Artículos científicos
Seasonal occurrence and biological parameters of Aedes albopictus (Skuse) (Diptera: Culicidae) as management tactics in Faisalabad, Punjab, Pakistan
Ocurrencia estacional y parámetros biológicos de Aedes albopictus (Skuse) (Diptera: Culicidae) como tácticas de manejo en Faisalabad, Punjab, Pakistán
Seasonal occurrence and biological parameters of Aedes albopictus (Skuse) (Diptera: Culicidae) as management tactics in Faisalabad, Punjab, Pakistan
Revista de la Sociedad Entomológica Argentina, vol. 81, núm. 4, 2022
Sociedad Entomológica Argentina
Recepción: 10 Octubre 2022
Aprobación: 21 Noviembre 2022
Abstract: Understanding the seasonal prevalence and biological parameters of mosquito Aedes albopictus (Skuse) are indispensable for population estimates, efficient vector control and dengue prevention tactics. The sampling of Ae. albopictus was completed through the collection of eggs, larvae, pupae and adults throughout the study seasons by use of nets and ovitraps at least once a week. Occurrence of Ae. albopictus was distinguished heterogeneously and populations were observed maximum in October post-monsoon season. The populations increased progressively from January-May and sharply deteriorated in June-July and September-November months revealing the highest breeding. However, the lowest density was observed during December-February. Growth of Ae. albopictus evaluated under standardized laboratory conditions with the provision of Balb/C mice as blood meal to adult mosquitoes, exhibited eggs hatching, and first, second, third and fourth instar larvae development in 3, 1.3, 1.2, 1.7 and 2.8 days, respectively. Nevertheless, pupation development, adult emergence, total life duration from egg hatching to adult emergence, male and female life spans persisted for 7, 3, 20, 25 and 30 days, respectively. Aedes albopictus accounted for 1% density of other mosquito genera collected (Culex and Anopheles), while, Aedes aegypti (L) was 3% abundant and indicated that both Aedes species had coexistence.
Keywords: Aedes albopictus, Biology, Dengue Vector Monitoring, Mosquitoes, Population Dynamics.
Resumen: Comprender la prevalencia estacional y los parámetros biológicos del mosquito Aedes albopictus (Skuse) es indispensable para las estimaciones de población, el control vectorial eficiente y la implemntación de tácticas de prevención del dengue. El muestreo de Ae. albopictus se completó mediante la colecta de huevos, larvas, pupas y adultos a lo largo de las temporadas de estudio mediante el uso de redes y ovitrampas al menos una vez por semana. La ocurrencia de Ae. albopictus se distinguió heterogéneamente, y las poblaciones se observaron al máximo en octubre después de la temporada del monzón. Las poblaciones aumentaron progresivamente de enero a mayo y se deterioraron bruscamente en los meses de junio a julio y de septiembre a noviembre, revelando la mayor reproducción. Sin embargo, la densidad más baja se observó durante diciembre-febrero. Crecimiento de Ae. albopictus evaluados bajo condiciones estandarizadas de laboratorio con la provisión de ratones Balb/C como harina de sangre a mosquitos adultos, exhibieron eclosión de huevos, desarrollo de larvas de primer, segundo, tercer y cuarto estadio en 3, 1.3, 1.2, 1.7 y 2.8 días, respectivamente. Sin embargo, el desarrollo de la pupa, la emergencia del adulto, la duración total de la vida desde la eclosión del huevo hasta la emergencia del adulto, la duración de la vida del macho y la hembra persistieron durante 7, 3, 20, 25 y 30 días, respectivamente. Aedes albopictus representó el 1% de la densidad de otros géneros de mosquitos recolectados (Culex y Anopheles), mientras que Aedes aegypti (L) fue abundante en un 3% e indicó que ambas especies de Aedes coexisten.
Palabras clave: Aedes albopictus, Biología, Dinámica de poblaciones, Monitoreo de vectores del dengue, Mosquitos.
INTRODUCTION
Among the entire diversified insect fauna, mosquitoes are of countless significance in terms of human health as they spread various diseases to millions of people each year. Currently, mosquito-transmitted diseases are a symbol of great health troubles and no fraction of humankind's dwellings is protected from this danger (Sarwar, 2014a; 2015a; 2016a). Public health authorities from recently mosquito-infested countries have shown much concern about Aedes albopictus (Skuse), which is the vector of at least 22 arboviruses that could lead to serious occurrences of diseases, such as the Eastern equine encephalitis and the dengue fever (Mitchell et al., 1992; Gubler, 2002). Aedes albopictus is well established in urban environments being tropical forests its original habitat. The ability of Ae. albopictus to settle in used tires is the basis of its recent prompt existence in this new habitat (Reiter, 1998). This species can lay eggs on the interior surface of watery vessels in urban and rural areas, as well as close boundaries of forest localities in vegetated areas within and around the homes. The larvae and pupae (immature forms) can be found in both artificial and natural breeding sites, such as buckets, tin cans, drums, tires, standing water, hollow bamboo stumps, rock holes and tree holes. Additionally, Ae. albopictus can instigate in temperate zones with cold winter (Russell et al., 2005; Sarwar, 2014b; 2015b; 2016b). It is a daytime feeder, with feeding peaks in the early morning and late afternoon. It is an opportunistic and belligerent species with an extensive host variety of wild animal fauna including man (Hawley, 1988).
