Articulos
Communities of ectoparasites associated with cricetid rodents in Pereyra Iraola Biosphere Reserve, Argentina, with an analysis of Rickettsia infection
Comunidades de ectoparásitos asociados a roedores cricétidos en la Reserva de Biósfera Pereyra Iraola, Argentina, con un análisis de la infección por Rickettsia
Communities of ectoparasites associated with cricetid rodents in Pereyra Iraola Biosphere Reserve, Argentina, with an analysis of Rickettsia infection
Revista de la Sociedad Entomológica Argentina, vol. 84, núm. 1, e0106, 2025
Sociedad Entomológica Argentina
Recepción: 18 Octubre 2024
Aprobación: 15 Enero 2025
Abstract: Cricetid rodents are associated with ectoparasites, which are components of biodiversity. They may also cause diseases and act as potential vectors of microorganisms. The Pereyra Iraola Park (PPI) is situated in the area of greatest biodiversity of Buenos Aires Province. Our goal was to characterize the communities of ectoparasites associated with cricetids from the Ecological Santa Elena Farm, situated at the PPI, and to analyze the presence of Rickettsia spp. in these arthropods. Samplings were conducted during 2017-2018. A subsample of ectoparasites was prepared and identified with the optical microscope, and others were subjected to molecular analysis using the PCR technique in search of Rickettsia spp. A total of 106 cricetids were trapped, and 865 ectoparasites were collected. Mesostigmata mites presented the highest richness and diversity of species, abundance and prevalence; fleas presented higher values than ticks. These results agree with researches from nearby localities. Only the fleas Polygenis (P.) atopus (Jordan & Rothschild) and Polygenis (P.) a. axius (Jordan & Rothschild) were positive for Rickettsia, with sequences corresponding to Rickettsia felis, while mites and ticks were negative. The results obtained contribute to the knowledge of the biodiversity of the PPI and to the zoonotic importance of its fauna.
Keywords: Ixodida, Mesostigmata, Rickettsiaceae, Sigmodontinae, Siphonaptera.
Resumen: Los roedores cricétidos están asociados a ectoparásitos que son componentes de la biodiversidad, pueden causar enfermedades y actuar como potenciales vectores de microorganismos. El Parque Pereyra Iraola (PPI) se ubica en el área de mayor biodiversidad de la provincia de Buenos Aires. Nuestro objetivo fue caracterizar las comunidades de ectoparásitos de cricétidos en la Huerta Ecológica Santa Elena, ubicada en el PPI, y analizar la presencia de Rickettsia spp. en estos artrópodos. Los muestreos se realizaron durante 2017-2018. Se preparó una submuestra de ectoparásitos que se identificó al microscopio óptico y otras fueron sometidas a análisis molecular mediante la técnica de PCR en busca de Rickettsia spp. Se capturaron 106 cricétidos y se recolectaron 865 ectoparásitos. Los ácaros Mesostigmata presentaron la mayor riqueza y diversidad específica, abundancia y prevalencia, y las pulgas presentaron valores superiores que las garrapatas. Estos resultados concuerdan con investigaciones realizadas en localidades cercanas. Solamente las pulgas Polygenis (P.) atopus (Jordan & Rothschild) y Polygenis (P.) a. axius (Jordan & Rothschild) resultaron positivas a Rickettsia, con secuencias correspondientes a Rickettsia felis; los ácaros y garrapatas fueron negativos. Los resultados obtenidos contribuyen al conocimiento de la biodiversidad en el IPP y a la importancia zoonótica de su fauna.
Palabras clave: Ixodida, Mesostigmata, Rickettsiaceae, Sigmodontinae, Siphonaptera.
INTRODUCTION
Fleas, mites, and ticks are common ectoparasites associated with wild rodents (Cricetidae) worldwide (Morand et al., 2006). Ectoparasites are an important component of biodiversity and hold significant sanitary relevance as they can cause diseases such as pulicosis and acariasis. Moreover, they may act as potential vectors of bacteria whose transmission cycles involve rodent hosts (Tsai et al., 2011; Gutiérrez et al., 2015; Tomassone et al., 2018). Worldwide, zoonotic research on ectoparasites has primarily focused on ticks (e.g. Nava et al., 2017), while studies on fleas and other Acari, such as Mesostigmata, remain scarce (Chaisiri et al., 2015; Moreno Salas et al., 2019, 2020). A similar trend is seen in Argentina, where substantial knowledge has been gained about ticks, encompassing numerous species and geographic regions (Nava et al., 2017). In contrast, researches on fleas are still preliminary (e.g. Urdapilleta et al., 2020, 2021; Melis et al., 2020a), and studies on Mesostigmata mites remain unexplored.
Ticks and fleas were reported to be associated with bacteria of the genus Rickettsia, among others. Rickettsias present a complex cycle that includes the participation of mammals (domestic and wild) as hosts and arthropods as vectors. These bacteria produce diseases considered emerging and collectively called rickettsiosis, which can affect humans and are characterized by high fever, headaches, skin rashes, and muscle pain (Moreno Salas et al., 2020; Moraga-Fernandez et al., 2023).
Northeastern Buenos Aires Province is one of the areas with the greatest biodiversity in Argentina, where cricetid rodents are an important ecosystem component (Burkart et al., 1999; Patton et al., 2015). Several ectoparasites associated with cricetids in the area have been studied, leading to the description of new species (e.g.Lareschi & Gettinger, 2009; Lareschi, 2020; Savchenko & Lareschi, 2022), thereby increasing the known diversity for the region and enhancing its contribution to the overall diversity of the country. Additionally, ticks and fleas parasitizing cricetids in the area have been recorded in association with Rickettsia (Nava et al., 2017; Melis et al., 2020a, 2020b), underscoring the importance of continuing these studies.
