Artículos científicos
Insecticidal and Repellent Actions of Methanolic Extracts from Five Medicinal Plants against the Saw-Toothed Grain Beetle Oryzaephilus surinamensis (Linn.) (Coleoptera: Silvanidae)
Acciones insecticidas y repelentes de extractos metanólicos de cinco plantas medicinales contra el escarabajo de los cereales con dientes de sierra Oryzaephilus surinamensis (Linn.) (Coleoptera: Silvanidae)
Insecticidal and Repellent Actions of Methanolic Extracts from Five Medicinal Plants against the Saw-Toothed Grain Beetle Oryzaephilus surinamensis (Linn.) (Coleoptera: Silvanidae)
Revista de la Sociedad Entomológica Argentina, vol. 81, núm. 3, 2022
Sociedad Entomológica Argentina
Recepción: 25 Agosto 2021
Aprobación: 05 Agosto 2022
Abstract: The insecticidal and repellent activities of methanolic extracts of Ruta chalepensis L. (Rutaceae: Sapindales), Rhazya stricta Decne (Apocynaceae: Gentianales), Heliotropium bacciferum Forssk (Boraginales: Boraginaceae), Salvadora persica L. (Salvadoraceae: Brassicales), and Moringa oleifera L. (Moringaceae: Brassicales) were investigated against the saw-toothed grain beetle Oryzaephilus surinamensis L. (Coleoptera: Silvanidae). Results demonstrated that all these plant extracts had variable toxicity against the beetle. The effectiveness of the tested plant extracts was shown by increasing the used concentrations. The methanolic extract from R. stricta performed the best and was able to cause 93.3% and 89.2% mortality for the adult and larval stage of O. surinamensis, respectively, at 500 ppm after six days of plant extract treatment. The tested extract of S. persica at 500 ppm caused 80.8% and 82.5% mortality on larvae and adults of O. surinamensis after six-day of treatment. On the other hand, repellent effects of extracts of R. stricta, S. persica, and .. chalepensis at 500 ppm were recorded, with higher values of 91.7%, 83.3%, and 78%, respectively, against the adults of O. surinamensis, which were under 500 ppm. These extracts especially from R. stricta, S. spinosa, and .. chalepensis could be useful to prevent damages and store grain production losses for O. surinamensis.
Keywords: Bioassay, Heliotropium bacciferum, Methanolic plant extracts, Moringa oleifera, Oryzaephilu surinamensis, Repellency, Rhazya stricta, Ruta chalepensis, Salvadora persica.
Resumen: Se investigó la actividad insecticida y repelente de los extractos metanólicos de Ruta chalepensis L. (Rutaceae: Sapindales), Rhazya stricta Decne (Apocynaceae: Gentianales), Heliotropium bacciferum Forssk (Boraginales: Boraginaceae), Salvadora persica L. (Salvadoraceae: Brassicales) y Moringa oleifera L. (Moringaceae: Brassicales) contra el escarabajo de los cereales Oryzaephilus surinamensis L. (Coleoptera: Silvanidae). Los resultados demostraron que todos estos extractos vegetales tenían una toxicidad variable contra el escarabajo. La eficacia de los extractos ensayados se puso de manifiesto al aumentar las concentraciones utilizadas. El extracto de R. stricta fue el que mejor funcionó y fue capaz de causar una mortalidad del 93,3% y del 89,2% en el estado de adulto y de larva de O. surinamensis respectivamente a 500 ppm tras seis días de tratamiento. El extracto de S. persica a 500 ppm causó un 80,8% y un 82,5% de mortalidad en larvas y adultos de O. surinamensis respectivamente tras seis días de tratamiento. Por otra parte, se registraron efectos repelentes de los extractos de R. stricta, S. persica y R. chalepensis a 500 ppm, con valores superiores al 91,7%, 83,3% y 78% respectivamente contra los adultos de O. surinamensis, que estaban por debajo de 500 ppm. Estos extractos, especialmente los de R. stricta, S. spinosa y R. chalepensis, podrían ser útiles para prevenir los daños y las pérdidas de producción de grano almacenado de O. surinamensis.
Palabras clave: Bioensayo, Extracto metanólico de plantas, Heliotropium bacciferums, Moringa oleifera, Oryzaephilu surinamensis, Repelencia, Rhazya stricta, Ruta chalepensis, Salvadora persica.