Aedes albopictus had been validated in all four dengue serotypes of transovarial transmissions with variability in serotype and strain diversification of virus and normally with strains type 1 highest to lowest in type 3 (Rosen et al., 1983). The infection is transmitted to female Ae. albopictus through copula, whereas infected Ae. albopictusfemales do not transmit their infection sexually to males. However, male mosquitoes transmit the virus vertically to their F1 generation albeit not directly to ova but, rather, through prior replication in the female genital tract (Rosen, 1987). The continuous increase of Ae. albopictus populations worldwide during the last thirty years has amplified human concern regarding public health and highlighted the need to improve our understanding of dengue surveillance and management (Lambrechts et al., 2010).
Given the need to protect human health, surveillance of mosquitoes in all their life stages, but mainly adults and their relevant immature stages, permits the evaluation of probable diffusion hazards and appropriate accomplishment of suitable mosquito control measures. In spite of this, a literature search revealed that few studies address this issue and a lot of entomologists lack the taxonomic skills needed to precisely classify vectors beyond the genus level; particularly in certain regions. The community health workers who are regularly assigned to perform entomological surveillance, in general, have even fewer skills in species identification. Therefore, the first step in mosquito control is its surveillance. By studying its biology and population fluctuation, one can get to know the population structure of this vector enabling recommendations to control the transmission of dengue and other arboviruses (Sarwar, 2014c; 2015c). To this end, the present work has the objective of collecting information on the population fluctuation and biology of Ae. albopictus in Faisalabad, located at the Punjab province of Pakistan.
MATERIALS AND METHODS
Sampling area for mosquito collection
The surveys were accomplished from January to December (2019-2020) aiming to determine the species composition of the mosquito community and the seasonal population fluctuations of Ae. albopictus at the National Institute for Agriculture and Biotechnology (NIBGE) and the Department of Zoology, Government College University Faisalabad (GCUF) new campus. Both institutes are situated at Jhang Road, Faisalabad, which is the metropolitan city of Punjab, Pakistan. Faisalabad is the third-largest metropolis in Pakistan and is located at an altitude of 184 meters above sea level (Fig. 1). The climate of Faisalabad is characterized by high temperatures during eight months of the year and high humidity and rainfall during summer. No insecticide sprays were applied in the area during the sampling period.
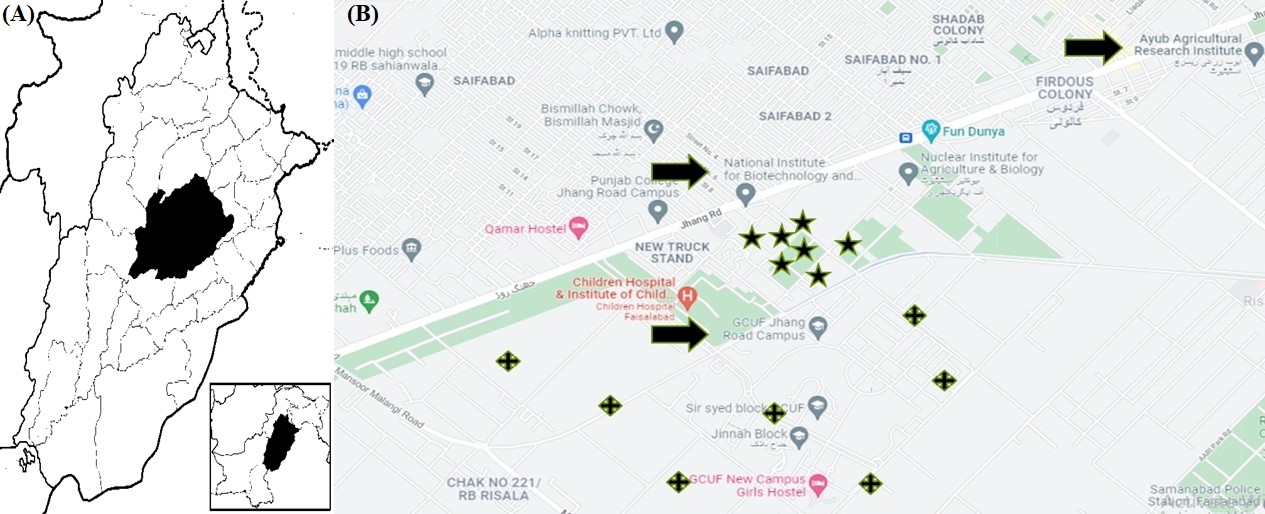
Figure 1.
A) Map of Faisalabad, Punjab, Pakistan, B) Map showing the sampling localities of secondary dengue vector mosquito Aedes albopictus (Diptera: Culicidae) from the institutes (NIBGE and GCUF) and metrological data collection institute (AARI) in Faisalabad, Punjab, Pakistan (Source: Google satellite images).
Collection of mosquito samples
Sampling for Ae. albopictus was performed at least once a week and accomplished by counts of eggs, larvae and adults. All stages (eggs to adults) of vector were collected. The samples were collected from small water reservoirs, plant pots, water tanks, water trenches, shady places of shrubs, tree holes, ponds, rainy water drainage areas, weeds and grasses of the gardens. Seven sampling sites in each of the institutes were chosen. Samples were collected with a fine brush, a siphon, an aspirator or orhard nets according to the stage and then transferred into plastic containers. The collection of adults was carried out with an aspirator from the bushes inside and outside of each mentioned collection site. Larvae and pupae were collected from water-holding containers and reachable water sources by using a pipette or funnel depending on the type of container. Eggs were collected from specially designed ovitraps (plastic vase with a wooden ovipositional paddle and hay infusion) installed across vegetation coverage in each institute. A total of seven ovitraps were placed in each institution. All of the specimens collected were properly placed into plastic containers covered with cloth and transported to the Entomological Laboratory.
