Artículos científicos
Recepción: 30 Marzo 2022
Aprobación: 02 Agosto 2022
Abstract: Sericulture, often known as silk farming is the practice of rearing silkworms for production of raw silk. One of the major impediments faced by the sericulture community is the silkworm diseases that cause significant crop losses. In the present study, the efficacy of phototrophic bacteria as feed supplements in improving the economic traits as well as disease resistance in mulberry silkworm, Bombyx mori L. was assessed. A total of three phototrophic bacteria were isolated and identified as belonging to the genera Marichromatium, Rhodobacter and Rhodopseudomonas based on 16S rRNA gene sequencing analysis. Their morphological, physiological and biochemical characters were studied, grown under ambient conditions and biomass was harvested. Mulberry fortified with the aforementioned phototrophic bacteria in solitude and combinations at varying concentrations (0.5, 1, 2 and 5%) were found to be innocuous to the mulberry silkworm. The best results were observed when mulberry leaf was supplemented with Rhodopseudomonas sp. at 2% concentration which improved survival, cocoon weight, shell weight, shell%, filament length and non-breakable filament length by 5.18, 0.27, 3.86, 3.5, 1.5 and 1.2%, respectively under normal rearing conditions. Under Staphylococcus sp. infected conditions, the aforesaid diet also enhanced survival in silkworms by 16%. On the other hand, the same diet didn’t exhibit any discernible influence on survival against fungal, viral, or microsporidian infections in silkworm.
Keywords: Bombyx mori, Feed supplements, Fortification, Mulberry, Phototrophic bacteria.
Resumen: La sericultura, a menudo conocida como cultivo de la seda, es la práctica de criar gusanos de seda para la producción de seda cruda. Uno de los principales impedimentos a los que se enfrenta la sericultura son las enfermedades de los gusanos de seda que causan importantes pérdidas en la cosecha. En el presente estudio, se evaluó la eficacia de las bacterias fototróficas como suplementos alimenticios para mejorar los rasgos económicos así como la resistencia a las enfermedades en el gusano de seda de morera, Bombyx mori L. Se aislaron tres bacterias fototróficas y se identificaron como pertenecientes a los géneros Marichromatium, Rhodobacter y Rhodopseudomonas, basándose en el análisis de secuenciación del gen 16S rRNA. Se estudiaron sus caracteres morfológicos, fisiológicos y bioquímicos, se cultivaron en condiciones ambientales y se cosechó la biomasa. Se comprobó que la morera enriquecida con las mencionadas bacterias fototróficas en soledad y en combinaciones a distintas concentraciones (0, 5, 1, 2 y 5%) era inocua para el gusano de seda de la morera. Los mejores resultados se observaron cuando se complementó la hoja de morera con Rhodopseudomonas sp. a una concentración del 2%, que mejoró la supervivencia, el peso del capullo, el peso de la cáscara, el porcentaje de cáscara, la longitud del filamento y la longitud del filamento no rompible en un 5,18, 0,27, 3,86, 3,5, 1,5 y 1,2%, respectivamente, en condiciones normales de cría. En condiciones de infección por Staphylococcus sp., la citada dieta también aumentó la supervivencia de los gusanos de seda en un 16%. Por otra parte, la misma dieta no mostró ninguna influencia discernible en la supervivencia contra las infecciones fúngicas, víricas o de microsporidios en los gusanos de seda.
Palabras clave: Bacterias fototróficas, Bombyx mori, Fortificación, Morera, Suplementos alimenticios.
INTRODUCTION
The silk industry, an agro based venture is distributed over sixty nations across the globe and mulberry silk produced by Bombyx mori L. is predominant in comparison to the other natural silks (Eri, Muga, and Tasar) (Dewangan, 2013; Giacomin et al., 2017). Bombyx mori L. is a monophagous, domesticated lepidopteran insect which derives its nutrition from mulberry leaves. Major biomolecules such as proteins, carbohydrates, lipids as well as minerals, vitamins, amino acids and water derived from mulberry play a significant role in biochemical and physiological processes underlying growth, development and disease resistance in silkworm (Dong et al., 2017). However, a range of abiotic and biotic factors influence the quality of mulberry which in turn will have a severe impact on the aforesaid biological mechanisms in silkworm, thereby affecting the silk productivity (Mahadeva, 2018).
Apart from the mulberry leaf attributes, the other imperative factor that plays a significant role in the success of a cocoon crop is management of silkworm diseases which account roughly for about 15-20% of crop losses (Reddy and Rao, 2009). The most frequently encountered diseases include grasserie, caused by a virus; flacherie, caused by bacteria, non-occluded viruses [Bombyx mori infectious flacherie virus (BmIFV), Bombyx mori densonucleosis virus (BmDNV)] and both in combination; muscardine, caused by fungi; and pebrine, a microsporidian parasitic disease (Rahul et al., 2019). Researchers across the world have and are exploring a variety of regimens aimed to improve the economic traits as well as survival in silkworm, each with its own set of benefits and drawbacks. Fortification of mulberry leaves with diverse supplements is one among the most investigated approaches over the last three decades aimed at fulfilling the aforementioned purpose (Etebari et al., 2004).
Mulberry leaves sprayed with vitamins, minerals, amino acids, nitrogenous compounds, plant extracts, antibiotics, probiotics, hormones and hormone analogues are tested and most of the compounds were found to have a better influence on developmental aspects as well as survival in silkworm (Etebari et al., 2004; Savithri, 2007; Muthuswami et al., 2011; Bai & Bai, 2012; Konala et al., 2013; Kumar & Balasubramanian, 2014; Rahul et al., 2017a; Rahul et al., 2017b; Saad et al., 2019). However, a few of our earlier studies also cited that the productive parameters were not significant (Rahul et al., 2017a; Rahul et al., 2017b). Owing to the high cost of most of the aforementioned compounds, their practical usage at the field level is very limited. Hence, an economically viable method that could improve the economic traits and instigate disease resistance in silkworm is considered necessary. In this direction, the present study was designed to explore the role of phototrophic bacteria as feed supplements.