INTRODUCTION
Damage of cereal grains through infestation of insects during the storage phase has become a severe challenge, predominantly in developing countries. Studies indicated that the stored grains, as well as products' damage due to insect pests, may amount to about 5% to 10% losses in temperate areas, whereas losses may range from 20% to 30% in tropical areas (Ileke & Oni, 2011; Akinneye & Ogungbite, 2013). Therefore, crop protection in this field must play vital and essential roles in contemporary agronomic production; the ever-lasting yield demands, as well as increased farming practices, have resulted in a rise in pest damage and control (Martins et al., 2012). The saw-toothed grain beetle Oryzaephilus surinamensis L. (Coleoptera: Silvanidae) is amongst serious pests of different categories of food, including nuts, dry fruits, spices, and cereal, in addition to other foodstuffs (Huda & Noor, 2019). The saw-toothed grain beetle O. surinamensis has a global distribution, especially in tropical and subtropical areas (Huda & Noor, 2019). Stored grain infestation usually occurs from top to bottom or vice versa through migration of pests such as rice weevils Sitophilus oryzae L. (Coleoptera: Curculionidae), saw-toothed grain beetle O. surinamensis, and red flour beetle Tribolium castaneum Herbs (Coleoptera: Tenebrionidae), known as vertical infestation (Kachhwaha et al., 2015). Studies reported that spoiled kernels due to insect attack by O. surinamensis are more susceptible than the whole grains. Hence, seeking more suitable methods of controlling stored-grains pests is required (Kachhwaha et al., 2015). Indeed, managing the infestation of pests in stored grains is predominantly attained by applying synthetic chemicals together with phosphine and methyl bromide (Pimentel et al., 2008). However, quite a lot of chemical insect repellents have been outlawed or regulated in many nation-states due to environmental concerns and hazards associated with human healthiness (Tapandjou et al., 2002).
Synthetic insecticides including artificial pyrethroids, carbamates, organophosphorus, and organochlorides are usually utilized for controlling stored-grain pests. However, applying these chemicals can lead to decreased grain quality, smelly and hazardous substances to human, and air pollution as a side effect of insecticides on the environment. The new chemical insect repellents' adverse effects have guided researchers in finding innovative pathways of controlling insects, resulting in the discovery of plants' products as a substitute method of managing insect pests. This method can be more effective against specific target pest species, and biodegradable into nontoxic produces and are suitable for application in integrated pest management programs (Akinkurolere et al., 2006; Sutherland et al., 2002; Zibaee, 2011).
Several studies demonstrated that some plant extracts such as Neem Azadirachta indica L. (Sapindales: Meliaceae) have insecticidal effects on Khapra beetle Trogoderma granarium Everts (Coleoptera: Bostrichidae) with variable levels (Mahmoud et al., 2015). Correspondingly, Haj et al. (2016) reported that Usher Calotropis procera Aiton (Apocynaceae), Argel Solenostemma argel Hayne (Asclepiadaceae), and Datura stramonium L. (Solanaceae) were more effective against the adults' saw-toothed grain beetles. Khemais et al. (2018) concluded that the essential oils of the flowering aerial portions of Ruta chalepensis were confirmed to be toxic and had physiological effects on Tribolium confusum Duval (Coleoptera: Tenebrionidae) as well. Their results also demonstrated that the maximum exposure time resulted in a maximum repellency of this pest. Moreover, Alvi et al. (2018) concluded that the leaf and seed extracts of R. chalepensis were toxic and repellent to T. granarium and Rhyzopertha dominica F.(Coleoptera: Bostrichidae) under research laboratory settings.
Moreover, Eldidamony et al. (2020) recorded that the extracts of Heliotropium curassavicum could be considered as rodenticides in the framework of IPM programs. Similarly, sprayings Moringa (Moringa oleifera L.) plant's extracts (seed and root extracts) within cowpea crop were capable of reducing several field insect pests, including Megalurothrips sjostedti Trybom, Aphis craccivora Koch., and Maruca vitrata F., as well as the infestation by stored insect Callosobruchus maculatus L. which is reduced after harvesting cowpea (Ojiako et al., 2013).
The present study was carried out in an attempt to explore alternative, effective, and environmentally safe insecticides, specifically of botanical origins against the larval and adult stages of the saw-toothed grain beetle O. surinamensis. We investigated the biological effects (i.e., toxicity and repellency) of five medicinal plant extracts, including Ruta chalepensis L. (Rutaceae: Sapindales), Rhazya stricta Decne (Apocynaceae: Gentianales), Heliotropium bacciferum Forssk (Boraginales: Boraginaceae), Salvadora persica L. (Salvadoraceae: Brassicales), and Moringa oleifera L. (Moringaceae: Brassicales) against this dangerous stored-grain pest.
MATERIALS AND METHODS
Insect Culture
The adults and larvae of the saw-toothed grain beetles were obtained from permanent colonies of the saw-toothed grain beetle O. surinamensis in the Laboratory of Plant Protection, the Department of Arid Land Agriculture, King Abdulaziz University. The culture was kept at about 28±2.0.C and arranged 75±5% relative dampness inside jars enclosed with a muslin material. The insects were sustained by interchanging the consumed grains with new uncontaminated paddy rice collected from the market.
