Artículos
Contribution to the knowledge of larval chaetotaxy of the genus Enochrus Thomson: description of five New World species (Coleoptera: Hydrophilidae)
Contribución al conocimiento de la quetotaxia larval del género Enochrus Thomson: descripción de cinco especies del Nuevo Mundo (Coleoptera: Hydrophilidae)
Contribution to the knowledge of larval chaetotaxy of the genus Enochrus Thomson: description of five New World species (Coleoptera: Hydrophilidae)
Revista de la Sociedad Entomológica Argentina, vol. 82, núm. 4, 2023
Sociedad Entomológica Argentina
Recepción: 08 Agosto 2023
Aprobación: 08 Noviembre 2023
Abstract: The primary and secondary chaetotaxy of the larval head capsule and head appendages of five New World species of Enochrus Thomson are described for the first time. These species belong to three subgenera: Enochrus (Hugoscottia) tremolerasi (Knisch), E. (Lumetus) hamiltoni (Horn), E. (Methydrus) barituensis Fernández, E. (Methydrus) ochraceus (Melsheimer) and E. (Methydrus) pygmaeus (Fabricius). Morphometric characters derived from the head capsule and mouthparts are also included, together with detailed illustrations of all characters. A comparative study of the chaetotaxy of four subgenera of Enochrus yields an important number of characters useful to diagnose the larvae of these subgenera, and could be used in future phylogenetic analyses. Several characters showing variation within a subgenus are discussed; additionally, possible diagnostic characters for Enochrus and the subfamily Enochrinae are discussed. The chaetotaxy of two subgenera, Enochrus Thomson and Hydatotrephis MacLeay, still remains unknown, and should be a priority to complete the larval knowledge of the genus Enochrus.
Keywords: Aquatic beetles, Enochrinae, Morphology, New World, Primary chaetotaxy.
Resumen: Se describe por vez primera la quetotaxia primaria y secundaria de la cápsula cefálica y de los apéndices cefálicos de cinco especies de Enochrus Thomson del Nuevo Mundo. Estas especies pertenecen a tres subgéneros: Enochrus (Hugoscottia) tremolerasi (Knisch), E. (Lumetus) hamiltoni (Horn), E. (Methydrus) barituensis Fernández, E. (Methydrus) ochraceus (Melsheimer) y E. (Methydrus) pygmaeus (Fabricius). Caracteres morfométricos derivados de diferentes medidas de la cápsula cefálica y de las piezas bucales también se incluyen, así como ilustraciones detalladas de todos los caracteres. El estudio comparado de la quetotaxia de cuatro subgéneros de Enochrus provee un número importante de caracteres para separar las larvas de estos subgéneros, y estos caracteres pueden llegar a ser útiles para futuros estudios filogenéticos. Se discuten algunos caracteres que presentan variación entre especies del mismo subgénero y también algunos caracteres que pueden ser diagnósticos para el género Enochrus y la subfamilia Enochrinae. La quetotaxia de dos subgéneros aún se desconoce, Enochrus Thomson y Hydatotrephis MacLeay, y su descripción debería ser una prioridad para completar el conocimiento larval de este género.
Palabras clave: Américas, Enochrinae, Escarabajos acuáticos, Morfología, Quetotaxia primaria.
INTRODUCTION
Enochrinae is a small subfamily of water scavenger beetles which includes just four genera: Cymbiodyta Bedel, Enochrella Hansen, Enochrus Thomson and Notionotus Spangler. Larvae have been described for all genera except for Enochrella, on the other hand Enochrus is the best known so far. The genus Enochrus has a worldwide distribution and is one of the most speciose genera within Hydrophilidae, including around 225 species (Short & Fikáček, 2011, 2013). Enochrus is subdivided into six subgenera: Enochrus (s. str.) Thomson, E. (Holcophilydrus) Kniz, E. (Hugoscottia) Knisch, E. (Hydatotrephis) MacLeay, E. (Lumetus) Zaitzev, and E. (Methydrus) Rey. Larvae have been described for most of the subgenera and many general morphological descriptions are available in the literature (for a comprehensive list of morphological descriptions see Byttebier & Torres, 2009, Table I).
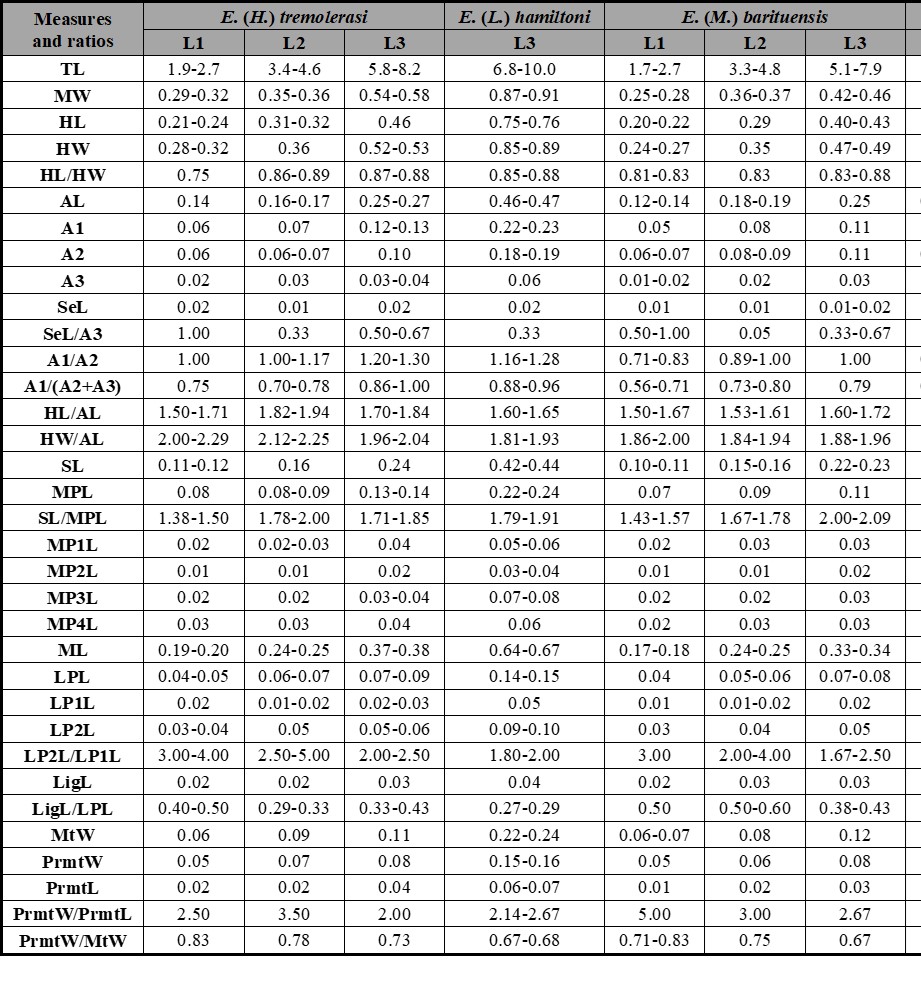
Table I.
Measurements (in mm) and ratios for different larval instars of described Enochrus species. Abbreviations: see Material and Methods section.
Nonetheless, only six detailed chaetotaxic descriptions have been published so far: E. (Methydrus) coarctatus (Gredler), E. (Methydrus) japonicus (Sharp), E. (Methydrus) vulgaris (Steinheil), E. (Hugoscottia) variegatus (Steinheil), E. (Holcophilydrus) simulans (Sharp), E. (Holcophilydrus) umbratus (Sharp) (Fikáček, 2006; Byttebier & Torres, 2009; Minoshima & Hayashi, 2011). Some chaetotaxic characters of a putative third instar larva of E. (Holcophilydrus) niisatoi Minoshima were also briefly mentioned by Minoshima (2017).