Processing and identification of the samples
Once in the laboratory, the specimens were transferred into 500 ml plastic containers, covered with mosquito nets and allowed for further development. Eggs were observed each day to determine whether larvae hatched. For dry samples with eggs, water was added to the plastic containers to allow embryo development. After the larvae hatched from the eggs, they were allowed to develop (see below). Upon reaching the 4th instar, they were placed onto glass slides to be examined under a compound binocular microscope for further identification (Cheong, 1986; Rueda, 2004). Larvae and pupae were identified up to genus level and only those belonging to the genus Aedes were transferred to separate perspex cages until adult’s emergence. Subsequently, they were identified up to the species level.
Laboratory rearing procedure
The mosquitoes were upraised and observed for their progression through different life stages (eggs, larval, pupal and adult periods) in insect rooms under standardized conditions (24 ± 2 .C temperature and 72 ± 4% relative humidity associated with L:D 12:12 photoperiod). Cotton balls saturated with 10% sugar solution were placed on the bottom of the rearing cages chambers after sequencing the excess of water. A laboratory-bred mouse (Balb/C) was offered every day for containment of the blood meal. Larval diet for culturing of larvae was composed of rice polish (55%), maize gluten (30%), sunflower meal (10%) and fishmeal (5%) in the form of small pellets. Durations of egg hatching, 1st to 4th larval instars and pupae were recorded. Male and female longevities were also recorded. The datasets were observed in 9 replicates. Practical procedures were followed from Aida et al. (2011) and Cui et al. (2021) for study.
Environmental data
Climatic datasets containing relative humidity, rainfall and temperature records were obtained from Physiology Section, the meteorological observatory wing located at Ayub Agricultural Research Institute (AARI), Faisalabad, Punjab, Pakistan, to study the weather impact on mosquito population. The data were aggregated to provide monthly estimates for each variable.
Statistical analysis
Datasets of all the treatments were evaluated statistically through the software Statistix 8.1(2003). The developmental time periods of life parameters (from embryo development to adult emergence) of A. albopictus were determined using the ANOVA (analysis of variance), through the LSD (least significance difference) test at the significant level of p ≤ 0.05. Further, the mosquito abundance related to environmental factors (temperature, relative humidity and rainfall) was determined by means of a multiple linear regression and Pearson correlation coefficient analysis in order to measure the overall effect of all weather variables on mosquito developmental stages and abundance (Schober et al., 2018; Drakou et al., 2020).
RESULTS
Species composition
The predominant mosquito genera captured in the whole period were Culex and Anopheles 96% with Aedes representing only 4% of the individuals. Among these 4% individuals, Ae. albopictus constituted 1% and Ae. aegypti 3% of the total sample.
Life cycle of Aedes albopictus
The laboratory-bred female of Ae. albopictus produced eggs 3 days after feeding on the blood of adult Balb/C mouse. Under adequate humidity conditions, the eggs hatched and evolved into the larval stage in about 3 days. The larval developmental progress for the first, second, third and fourth larval instars lasted for 1.3, 1.2, 1.7 and 2.8 days, respectively (Table I). The hatching of eggs per female, pupation, adult emergence, duration from egg to adult emergence, male and female life span persisted for 3, 7, 3, 20, 25 and 30 days, respectively (Table I) in standardized laboratory conditions.
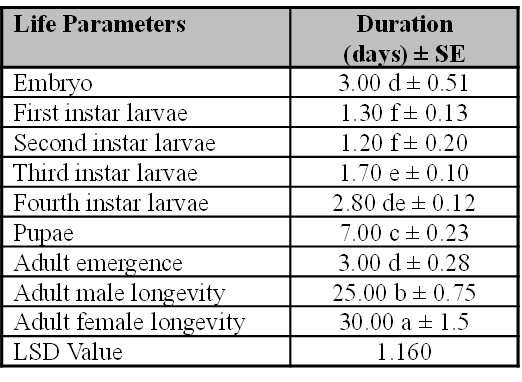
Table I.
The total developmental time-periods of life parameters (egg hatching to adult) of Aedes albopictus.
Mean values represented inside a column followed by similar letters are not statically different using the LSD test (P= 0.05), SE (Standard Error), number of eggs, n (15).
Seasonal prevalence of Aedes albopictus
Weekly data collection of Ae. albopictus mosquitoes from the study area are presented in Figure 2. The results showed that in both years of vector monitoring, the occurrence of Ae. albopictus adults varied throughout the seasons and the population attained maximum levels during October. Vector proliferation enhanced gradually from January to May with a peak population observed in March. By June and July, it declined sharply and started increasing until reaching its second peak in October. The lowest vector abundance was recorded from December to February (Fig. 2). Egg seasonal fluctuation (Fig. 2) also exhibited two peaks; the first was during September to November and the second, during March and April. The lowest abundance was recorded in December, January and February, while May, June, July and August presented intermediate values (Fig. 2).
As for larvae and pupae, their two peaks were also observed. The first one was during September to November; while the second, found in March and April. The lowest abundance was observed in December, January and February (Fig. 2).
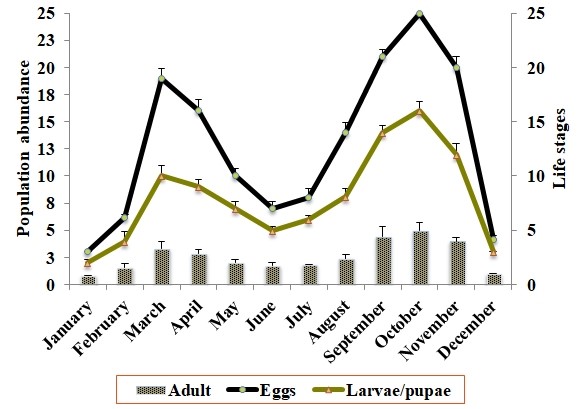
Figure 2.