Pereyra Iraola Park (PPI) harbors the greatest biodiversity in the province of Buenos Aires. Located in the northeast of the province, the PPI spans the territory of four municipalities (La Plata, Berazategui, Ensenada, and Florencio Varela), though its jurisdiction is mainly provincial. In 2007, the United Nations Educational, Scientific and Cultural Organization (UNESCO) designated the PPI as a World Biosphere Reserve. The PPI forms the Pereyra Iraola Biosphere Reserve with the Punta Lara Nature Reserve (Paolocá, 2023). Prior to the founding of the PPI in 1949, over 10,000 hectares of the park were owned by two large families and were expropriated to protect their rich biodiversity (Morosi et al., 1992). Due to its origin as a family estate, the park features more than 132 introduced forest species, along with important native flora (Vitalone & Delgado, 1994). Currently, the PPI offers a recreational space for the inhabitants of the province while also serving as a buffer between the two major metropolitan areas: Great La Plata to the south and Great Buenos Aires to the north, where one-third of the population of Argentina lives, and the most important industrial and economic hub of the country is located (Del Rio et al., 2007). Due to its diverse uses, the PPI is included in the Man and Biosphere Program (MAB-UNESCO). The park has schools, scientific institutes, experimental nurseries, state agencies, park ranger bases, police training institutions, and numerous agroecological production farms. Among these farms, the Ecological Santa Elena Farm is noteworthy, as it also hosts the headquarters of the Community Centre for University Extension (CCEU) N°10 of the National University of La Plata (UNLP). In addition, the PPI is a popular destination for visitors from across the Metropolitan Area of Buenos Aires (AMBA), particularly on weekends (Domínguez, 2008)
For these reasons, the PPI is of significant interest for conducting various studies. Our research focuses on characterizing the ectoparasite communities associated with cricetid rodents in the area and analysing the presence of Rickettsia in these arthropods. The results will contribute to our understanding of the biodiversity of the region and the zoonotic significance of its fauna.
MATERIALS AND METHODS
Study area
Sampling was conducted at the Ecological Santa Elena Farm (34º50’S, 58º5’W) (Fig. 1), an agroecological production farm located in the southern part of the PPI, a few meters from the Pereyra Train Station next to the General Roca Railway Train Stations and adjacent to the María Teresa School. This farm is the headquarters of the CCEU N° 10 of the UNLP. For this reason, in addition to marketing pesticide-free vegetables through home deliveries, different teams carry out extension practices there and receive numerous visits from national and international schools, universities and research centers. The CCEU itself is inhabited by two caretakers living in a 60 m2 house at the entrance of the property. Additionally, agricultural workers, as well as school and university students and researchers, regularly visit the premises to carry out extension and research tasks. Besides, the farm is home to numerous pigs, chickens in pens, and free-roaming horses, cows, a pony, dogs, and cats.
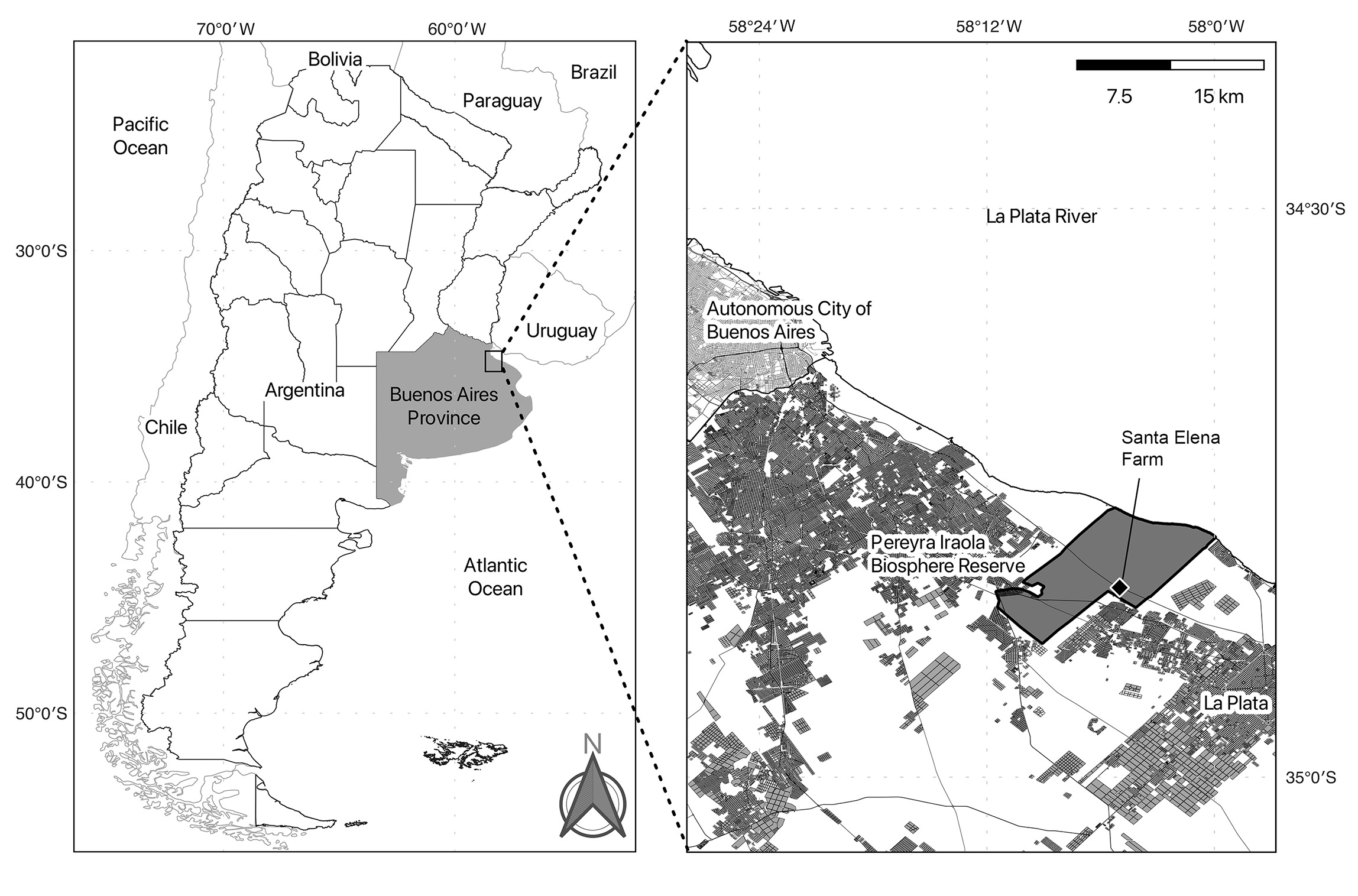
Figure 1.
Map of the Pereyra Iraola Biosphere Reserve and the sampling location (Santa Elena Farm).