Larval characters in Hydrophiloidea, including chaetotaxy, have proved to be a valuable tool for systematic and phylogenetic studies (e.g. Archangelsky, 2004; Fikáček, 2006; Fikáček et al., 2013; Minoshima et al., 2013; Archangelsky et al., 2021; Rodriguez et al., 2021). However, the limited number of chaetotaxic descriptions poses two difficulties: 1- limits our understanding of intrageneric variation; 2- it also limits the use of this source of characters in phylogenetic research since most generic larval chaetotaxic diagnoses are based on just one or two species. I have gathered a significant amount of larval material of Enochrus species through the years. Part of this material has been described following a general morphological approach (Archangelsky, 1997, 1999, 2002), but chaetotaxic descriptions of these species were never presented before since the chaetotaxic and morphometric systems were not developed at the time (Fikáček, 2006; Fikáček et al., 2008; Torres, 2008; Byttebier & Torres, 2009). The aim of this contribution is to increase larval knowledge of Hydrophilidae by describing in detail the chaetotaxy and morphometry of five species of Enochrus belonging to three subgenera: Hugoscottia, Lumetus and Methydrus. This information will be very useful to researchers working in hydrophiloid phylogeny using larval characters; additionally, this is the first chaetotaxic description of larvae of the subgenus Lumetus.
MATERIAL AND METHODS
This paper focuses on the description of the chaetotaxic and morphometric characters of the head capsule and head appendages; for general morphological characters of these species the following descriptions should be consulted (Richmond, 1920; Wilson, 1923; Gundersen, 1967; Archangelsky, 1997, 2002).
Source of material. Enochrus (Hugoscottia) tremolerasi (Knisch) (Fig. 1c): Argentina, La Rioja province; Departamento Castro Barros, Arroyo Santa Vera Cruz, 1900 m, 28° 40’ 34” S, 66° 59’ 23” W, 5.IX.1999, M. Archangelsky leg. (4 L1, 2 L2, 4 L3). E. (Lumetus) hamiltoni (Horn): USA, Ohio, Holmes Co., Killbuck Creek, near Killbuck, 21.VII.1990, M. Archangelsky leg. (3 L3). E. (Methydrus) barituensis Fernández (Fig. 1a-b): Argentina, La Rioja province; Departamento Sanagasta, Arroyo Tambito, 1340 m, 29° 09’ 47” S, 67° 04’ 47” W, 29.XI.1999, M. Archangelsky leg. (4 L1, 2 L2, 4 L3). E. (Methydrus) ochraceus (Melsheimer): USA, Ohio, Franklin Co., OSU West Campus pond, 10.VII.1990, M. Archangelsky leg. (3 L1, 2 L3). Enochrus (Methydrus) pygmaeus (Fabricius): (3 L1, 1 L2, 4 L3). The material studied is kept in the larval collection of the author and will be deposited in the larval collection of the Laboratory of Entomology, Buenos Aires University, Argentina (LEBA).

Fig. 1.
Habitus of third instar larvae. a. Enochrus (M.) barituensis, dorsal view. b. Enochrus (M.) barituensis, lateral view, ventral view. c. Enochrus (H.) tremolerasi, dorsal view. Scale bars = 1 mm.
Methods. Adults of all studied species were reared in laboratory, for most species the complete life cycle was obtained; for E. (M.) ochraceus only first and third instar larvae were available and for E. (L.) hamiltoni only third instar larvae were available. Larval specimens were cleared in warm lactic acid, dissected and mounted on glass slides with Hoyer’s medium. Observations (up to 1000×), photographs and drawings were made with a Leica DMLB compound microscope with a camera lucida and a Leica S6D dissecting microscope with a photographic camera attached. Drawings were scanned and digitally edited, photographs were assembled using the freeware program CombineZP (Hadley, 2010).
Morphometry. Diverse measurements of the head capsule and head appendages were taken with a micrometer and were used to calculate ratios. Measured structures were adjusted as parallel as possible to the plane of the objective. The following measurements were taken; TL: total body length, measured in dorsal view, from anterior margin of frontoclypeus to posterior margin of the median lobe of spiracular atrium; MW: maximum body width, measured at level of prothorax; HL: head length, medially along epicranial stem from anterior margin of frontoclypeus to occipital foramen; HW: maximum head width; AL: length of antenna, derived by adding the lengths of the first (A1L), second (A2L) and third (A3L) antennomeres; SeL: length of antennal sensorium; SL: length of stipes; MPL: length of maxillary palpus, obtained by adding the lengths of the first (MP1L), second (MP2L), third (MP3L) and fourth (MP4L) palpomeres; ML: length of maxilla, derived by adding SL and MPL, cardo omitted; LPL: length of labial palpus, obtained by adding the lengths of the first (LP1L) and second (LP2L) palpomeres; LigL: length of ligula; MtW: maximum width of mentum; PrmtL: length of prementum, measured medially on dorsal side; PrmtW: maximum width of prementum.
Chaetotaxy. Primary (present in first-instar larva) and secondary (arising in later instars) setae and pores were identified in the cephalic capsule and head appendages. Primary sensilla were labeled by comparison with the ground plan of chaetotaxy of Hydrophilidae (Fikáček et al., 2008; Byttebier & Torres, 2009). Homologies were established using the criterion of similarity of position (Wiley, 1981). Sensilla are coded with a number and two capital letters, usually corresponding to the first two letters of the name of the structure on which they are located. Chaetotaxic descriptions of first and third larval instars are presented, second instar larvae are very similar to third instars. The following abbreviations were used. AN: antenna; FR: frontale; LA: labium; MN: mandible; MX: maxilla; PA: parietale; gAN: group of antennal sensilla; gAPP: group of sensilla on the inner appendage of the maxilla; gFR1, gFR2: group of sensilla on the frontale; gLA: group of sensilla on the labial palp; gMX1: group of sensilla on the maxillary palpomere 4; gMX2: group of sensilla on inner margin of maxillary stipes.
RESULTS
Genus Enochrus Thomson
Primary chaetotaxic diagnosis
Head capsule: Frontale with 40-48 sensilla (Figs. 2a, 8a, 12a, 16a): two setae close to frontal lines (FR1) at midlength; two pores (FR2) situated anteromesally to FR1; two very short setae (FR3) placed more anteriorly and mesally than FR2; five setae (FR5-7, FR9-10) and two pores (FR4, FR14) near inner margin of antennal socket: FR5 and FR6 posterior to antennal socket, FR7 on inner margin of antennal socket, FR4 mesal to FR7, FR9 and FR10 situated mesoanteriorly to antennal socket, FR14 anterior to antennal socket; nasale with six dorsal stout setae (gFR1), sometimes with one or two minute ventral setae (Fig. 2c); anterior margin of each epistomal lobe with a pair of very short setae and one or two pores (gFR2, pores difficult to see in some species); one pair of pores (FR15 and two long setae (FR8) posterior to gFR1, FR8 closer to FR15 on left side of frontale; two pores (FR11, FR13) and one very short seta (FR12 posterior to epistomal lobes, right side with FR12 posterior to FR11 and FR13, left side with FR12 between FR11 and FR13. Each parietale with 30 sensilla (Figs. 2a-b, 8a-b, 12a-b, 16a-b): dorsal surface basally with four short setae (PA1-2, PA4-5 and one pore (PA3) arranged in an irregular longitudinal line at midwidth; one subbasal pore (PA6) close coronal line; one long seta (PA7) and a shorter one (PA12) on central area of dorsal surface of parietale; one pore (PA10) distal to PA12; two long setae (PA8, PA9) distal to PA10, posterior to antennal socket; three setae (PA11, PA13-14) and one pore (PA15) at midlength on lateral side of parietale; anterolateral corner of parietale with one pore (PA19) and two long setae (PA20-21). Ventral surface with two pores (PA29-30) at basal fourth, PA29 at midwidth, PA30 lateral; distal to pore PA29 two long setae (PA28 more basal, PA26 more distal) and one pore (PA27) between PA28 and PA26 arranged in a longitudinal row; two long setae (PA18 more basal, PA16 more distal) and one pore (PA17) near seta PA16 along lateral side of parietale; anterolateral corner of parietale with one long seta (PA22) and three pores (PA23-25) on mandibular acetabulum.
Antenna (Figs. 3a, 9a, 13a, 17a): A1 bearing five pores (AN1-5), AN1 dorsal, at midlength close to outer face, AN2 dorsal, distal, at midwidth, AN3 apical on inner face, AN4 and AN5 apical, on ventral side, AN4 close to outer face, AN5 at midwidth; A2 with one dorsal distal pore (AN6) at midwidth and five setae (AN7-11), AN7-9 dorsal, at base of sensorium (SE1), long seta AN10 and short seta AN11 apical or subapical, on inner face; A3 bearing a group of six sensilla (gAN).