Extraction and Preparation of Plants' Material
The fresh twigs of R. chalepensis, R. stricta, H. bacciferum, S. persica, and M. oleifera were obtained from different places in Riyadh region (Al-Duwadimi, Al-Aflaj, Al-Qaseem, and Shagra) which is located in the center part of Saudi Arabia. These plants grown naturally without any treatment of pesticides. The plants were identified and collected by the expert staff of the Department of Arid Land Agriculture, Faculty of Meteorology, Environment and Arid Land Agriculture, King Abdulaziz University. The abovementioned plants were dried under dark condition for four weeks, ground separately into fine particles, put through a sieve (1 mm mesh), and then preserved in distinct plastic bottles at 4.C with close-fitting lids before use. Methanolic extracts of R. chalepensis, R. stricta, H. bacciferum, S. persica, and M. oleifera were obtained through the cold extraction technique. Briefly, around 150 g of the powder was mixed discretely in extraction bottles with 500 ml of methanol. After that, the mixture was stirred using a glass rod; after seventy-two hours, the extraction was prepared to use. The resultant mix was passed through a double layer (Whatman No. 1) filter paper. The solvent was placed in a rotary evaporator at temperatures ranging from 30.C to about 40.C with a three to six rpm rotary speed for eight hours. Then, subsequent solid substances produced by the evaporation of methanol from used plants were separately air-dried in order to remove all traces of the used solvent (methanol), resulting in a final stock solution of each tested plant. From these stock solutions, different concentrations of 200, 300, 400, and 500 ppm were separately prepared for each tested plant through the use of distilled water. Preliminary tests were applied on larval and adult stages of O. surinamensis in order to determine these experimental concentrations.
Bioassays with Plant Extracts on O. surinamensis
About 50 g of paddy rice was weighed into 250 ml plastic jars. Afterwards, 1 ml of tested plant extracts of 200, 300, 400, and 500 ppm was separately mixed with the paddy. The mixtures were left for an hour in order to certify volatile solvent evaporation. Control treatment received only 1 ml of distilling water. Thirty of the 3rd instars of larvae of O. surinamensis were separately moved into separate jars which included the mixtures of paddy rice and tested concentrations of each plant as well as the control treatment. Furthermore, this procedure was applied on thirty adults of O. surinamensis following the same method for the larval stage. Four replicates of each concentration from the investigated plants and untreated plants (control) were carried out in a Randomized Complete Block Design (RCBD). Both larvae and adult mortality were separately observed and recorded at 2, 4, and 6 days after treatments.
Repellence Plant Extract Assessment
The methodology employed in testing repellency was in line with the techniques of Talukder & Howse (1994) and Kundu et al. (2007). The Petri dishes contained treated as well as untreated new grain portions. Filter papers were then cut into two uniform halves. One ml solution of each concentration from each plant extract was added to one half using a pipette. Consequently, treated filter papers were then air-dried. They were brought together with the untreated half through the use of a cello-tape at the center, and they did not inhibit the beetles' free movements from a single half of the paper to the other portion of the filter-paper. Every filter paper was then placed in a Petri dish of about nine centimeters in diameter. Newly emerged O. surinamensis adults were then unconfined at the middle of each filter paper in groups of 20, and the Petri dish was closed. Each concentration was replicated three times for each plant extract. The insects on each half of the paper strip were then tallied after every two hours. The obtained data were finally presented as a percentage of repulsion (P.R.) according to the method of Jilani et al. (1988).
Data Analyses
The percentages of mortality of the larvae and adults of O. surinamensis were calculated for each observed point (2, 4, and 6 days). Then, each of them was separately analyzed by one-way ANOVA in order to assess the effect of concentrations of five plants (20 experimental units with control). Before applying ANOVA, data transformation using a Log 10 (Max+1-X) was performed in order to meet the normality and improve variances. Where significant treatment differences (P≤0.05) were observed, Fisher's least significant difference (LSD) tests were conducted in order to identify differences between treatment means. All data analyses were carried out within SPSS version 2.0 (IBM Corporation, 2011).
Moreover, the values of LC50 (the lethal concentration to cause 50% mortality) were separately calculated for the larvae and adults of O. surinamensis, which is in line with the study of Finney (1971), while data correction for controlled mortality was adopted from Abbott's formula (1925). In order to determine the best of the five used plants with tested concentrations (20 experimental units) as a repellent of emerged adults of O. surinamensis, the calculated percentages of repellency test were subjected to one-way ANOVA. However, untransformed means and standard errors are illustrated in order to simplify interpretation, and a significance difference is considered at P≤0.05.