Anoxygenic phototrophic bacteria are a physiologically and phylogenetically diverse clade of Gram negative bacteria (George et al., 2020). These bacteria have a general ability to photosynthesize under anoxic conditions in the presence of light employing bacteriochlorophylls and without emancipation of oxygen. In contrast to oxygenic phototrophs (cyanobacteria, algae and green plants), they make use of a single photosystem and are not capable of using water as an electron donor (Imhoff, 1995). They are widely distributed across diverse habitats and the presence of varied photosynthetic pigments such as carotenoids bestow them with distinct coloration (green, yellowish-green, brown, brownish-green, brownish-red, red, pink, purple, pink to red and purple-violet depending on the pigment content). Anoxygenic phototrophic bacteria encompass members of the groups of purple sulfur bacteria (PSB), purple non sulfur bacteria (PNSB), green sulfur bacteria, green non sulfur bacteria, heliobacteria and aerobic anoxygenic phototrophic bacteria (Imhoff, 1995).
The bacterial biomass of anoxygenic phototrophic bacteria is widely exploited for diverse applications. They are widely employed as feed fortifiers in aquaculture, pisciculture and poultry; single cell proteins, biocolorants, antioxidants, source of vitamins, hormones, quinones, and in biodegradation (Sasikala & Ramana, 1995). Presence of biological cofactors, digestible cell wall, carotenoids and being rich sources of protein, fat and vitamins make them possible candidates for being explored as feed supplements. Considering the aforesaid advantages, this study has been aimed to explore the efficacy of phototrophic bacteria treated mulberry leaves in improving the economic traits and disease management in silkworm.
MATERIALS AND METHODS
Culturing of purple phototrophic bacteria (PSB/PNSB)
Sediment and water samples from Chilika Lagoon, Odisha, India were collected in zip-lock polythene covers and sterile screw cap glass tubes (20 ml), respectively. Salinity (Salinity meter, ILS Corp, India) and pH (pH indicator strips, Merck Millipore) of the samples were measured at the collection site. The samples were brought to the laboratory within two days of collection and refrigerated.
For enriching PNSB, sediments (0.5 g) or water samples (0.5 ml) were inoculated in modified mineral salts medium (Biebl & Pfennig, 1981) {(g.l-1): KH.PO. - 0.5. MgSO..7H.O - 0.5; NaCl - 0.5/20 (depending on salinity of sample); NH.Cl - 0.6; CaCl..2H.O - 0.05; Sodium pyruvate - 3.0; Yeast extract - 0.3; Ferric citrate solution (0.1%, w/v) - 5 ml; Vitamin B12 solution (2 mg/100 ml of distilled water followed by filter sterilization employing a 0.2 µm cellulose acetate membrane filter) - 1 ml; Micronutrient solution SL7 [(mg.l-1): HCl (25%, v/v) - 1 ml; ZnCl. - 70; MnCl..4H.O - 100; H.BO. - 60; CoCl..6H.O - 200; CuCl..H.O - 20; NiCl..6H.O - 20; NaMoO..2H.O - 40. pH was adjusted to 6.8-7 with 1N NaOH/1N HCl before autoclaving] - 1 ml}. Final pH of the medium was adjusted to 7-7.5 with sterile HCl (1N)/NaOH (1N). Whereas for enriching PSB, apart from the above ingredients, sodium bicarbonate (10%, w/v) - 10 ml and Na.S.9H.O (1 M; prepared in screw cap glass tubes, flushed with nitrogen gas for two minutes and sterilized by autoclaving) - 1 ml were also supplemented. All the aforementioned chemicals used in the preparation of media were obtained from Himedia, India.
The inoculated fully filled screw cap bottles (to maintain anaerobic conditions) of 42 ml capacity were incubated under illumination (2,400 lux) at 30+2 °C. Enrichment cultures of PSB and PNSB were purified by streaking repeatedly on agar slants with modified mineral salts medium under Nitrogen gas phase. The purity of the isolated bacteria was determined through evaluation of its attributes which included color, colony morphology, and microscopic examination. Pure cultures were stored at 4 °C in refrigerator and intermittent transfer to fresh media was performed every ten days.
Morphological characterization
The colony characters on the agar slants were recorded by visual observation whereas cellular morphological properties of the isolated strains were determined upon Gram staining followed by visualization under a phase contrast microscope (Leitz). Gram-staining of the bacterial isolates was performed using a Gram-staining kit [HiMedia (Cat No: K001)] following manufacturer’s instructions.
Physiological and biochemical characterization
Saline requirement and tolerance
The saline tolerance of the isolated phototrophic bacteria was tested using varying concentrations of sodium chloride in mineral salts medium with pyruvate (0.3%, w/v) and NH.Cl (0.06%, w/v) as carbon and nitrogen sources, respectively. Media devoid of NaCl was used as control. Growth was measured turbidometrically (OD540) upon incubation.
Growth at different temperatures
The phototrophs isolated were cultured in growth media by incubating at varying temperatures (4, 25, 30, 35, 40, 45 and 50 °C) and growth was examined turbidometrically (OD540).
Growth at different pH
The pH range for growth was determined by inoculating the bacterial isolates in buffered medium (Xu et al., 2005) adjusted to different pH values (pH 4.0-11.0, intervals of 1 units) as delineated earlier (Rahul et al., 2014) at optimal salinity followed by incubation at appropriate temperature. The optical density was measured at 540 nm upon incubation.
Utilization of carbon sources
The ability of the isolated phototrophs to utilize acetate, arabinose, citrate, ethanol, fructose, glucose, glutamate, melibiose, malate, proline, pyruvate, succinate, sucrose and xylose was examined by inoculating the axenic cultures (1%) in mineral salts medium with ammonium chloride (0.06%, w/v) and yeast extract as sources of nitrogen and growth factor, respectively along with the aforementioned test compounds as the solitary source of carbon and energy. Growth was measured turbidometrically (OD540) upon incubation.
Utilization of nitrogen sources
In mineral salts medium with pyruvate (0.3%, w/v) as only source of carbon, a range of compounds which include NaNO./NaNO./NH.Cl (0.06-0.068% w/v); C.H10N.O./C.H.NO./CH₄N₂O (0.06-0.068%, w/v) as nitrogen sources was tested. Control consisted media devoid of nitrogen source. Growth was assessed turbidometrically (OD540) upon incubation.
