73 ± 1.50 µl/bee/day) of L. junelliana (P = 0.0012), when compared to the control (38.80 ± 1.06 µl/bee/day). The E. muticus treatments showed no significant differences from the control (P > 0.05) (Fig. 2). The lowest concentrations tested of L. junelliana (500 and 1000 μl/l) were consumed more than the control. The rates of survival of these treatments were the same as for controls.
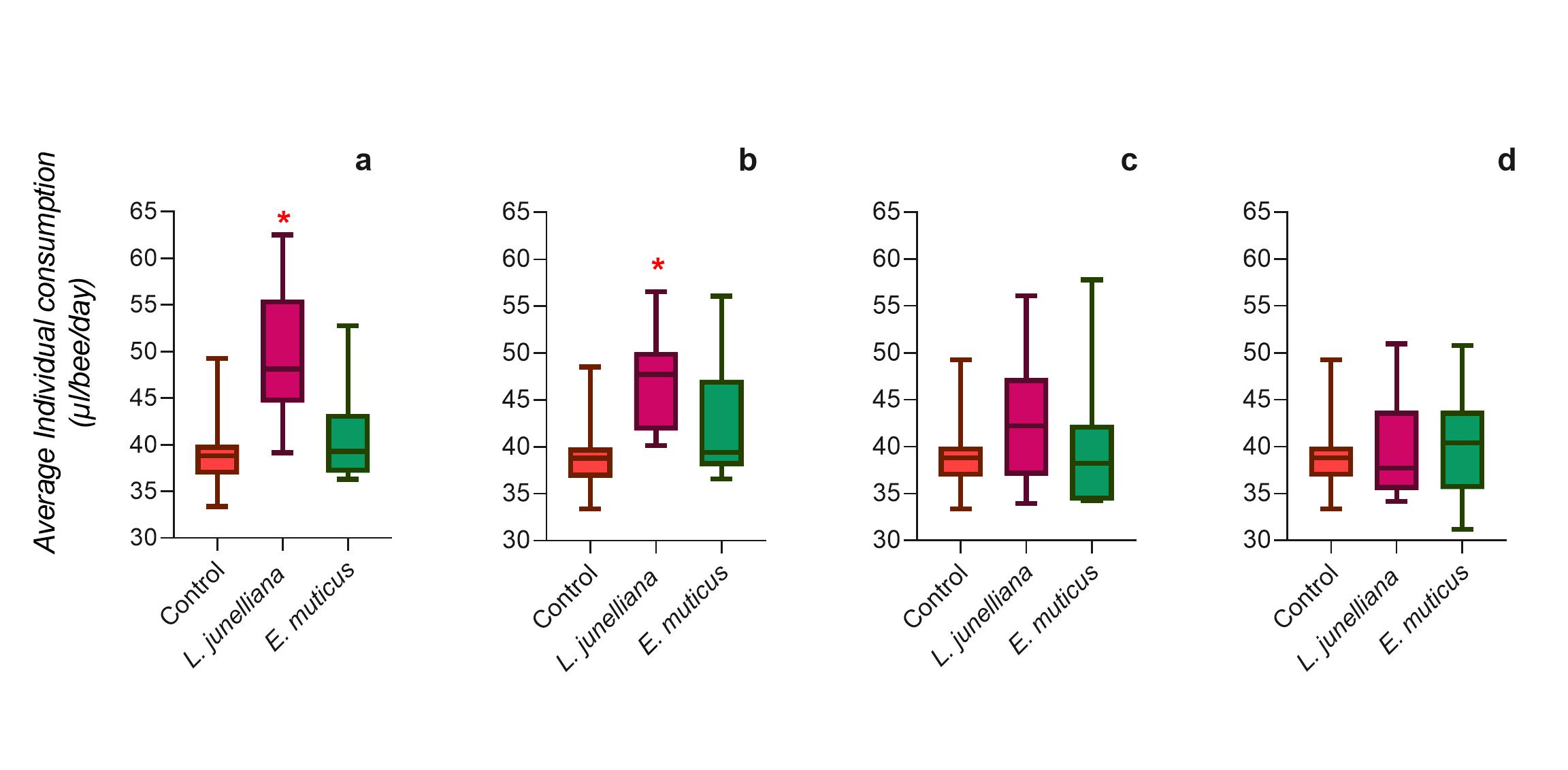
Fig. 2. Box- plot of consumption (µl/bee/day) for treatments. (a) Concentration of 500µl/l; (b) Concentration of 1000 µl/l; (c) Concentration of 2000 µl/l; (d) Concentration of 4000µl/l. Statistical differences between treatments (P < 0.05) denoted with an asterisk.