Mean seasonal abundance of the different life stages of secondary dengue vector mosquito Aedes albopictus (Diptera: Culicidae) during the years (2019-2020).
(Eggs) the abundance of eggs per trap per week, (Larvae/ pupae), the abundance of larvae/ pupae per site per week, (Adult) adult population per site per week.
Weather conditions during the study period are presented in Figure 3. Average minimum temperature ranged between 4.1oC and 27.9 oC; while maximum temperature ranged between 19.5 .C and 41.6 oC). Minimum relative humidity ranged from 14.0 and 47.0% and maximum relative humidity ranged from 51.0 to 100.0%. Rainfall varied across the years and the mean heavier precipitations occurred in September (147.9 mm) followed by July (63.0 mm) and after that subsequent peak (26.0 mm) occurred in April (Fig. 3).
When the maximum temperature of the day exceeded 28 ºC or was lower than 16 ºC, a decrease in the mosquito abundance was observed. Based on the Pearson correlation coefficient and multiple linear regression analysis, our results showed a significant correlation of mosquito abundance and mean monthly temperature, humidity and rainfall (Table II). The overall mean monthly maximum temperature exhibited weak negative correlation with eggs (- 0.124), a moderate negative correlation with larvae/ pupae (- 0.570) and adults (- 0.641). Further, the overall mean monthly minimum temperature presented a weak negative correlation with eggs (- 0.040), larvae/ pupae (- 0.372) and a moderate negative correlation (- 0.424) with adults. Monthly average maximum relative humidity showed a moderate positive correlation with eggs (0.544), larvae/ pupae (0.620) and adults (0.662). Whereas, monthly average minimum relative humidity presented weak correlation with eggs (0.202), larvae/ pupae (0.312) and adults (0.394). A significant strong positive correlation was recorded between rainfall values with eggs (0.841), larvae/ pupae (0.870) and adult populations (0.780).
Extremely high rainfall levels in September in association with high temperature led to the large population abundance of Ae. albopictus.
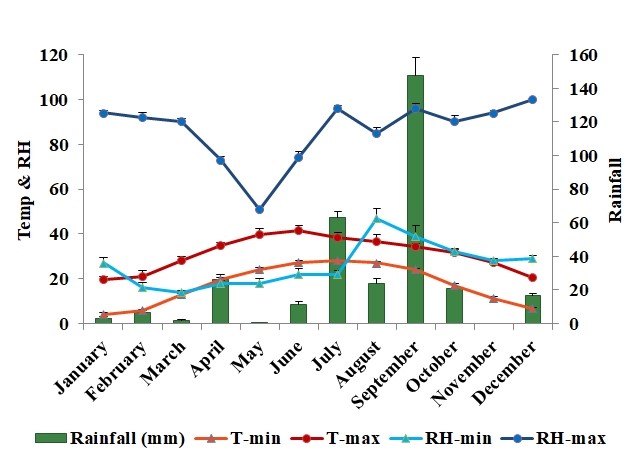
Figure 3.
Mean meteorological observations noted at the Observatory of Plant Physiology Section, Ayub Agricultural Research Institute, (AARI), Faisalabad, during the years (2019-2020).
Temperature (°C), Relative Humidity (%), Rainfall (millimeter).
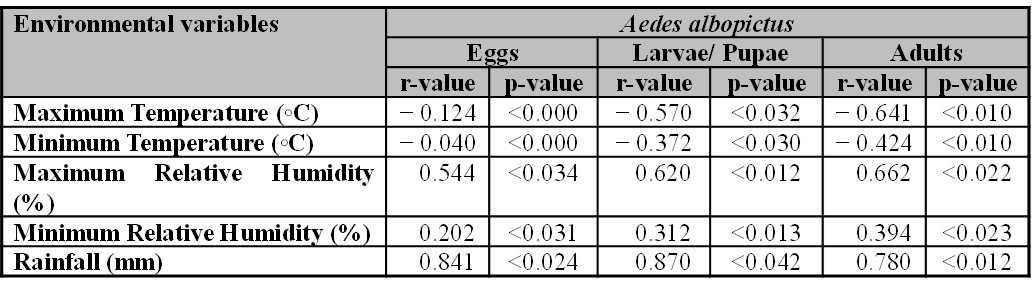
Table II.
Mosquito abundance and monthly average environmental variable (temperature, relative humidity, and rainfall) presented through Pearson coefficient correlation (r-values) and multiple linear regression (p-values) from January 2019 to December 2020.
r = 1.0-0.9 (very strong correlation), r = 0.89-0.7 (strong correlation), r = 0.69-0.4 (moderate correlation), r = 0.39-0.1 (weak correlation), p < 0.05 (significant).
DISCUSSION
In this study we evaluated the occurrence of Ae. albopictus in the area of Faisalabad aiming to determine the species composition of the mosquito community, surveillance mechanisms, prevalence and the seasonal population fluctuation. We found that different environmental factors had marked influence on Ae. albopictus developmental parameters such as hatching of eggs, larval and pupal growth, emergence rates, adult mosquito survivorship and reproduction capacity. The rainfall, humidity and temperature were significant impelling overall Ae. albopictus abundance in the studied area. All these results contribute to our understanding of Ae. albopictus and provide valuable information regarding the considerable variations of seasonal abundance throughout the study period.