Identification of phototrophic bacteria
DNA from pure cultures of phototrophic bacteria was extracted with DNeasy Blood and Tissue kit (Qiagen) following instructions of the manufacturer. PCR amplification was performed in a DNA thermal cycler (Eppendorf) and the reaction mixture consisted forward (Eub27F: 5'-GAGTTTGATCCTGGCTCAG-3') and reverse (Univ1492R: 5' GGTTACCTTGTTACGACTT-3') primers (1 μl each), template DNA (2.5 μl), nuclease free water (8 μl) and PCR master mix (12.5 μl; Himedia) (Rahul et al., 2014). The mixture was subjected to a single cycle of initial denaturation at 96 °C for 2 min, followed by 32 cycles of denaturation at 96 °C for 40 sec, annealing at 50 °C for 40 sec, extension at 72 °C for 1 min and final extension at 72 °C for 5 min. The PCR amplicons thus obtained were purified by using a QIA quick PCR purification kit (Qiagen). Horizontal agarose gel electrophoresis [2% (w/v) in TAE buffer at 15 V cm-1] of PCR amplicons (5 μl) and DNA marker (5 μl, Himedia) was performed to assess the quality of the amplified DNA. The electrophoresed gel was subjected to ethidium bromide (0.5 μg ml-1) staining and visualized in a gel documentation system (Biorad). The PCR amplicons were sequenced with the aforementioned primers at M/s. Xcelris Labs Limited, Gujarat, India. Nearest phylogenetic relatives were determined by aligning the obtained sequences against EZ BioCloud 16S rRNA gene database (Yoon et al., 2017). The neighbor joining algorithm in MEGA 6 software was used to create a phylogenetic tree using the Kimura-2 parameter in a pairwise deletion process. Percentage support values were acquired by means of a bootstrap process with 1000 iterations (Tamura et al., 2013).
Culturing of major silkworm pathogens
The type cultures of causal micro organisms of major silkworm diseases viz., bacteria (Staphylococcus sp.), fungi (Beauveria bassiana (Balsamo) Vuillemin),virus [Bombyx mori nuclear polyhedrosis virus (BmNPV)] and microsporidian (Nosema bombycis Nageli) available at Central Sericultural Research & Training Institute-Berhampore, India were used in this study. The culturing conditions as delineated (Sivaprasad et al., 2021) were followed. In brief, Staphylococcus sp. and B. bassiana were cultured in nutrient broth and potato dextrose broth (Himedia), respectively. The incubation conditions for the pathogenic bacterium were 30±2 °C for 36 hours whereas for the fungal pathogen, it was 30 °C for 96 hours. Bacterial enumeration was determined by serial dilution technique while fungal conidial concentration was counted upon employing a Neubauer haemocytometer.
Haemolymph from grasserie infected silkworm larvae was collected in sterile distilled water, left undisturbed for overnight at room temperature and centrifuged (10000 rpm, 10 min) to isolate BmNPV polyhedra. The pellet thus obtained was thoroughly rinsed thrice with distilled water before being washed a couple of times with sterile saline solution. Gradient centrifugation was employed for subsequent purification of the BmNPV Polyhedra and counts of the same was determined with the aid of a Neubauer haemocytometer. Pebrine inoculated larvae were macerated in potassium carbonate solution (0.6%; 3 ml/g of fresh weight) and filtered using a muslin cloth. The filtrate was subjected to centrifugation (2500 rpm, 10 min) and the resulting microsporidian spore pellet was rinsed thrice with saline before being purified using Percoll based iso-density equilibrium centrifugation. Neubauer haemocytometer was used to assess the spore count.
Silkworm rearing
Disease free layings (DFLs) of the silkworm hybrid, SK6 x SK7 were obtained from the Bivoltine silkworm breeding laboratory of CSRTI-Berhampore. Silkworms were reared on plastic trays in a 5% bleaching powder disinfected silkworm rearing room according to the standard conditions as delineated earlier (Datta, 1992). They were fed the mulberry leaves of the variety S-1635. The optimum temperature and relative humidity (RH) maintained during the rearing of I-III instar larvae were 26-28 °C and 80-90%, respectively whereas for the late stage larvae (IV and V instar), it was 24-25 °C with 70-75% RH. Light and dark photoperiods of 12 h each were maintained during the entire course of larval rearing. A temperature of 25 °C and RH of 70% was maintained throughout the spinning process.
Bioassay studies
The isolated phototrophic bacteria were grown under standard conditions based on the physiological and biochemical characterization. Upon attaining the log phase, the bacterial biomass was harvested by centrifugation at 10,000 rpm for 10 min. The cell pellet was washed thrice with sterile water to remove traces of media components and spent wastes. The pellet was dried and diluted to different concentrations (0.5, 1, 2 and 5%) in sterile water. The same were sprayed (1, 3 and 5 ml/100 larvae during the third, fourth and fifth instars respectively) on to the mulberry leaves and allowed to air dry. The fortified leaves were fed to the silkworm larvae (SK6 x SK7) once daily from the first day of third instar. The mulberry fed to the control worms was sprayed with sterile water. Three replicates of hundred larvae each were maintained per treatment. Various rearing (mature larval weight, effective rate of rearing (ERR), cocoon weight, shell weight and shell ratio) and reeling traits (filament length, non-breakable filament length and denier) were recorded as delineated earlier with minor modifications (Rahmathulla et al., 2007; Chattopadhyay et al., 2018). Mature larval weight was determined by measuring the weight of V instar silkworm larvae (5 Nos each of male and female) at the maturation stage, using an electronic balance (Sartorius). In a similar way, the cocoon and shell weights were measured whereas the shell percentage was calculated by using the formula (shell weight/cocoon weight) x 100. ERR% or effective rate of rearing percentage which implies survival in silkworms was determined by measuring the ratio of the number of good cocoons harvested to the number of larvae retained in each treatment multiplied by 100. The total length of silk filament unwound from a single cocoon, measured in meters indicates the filament length which is measured by subjecting the cocoons from each treatment to stifling in boiling water and threads reeled using an epprouvette and observed for silk filament length whereas non-breakable filament length is the length of filament that can be unwound from the cocoon under test without a break and is calculated using the formula [Filament length in m/ (1 + no. of breaks)]. Denier represents the thickness of silk filament which is calculated using the formula [(Weight of the filament (g)/ Length of the filament (m)) x 9000]. The feed supplement performing better in terms of improving the aforementioned rearing and reeling traits in silkworm was only chosen to study its efficacy against silkworm pathogens through in vivo studies, the methodology of which is as follows.