DISCUSSION
Essential oils are among the ecological alternatives that have been studied to control pathogens (Ellse & Wall, 2014; Chaudhari et al., 2021), and also as natural preservatives, additives or supplements in animal feed given their content of bioactive compounds and aromatic characteristics (Zeng et al., 2015; Stevanović et al., 2018). In honey bees, the effects of EOs to control pathogens have been tested with a great variety of methodologies and diverse results (Albo et al., 2010; Porrini et al., 2017; Borges et al., 2020). Here we tested two different EOs at four different concentrations and found that the survival and consumption was affected both by the EO and the concentration given.
Our results show that the highest tested concentrations of both EOs (4000 µl/l) resulted in the lowest survival, even when consumption was similar compared to the control. Several authors have found that high oral concentrations of EOs cause acute toxicity in bees (Albo et al., 2003; Albo et al., 2010; Borges et al., 2020), as do long-term treatments in adult honey bees (Bravo et al., 2017; Borges et al., 2020). In general, high concentrations of EOs have been found to cause high toxicity and low consumption (Albo et al., 2003), possibly due to the low palatability of toxic EO. Here, the EOs of L. junelliana and E. muticus showed good palatability, even at toxic levels.
With regard to L. junelliana, recent in vitro studies have shown that it effectively controls several fungi, such as Candida krusei (Castellani) Berkhout, C. albicans (C.P.Robin) Berkhout and Aspergillus spp.fumigati section, although it was not effective to control Fusarium spp. (Córdoba et al., 2016, 2019). Lippia junelliana EO showed a Mean Lethal Doses (LD50) > 100 µg a.p. per bee at 24, 48 and 72 hours on adult bees, which places it within the range of “virtually non-toxic” products (Albo et al., 2014). We also observed this non-toxic trend in our long-term survival trials (15 days) at low concentrations. Previous reports of several EOs, such as Laurus nobilis L, Origanum vulgare L, and Salvia rosmarinus Spenns (Rosmarinus officinalis), proved to be non-toxic at similar concentrations to the ones used in our trial (Porrini et al., 2017). In addition, L. junelliana EO showed higher consumption at low concentrations (500 and 1000 μl/l) respect to control, which indicates a good palatability without toxic effects for adult honey bees.
With regard to E. muticus, there are some preliminary results of in vitro tests exploring its capacity to control human and animal pathogens (Lameck et al., 2016; Puppin et al., 2018; Lazzarini et al., 2018). However, studies of this EO on honey bee pathogens have not been reported yet. In relation to in vivo tests to date, only a few trials have been reported on the control of human diseases. Lameck et al. (2016) have demonstrated that E. muticus was effective in vitro for the control of some yeasts such as C. albicans, C. krusei and C. neoformans Vuillemin, using the broth diffusion technique, with Minimal Inhibitory Concentration (MIC) values ranging from 0.5 to 5 μg/ml and Minimal Fungicidal Concentration (MFC) values of 2.5 to 10 μg/ml. Recent studies showed that the effect of this EO was not fungicidal, but fungistatic against C. krusei, C. utilis with MIC = 0.0312 mg/ml and MFC = 0.125 mg/ml, and against C. parapsilosis with MIC = 0.0625 mg/ml and MFC = 0.5 mg/ml (Puppin et al., 2018).
Also, Lazzarini et al. (2018) showed that this EO had no inhibitory effect against the thermal-dimorphic fungi Sporothrix schenckii and Sporothrix brasiliensis in dilutions ranging from 8.14 to 1.041 μg/ml. In relation to bacteria, this EO has antimicrobial activity against Escherichia coli, Staphylococcus coagulase negative, S. aureus, S. pseudintermedius, S. uberis, P. aeruginosa and Bacillus cereus through micro-dilution techniques in broth and disk diffusion, with MIC values ranging from 1 mg/ml to 150 mg/ml (Hess, 2007; Puppin, 2018). In vivo tests demonstrated a fumigant action on the agricultural pest Tetranychus urticae C.L.Koch (Acari) at low concentrations (0.03-1 µl/l) (Hauschild et al., 2020). To our knowledge, there is no report of the use of E. muticus for controlling bee pathogens.