The occurrence of Ae. albopictus in the experimental localities represented 1% of the total mosquito collection; while, Ae. aegypti represented 3% and both species coexisted. There are various possible reasons for the lower abundance of Ae. albopictus compared to Ae. aegypti: (1) inter-specific mating offspring sterility, (2) increased fitness of Ae. albopictus for parasites fetched in the locality and (3) Ae. aegypti dominance in larval resource competition (Costanzo et al., 2005). Aedes albopictus abundance was the highest at cool sites and significantly declined with the increasing mean temperature and length of the dry season. Whereas, Ae. aegypti was lowest at cool sites and a significant enhanced population increase was observed with increasing mean temperature and length of the dry period. Moreover, in the active season, egg-laying started earlier at low altitude and ended earlier within the highest altitudinal range (Romiti et al., 2022). The coexistence of these species is likely because warm, dry climates favor Ae. aegypti and lessen effects of competition from Ae. albopictus via differences in eggs mortality (Juliano et al., 2002). Honorio et al. (2009) already point that both species, Ae. aegypti andAe. albopictus peaked during the wet season, therefore distribution configuration and adults flight range may indulgence to the co-existence of larvae of these species in urban areas or urban-forest transition zone. Further, the co-occurrence of both species corresponded to the seasonality of Ae. aegypti with Ae. albopictus, where Ae. aegypti was found in tires in correspondence to its better adaptation to urban areas, while Ae. albopictus was common in forest areas and suffered from the effects of interspecific competition.
Because the biology of Ae. albopictus differs from season to season and region to region (Giatropoulos et al., 2012; Delatte et al., 2009; Honorio et al., 2009; Cheong, 1986), current knowledge about its biology is essential for adopting appropriate control measures in a particular season and region. There are some effective ways to prevent mosquito breeding at home and outside during the rainy season in stagnant water. These preventive measures include removing stagnant water from flowerpots, plates, plant saucers and discarding unwanted containers. Here we found that the Ae. albopictus was frequent in vegetation areas; therefore, its population depression strategies should be mainly focused on such locations. Further, additional preventive measures are advisable, such as storing kids’ pools indoors when not in use, keeping rain gutters clean and free-flowing, repairing any leaky pipes and stopcocks, making sure that the fish pond and pool water are moving, emptying the pet’s drinking bowl outside and keeping the outdoor garbage bins covered, among others (Sarwar, 2014d).
The total time required for juvenile growth from the egg stage until adult emergence averaged 20 days in standardized laboratory conditions. This value falls within the range reported in previous studies, which is around 10 to 24 days. For example, Lee (1994) estimated the development from egg to adult as 12.1 days under laboratory conditions. Gomes et al. (1995) reared this species in bamboo stumps, tree holes and a car tire as diverse vessels and calculated 19.6, 27.3 and 37.5 days from egg stage to adult appearance, respectively. As in most insects and other poikilotherms, different variables such as temperature, relative humidity, food, and photoperiod are known to affect the life cycle of any mosquito species (Estrada-Franco & George, 1995). Usually, the initial and medium hatching age of the eggs was a significant factor that influenced the hatching rate (Christopher & Livdahl, 2006). Larvae presence in hatching medium may also alter hatch rates; and some stimulatory effects have been found at low densities, though as the density of larvae increased, egg hatch is inhibited (Edgerly & Marvier, 1992).
Here we found that the occurrence of Ae. albopictus adults varied throughout the seasons and the population attained maximum levels during October, enhanced gradually from January to May with a peak population observed in March. The population dropped severely by June and July, and started increasing until it reached its second peak in October. The lowest Ae. albopictus abundance was observed from December to February. Further, the present research also aimed to determine surveillance mechanism for detection of Ae. albopictusmosquitoes. The density of Ae. albopictus developmental stages was correlated with weather factors to identify which of them might predict mosquito activity and species distribution. Temperature, relative humidity and rainfall are important environmental variables, which impact mosquito activity, survival and distribution (Roiz et al., 2014; Yoo et al., 2016; Asigau & Parker 2018; Khan et al., 2018). Our results demonstrate that temperature significantly marks the host-searching activity of Ae. albopictus population. Specifically, temperatures between 16 °C to 28 °C seem to be more suitable for their host searching activity. Higher abundance in that period might be the direct consequence of the preceding rainfall providing the multiple breeding sites. Temperatures below 16 °C and above 28 °C lead to a decrease in population abundance. These results are consistent with earlier literature that showed temperatures between 15 °C to 28 °C to be more advantageous for mosquito activities (Asigau & Parker, 2018). The average relative humidity in the study area ranged between 35% to 68%. It has been described that high humidity increases egg production, larval growth and marks mosquito activities (De Almeida et al., 2010; Khan et al., 2018). Further, proper humidity between 44% and 68% stimulates mosquito flight activities (Jemal et al., 2018; Khan et al., 2018). Our results showed a vigorous positive significant correlation between rainfall and mosquito population growth, and are consistent with earlier findings (Roiz et al., 2014; Yoo et al., 2016; Khan et al., 2018; Drakou et al., 2020). It is documented that environmental variables are interrelated and for this particular reason, these factors should be considered keenly for the estimation of mosquito population assessments.