RESULTS AND DISCUSSION
Plant Extracts' Toxicity on O. surinamensis larvae and adults
The present study demonstrated that the used plants with different concentrations for each observation time had higher significant differences on the mortality of larvae of the saw-toothed grain beetle O. surinamensis after 2 days (F20.63=13.04, P˂0.001), 4 days (F20.63=14.28, P˂0.001), and 6 days (F20.63=13.26, P˂0.001). The larvae of O. surinamensis indicated the highest mortality (89.2%) when larvae were treated with the extract of R. stricta at the concentration of 500 ppm after 6 days followed by 80.83, 72.5, 71.67, and 68.33% mortality when larvae were treated with the extract of S. persica, R. chalepensis, M. oleifera, and H. bacciferum, respectively, at the same concentration and observed time (Fig. 1). Both concentrations of R. stricta and S. persica were the most effective, and the mortality of beetle larvae ranged from 60.8 to 89.2% and from 55.0 to 80.8% at 6 days, respectively (Fig. 1). Similarly, the results of toxicity presented in (Table I) indicated that the significant lower LC50 values of O. surinamensis larvae were observed when both R. stricta (160.77, 137.26, and 110.41 ppm) and S. persica (200.64, 168.68, and 126.08 ppm) were applied after 2, 4, and 6 days, respectively, in comparison with other used plant extracts. The values of LC50 for other plant extracts of R. chalepensis, M. oleifera, and H. bacciferum after 6 days following exposure were 130.91, 206.83, and 285.11 ppm, respectively. Comparing the LC50 values, it was observed that the methanolic extracts of R. stricta,S. persica, and R. chalepensis showed better performance than other plant extracts.
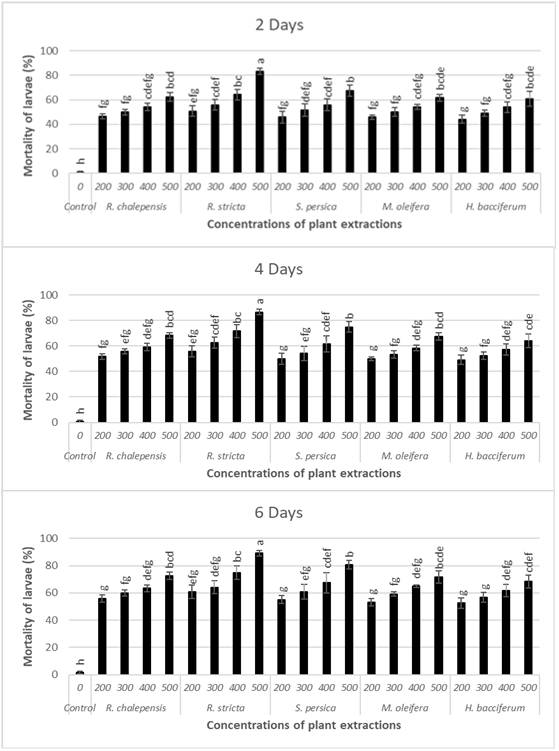
Fig. 1.
Mortality of Oryzaephilus surinamensis larvae by five plant extracts after three observed times (2,4 and 6 days). Means and S.E. of (%) mortalities. Different letter denotes significant differences between treatments (t ˂ 0.05), according to the LSD test.
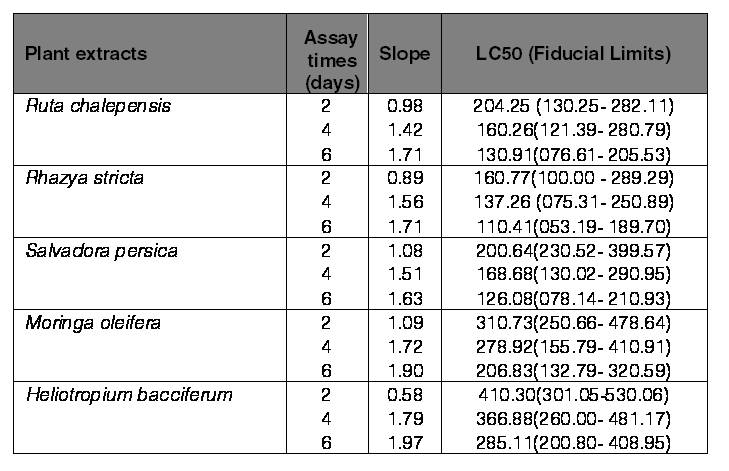
Table I.
LC50 values (ppm) and 95% confidence limits for Oryzaephilus surnamensis larvae reared in media containing methanolic extracts from five plant materials.