Samples
Rodent traps were placed in an area characterized by three greenhouses where corn, tomato, and legumes are grown, surrounded by a Celtis sp. forest. Rodents were captured alive using Sherman-like traps baited with oats. The traps were placed in the vicinity of the greenhouses and the house. Sampling occurred in 2017 (October) and 2018 (January, February, March, August and October). The sampling effort involved 80 traps per night spaced five meters apart and left in the field for 24 hours. Captured rodents were anesthetized with sulphuric ether and euthanized by cervical dislocation. All procedures were conducted following the ethical guidelines established by the American Society of Mammalogists (Sikes, 2016).
Ectoparasites were collected in the field by examining the fur of the hosts using brushes and forceps, and then stored in alcohol 96 % at -18 °C. A subsample of the ectoparasites was prepared for identification using conventional optical microscopic techniques. Other specimens were subjected to molecular analysis using the PCR technique to detect Rickettsia spp., as described below.
Taxonomic identifications
After DNA extraction, the exoskeletons of the fleas were prepared for their identification under an optical microscope. They were washed with distilled water, cleared in 10 % KOH, dehydrated in a series of ethanol concentrations (80 % to 100 %), diaphanized in eugenol, and mounted in Canada balsam and identified following descriptions and keys provided by Smit (1987) and Linardi & Guimarães (2000). Mites were cleared in lactophenol and mounted in Hoyer’s medium and identified following Furman (1972), Savchenko & Lareschi (2019, 2022), Lareschi (2020), and Radovsky (2010). Ticks were identified under a stereoscopic binocular microscope and identified following Nava et al. (2017). Moreover, voucher specimens of every species fleas were deposited at the Colección de Entomología, and mites at the Colección de Invertebrados, both of the Museo de la Plata, Argentina. Rodents were identified following Patton et al. (2015) and deposited at the Colección de Mamíferos del Centro Nacional Patagónico (CENPAT), Puerto Madryn, Argentina.
Ecological analyses
For ectoparasites collected from each host species indices and parameters were calculated as follows: Ectoparasite specific richness (S= number of species), Shannon specific diversity index [H= -Σ (pi ln pi)], equitability index (J= H/ln S), mean abundance (MA= total number of individuals of a particular parasite species in a sample of a particular host species/total number of hosts of that species, including both infected and non-infected hosts), mean intensity (MI= total number of individuals of a particular parasite species in a sample of a particular host species/total number of infected hosts of that species); and prevalence [P= (number of hosts infected with one or more individuals of a particular parasite species/the number of hosts examined for that parasite species) x 100] (Begon et al., 1988; Bush et al., 1997). The significance (p) of differences between mean abundances and prevalences was tested by using Fisher’s exact test and Bootstrap test (20000 replications), respectively. The software Quantitative Parasitology 3.0 (Reiczigel et al., 2019) was used for the analyses. Only host species with 10 or more individuals captured were considered for the analyses.
Prospecting for Rickettsia
From the only tick collected and from a subsample of mites organized in pools (each pool consisting of three to five specimens collected from the same individual host), genomic DNA extraction was performed by a destructive method through proteolytic digestion with the enzyme proteinase K, placing each specimen in a solution consisting of 340 μl of a solution (10 mM TRIS at pH 8; 100 mM EDTA; 100 mM NaCl), 40 μl of 20 % SDS (sodium dodecyl sulfate) and 20 μl of proteinase K, 500 μg/ml (InvitrogenTM). DNA purification was performed with phenol and chloroform-isoamyl alcohol to denature and precipitate proteins, and precipitation was done with absolute ethanol. Finally, the extracted DNA was reconstituted in a TE buffer solution (10 mM Tris at pH 7.6; 0.1 mM EDTA) (Mangold et al., 1998).
In the case of fleas, it was necessary to preserve the exoskeleton for later preparation and identification under a microscope. Thus, individual DNA extraction was performed with Chelex®-100 (Bio-Rad Laboratories, CA, US) after an incision at the abdominal level to preserve the exoskeleton for further identification under an optical microscope. All collected fleas were analyzed. Genomic DNA extraction was adapted from the procedure described by Miura et al. (2017)as follows: a 5 % solution of Chelex®-100 resin in sterile distilled water was prepared, 100 μL of the homogenized mixture was placed in a microcentrifuge tube together with the individual flea sample, and 5 μL of proteinase K were added; it was left incubating for 18 h at 56 °C, then the enzyme was inactivated at 95 °C for 10 min, and once cooled it was centrifuged for 5 min at 14000 rpm.
In the first instance, a conventional PCR targeting a ca. 800-bp gltA fragment (citrate synthase) was performed to detect the presence of Rickettsia genus by using primers: CS-239 5’-CTCTTCTCATCCTATGGCTATTAT-3’ and CS-1069 5’-CAGGGTCTTCGTGCATTTCTT-3’ (Labruna et al., 2004). To identify Rickettsia to a specific level, samples shown to be positive to gltA were used to amplify: a ca. 800-bp fragment of the outer membrane protein B (OmpB) with the primers 120-M59: 5’-CCGCAGGGTTGGTAACTGC-3’ and 120-807 5’-CCTTTTAGATTACCGCCTAA-3’ (Regnery et al., 1991; Roux & Raoult, 2000). A negative control with ultrapure water and positive control consisting of DNA from Rickettsia vini was included for each reaction. The PCR conditions were: for gltA, 95 ºC for 3 min, 40 cycles of: denaturation at 95 ºC for 15 s, annealing at 52 ºC of 30 s, extension at 72 ºC for 30 s; and a final extension at 72 ºC for 7 min; for ompB, an initial denaturation at 95 ºC for 5 min, 40 cycles of: denaturation at 95 ºC for 30 s, annealing at 50 ºC of 30 s, extension at 72 ºC for 50 s; and a final extension at 72 ºC for 5 min. Afterward, PCR products of positive samples were purified with Wizard® Genomic DNA Purification kit (Promega®) and sequenced using ABI 3730XLs genetic analyzer, Macrogen Inc. (Korea).
