Artículos
First sight at entomotoxicology using Peckia intermutans (Diptera: Sarcophagidae) in Ecuador and its potential as a marker for methanol determination in biological samples
Primer vistazo a la entomotoxicología utilizando Peckia intermutans (Diptera: Sarcophagidae) en Ecuador y su potencial como marcador para la determinación de metanol en muestras biológicas
First sight at entomotoxicology using Peckia intermutans (Diptera: Sarcophagidae) in Ecuador and its potential as a marker for methanol determination in biological samples
Revista de la Sociedad Entomológica Argentina, vol. 83, núm. 1, 2024
Sociedad Entomológica Argentina
Recepción: 18 Agosto 2023
Aprobación: 09 Febrero 2024
Abstract: Forensic entomotoxicology, which analyzes necrophagous insects to detect toxic substances, is crucial in determining causes of death and estimating the post-mortem interval. This study focused on Peckia intermutans (Walker) (Diptera: Sarcophagidae) as a marker for detecting methanol, a potentially lethal alcohol often added as an adulterant in alcoholic beverages due to its low cost and similarity to ethanol. A gas chromatography-mass spectrometry (HS-GC-MS) method was developed to detect methanol, and its effects on the development time, survival, and morphology of flesh flies were investigated. Larvae were reared on liver substrates enriched with methanol (300, 500, and 1000 mg/kg). Results showed: the developed method detected methanol in all instars; methanol affected the developmental time of the flies; survival increased with higher methanol concentration; and morphological changes were evident in weight. This study provides valuable information on entomotoxicological research in Ecuador, emphasizing the need to consider morphological changes in P. intermutans to avoid inaccurate estimations of post-mortem intervals.
Keywords: Entomology, Gas chromatography-mass spectrometry, Morphology, Necrophagous, Toxicology.
Resumen: La entomotoxicología forense, que analiza insectos necrófagos para detectar sustancias tóxicas, es crucial en la determinación de causas de muerte y la estimación del intervalo post-mortem. Este estudio se centró en Peckia intermutans (Walker) (Diptera: Sarcophagidae) como marcador para la detección de metanol, un alcohol potencialmente letal presente como adulterante en bebidas alcohólicas debido a su bajo costo y parecido con el etanol. Se desarrolló un método de cromatografía de gases-espectrometría de masas (HS-GC-MS) para detectar metanol y se estudió su efecto sobre el tiempo de desarrollo, supervivencia y morfología de las moscas de la carne. Las larvas se criaron en sustratos de hígado enriquecidos con metanol (300, 500 y 1000 mg/kg). Los resultados mostraron: el método desarrollado detectó metanol en todos los estadios; el metanol afectó el tiempo de desarrollo de las moscas; la supervivencia aumentó con una mayor concentración de metanol; y los cambios morfológicos fueron evidentes en el peso. Este estudio ofrece valiosa información sobre la investigación entomotoxicológica del Ecuador, por lo que se debe considerar los cambios morfológicos de P. intermutans para evitar estimaciones inexactas de los intervalos post-mortem.
Palabras clave: Cromatografía de Gases-Espectrometría de Masas, Entomología, Morfología, Necrófagos, Toxicología.
INTRODUCTION
Flesh flies (Diptera: Sarcophagidae) encompass around 3100 species, with some having a global distribution (Pape et al., 2011), and approximately six hundred twenty-two species in the Neotropical Region (Amorin et al., 2002). This family holds significant importance in forensic entomology due to their scavenging behavior on decaying matter. Nevertheless, their potential is constrained by limited taxonomic knowledge and insufficient research on their development in various geographical areas (Buenaventura & Pape, 2013).
Peckia Robineau-Desvoidy,1830, a genus exclusive to the New World, encompasses 67 identified species, with some introduced in Oceania (Buenaventura & Pape, 2013). Several Peckia species thrive in both urban and natural environments, including coastal areas where they breed in vertebrate excrement and carrion. These species are considered crucial forensic indicators because, unlike other flies, Sarcophagidae eggs hatch in the uterus of the female (Garcés et al., 2020). Among them isPeckia (Pattonella) intermutans (Walker, 1861), distributed from Mexico to Paraguay, with documented occurrences in Ecuador’s Napo, Orellana, and Imbabura provinces (Buenaventura & Pape, 2013; Salazar & Donoso, 2015). Prior research has linked this species to human corpses and forensic biomodels, such as pigs in Brazil (Carvalho et al., 2000; Barros et al., 2008).
Global fatalities from drug abuse, pesticides, and toxins highlight the need for effective forensic tools (Nekoukar et al., 2021). Insects, equipped with a remarkable ability to detect volatile organic compounds (VOCs) during decomposition (Martin et al., 2019), play a vital role in entomotoxicology forensics. Specially, the empty puparium (EP) of insects is used because of their high chitin content. It provides a protective barrier, helping to preserve any residual substances or toxic compounds that may be present on or within the puparium, also EP's long lifetime in the environment provides reliable data for entomotoxicological analyses (Chophi et al., 2019;Aly & Aldeyarbi, 2020)
Utilizing insects as indirect indicators of toxic substances in human remains, specifically drugs and other toxins that may have been present prior to death, is the primary objective of entomotoxicology forensics (Da Silva et al., 2017). Methanol, a VOC released during decomposition, raises concerns, particularly in cases of methanol intoxication resulting from adulterated alcoholic beverages.
Methanol poses a significant risk when present in excessive amounts, often in adulterated alcoholic beverages due to inadequate regulatory controls in low-income countries (Costa & Aschner, 2014). This illicit practice, driven by cost-effectiveness compared to ethanol, has led to methanol-related deaths (PHE, 2015).
The impact of alcohols on necrophagous insects has prompted the exploration of innovative methodologies, such as using insects as toxicological samples. While insects can detect methanol through their chemosensory systems, their attraction to decomposing bodies primarily arises from organic compounds, rather than methanol itself. Post-mortem, methanol accumulation occurs as the elimination mechanisms cease, affecting various tissues (Wu Chen et al., 1985; Andresen et al., 2008). Flies may be exposed to methanol through ingestion, exhalation, evaporation, and decomposition products.
