Composition and structure of the Chironomidae (Insecta: Diptera) assemblage of Ventania hill system streams (Argentina) along an altitudinal gradient
Composición y estructura del ensamble de Chironomidae (Insecta: Diptera) del sistema serrano de Ventania (Argentina) a lo largo de un gradiente altitudinal
Composition and structure of the Chironomidae (Insecta: Diptera) assemblage of Ventania hill system streams (Argentina) along an altitudinal gradient
Revista de la Sociedad Entomológica Argentina, vol. 81, núm. 2, 2022
Sociedad Entomológica Argentina
Recepción: 03 Enero 2022
Aprobación: 20 Junio 2022
Abstract: A two-year study was conducted in a conservation area of the Ventania hill system where the Pampean grassy steppes and their headwater springs maintain conditions close to natural ones. The composition and the structure of Chironomidae (Insecta: Diptera) along two short hill pristine streams and their relationship with the physicochemical variables were analyzed. A total of 15444 individuals belonging to 37 species of four subfamilies were identified. An assemblage structure more similar to those described for Andean or Argentine extra-Andean hill streams than to those of the surrounding Pampas was observed. Orthocladiinae showed the highest and Podonominae the lowest taxonomic richness and density. Chironominae was best represented downstream by genera with wide distribution. From the five Podonominae genera reported in the Andean-Patagonic region, three of them (Podonomus Philippi, Parochlus Enderlein and PodonomopsisBrundin) were collected. The presence of Podonomus and Podonomopsis represent the reports at lower altitudes for both genera. The physicochemical variables differed between seasons, and only water conductivity showed an altitudinal gradient. Those species prevailing in piedmont sites correlated positively with conductivity. With few exceptions, the species collected were recorded in both seasons. In Toro stream, density was significantly highest in Spring of the second year. This study provides the ecological and systematic baseline knowledge of an insect group widely used in biomonitoring of aquatic environments, to provide biological information that supports the importance of preserving this area.
Keywords: Limnology, Neotropics, Podonominae, Ventania hill system.
Resumen: Se llevó a cabo un estudio bianual en un área protegida del Sistema serrano de Ventania donde la estepa pampeana y las cabeceras de los arroyos mantienen las condiciones cercanas a las naturales. Se analizó la composición y la estructura del ensamble de Chironomidae (Insecta: Diptera) en dos arroyos prístinos y su relación con las variables fisicoquímicas. Se identificaron 15444 especímenes, correspondientes a 37 especies distribuidas en 4 subfamilias. El ensamble presentó una estructura más similar a las reportadas para los arroyos de la región Andina o extra Andina, que la de otras áreas de la región pampeana. Orthocladiinae presentó la mayor riqueza taxonómica y densidad, mientras que Podonominae las menores. Chironominae estuvo mejor representada aguas abajo, presentando géneros con distribución amplia. De los 5 géneros de Podonominae reportados en la región Andina-Patagónica, 3 de ellos (Podonomus Philippi, ParochlusEnderlein y Podonomopsis Brundin) fueron colectados. La presencia de Podonomus y Podonomopsis representa el registro de menor altitud para ambos géneros. Las variables fisicoquímicas difirieron entre las estaciones, y la conductividad fue la única que mostró un gradiente altitudinal. Las especies que prevalecieron en los sitios pedemontanos, correlacionaron positivamente con la conductividad. Con pocas excepciones, las especies recolectadas se registraron en otoño y primavera. En el arroyo Toro, la densidad fue significativamente mayor en la primavera del segundo año. Nuestro estudio aporta información de base sobre la ecología y la sistemática de un grupo de insectos ampliamente utilizado en estudios de biomonitoreo de ambientes acuáticos, lo cual resulta muy importante para respaldar la importancia de preservar esta área.
Palabras clave: Limnología, Neotrópico, Podonominae, Sistema serrano de Ventania.
INTRODUCTION
The cosmopolitan Chironomidae (Insecta: Diptera) inhabits an enormous diversity of aquatic ecosystems, frequently dominating the insect communities in both abundance and species richness in these environments (Ferrington, 2008). The composition of the chironomid assemblage is strongly related with the susceptibility or affinity of the species to the prevailing environmental conditions. As a result of the adaptations of the major subfamilies to the prevailing environmental conditions, the proportion of each subfamily varies from high latitudes to low latitudes, as well from high mountains to lowlands (Ashe et al., 1987).
The grassland in the Eastern plains of Argentina between 30° to 39°S comprises the Pampean biogeographic province (Cabrera & Willink, 1973), a vast and continuous plain where flat landscapes alternate with slightly undulating reliefs over long distances. The highest elevations are represented by two hill systems named Tandilia and Ventania older than the origin of the Pampean biome, located in the center and the south of the province of Buenos Aires respectively. The temperate climate shows mesothermic characteristics with average annual temperature that ranges between 14 °C in the south and 18 °C in the north, having fewer extreme conditions than other regions at similar latitudes by the moderating influence of the Atlantic Ocean. Towards the west, the moderating effect of the ocean decreases and the degree of continentality increases, which produces greater ranges of thermal amplitude (Soriano et al., 1992). A northeast to southwest precipitation gradient is present, which ranges from 1300 mm to 500 mm per year respectively.
The Ventania hill system extends 170 km in Buenos Aires Province, with a highest altitude of 1243 m a.s.l., and represents one of the few areas where the Pampean grassy steppes with some spring headwaters streams maintain conditions close to natural ones. The rainy season in this area occurs during spring-summer, winters are severe with occasional snow and a strong annual temperature variation occurs (Kristensen & Frangi, 1995). The headwater springs are placed in the high areas of the hills, and streams with bedrock bottom and clear oligotrophic waters run for less than 4 km until reaching the piedmont area. The 'Parque Provincial Ernesto Tornquist' (PPET) is a conservation area located in the Ventania hill system covering 6707 ha, which was created in 1937 to preserve this unique upland ecosystem.
The insect knowledge from the Ventania hill system is based on the species lists of aquatic Heteroptera (Konopko et al., 2009), Coleoptera (Fernández et al., 2010) and the dipteran Ceratopogonidae (Marino et al., 2011), on the description of new species and new records (Siri & Donato 2012; Donato et al., 2012; Mauad et al., 2013; Siri et al., 2014, 2015) and functional feeding groups of Chironomidae (Ocon et al., In press) and, on an ecological study of those phytotelm insects breeding in the impounded water between the axils of Eryngiumplants (Campos, 2015). Besides insects, studies on arthropods were conducted on harvestmen (Arachnida: Opiliones; Ringuelet, 1961a; Guerrero, 2019), spiders (Arachnida: Araneae; Ferreti et al., 2012; Pompozzi et al., 2014) and scorpions (Arachnida: Scorpionida; Schwerdt, 2016).
Due to the conservation areas are established to protect the valuable biodiversity distributed along patches of unique natural landscapes their management is fundamental (Naughton-Treves et al., 2005). For that, it is essential to know the biodiversity present in the area. In order to increase the knowledge of the chironomids of the Ventania hill system, the objectives of the present study were: 1- to describe and analyze the composition and structure of the Chironomidae immature assemblages along two hill pristine streams in the high-water periods (Autumn and Spring); 2- to analyze the relationship between the physicochemical variables and the structure of the Chironomidae assemblages.
MATERIAL AND METHODS
Study area. The present study was conducted in the 'Parque Provincial Ernesto Tornquist' (PPET) located in the Ventania hill system. The two main fluvial streams (Fig. 1), with their spring headwaters located at the PPET were studied during the two annual high-water periods of 2011 and 2012: Autumn (A) and Spring (S).
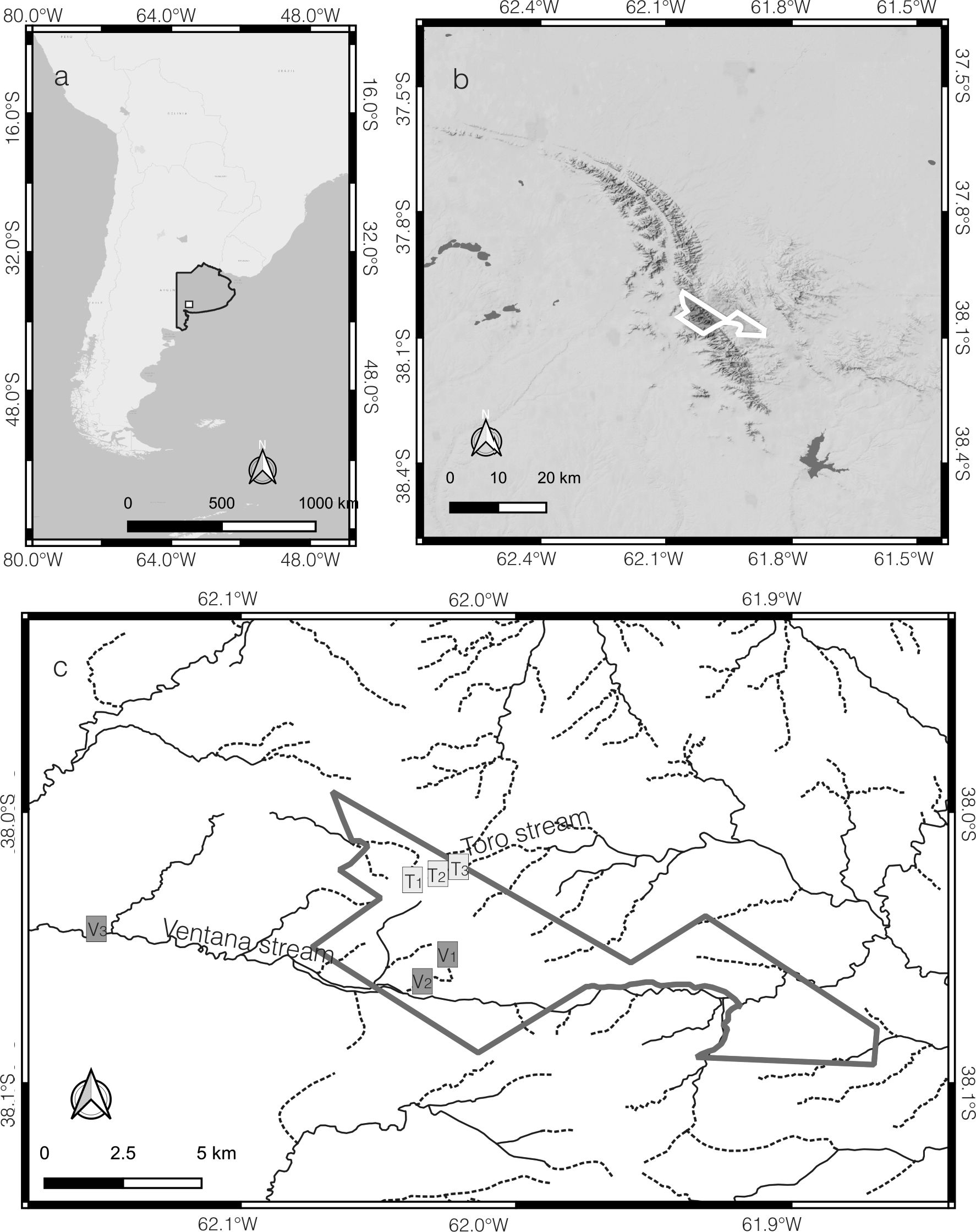
Fig. 1.