The dominant trend in the population of Ae. albopictus during post-monsoon rainfall season is similar to previous finding, where seasonal conditions have a great deal of influence on the dynamics of mosquito appearance (Sarwar & Rasool, 2022). Seasonal population dynamics differed between years, and mosquito species in relation to wetland, urban and forest habitats were mainly influenced by the water level, precipitation and temperature (Vujic et al., 2010). The abundance pattern stated by Waldock et al. (2013) showed that population densities depend on variable environmental conditions and the influence of climate change impacts on the geographic distributions of previously known tropical species. The population fluctuation of Ae. albopictus significantly depends on occurrence and distribution of the temporary habitat, environmental conditions, species biology and implementation of mosquito control campaigns (Nargis et al., 2012). This is also unpredictable with distribution patterns of the seasonal observation displaying that mosquito is constantly active from mid-spring to the end of December with high oviposition (Giatropoulos et al., 2012).
The populations of Ae. albopictus have been found increasing comparatively and summer temperatures are the possible reason of population increase during the season. Further, the populations gowth during low winter temperatures are likely to occur slowly and keep constant the emergence of adults during the season. Climate change associated with high temperature or without a reproductive diapausing phase, may enhance the rate of mosquito spread (Hawley et al., 1987; Barry & Juliano, 2001; Lounibos et al., 2003). Certainly, it is agreed that the population dynamics of Ae. albopictus is mainly driven by two factors (temperature and precipitation) in which temperature has a strong impact on the survival of mosquito populations and the growth of aquatic phases (Delatte et al., 2009). Secondly, the breeding sites are influenced by precipitation concentrations. Moreover, it is anticipated that the hatching of eggs is prompted by rainfall events through water supply by humans (Roiz et al., 2010). It is more likely that the temperature has a robust effect on the existence of terrestrial stages of mosquitoes and precipitations affect the carrying capacity of aquatic stages, thus raising the number of breeding sites accessible for Ae. albopictus (Sarwar, 2020; Sarwar & Roohi, 2020).
For winding up the results, the population dynamics of the vector displayed a distinct escalating style during the post-monsoon rainfall season. Subsequently, observations made found a fast turn down in its abundance during the winter months to point out a promising positive relationship between vector densities and weather prevalence. Since the vector abundance specifies disease occurrence, the management of a passable understanding of species diversification, dispersal arrangement and preferential habitat selection of vector species will help to advance an appropriate approach to manage mosquito populations and disease outbreak prevention. The dengue vector surveillance can be used to control population densities of mosquitoes and monitoring their quantities may offer information concerning the probabilities of disease activity in the state. Even then, the study showed general trends for Ae. albopictus reproductive activity in this locality and specific stimuli were noted, there can be enormous fluctuations from year to year. Since the mosquito specimens were accomplished on a restricted habitat type and using only usual sampling methods, it can be expected that not all species occurring in the studied locality would have been identified. Future comparative studies between adjacent fields and domestic precincts are needed to determine the effects of various factors that could provide insight into the origin or fall in the populations of Ae. albopictus to combat a public health threat. To study the feasible development of a dengue epidemic in an area, the recurring variation of mosquito population should be taken into account seeing as the abundance of adult males and females is the prime aspect for the spread of the disease. The present research implications suggest the effective regular monitoring of this important mosquito species for disease prevention activities further to determine the growth stage of the mosquitoes, which is helpful in decision making, whether vector control is necessary or at what time the control strategy is needed to be applied.
CONCLUSION
The results of this study indicated that different environmental factors had marked influences on Ae. albopictus developmental parameters such as hatching of eggs, larval and pupal growth, emergence rates, adult mosquito survivorship and reproduction capacity. Rainfall, humidity and temperature were significant factors influencing overall Ae. albopictus abundance in the studied area. Considering the whole seasonal dynamic observations, the peak breeding activities of mosquito were detected during the months of September, October and November owing to optimum environmental factors. The findings are significantly supportive in providing baseline information and revealed the seasonal abundance of Ae. albopictus enduring considerable variations throughout the study period. The present study might have vital contributions to control the population densities besides the growth stage of mosquito vectors and is useful for disease prevention activities.
Acknowledgments
We are highly thankful to all staff members and colleagues for their valuable contributions.
REFERENCES
Aida, H.N., Dieng, H., Ahmad, A.H., Satho, T., Nurita, A.T., Salmah, M.R.C., Miake, F. & Norasmah, B. (2011) The biology and demographic parameters of Aedes albopictus in northern peninsular Malaysia. Asian Pacific Journal of Tropical Biomedicine, ., (6): 472-477.
Asigau, S. & Parker, P.G. (2018) The influence of ecological factors on mosquito abundance and occurrence in Galápagos. Journal of Vector Ecololgy, 43, 125-137.
Barry, W.A. & Juliano, S.A. (2001) Temperature Effects on the Dynamics of Aedes albopictus (Diptera: Culicidae) Populations in the Laboratory. Journal of Medical Entomology, 38, 548-556.
Cheong, W.H. (1986) The vector of dengue and dengue hemorrhagic fever in Malaysia. In: Rudnick, A. & Lim, T.W. Dengue fever studies in Malaysia. Malaysian Institute of Medical Research, Kuala Lumpur, Bulletin, 23, 155-167.
Christopher, J.V. & Livdahl, T.P. (2006) Field and Laboratory Comparison of Hatch Rates in Aedesalbopictus (Skuse). Journal of the American Mosquito Control Association, 22, 609-614.
Costanzo, K.S., Mormann, K. & Juliano, S.A. (2005) Asymmetrical competition and patterns of abundance of Aedes albopictus and Culex pipiens (Diptera: Culicidae). Journal of Medical Entomology, 42, 559-570.