The major silkworm pathogen suspensions [(Staphylococcus spp. (1.7 x 107 cfu/ml), BmNPV (1 x 107 polyhedra/ml) and N. bombycis (1 x 107 spores/ml)] were smeared onto the mulberry leaves individually, air-dried and fed to the III instar silkworm larvae [(SK6 x SK7); 1 ml/100 larvae]. B. bassiana (2.5 x 106 conidia/ml), the fungal pathogen suspension was smeared directly onto the silkworm larval surface as it infects via per cutaneous route. Mulberry leaf fortified with phototrophic bacteria (Rhodopseudomonas sp.) at 2% concentration was fed as delineated above. Another batch of larvae inoculated with pathogens and fed with mulberry leaves sprayed with sterile water was maintained as control. Five replicates of 50 larvae each were maintained per treatment. The experiment was conducted across three seasons/crops during December, 2018; March, May, 2019. The silkworm larvae were reared under standard rearing conditions as mentioned above. The observations recorded include the development of disease symptoms and larval mortality in each treatment. The incidence of silkworm diseases was recorded for respective treatments based on typical disease symptoms, microscopic examination of haemolymph/midgut.
Statistical analysis
The data recorded with respect to various rearing and reeling traits of B. mori being fed feed supplement at different concentrations and devoid of feed supplement was analyzed using an online software tool ‘OPSTAT’ (Sheoran et al., 2020) and expressed as mean ± standard error of the mean. The significant differences between the means at 1% probability of all experimental groups were determined by one-way analysis of variance (ANOVA). Using the aforementioned tool, Student's t-test was performed to determine the significant difference in ERR% values recorded between the two groups (normal and fortified diet). Significance levels were set at P< 0.05.
RESULTS AND DISCUSSION
A total of six samples were collected from Chilika Lagoon in order to enrich phototrophic bacteria. Phototrophic bacteria were reported to be isolated from a wide variety of habitats including Chilika Lagoon earlier (Imhoff, 2017). Of the six samples subjected to enrichments and subsequent purification, three phototrophic bacterial strains (one purple sulfur bacterium and two purple non sulfur bacteria) were isolated in axenic form and were identified by 16S rRNA gene sequencing analysis. Based on the same, the purple sulfur bacterium belonged to the genera Marichromatium exhibiting a similarity of 100% with Marichromatium indicum Arunasri et al. JA100T. Phylogenetic analysis on the basis of 16S rRNA gene sequences exhibited that the strain clustered with species of the genus Marichromatium and formed a distinct clade with type strains of M. indicum and Marichromatium purpuratum (Imhoff & Trüper) Imhoff et al. (Fig. 1). One strain of the purple non sulfur bacteria belonged to the genus Rhodobacter Imhoff et al. with 99.56% similarity to Rhodobacter vinaykumarii Srinivas et al. DSM 18714T whereas the other strain exhibited a 100% similarity to Rhodopseudomonas thermotolerans Kumar et al.JA576T. A neighbor joining tree constructed based on the 16S rRNA gene sequences of the nearest type strains of the genus Rhodobacter indicated that the isolated strain clustered distinctly with the type strain of R. vinaykumarii. Rhodopseudomonas sp. clustered with the type strains of R. thermotolerans, Rhodopseudomonas pentothenatexigens Kumar et al. and Rhodopseudomonas palustris (Molisch) van Niel. The results of phylogenetic analysis further justified their assignment to the concerned genera (Fig. 2 & 3). The 16S rRNA gene sequences of Marichromatium sp., Rhodobacter sp. and Rhodopseudomonas sp. isolated in the present study were deposited at the European Molecular Biology Laboratory with the accession numbers LR797537, LR797538 and LR797539 respectively.
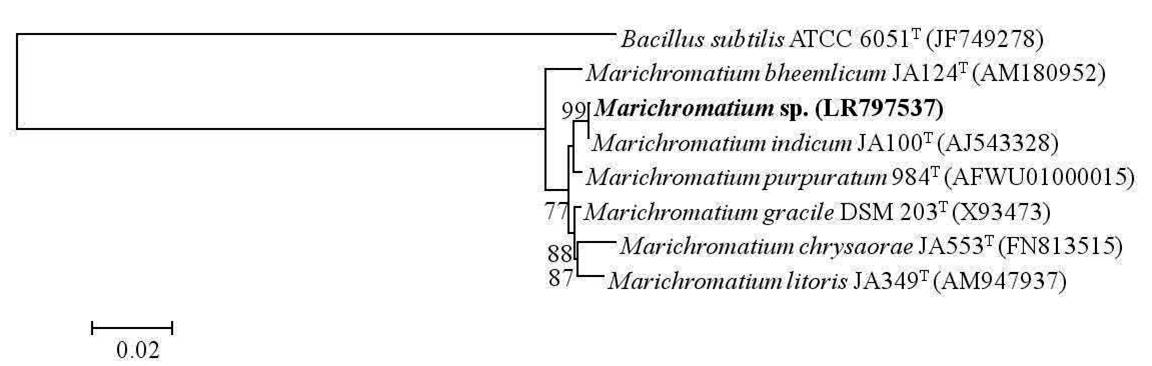
Fig. 1
Dendrogram depicting the phylogenetic relationship of Marichromatium sp. isolated in the present study with its closest phylogenetic neighbors within the genus Marichromatium determined using 16S rRNA gene sequence analysis. Bacillus subtilis ATCC 6051T is used as an out-group. Bar, 2 nucleotide substitution per 100 nucleotides
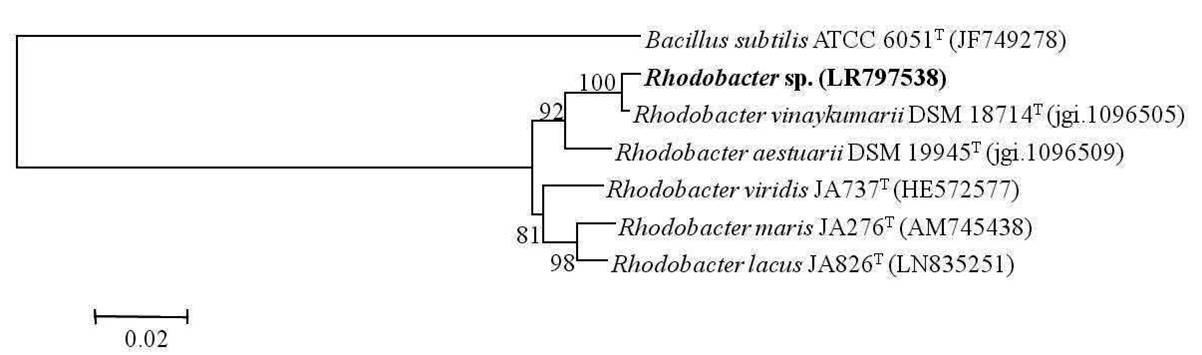
Fig. 2.