Likewise, the used plants with tested concentrations showed higher significant differences on the mortality of the adult stage of O. surinamensis after 2 days (F20.63=13.15, P˂0.001), 4 days (F20.63=14.70, P˂0.001), and 6 days (F20.63=16.46, P˂0.001). The used plant extracts were found to be more effective against the adult stage of O. surinamensis in comparison with the larval stage in which the extract of R. stricta caused 93.33% adult mortality at the concentration of 500 ppm after 6 days. Meanwhile, the extracts of S. persica, R. chalepensis,M. oleifera, and H. bacciferum caused 82.50, 74.17, 73.33, and 69.17% mortality of O. surinamensis adults, respectively, at the same concentration and observed time (Fig. 2). Consequently, the data revealed that concentrations of R. stricta, S. persica, and R. chalepensis were the most effective plant extracts on controlling the adult stage of O. surinamensis where the mortality in adult beetle ranged from 62.5 to 93.3%, 57.5 to 82.5%, and 56.7 to 74.2% after 6 days from applying the treatments, respectively. The used plant extracts showed time- and concentration-dependent effects against the O. surinamensis adults in which their mortality percentage increased after 6 days of exposure for all the plant extracts. Throughout 6 days of exposure, adulticides of all the plant extracts were approximately improved showing higher mortality of the O. surinamensis adults with increasing exposure time and concentrations. Mortality of control was less than 5% along the exposure periods. The toxicity analyses with LC50s and 95 % confidence limits for each plant extract against O. surinamensis adults are shown in (Table II). The lowest LC50s were for R. stricta,S. persica, and R. chalepensis after 6 days from the treatment with the values being 108.47, 121.54, and 128.14 ppm, respectively. The respective values of LC50s of the other plant extracts after the same period of exposure were 201.47 and 292.49 ppm for M. oleifera and H. bacciferum, respectively. The obtained results demonstrated that the plant extracts of R. stricta, S. persica, and R. chalepensis were generally more toxic than other plant extracts and possessed lower LC50s. This finding is in agreement with the results of Qureshi et al. (2018), who found that the extract of S. persica was very effective against T. confusum in which the larval growth of this pest was significantly inhibited when extract was incorporated to larval diet. Moreover, Al-Qahtani et al. (2012) reported the lethal effect of three plants named ginger (Zingiber officinale), cardamom (Elettaria cardamomum), and fennel (Foeniculum vulgare) against O. surinamensis, describing the most dangerous pest recorded in Saudi Arabia. In addition, our results supported the results of Alvi et al. (2018), who found that R. stricta leaf and seed extracts caused a significant mortality and repellency against R. dominica and T. granarium. In their study, mortality and repellency were increased as the concentrations and extract exposure time increased. More recently, Asiry & Zaitoun (2020) reported that the extract from R. stricta was very effective against T. granarium as an insecticide. The toxicity and repellency properties of R. stricta against larval and adult stages of O. surinamensis could be attributed to the presence of different glycosides, triterpenes, alkaloids, and volatile basis (Ali et al., 2000; Marwat et al., 2012).
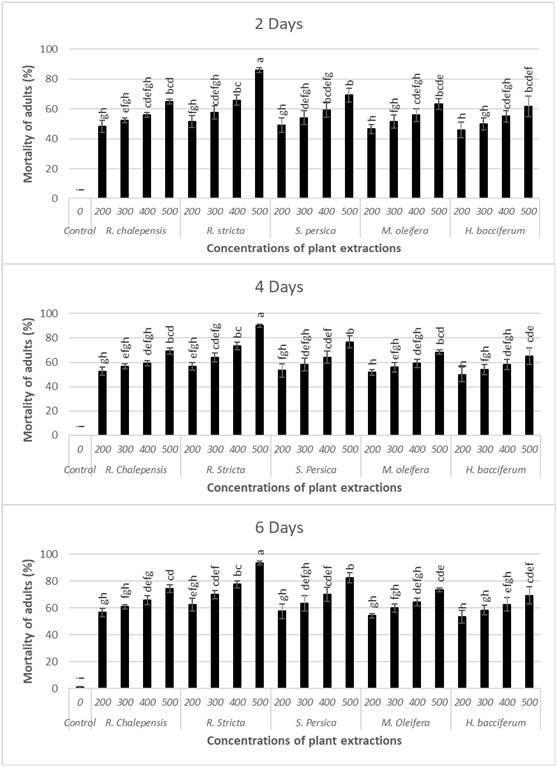
Fig. 2.
Mortality of Oryzaephilus surinamensis adults by five plant extracts after three observed times (2,4 and 6 days). Data are Means±SE. Different letter denotes significant differences between treatments (p ˂ 0.05), according to the LSD test.
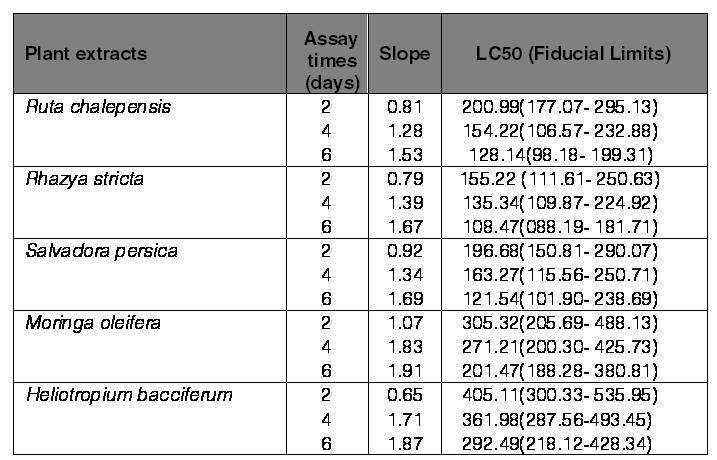
Table II.
LC50 values(ppm) and 95% confidence limits for Oryzaephilus surnamensis adults reared in media containing methanolic extracts from five plant materials.