Other authors have used the EO of Litea cubeba (Lour.) Pers (Lauraceae), which has a similar composition to E. muticus, for the control of same toxigenic fungi, including Aspergillus flavus, A. carbonarius, and Penicillium viridicatum that contaminated food and agricultural commodities (Wang et al., 2018), and also for controlling A. apis and A. flavus (aetiological agents of chalkbrood and stonebrood, respectively) (Nardoni et al., 2018). This information is relevant to investigate which could be the responsible compounds and to select other EOs to be tested.
In all, our study provides novel information about the EOs tested. Lippia junelliana produces higher survival and better consumption at lower concentrations than E. muticus. Further studies are needed to evaluate E. muticus at a lower concentration for control of the most prevalent fungal honey bee pathogens, as A. fumigatus, A. apis, or even Vairimorpha spp. (Tokarev et al., 2020). Our results show that certain concentrations of L. junelliana do not affect survival, opening the possibility to consider its use as an agent to control pathogens. Nevertheless, it will be necessary to carry out a more detailed evaluation of the impact of L. junelliana on the pathogen community and the possibility of providing lower doses or for less time, to determine its use in honey bee hives. Although we used only one colony of honey bees during our study, results allowed us to determine a different response associated with the EO tested but the variability in the response associated to the origin of the bees could not be determined. Future studies involving more colonies are required to test colony associated variability.
REFERENCES
Albo, G.N., Altamirano, R., Reynaldi, F. J., & Córdoba, S.B. (2017) Lippia alba essential oil as potential control agents of chalkbrood. 45th Apimondia International Apicultural Congress. 2017, Istambul, Turkey. Pp. 234.
Albo, G.N., Reynaldi, F. J, Elder, H., Juárez, M.A., & Córdoba, S.B. (2014) Control in vitro de Ascosphaera apis con Schinus molle y Lippia junelliana. Efectos tóxicos potenciales en abeja melífera. XI Congreso Latinoamericano de Apicultura FILAPI 2014. Misiones, Argentina.
Albo, G.N., Henning, C., Reynaldi, F.J., Ringuelet, J., & Cerimele, E. (2010) Median Lethal Dose (LD50) of some essential oils and biocides effective for the control of Ascosphaera apis on Apis mellifera L. REDVET, 11(10).
Albo, G.N., Henning, C., Ringuelet, J., Reynaldi, F.J., Giusti, M.R.D., & Alippi, A.M. (2003) Evaluation of some essential oils for the control and prevention of American Foulbrood disease in honey bees. Apidologie, 34, 417-427.
Aljaafari, M.N., AlAli, A.O., Baqais, L., Alqubaisy, M., AlAli, M., Molouki, A., Ong-Abdullah, J., Abushelaibi, A., Lai, K.-S., & Lim, S.-H.E. (2021) An overview of the potential therapeutic applications of essential oils. Molecules,26, 628.
Altieri, M.A., & Toledo, V.M. (2011) The agroecological revolution in Latin America: rescuing nature, ensuring food sovereignty and empowering peasants. The Journal of Peasant Studies,38, 587-612.
Borges, D., Guzman-Novoa, E., & Goodwin, P.H. (2020) Control of the microsporidian parasite Nosema ceranae in honey bees (Apis mellifera) using nutraceutical and immuno-stimulatory compounds. PLOS ONE, 15(1), e0227484.
Bravo, J., Carbonell, V., Sepúlveda, B., Delporte, C., Valdovinos, C.E., Martín-Hernández, R., & Higes, M. (2017) Antifungal activity of the essential oil obtained from Cryptocarya alba against infection in honey bees by Nosema ceranae. Journal of Invertebrate Pathology, 149, 141-147.
Brzozowski, L., & Mazourek, M. (2018) A sustainable agricultural future relies on the transition to organic agroecological pest management. Sustainability, 10(6), 2023.
Chaudhari, A.K., Singh, V.K., Kedia, A., Das, S., & Dubey, N.K. (2021) Essential oils and their bioactive compounds as eco-friendly novel green pesticides for management of storage insect pests: prospects and retrospects. Environmental Science and Pollution Research, 28(15), 18918-18940.