Mandibles (Figs. 3b-c, 9b-c,13b-c, 17b-c): Bearing two setae (MN1, MN5) and four pores (MN2-4, MN6); seta MN1 long, on outer face, seta MN5 minute, distal, on outer margin; pores MN2-4 forming a triangle on dorsal surface at about midlength of mandible, pore MN6 subapical on inner face.
Maxilla (Figs. 3d-e, 9d-e, 13d-e, 17d-e): Cardo bearing one ventral seta (MX1). Stipes with a row of five setae (MX7-11) along inner face, MX7 subbasal, MX8-11 stouter, distal to MX7, distance between setae variable, simple or bifid at the apex; outer face with two long setae (MX5-6) and one pore (MX4); ventrally with two pores at basal third (MX2-3), MX3 closer to inner face, MX2 closer to outer face. MP1 with a stout basal seta (MX16) on dorsal inner face; ventrally with three distal sensoria, two long setae (MX13-14) and one pore (MX12), MX13 between MP12 and MP14; two pores (MX15, MX17) at base of inner appendage, on border between appendage and membrane, inner appendage bearing a three sensoria (gAPP). MP2 with two distal pores (MX18-19) and one minute basal seta (MX27), MX19 dorsal on inner face, MX18 closer to outer face, MX27 on outer face. MP3 with two long setae (MX21, MX23) and two pores (MX20, MX22), seta MX23 and pore MX20 on outer face, seta MX21 and pore MX22 closer to inner face. MP4 bearing a basal long seta (MX24) and two subapical sensoria on outer face (pore MX26 and digitiform sensillum MX25), apical area of palpomere with several minute sensilla (gMX).
Labium (Figs. 2b, 3f-g, 8b, 9f-g, 12b, 13f-g, 16b, 17f-g): Submentum with two pairs of setae (LA1-2), LA1 long, on lateral margins, LA2 very short, on anterior lobes. Mentum with two setae (LA3) and two pores (LA4) at about midlength, LA3 below LA4. Prementum dorsally with two pores (LA8) and two minute seta-like sensilla (LA9) on membrane connecting prementum with palpi; ventrally with two minute setae (LA5) on basal corners, two long setae (LA6) at midlength and two pores (LA7) on distal corners. Ligula dorsally with two apical sensilla (LA12) and a pair of long setae (LA10) on base of ligula or on membrane at base of ligula; ventrally with a pair of pore-like sensilla (LA11) of variable position. LP1 with a minute basal seta (LA13) ventrally and one dorsal pore (LA14) on membrane connecting with LP2. LP2 with one dorsal subapical pore (LA15) on outer face and a group of several apical sensoria (gLA).
Enochrus (Hugoscottia) tremolerasi (Knisch)
First instar larva
Head capsule (Fig. 2a-c): Frontale with 43 sensilla; seta FR1 moderately long, setae FR5 and FR6 subequal in length; gFR1 with six dorsal setae and one minute ventral seta below third seta from the right; first and second setae on right side shorter than remaining others; each gFR2 with two short setae and one small mesal pore. Parietale with setae PA7, PA12-14 and pore PA15 arranged in an almost transverse line; setae PA26, PA28 and pore PA27 not closely aggregated, PA27 closer to PA28 than to PA26; setae PA26 and PA16 at same distance from anterior margin of parietale.
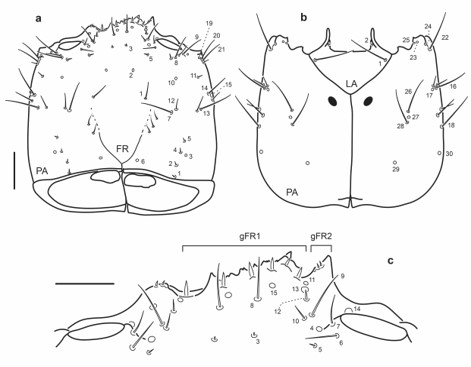
Fig. 2.
Chaetotaxy of first instar larva of E. (H.) tremolerasi. a. Head capsule, dorsal view. b. Head capsule, ventral view. c. Detail of clypeolabrum, dorsal view. Scale bars: a-b = 0.1 mm, c = 0.025 mm.
Antenna (Fig. 3a): Setae AN10-11 on membrane connecting A2 and A3.
Mandibles (Fig. 3b-c): Seta MN1 very long, distinctly projecting past pore MN4; pore MN2 equidistant to pore MN4 and seta MN1; seta MN5 equidistant to pore MN4 and apex of mandible.
Maxilla (Fig. 3d-e): Seta MX 1 rather short; setae MX8-9 closely aggregated; pore MX4 at same level than seta MX5.
Labium (Fig. 3f-g): Distance between pores LA7 greater than that between setae LA6; distance between pores PA8 shorter than that between seta-like sensilla LA9; pores LA11 at midlength of ligula; setae LA10 at base of ligula.
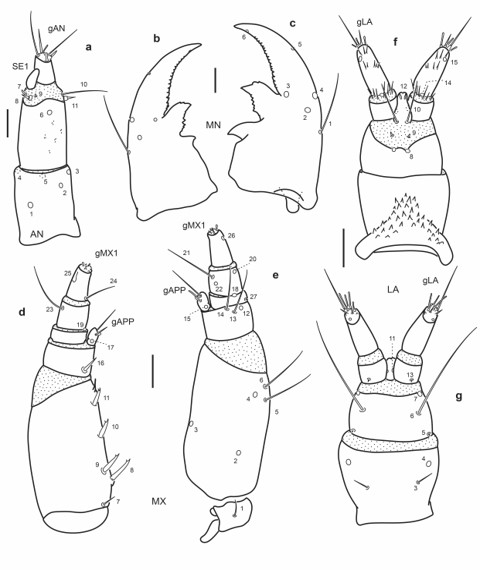
Fig. 3.
Chaetotaxy of first instar larva of E. (H.) tremolerasi. a. Left antenna, dorsal view. b. Left mandible, dorsal view. c. Right mandible, dorsal view. d. Left maxilla, dorsal view. e. Left maxilla, ventral view. f. Labium, dorsal view. g. Labium, ventral view. Scale bars = 0.025 mm.
Third instar larva
Head capsule (Fig. 4a-c): Frontale without secondary sensilla; right two setae of gFR1 proportionally larger than in first instar larvae. Parietale with several secondary sensilla distributed as follows: dorsally with four short setae between pore PA6 and seta PA7, one minute seta near PA8, one rather short seta between PA8 and PA9, one pore close to PA9, two rather short setae near PA13 and PA14; ventrally with one seta posterior to PA18 and on seta distal to PA16.
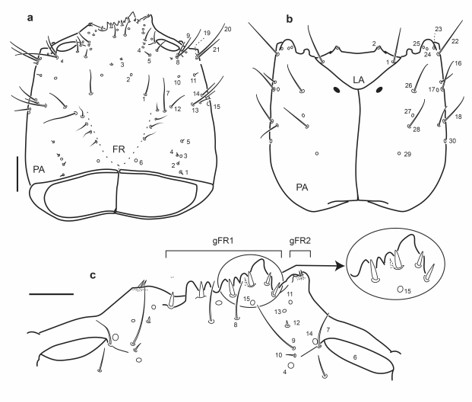
Fig. 4.
Chaetotaxy of third instar larva of E. (H.) tremolerasi. a. Head capsule, dorsal view. b. Head capsule, ventral view. c. Detail of clypeolabrum, dorsal view. Scale bars: a-b = 0.1 mm, c = 0.05 mm.
Antenna (Fig. 5a): Antennal sensorium (SE1) proportionally shorter than A3.
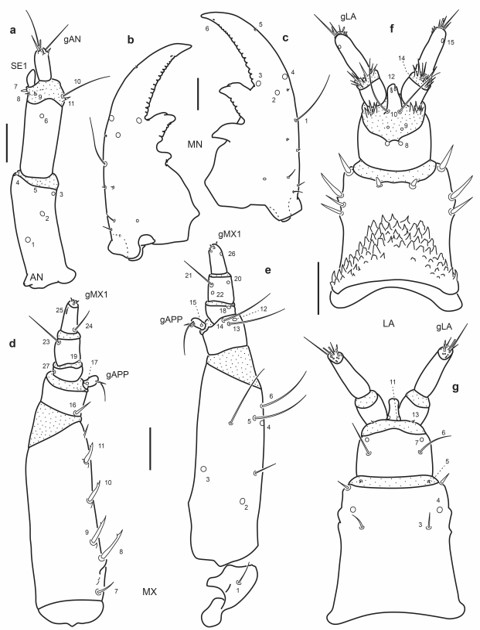
Fig. 5.