The study of Ahmad et al. (1983) regarding the alkaloidal constituents of R. stricta leaves had resulted in the isolation of rhazimal (16-formylstrictamine), rhazimol (deacetylakuammiline), and rhazinol (a hydroxymethyl analogue of strictamine). In addition, two more alkaloids stricticine and strictine had also been isolated. Jeon et al. (2013) indicated that the insecticidal constituent of R. chalepensis leaves was isolated by chromatographic techniques and identified as quinoline-4-carbaldehyde (C10H.NO) which was very effective against Sitophilus oryzae L. (Coleoptera: Curculionidae). The flavonoids presented in plant extracts possess a catecholic B-ring that seems to be responsible for the toxicity to insects (Onyilagha et al., 2004). Moreover, Jabilou et al. (2006) found insecticidal activities from four medicinal plant extracts, including Peganum harmala (Sapindales: Nitrariaceae), Ajuga iva (Lamiales: Labiatae), Aristolochia baetica (Piperales: Aristolochiaceae), and Raphanus raphanistrum (Brassicales: Brassicaceae) against O. surinamensis. Furthermore, Haj et al. (2015) demonstrated that the three doses utilized (5, 10, and 15%) of three plant extracts of Calotropis procera (Gentianales: Apocynaceae), Solenostemma argel (Gentianales: Apocynaceae), and Datura stramonium (Solanales: Solanaceae) revealed some potential insecticidal effects to the adult of O. surinamensis and a high repellency action against this pest.
Repellency Rate of Plant Extracts against the Adults of O. surinamensis
The one-way ANOVA showed a higher significant difference between the tested plant extracts on the repellency rate (F20.41=40.65, P=0.001) against the adults of O. surinamensis. Statistical analysis revealed that R. stricta exhibited the most extraordinary repellent action against the adult of O. surinamensis (Fig. 3). At a concentration of 500 ppm, the repellent percentage of R. stricta was 91.7%, followed by S. persica (83.3%) and R. chalepensis (78.3%) using the same concentration (Fig. 3).
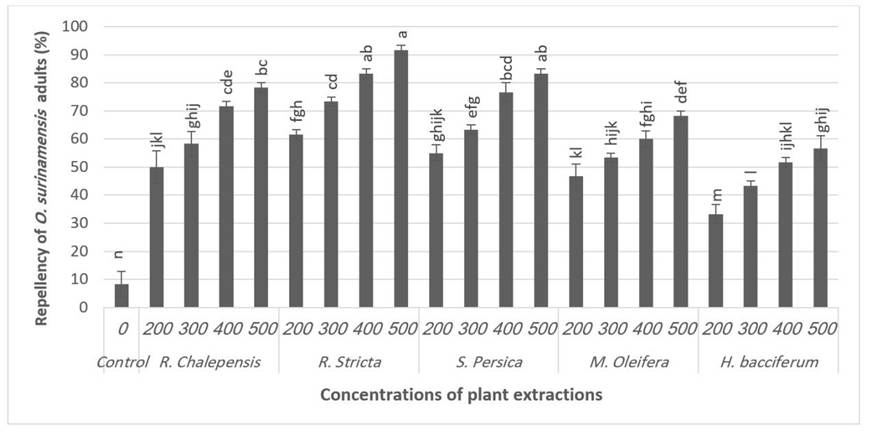
Fig. 3.
Repellence of Oryzaephilus surinamensis adults by five plant extracts after three observed times (2,4 and 6 days). Data are Means±S.E. Different letter denotes significant differences between treatments (P ˂ 0.05), according to the LSD test.
The present results supported the finding of Kundu et al. (2007) and Elumalai et al. (2015), whose repellent activities of some plant extracts, Polygonum hydropiper L. (Caryophyllales: Polygonaceae) and Rivina humilis Linn (Caryophyllales: Phytolaccaceae) against T. castaneum, reported that the rate of repellency increased proportionately with increasing concentrations of the plant extracts. Furthermore, Abdel-Sattar et al. (2009) suggested that the leaf and fruit essential oils of Schinus molle L. have an insecticidal and repellent effect on T. castaneum and T. granarium. The outcomes attained from this study revealed the importance of the toxicity besides the repellent influence of the extracted plant materials, particularly R. stricta and S. persica, in regulating the stored-product insects, particularly O. surinamensis. Aliv et al. (2018) declared that R. stricta worked as a repellent material against R. domina and T. granarium. Elmhalli et al. (2019) reported that S. persica has a repellent effect against nymphs of I. ricinus. Kachhwaha et al. (2015) demonstrated that repellent behavior could be utilized as an essential measure as it does not kill insects but protects the grain from their infestation.
CONCLUSION
The discovery of natural and inexpensive insecticides from local resources that could be applied to prevent stored products from insect infestation is an essential goal for food security and safety. The plant extracts application is commonly expected not to leave harmful residues in the surroundings as they occur naturally amongst the native flora. The results of this study demonstrated that methanolic extracts of R. stricta, S. persica, and R. chalepensis plants possess toxic and repellent activity that could serve adequately as a potential pest control strategy for the stored-grain protection against the saw-toothed grain beetle O. surinamensis.