The sequences were edited and aligned using CodonCode Aligner (CodonCode Corporation). The DNA sequences obtained were subjected to an analysis of comparison of sequences by using the Basic Local Alignment Search Tool (BLAST; https://blast.ncbi.nlm.nih.gov/Blast.cgi) to determine similarities with species of the genus Rickettsia. A phylogenetic tree was constructed with MrBayes 3.2.6 after 10 million generations, using a GTR+G model of substitution and including other sequences of Rickettsia spp. available in Genbank to determine their phylogenetic relationships. Only sequences where Rickettsia and host were identified at the species level were considered. The tree was visualized with FigTree v.1.4.4. The Bayesian posterior probabilities were transformed into percentages and indicated on the nodes of phylogenetic trees. GenBank accession numbers were indicated on the label of the tree, and samples obtained in this study were in bold (OL961553, OL961554).
RESULTS
A total of 106 cricetid rodents were trapped and identified as Oxymycterus rufus (Fischer) (n= 44), Akodon azarae (Fischer) (n= 41), Oligoryzomys flavescens (Waterhouse) (n= 19), Calomys laucha (Fischer) (n= 1) and Holochilus brasiliensis (Desmarest) (n= 1). Ectoparasites associated only with the three most abundant rodent species were studied, and a total of 859 ectoparasites were collected from them. Out of these, 826 specimens were mites (Acari, Mesostigmata), 32 were fleas (Hexapoda, Siphonaptera), and only one was a tick (Acari, Ixodida). The collected ectoparasites were identified as: Acari, Mesostigmata, Laelapidae: Androlaelaps azarae Lareschi (n= 376), Androlaelaps fahrenholzi (Berlese) (n= 185), Mysolaelaps microspinosus Fonseca (n= 118), Gigantolaelaps wolffsohni Oudemans (n= 39), Laelaps schatzi Savchenko & Lareschi (n= 27), Laelaps galliarii Savchenko & Lareschi (n=17); Acari, Macronyssidae: Ornithonyssus bacoti (Hirst) (n= 64); Acari, Ixodida, Ixodidae: Amblyomma triste Koch (n= 1) (Fig. 2); Hexapoda, Siphonaptera, Rhopalopsyllidae: Polygenis (Neopolygenis) atopus (Jordan & Rothschild) (n= 10), Polygenis (Neopolygenis) massoiai Del Ponte (n= 1), Polygenis (Polygenis) axius axius (Jordan & Rothschild) (n= 13), Polygenis (Polygenis) platensis (Jordan & Rothschild) (n= 8) (Fig. 3).

Figure 2.
Acari.
Mesostigmata, Laelapidae: a. Androlaelaps azarae female. b. Androlaelaps fahrenholzi female. c. Mysolaelaps microspinosus female. d. Gigantolaelaps wolffsohni female. g. Laelaps schatzi female. h. Laelaps galliarii female. Mesostigmata, Macronyssidae: e. Ornithonyssus bacoti female. Ixodida, Ixodidae: f. Amblyomma triste female.

Figure 3.
Hexapoda, Siphonaptera.
a. Polygenis (Neopolygenis)atopus male. b. Polygenis (Neopolygenis) massoiai male. c. Polygenis (Polygenis) axius axius male. d. Polygenis (Polygenis) platensis male.
Total species richness was S= 12, diversity was H= 1.65, and equitability was J= 0.66. In comparison, mites included more species and were more prevalent and abundant (S= 7; P= 83.6%; MA= 7.95) than fleas (S= 4; P= 22%, p< 0.0001; MA= 0.31, p< 0.0001) and ticks (S= 1; P= 1%, p< 0.0001; MA= 0.01, p< 0.0001). Also, fleas were more prevalent and abundant than ticks (P, p< 0.0001; MA, p<0.0005). When comparing among host species, O. rufus showed the highest diversity (H= 1.18), specific richness (S= 7) and equitability (J= 0.61) of ectoparasites, while Ak. azarae showed the lowest diversity (H= 0.58) and equitability (J= 0.32) (Table I).
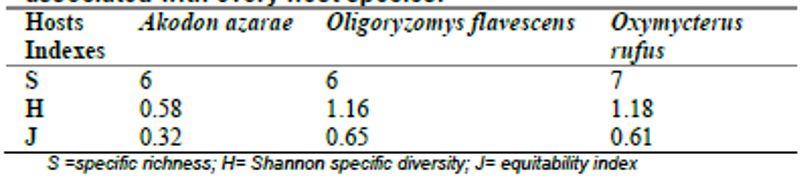
Table I.
Comparison of the communities of ectoparasites associated with every host species.
S =specific richness; H= Shannon specific diversity; J= equitability index
When comparing total ectoparasites associated with every host species, the mean abundances were higher for O. flavescens (MA= 11.16) and Ak. azarae (MA= 11.39) compared to O. rufus (MA= 4.11, p= 0.004, p= 0.007). There were no differences between O. flavescens and Ak. azarae (p= 0.93). Also, there were no differences between the prevalences of O. flavescens (P= 94.7) and Ak. azarae (P= 92.7, p=0.9), O. flavescens and O. rufus (P= 81.8, p= 0.25), and Ak. azarae and O. rufus (p= 0.19).
Considering only the fleas, O. rufus showed the highest P (38.6%) and MA (0.5), compared to Ak. azarae (P= 12%, p= 0.007; MA= 0.2, p= 0.045) and O. flavescens (P= 5%, p= 0.007; MA= 0.05, p= 0.0008). There were no differences between Ak. azarae and O. flavescens for P (p= 0.65) and MA (p=0.16). Concerning mites, O. flavescens (P= 95%; MA= 11.1) and Ak. azarae (P= 92%; MA= 11.2) showed prevalences and mean abundances higher than O. rufus (P= 70%, p= 0.047, p= 0.012; MA= 3.6, p= 0.002, p= 0.004). There were no differences between Ak. azarae and O. flavescens for P (p= 0.99) and MA (p= 0.98).