Chaetotaxy of third instar larva of E. (H.) tremolerasi. a. Left antenna, dorsal view. b. Left mandible, dorsal view. c. Right mandible, dorsal view. d. Left maxilla, dorsal view. e. Left maxilla, ventral view. f. Labium, dorsal view. g. Labium, ventral view. Scale bars = 0.05 mm.
Mandibles (Fig. 5b-c): Several secondary sensilla distributed as follows: one minute seta posterior to MN4, two minute setae posterior to MN1, a group of three short basal setae close to outer face and one pore placed more mesally than this group.
Maxilla (Fig. 5d-e): Outer face of stipes with two ventral secondary setae, one long mesal to MX5, the other short, halfway between pores MX2 and MX4.
Labium (Fig. 5f-g): Seta LA3 positioned more distally; mentum with nine or ten stout secondary setae dorsally, four on distal margin and two or three on each lateral face; ventrally with one short seta on each anterolateral corner.
Enochrus (Lumetus) hamiltoni (Horn)
Third instar larva
Head capsule (Fig. 6a-c): Frontale with 44 primary sensilla; seta FR1 moderately short, seta FR5 slightly shorter than FR6; gFR1 with six stout dorsal setae (specimen illustrated with one supernumerary seta, therefore bearing seven dorsal setae), first seta on right side shorter than remaining ones, ventrally with two minute setae between middle dorsal setae; each gFR2 with two minute setae and a small mesal pore; frontale with short secondary setae distal to FR1 (illustrated specimen with one on right side and two on left side). Parietale with numerous secondary sensilla distributed as follows: dorsally with seven to eight short setae between pore PA6 and seta PA7, one short seta near PA8, one rather long seta near PA9, one seta near PA13, one pore between seta PA9 and pore PA19, one short seta posterior to PA21; ventrally with four short setae between pore PA30 and seta PA16, one more seta distal to pore PA17.
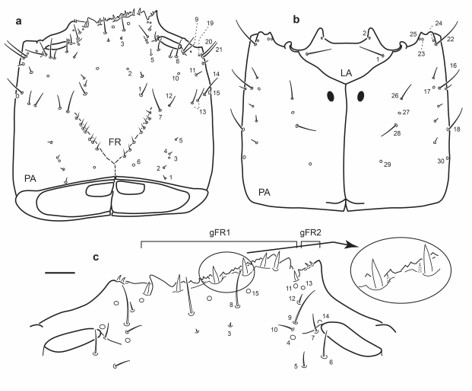
Fig. 6.
Chaetotaxy of third instar larva of E. (L.) hamiltoni. a. Head capsule, dorsal view. b. Head capsule, ventral view. c. Detail of clypeolabrum, dorsal view. Scale bars: a-b = 0.1 mm, c = 0.05 mm.
Antenna (Fig. 7a): Antennal sensorium (SE1) very short; setae AN10-11 subapical.
Mandibles (Fig. 7b-c): Seta MN1 rather short; seta MN5 closer to apex of mandible than to MN4; one minute secondary seta between MN1 and MN4, five short secondary setae posterior to MN1 two closer to MN1, remaining three more basal.
Maxilla (Fig. 7d-e): Outer face of stipes with two ventral secondary setae, one long mesal to MX5, the other shorter, distal to pore MX2.
Labium (Fig. 7f-g): Dorsal surface of mentum with 14 stout secondary setae, four on distal margin and five on each lateral face; ventrally with one short seta on each anterolateral corner.
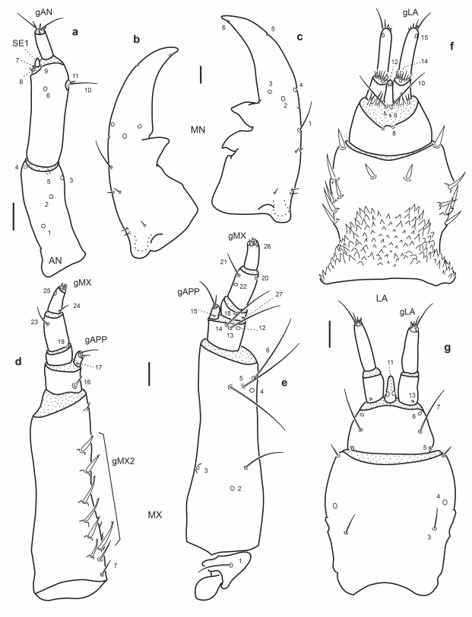
Fig. 7.
Chaetotaxy of third instar larva of E. (L.) hamiltoni. a. Left antenna, dorsal view; b. Left mandible, dorsal view. c. Right mandible, dorsal view. d. Left maxilla, dorsal view. e. Left maxilla, ventral view. f. Labium, dorsal view. g. Labium, ventral view. Scale bars = 0.05 mm.
Enochrus (Methydrus) barituensis Fernández
First instar larva
Head capsule (Fig. 8a-c): Frontale with 42 sensilla; seta FR1 moderately long, setae FR5 much shorter than FR6; gFR1 with six setae, second seta on right side shorter than remaining others; each gFR2 with two short setae and one small mesal pore. Parietale with setae PA7 and PA12 closely aggregated, seta PA14 and pore PA15 distal to PA13; setae PA26, PA28 and pore PA27 closely aggregated; seta PA16 and pore PA17 more distally placed than PA26.
Antenna (Fig. 9a): Setae AN10-11 at distal two thirds on inner face of A2.
Mandibles (Fig. 9b-c): Seta MN1 long reaching pore MN4; pore MN2 distinctly closer to pore MN4 than to seta MN1; seta MN5 closer to apex of mandible than to pore MN4.
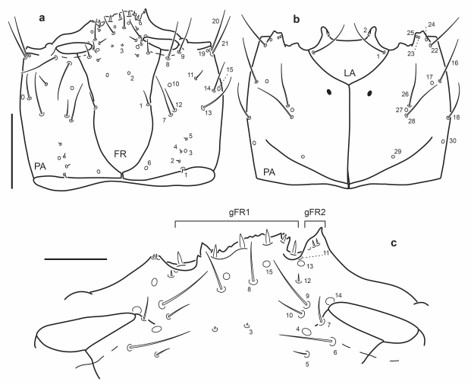
Fig. 8.
Chaetotaxy of first instar larva of E. (M.) barituensis. a. Head capsule, dorsal view. b. Head capsule, ventral view. c. Detail of clypeolabrum, dorsal view. Scale bars: a-b = 0.1 mm, c = 0.025 mm.
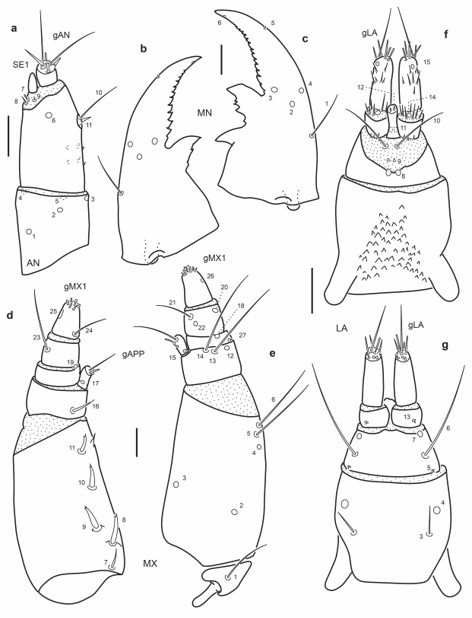
Fig. 9.
Chaetotaxy of first instar larva of E. (M.) barituensis. a. Left antenna, dorsal view. b. Left mandible, dorsal view. c. Right mandible, dorsal view. d. Left maxilla, dorsal view. e. Left maxilla, ventral view. f. Labium, dorsal view. g. Labium, ventral view. Scale bars = 0.025 mm.
Maxilla (Fig. 9d-e): Seta MX1 long; setae MX8–9 not too closely aggregated; pore MX4 posterior to seta MX5; setae MX13-14 very long.
Labium (Fig. 9f-g): Distance between pores LA7 subequal or less than that between setae LA6; distance between pores PA8 subequal to that between seta-like sensilla LA9; pores LA11 at base of ligula; setae LA10 on membrane below ligula.