Chouhan, S., Sharma, K., & Guleria, S. (2017) Antimicrobial activity of some essential oils - Present status and future perspectives. Medicines, 4(3), 58.
Córdoba, S., Vivot, W., Szusz, W., & Albo, G. (2019) Antifungal activity of essential oils against Candida species isolated from clinical samples. Mycopathologia, 184(5), 615-623.
Córdoba, S., Vivot, W., Szusz, W., Isla, G., Hevia, A., Abrantes, R., Della Vedova, R., & Albo, G. (2016) In vitro activity of essential oils extracted from aromatic plants against Aspergillus spp., and Fusarium spp., of clinical origin. In Proceedings of XXIII Latin American Congress of Microbiology, XIV Argentine Congress of Microbiology, IV Latin American Congress of Microbiology of Medicines and Cosmetics (CLAMME), Meeting of the Latin American Society of Tuberculosis and other Mycobacteriosis (SLAMTB), 2016, Rosario, Santa fé, Argentina.
Di Rienzo, J. A., Casanoves, F., Balzarini, M., Gonzalez, L., Tablada, M., & Robledo, C. (2019) InfoStat statistical software student version. Córdoba, Argentina: InfoStat Group, FCA, National University of Córdoba, Argentina. http://www. mfos-tat. com. ar/index. php.
Ellse, L., & Wall, R. (2014) The use of essential oils in veterinary ectoparasite control: a review. Medical and Veterinary Entomology, 28(3), 233-243.
Füller, T.N., Bertrand, C., Simon, A., Inchausti de Barros, I.B., & Barbosa Neto, J.F. (2014) Elionurus muticus as an alternative source of citral from Pampa biome, Brazil. Journal of Oleo Science, 63(11), 1109-1116.
Genchi Garcia, M.L., Plischuk, S., Bravi, C.M., & Reynaldi, F.J. (2019) An overview on honeybee colony losses in Buenos Aires Province, Argentina. Sociobiology, 66(1), 75-80.
Gende, L.B., Maggi, M.D., Fritz, R., Eguaras, M.J., Bailac, P.N., & Ponzi, M.I. (2009) Antimicrobial activity of Pimpinella anisum and Foeniculum vulgare essential oils against Paenibacillus larvae. Journal of Essential Oil Research, 21(1), 91-93.
Giacobino, A., Pacini, A., Molineri, A., Rodríguez, G., Crisanti, P., Bulacio Cagnolo, N., Merke, J., Orellano, E., Bertozzi, E., et al. (2018) Potential associations between the mite Varroa destructor and other stressors in honeybee colonies (Apis mellifera L.) in temperate and subtropical climate from Argentina. Preventive Veterinary Medicine, 159, 143-152.
Giraldo, O.F., & McCune, N. (2019) Can the state take agroecology to scale? Public policy experiences in agroecological territorialization from Latin America. Agroecology and Sustainable Food Systems, 43(7-8), 785-809.
Gontijo, P.C., Picanço, M.C., Pereira, E.J.G., Martins, J.C., Chediak, M., & Guedes, R.N.C. (2013) Spatial and temporal variation in the control failure likelihood of the tomato leaf miner, Tuta absoluta. Annals of Applied Biology, 162(1), 50-59.
GraphPad Software Inc. (2019). Graphpad prism. Version 8.0. For windows, San Diego, California, EE. UU. Www.graphpad.com.
Gregorc, A., & Sampson, B. (2019) Diagnosis of Varroa Mite (Varroa destructor) and sustainable control in honey bee (Apis mellifera) colonies. A review. Diversity. 11(12), 243.
Hauschild, R., Ott, A., & da Silva, M.A.S. (2020) Ação fumigante de óleos essenciais de Baccharis dracunculifolia e Elionurus muticus contra o Ácaro rajado (Tetranychus urticae, Koch 1836) em morangueiro. Anais do XI Congresso Brasileiro de Agroecologia, São Cristóvão, Sergipe. 2020. Cadernos de Agroecologia. 15(2).
Hess, S.C., Peres, M.T.L.P., Batista, A.L., Rodrigues, J.P., Tiviroli, S.C., Oliveira, L.G.L., Santos, C.W.C., Fedel, L.E.S., Crispim, S.M.A., et al. (2007) Evaluation of seasonal changes in chemical composition and antibacterial activity of Elyonurus Muticus (Sprengel) O. Kuntze (Gramineae). Química Nova. 30(2), 370-373.