Dendrogram depicting the phylogenetic relationship of Rhodobacter sp. isolated in the present study with its closest phylogenetic neighbors within the genus Rhodobacter determined using 16S rRNA gene sequence analysis.Bacillus subtilis ATCC 6051T is used as an out-group. Bar, 2 nucleotide substitution per 100 nucleotides
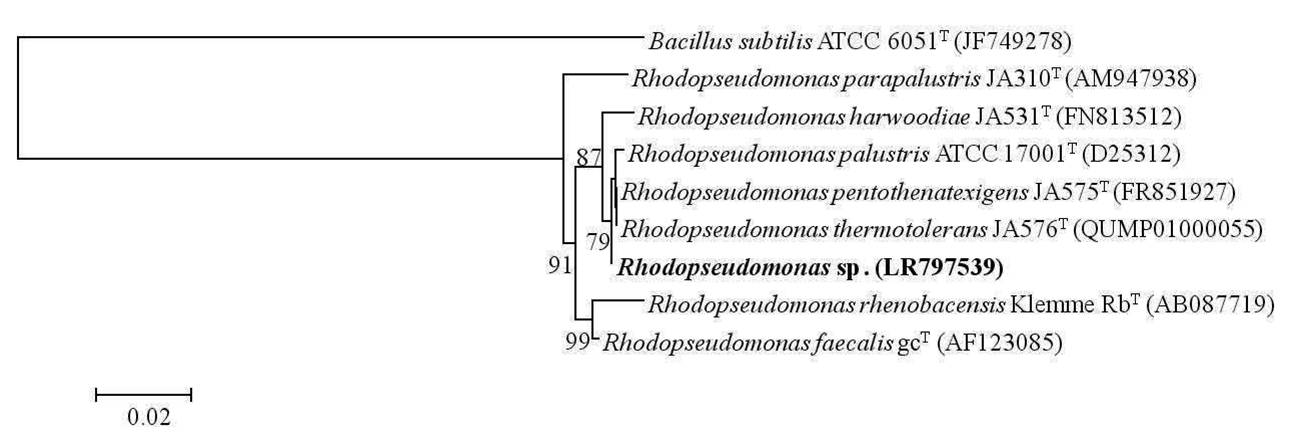
Fig. 3.
Dendrogram depicting the phylogenetic relationship of Rhodopseudomonas sp. isolated in the present study with its closest phylogenetic neighbors within the genus Rhodopseudomonas determined using 16S rRNA gene sequence analysis. Bacillus subtilis ATCC 6051T is used as an out-group. Bar, 2 nucleotide substitution per 100 nucleotides.
The morphological, physiological and biochemical characterization of the isolates was carried out and the results of the same was presented in Table I & Fig. 4. All the three phototrophic bacterial strains experimented in the present study were Gram negative rods. All the strains were mesophiles. The purple non sulfur bacterial strains were neutrophiles where as good growth of Marichromatium sp. was observed at a slightly acidic pH. Rhodobacter sp. and Marichromatium sp. have obligate requirement for sodium chloride where as Rhodopseudomonas sp. can grow devoid of the same. The purple non sulfur bacterial strains grew with pyruvate as a carbon source where as Marichromatium sp. could utilize fructose. Ammonium chloride as a nitrogen source supported growth of all the three strains.
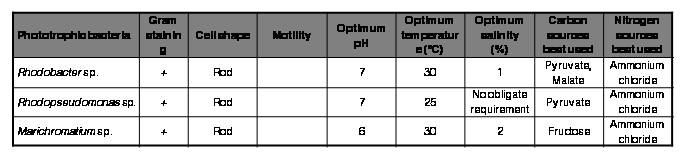
Table I.
Morphological, physiological and biochemical characteristics of the isolated phototrophic bacterial strains.
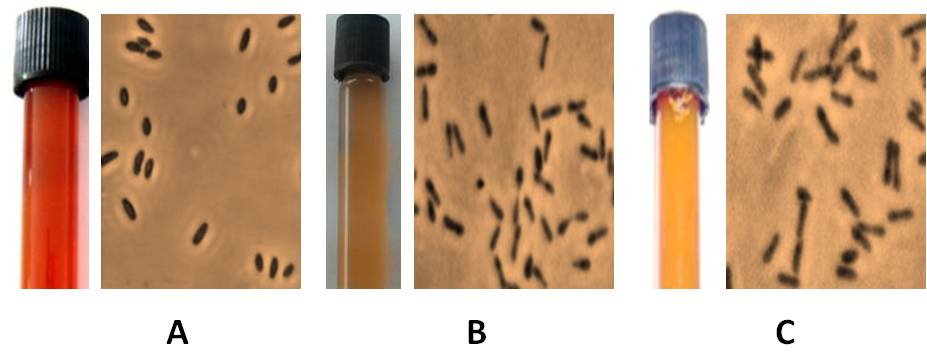
Fig. 4.
Broth culture and phase contrast micrograph of Marichromatium sp. (A), Rhodobacter sp. (B) and Rhodopseudomonas sp. (C) isolated in the present study.
The feed supplements (FS) individually and in combination at 0.5, 1, 2 and 5% did not exert any toxic effect on silkworms. This is evident from the ERR% values which did not significantly deter with respect to the control silkworms (Tables II, III, IV and V). Mulberry fortified with Marichromatium sp. and a combination of all the three strains couldn’t exert any beneficial effect in silkworms as there is no enhancement in terms of survival, rearing and reeling traits recorded in comparison to the control group of larvae (Tables II and V).
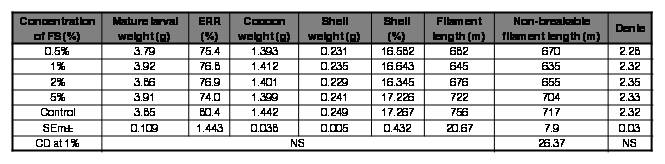
Table II.
Effect of FS (Marichromatium sp.) on rearing and reeling traits of Bombyx mori [SK6 x SK7].
FS: feed supplement; ERR: effective rate of rearing; NS: Non-significant.