Study area. a. Map of South America. Buenos Aires province in gray, Ventania hill system in white square. b. Detail of Ventania hill system. Ernesto Tornquist provincial park (white line). c. Ernesto Tornquist provincial park (gray line) and sample sites.
The Ventana stream (V) is placed in the SW slope and belongs to the Sauce Chico basin. During most of the year, the stream section located in the hill area before the piedmont, is represented by shallow pools with permanent hyporheic connection and only superficially after rainfall. Three sampling sites were set up along this stream: site 1 (V.) at 38°03´31.7´´S / 62°01´11.8´´W, 782 m a.s.l; site 2 (V.) at 38°03´42.4´´S / 62°01´21.4´´W, 518 m a.s.l.; site 3 (V.) at 38°02´53.1´´S / 62°07´41.5´´W, 342 m a.s.l.
Toro stream (T) is placed in the NE slope and belongs to the Sauce Grande basin. This stream usually has superficial water continuity from the hill area to the piedmont. Three sampling sites were selected along the stream: site 1 (T.) at 38°01´26.1´´S / 62°01´45.8´´W, 800 m a.s.l.; site 2 (T.) at 38°01´19.0´´S / 62°01´33.3´´W, 672 m a.s.l.; site 3 (T.) at 38°01´09.6´´S / 62°00´56.27´´W, 561 m a.s.l. Along the text, the sites are denoted by the initial of the stream Ventana (V) or Toro (T), followed by sampling sites (1 to 3), the season Autumn (A) or Spring (S) and year 2011 (11) or 2012 (12).
Physicochemical water variables. For each site, the following water variables were measured in situ with a multiparameter probe: temperature (WT; °C), electrical conductivity (WC; µS.cm−1), pH and dissolved oxygen(DO; mg.l−1). In addition, water sub-surface samples were collected, using 500 ml plastic bottles, and kept refrigerated and transported to the laboratory to analyze nitrate (N-NO.), nitrite (N-NO.), ammonia (N-NH.) and phosphates (P-PO.) concentrations (mg.l−1), biochemical oxygen demand (BOD.) and chemical oxygen demand (COD) using standard methods (APHA, 1998) to be correlated with biological variables. Shapiro-Wilks test applied in ''Agricolae'' package (de Mendiburu, 2019) in R-language showed that physicochemical variables did not meet the variance assumption of normality. The Levene test applied in the "car" package R-language showed data without variance homogeneity. Therefore, Kruskal-Wallis non-parametric tests (H-test) were applied to detect significant differences on the physicochemical variables along the sample sites of each stream, between the streams and between seasons. A posteriori Dunn Bonferroni’s test using the ''FSA'' package (Ogle et al., 2022) was applied in R-language.
Sampling and identification of chironomids. Quantitative samples were obtained with a 250-µm-pore size mesh D-net in triplicate from an area of 1m. and fixed with 70% ethanol. In the laboratory, immature chironomids were separated, identified up to morphospecies under stereoscopic microscope and counted. To help in the identification of the collected larvae at specific level, we performed qualitative samples using manual or drift net for immatures and pupal exuviae, respectively. The larvae and pupa collected were individually reared in the laboratory up to the adult stage following Epler (2001).
Identifications were corroborated by permanent microscope slides of some specimens of each morphospecies. Larvae were mounted in Canada Balsam following Epler (2001). Identification of immature was performed using the following keys and papers: Brundin (1966), Epler (2001), Spies et al. (2009), Siri & Donato (2012, 2015), Andersen et al. (2013) and Silva et al. (2018a). Studied specimens were deposited at the ILPLA (Instituto de Limnología “Dr. Raúl A. Ringuelet” collection).
Composition, structure, and multivariate analyses of Chironomidae assemblages. The structural attributes of the chironomid assemblage such as Taxa Richness (S), Density (ind.m−1), Diversity index of Shannon-Weaver (H´), Simpson index (D), and evenness of Pielou (J´) were obtained (Moreno, 2001). Kruskal-Wallis non-parametric tests (H-test) was applied to detect significant differences of these structural attributes along the streams, as well as between the two analyzed seasons. A posteriori Dunn Bonferroni´ test using the ''FSA'' package (Ogle et al., 2022) in R-language was applied.
Non-metric multi-dimensional scaling analysis (nMDS) using Bray-Curtis similarity index were performed to analyze sampling sites and species distribution in the multi-dimensional space and toidentify the physicochemical variables that fit the ordination space of the chironomid sites. In the nMDS analyses, the density transformed to log10(x+1) of each species of the three replicates was included for each sample.
Prior to the nMDS analysis, physicochemical variables were log10(x+1)-transformed except for pH. Spearman correlation analysis among physicochemical data was applied to determine collinearity among variables. The physicochemical PO., NO. and BOD. showed multicollinearity and were not included in the nMDS analysis. The function “envfit” was applied to fit the scaling of the assemblage dissimilarities with environmental variables, where each vector has the maximum correlation with the corresponding environmental variables. The environmental factor association (R.) was calculated, and the significance level tested by permutation with 999 simulations.
One-way Analysis of Similarity (ANOSIM) was applied to test differences between sample sites and between the groups obtained in the nMDS analyses using Bray-Curtis similarity index and 999 permutations to test significance. To estimate those morphospecies mostly contributing to the dissimilarities among sample sites or between those groups obtained in nMDS analyses, a similarity percentages-species contributions analysis (SIMPER) was conducted.
Spearman correlation between the physicochemical variables with the chironomid density and, with the taxonomic richness was conducted to test their significant (p<0.05) influence.
The nMDS analyses were performed using the ‘‘vegan’’ package (Oksanen et al., 2013); correlations with “correlation” packages (Makowski et al., 2021) and some of the default functions in the R environment (R Core Team, 2020). ANOSIM and SIMPER analyses were performed using Paleontological Statistics (PAST) software package Version 4.03 (Hammer et al., 2001).
RESULTS
Environmental characterization. Stream substrates consisted in rocky bottom with clear water and poor riparian forest vegetation or macrophytes communities. The water showed oligotrophic characteristics with high DO, pH neutral to slightly alkaline, low WC and relatively low concentrations of dissolved phosphorus and nitrates (Table I).
The WC varied from 20 µS.cm−1 (V1S12) to 124.89 µS.cm−1 (V3S12); V3 always had the highest values. The WT varied from 8 °C (V1A12) to 18 °C (V1S11, T1S11, T2S11, T3S11, V3S12). The DO displayed high values from 8.95 mg.l−1 (T3S11) to 16.58 mg.l−1 (V1A12). The pH varied from 6.35 (T2S12) to 8.02 (T2S11). BOD5 varied from 1 to 11; sites V2 and V3 (except V2S12) showing lower values than V1. COD varied from 4 (V3S11) to 31 (T2A12). PO4 ranged from 0.008 mg.l−1 (T1S11) to 0.109 mg.l−1 (T2A12); NO3 from 0.007 mg.l−1 (V3S11) to 0.321 mg.l−1 (T2A12); NH4 from 0.001 mg.l−1 (V1S12, T1S12) to 0.127 mg.l−1 (T2A12). NO2 varied between 0.001 mg.l−1 and 0.003 mg.l−1.
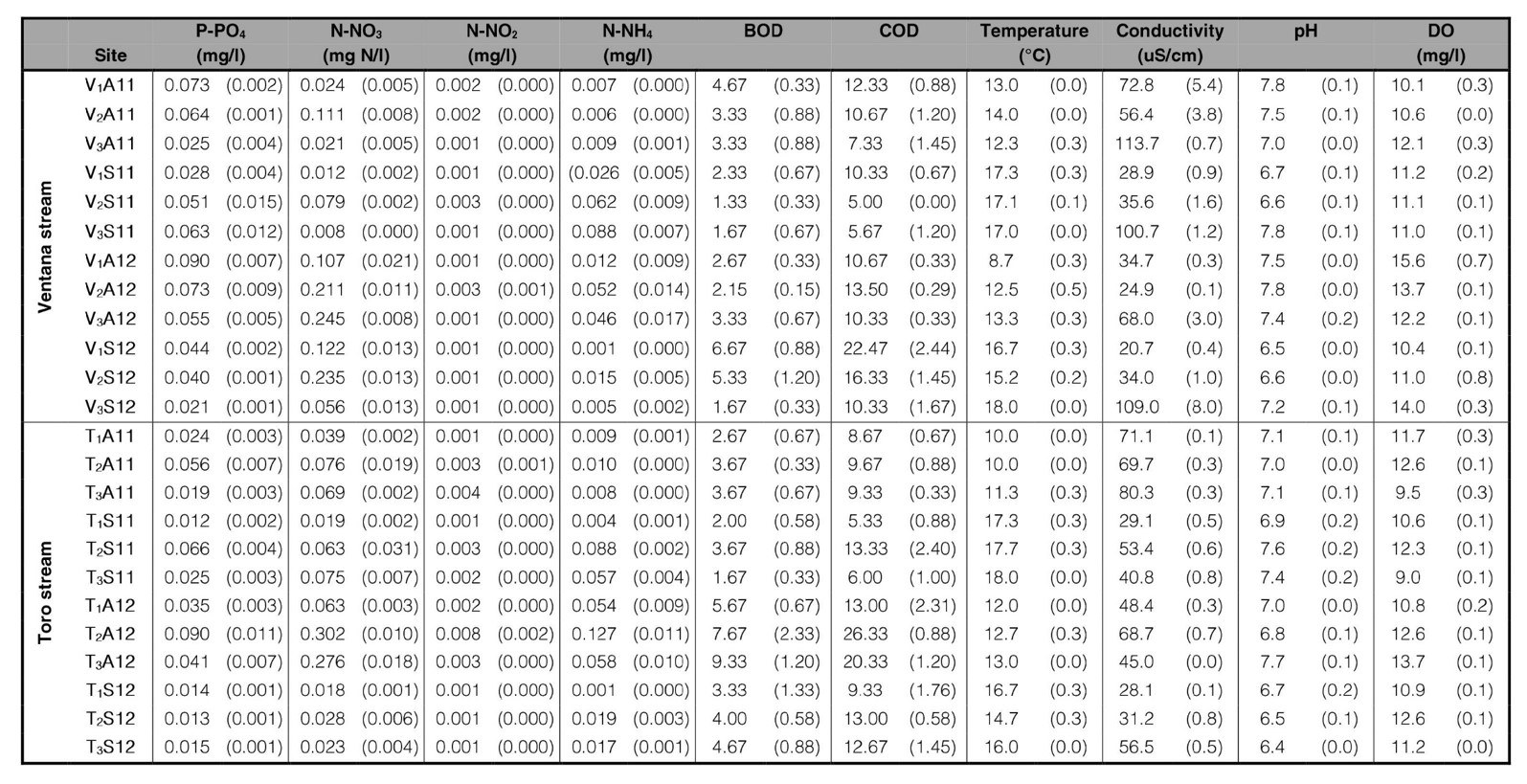
Table I.