Hinkel, J., Lincke, D., Vafeidis, A.T., Perrette, M., Nicholls, R.J., Tol, R.S., Marzeion, B., Fettweis, X., Ionescu, C., & Levermann, A. (2014) Coastal flood damage and adaptation costs under 21st century sea-level rise. Proceedings of the National Academy of Sciences. 111(9), 3292-3297.
Juárez, M.A., Elechosa, M.A., Viturro, C.I., Molina, A.C., & Martinez, A.J. (2015) Chemical diversity in the essential oils of Lippia junelliana (Verbenaceae) in 30 towns in central and northwestern Argentina IV National Conference on native aromatic plants and their essential oils Lilloa 52 (Suppl 1): 42-44.
Kammon, A.M. (2017) The future use of medicinal plants as alternatives to antibiotics in animal health and production. Approaches in Poultry, Dairy & Veterinary Sciences. . 1(1). 1-2.
Kulhanek, K., Steinhauer, N., Rennich, K., Caron, D.M., Sagili, R.R., Pettis, J.S., James, D., Ellis, J.D., Wilson M.E., et al. (2017) A national survey of managed honey bee 2015-2016 annual colony losses in the USA. Journal of Apicultural Research, 56(4), 328-40.
Lameck, S., Chagonda, L., & Fungirayi, B. (2016) Antifungal activity of the essential oil of Elionurus Muticus (Spreng) Kunth from Zimbabwe against Candida albicans, C. krusei and Cryptococcus neoformans. Journal of Multidisciplinary Engineering Science and Technology, ., 2458-9403.
Lazzarini, J. de A., Rezende, N., Raposo, B., Lima, R.M., Brandão, A., Leite, M.N., & Crotti, A.E.M. (2018) Pimenta pseudocaryophyllus Gomes Landrum and Elionurus muticus (Spreng) Kunth and their antifungal properties against Sporothrix schenckii and Sporothrix brasiliensis. Journal of Multidisciplinary Engineering Science and Technology (JMEST) 5(12), 9.
Lin, Z., Su, X., Wang, S., Ji, T., Hu, F.-L& Zheng, H.-Q. (2020) Fumigant toxicity of eleven Chinese herbal essential oils against an ectoparasitic mite (Varroa destructor) of the honey bee (Apis mellifera). Journal of Apicultural Research, 59(2), 204-210.
Maggi, M., Quintana, S., Revainera, P.D., Porrini, L.P., Meroi Arcerito, F.R. Fernandez de Landa, G., Brasesco, C., Di Gerónimo, V.., Ruffinengo, S.R., & Eguaras, M.J. (2020) Biotic stressors affecting key apiaries in Argentina. Bee World. 97(2), 45-52.
Maggi, M., Antúnez, K., Invernizzi, C., Aldea, P., Vargas, M., Negri, P., Brasesco, C., De Jong, D., Teixeira, E.W., & Principal, J. (2016) Honeybee health in South America. Apidologie, 47(6), 835-854.
Moreira, L.T., Falcão, L.A.D., & de Araújo, W.S. (2020) Geographical patterns in the architecture of neotropical flower-visitor networks of hummingbirds and insects. Zoological Studies. e59.
Nardoni, S., D’Ascenzi, C., Rocchigiani, G., Papini, R.A., Pistelli, L., Formato, G., Najar, B., & Mancianti, F. (2018) Stonebrood and chalkbrood in Apis mellifera causing fungi: in vitro sensitivity to some essential oils. Natural Product Research, 32(4), 385-390.
Ollerton, J., Winfree, R., & Tarrant, S. (2011) How many flowering plants are pollinated by animals? Oikos, 120(3), 321-326.
Padula, L.Z., Collura, A.M., Rondina, R.V.D., Mizrahi, I., Coussio, J.D., & Juarez, M.A. (1977) Experimental cultivation of Elyonurus muticus (Sprengel) O. Kuntze in Argentina. Qualitative and quantitative analysis of its essential oil. Rivista italiana essenze, profumi, piante officinali, aromi, saponi, cosmetici, aerossol, 59(2), 58-63.
Pellegrini, M.C., Alonso‐Salces, R.M., Umpierrez, M.L., Rossini, C., & Fuselli, S.R. (2017) Chemical composition, antimicrobial activity, and mode of action of essential oils against Paenibacillus larvae, etiological agent of American foulbrood on Apis mellifera. Chemistry & Biodiversity. 14(4), e1600382.