Cui, G., Zhong, S., Zheng, T., Li, Z., Zhang, X. Li, C., Hemming-Schroeder, E, Zhou G, & Li, Y. (2021) Aedes albopictus life table: environment, food, and age dependence survivorship and reproduction in a tropical area. Parasites . Vectors, ., 14 (1): 568.
De Almeida Costa, E.A.P., De Mendonça Santos, E.M., Correia, J.C. & De Albuquerque, C.M.R. (2010) Impact of small variations in temperature and humidity on the reproductive activity and survival of Aedes aegypti (Diptera, Culicidae). Revista Brasleria de Entomolgia, 54, 488-493.
Delatte, H., Gimonneau, G., Triboire, A. & Fontenille, D. (2009) Influence of temperature on immature development, survival, longevity, fecundity, and gonotrophic cycles of Aedes albopictus, vector of chikungunya and dengue in the Indian Ocean. Journal of Medical Entomology, 46, 33-41.
Drakou, K., Nikolaou, T., Vasquez, M., Petric, D., Michaelakis, A., Kapranas, A., Papatheodoulou, A., Koliou, M. (2020) The Effect of Weather Variables on Mosquito Activity: A Snapshot of the Main Point of Entry of Cyprus. International Journal of Environmental Research and Public Health, 21, 17 (4): 1403.
Edgerly, J.S. & Marvier, M.A. (1992) To hatch or not to hatch? Egg hatch in response to larval density and larval contact in a tree hole mosquito. Ecological Entomology, 17, 28-32.
Estrada-Franco, J.G. & George, B.C.J. (1995) Biology, disease relationships, and control of Aedes albopictus. Pan American Health Organization, Washington, D.C., Technical Paper, 42, 61.
Giatropoulos, A., Emmanouel, N., Koliopoulos,G. & Michaelakis, A. (2012) A study on distribution and seasonal abundance of Aedes albopictus (Diptera: Culicidae) population in Athens, Greece. Journal of Medical Entomology, 49, 262-269.
Gomes, A.C., Gotlieb, S.L., Marques, C.C., De Paula, M.B. & Marques, G.R. (1995) Duration of larval and pupal development stages of Aedes albopictus in natural and artificial containers. Revista de Saúde Pública, 29, 15-19.
Gubler, D.J. (2002) Epidemic dengue/ dengue hemorrhagic fever as public health, social, and economic problem in the 21st century. Trends in Microbiology, 10, 100-103.
Hawley, W. A., Reiter, P., Copeland, R.S., Pumpuni, C.B. & Craig. G.B.Jr. (1987). Aedes albopictus in North America probable introduction in tires from northern Asia. Science, 236, 1114-1116.
Hawley, W.A.(1988) The biology of Aedes albopictus. Journal of American Mosquito Control Association, ., 1-40.
Honorio, N.A., Castro, M.G., Barros, F.S., Magalhães, M.A. & Sabroza, P.C. (2009) The spatial distribution of Aedes aegypti and Aedes albopictus in a transition zone, Rio de Janeiro, Brazil. Cad Saude Publica, 25, 1203-1214.
Jemal, Y.& Al-Thukair, A.A. (2018) Combining GIS application and climatic factors for mosquito control in Eastern Province, Saudi Arabia. Saudi Journal of Biological Sciences, 25, 1593-1602.
Juliano, S.A., O'Meara, G.F., Morrill,J.R. & Cutwa, M.M. (2002) Desiccation and thermal tolerance of eggs and the coexistence of competing mosquitoes. Oecologia, 130, 458-469.
Khan, M.A., Elhossary, S., Khan, I.A., Al Zahrani, M.H., Al Zahrani, F.S. & Al Bashri, F.M. (2018) The impact of climatic variables with GIS application on the abundance of medically important mosquitoes (Diptera: Culicidae) in Jeddah, Saudi Arabia. International Journal of Mosquito Research, ., 12-18.
Lambrechts, L., Scott, T.W. & Gubler, D.J. (2010) Consequences of the Expanding Global Distribution of Aedes albopictus for Dengue Virus Transmission. PLoS. Neglected Tropical Diseases, ., e646.
Lee, S.J. (1994) Development of Eggs, Larvae and Pupae of Aedes albopictus (Skuse) (Diptera: Culicidae). China Journal of Entomology, 14, 13-32.
Lounibos, L.P., Escher, R.L., Lourenço-De-Oliveira, R. (2003) Asymmetric evolution of photoperiodic diapause in temperate and tropical invasive populations of Aedes albopictus (Diptera: Culicidae). Annals of Entomological Society of America, 96, 512-518.
Mitchell, C.J., Niebylski, M.L., Smith, G.C., Karabatsos, N., Martin, D., Mutebi, J.P., Craig, G.B.J. & Mahler, M.J. (1992) Isolation of eastern equine encephalitis virus from Aedes albopictus in Florida. Science, 257, 526-527.
Nargis, S., Tanginm, A. & Shefalim, B. (2012) Population Studies of Tree Hole Breeding Aedes Species (Diptera: Culicidae) In Dhaka University Campus and Its Adjacent Suhrawardi Park, Dhaka City, Bangladesh. Bangladesh Journal of Zoology, 40, 1-11.
Reiter, P. (1998) Aedes albopictus and the world trade in used tires, 1988-95: the shape of things to come? Journal of American Mosquito Control Association, 14, 83-94.
Roiz, D., Rosa, R., Arnoldi, D. & Rizzoli, A. (2010) Effects of temperature and rainfall on the activity and dynamics of host-seeking Aedes albopictus females in northern Italy. Vector Born Zoonotic Diseases, 10, 811-816.