In Ecuador, studies in forensic entomology are limited and primarily focused on faunistic knowledge, although they have been increasingly used as evidence in criminal investigations and autopsies involving human remains (Andresen et al., 2008; García-Ruilova et al., 2020; Salazar & Donoso, 2015). However, to the best of our knowledge, there are currently no records of toxicological analysis of insect evidence in the country.
Due to the importance of Peckia species in forensic entomology, this study aimed to determine the potential of P. intermutans in entomotoxicology as a marker for detecting methanol on experimental substrates using gas chromatography-mass spectrometry (GC-MS) techniques. This pioneering effort in Ecuador seeks to establish an analytical technique for the effective identification of methanol in all stages of P. intermutans using GC-MS. Additionally, the study investigates the impact of methanol exposure on the development time, survival rate, and physical characteristics (length and weight) of P. intermutans. Through these efforts, the study contributes to advancing the field of entomotoxicology and emphasizes the crucial role of Peckia intermutans in forensic investigations involving toxic substances.
MATERIAL AND METHODS
Sample selection and identification
Adult wild flies were collected according to “Ministerio del Ambiente, Agua y Transición Ecológica” (MAATE) and the “autorización de recolección sin fines comerciales” (ARSFC) permission (MAATE-ARSFC-2023-3006) using bottle traps containing beef offal. Six bottle traps were hung on tree branches, in the surroundings of Ikiam University, Tena, Ecuador (0°57′01″S, 77°51′46″O). Each bottle trap contained 100 g of beef viscera, with damp paper at the base to prevent dehydration of the viscera. The traps were suspended at a height of 1.5 meters. Peckia (Pattonella) intermutans was identified using the keys provided by (Carvalho & Mello-Patiu, 2008; Vairo et al., 2011; Buenaventura & Pape, 2013; De Camargo et al., 2018). Adults were placed in 900 cm. containers and 15 days replenished to prevent inbreeding. The 900 cm. containers consist of wire cages covered with a fine mesh, with an opening used to feed the samples. P. intermutans species used in this experiment were harvested from a third generation.
Establishment of the colony and rearing of P. intermutans
Twelve hatcheries (900 cm. containers) were used to get the first generation (F1) of P. intermutans. Each hatcheries contained 10 adult flies (5 males and 5 females), and for 5 days, adults were supplied with water and sugar ad libitum. On day 6, each fly container was provided with a small plastic tray (23.8 cm x 18.7 cm x 12.3 cm) containing 200 g of fresh beef liver on water-moistened paper, allowing females to deposit larvae directly on the liver. The liver was checked every 8 h. When larviposition was observed, to identify the new generation, the larvae deposited by P. intermutans were removed using a fine paintbrush and transferred to a new hatchery with a plastic tray containing 3 cm of moist soil (substrate for the pupation) and beef liver. The larvae were placed directly on the liver. When larvae emerged as adults, the first generation (F1) was collected. F1 individuals were transferred to another container. Procedures described to get F1 were replicated for the second (F2) and third (F3) generations.
To closely resemble real conditions, the samples were exposed to the natural conditions of the city. To record the temperature and humidity in which the samples were bred, a thermo-hygrometer was used, and the temperature and relative humidity were recorded three times a day. The results were averaged, revealing that P. intermutans were reared at a temperature of 27±2 °C, a relative humidity of 73±6% and the photoperiod (h) was 12:12 (L: D).
Preparation of foodstuff and rearing of flesh flies
Four experimental samples were prepared. For each sample, 200 g of ground beef liver was used, and using a spatula was homogenized with methanol. Concentrations of 300, 500, and 1000 mg/kg of methanol, referred to as M1, M2, and M3, respectively, were added. Another liver substrate containing no methanol was used as a control (M0).
Each experimental sample (M0, M1, M2, and M3) was placed in airtight containers. The samples were allowed to rest for 24 hours at 4 °C in a refrigerator. Later, livers were placed on water-moistened paper to avoid desiccation, inside a plastic tray containing 3 cm of moist soil. To keep the paper moist, water was added twice a day using a dropper. Each tray was placed inside cages covered with a fine cloth. The larvae obtained from F3 were removed with a paintbrush and placed in samples M0, M1, M2, and M3. In each substrate, 120 larvae were added, all in the first larval stage.
For the preparation of foodstuff and rearing of flesh flies, the amount of methanol was based on the largest permissible limits of alcoholic degree established by NTE INEN 362:2014 (Instituto Ecuatoriano de Normalización, 2014). The lethal dose is estimated at 300-1000 mg/kg (PHE, 2015).
In natural decomposition scenarios, direct access to a decedent's liver by insects is unlikely, especially in the absence of trauma. Insects typically access the decomposing tissues through natural orifices, wounds, or openings in the body. However, entomotoxicology uses viscera as a standardized substrate to study the interactions between insects and toxic substances in a controlled environment. Liver was used because is the typical medium for forensic entomology experiments and is the major site of metabolism and storage for various substances and xenobiotics in the body (Gosselin et al., 2011; Da Silva et al., 2017; Chophi et al., 2019; Aly & Aldeyarbi, 2020).
Sample collection
Two sets of samples, from each treatment at each life stage were collected. One consisting of 10 individuals and another amounting to 1 g. Flesh flies were collected when they reached second (L2), third (L3), post-feeding (PF), pupal (P), and adult (A) instars (Amendt et al., 2007; Magni et al., 2018). Empty puparium (EP) was also collected. The liver was checked every 8 h.
The samples of 10 individuals of each treatment were used for morphological analyses (length and weight). The specimens were collected using metallic forceps and preserved following guidelines for forensic entomology (Amendt et al., 2007). The larvae (L2, L3, PF) and pupa from each stage were sacrificed in hot water (>80 °C) for 30 s and then stored in 70% ethanol at 4 °C until the time of measurement and weighing. Adults were placed in plastic containers and sacrificed 2 days after emergence by placing them in the freezer (-80 °C) for one hour. Once dead, they were stored in 70% ethanol at 4 °C.