Some ectoparasite species were associated with only one host species, such as An. azarae, which was specific to Ak. azarae; G. wolffsohni, L. schatzi, L. galliarii and M. microspinosus only to O. flavescens; P. massoiai to O. rufus; and A. triste to O. rufus (Table II). On the other hand, some ectoparasite species were shared by two or the three cricetid species; A. fahrenholzi was significantly different among host species, with higher mean abundance on O. rufus and Ak. azarae compared to O. flavescens (p= 0.002; p= 0.004), while there were no significant differences between Ak. azarae and O. rufus (p= 0.74). Neither were there differences in prevalences between Ak. azarae–O. flavescens (p= 0.1), Ak. azarae–O. rufus (p= 0.9), and O. flavescens–O. rufus (p= 0.2) (Table II). For O. bacoti, the prevalence was higher in O. rufus compared to Ak. azarae (p= 0.0001), but there was no difference for MA (p= 0.28). For P. (P.) a. axius the prevalence and mean abundance were higher in O. rufus then in Ak. azarae (P, p= 0.03; MA, p= 0.03). The prevalences and mean abundances for P. (N.) atopus and P. platensis were no different between cricetid rodents (Table II).
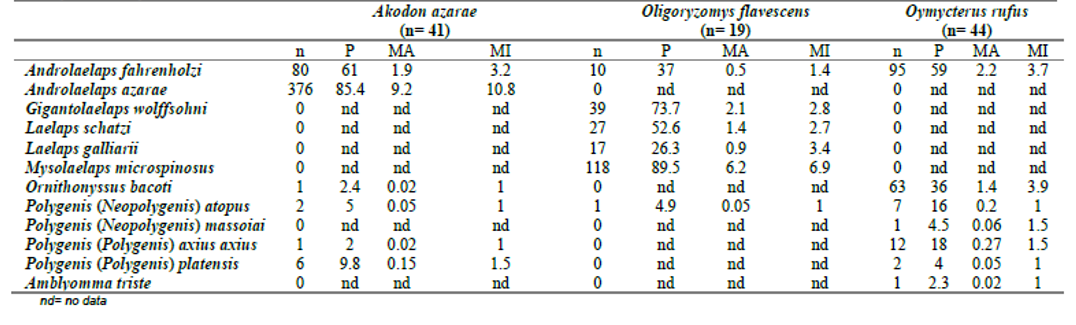
Table II.
Comparison of the number of specimens (n), prevalence (P), mean abundance (MA), and mean intensity (MI) of every ectoparasite species associated with each host species.
nd= no data
Concerning prospection of Rickettsia spp., only two fleas were positive, while mites and ticks were negative. Of the two positive fleas, one was identified as P. (P.) atopus from O. rufus and the other one, as P. (P.) a. axius from A. azarae. The sequence analysis indicated that the two positive findings corresponded to Rickettsia felis (Table III; Fig. 4).
The phylogenetic tree showed a clade including all ompB sequences of R. felis and separated from the other Rickettsia species. These clades were structured independently of the arthropod, host, or site of sampling. The clade of R. felis was a sister clade of R. prowazekii, R. rickettsii, and R. typhi, and both clades were sisters of the clade including to R. aeschlimanni, R. massiliae, R. parkeri, and R. raoultii (Fig. 4).

Figure 4.
Unrooted phylogenetic tree from ompB sequences of Rickettsia spp.
The nodes indicated the Bayesian probabilities. Labels indicated the species of Rickettsia, followed by the arthropod (except for human samples), the host or site of sampling and the country of the sample. GenBank accession numbers are indicated in parentheses for each taxa.
DISCUSSION
While 12 species of ectoparasites were identified, none of the host species reached this specific richness because several of the ectoparasites were host-specific. Although studies on the ectoparasites of the rodents herein considered were carried out in close localities years ago, comparisons are difficult to make because those researches do not consider all the ectoparasites identified at a specific level, such as fleas (Polygenis spp.) (e.g. Lareschi, 1996), or they include the Phthiraptera species, not considered in the present study (e.g. Liljesthröm & Lareschi, 2002). In addition, in recent years the systematics of some ectoparasite species has been revised and these changes make comparisons difficult, so it would be necessary to review those specimens. In this sense, the taxonomic review of Mesostigmata mites associated with cricetids from the PPI and its surroundings allowed the identification of cryptic species and, in some cases, they were described as new species for science. Such is the case of L. schatzi, which was previously considered included in the complex Laelaps paulistanensis Fonseca, An. azarae in Androalelaps rotundus (Fonseca) complex, and L. galliarii and Laelaps scapteromyos (Savchenko & Lareschi) in Laelaps manguinhosi Fonseca (Savchenko & Lareschi, 2019, 2022; Lareschi, 2020). These studies not only expanded the number of known species of the Laelapidae family, but also increased the known biodiversity for the area. Moreover, some of these species previously considered generalists (e.g. An. rotundus and L. manguinhosi), were then considered specific to their hosts based on these studies. These findings not only highlight the importance of the area in biodiversity studies but also suggest the need for its protection, given that being host-specific, the extinction of the rodent hosts would, in turn, lead to the extinction of their parasites.
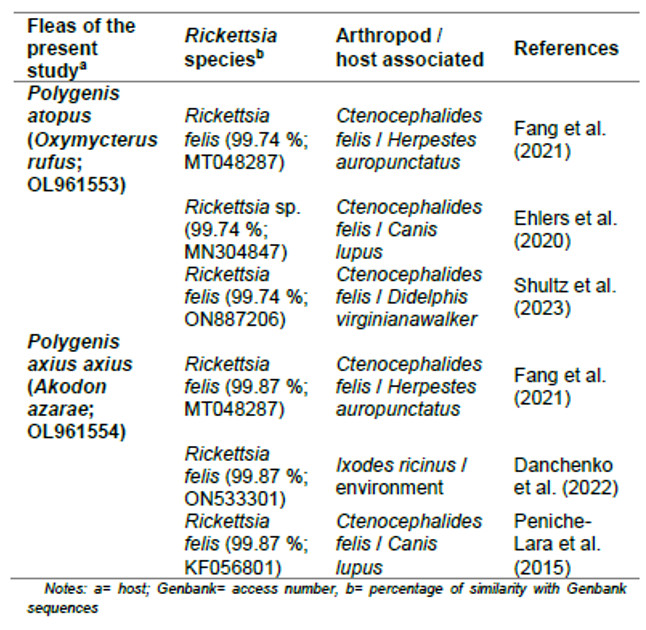
Table III.
Similarity percentage for obtained sequences with BLAST analyses.