Mean environmental variables of Ventana and Toro streams followed by standard error in parenthesis.
Sites are denoted by the initial of the stream Ventana (V) or Toro (T); sampling sites (1 to 3), season (Autumn: A, Spring: S) and year (2011: 11, 2012: 12).
According to the Kruskal-Wallis non-parametric test, the physicochemical variables did not show significant differences in an altitudinal gradient except when otherwise indicated following (Table II). In the Ventana stream, the WC and COD showed significant differences between hill sites (sites 1 and 2) with piedmont site (site 3). The variables NO3 and NO2 in the Ventana stream showed significant differences between sites V1 and V2, as well between sites V1 and V3, while no differences were observed between sites V2 and V3. In the Toro stream these variables showed significant differences between T1 and T2, as well between T1 and T3, but not between T2 and T3.Besides, the physicochemical variables differ significantly between the two analyzed seasons, with highest values in Autumn except for WT whose values were higher in spring (Table II).
Composition, structure, and multivariate analyses of Chironomidae assemblages. A total of 15.444 immature individuals belonging to 37 species were identified from four subfamilies (Table III). The density of specimens varied between 4 (V1A11) to 1165 (V3S11) ind.m−2. The minimal taxonomic richness (S) was 2 at V1A11, while the maximal was 21 at T3S12. According to Kruskal Wallis analysis, there were no significant differences in the structural attributes of the assemblage between the two years (Table IV). There were significant differences in the taxonomic richness between the sites along the streams, with lowest values occurring in the upper sites (V1, T1). The density differed significatively between Autumn and Spring, with highest values occurring in Spring.
The taxonomic richness between subfamilies showed Orthocladiinae represented by 16 species (41.7%), Tanypodinae and Chironominae each by 9 species (25%) and Podonominae by 3 species (8.3%). Orthocladiinae also showed the highest abundance of immatures (68.5%), followed by Chironominae (14.5%), Tanypodinae (8.85%) and Podonominae (8.42%).
Ventana stream. In most sample dates, S was lowest at V1 and highest at V2. The highest S value was found in V2A11 (18); the highest density (1165 ind.m−2) at V3S11; the highest H´ (2.39) and D (0.87) at V3S12. In contrast, the sample site V1A11 showed the lowest density (4 ind.m−2), and S (2). All sites showed values of J´ higher than 0.5. Kruskal Wallis analysis showed that there were no differences in the structural attributes of the assemblage between the two years nor between seasons (Table IV). Along the Ventana stream, site V2 showed highest significative values of S and H´. Density of site V1 was significatively lower than V3. Site V1 showed a highest taxonomic richness proportion of Orthocladiinae, followed by Podonominae or Tanypodinae and lowest of Chironominae. Sites V2 and V3 showed a decreasing ratio of Orthocladiinae, Tanypodinae or Chironominae and Podonominae, while the subfamily Podonominae was absent in V3. In all sites, Orthocladiinae was the subfamily with the highest number of species.
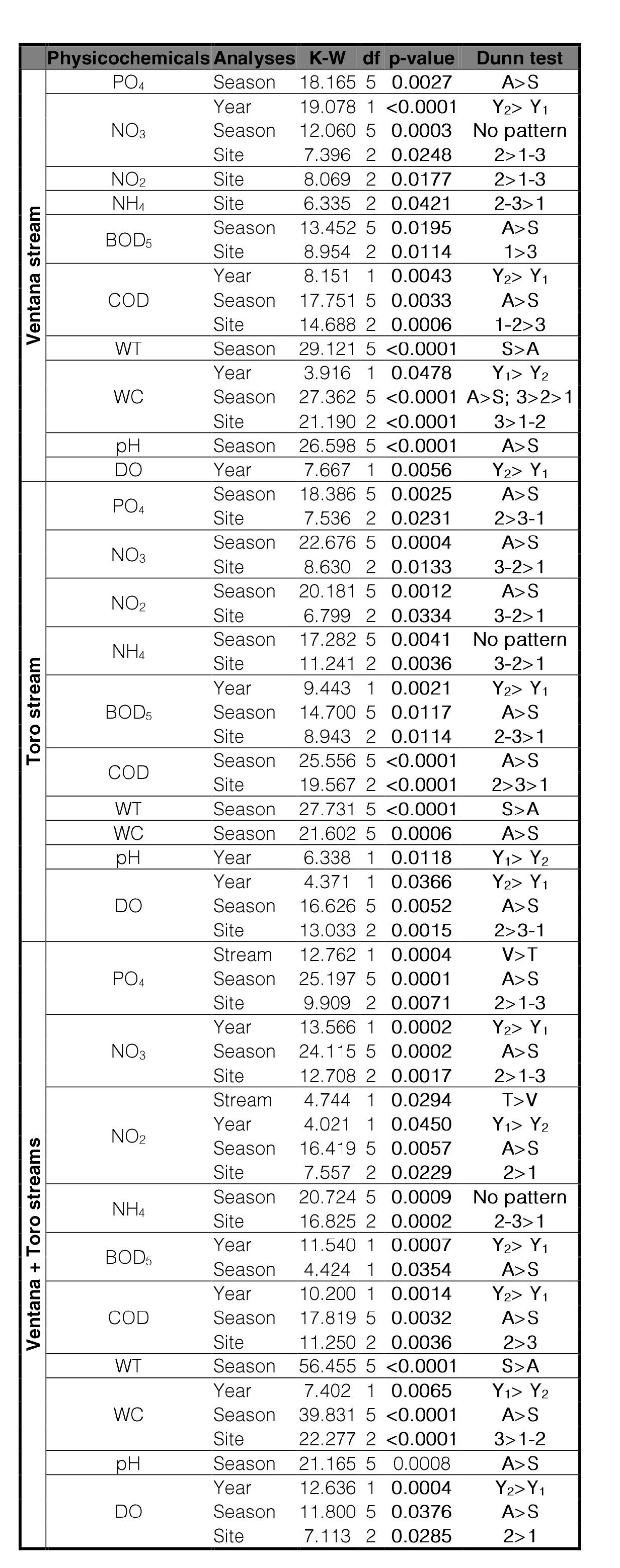
Table II.
Kruskal-Wallis non-parametric test on the physicochemicalvariables (PC) along the sampling sites and, between seasons.
Only significant values (p ≤ 0.05) are presented. Dunn posteriori test is included. Analyses were conducted by stream (V = Ventana; T = Toro); year (Y1 = 2011; Y2 = 2012), season (A = Autumn; S = Spring) and sites (1 = Upstream; 2 = Middle; 3 = Downstream).
Toro stream. The highest S value was found in at T3S12 (21); the highest density (414) at T2S12; the highest H´ (2.29) and D (0.88) occurred in T3S11. In all sample dates H´ and D were lowest at site T1, with lowest values (at T1S11) of 0.91 and 0.48 respectively. All sites showed high values of equitability (J´), varying from 0.61 at T1S11 and T1S12 to 0.88 at T2A11. Kruskal Wallis analysis showed that there were no differences in the structural attributes of the assemblage between the two years (Table IV). Along the Toro stream, site T1 had significant lowest values of H´, D and J´. The lowest density values were observed in Autumn, although differing significantly only with the second-year spring samples.
Orthocladiinae, Podonominae, Tanypodinae and Chironominae were present in all samples, but Podonominae in T3A11. In all samples, Orthocladiinae showed the highest S value, while Chironominae usually showed the lowest values. In the hill section sites (T1 and T2) there was a decreasing ratio in the taxonomic richness of Orthocladiinae, Podonominae or Tanypodinae and Chironominae. In the piedmont site (T3), the proportion of the taxonomic richness was Orthocladiinae, Tanypodinae or Chironominae and Podonominae.
The nMDS analysis (stress = 0.1137) withboth streams is shown in Fig. 2a, in which the sites are grouped following an altitude gradient.Each of the piedmont sites (T3; V3) was individually grouped, but sites of the hill section (T1; T2; V1; V2) were not separated independently and convex hulls overlapped. Upper sites (T1; V1) were located mainly at quadrants Q1 and Q4, middle sites (T2; V2) mainly at Q4 close to the origin center of coordinates and, piedmont sites (T3; V3) at quadrants Q2 and Q3. The site V1 showed the most widespread spatial dispersion, perhaps due to its high hydrological instability. In descending order, the WC, WT, COD and pH were the water variables that significantly (p <0.05) fitted the ordination space of chironomid samples.
The nMDS analysis with species distribution (Fig. 2b) showed that Dicrotendipesembalsensis Paggi, Parachironomus Lenz sp., Paratanytarsus Thienemannet Bause sp., Cryptochironomus Kieffer sp., Apedilum Townes sp., Rheotanytarsus Thienemannet Bause sp. (Chironominae), Corynoneura Winnertz sp.2, Cricotopusv.d. Wulp sp.3, Cricotopus sp.4, Metriocnemini sp., Nanocladius Kieffer sp., Onconeuraanaliae Donato et al., Thienemanniellaliae Paggi, Parametriocnemus Goetghebuer sp. (Orthocladiinae), Metapelopia Silva et al. sp., Labrundinia Fittkau spp., Procladiini sp. and Tanypus Meigensp. (Tanypodinae) prevailed at piedmont sites. Most of these species have been found only in the piedmont. Corynoneura sp.1, Cricotopus sp.1, Limnophyes Eaton sp. (Orthocladiinae), representatives of Podonominae and,Larsia Fittkau sp., Pentaneura Philippi sp., and Paggipelopia spaccesi Siriet Donato (Tanypodinae) were collected in all sites but prevailed on the hill section.
Results from one-way ANOSIM (R= 0.4446; p<0.0001) and SIMPER analyses are presented in Table V. Supporting nMDS analysis, the one-way ANOSIM analysis showed that there is no significant differentiation between the sites 1 and 2 of the hill section (R=0.32; p=0.001), whilst each sample site of the hill section showed significantdifferences with the piedmont one. SIMPER analysis showed that higher densities of Onconeura analiae and Cricotopus sp.3 in piedmont sites and, highest densities of Podonomus tehuelche and Limnophyes sp., and the absence of Dicrotendipes embalsensis in the hill sites, mostly contributes to dissimilarities between the altitudinal groups.ANOSIM analysis showed significant differences between the autumn and spring samples (R=0.1029; p=0.0004), although it is not evident in the nMDS (Fig. 2a). SIMPER analysis showed that Limnophyes sp., Corynoneura sp.1 and Cricotopus sp.2 were the species mostly contributing to dissimilarities between the seasons, with highest density values occurring in Spring.
Spearman correlation analyses of the Chironomidae density (Table VI) showed that those species prevailing in piedmont sites correlated positively with WC, whilst Limnophyes sp. and P. spaccesi prevailing at the hill section correlated negatively. The total number of species, as well as the number of species of each subfamily were not correlated with the physical-chemical variables analyzed.