Following preservation, a stereomicroscope (AmScope SM-1TSL-64S-V331) was used to determine the stage of P. intermutans based on taxonomic descriptions (Fig. 1) (Carvalho & Mello-Patiu, 2008; Buenaventura et al., 2009; Vairo et al., 2011; Buenaventura & Pape, 2013). For the P. intermutans identification, the recognition of larval stages was based on the number of spiracles. Two spiracles for the second larval instar and three for the third larval instar. Regarding adults, dichotomous keys mentioned in the literature were followed, identification based on male genitals, veins in the wings, and antennae. Besides, the length and weight of each instar were recorded. The length of each individual was measured using a stereomicroscope (AmScope SM-1TSL-64S-V331) equipped with a 24 px camera and measurement software. An analytical balance was used for weight.
The 1 g samples were used for toxicological analysis by headspace gas chromatography-mass spectrometry (HS-GC-MS). After cleaning with deionized water to drop external contamination, samples were stored at -80 °C (Li & Tennessen, 2018).
The analytical method validation was performed using 1 g of control Empty puparium. Empty puparium was chosen because of its high chitin content, also EP's long lifetime in the environment provides reliable data for entomotoxicological analyses (Magni et al., 2014, 2016, 2018).
To consider the methanol effects on the life cycle and survival of P. intermutans, 100 individuals in PF instar from each treatment were placed in separate boxes. The time to pupation, the total number of individuals pupating and emerging adults were recorded.
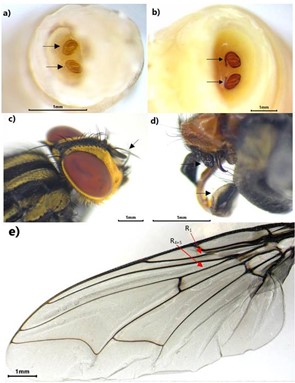
Fig. 1.
Peckia intermutans immature and adult characters. a. Second instar larvae showing posterior spiracles. b. Third instar larvae showing posterior spiracles. c. Adult showing antenna arista with long plumosity. d. Male genialia structures showing surstylus round. e. Wing venation: vein R1 and R4+5 bare dorsally.
Toxicological Analysis
Chemicals and reagents - Methanol (HPLC grade) and sodium chloride 99.9% (NaCl) were purchased by J.T Baker (USA). Deionized water for preparation of all the solution was purified (18.2 μΩ·cm) using Smart2 Pure of Thermo Scientific.
Sample preparation for HS-GC-MS analysis - 1 g of P. intermutans samples (L2, L3, PF, P, EP, and A) were placed separately in 15 ml falcon tubes, and 1 ml of ice-cold 0.9 % NaCl was added as part of the preliminary wash (Li & Tennessen, 2018). The tubes with samples were placed in a vortex for 2 min and the solvent was discarded. Empty puparia and adults were dried at room temperature under nitrogen. Frozen samples (liquid N.) were crushed using a mortar and pestle and placed in a 20 ml glass vial (Vial-20-ND18-CG-100) (Fig. 2). Pupa, empty puparia, and adults were diluted 1:4 with deionized water (Chun et al., 2016).
The methanol detection method was validated using 1 g of control P. intermutans. For the toxicological analysis, the whole specimen was used.
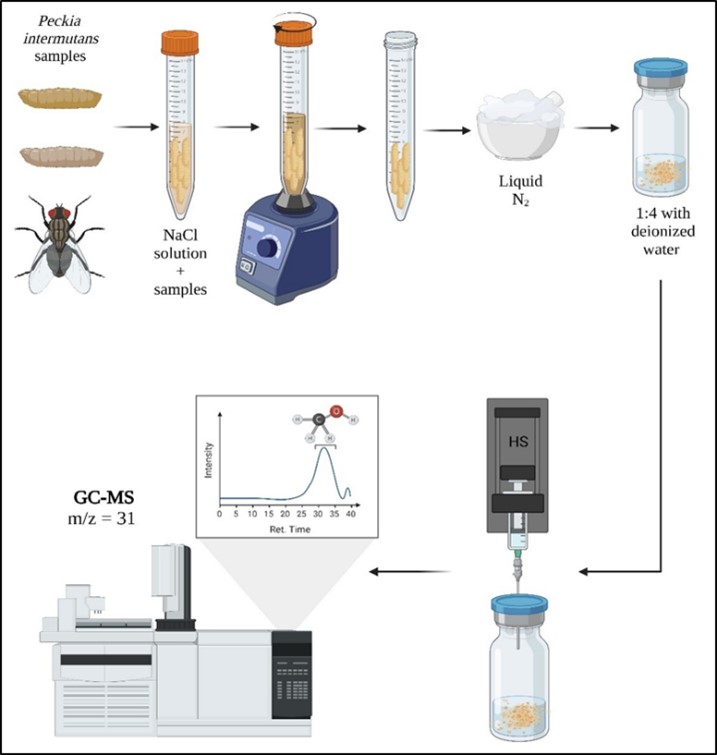
Fig. 2.
Peckia intermutans samples preparation for toxicological analysis in HS-GC-MS. One gram of P. intermutans samples (L2, L3, PF, P, EP, and A) is placed in Falcon tubes, washed with NaCl, and vortexed. Once the samples are clean, they are macerated, transferred to a vial with deionized water, and subjected to HS-GC-MS.
Headspace conditions - The automated headspace autosampler AOC-6000 (Switzerland) was coupled with the GC-MS for sample preparation and introduction into the GC. The samples were automatically shaken and incubated for 5 min at 80 °C. Then, 300 µL headspace aliquot was sampled for the analysis with a fill speed of 35 mL/min. To obtain homogenous sampling, a fill stroke count of 3 was used. The sample was injected into the GC at an injection speed of 25 mL/min. After each analysis, the syringe was flushed with nitrogen for 5 min. The run cycle time was set at 15 min.