Roiz, D., Ruiz, S., Soriguer, R. & Figuerola, J. (2014) Climatic effects on mosquito abundance in Mediterranean wetlands. Parasites & Vectors, ., 1-13.
Romiti, F., Casini, R., Magliano, A., Ermenegildi, A. & De Liberato, C. (2022) Aedes albopictus abundance and phenology along an altitudinal gradient in Lazio region (central Italy). Parasites & Vectors, 18, 92.
Rosen, L. (1987) Sexual transmission of dengue viruses by Aedes albopictus. American Journal of Tropical Medicine Hygine, 37, 398-402.
Rosen, L., Shroyer, D.A.,Tesh, R.B., Freier, J.E. & Lien, J.C. (1983) Transovarial transmission of dengue viruses by mosquitoes: Aedes albopictus and Aedes aegypti. American Journal of Tropical Medicine Hygine, 32, 1108-1119.
Rueda, L. M. (2004). Pictorial keys for the identification of mosquitoes (Diptera: Culicidae) associated with Dengue Virus Transmission. Zootaxa, 589(1), 1-60.
Russell, R.C., Williams, C.R., Sutherst, R.W. & Ritchie, S.A. (2005) Aedes .Stegomyia. albopictus - a dengue threat for southern Australia. Communicable Disease Intelligence Quarterly Report, 29, 296-298.
Sarwar, M. (2014 a) Proposing Solutions for the Control of Dengue Fever Virus Carrying Mosquitoes (Diptera: Culicidae) Aedes aegypti (Linnaeus) and Aedesalbopictus (Skuse). Journal of Pharmacology and Toxicological Studies, 2 (1) 1-6.
Sarwar, M. (2014 b) Proposals for the Control of Principal Dengue Fever Virus Transmitter Aedes aegypti (Linnaeus) Mosquito (Diptera: Culicidae). Journal of Ecology and Environmental Sciences, 24-28.
Sarwar, M. (2014 c) Dengue Fever as a Continuing Threat in Tropical and Subtropical Regions around the World and Strategy for Its Control and Prevention. Journal of Pharmacology and Toxicological Studies, ., 1-6.
Sarwar, M. (2014 d) Defeating Malaria with Preventative Treatment of Disease and Deterrent Measures against Anopheline Vectors (Diptera: Culicidae). Journal of Pharmacology and Toxicological Studies, 2 (4)1-6.
Sarwar, M. (2015 a) Elimination of Dengue by Control of Aedes Vector Mosquitoes (Diptera: Culicidae) Utilizing Copepods (Copepoda: Cyclopidae). International Journal of Bioinformatics and Biomedcal Engneering, 1, 53-58.
Sarwar, M. (2015 b) Reducing Dengue Fever through Biological Control of Disease Carrier Aedes Mosquitoes (Diptera: Culicidae). International Journal of Preventive Medicine Research, 1,161-166.
Sarwar, M. (2015 c) Controlling Dengue Spreading Aedes Mosquitoes (Diptera: Culicidae) Using Ecological Services by Frogs, Toads and Tadpoles (Anura) as Predators. American Journal of Clinical Neurology and Neurosurgery, 1, 18-24.
Sarwar, M. (2016 a) Mosquito-Borne Viral Infections and Diseases among Persons and Interfering with the Vector Activities. International Journal of Vaccines and Vaccination, 3, 00063.
Sarwar, M. (2016 b) Mosquitoes (Diptera: Culicidae) as Malaria Transmitters and Procedures for Suppression to Exposure and Spread of Vectors. Biomedical and Health Informatics, 1, 38-43.
Sarwar, M. (2020) Typical Flies: Natural History, Lifestyle and Diversity of Diptera. p. 50. In: Life Cycle and Development of Diptera (M. Sarwar, Editor). Intech Open Ltd., London, UK. 170.
Sarwar, M. & Roohi, A. (2020) New advances in insect vector biology and virus epidemiology. p. 301-311. In: Applied Plant Virology: Advances, Detection, and Antiviral Strategies, L. P. Awasthi (Ed.). Elsevier Inc., London. pp. 682.
Sarwar, M. & Rasool, B. (2022) Seasonal Prevalence and Phenomenal Biology as Tools for Dengue Mosquito Aedes aegypti (Linnaeus) (Diptera: Culicidae) Management. Brazilian Archives of Biology and Technology,65, e22220050.
Schober, P., Boer, C., Schwarte, L. (2018) Correlation coefficients: Appropriate use and interpretation. Anesthesia & Analgesia, 126, 1763-1768.
Statistix 8.1. (2003) User’s Manual. Analytical Software, Tallahassee, Florida, USA.
Vujić, A., Stefanović, A., Dragičević, I., Matijević, T., Pejčić, L., Knežević, M., ... & Veselić, S. (2010). Species composition and seasonal dynamics of mosquitoes (Diptera: Culicidae) in flooded areas of Vojvodina, Serbia. Archives of Biological Sciences, 62(4), 1193-1206.
Waldock, J., Chandra, N.L., Lelieveld, J., Proestos, Y., Michael, E., Christophides, G. & Parham, P.E. (2013) The role of environmental variables on Aedes albopictus biology and chikungunya epidemiology. Pathology and Global Health,107, 224-241.
Yoo, E.H., Chen, D. & Diao, C. (2016) The effects of weather and environmental factors on west nile virus mosquito abundance in greater toronto area. Earth Interactions, 20, 1-22.
Notas de autor
bilalisb2001@yahoo.com