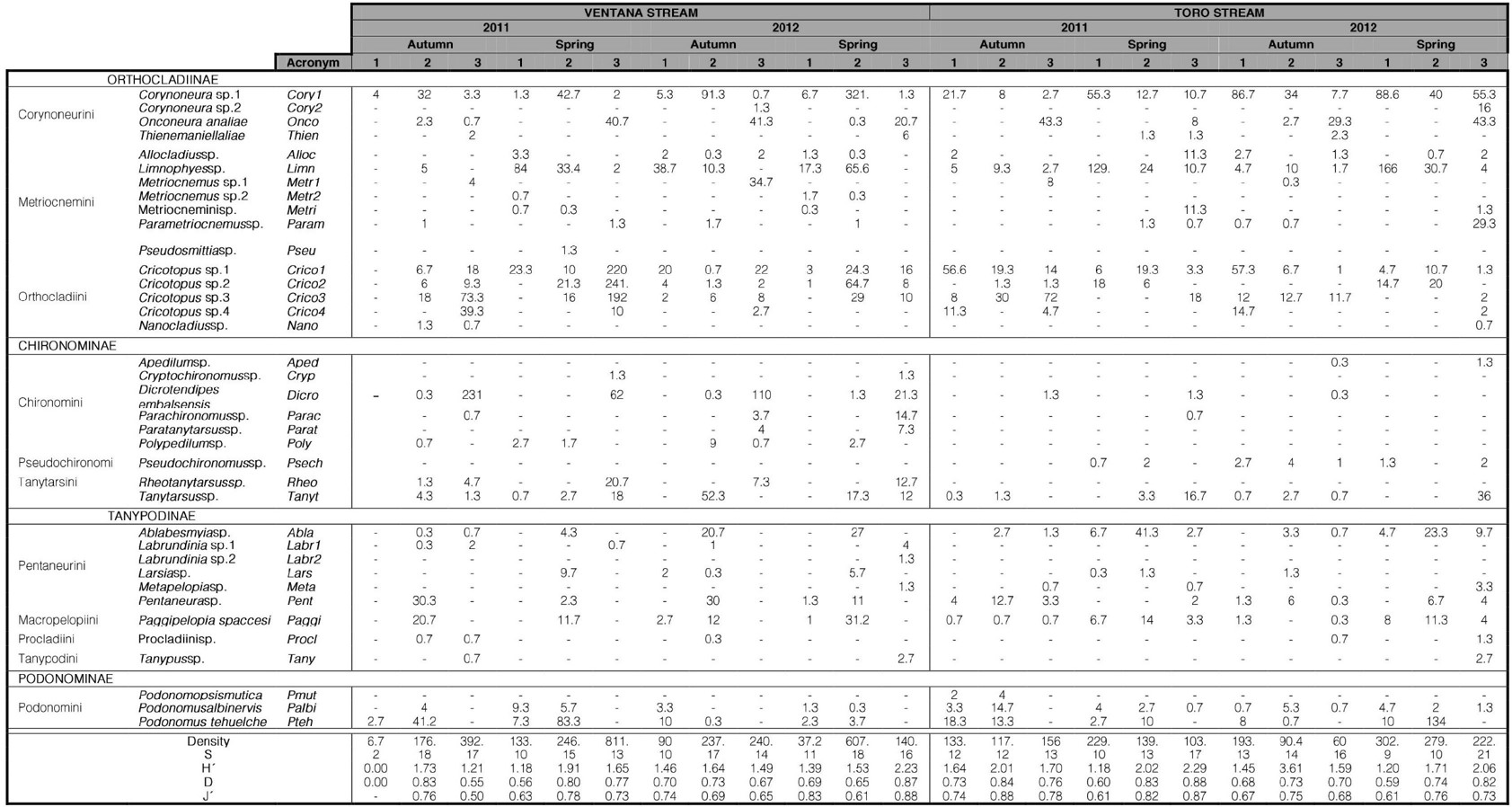
Table III.
Composition and density of Chironomidae in two streams of Ventania hill system.
Total Density (ind.m-2), Shannon-Wiener (H´) and Pielou (J) diversity is expressed as the mean of the 3 sample sites. The taxonomic richness (S) is expressed as the total of the species of the 3 sample sites.
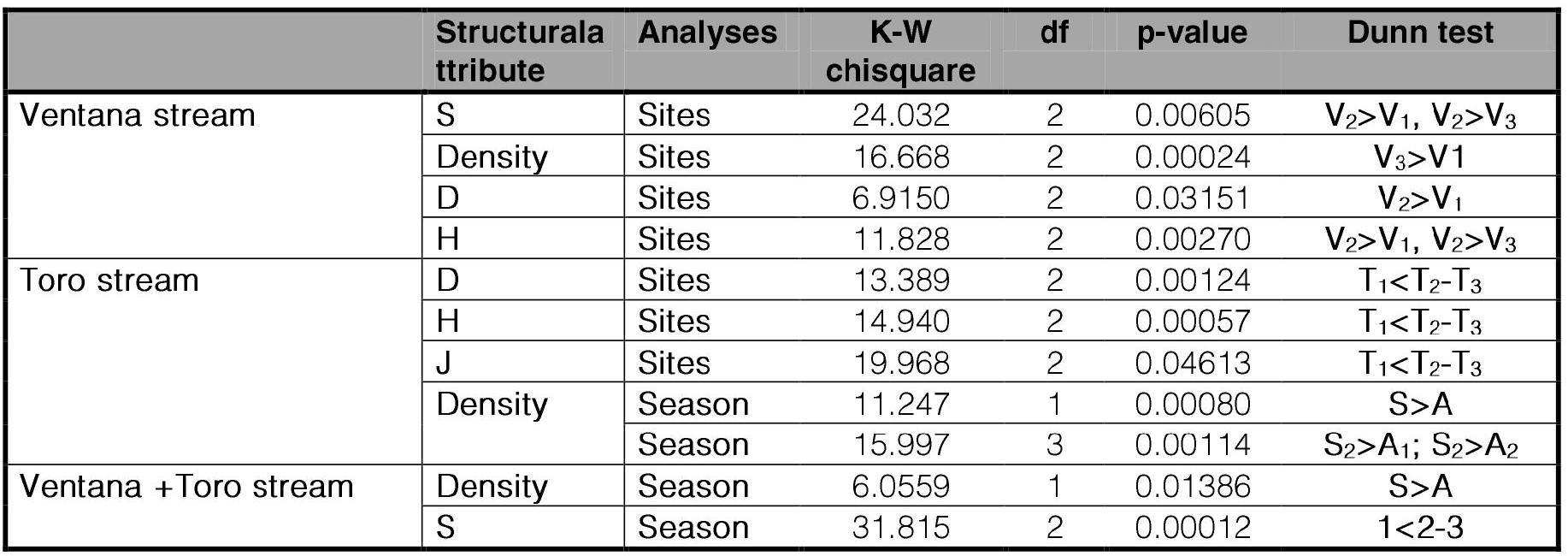
Table IV.
Kruskal-Wallis non-parametric test on the structural attributes of the assemblage along the sampling sites and between seasons.
Only significant values (p ≤ 0.05) are presented. Dunn posteriori test is included. Analyses were conducted by stream (V = Ventana; T = Toro); season (A = Autumn; A1 = Autumn 2011; A2 = Autumn 2012; S = Spring; S1 = Spring 2011; S2 = Spring 2012) and sites (1 = Upstream; 2 = Middle; 3 = Downstream).
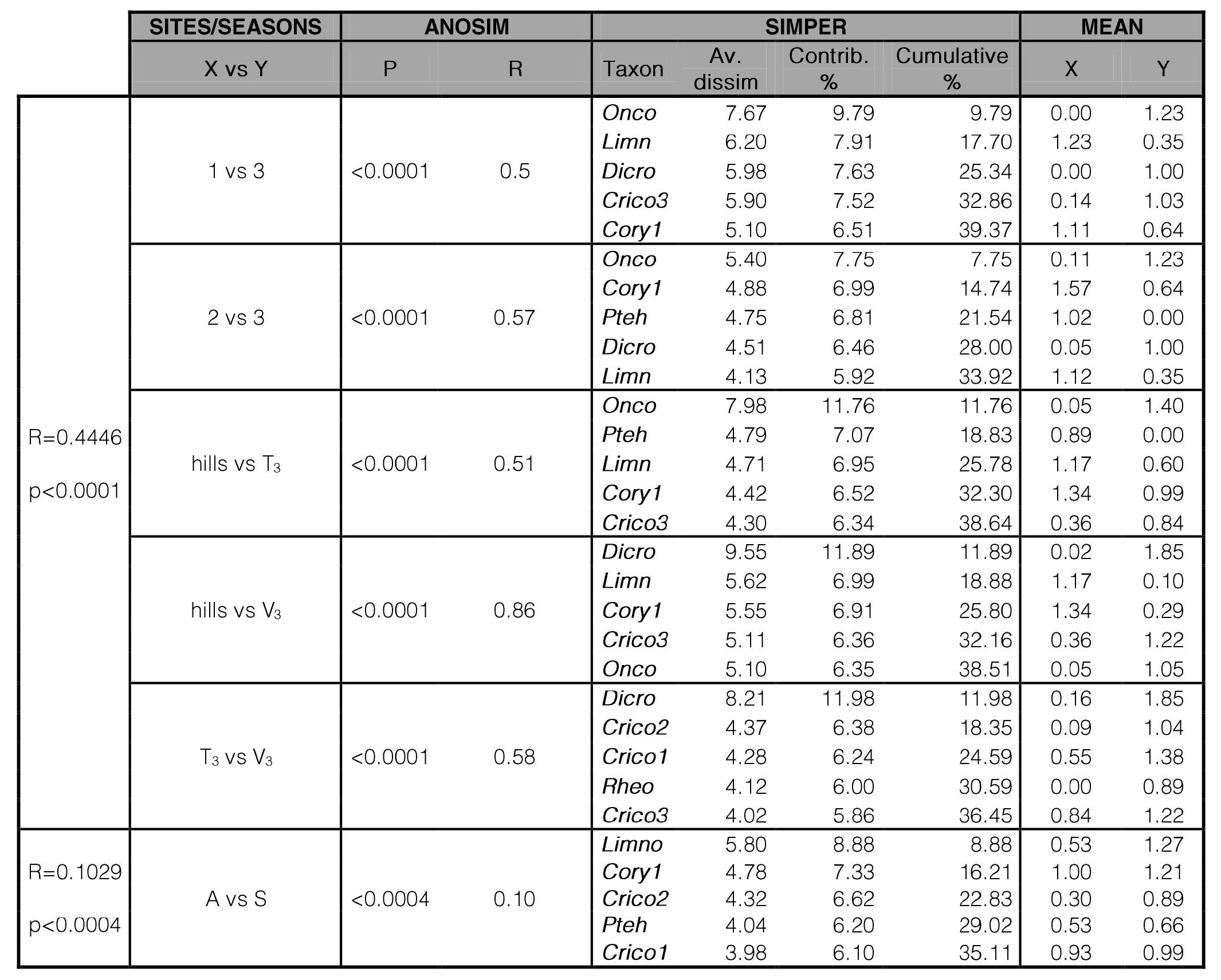
Table V.
One-way ANOSIM and SIMPER analyses of Ventana and Toro streams.
Sites (or groups of sites) with significant differences (p<0.001) are included. V = Ventana stream; T = Toro stream; 1 = Sites V1+T1; 2 = Sites V2+T2; 3 = Sites V3+T3; hills = Sites V1+T1+V2+T2; A = Autumn; S = Spring. For taxa acronyms, see Table III.