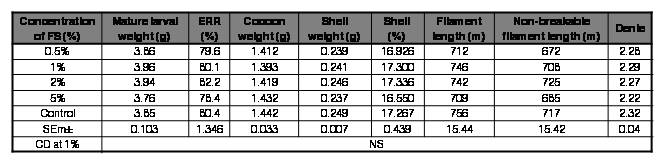
Table III.
Effect of FS (Rhodobacter sp.) on rearing and reeling traits of Bombyx mori [SK6 x SK7].
FS: feed supplement; ERR: effective rate of rearing; NS: Non-significant.
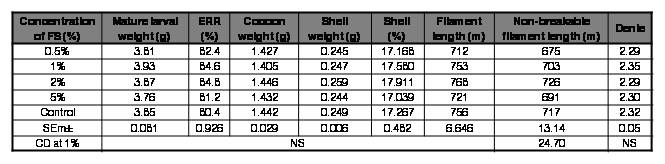
Table IV.
Effect of FS (Rhodopseudomonas sp.) on rearing and reeling traits of Bombyx mori [SK6 x SK7]
FS: feed supplement; ERR: effective rate of rearing; NS: Non-significant
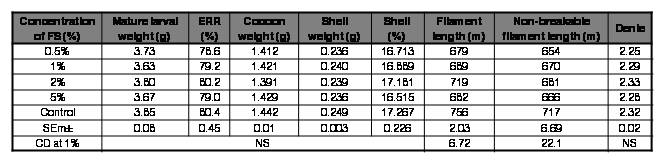
Table V.
Effect of FS (Marichromatium/Rhodobacter/Rhodopseudomonas sp.) on rearing and reeling traits of Bombyx mori [SK6 x SK7]
FS: feed supplement; ERR: effective rate of rearing; NS: Non-significant
However, mulberry fortified with Rhodopseudomonas sp. at all the concentrations tested improved survival in silkworm in the ranges of 0.98-5.18%, with 2% being the best. There was also a marginal enhancement in all the rearing and reeling traits with the mulberry diet supplemented with Rhodopseudomonas sp. at 2% (Table IV). Rhodobacter sp. at 2% improved survival by 2.1% and also exhibited a slender enhancement in shell%, non-breakable filament length and denier. Although the improvement in values of the traits seems to be trivial, they can have a profound influence at the field level; where in the farming fraternity rear a huge quantum of DFLs.
Since, the performance of experimental batches that were fed mulberry fortified with Rhodopseudomonas sp. at 2% performed relatively better in terms of all the traits tested, the same was chosen for further experimentation. The efficacy of the aforementioned diet against major silkworm pathogens was assessed by in vivo studies. The results were depicted in Table VI. The feed supplement was found to be effective against the bacterial pathogen Staphylococcus sp. The survival increased by 16% in batches that were inoculated with the pathogen and fed a feed supplement over control batches reared on normal diet. Though the survival of batches inoculated with BmNPV and fed a feed supplement was high over control batches, it was not statistically significant. The feed supplement was not effectual against fungal and microsporidian pathogens tested. The phototrophic bacterial cellular components (proteins, lipids, vitamins, biological co-factors) and not the bacterial metabolites extruded in the nutrient medium may indeed be accountable for the feed supplement's enhancement in rearing and reeling traits of silkworm. However, the resistance against bacterial infection may be attributed to the diverse bioactive components, most notably carotenoids encompassed by phototrophic bacteria. The antibacterial and antioxidant potential of carotenoids is previously emphasized by other researchers (Keceli et al., 2013).
Researchers spanning the domains of aquaculture, pisciculture and allied fields have earlier demonstrated the usage of phototrophic bacteria as feed supplements and their beneficial effects. To list a few, Rhodovulum sulfidophilum (Hansen & Veldkamp) Hiraishi & Ueda, fortified fish feed improved survival and growth characteristics in Oreochromis niloticus (L)(Banerjee et al., 2000). Penaeus monodon F., the giant tiger prawn being fed with R. sulfidophilum fortified diet also exhibited improvement in growth and survival (Azad, 2002). The author also reported that supplementation of higher concentrations of phototrophic bacterial biomass (>3%) didn’t result in any significant beneficial effects in Penaeus monodon. The results in the present study also corroborate with the same although the organisms tested were different. The role of photosynthetic bacteria (strains belonging to the genera Rhodopseudomonas, Rubrivivax, Rhodobacter and Phaeospirillum) since 1980s in transformation of Chinese aquaculture was reported (Qi et al., 2009). The Asian Seabass, Lates calcarifer (Bloch) on feeding Rhodovulum sp. fortified diet exhibited better growth rate with a substantial increase in weight (Shapawi et al., 2012). Significant improvements in rotifer (Brachionus rotundiformis Tschuqunoff) density was observed when R. palustris, Rhodobacter sphaeroides (van Niel) Imhoff et al. and R. sulfidophilum were employed as feed supplements (Loo et al., 2013). Malaysian Mahseer, Tor tambroides (Bleeker) on being fed Marichromatium sp. supplemented diet exhibited improvement in survival, growth as well as proximate composition (Chowdhury et al., 2016). Improvement in weight, feed conversion ratio, growth rate and tolerance to ammonia stress was observed in Penaeus vannamei (Boone) on feeding shrimp feed fortified with Rhodopseudomonas sp. The same phototrophic bacteria also exhibited inhibitory activity against vibrio pathogens (in vitro) (Alloul et al., 2021). All the aforementioned beneficial aspects of phototrophic bacteria across diverse organisms are attributable to the rich nutritive values possessed by them (Qi et al., 2009). Also, most of the aforementioned researchers have emphasized the economic viability of the phototrophic bacterial feed supplements.
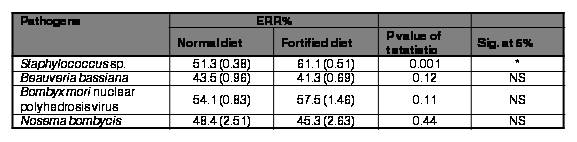
Table VI.
Effect of feed supplement (Rhodopseudomonas sp. at 2%) on survival of Bombyx mori against major silkworm pathogens.