GC-MS analysis - Analytical determinations for the detection of methanol were performed using a gas chromatograph coupled with a mass spectrometer Shimadzu GCMS-QP2020NX fitted with a split/splitless injector and equipped with a capillary column Rtx-50 (50% methylphenylsiloxane) 30 m x 0.25 mm i.d. x 0.25 μm df. The oven temperature was programmed as follows: initial column temperature was 45 °C for 5 min, then increased by 10 °C/min to 80 °C for 1 min for a total run time of 9.5 min. The injection port and transfer line were kept at T = 200 °C using helium ultrapure as carrier gas at an initial flow of 1 mL/min. Electron ionization mode and full mass spectra were acquired to obtain a comprehensive view of the sample composition, confirming the presence of methanol through its mass spectrum. Additionally, this mode facilitates the identification of potential contaminants or impurities within the working range of 20 to 80 Da. Next, a selected ion monitoring (SIM) analysis was conducted to improve sensitivity and selectivity in the quantitative analysis, using the ion m/z 31 characteristic of methanol. The background-subtracted mass spectrum for methanol (using EI in full scan mode) is given in Fig. 3. The peak identification was performed using NIST 2020 (Wiley Registry 12th Edition/NIST 20). All the analyses were performed in triplicate.
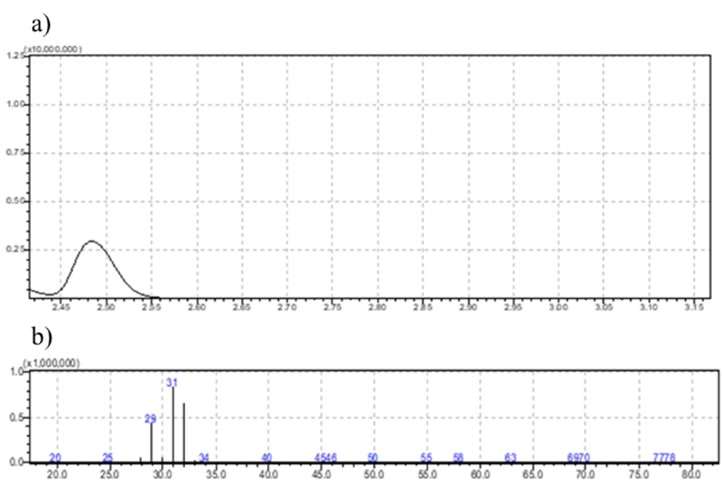
Fig. 3.
Chromatogram of methanol obtained with electronic impact (EI) ionization.
Method validation - The following parameters were obtained: coefficient of linearity (R2), specificity, limit of detection (LOD), limit of quantification (LOQ), extraction recovery (ER %), repeatability and carryover (Chun et al., 2016; Magni et al., 2016, 2018). The validation included the quantitative determination of methanol in larvae, P, EP and A.
Specificity - A total of ten samples of the control EP were used to ascertain the specificity of the method, of which five were fortified with 126 μl of methanol. Substances with similar characteristics, such as ethanol and isopropanol, were evaluated for specificity analysis.
Linearity - Calibration models were determined from five-point calibration curves with calibrators prepared in triplicate in homogenized EP. The linear calibration model was checked by analyzing control EP samples (1 g) fortified with methanol at concentrations of 300, 400, 500, 700 and 100 mg/kg. The linear calibration parameters were calculated by least-squares regression, and the squared correlation coefficient (R2) was used to estimate linearity.
LOD and LOQ - The LOD and LOQ were administratively set at and verified with the lowest calibrator prepared in triplicate in three lots of EP homogenates. For the limit of detection, the lowest concentration of methanol found in corpses was used. A relationship was established between the theoretical quantity and the lowest amount found in samples contaminated with methanol.
Extraction recovery – ER % was evaluated using three methanol concentrations in control EP: 50, 100 and 200 mg/mg. ER % was calculated by the average ratio between the analyte concentration determined after its extraction (first set) and the one determined on the fortified extract (second set).
Repeatability - Repeatability was calculated as the percent coefficient of variance (CV %), after fortifying ten samples of control EP with two methanol concentrations: 350, 600 and 900 mg/kg. Repeatability was considered acceptable when the CV % is lower than 25 % at low analyte concentrations and lower than 15 % at high concentrations (Magni et al., 2014, 2016, 2018).
Carry Over - Carry-over effect was evaluated by injecting an alternate sequence of five negative EP samples and ten blank EP samples fortified with methanol at 1000 mg/kg concentration to ensure the absence of any carry-over effect. The sequence was: blank, methanol, blank, methanol, methanol, blank, methanol, methanol, methanol, blank, methanol, methanol, methanol, methanol, blank.
Statistical Analysis
Concentrations of methanol in P. intermutans and their remains, as well as their respective lengths and weights in different treatments, were analyzed by one-way analysis of variance (ANOVA) and Tukey test. Pupation and eclosion rates were analyzed by one-way ANOVA and Pearson’s chi-squared test. The level of statistical significance was set at p < 0.05. Calculations were done using Microsoft Excel and IBM SPSS Statistics 29 software package.
RESULTS
Method validation
The validation of methanol involved measuring various parameters, including the coefficient of linearity (R.), limit of detection (LOD), limit of quantification (LOQ), ER %, and repeatability (CV %). Table I presents a summary of these validation parameters, indicating that specificity was adequate and that there were no carry-over effects.
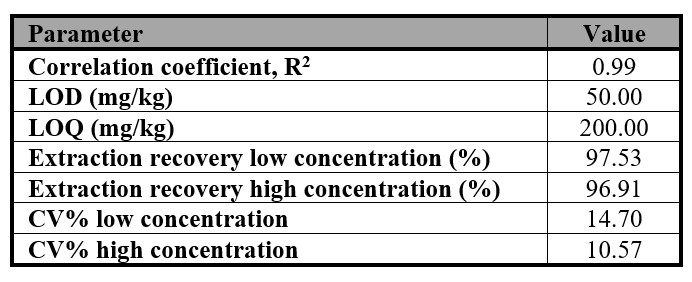
Table I.