Porrini, M.P., Garrido, P.M., Gende, L.B., Rossini, C., Hermida, L., Marcángeli, J.A., & Eguaras, M.J. (2017) Oral administration of essential oils and main components: Study on honey bee survival and Nosema ceranae development. Journal of Apicultural Research, 56(5), 616-624.
Potts, S.G., Biesmeijer, J.C., Kremen, C., Neumann, P., Schweiger, O., & Kunin, W.E. (2010) Global pollinator declines: trends, impacts and drivers. Trends in Ecology & Evolution, 25(6), 345-353.
Puppin, D. de G.P.B. (2018) Ação antifúngica e toxicidade do óleo essencial de Elionurus muticus (capim-limão brasileiro). Tesis de Maestria. Universidade Estadual de Campinas. Disponible en:
Requier, F., & Leonhardt, S.D. (2020) Beyond flowers: including non-floral resources in bee conservation schemes. Journal of Insect Conservation, 24(1), 5-16.
Requier, F., Antúnez, K., Morales, C.L., Aldea Sánchez, P., Castilhos, D., Garrido, P.M., Giacobino, A., Reynaldi, F.J., Rosso Londoño, J.M., et al. (2018) Trends in beekeeping and honey bee colony losses in Latin America. Journal of Apicultural Research, 57(5), 657-662.
Reynaldi, F.J., López, A.C., Albo, G.N., & Alippi, A.M. (2003) Differentiation of Ascosphaera apis isolates by rep-PCR fingerprinting and determination of chalkbrood incidence in Argentinean honey samples. Journal of Apicultural Research, 42(4), 68-76.
Salina, M.D., Garcia, M.L.G., Bais, B., Bravi, M.E., Brasesco, C., Maggi, M., Pecoraro, M., Larsen, A., Sguazza, H.G., & Reynaldi, F.J. (2021) Viruses that affect Argentinian honey bees (Apis mellifera). Archives of Virology, 166, 1533-1545.
Sanchez-Bayo, F., & Goka, K. (2014) Pesticide residues and bees - A risk assessment. PLOS ONE, 9(4), e94482.
Simitzis, P.E. (2017) Enrichment of animal diets with essential oils-a great perspective on improving animal performance and quality characteristics of the derived products. Medicines, 4(2), 35.
Stevanović, Z.D., Bošnjak-Neumüller, J., Pajić-Lijaković, I., Raj, J., & Vasiljević, M. (2018) Essential oils as feed additives - Future perspectives. Molecules, 23(7), 1717.
Tokarev, Y.S., Huang, W.-F., Solter, L.F., Malysh, J.M., Becnel, J.J., & Vossbrinck, C.R. (2020) A formal redefinition of the genera Nosema and Vairimorpha (Microsporidia: Nosematidae) and reassignment of species based on molecular phylogenetics. Journal of Invertebrate Pathology, 169, 107279.
Tutun, H., Koç, N., & Kart, A. (2018) Plant essential oils used against some bee diseases. Turkish Journal of Agriculture - Food Science and Technology, 6(1), 34-45.
Tylianakis, J.M., Didham, R.K., Bascompte, J., & Wardle, D.A. (2008) Global change and species interactions in terrestrial ecosystems. Ecology Letters, 11(12), 1351-1363.
Wang, H., Yang, Z., Ying, G., Yang, M., Nian, Y., Wei, F., & Kong, W. (2018) Antifungal evaluation of plant essential oils and their major components against toxigenic fungi. Industrial Crops and Products, 120, 180-186.
Wińska, K., Mączka, W., Łyczko, J., Grabarczyk, M., Czubaszek, A., & Szumny, A. (2019) Essential oils as antimicrobial agents - Myth or real alternative? Molecules, 24(11), 2130.
Yilmaz, F., Kuvanci, A., Konak, F., Öztürk, S.H., & Şahi̇n, A.E. (2020) The effects of some essential oils against Nosemosis. Bee Studies, 12(2), 37-41.
Zeng, Z., Zhang, S., Wang, H., & Piao, X. (2015) Essential oil and aromatic plants as feed additives in non-ruminant nutrition: a review. Journal of Animal Science and Biotechnology, 6(1), 7.