In comparison with conventional insecticides, plants' extracts are confirmed to be relatively harmless. They also tend to degrade quickly in the surroundings (Oparaeke et al., 2005). This study put forward that there might be diverse compounds in plant extracts possessing various bioactivities. Further studies are required to isolate, identify, and assess the bioactivity of insecticide compounds present in studied plants against stored products' pests.
Acknowledgments
This project was funded by the Deanship of Scientific Research (DSR) at King Abdulaziz University, Jeddah, Saudi Arabia, under Grant G-1364-155-1440. The authors, therefore, acknowledge with thanks DSR for technical and financial support.
REFERENCES
Abbott, W.S. (1925) A method of computing the effectiveness of an insecticide. Journal of Economic Entomology,18, 265-267.
Abdel-Sattaral, E., Zaitoun A. A., Farag M. A., El-Gayed S. H., & Harraz F. M. (2009) Chemical composition, insecticidal and insect repellent activity of Schinus molle L. leaf and fruit essential oils against Trogoderma granarium and Tribolium castanium. Natural Product Research, ., 1-10.
Ahmad, Y., Fatima K, LeQuensne P. W., & Rahman A. (1983) Further alkaloidal constituents of leaves of Rhazya stricta. Phytochemistry, 22, 1017-1019
Akinkurolere, R.O.; Adedire, C.O., & Adeyemi, O.O. (2006) Laboratory evaluation of the toxic properties of forest Anchomanes, Anchomanes difformis against pulse beetle, Callosobruchus maculatus (Coleoptera: Bruchidae). Insect Science, 13, 25-29.
Akinneye J.O. & Ogungbite O.C. (2013) Insecticidal activities of some medicinal plants against Sitophilus zeamais (Motschulsky) (Coleoptera: Curculionidae) on stored maize, Archives of Phytopathology and Plant Protection, 46(10), 1206 - 1213.
Ali, B.H., Qarawi, A.A.A., Bashir, A.K., & Tanira, M.O. (2000). Phytochemistry, pharmacology, and toxicity of Rhazya stricta Decne: A Review. Phytotherapy Research, 14, 229–234.
Al-Qahtani, A.M., Al-Dafar Z.M., & Rady, M.H. (2012) Insecticidal and biochemical effect of some dried plants against Oryzaephilus surinamensis (Coleoptera-Silvanidae). The Journal of Basic & Applied Zoology, 65(1), 88-93
Alvi, A.M., Naeem N., Bashir, M.A., Rehmani, M.A., Ullah, Z. Saeed, O., & Latif, A. (2018) Efficacy of Rhazya stricta leaf and seed extracts against Rhyzopertha Dominica and Trogoderma granarium, Kuwait Journal of Science ,45 (3), 64-71.
Asiry, K.A., & Zaitoun, A.A (2020) Evaluation of three plant extracts' toxicity against the Khapra beetle Trogoderma granarium Everts (Coleoptera: Dermestidae) under laboratory conditions. Revista de la Sociedad Entomológica Argentina, 79(1), 5-12.
Eldidamony, A., Moustafa, G.G., Mead, H., & Elshafeiy, S. (2020) New Insight on Heliotropium curassavicum L. Extract as a Rodenticide. Annals of Biology, 36 (1), 102- 111.
Elmhalli, F., Garboui, S.S., Borg-Karlson, A.K., Mozuraitis, R., Baldauf, S.L. & Grandi, G. (2019) The repellency and toxicity effects of essential oils from the Libyan plants Salvadora persica and Rosmarinus officinalis against nymphs of Ixodes ricinus. Experimental and Applied Acarology, 77, 585-599.
Elumalai, A., Krishnappa, K., Kalaichelvi, N., & Elumalai, K. (2015) Insecticidal, ovicidal and repellent activities of different solvent extracts of Rivina humilis Linn (Phytolaccaceae) against the selected stored grain pest, Tribolium castaneum Herbs. (Coleoptera: Tenebrionidae). International Journal of Advanced Research in Biological Sciences, .(10), 161–169.
Finney, D. J. (1971) Probit analysis 3rd edition. Cambridge University Press. Cambridge, UK.
Haj, R.E.; Yousif, A., & Taha, A.K. (2016) Evaluation of Some Plant Extracts against Adults of the Saw-toothed Grain Beetle, Oryzaephilus surinamensis (Linn.) (Coleoptera: Silvanidae). Universal journal of agricultural research, .(5), 170-174.
Huda, A.N., & Noor A. M. (2019) Food Preference of Oryzaephilus Surinamensis (Coleoptera: Silvanidae) to Different Types of Plant Products. Malaysian Journal of Halal Research Journal, .(2), 53-57.