Validation parameters calculated for methanol.
LOD = detection limit; LOQ = limit of quantification
Methanol concentration
According to the results of the GC-MS analysis, it was found that in the PF stage of both M1 and M2 treatments, methanol could not be measured. As for the M3 treatment, the amount of methanol detected was below the limit of detection (LOD), as indicated in Table II. However, in other stages of P. intermutans, such as L2, L3, P, and A, methanol was quantifiable. Additionally, it is reflected that in the P, EP, and A stages, the concentration of methanol gradually increases, with M3 showing the highest concentrations. As the concentration increases in each treatment, namely from M1 to M3, the detected amount decreases as the stage progresses (from P to A). On the other hand, in the M0 treatment, methanol was detected in different development stages of P. intermutans.
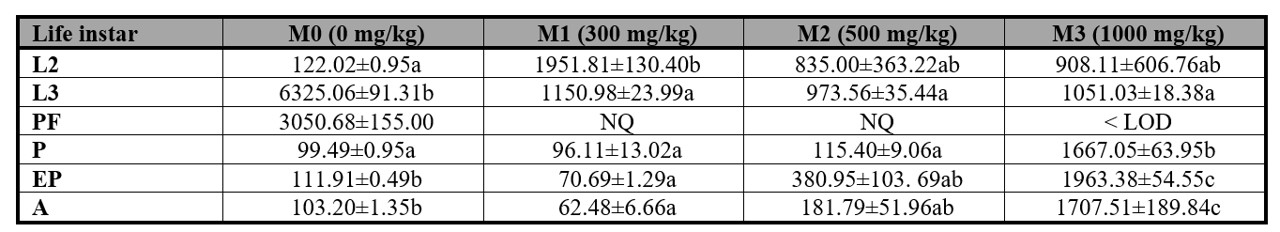
Table II.
Methanol quantification (mg/kg ± SE) in Peckia intermutans by HS-GC-MS analysis.
Quantification was calculated using three replicates. Means followed with different letter are statistically different (p < 0.05), Tukey test. LOD = 50 mg/kg. NQ = not quantifiable. L2 = second instar, L3 = third instar, PF = post-feeding instar, P = pupa instar, EP = empty puparium, A = adult instar
Developmental time and survival
The survival and developmental time of P. intermutans were significantly impacted by the existence of methanol. As per Table III, larvae that consumed liver with methanol exhibited a shorter period between larviposition and eclosion compared to the control group.
As shown in Table III, during the PF stage, the mortality rate of larvae before pupation was 10/100 for the 0 mg/kg treatment, 9/100 for the 300 mg/kg treatment, 9/100 for the 500 mg/kg treatment, and 10/100 for the 1000 mg/kg treatment. Following metamorphosis, a lower number of adult flies emerged in the M0 treatment (55/100) in comparison to the M3 treatment (88/100). These findings indicate that the consumption of liver containing methanol can significantly impact the survival of P. intermutans, particularly during the metamorphosis stage.
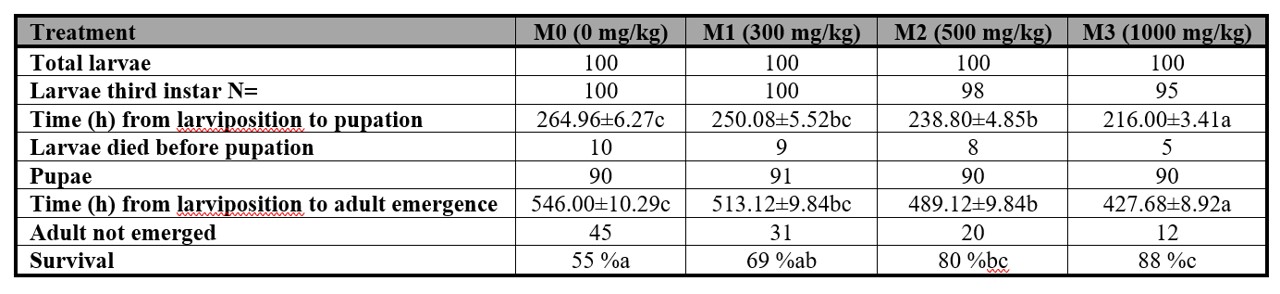
Table III.
Time (h mean ± S.E.) from larviposition to pupation and from larviposition to adult emergence of Peckia intermutans larvae, which were exposed to either liver containing different amounts of methanol and control.
The table shows the number of larvae dead prior to pupation, the number of not emerged adults, and the survivors. Means followed with different letter are statistically different (p < 0.05), Tukey test.
Peckia intermutans length and weight
The presence of methanol had a significant impact only on the length of pupae reared in the M3 treatment, which were larger than those in the M0, M1, and M2 treatments. However, no significant differences were observed in the mean length of larvae or adults. The effects of methanol on the length of P. intermutans larvae, pupae, and adults are summarized in Table IV.
Furthermore, the weight of P. intermutans adults was found to differ significantly in samples that consumed liver with methanol (Table V). Adult flies in the M1, M2, and M3 treatments exhibited greater weight (mg) than those in the control group. Additionally, significant differences were observed in the weight of L3 larvae that consumed liver containing 1000 mg/kg methanol, as compared to the M1, M2, and control groups.
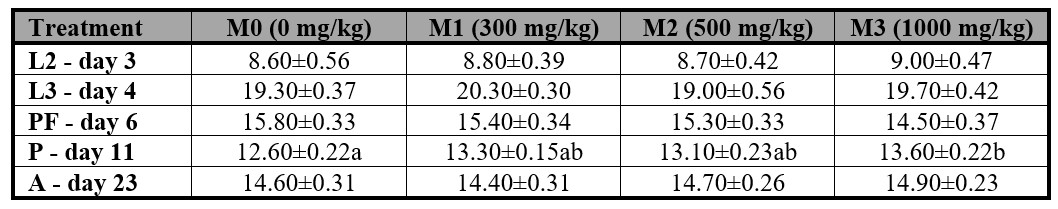
Table IV.