Figures in () indicate log transformed values
Although numerous reports exist with respect to application of phototrophs as feed fortifiers in numerous sectors, there are a very few reports on using phototrophic bacteria as feed additives in sericulture to our knowledge. However, employing a photosynthetic bacterium as a component of artificial feed for silkworms is cited in a European patent (Kobayashi et al., 2009). A Japanese patent (JP59071650) possibly from the same author as cited above indicated that Rhodopseudomonas sphaeroides van Niel, a phototrophic (purple non sulfur) bacterium was employed in artificial feed for silkworms. This helped in attaining uniform growth and disease resistance in silkworms as well as increased ovi-position ratio of moths. Although this Japanese patent cited was not accessible at present, but the information was accessible from another patent document (Kumar et al., 2011). The results obtained from the present study corroborates with the findings as cited in the above patent documentation and the phototrophic bacterium isolated in the present study (Rhodopseudomonas sp.) can be a potential component of silkworm feed, either fresh mulberry or more importantly in the artificial diets considering the attributes which include palatability, on par or improved economic traits and resistance to bacterial infections.
Although artificial diets in silkworm rearing were first conducted in the 1960s in Japan (Ito, 1980; Shinbo & Yanagaw, 1994), their implementation at the field level in countries like India has been largely unsuccessful due to the high expenses involved. A few farmers although use artificial feed, they restrict its usage to the chawki stage (young age larvae). As a result, developing a low-cost artificial feed for silkworm rearing is a viable option, and phototrophic bacterium such as Rhodopseudomonas sp. can undoubtedly play a significant role as the cost involved in its culturing is low.
To our knowledge, this is the first comprehensive report on employing phototrophic bacterial feed supplementation and assessing the silkworms’ economic traits as well as disease resistance. The findings from this study clearly indicated that Rhodopseudomonas supplemented mulberry diet was effective in improving economic features and reducing Staphylococcus sp. induced bacteriosis in silkworms. This feed supplement can play a significant role in silkworm rearing across the tropics, wherein bacterial infections are high and cause considerable cocoon crop losses to the sericulture fraternity. Although, it is evident from this study about the beneficial aspects of phototrophic bacterial supplemented diet in mulberry silkworm rearing, the contributing metabolites pertaining to the same are yet to be identified. Detailed studies on metabolite profiling employing analytical techniques such as mass spectroscopy between the silkworms reared on normal and phototrophic bacterial enriched diets might portray necessary information in the future.
Acknowledgments
The financial assistance received from Central Silk Board, Ministry of Textiles: Govt. of India for carrying out the present work (Project ARP3590) is gratefully acknowledged. KR thanks Dr. Zakir Hossain and Dr. Satadal Chakrabarty for their guidance and support.
REFERENCES
Alloul, A., Wille, M., Lucenti, P., Bossier, P., Stappen, G.V., & Vlaeminck, S.E. (2021) Purple bacteria as added-value protein ingredient in shrimp feed: Penaeus vannamei growth performance, and tolerance against Vibrio and ammonia stress. Aquaculture, 530, 735788.
Azad, S.A. (2002) Phototrophic bacteria as feed supplement for rearing Penaeus monodon larvae. Journal of the World Aquaculture Society, 33(2), 158-168.
Bai, P.K.K.S., & Bai, M.R. (2012) Studies on the effect of a probiotic and a neutraceutical agent on growth, development and commercial characteristics of silkworm, Bombyx mori L. Indian Journal of Sericulture, 5(1), 37-42.
Banerjee, S., Azad, S.A., Vikineswary, S., Selvaraj, O.S., & Mukherjee, T.K. (2000) Phototrophic bacteria as fish feed supplement. Asian-Australasian Journal of Animal Sciences, 13(7), 991-994.
Biebl, H., & Pfennig, N. (1981) Isolation of members of the family Rhodospirillaceae. The Prokaryotes - a handbook on habitats, isolation and identification of bacteria (ed. Starr, M.P., Stolp, H., Trüper, H.G., Balows, A., & Schlegel, H.G.), pp. 267-273. Springer-Verlag, USA.
Chattopadhyay, D., Munshi, R., & Chakravorty, D. (2018) Studies on distribution of filament length and non-broken filament length for tropical tasar and muga silk cocoons vis-à-vis mulberry silk cocoons. Journal of the Textile Institute, 109(9), 1202-1207.
Chowdhury, A.J.K., Zakaria, N.H., Abidin, Z.A.Z., & Rahman, M.M. (2016) Phototrophic purple bacteria as feed supplement on the growth, feed utilization and body composition of Malaysian Mahseer, Tor tambroides juveniles. Sains Malaysiana, 45(1), 135-140.
Datta, R.K. (1992) Guidelines for bivoltine rearing. Central Silk Board, Bangalore, India.
Dewangan, S.K. (2013) Livelihood opportunities through sericulture a model of Gharghoda tribal block, Raigarh Dist. American Journal of Environmental Science, 9(4), 343-347.
Dong, H.L., Zhang, S.X., Tao, H., Chen, Z.H., Li, X., Qiu, J.F., Cui, W.Z., Sima, Y.H., Cui, W.Z., & Xu, S.Q. (2017) Metabolomics differences between silkworms (Bombyx mori) reared on fresh mulberry (Morus) leaves or artificial diets. Scientific Reports, 7(1), 10972.
Etebari, K., Kaliwal, B.B., & Matindoost, L. (2004) Different aspects of mulberry leaves supplementation with various nutritional compounds in sericulture. International Journal of Industrial Entomology, 9(1), 15-28.
George, D.M., Vincent, A.S., & Mackey, H.R. (2020) An overview of anoxygenic phototrophic bacteria and their applications in environmental biotechnology for sustainable resource recovery. Biotechnology Reports, 28, e00563.
Giacomin, A.M., Garcia Jr., J.B., Zonatti, W.F., Silva-Santos, M.C., Laktim, M.C., & Baruque-Ramos, J. (2017) Brazilian silk production: economic and sustainability aspects. Procedia Engineering, 200, 89-95.
Imhoff, J.F. (1995) Taxonomy and physiology of phototrophic purple bacteria and green sulfur bacteria. Anoxygenic photosynthetic bacteria. (ed. Blankenship, R.E., Madigan, M.T., & Bauer, C.E.), pp. 1-15. Kluwer Academic Publishers, The Netherlands.
Imhoff, J.F. (2017) Diversity of anaerobic anoxygenic phototrophic purple bacteria. Modern topics in the phototrophic prokaryotes. (ed. Hallenbeck, P.), pp. 47-85. Springer, Cham.
Ito, T. (1980) Application of artificial diets in sericulture. Japan Agricultural Research Quarterly, 14(3), 163-168.