Third instar larva
Head capsule (Fig. 10a-c): Frontale lacking secondary sensilla. Parietale with several secondary sensilla distributed as follows: dorsally with two to four rather short setae between pore PA6 and seta PA7, one very short seta mesal to PA8, another short seta between PA8 and PA9, one pore close to PA9, one minute seta behind PA21, one short seta near PA13 and another posterior to pore PA15; ventrally with two setae between PA16 and PA18 and two short setae distal to PA16 and PA17.
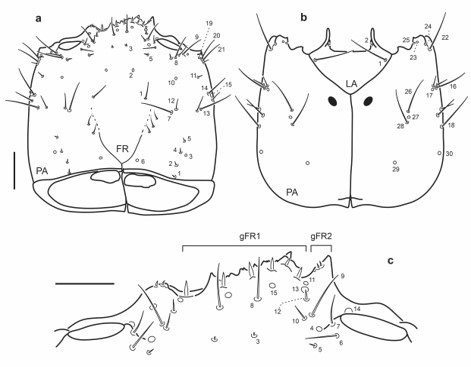
Fig. 10.
Chaetotaxy of third instar larva of E. (M.) barituensis. a. Head capsule, dorsal view. b. Head capsule, ventral view. c. Detail of clypeolabrum, dorsal view. Scale bars: a-b = 0.1 mm, c = 0.05 mm.
Antenna (Fig. 11a): Antennal sensorium (SE1) proportionally shorter than A3.
Mandibles (Fig. 11b-c): Several secondary sensilla distributed as follows: one minute seta between MN4 and MN1, two minute setae posterior to MN1, a group of four short basal setae close to outer face.
Maxilla (Fig. 11d-e): Outer face of stipes with two secondary setae, one long mesal to pore MX4, the other short, near pore MX2; setae MX16 and MX23 proportionally shorter.
Labium (Fig. 11f-g): Mentum with eight or nine stout secondary setae dorsally, three or four on distal margin and two or three on each lateral face; ventrally with one short and slender seta on each anterolateral corner.
Enochrus (Methydrus) ochraceus (Melsheimer)
First instar larva
Head capsule (Fig. 12a-c): Frontale with 42 sensilla; seta FR1 rather short, setae FR5 much shorter than FR6; gFR1 with six setae, second seta on right side distinctly shorter than remaining others; each gFR2 with two short setae and one small mesal pore. Parietale with setae PA7 and PA12 closely aggregated, seta PA12 much shorter than PA7; seta PA14 and pore PA15 distal to PA13; seta PA20 much shorter than PA21; setae PA26, PA28 and pore PA27 closely aggregated; seta PA16 and pore PA17 more distally placed than PA26.
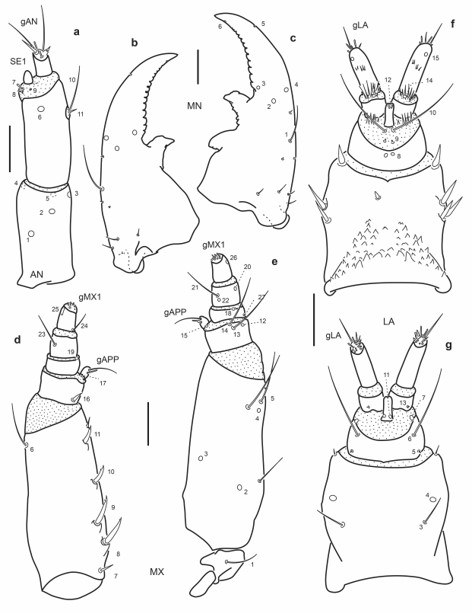
Fig. 11.
Chaetotaxy of third instar larva of E. (M.) barituensis. a. Left antenna, dorsal view. b. Left mandible, dorsal view. c. Right mandible, dorsal view. d. Left maxilla, dorsal view. e. Left maxilla, ventral view. f. Labium, dorsal view. g. Labium, ventral view. Scale bars = 0.05 mm.
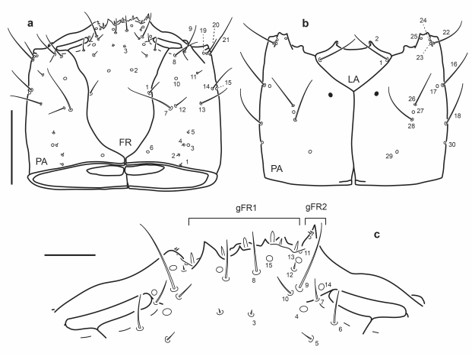
Fig. 12.
Chaetotaxy of first instar larva of E. (M.) ochraceus. a. Head capsule, dorsal view. b. Head capsule, ventral view. c. Detail of clypeolabrum, dorsal view. Scale bars: a-b = 0.1 mm, c = 0.025 mm.
Antenna (Fig. 13a): Setae AN10-11 at distal two thirds on inner face of A2.
Mandibles (Fig. 13b-c): Seta MN1 long reaching pore MN4; pore MN2 distinctly closer to pore MN4 than to seta MN1; seta MN5 closer to apex of mandible than to pore MN4.
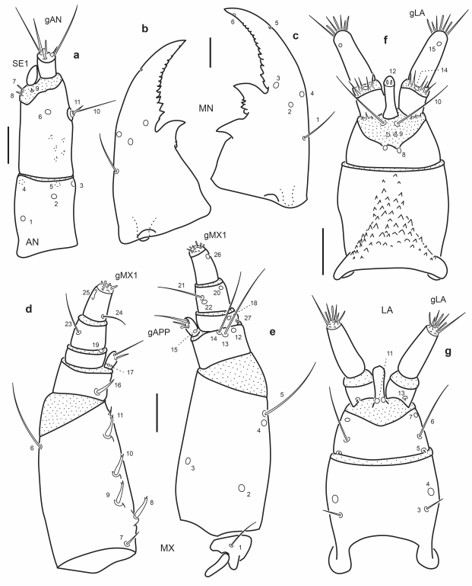
Fig. 13.
Chaetotaxy of first instar larva of E. (M.) ochraceus. a. Left antenna, dorsal view. b. Left mandible, dorsal view. c. Right mandible, dorsal view. d. Left maxilla, dorsal view. e. Left maxilla, ventral view. f. Labium, dorsal view. g. Labium, ventral view. Scale bars = 0.025 mm.
Maxilla (Fig. 13d-e): Seta MX1 long; setae MX8–9 not too closely aggregated; pore MX4 posterior to seta MX5; setae MX13-14 very long.
Labium (Fig. 13f-g): Distance between pores LA7 subequal or less than that between setae LA6; distance between pores PA8 slightly larger than that between seta-like sensilla LA9; pores LA11 at base of ligula; setae LA10 on membrane below ligula.
Third instar larva
Head capsule (Fig. 14a-c): Frontale lacking secondary sensilla. Parietale with several secondary sensilla distributed as follows: dorsally with one to three short setae between pore PA6 and seta PA7, one very short seta mesal to PA8, another long seta between PA8 and PA9, one pore close to PA9, one minute seta behind PA21, one short seta mesal to PA13 and another laterad to PA13; one short seta posterior to pore PA15; ventrally with one short seta between distal to PA18, two short setae distal to PA16 and PA17.
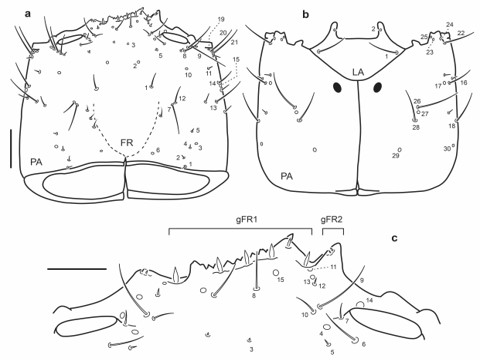
Fig. 14.
Chaetotaxy of third instar larva of E. (M.) ochraceus. a. Head capsule, dorsal view. b. Head capsule, ventral view. c. Detail of clypeolabrum, dorsal view. Scale bars: a-b = 0.1 mm, c = 0.05 mm.
Antenna (Fig. 15a): Antennal sensorium (SE1) proportionally shorter than A3.
Mandibles (Fig. 15b-c): Several secondary sensilla distributed as follows: one minute seta between MN4 and MN1, one or two very short setae posterior to MN1, a group of four short basal setae close to outer face.