Peckia intermutans mean (± S.E.) larvae, pupae, and adult length (mm) for each methanol concentration at the different instars and stages.
L2 = second instar, L3 = third instar, PF = post-feeding instar, P = pupa instar, A= adult instar. Means followed with different letter are statistically different (p < 0.05), Tukey test.
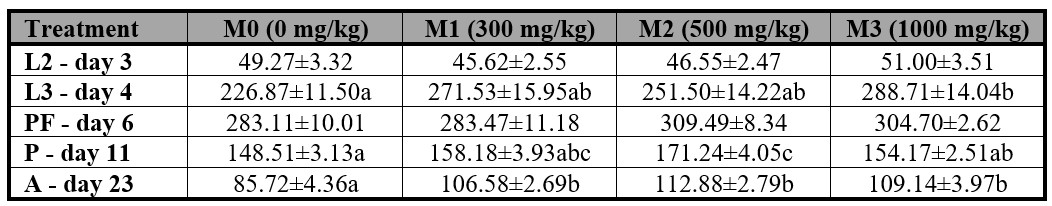
Table V.
Mean weight (mg ± S.E.) of Peckia intermutans larvae, pupae, and adult related to the time of exposure (day) and instar of life.
L2 = second instar, L3 = third instar, PF = post-feeding instar, P = pupa instar, A= adult instar. Means followed with different letter are statistically different (p < 0.05), Tukey test.
DISCUSSION
Methanol, commonly known as methyl alcohol, is a potent poison frequently employed in the production of counterfeit alcoholic beverages (Costa & Aschner, 2014). The rapid absorption of methanol by body fluids and its accumulation in biological compartments, such as urine, blood, and vitreous humor, make these samples critical in forensic post-mortem alcohol analysis (Andresen et al., 2008). In this regard, insects can be a useful alternative to conventional biological samples for post-mortem alcohol analysis in forensic investigations (Tabor et al., 2005). This study demonstrated the potential of P. intermutans as a valuable alternative to conventional biological samples for investigating the effects of methanol. Particularly, considering that the determination of methanol can be challenging due to the sample matrix, the analytical method underwent successful validation using recovery percentages, simulated by mimicking the studied matrix with empty puparium (SOFT / AAFS (Society of Forensic Toxicologists, & American Academy of Forensic Sciences), 2006; Bursová et al., 2015; Chun et al., 2016). These results consistently hovered around 96%, signifying the insignificance of the matrix effect for this type of analysis. This discovery emphasizes the resilience of our methodology and affirms the reliability of the obtained results, even under conditions that could present analytical challenges. It serves as a demonstration of the method’s robustness in methanol determination.
The findings suggest that methanol affects the developmental time, survival, and morphology of P. intermutans, with higher concentrations leading to increased survival rates. Intriguingly, the experimentally placed methanol concentrations were surpassed by the actual levels found in the samples, sparking discussion on the potential reasons behind these outcomes.
While existing research lacks information on methanol's effects on flesh flies, studies using HS-SPME-GC-MS have detected volatile compounds in various arthropods (Chen, 2017). The study reveals that methanol concentrations in the liver influence its detection in different instars, showing variations in response to treatments. Additionally, methanol was detected in larvae, pupa, and adults of P. intermutans that consumed the control sample (M0), raising questions about endogenous metabolic processes leading to post-mortem alcohol formation. The presence of methanol in the control samples (M0) is attributed to the formation of alcohol during the post-mortem stages (Chun et al., 2016). The production of methanol, especially in the initial hours, results in an increase in methanol concentrations in the samples (Table II). Therefore, the detected amount in the samples was higher than the initially introduced amount (M1, M2, M3).
Considering the detectable concentrations of methanol, and the survival and development time of P. intermutans, it is possible to speculate that, given that methanol is a byproduct formed during corpse degradation, necrophagous insects have evolved a tolerance system to alcohols, making them easily metabolized. Malpighian tubules represent one of the main detoxification mechanisms in insects, especially during their larval stage (Pal & Kumar, 2012). Early exposure to the xenobiotic triggers detoxification mechanisms, leading to increased survival at higher methanol concentrations (Table III).
The research suggests that P. intermutans possesses effective mechanisms to excrete methanol, allowing them to develop and survive despite its presence (Pal & Kumar, 2012). The decreasing concentration of methanol in the final stage of the fly's life cycle (adults), with the highest levels detected in adults fed with M3, indicates the larvae's early activation of detoxification mechanisms.
Previous studies have highlighted insects' capability to efficiently excrete drugs, with Malpighian tubules serving as essential organs for metabolism and detoxification. Observations on Sarcophagidae suggest the involvement of peroxisomes in the Malpighian tubules in eliminating toxic molecules (Pal & Kumar, 2012; Nauen et al., 2021).
The experimental results underscore the impact of methanol on the survival and development time of P. intermutans. Higher concentrations of exogenous methanol result in more significant effects, as illustrated in Table III. Changes in developmental time can affect PMI accuracy, and the study notes that P. intermutans reared on food containing methanol had a shorter development time compared to those reared-on ethanol-spiked food (Tabor et al., 2005). This becomes crucial in forensic investigations, where insects found on corpses play a vital role in estimating the post-mortem interval (PMI).
The results shed light on the complexity of methanol's impact on fly larvae, highlighting their adaptability and tolerance to potentially toxic substances. The discrepancies between experimentally placed and detected methanol concentrations open avenues for further research, emphasizing the need for a comprehensive understanding of the mechanisms governing methanol-related processes in flesh flies. This research holds implications for forensic investigations, urging examiners to consider various factors when interpreting evidence obtained from insect specimens found on corpses, ultimately contributing to the advancement of forensic entomology.