Genbank access number is included for every sequence
Notes: a= host; Genbank= access number, b= percentage of similarity with Genbank sequences
Although, as stated, comparisons between different studies are not exact, in general terms, within the ectoparasites, the Mesostigmata mites presented the greatest richness and diversity of species and were the most abundant and prevalent. In turn, fleas presented higher values in all these parameters and indices than ticks. These results agree with similar studies in nearby areas such as Punta Lara Nature Reserve (Lareschi, 1996). Likewise, they also agree with studies in other locations in the Argentinean provinces of Buenos Aires (e.g. Navone et al., 2009; Colombo et al., 2014), Entre Ríos (Abba et al., 2001) and Misiones (Lareschi et al., 2019), as well as in southern Brazil (e.g. Barros et al., 1993; Sponchiado et al., 2015). Similarly, high species richness and diversity (e.g. in O. rufus) were not associated with high abundance and prevalence (which were higher in O. flavescens and A. azarae). Comparing the higher taxa of ectoparasites, in O. rufus fleas had the highest abundance and prevalence values, while in the remaining host species, Mesostigmata mites presented the highest ones. Although these values for the communities associated with each host species in some cases agree with results from nearby areas and in others disagree (e.g. Lareschi, 1996), the dominance of Mesostigmata mites remains constant.
Concerning molecular results, they indicate that the sequences of this study have high similarity with R. felis isolated from the flea Ctenocephalides felis (Bouché) from America and Asia. Previously, in Argentina, R. felis was reported for P. (P.) a. axius from Arana, a semiurbanized area located 14 km south of the city of La Plata (northeast of Buenos Aires Province) (Melis et al., 2020a), and C. felis from Rafaela city in Santa Fe Province, Argentina (Nava et al., 2008).
Our study reported the association of R. felis with the flea P. (P.) atopus for the first time and extended the association with P. (P.) a. axius to a nearby locality. Since these fleas have a wide distribution and are also host generalists (Lareschi et al., 2016), the results obtained are epidemiologically important. Rickettsia felis is associated with the spotted fever group rickettsiae (SFG). This bacterium is an emergent and widely distributed flea-borne human pathogenic species, associated with domestic and peridomestic animals and ectoparasites (Stevenson et al., 2005; Merhej & Raoult, 2011; Panti-May et al., 2015). Its main vector is the flea C. felis, but mosquitoes, Mesostigmata mites and ticks may also be associated with this bacterium (Shpynov et al., 2018). Although in this study, Mesostigmata mites were negative for Rickettsia presence, this bacterium was reported in the families Laelapidae and Macronyssidae from Brazil, Lithuania, and China (e.g. Nieri-Bastos et al., 2011; Kuo et al., 2020; Yin et al., 2021; Radzijevskaja et al., 2018). Thus, the epidemiologic relevance of Mesostigmata in the cycle of Rickettsia should not be discarded.
The only tick collected was negative to Rickettsia. However, the association of A. triste with Rickettsia parkeri was reported in nearby localities (Venzal et al., 2012; Nava et al., 2017; Romer et al., 2020). Thus, more samples are needed to evaluate the presence of Rickettsia in this tick in PPI area.
The results obtained present a scenario where it is necessary to continue the research on the ectoparasites of cricetid rodents in the PPI in order to elucidate the taxonomic status of the different species and their host affinity, and consequently, the biodiversity in the area. Likewise, it is necessary to increase research on the Rickettsia-ectoparasite association in order to elucidate the possible role of fleas, mites and ticks in the enzootic cycle of rickettsiae with potential risk for humans mainly in environments where human activities are frequent.
Acknowledgments
We are especially grateful to Elena Senattori (Santa Elena Farm) and Fernando Glenza (Cátedra Libre de Soberanía Alimentaria, Universidad Nacional de La Plata) for providing the guidance and logistics necessary to carry out the sampling at the Farm; to Graciela Minardi (CEPAVE) for her collaboration with the statistical analyses; and to Ulyses Pardiñas (IDEAus, CONICET, Argentina) for his collaboration in the identification of the rodents. The study was supported by Agencia Nacional de Promoción Científica y Tecnológica (PICT 2015-1564), Universidad Nacional de La Plata, Argentina (N992) and Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET) (PIP 628) (all to M. Lareschi). Collecting permits were provided by the Dirección de Flora y Fauna del Ministerio de Desarrollo Agrario de la Provincia de Buenos Aires. This study is part of the doctoral dissertation of M. Melis at the Facultad de Ciencias Naturales y Museo, Universidad Nacional de La Plata, Argentina.
REFERENCES
Abba, A.M., Udrizar Sauthier, D.E., Bender, J.B., & Lareschi, M. (2001) Mites (Acari: Laelapidae) associated with Sigmodontinae rodents in Entre Ríos Province, Argentina. Memórias Do Instituto Oswaldo Cruz, 96, 1171-1172.
Barros, D.M., Linardi, P.M., & Botelho, J.R. (1993) Ectoparasites of some wild rodents from Paraná State, Brazil. Journal of Medical Entomology, 30, 1068-1070. https://doi.org/10.1093/jmedent/30.6.1068
Begon, M. (1988) Ecología: individuos, poblaciones, comunidades. Omega, Barcelona, España.
Burkart, R., Bárbaro, N., Sánchez, R., & Gómez, D. (1999) Eco-regiones de la Argentina. Administración de Parques Nacionales, Presidencia de la Nación, Argentina.
Bush, A., Lafferty, K,, Lotz, J.M., & Shostak, A. (1997) Parasitology meets ecology on its own terms: Margolis et al. revisited. Journal of Parasitology, 83, 575-583.
Chaisiri, K., McGarry, J., Morand, S., & Makepeace, B. (2015) Symbiosis in an overlooked microcosm: a systematic review of the bacterial flora of mites. Parasitology, 142, 1152-1162. https://doi.org/10.1017/S0031182015000530
Colombo, V.C., Lareschi, M., Monje, L.D., Nava, S., Antoniazzi, L.R., Beldomenico, P. M., & Guglielmone, A.A. (2014) Garrapatas (Ixodida) y ácaros (Mesostigmata) parásitos de roedores sigmodontinos del delta del Paraná, Argentina. FAVE Sección Ciencias Veterinarias, 12, 39-50.
Danchenko, M., Benada, O., & Sekeyová, Z. (2022) Culture isolate of Rickettsia felis from a tick. International Journal of Environmental Research and Public Health, 19, 4321. https://doi.org/10.3390/ijerph19074321
Del Rio, J.P., Maidana, J.A., Molteni, A., Pérez, M., Pochettino, M.L., Souilla, L., & Turco, E. (2007) El rol de las “quintas” familiares del Parque Pereyra Iraola (Bs.As., Argentina) en el mantenimiento de la agrobiodiversidad. Kurtziana, 33, 217-226.