IBM Corporation (2011) PASW Statistics, 20th ed. IBM, Chicago, IL, USA. Available from: https://www.ibm.com/software/analytics/spss/
Ileke K.D. & Oni M.O. (2011) Toxicity of some plant powders to maize weevil, Sitophilus zeamais (Motschulsky) (Coleoptera: Curculionidae) on stored wheat grains (Triticum aestivum). African Journal of Agricultural Research, ., 3043-3048.
Jabilou, R., Ennabili A., & Sayah F. (2006) Insecticidal activity of four medicinal plant extracts against Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae). African Journal of Biotechnology, .(10), 936-940.
Jeon, J.H., Kim, M.G. & Lee, H.S. (2013) Insecticidal activities of Ruta chalepensis leaves isolated constituent and structure-relationships of its analogues against Sitophilus oryzae. Journal of the Korean Society for Applied Biological Chemistry, 56, 591-596.
Jilani, G., Saxena R. C., & Rueda B. P. (1988) Repellent and growth- inhibiting effect of turmeric oil, sweet flag oil, neem oil, and margosa oil on red flour beetle (Coleoptera: Tenebrionidae). Journal of Economic Entomology, 81, 1226-1230.
Kachhwaha, N., Meena G. D. & Meena, S. (2015) Plant extracts control Oryzaephilus surinamensis by showing repellency behaviour. European Journal of Experimental Biology, .(5), 98-101.
Khemais A., Acheuk, F., Miladi, M., Boughattas, I. Omri, G. (2018). Phytochemistry, biochemical and insecticidal activities of Ruta chalepensis essential oils on Tribolium confusum. Agriculture & Forestry, 64 (3), 31-45.
Kundu, B. R., Ara R., Begum M. M., & Sarker Z. I. (2007) Effect of Bishkatali, Polygonum hydropiper L. plant extracts against the red flour beetle, Tribolium castaneum Herbst. University Journal of Zoology, Rajshahi University, 26, 93-97.
Mahmoud, A. K, Bedawi, S. M., & Satti, A. A. (2015) Efficacy of some Botanical Extracts in the control of Khapra Beetle (Trogoderma granarium). Journal of science, . (4), 213- 217.
Martins C.H.Z, Freire M.G.M, Parra J.R.P & Macedo M.LR. (2012) Physiological and biochemical effects of an aqueous extract of Koelreuteria paniculata (Laxmi.) seeds on Anticarsia gemmatalis (Huebner) (Lepidoptera: Noctuidae). SOAJ Entomology Studies. ., 81 – 93.
Marwat, S.K., Rehman, F.U., Usman, K., Shah, S.S., Anwar, N., & Ullah, I. (2012). A review of phytochemistry, bioactivities, and ethnomedicinal uses of Rhazya stricta Decne (Apocynaceae). African Journal of Microbiology Research, .(8), 1629-1641.
Ojiako, F. O, Agu, C. M., & Ahuchaogu, C. E. (2013) Potentiality of Moringa oleifera Lam. Extracts in the Control of some Field – Store Insect Pests of Cowpea. International Journal of Plant Production, ., 3537-3542
Onyilagha, J. C., Lazorko, J., Gruber, M.Y., Soroka J. J. & Rrlandson, M.A. (2004) Effect of flavonoids on feeding performance and development of the crucifer pest. Journal of Chemical Ecology,30, 109-124.
Oparaeke, A. M., Dike, M.C., & Amatobi, C.L. (2005). Evaluation of botanical mixtures for insect pests management on cowpea plants. Journal of Agriculture and Rural Development in the Tropics and Subtropics,106(1), 41- 48.
Pimentel, M.; Faroni, L.R.A.; Batista, M.D., & da Silva, F.H. (2008) Resistance of stored-product insects to phosphine. Pesquisa Agropecuária Brasileira , 43(12), 1671- 1676.
Qureshi, N., Khan, M.F., Ali, U., & Javed, T. (2018). Pest control potential of halophyte medicinal plant Salvadora persica L. against wheat pest Tribolium confusum Jacquelin. International Journal of Biology and Biotechnology, 15 (4), 735-741.
Sutherland J.P., Baharally V. & Permaul D. (2002) Use of the botanical insecticide, neem to control the small rice stinkbug Oebalus poecilus (Dallas, 1951) (Hemiptera: Pentatomidae) in Guyana. Entomoltropica, 17, 97 - 101.
Talukder, F.A., & Howse P. E. (1994). Laboratory evaluation of toxic and repellent properties of pithraj, A. polystachya Wall. against Sitophilus oryzae L. International journal of pest management,40(3), 274-279.
Tapandjou I.A., Alder A., Fontem H. & Fontem D.A. (2002) Efficacy of powder and essential oil from Chenopodium ambrosioides leaves post-harvest grain protectants against six stored products beetles. Journal of Stored Products Research,38,395-402.
Zibaee A. (2011) Botanical insecticides and their effects on insect biochemistry and immunity. Pesticides in the world. In: Stoytcheva M. (Ed), Pests Control and Pesticides Exposure and Toxicity Assessment, pp.55-68. In Tech, Croatia.