CONCLUSIONS
The global concern of methanol adulteration in alcoholic beverages, particularly in low- and middle-income countries with inadequate regulations, underscores a significant public health challenge. In the current study, P. intermutans emerges as a potential biomarker for methanol detection in biological samples, utilizing the valuable HS-GC-MS technique. The research successfully detected methanol in all stages of P. intermutans reared on varied methanol concentrations, revealing adverse effects on morphology, survival, and development time, extending to offspring consuming the contaminated substrate.
This study contributes significantly to entomotoxicology knowledge in Ecuador, highlighting the necessity for further investigations into the impact of methanol on flesh flies. Future research avenues include exploring the impact of alcoholic beverage-related deaths on flesh fly development, considering both immediate and subsequent generations, and its implications for minimum post-mortem interval estimation. Additionally, investigating higher methanol doses in other fly genera, understanding resistance mechanisms and oviposition patterns, examining the interaction of methanol with other drugs in flesh flies, quantifying post-mortem endogenous alcohol formation to distinguish ante-mortem ingestion from post-mortemproduction, and studying the effects of methanol on corpse degradation are recommended.
REFERENCES
Aly, S.M., & Aldeyarbi, H. (2020) Zastosowanie entomologii sądowej: przegląd stanu wiedzy i najnowsze doniesienia. Archiwum Medycyny Sadowej i Kryminologii, 70(1), 44-77. https://doi.org/10.5114/amsik.2020.97832
Amendt, J., Campobasso, C.P., Gaudry, E., Reiter, C., LeBlanc, H.N., & Hall, M.J.R. (2007) Best practice in forensic entomology - Standards and guidelines. International Journal of Legal Medicine, 121(2), 90-104. https://doi.org/10.1007/s00414-006-0086-x
Amorin, D. de S., Silva, V.C., & Balbi, M.I.P.A. (2002). Estado do conhecimento dos diptera neotropicais. Sociedad Entomológica Aragonesa - SEA, Monografías Tercer Milenio, 2, 29-36. http://sea-entomologia.org/PDF/M3M_PRIBES_2002/029_036_Dalton.pdf
Andresen, H., Schmoldt, H., Matschke, J., Flachskampf, F.A., & Turk, E.E. (2008) Fatal methanol intoxication with different survival times-Morphological findings and postmortem methanol distribution. Forensic Science International, 179(2-3), 206-210. https://doi.org/10.1016/j.forsciint.2008.05.014
Barros, R.M., Mello-Patiu, C.A., & Pujol-Luz, J.R. (2008) Sarcophagidae (Insecta, Diptera) associados à decomposição de carcaças de Sus scrofa Linnaeus (Suidae) em área de Cerrado do Distrito Federal, Brasil. Revista Brasileira de Entomologia, 52(4), 606-609. https://doi.org/10.1590/S0085-56262008000400011
Buenaventura, E., Camacho, G., García, A., & Wolff, M. (2009) Sarcophagidae (Diptera) de importancia forense en Colombia: claves taxonómicas, notas sobre su biología y distribución Sarcophagidae (Diptera) of forensic importance in Colombia: taxonomic keys, notes on biology, and distribution. Revista Colombiana de Entomología, 35(2), 189-196. https://doi.org/10.25100/socolen.v35i2.9216
Buenaventura, E., & Pape, T. (2013) Revision of the New World genus Peckia Robineau-Desvoidy (Diptera: Sarcophagidae). Zootaxa, 3622(1), 1-87. https://doi.org/10.11646/zootaxa.3622.1.1
Bursová, M., Hložek, T., & Čabala, R. (2015) Simultaneous determination of methanol, ethanol and formic acid in serum and urine by Headspace GC-FID. Journal of Analytical Toxicology, 39(9), 741-745. https://doi.org/10.1093/jat/bkv075
Carvalho, C.J., & Mello-Patiu, C. (2008) Key to the adults of the most common forensic species of Dipterain South America. Revista Brasileira de Entomologia, 52(3), 390-406. https://doi.org/10.1590/S0085-56262008000300012
Carvalho, L., Thyssen, P.J., Linhares, A.X., & Palhares, F. (2000) A Checklist of Arthropods Associated with Pig Carrion and Human Corpses in Southeastern Brazil. Mem Inst Oswaldo Cruz, 95(1), 135-138. https://doi.org/10.1590/S0074-02762000000100023
Chen, J. (2017) Freeze-Thaw Sample Preparation Method Improves Detection of Volatile Compounds in Insects Using Headspace Solid-Phase Microextraction. Analytical Chemistry, 89(16), 8366-8371. https://doi.org/10.1021/acs.analchem.7b01622
Chophi, R., Sharma, S., Sharma, S., & Singh, R. (2019) Forensic entomotoxicology: Current concepts, trends, and challenges. Journal of Forensic and Legal Medicine, 67, 28-36. https://doi.org/10.1016/j.jflm.2019.07.010
Chun, H.J., Poklis, J.L., Poklis, A., & Wolf, C.E. (2016) Development and validation of a method for alcohol analysis in brain tissue by headspace gas chromatography with flame ionization detector. Journal of Analytical Toxicology, 40(8), 653-658. https://doi.org/10.1093/jat/bkw075
Costa, L.G., & Aschner, M. (2014) Methanol. Encyclopedia of the Neurological Sciences (ed. Aminoff, M.J., & Daroff, R.B.), pp. 134-135. Elsevier, The Netherlands. https://doi.org/10.1016/B978-0-12-385157-4.00265-7
Da Silva, E.I.T., Wilhelmi, B., & Villet, M.H. (2017) Forensic entomotoxicology revisited-towards professional standardisation of study designs. International Journal of Legal Medicine,131(5), 1399-1412. https://doi.org/10.1007/s00414-017-1603-9