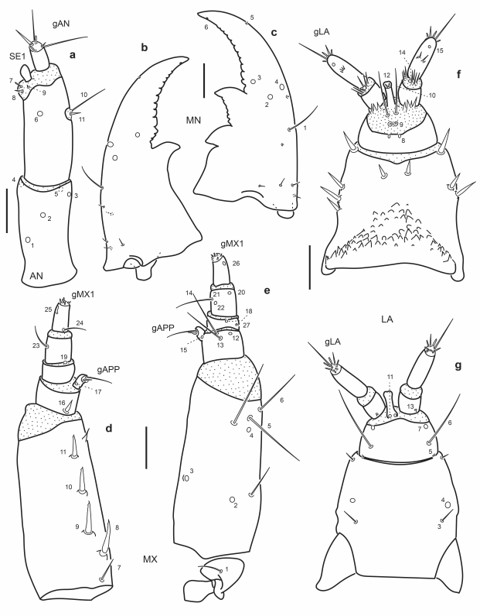
Fig. 15.
Chaetotaxy of third instar larva of E. (M.) ochraceus. a. Left antenna, dorsal view. b. Left mandible, dorsal view. c. Right mandible, dorsal view. d. Left maxilla, dorsal view. e. Left maxilla, ventral view. f. Labium, dorsal view. g. Labium, ventral view. Scale bars = 0.05 mm.
Maxilla (Fig. 15d-e): Stipes with two secondary setae, one long mesal to pore MX4, the other short, near pore MX2 on outer face; setae MX16 and MX23 proportionally shorter.
Labium (Fig. 15f-g): Mentum with nine or ten stout secondary setae dorsally, four on distal margin and two or three on each lateral face; ventrally with one short and slender seta on each anterolateral corner.
Enochrus (Methydrus) pygmaeus (Fabricius)
First instar larva
Head capsule (Fig. 16a-c): Frontale with 40 sensilla; seta FR1 rather short, setae FR5 much shorter than FR6; gFR1 with six setae, second seta on right side distinctly shorter than remaining others; each gFR2 with two short setae, small mesal pore present in other species not found. Parietale with setae PA7 and PA12 closely aggregated, seta PA12 much shorter than PA7; seta PA14 and pore PA15 distal to PA13; seta PA20 much shorter than PA21; setae PA26, PA28 and pore PA27 closely aggregated; seta PA16 and pore PA17 more distally placed than PA26.
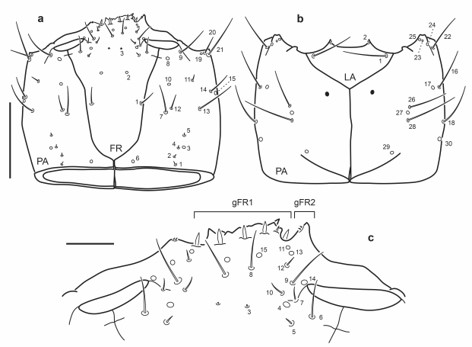
Fig.16.
Chaetotaxy of first instar larva of E. (M.) pygmaeus. a. Head capsule, dorsal view. b. Head capsule, ventral view. c. Detail of clypeolabrum, dorsal view. Scale bars: a-b = 0.1 mm, c = 0.025 mm.
Antenna (Fig. 17a): Setae AN10-11 at distal two thirds on inner face of A2.
Mandibles (Fig. 17b-c): Seta MN1 rather short, not reaching pore MN4; pore MN2 distinctly closer to pore MN4 than to seta MN1; seta MN5 closer to apex of mandible than to pore MN4.
Maxilla (Fig. 17d-e): Seta MX1 long; setae MX8-9 not rather closely aggregated; pore MX4 posterior to seta MX5; setae MX13-14 very long.
Labium (Fig. 17f-g): Distance between pores LA7 subequal or less than that between setae LA6; distance between pores PA8 subequal to that between seta-like sensilla LA9; pores LA11 at base of ligula; setae LA10 on membrane below ligula.
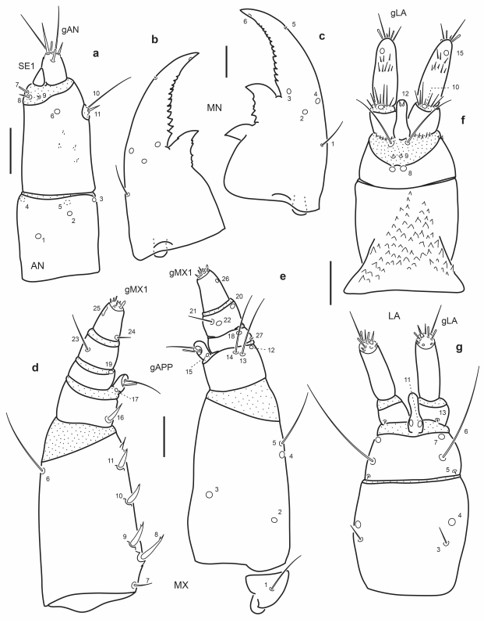
Fig. 17.
Chaetotaxy of first instar larva of E. (M.) pygmaeus. a. Left antenna, dorsal view. b. Left mandible, dorsal view. c. Right mandible, dorsal view. d. Left maxilla, dorsal view. e. Left maxilla, ventral view. f. Labium, dorsal view. g. Labium, ventral view. Scale bars = 0.025 mm.
Third instar larva
Head capsule (Fig. 18a-c): Frontale lacking secondary sensilla, specimen illustrated missing right seta FR8. Parietale with several secondary sensilla distributed as follows: dorsally with three short setae between pore PA6 and seta PA7, one very short seta mesal to PA8, another long seta between PA8 and PA9, one pore close to PA9, one short seta mesal to PA13 and one short seta posterior to pore PA15; ventrally with one short seta between distal to PA18, two short setae distal to PA16 and PA17.
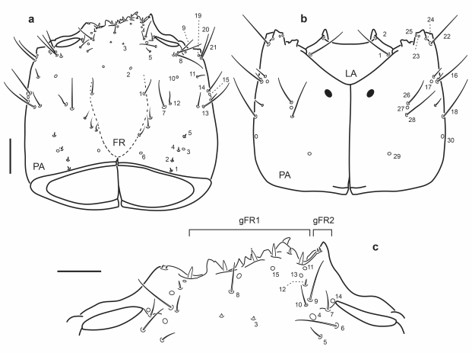
Fig. 18.
Chaetotaxy of third instar larva of E. (M.) pygmaeus. a. Head capsule, dorsal view. b. Head capsule, ventral view. c. Detail of clypeolabrum, dorsal view. Scale bars: a-b = 0.1 mm, c = 0.05 mm.
Antenna (Fig. 19a): Antennal sensorium (SE1) proportionally shorter than A3.
Mandibles (Fig. 19b-c): Several secondary sensilla distributed as follows: one minute seta between MN4 and MN1, one very short seta posterior to MN1, a group of three short basal setae close to outer face.
Maxilla (Fig. 19d-e): Stipes with two long secondary setae, one mesal to pore MX4, the other near pore MX2 on outer face; seta MX16 proportionally shorter, seta MX24 proportionally longer.
Labium (Fig. 19f-g): Mentum with twelve stout secondary setae dorsally, four on distal margin and four on each lateral face; ventrally with one short and slender seta on each anterolateral corner.
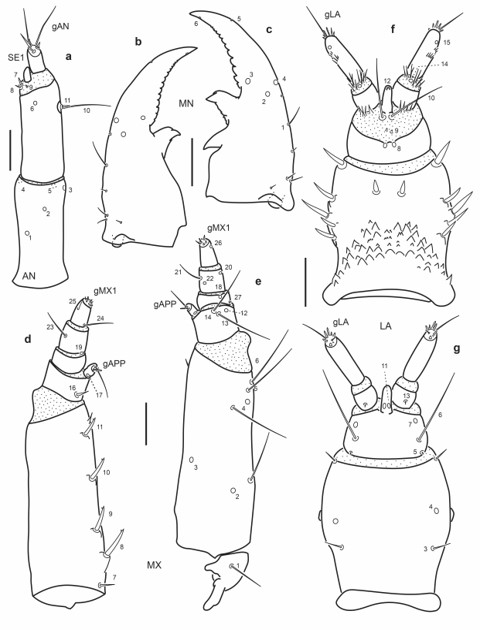
Fig. 19.