Domínguez, A. (2008) El contexto cultural en la implementación de proyectos de desarrollo rural. El caso del Parque Pereyra Iraola. Mundo Agrario, 9, 1-16.
Ehlers, J., Krüger, A., Rakotondranary, S., Ratovonamana, R., Poppert, S., Ganzhorn, J., & Tappe, D. (2020) Molecular detection of Rickettsia spp., Borrelia spp., Bartonella spp. and Yersinia pestis in ectoparasites of endemic and domestic animals in southwest Madagascar. Acta Tropica, 205, 105339. https://doi.org/10.1016/j.actatropica.2020.105339
Fang, K., Philpot, K., Chi, X., Ketzis, J., Du, A., & Yao, C. (2021) Small indian mongooses (Herpestes auropunctatus) serve as reservoirs of Bartonella henselae and Rickettsia felis vectored by Ctenocephalides felis. Vector-Borne and Zoonotic Diseases, 21, 422-431. https://doi.org/10.1089/vbz.2020.2733
Furman, D.P. (1972) Laelapid mites (Laelapidae: Laelapinae) of Venezuela. Brigham Young University Science Bulletin Biological Series, 27, 1-58.
Gutiérrez, R., Krasnov, B., Morick, D., Gottlieb, Y., Khokhlova, I., & Harrus, S. (2015) Bartonella infection in rodents and their flea ectoparasites: an overview. Vector-Borne and Zoonotic Diseases, 15, 27-39. https://doi.org/10.1089/vbz.2014.1606
Kuo, C.C., Lee, P.L., & Wang, H.C. (2020) Molecular detection of Rickettsia species and host associations of Laelaps mites (Acari: Laelapidae) in Taiwan. Experimental & Applied Acarology, 81, 547-559. https://doi.org/10.1007/s10493-020-00522-1
Labruna, M.B., Whitworth, T., Horta, M.C., Bouyer, D.H., McBride, J.W., Pinter, A., Popov, V., Gennari, S.M., & Walker, D.H. (2004) Rickettsia species infecting Amblyomma cooperi ticks from an area in the state of São Paulo, Brazil, where Brazilian spotted fever is endemic. Journal of Clinical Microbiology, 42, 90-98. https://doi.org/10.1128/jcm.42.1.90-98.2004
Lareschi, M. (1996) Estudio preliminar de la comunidad de roedores (Rodentia: Muridae) y sus ectoparásitos (Acari, Phthiraptera y Siphonaptera) en Punta Lara (Buenos Aires). Revista de la Sociedad Entomológica Argentina, 55, 113-120.
Lareschi, M. (2020) Three new species of Laelapidae mites (Mesostigmata) parasitic of species of Akodon (Rodentia: Cricetidae: Sigmodontinae) on the basis of female, male and deutonymph specimens. Veterinary Parasitology: Regional Studies and Reports, 22, 100500. https://doi.org/10.1016/j.vprsr.2020.100500
Lareschi, M., & Gettinger, D. (2009) A new species of Androlaelaps (Acari: Parasitiformes) from the akodontine rodent Deltamys kempi Thomas, 1919, in La Plata River Basin, Argentina. Journal of Parasitology, 95, 1352-1355. https://doi.org/10.1645/GE-2045.1
Lareschi, M., Sanchez, J., & Autino, A. (2016) A review of the fleas (Insecta- Siphonaptera) from Argentina. Zootaxa, 4103, 239-258.
Lareschi, M., Savchenko, E., & Urdapilleta, M. (2019) Ectoparasites associated with sigmodontine rodents from northeastern Argentina. Therya, 10, 103-108. https://doi.org/10.12933/therya-19-758
Liljesthröm, G., & Lareschi, M. (2002) Estudio preliminar de la comunidad ectoparasitaria de roedores sigmodontinos en el partido de Berisso, provincia de Buenos Aires. Actualizaciones en artropodología sanitaria Argentina (ed. Salomón, O.D.) pp. 257-260. Red Argentina de Estudio de Artrópodos Vectores de Enfermedades Humanas (RAVE). Serie Enfermedades Transmisibles. Publicación monográfica 2. Fundación Mundo Sano, ISBN 987-20421-0-1. Buenos Aires.
Linardi, P.M., & Guimarães, L.R. (2000) Sifonapteros do Brasil. Museu Zoologia USP, FAPESP, São Paulo, Brazil.
Mangold, A., Bargues, M., & Mas-Coma, S. (1998). Mitochondrial 16S rDNA sequences and phylogenetic relationships of species of Rhipicephalus and other tick genera among Metastriata (Acari: Ixodidae). Parasitology Research, 84, 478-484. https://doi.org/10.1007/s004360050433
Melis, M., Espinoza-Carniglia, M., Savchenko, E., Nava, S., & Lareschi, M. (2020a) Molecular detection and identification of Rickettsia felis in a Polygenis flea (Siphonaptera, Rhopalopsyllidae, Rhopalopsyllinae) associated with cricetid rodents from a rural area in central Argentina. Veterinary Parasitology: Regional Studies and Reports, 21, 100445. https://doi.org/10.1016/j.vprsr.2020.100445
Melis, M.E., Sebastian, P.S., Balcazar, D.E., Lareschi, M., & Nava, S. (2020b) Molecular detection of Rickettsia bellii in Ixodes loricatus (Acari: Ixodidae) ticks associated with rodents from Buenos Aires Province, Argentina. Revista de la Sociedad Entomológica Argentina, 79, 51-55. https://doi.org/10.25085/rsea.790409
Merhej, V., & Raoult, D. (2011) Rickettsial evolution in the light of comparative genomics. Biological Reviews, 86, 379-405.
Miura, K., Higashiura, Y., & Maeto, K. (2017) Evaluation of easy, non-destructive methods of DNA extraction from minute insects. Applied Entomology and Zoology, 52, 349-352. https://doi.org/10.1007/s13355-017-0481-4
Moraga-Fernandez, A., Munoz-Hernandez, C., Sanchez-Sanchez, M., de Mera, I.G.F., & de la Fuente, J. (2023) Exploring the diversity of tick-borne pathogens: The case of bacteria (Anaplasma, Rickettsia, Coxiella and Borrelia) protozoa (Babesia and Theileria) and viruses (Orthonairovirus, tick-borne encephalitis virus and louping ill virus) in the European continent. Veterinary Microbiology, 286, 109892.