De Camargo, S.L.L.X., Da Silva Carvalho-Filho, F., & Cristina Esposito, M. (2018). The genus Peckia Robineau-Desvoidy (Diptera: Sarcophagidae) in the Brazilian Amazon: A new species, new records, descriptions of female terminalia and key to species. Zootaxa, 4483(1), 1-35. https://doi.org/10.11646/zootaxa.4483.1.1
Garcés, P.A., Arias, L.N., & Medina, M. (2020) Sarcophagidae de interés forense en el Parque Nacional Soberanía, Provincia de Panamá. Periodicidad: Semestral, 22(2), 1-11. http://portal.amelica.org/ameli/journal/224/2241149007/2241149007.pdf
García-Ruilova, A.B., Barragán, A., Ordoñez, S. del C., García, J.F., Mazón, J.D., Cueva, R., & Donoso, D.A. (2020) First records of Diptera associated with human corpses in Ecuador. Neotropical Biodiversity, 6(1), 197-202. https://doi.org/10.1080/23766808.2020.1845009
Gosselin, M., Wille, S.M.R., Fernandez, M. del M.R., Di Fazio, V., Samyn, N., De Boeck, G., & Bourel, B. (2011) Entomotoxicology, experimental set-up and interpretation for forensic toxicologists. Forensic Science International, 208(3), 1-9. https://doi.org/10.1016/j.forsciint.2010.12.015
Instituto Ecuatoriano de Normalización (2014). Bebidas alcohólicas. Aguardiente de caña. https://www.normalizacion.gob.ec/buzon/reglamentos/RTE-107.pdf(Accessed 15 February 2024)
Li, H., & Tennessen, J.M. (2018) Preparation of Drosophilalarval samples for gas chromatography-mass spectrometry (GC-MS)-based metabolomics. Journal of Visualized Experiments, 136, e57847. https://doi.org/10.3791/57847
Magni, P.A., Pacini, T., Pazzi, M., Vincenti, M., & Dadour, I.R. (2014) Development of a GC-MS method for methamphetamine detection in Calliphora vomitoria L. (Diptera: Calliphoridae). Forensic Science International, 241, 96-101. https://doi.org/10.1016/j.forsciint.2014.05.004
Magni, P.A., Pazzi, M., Vincenti, M., Alladio, E., Brandimarte, M., & Dadour, I.R. (2016) Development and validation of a GC-MS method for nicotine detection in Calliphora vomitoria (L.) (Diptera: Calliphoridae). Forensic Science International, 261, 53-60. https://doi.org/10.1016/j.forsciint.2015.11.014
Magni, P.A., Pazzi, M., Vincenti, M., Converso, V., & Dadour, I.R. (2018) Development and Validation of a Method for the Detection of α- And β-Endosulfan (Organochlorine Insecticide) in Calliphora vomitoria (Diptera: Calliphoridae). Journal of Medical Entomology, 55(1), 51-58. https://doi.org/10.1093/jme/tjx177
Martin, C., Vanderplanck, M., Boullis, A., Francis, F., Haubruge, E., & Verheggen, F. (2019) Impact of necrophagous insects on the emission of volatile organic compounds released during the decaying process. Entomologia Generalis, 39(1), 19-31. https://doi.org/10.1127/entomologia/2019/0663
Nauen, R., Bass, C., Feyereisen, R., & Vontas, J. (2021) The Role of Cytochrome P450s in Insect Toxicology and Resistance. Annual Review of Entomology, 67, 105-124. https://doi.org/10.1146/annurev-ento-070621
Nekoukar, Z., Zakariaei, Z., Taghizadeh, F., Musavi, F., Banimostafavi, E. S., Sharifpour, A., Ghuchi, N. E., Fakhar, M., Tabaripour, R., & Safanavaei, S. (2021) Methanol poisoning as a new world challenge: A review. Annals of Medicine & Surgery, 66. https://doi.org/10.1016/j.amsu.2021.102445
Pal, R., & Kumar, K. (2012) Ultrastructural features of the larval Malpighian tubules of the flesh fly Sarcophaga ruficornis (Diptera: Sarcophagidae). International Journal of Tropical Insect Science, 32(3), 166-172. https://doi.org/10.1017/S1742758412000252
Pape, T., Blagoderov, V., & Mostovski, M. (2011) Order Diptera Linnaeus, 1758. Animal biodiversity: An outline of higher-level classification and survey of taxonomic richness. Zootaxa, 3148(1), 222-229. https://doi.org/10.11646/zootaxa.3148.1.42
PHE (Public Health England) (2015) Compendium of Chemical Hazards: Methanol. https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/456293/Methanol_TO_PHE_260815.pdf (Accessed 15 February 2024)
Salazar, F., & Donoso, D. (2015) Catálogo de insectos con valor forense en el Ecuador. Revista Ecuatoriana de Medicina y Ciencias Biológicas, 36, 49-59. http://theantsofecuador.com/david/publications/Salazar%20&%20Donoso_2015_Catalogo%20Forense.pdf (Accessed 15 February 2024)
SOFT / AAFS (Society of Forensic Toxicologists, & American Academy of Forensic Sciences) (2006) Forensic toxicology laboratory guidelines, 2006 version. http://www.the-ltg.org/data/uploads/guidelines/soft-guidelines_2006.pdf (Accessed 15 February 2024 )
Tabor, K.L., Fell, R.D., Brewster, C.C., Pelzer, K., & Behonick, G.S. (2005) Effects of Antemortem Ingestion of Ethanol on Insect Successional Patterns and Development of Phormia regina (Diptera: Calliphoridae). Journal of Medical Entomology, 42(3), 481-489. https://academic.oup.com/jme/article/42/3/481/850574
Vairo, K.P., Mello-Patiu, C.A., & Carvalho, C. (2011) Pictorial identification key for species of Sarcophagidae (Diptera) of potential forensic importance in southern Brazil. Revista Brasileira de Entomologia, 55(3), 333-347. https://doi.org/10.1590/S0085-56262011005000033
Wu Chen, N.B., Donoghue, E.R., & Schaffer, M.I. (1985) Methanol intoxication: distribution in postmortem tissues and fluids including vitreous humor. Journal of Forensic Sciences, 30(1), 213-216. https://doi.org/10.1520/JFS10983J