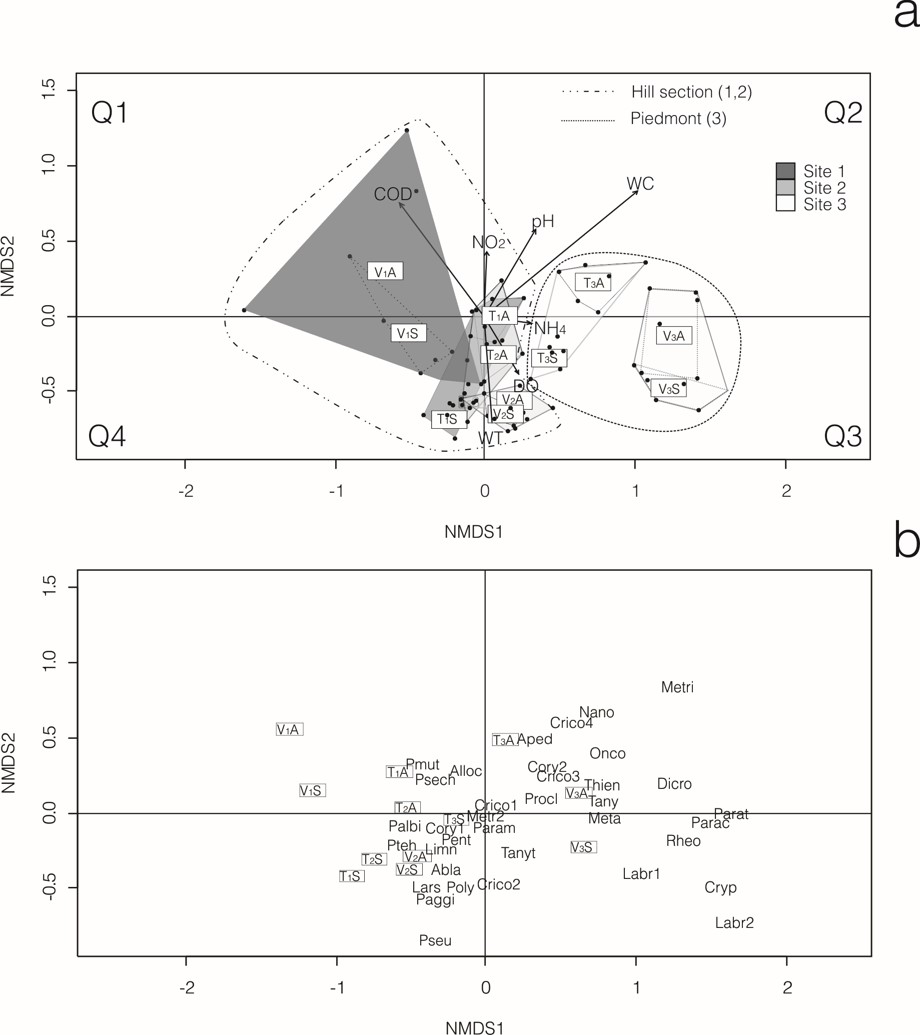
Fig. 2.
nMDS analysis including all sample sites. a. Triplot showing the results of non Metric Multi-Dimensional Scaling (nMDS). Convex hulls (polygons) for each sample site are included. b. Biplots of sampling sites and species. For taxa acronyms, see Table III.
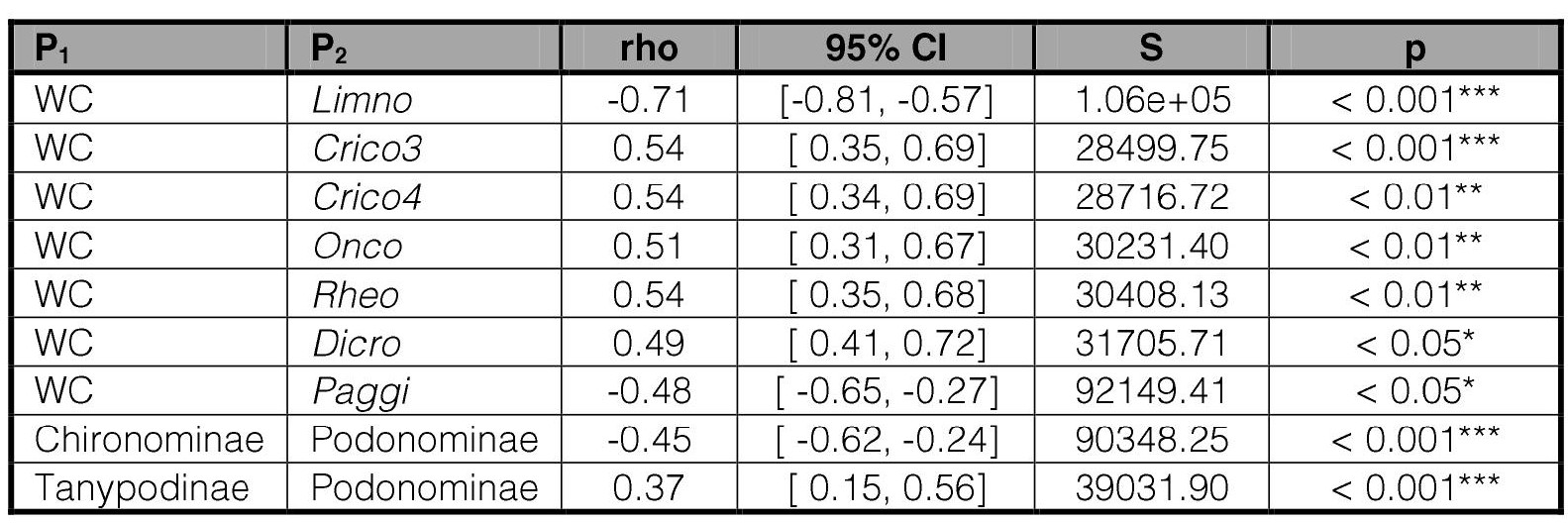
Table VI.
Spearman’s correlations.
Those significant correlation values are only presented. P1: parameter 1, P2: parameter 2. Acronyms in Tables I and III.
The individual nMDS analysis of Ventana stream (stress: 0.0754) resulted in a site distribution with three main groups, ordered according to their altitude (Fig. 3). The WC and COD were the physicochemical variables that significantly fitted chironomid samples ordination (Fig. 3). One-way ANOSIM analysis (Table VII) supported the 3 main groups observed in the nMDS analysis. Site V1 differed from V2 (R=0.75; p<0.0001) and from V3 (R=0.74; p<0.0001), while V2 also differed from V3 (R=0.93; p<0.0001). According to the SIMPER analysis (Table VII), the highest densities of P. spaccesi and Limnophyes sp. together with the absence of Dicrotendipes embalsensis in hill sites, and the highest densities of Cricotopus sp.3 together with the absence of Pentaneura sp. in V3, mostly contributed to dissimilarities between the three groups.In the nMDS analysis —also supported by the one-way ANOSIM—, sites were not grouped according to the two analyzed seasons.
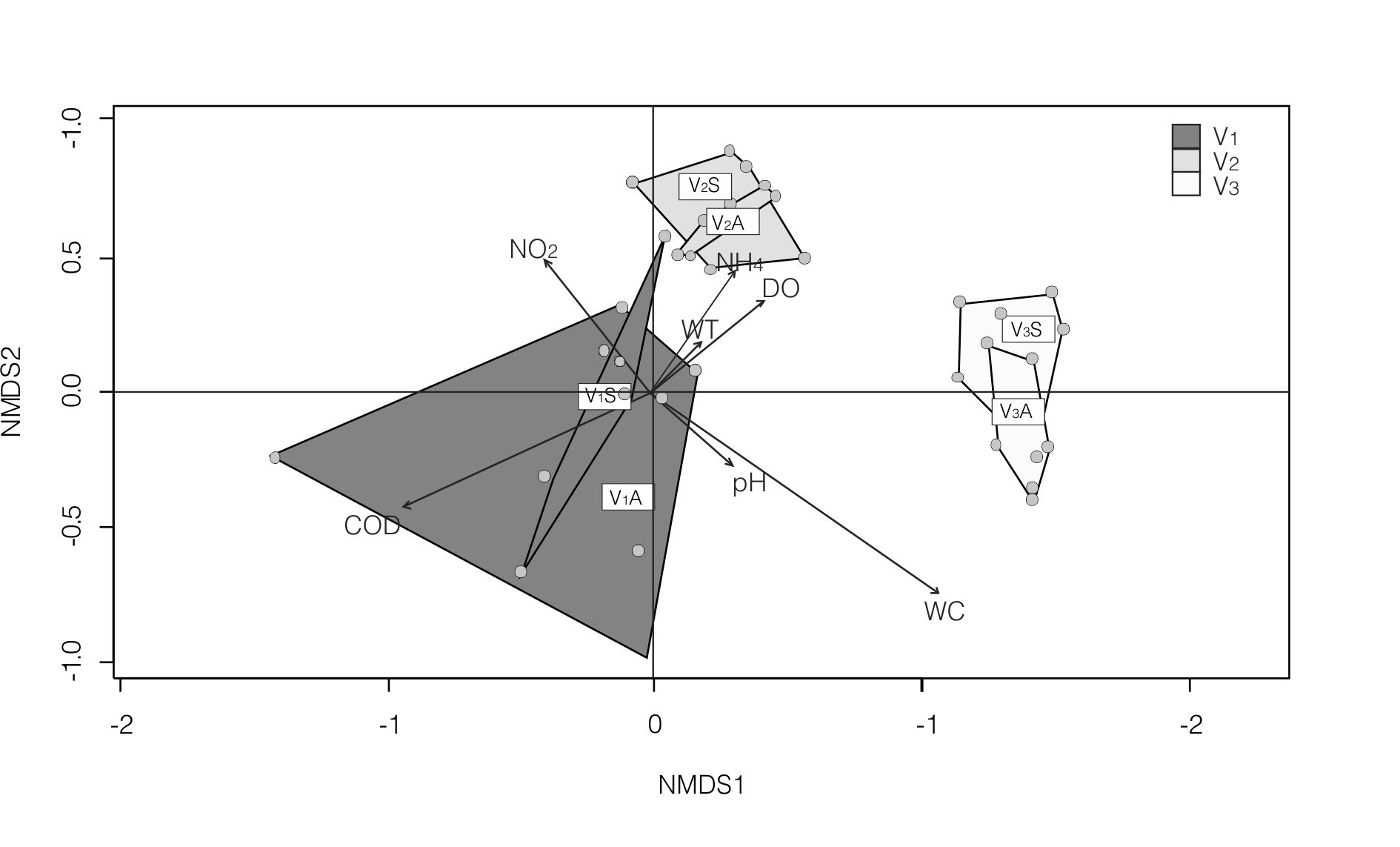
Fig. 3.
Biplot showing the results of non Metric Multi-Dimensional Scaling (nMDS) of Ventana stream. Convex hulls (polygons) for each sample site by season are included.