Keceli, T.M., Erginkaya, Z., Turkkan, E., & Kaya, U. (2013) Antioxidant and antibacterial effects of carotenoids extracted from Rhodotorula glutinis strains. Asian Journal of Chemistry, 25(1), 42-46.
Kumar, S.N., Trivedy, K., Ramesh, M., & Dandin, S.B. (2011) Universal semi synthetic diet for young instar silkworm, multi x bivoltine hybrids of silkworm Bombyx mori. Indian patent, 247304.
Kumar, K., & Balasubramanian, U. (2014) Supplementary effect of Spirulinaon lipids and enzymes in silk gland of silkworm, Bombyx mori (L.). Journal of Entomology and Zoology Studies, 2(4), 279-282.
Kobayashi, J., Nakamura, M., & Yokoyama, J. (2009) Artificial diet for lepidopteran and method for producing the same, lepidopteran and method for producing the same, and biomaterial. European patent, 2022339 - EP07743580A1 - EPO.
Konala, N., Abburi, P., Bovilla, V.R., & Mamillapalli, A. (2013) The effect of bovine milk on the growth of Bombyx mori. Journal of Insect Science, 13, 98.
Loo, P.L., Vikineswary, S., & Chong, V.C. (2013) Nutritional value and production of three species of purple non-sulphur bacteria in palm oil mill effluent and their application in rotifer culture. Aquaculture Nutrition, 19, 895-907.
Mahadeva, A. (2018) Insect pest infestation, an obstacle in quality mulberry leaves production. Asian Journal of Biological Sciences, 11, 41-52.
Muthuswami, M., Bhaskar, R.N., & Naveen, V. (2011) Role of food additives on young age silkworm (Bombyx mori L) rearing. International Journal of Pure and Applied Sciences and Technology, 7(2), 132-140.
Qi, Z., Zhang, X.H., Boon, N., & Bossier, P. (2009) Probiotics in aquaculture of China - Current state, problems and prospect. Aquaculture, 290, 15-21.
Rahmathulla, V.K., Das, P., Ramesh, M., & Rajan, R.K. (2007) Growth rate pattern and economic traits of silkworm, Bombyx mori L under the influence of folic acid administration. Journal of Applied Sciences and Environmental Management, 11(4), 81-84.
Rahul, K., Moamongba, K., Rabha, M., & Sivaprasad, V. (2019) Identification and characterization of bacteria causing flacherie in mulberry silkworm, Bombyx mori L. Journal of Crop and Weed, 15(3), 178-181.
Rahul, K., Roy, G., Hossain, Z., & Trivedy, K. (2017a) Impact of probiotics Lactobacillus rhamnosus ATCC 9595 and Lactobacillus acidophilus ATCC 4356 on the economic traits of silkworm Bombyx mori L. Imperial Journal of Interdisciplinary Research, 3(3), 1115-1117.
Rahul, K., Roy, G., Hossain, Z., & Trivedy, K. (2017b) Effect of mulberry leaves enriched with yeast extract on silkworm Bombyx mori L. Journal of Research in Science Technology Engineering and Management, 3(1), 14-16.
Rahul, K., Sasikala, C., Tushar, L., Debadrita, R., & Ramana, C.V. (2014) Alcanivorax xenomutans sp. nov., a hydrocarbonoclastic bacterium isolated from a shrimp cultivation pond. International Journal of Systematic and Evolutionary Microbiology, 64, 3553-3558.
Reddy, B.K., & Rao, J.V.K. (2009) Seasonal occurrence and control of silkworm diseases, grasserie, flacherie and muscardine and insect pest, uzi fly in Andhra Pradesh, India. International Journal of Industrial Entomology, 18(2), 57-61.
Saad, M.S.I., Elyamani, E.M.Y., & Helaly, W.M.M. (2019) Controlling of bacterial and fungal diseases that contaminating mulberry silkworm, Bombyx mori by using some plant extracts. Bulletin of the National Research Centre, 43, 172.
Sasikala, C., & Ramana, C.V. (1995) Biotechnological potentials of anoxygenic phototrophic bacteria. I. Production of single-cell protein, vitamins, ubiquinones, hormones, and enzymes and use in waste treatment. Advances in Applied Microbiology, 41, 173-226.
Savithri, G. (2007) In vivo effects of antibiotics on silkworm Bombyx mori L infected with Bacillus coagulans. International Journal of Industrial Entomology,15(1), 9-16.
Shapawi, R., Ting, T.E., & Azad, S.A. (2012) Inclusion of purple non sulfur bacterial biomass in formulated feed to promote growth, feed conversion ratio and survival of Asian Seabass Lates calcarifer juveniles. Journal of Fisheries and Aquatic Science, 7(6), 475-480.
Sheoran, O.P., Kumar, V., & Tonk, M. (2020) Web based statistical analysis tool for series of experiments in randomized block design cum pooled analysis. International Journal of Agriculture and Statistical Science, 16(1), 1101-1108.
Shinbo, H., & Yanagaw, H.A. (1994) Low-cost artificial diets for polyphagous silkworms. Japan Agricultural Research Quarterly, 28, 262-267.
Sivaprasad, V., Rahul, K., & Makwana, P. (2021) Immunodiagnosis of silkworm diseases. Methods in silkworm microbiology. (ed. Gurtler, V., & Subrahmanyam, G.), pp. 27-46. Elsevier Academic Press, United Kingdom.
Tamura, K., Stecher, G., Peterson, D., Filipski, A., & Kumar, S. (2013) MEGA 6: Molecular Evolutionary Genetics Analysis version 6.0. Molecular Biology and Evolution, 30(12), 2725-2729.
Xu, P., Li, W.J., Tang, S.K., Zhang ,Y.Q., Chen, G.Z., Chen, H.H., Xu, L.H., & Jiang, C.L. (2005) Naxibacter alkalitolerans gen. nov., sp. nov., a novel member of the family ‘Oxalobacteraceae’ isolated from China. International Journal of Systematic and Evolutionary Microbiology, 55, 1149-1153.
Yoon, S.H., Ha, S.M., Kwon, S., Lim, J., Kim, Y., Seo, H., & Chun, J. (2017) Introducing EzBioCloud: A taxonomically united database of 16S rRNA and whole genome assemblies. International Journal of Systematic and Evolutionary Microbiology; 67, 1613-1617.
Notas de autor
rahulk.csb@nic.in