Chaetotaxy of third instar larva of E. (M.) pygmaeus. a. Left antenna, dorsal view. b. Left mandible, dorsal view. c. Right mandible, dorsal view. d. Left maxilla, dorsal view. e. Left maxilla, ventral view. f. Labium, dorsal view. g. Labium, ventral view. Scale bars = 0.05 mm.
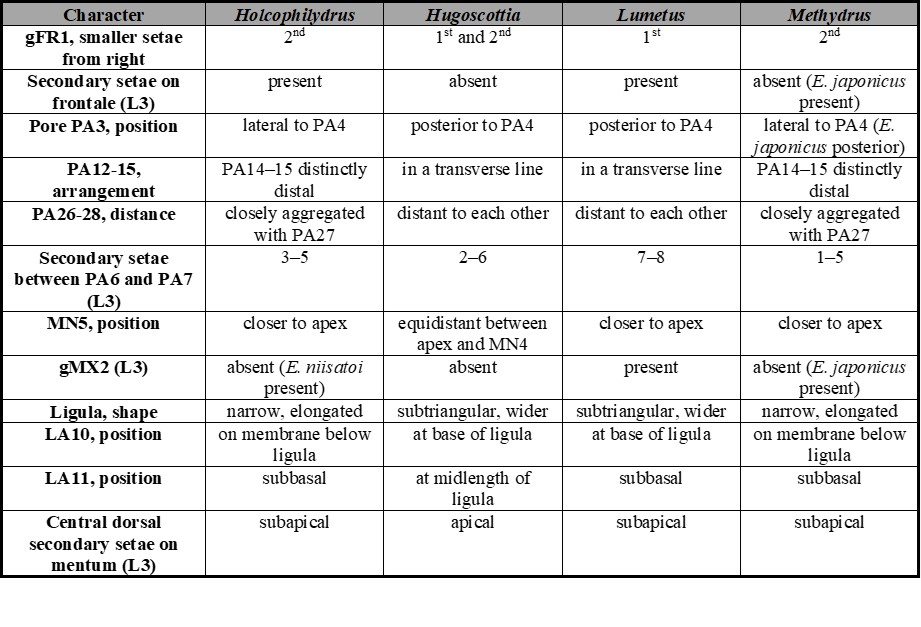
Table II.
Chaetotaxic and morphometric characters useful to distinguish among larvae of known subgenera of Enochrus.
DISCUSSION
Comparative notes among subgenera
A comparative study of the chaetotaxy among known species of Enochrus provides an important number of characters useful to distinguish among subgenera. Table II summarizes all chaetotaxic characters useful to tell apart larvae of the subgenera Holcophilydrus, Hugoscottia, Lumetus and Methydrus; the chaetotaxy of the subgenera Enochrus and Hydatotrephis remains unknown.
An additional, and useful morphological character not included in Table II, is the number of inner teeth on the left mandible, known Hugoscottia and Hydatotrephis larvae have two teeth while those of Holcophilydrus, Lumetus and Methydrus have only one (Archangelsky, 1997, 2002; Watts, 2002; Fikáček, 2006; Byttebier & Torres, 2009; Minoshima & Hayashi, 2011).
The shape of the nasale, which bears a number of small irregular teeth, was used to distinguish between species by different authors (Richmond, 1920; Wilson, 1923; Böving & Henriksen, 1938; Fernández, 1992; Archangelsky, 2002; Watts, 2002; Byttebier & Torres, 2009). However, the number of teeth on the nasale does not seem useful when comparing larvae of several species for the following reasons: in first place there is some overlap in the number of teeth among species, for example both E. (H.) variegatus and E. (M.) vulgaris have 6-7 teeth while E. (H.) tremolerasi has 6 (Archangelsky, 2002; Byttebier & Torres, 2009); secondly in some species the nasale does not have a definite number of teeth and appears as irregularly serrated (e.g. E. hamiltoni, Fig. 6c); and finally in many specimens the nasale is worn out and it is difficult to count the teeth.
The number of stout lateral setae on the mentum of third instar larvae was considered a good character to separate subgenera (Byttebier & Torres, 2009). However, after considering the new descriptions included in the present work, this character shows some degree of overlapping, making it less reliable: Holcophilydrus larvae have 4–5, Hugoscottia larvae have 3-4, Lumetus larvae have 4-6 and Methydrus larvae have 3-5 (Fikáček, 2006; Byttebier & Torres, 2009; Minoshima & Hayashi, 2011).
A special paragraph addressing the minute ventral setae of gFR1 should be made here. Two ventral minute setae in gFR1 were mentioned for E. (Hugoscottia. variegatus by Byttebier & Torres (2009) and after this mention they were not recorded for other species until now. Enochrus (Hugoscottia) tremolerasi has apparently only one (Figs. 2c and 4c), below the third dorsal tooth of gFR1 from the right; this seta was found in all instars. Enochrus (Lumetus) hamiltoni has two ventral setae on gFR1 (Fig. 6c). Minoshima (pers. com.) confirmed that larvae of E. (Holcophilydrus) simulans have at least one minute ventral seta on gFR1, and that larvae of E. (Methydrus) japonicus have two minute ventral setae. I have not been able to find them in the three Methydrus larvae described in this paper. Apparently due to the small size of these ventral setae and the irregular shape of the serrated nasale of Enochrus larvae, these setae are very difficult to visualize, therefore their presence cannot be ruled out.
Variation within subgenera
Larvae within the same subgenus are very similar to each other, nevertheless some characters show variability (Table II). One interesting finding is that larvae of E. (Methydrus) japonicus display several differences with other known Methydrus larvae. The frontale in third instar larvae of E. (M.) japonicus has one or two secondary setae distal to FR1 (Fig. 25A in Minoshima & Hayashi, 2011); all other known third instars of Methydrus lack secondary setae on the frontale. Additionally, gFR1 of E. (M.) japonicus has two minute ventral setae (Minoshima, pers. com.), these are apparently absent in other known Methydrus species. Pore PA3 in E. (M.) japonicus, is posterior to seta PA4, while in the remaining Methydrus species pore PA3 is located laterally to seta PA4. Third instar larvae of E. (M.) japonicus have secondary setae on the inner margin of the stipes, therefore conforming a distinctive gMX2 (Minoshima & Hayashi, 2011); these secondary setae are absent in all other known third instar Methydrus larvae. Secondary setae on the inner margin of the stipes (gMX2) are also found in third instar larvae of Enochrus (Lumetus) hamiltoni and E. (Holcophilydrus) satoi (Minoshima, 2017). Within Holcophilydrus, only third instar larvae of E. (H.) satoi have gMX2 on the stipes; third instars of two other known species, E. (H.) simulans and E. (H.) umbratus lack this character.
Possible diagnostic characters of Enochrus and Enochrinae
One interesting character that all described species of Enochrus share is the asymmetry in the distribution of sensilla FR11-13, already mentioned by Minoshima & Hayashi (2011). On the right side pore FR13 is located between pore FR11 and seta FR12 (e.g.Figs. 2c, 6c), whereas on the left side seta FR12 is positioned between pores FR11 and FR13 (e.g.Figs. 2c, 6c). This character seems unique within Enochrinae since it is absent in the other known larvae of this subfamily, Cymbiodyta and Notionotus (Perkins, 1972; Archangelsky, 1997; Fikáček, 2006; pers. obs.).
Another character that seems common to most described Enochrus larvae is the reduction in size of some of the right setae in gFR1 (e.g.Figs. 2c, 6c, 8c). In Enochrus (Hugoscottia) the first two setae of the right are smaller than the remaining ones; in E. (Lumetus) hamiltoni, the first seta on the right is smaller; in Enochrus (Methydrus) the second seta on the right is smaller. Apparently, Cymbiodyta and Notionotus show no reduction of setae on the right side of gFR1 (Perkins, 1972; Archangelsky, 1997; Fikáček, 2006; pers. obs.).
A character present in all known second and third instar larvae of Enochrus is a short ventral seta on each anterior corner of the mentum (e.g.Figs. 5g, 7g); this seta is also present in second and third instars of Cymbiodyta (pers. obs.); since Notionotus second and third instar larvae are unknown this character could not be evaluated for this genus. Finally, first instar larvae of all known Enochrus species bear a few small spicules on the ventral side of the second antennomere (Byttebier & Torres, 2009; Minoshima pers. com.). These spines are also present in first instars of Cymbiodyta and Notionotus larvae (Perkins, 1972; pers. obs.).