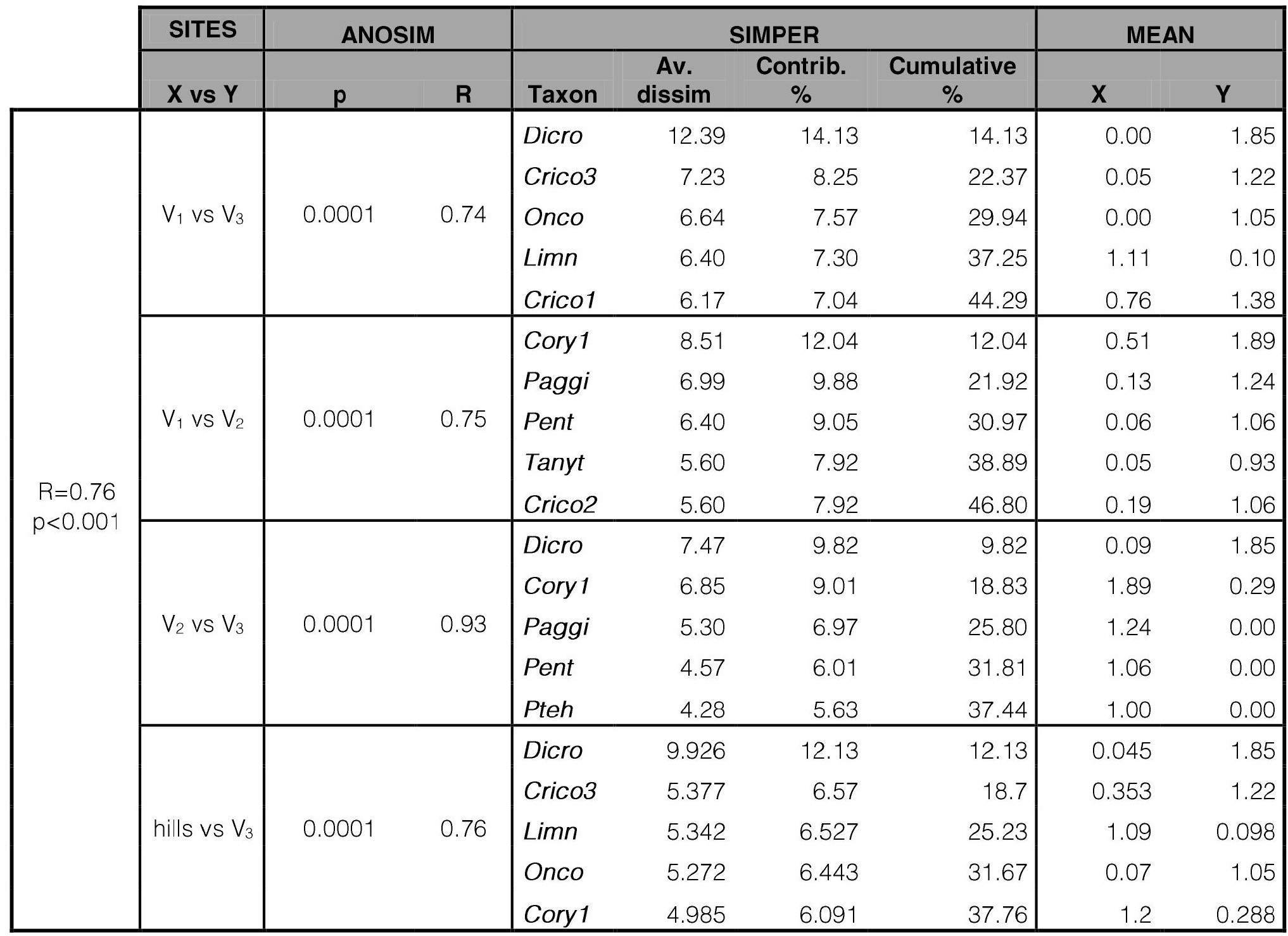
Table VII.
One-way ANOSIM and SIMPER analyses of Ventana stream.
Sites (or groups of sites) with significant differences are included. hills = Sites V1+V2. For taxa acronyms, see Table III.
In the nMDS analysis of Toro stream (stress = 0.1061) sites were ordered following an altitude gradient, although the convex hulls of the sites of the hill section overlapped (Fig. 4). Besides, sites were grouped according the two analyzed seasons, with autumn samples placed in quadrants Q1 and Q2, and spring samples placed in quadrants Q3 and Q4. The WT and WC significantly fitted the chironomid samples ordination. One-way ANOSIM analysis (Table VIII) did not show significant differences among sites of the hill section (T1; T2), while each of these sites differed from the piedmont site (R=0.66 and 0.60 respectively; p<0.001). SIMPER analysis (Table VIII) showed that the absence of O. analiae in the hill section and, the absence of P. tehuelcheand lower density of Limnophyes sp. in T3 mostly contributes to dissimilarities between the hill sites and piedmont in Toro stream. The groups formed by the samples ordered by seasons in the nMDS analysis of Toro stream were supported by the ANOSIM analysis. The SIMPER analysis showed that Limnophyes sp, Cricotopus sp.3, C. sp.2, Ablabesmyia sp., and P. tehuelche were the species mostly contributing to dissimilarities between the two analyzed seasons.
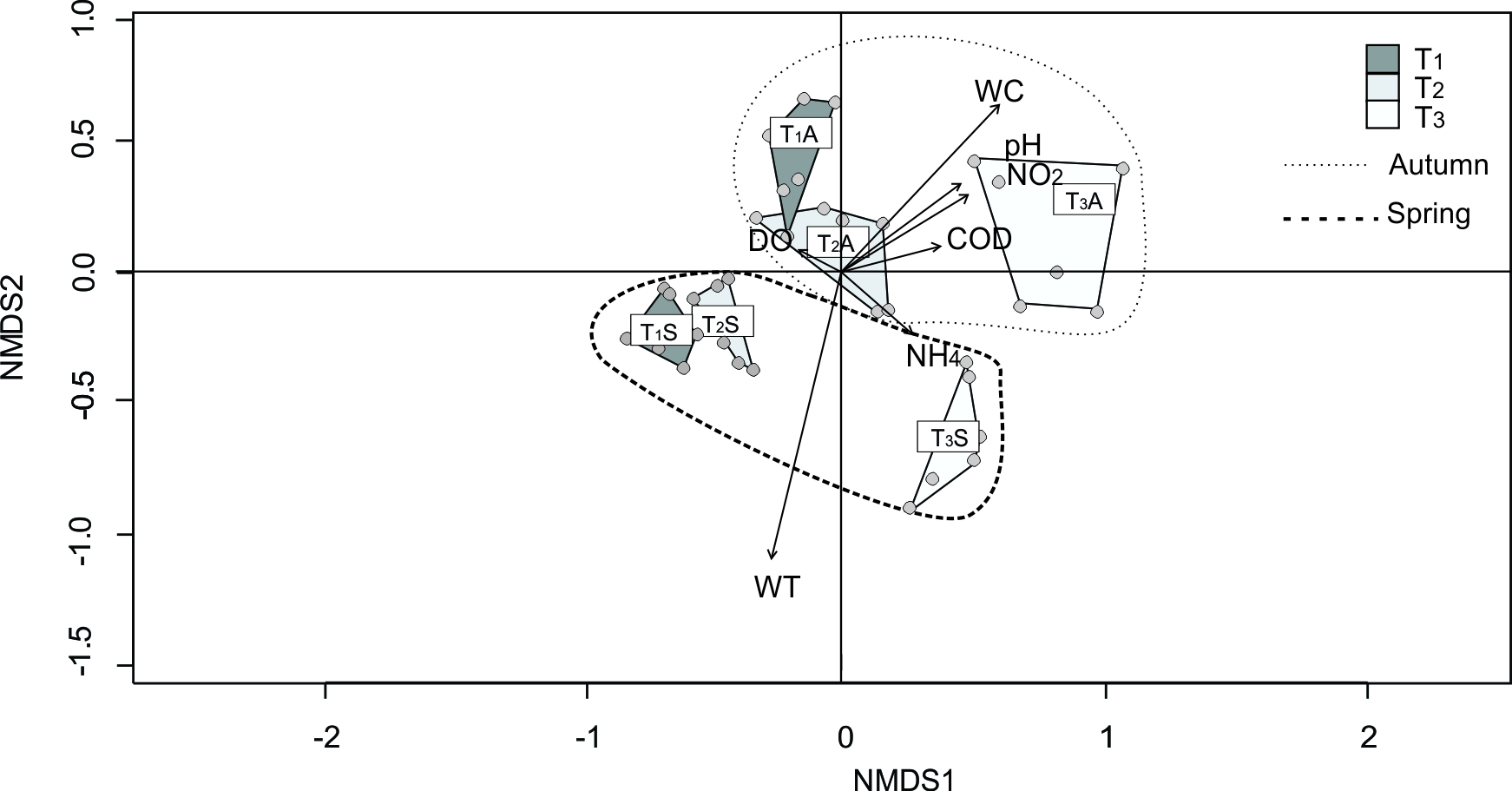
Fig. 4.
Biplot showing the results of non Metric Multi-Dimensional Scaling (nMDS) of Toro stream. Convex hulls (polygons) for each sample site by season are included.
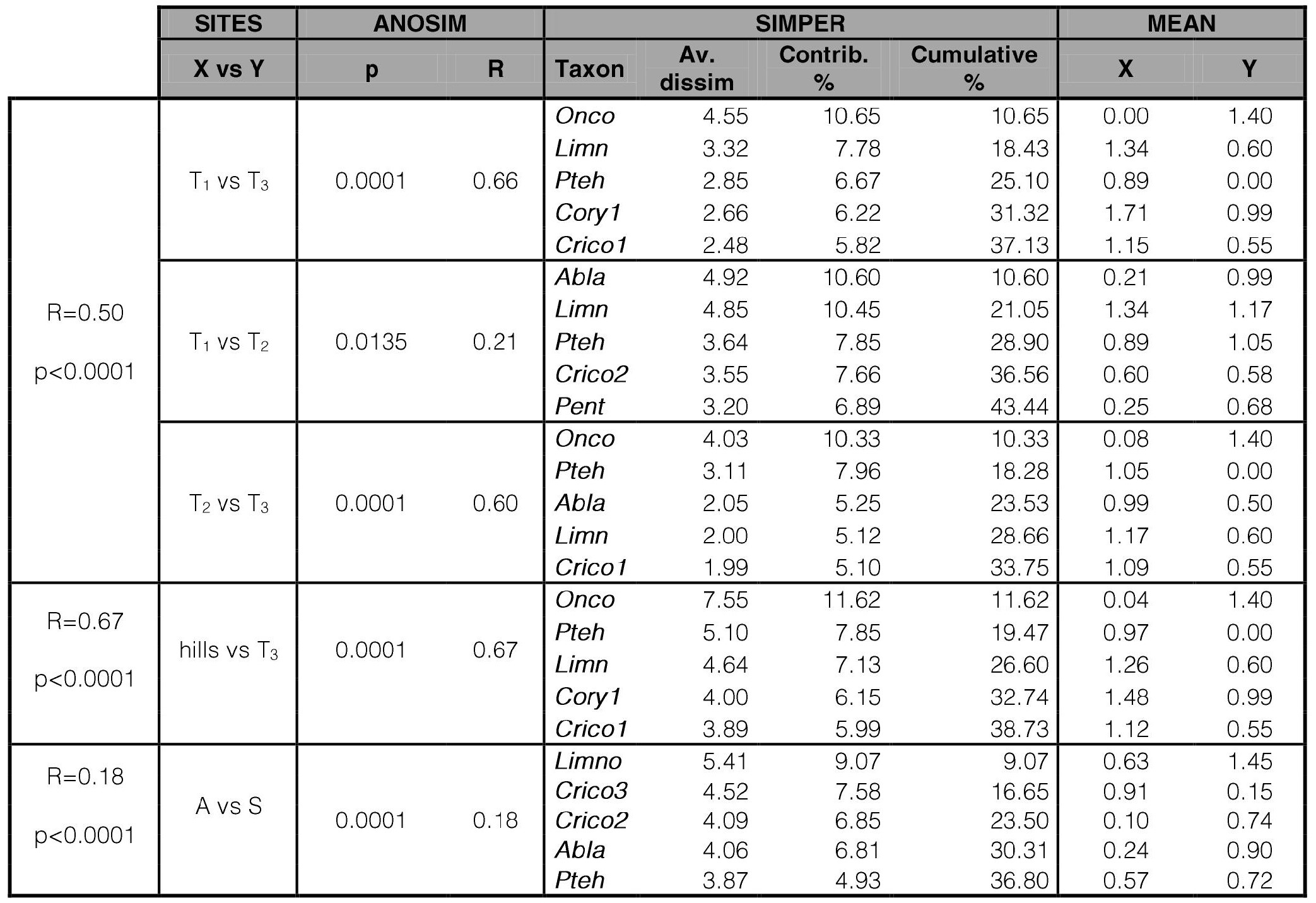
Table VIII.
One-way ANOSIM and SIMPER analyses of Toro stream.
Sites (or groups of sites) with significant differences are included. hills = Sites T1+ T2; A = Autumn; S = Spring. For taxa acronyms, see Table III.
DISCUSSION
The sections analyzed of both streams in the present study are located in the PPET, except V3, and are subjected to very low anthropogenic disturbances. These streams have their headwaters in the high areas of the hill and run through a relatively short section in the hill until they reach the piedmont section. The physical-chemical variables analyzed showed to be quite similar between the studied sites, except for conductivity. Although the WC showed significant differences between the upstream and downstream sites, the values did not present great differences between the hill sites (20.0-115.1 µS.cm−1) and the piedmont ones (40.0-124.9 µS.cm−1). Those physicochemical variables analyzed were significatively highest in autumn except for WT.
The present study considerably increases the knowledge of the diversity and ecology of Chironomidae from the Ventania hill system. Our results showed a high taxonomic diversity with differences in the chironomid assemblage composition and structure between the hill section of the stream and the piedmont site. The chironomid assemblage structure of the hill section showed Orthocladiinae with the highest taxonomic richness, Podonominae and Tanypodinae well represented and Chironominae scarcely represented. There was no exclusive species for the hill section, but some species such as Limnophyes sp., Corynoneura sp.1, Larsia sp., Pentaneura sp. and species of Podonominae were best represented in this section. In the piedmont, Chironominae and Tanypodinaewere well represented —although with a lower taxa richness than Orthocladiinae— and Podonominae was scarce or absent. In this stream section, high density of Chironomini (Chironominae), Corynoneurini (Orthocladiinae) and other cosmopolitan or widely distributed genera was observed.With few exceptions, the species collected in the present study were recorded in both seasons. In the analyzes that included both streams, density was significantly higher in Spring.
According to the subfamily proportion rule along an elevational gradient (Ashe et al., 1987) there is an increase in the diversity and abundance of Chironominae and Tanypodinae with a corresponding decrease in the Orthocladiinae and Podonominae, Diamesinae and Prodiamesinae from high mountains to lowlands. Despite the relatively low altitudinal range (less than 500 m) in the Ventania hill system, the chironomid assemblage structure follows this subfamily proportions rule along the elevational gradient. This chironomid assemblage structure in Ventania is similar to those described for Andean (Ashe et al., 1987) and to those reported for Argentine extra-Andean hill streams (Tejerina & Molineri 2007; Scheibler et al., 2014; Zanotto et al., 2015). The subfamily Diamesinaewas collected from some of those Andean and extra-Andean streams but not in Ventania, perhaps since higher cold requirements of this subfamily are not present in this hill system. On the other hand, this chironomid assemblage structure found in Ventania differs from the structure reported from other areas of the Pampean province (e.g. Cortelezzi et al., 2020; Zanotto et al., In press) where higher proportions of Chironominae and the absence of subfamily Podonominaeor Macropelopiini tribe (Tanypodinae subfamily) occurs.
Species reported in previous studies of Chironomidae from Ventania (Paggi, 2007; Donato et al., 2012; Siri & Donato, 2012, 2015; Mauad et al., 2013; Siri et al., 2014) as well as in our study, includes both Patagonian and Neotropical taxa. In the present study, we found that the assemblage structure is more similar to a structure associated with gradients of altitude than to that of the latitude in which this hill system is located. Besides of Chironomidae, the presence of both Patagonian and Neotropical insect species was previously reported for heteropterans (Konopko et al., 2009), coleopterans (Fernández et al., 2010), midges (Marino et al., 2011) as well as for other arthropods such as scorpions (Schwerdt et al., 2016). The presence of Patagonic taxa in Ventaniacould be explained in part by the current environmental condition in this hill system. It has also been attempted to explain by historical processes that determined the distributional patterns of these taxa. In this sense, Ventania hill system has been reported as one of the fragments currently persisting of the continuous corridor connecting the Andes and Brazil, defined by Frenguelli (1950) as the PeripampasicOrogenic Arc. The function as a biotic corridor, where Neotropical and Patagonian biota converge, was supported by several studies performed in different biotic groups (e.g.,Ringuelet, 1961b; Sota, 1967; Crisci et al., 2001; Acosta, 2002; Roig-Juñent et al., 2003; Silva et al., 2018b; Roig-Juñent & Quiroga, 2021).
Composition, bionomy and comments of the subfamilies collected in Ventania.
In our study, Orthocladiinae was the most abundant subfamily in number of species and individuals along the streams. This subfamily contains many cold stenothermic genera susceptible to elevated temperatures and reduced oxygen (Ashe et al., 1987). Within this subfamily, the tribe Metriocnemini with the genus Limnophyes was best represented upstream, and Orthocladiini downstream.
Podonominae was the least abundant in both number of species and total individuals. Immature stages of this subfamily require high levels of dissolved oxygen, relatively low temperature, and clear water (Brundin, 1966). In our study, this subfamily prevailed in the hill section of the Ventania streams, where the necessary conditions for development and survival of this subfamily are present. Historically, in South America Podonominae was associated with an Andean-Patagonic distribution (Brundin, 1966). However, in the last years, studies also showed an extra-Andean distribution including different sites along elevated mountain ranges in southern South America (Roque &Trivinho-Strixino 2004; Rodríguez et al., 2009; Siri & Donato, 2012; Shimabukuro et al., 2017; Pinho & Shimabukuro, 2018). From the five Podonominae genera reported in the Andean-Patagonic region, three of them (Podonomus Philippi, Parochlus Enderlein and Podonomopsis Brundin) were collected in the Ventania hill system. The present study represents the first extra Andean report of the Podonomopsis genus for South America. Moreover, the reports of Podonomus and Podonomopsis for this area represent the reports at lower altitudes for both genera.
Tanypodinae and Chironominae showed an intermediate number of species and individuals. Many species of these subfamilies tolerate high temperatures and reduced oxygen levels.Chironominae subfamily was well represented downstream, by many individuals belonging to widely distributed genera. Concerning the Tanypodinae subfamily, the Macropelopiini tribe and the genus Pentaneura reported in Ventaniawere previously collected in cold environments, while remaining genera of Tanypodinae have a wide distribution.
Implication for conservation policies
It is widely known that, in response to the reduction of habitats, declines in biodiversity and species richness have been documented for almost all freshwater taxa, with losses in freshwater communities being some of the most severe (Strayer & Dudgeon, 2010). Except for the PPET, the Ventania hill system and its proximities have been widely impacted through different land uses. The uses of agricultural lands are dominated by crops covering most of the area. Tourism has widely increased in the last decades, where the old villages have largely expanded, and new villages have been built. Due to the high ancestry, Ventania have been part of important evolutionary, geologic and climatic processes, then the biota current result of these events. Since Chironomidae is a set of spatially and temporally integrated taxa where their assemblage is the result of a common history, they could be used to characterize zones as well as in conservation studies of areas. Therefore, we are aware of the importance of the Ventania hill system, so we provide a broad and baseline contribution about the systematic and ecology knowledge of an insect group widely used in biomonitoring and provide biological information that supports the importance for conserving this area.
Acknowledgments
The authors wish to thank Jorge Donadelli (ILPLA) for the water physicochemical analysis, AnibalAreco and all the park rangers of PPET for helping us in the field work and to OPDS for logistic support and for granting authorization to collect. The paper is the Scientific Contribution Nº1119 of the Institute of Limnology ''Dr. R.A. Ringuelet'' (ILPLA), CCT-La Plata, CONICET, UNLP. This study was partially supported by CONICET and AgenciaNacional de PromociónCientífica y Tecnológica (PICT-2008-1910).
REFERENCES
Acosta, L.E. (2002) Patrones zoogeográficos de los opiliones argentinos (Arachnida: opiliones). RevistaIbérica de Aracnología, ., 69-84.
APHA (American Public Health Association). (1998) Standard methods for the examination of water and wastewater. (Hanover, USA: American Water Works Association).
Andersen, T., Cranston, P.S., &Epler, J.H. (2013). Chironomidae of the Holarctic Region. Keys and diagnoses-Larvae. Insect Systematics & Evolution, Supplement, 66, 573.
Ashe, P., Murray, D.A., & Reiss, F. (1987) The zoogeographical distribution of Chironomidae (Insecta: Diptera). Annales de Limnologie, 23, 27-60.
Brundin, L. (1966) Transantarctic relationships and their significance, as evidenced by chironomid midges. With a monograph of the subfamilies Podonominae and Aphroteniinae and the Austral Heptagyiae. KunglikaSvenskaVetenskapakadamiensHandlingar, 11, 1-474.
Cabrera, A.L., &Willink, A. (1973) Biogeografía de América Latina [Monografía 13], Serie de Biología. OEA, Washington, DC.
Campos, R.E. (2015) Aquatic Diptera assemblages in four sympatric Eryngium (Apiaceae) phytotelmata in flowering and senescent times. Journal of Natural History, 50, 1-17.
Cortelezzi, A., Simoy, M., Siri, A., Donato, M., Marinelli, C.B., Cepeda, R. &Berkunsky, I. (2020) New insights on bioindicator value of Chironomids by using occupancy modelling. Ecological Indicators, 117.