CONCLUSIONS
Even though no wide phylogenetic analysis of Enochrinae using molecular and/or adult characters has been performed, there is some evidence that the relationships within Enochrus are not completely resolved (see Fig. 3, Short & Fikáček, 2013). Enochrus larvae share several diagnostic characters that easily distinguish them from other genera within Enochrinae. Moreover, despite the great similarity they show, several morphological and chaetotaxic characters are useful to discriminate among larvae of the different subgenera. Regardless of this, some characters show variation within the same subgenus; this illustrates how important is the description of new larvae within genera that are supposed to be considerable well known (Rodriguez, 2021).
This study shows that Enochrus larvae display a great similarity in the chaetotaxy among species of the same subgenus, making it difficult to tell them apart; nevertheless, some species (e.g. E. (M.) japonicus) are quite distinctive within the subgenus and are easy to differentiate, therefore it is necessary to increase the number of larval descriptions in order to better understand this fact.
Description of the chaetotaxy and morphometry of larvae of the subgenera Enochrus (s. str.) and E. (Hydatotrephis) should be a priority with the aim to complete the larval knowledge of the genus. Knowledge of these larvae will give us a more complete picture of larval characters and will provide invaluable information for future phylogenetic analyses.
Acknowledgments
CONICET (Consejo Nacional de Investigaciones Científicas y Técnicas) is acknowledged for supporting systematic research. I am indebted to Yȗsuke Minoshima and Patricia L. M. Torres for their help checking some chaetotaxic characters in Enochrus larvae. The comments and suggestions of two anonymous reviewers and the Editors improved this contribution and are also acknowledged.
REFERENCES
Archangelsky, M. (1997) Studies on the biology, ecology, and systematics of the immature stages of New World Hydrophiloidea (Coleoptera: Staphyliniformia). Ohio Biology Survey Bulletin New Series, 12, 1-207.
Archangelsky, M. (1999) Immature stages of Paracymus rufocinctus Bruch, 1915 and Enochrus (Methydrus) vulgaris (Steinheil, 1869) (Coleoptera: Hydrophilidae: Hydrophilinae). Elytron,13, 87-99.
Archangelsky, M. (2002) Immature stages of Neotropical Enochrus (Coleoptera: Hydrophilidae): E. (Methydrus) lampros Knisch, 1924 and E. (Hugoscottia) tremolerasi Knisch, 1922. Aquatic Insects,24, 41-52.
Archangelsky, M. (2004) Higher-level phylogeny of Hydrophilinae (Coleoptera: Hydrophilidae) based on larval, pupal and adult characters. Systematic Entomology, 29, 188-214.
Archangelsky, M., Martínez-Román, N.R. & Fikáček, M. (2021) Larval chaetotaxy and morphology are highly homoplastic yet phylogenetically informative in Hydrobiusini water scavenger beetles (Coleoptera: Hydrophilidae). Zoological Journal of the Linnean Society,192, 416-452. https://doi.org/10.1093/zoolinnean/zlaa050
Böving, A.G. & Henriksen, K.L. (1938) The developmental stages of the Danish Hydrophilidae. Videnskabelige Meddelelser Dansk Naturhistorisk Forening102, 27-162.
Byttebier, B. & Torres, P.L.M. (2009) Description of the preimaginal stages of Enochrus (Hugoscottia) variegatus (Steinheil, 1869) and E. (Methydrus) vulgaris (Steinheil, 1869) (Coleoptera: Hydrophilidae), with emphasis on larval morphometry and chaetotaxy. Zootaxa,2139, 1-22.
Fernández, L.A. (1992) Descripción de la ooteca y estadios larvales de Enochrus (Hugoscottia) scutellaris (Bruch) y Enochrus (Methydrus) vulgaris (Steinheil) (Coleoptera, Hydrophilidae). Revista de la Sociedad Entomológica Argentina, 50, 343-352.
Fikáček, M. (2006) Primary chaetotaxy of the larval head of the hydrophiloid beetles (Coleoptera: Hydrophiloidea). Unpublished M.Sc. thesis, Department of Zoology, Faculty of Science, Charles University in Prague, Praha.
Fikáček, M., Archangelsky, M. & Torres, P.L.M. (2008) Primary chaetotaxy of the larval head capsule and head appendages of the Hydrophilidae (Coleoptera) based on larva of Hydrobius fuscipes (Linnaeus, 1758). Zootaxa,1874, 16-34.
Fikáček, M., Minoshima, Y., Vondráček, D., Gunter, N. & Leschen, R.A.B. (2013) Morphology of adults and larvae and integrative taxonomy of Gondwanan genera Tormus and Afrotormus (Coleoptera: Hydrophilidae). Acta Entomologica Musei Nationalis Pragae,53, 75-126.
Gundersen, R.W. (1967) Taxonomic revision of the genus Enochrus, subgenera Enochrusand Methydrus for the Nearctic region. (Hydrophilidae: Coleoptera). PhD thesis, University of Minnesota.
Hadley, A. (2010) Combine ZP 1.0. Available from https://combinezp.software.informer.com/ (accessed July 10, 2023)
Minoshima, Y.N. (2017). A new species of Enochrus Thomson from Laos (Coleoptera, Hydrophilidae). Special Bulletin of the Coleopterological Society of Japan, 1, 61-68.
Minoshima, Y. & Hayashi, M. (2011) Larval morphology of the Japanese species of the tribes Acidocerini, Hydrobiusini and Hydrophilini (Coleoptera: Hydrophilidae). Acta Entomologica Musei Nationalis Pragae, 51, 1-118.
Minoshima, Y., Hayashi, M., Kobayashi, N. & Yoshitomi, H. (2013) Larval morphology and phylogenetic position of Horelophopsis hanseni Satô et Yoshitomi (Coleoptera, Hydrophilidae, Horelophopsinae). Systematic Entomology, 38, 708-722.
Perkins, P.D. (1972) A study of the Hydraenidae and Hydrophilidae (Coleoptera) of the San Gabriel River, with emphasis on larval taxonomy. Unpublished M.Sc. thesis, California State University, Long Beach.
Richmond, E.A. (1920) Studies on the biology of aquatic Hydrophilidae. Bulletin of the American Museum of Natural History,42, 1-94.
Rodriguez, G. (2021) Sistemática filogenética de Hydrophilidae (Insecta: Coleoptera: Polyphaga) basada en el estudio del sistema sensorial de las larvas: morfología externa y ultraestructura, relaciones morfofuncionales y patrones de distribución de la quetotaxia. Unpublished Ph.D. thesis, Departamento de Biodiversidad y Biología Experimental, Facultad de Ciencias Exactas y Naturales, Universidad de Buenos Aires, Argentina.
Rodriguez, G., Fikáček, M., Yoshitomi, Y.N., Archangelsky, M. & Torres, P.L.M. (2021) Going underwater: multiple origins and functional morphology of piercing-sucking feeding and tracheal system adaptations in water scavenger beetle larvae (Coleoptera: Hydrophiloidea). Zoological Journal of the Linnean Society, 193, 1-30. https://doi.org/10.1093/zoolinnean/zlaa132
Short, A.E.Z. & Fikáček, M. (2011) World catalogue of the Hydrophiloidea (Coleoptera): additions and corrections II (2006– 2010). Acta Entomologica Musei Nationalis Pragae, 51, 83-122.
Short, A.E.Z. & Fikáček, M. (2013) Molecular phylogeny, evolution and classification of the Hydrophilidae (Coleoptera). Systematic Entomology, 38, 23-752.
Torres, P.L.M. (2008) Sistemática y Bionomía de Hydrophilina Argentinos (Coleoptera: Hydrophilidae). Unpublished Ph.D. thesis, Departamento de Biodiversidad y Biología Experimental, Facultad de Ciencias Exactas y Naturales, Universidad de Buenos Aires, Argentina.
Watts, C.H.S. (2002) The larvae of some Australian aquatic Hydrophilidae (Coleoptera: Insecta). Records of the South Australian Museum, 35, 105-138.
Wiley, E.O. (1981) Phylogenetics. The theory and practice of phylogenetic systematics. John Wiley & Sons, New York.
Wilson, C.B. (1923) Water beetles in relation to pondfish culture with life-histories of those found in fishponds at Fairport, Iowa. Bulletin of the Bureau of Fisheries, Washington,39, 231-345.
Notas de autor
hydrophilinae@gmail.com