Crisci, J.V., Freire, S.E., Sancho, G., &Katinas, L. (2001) Historical biogeography of the Asteraceae from Tandilia and Ventaniamountain ranges (Buenos Aires, Argentina). Caldasia, 23, 21-41.
de Mendiburu, F. (2019) Agricolae: Statistical Procedures for Agricultural Research. R package version 1.3-1. Available at https://CRAN.R-project.org/package=agricolae
Donato, M., Siri, A., & Mauad, M. (2012) Description of a new species of the genus Onconeura Andersen etSaether (Diptera: Chironomidae) from Argentina with a cladistic analysis of the genus. Zootaxa, 3580, 43-55.
Epler, J.H. (2001). Identification manual for the larval Chironomidae (Diptera) of North and South Carolina. A guide to the taxonomy of the midges of southeastern United States, including Florida. Available at http://johnepler.com/SEMidges.pdf.
Fernández, L.A., Torres, P.L.M, Michat, M.C, Fischer, S., Oliva, A., &Bachmann, A.O. (2010) Coleópteros acuáticos y semiacuáticos del Parque Provincial Ernesto Tornquist (Provincia de Buenos Aires, Argentina). Revista de la Sociedad EntomológicaArgentina, 69, 189-199.
Ferreti, N., Pompozzi, G, Copperi, S, Pérez-Miles, F, González, A. (2012) Mygalomorph spider community of a natural reserve in a hillysystem in Central Argentina. Journal of Insect Science, 12(31), 1-16.
Ferrington, Jr.L.C. (2008) Global diversity of non-biting midges (Chironomidae; Insecta-Diptera) in freshwater. Hydrobiologia, 595, 447-455.
Frenguelli, J. (1950) Rasgos generales de la morfología y la geología de la provincia de Buenos Aires. Laboratorio de Ensayo de Materiales e Investigaciones Tecnológicas, La Plata, Argentina.
Guerrero, E. (2019) Harvestmen (Arachnida: Opiliones) of the protected areas of the province of Buenos Aires and the Autonomous City of Buenos Aires, Argentina. Revista de la Sociedad Entomológica Argentina, 78(4), 4-13.
Hammer, Ø., Harper, D., & Ryan, P.D. (2001) PAST: Paleontological statistics software package for education and data analysis. Palaeontologia Electronica 4:1-9. Available at http://palaeo-electronica.org/2001_1/past/issue1_01.htm
Konopko, S., Mazzuconi, S., López Ruf, M., & Bachmann, A. (2009). Los heterópteros acuáticos y semiacuáticos del Parque Provincial Ernesto Tornquist (Provincia de Buenos Aires, República Argentina). Revista de la Sociedad Entomológica Argentina, 68(3-4), 271-282.
Kristensen, M. J., & Frangi, J.L. (1995). La Sierra de la Ventana: Una Isla de Biodiversidad. Ciencia Hoy, ., 25-34.
Makowski, D., Lüdecke, D., & Ben-Shachar, M.S. (2021) Package “correlation”: Methods for Correlation Analysis, version 0.6.1.Available at https://CRAN.R-project.org/package=correlation
Marino, P.I., Cazorla, C, Díaz, M.F. (2011) Los Ceratopogonidae (Diptera: Culicomorpha) del Sistema Serrano de Ventania (Buenos Aires, Argentina). Revista de la Sociedad Entomológica Argentina, 70(3-4), 197-205.
Mauad, M., Siri, A., & Donato, M. (2013) New species of Pseudosmittia Edwards, 1932 and new records of Allocladius Kieffer, 1913 (Diptera: Chironomidae, Orthocladiinae) from South America. Zootaxa, 3694, 445-460.
Moreno, C.E. (2001) Métodos para medir la biodiversidad. M&T–Manuales y Tesis SEA, vol. 1. Zaragoza.
Naughton-Treves, L., Holland, M.B., & Brandon, K. (2005) The role of protected areas in conserving biodiversity and sustaining local livelihoods. Annual Review of Environment and Resources, 30, 219-252.
Ocon, C., Siri, A., Altieri, P., & Donato, M. (In press) Functional feeding groups of Chironomidae (Diptera: Nematocera) and their spatial variation in an intermittent hill stream (Ventana stream, Buenos Aires, Argentina). Annals of the Brazilian Academy of Sciences.
Ogle, D., Doll, J., Wheeler, P., & Dinno, A. (2022) FSA: Simple Fisheries Stock Assessment Methods. R (≥ 3.5.0). Available athttps://cran.r-project.org/web/packages/FSA/
Oksanen, J., Blanchet, F. G., Kindt, R., Legendre, P., Minchin, P. R., O´hara, R.B., Simpson, G.L., Solymos, P., Henry, M., Stevens, H., Szoecs, E., & Wagner, H. (2013) Package ‘vegan’. Community ecology package, version, 2(9), 1-295. Available at http://CRAN.R-project.org/package=vegan
Paggi, A.C. (2007). A new Neotropical species of the genus ThienemanniellaKieffer, 1911 (Diptera: Chironomidae, Orthocladiinae). Contribution to systematics and ecology of aquatic Diptera - A Tribute to Ole A. Sæther. (ed. Trond, A.), pp. 247-254. The Caddis Press, Columbus, Ohio, USA.
Pinho, L.C., & Shimabukuro, E.M. (2018) Podonomusamarali n. sp., a new species of the albinervis group (Diptera: Chironomidae: Podonominae) from Atlantic Forest, southern Brazil. Zootaxa, 4402, 542-550.
Pompozzi, G., Copperi, G., Schwerdt, L., Ferreti, N. (2014) On the ecology of the cursorial spider Odobruchi (Araneae: Zoridae) in a grassland natural reserve from central Argentina. Revista de Biología Tropical, 62(1), 87-93.
R Core Team. (2020) R: A language and environment for statistical computing.R Foundation for Statistical Computing, Vienna, Austria. Available at http://www.r-project.org/index.html
Ringuelet, R.A. (1961a) Un nuevo opilión de Sierra de la Ventana, Physis, 21, 326-327.
Ringuelet, R.A. (1961b). Rasgos fundamentales de la zoogeografía de la Argentina, Physis, 22, 151-170.
Rodríguez, M., Paggi, A.C., & Medina, A.I. (2009) A new Neotropical species of the genus Parochlus Enderlein, 1912 (Chironomidae: Podonominae) and new distribution in Argentina. Zootaxa, 2300, 39-46.
Roig-Juñent, S.A, Flores, G.E., & Mattoni, C. (2003) Consideraciones biogeográficas de la Precordillera (Argentina), con base en artrópodos epígeos. Una perspectiva latinoamericana de la biogeográfica (eds. Morrone J.J., & Llorente, J.), pp. 275-288. Las Prensas de Ciencias, UNAM, México, D. F.
Roig-Juñent, S.A., & Quiroga, C.R. (2021) Nuevas especies de carábidos y consideraciones biogeográficas del arco peripampásico. Revista de la Sociedad Entomológica Argentina, 80(1): 10-22.
Roque, F. de O., & Trivinho-Strixino, S. (2004) Podonomuspepinellii n. sp., first record of thegenus and subfamilyfromBrazil (Diptera: Chironomidae: Podonominae). Zootaxa, 689, 1-7.
Scheibler, E.E., Roig-Juñent, S., & Claps, M. (2014) Chironomid (Insecta: Diptera) assemblages along an Andean altitudinal gradient. Aquatic Biology, 20, 169-184.
Schwerdt, G., Copperi, S., Pompozzi., G., & Ferreti, N. (2016) Diversity and seasonal composition of the scorpion fauna from a mountainous system on pampean grasslands in central Argentina. Studies on Neotropical Fauna and Environment, 51(3), 1-7.
Shimabukuro, E.M., Pepinelli, M., & Trivinho-Strixino, S. (2017) Two new species of Podonomus Philippi (Diptera: Chironomidae: Podonominae) from mountains of Southeastern Brazil. Zootaxa, 4290, 155-166.
Silva, F. da L., Pinho, L.C., Wiedenbrug, S., Dantas, G.P.S., Siri, A., Andersen, T., & Trivinho-Strixino, S. (2018a). Family Chironomidae. Freshwater Invertebrates (eds. Hamada, N., Thorp, J.H. & Rogers, D.C.), pp. 661-700. Elsevier, Academic Press.
Silva, G.T., San Blas, G., Peçanha, W.T., Moreira, G.R.P., & Gonçalves, G.L. (2018b) Phylogeography of the gall-inducing micromothEucecidosesminutanusBrèthes (Cecidosidae) reveals lineage diversification associated with the Neotropical Peripampasic Orogenic Arc. PLOS ONE, 13(8), e0201251.
Siri, A., & Donato, M. (2012) Two new species of Podonomus (Diptera: Chironomidae: Podonominae) of the Brundin’sdecarthrus group from Ventania system, Argentina. Zootaxa, 3848, 39-54.
Siri, A., Donato, M., & Fuentes, M.C. (2014) New record of Parochluscarolinae (Chironomidae: Podonominae) from the Ventania hill system with the description of the female. Revista de la Sociedad Entomológica Argentina, 73, 171-175.
Siri, A., & Donato, M. (2015). Phylogenetic analysis of the tribe Macropelopiini (Chironomidae: Tanypodinae): adjusting homoplasies. Zoological Journal of the Linnean Society, 174, 74-92.
Soriano, A., León, R.J.C., Sala, O.E., Lavado, R.S., Deregibus, V.A., Cahuepé, M.A., Scaglia, O.A., Velázquez, C.A., Lemcoff, J.H. (1992) Río de la Plata grasslands. Ecosystems of the world 8A. Natural grasslands. Introduction and western hemisphere. (ed. Coupland, R.T.), pp. 367-407, Elsevier, New York.
Sota, E.A. de la. (1967) Composición, origen y vinculación de la flora pteridológica de las sierras de Buenos Aires (Argentina). Boletín de la Sociedad Argentina de Botánica, 11, 105-128.
Spies, M., Andersen, T., Epler, J.H., & Watson, C.N.j. (2009) Chironomidae (non-bitingmidges). Manual of Central American Diptera (eds. Brown, B.V., Borkent, A., Cumming, J.M., Wood, D.M., Woodley N.E., & Zumbado, M.A.), pp. 437-480. NRC Research Press, Ottawa, Canada.
Strayer, D. L., & Dudgeon, D. (2010) Freshwater biodiversity conservation: recent progress and future challenges. Journal of the North American Benthological Society, 29, 344-358.
Tejerina, E.G., & Molineri, C. (2007) Comunidades de Chironomidae (Diptera) en arroyos de montaña del NOA: comparación entre Yungas y Monte. Revista de La Sociedad Entomológica Argentina, 66(3-4), 169-177.
Zanotto-Arpellino, J.P., Principe, R.E., Oberto, A.M., & Gualdoni, C.M. (2015) Variación espacio-temporal de Chironomidae (Diptera) bentónicos y derivantes en un arroyo serrano en Córdoba, Argentina. Iheringia. Série Zoologia, 105(1), 41-52.
Zanotto-Arpellino, J.P., Rodriguez Catanzaro, L.N.S, Montalto, L., Siri, A., Donato, M. (In press) Diversity, phenology and voltinism of Chironomidae (Diptera). Neotropical streams as a study model. Annals of the Brazilian Academy of Sciences.
Notas de autor
augusto@ilpla.edu.ar