Morand, S., Krasnov, B.R., Poulin, R., & Degen, A.A. (2006) Micromammals and macroparasites: Who is who and how do they interact?. Micromammals and Macroparasites (ed. Morand, S., Krasnov, B., & Poulin, R.), pp. 3-9. Springer-Verlag Tokyo, New York, USA.
Moreno Salas, L., Espinoza-Carniglia, M., Lizama-Schmeisser, N., Torres Fuentes, L.G., Silva-de la Fuente, M.C., Lareschi, M., & González-Acuña, D. (2019) Fleas of black rats (Rattus rattus) as reservoir host of Bartonella spp. in Chile. PeerJ, 7, e7371. https://doi.org/10.7717/peerj.7371
Moreno Salas, L., Espinoza-Carniglia, M., Lizama-Schmeisser, N., Torres Fuentes, L.G., Silva-de la Fuente, M.C., Lareschi, M., & González-Acuña, D. (2020) Molecular detection of Rickettsia in fleas from micromammals in Chile. Parasites & Vectors, 13, 523. https://doi.org/10.1186/s13071-020-04388-5
Morosi, J.A., Amarilla, B., Contin, M., Vitalone, C., Conti, A.L., Delgado, A., Vásquez, V., Coletti, R., Stornini, A., & Molinari, G. (1992) Parque Provincial Pereyra Iraola. Una introducción a la conservación y refuncionalización de su patrimonio. LINTA, CIC, Buenos Aires, Argentina.
Nava, S., Pérez-Martínez, L., Venzal, J., Portillo, A., Santibáñez, S., & Oteo, J.A. (2008) Rickettsia felis in Ctenocephalides felis from Argentina. Vector-Borne and Zoonotic Diseases,8, 465-466. https://doi.org/10.1089/vbz.2007.0243
Nava, S., Venzal, J.M., González-Acuña, D., Martins, T.F., & Guglielmone, A.A. (2017) Ticks of the southern cone of America. Elsevier, Academic Press, London, England.
Navone, G.T., Notarnicola, J., Nava, S., Robles, M., Galliari, C., & Lareschi, M. (2009) Ensamble de artrópodos y helmintos parásitos en roedores sigmodontinos de los humedales del Río de la Plata, Argentina. Mastozoología Neotropical, 16, 121-134.
Nieri-Bastos, F.A., Labruna, M.B., Marcili, A., Durden, L.A., Mendoza-Uribe, L., & Barros-Battesti, D.M. (2011) Morphological and molecular analysis of Ornithonyssus spp. (Acari: Macronyssidae) from small terrestrial mammals in Brazil. Experimental and Applied Acarology, 55, 305-327. https://doi.org/10.1007/s10493-011-9475-z
Panti-May, J.A., Torres-Castro, M., Hernández-Betancourt, S., Dzul-Rosado, K., Zavala-Castro, J., López-Avila, K., & Tello-Martin, R. (2015) Detection of Rickettsia felis in wild mammals from three municipalities in Yucatan, Mexico. Ecohealth, 12, 523-527. https://doi.org/10.1007/s10393-014-1003-2
Paolocá, I. (2023) Gubernamentalidad ambiental y resistencia. El caso del conflicto entre guardaparques y funcionarios de un parque provincial bonaerense. Revista del Museo de Antropología, 16, 165-176. http://doi.org/10.31048/1852.4826.v16.n2.41583
Patton, J., Pardiñas, U.F.J., & D’Elía, G. (2015) Mammals of South America Volume 2 Rodents. The University Chicago Press, Chicago, USA.
Peniche-Lara, G., Jimenez-Delgadillo, B., & Dzul-Rosado, K. (2015) Rickettsia rickettsii and Rickettsia felis infection in Rhipicephalus sanguineus ticks and Ctenocephalides felis fleas co-existing in a small city in Yucatan, Mexico. Journal of Vector Ecology, 40, 422-424. https://doi.org/10.1111/jvec.12185
Radovsky, F.J. (2010) Revision of genera of the parasitic mite Family Macronyssidae (Mesostigmata: Dermanyssidae) of the world (ed. Indira publishing house), pp. 1-170. West Bloomfield, US.
Radzijevskaja, J., Kaminskienė, E., Lipatova, I., Mardosaitė-Busaitienė, D., Balčiauskas, L., Stanko, M., & Paulauskas A. (2018) Prevalence and diversity of Rickettsia species in ectoparasites collected from small rodents in Lithuania. Parasites & Vectors, 11, 375. https://doi.org/10.1186/s13071-018-2947-9
Regnery, R.L., Spruill, C.L., & Plikaytis, B. (1991) Genotypic identification of rickettsiae and estimation of intraspecies sequence divergence for portions of two rickettsial genes. Journal of Bacteriology, 173, 1576-1589. https://doi.org/10.1128/jb.173.5.1576-1589.1991
Romer, Y., Borrás, P., Govedic, F., Nava, S., Carranza, J.I., Santini, S., Armitano, R., & Lloveras, S. (2020) Clinical and epidemiological comparison of Rickettsia parkeri rickettsiosis, related to Amblyomma triste and Amblyomma tigrinum, in Argentina. Ticks and Tick-borne Diseases, 11, 101436. https://doi.org/10.1016/j.ttbdis.2020.101436
Roux, V., & Raoult, D. (2000) Phylogenetic analysis of members of the genus Rickettsia using the gene encoding the outer-membrane protein rOmpB (ompB). International Journal of Systematic and Evolutionary Microbiology, 50, 1449-1455. https://doi.org/10.1099/00207713-50-4-1449
Reiczigel, J., Marozzi, M., Fábián, I., & Rózsa, L. (2019) Biostatistics for Parasitologists - A Primer to Quantitative Parasitology. Trends in Parasitology, 35, 277-281. doi: 10.1016/j.pt.2019.01.003. Epub 2019 Jan 31. PMID: 30713051
Savchenko, E., & Lareschi, M. (2019) A new species of Laelaps Koch, 1836 (Mesostigmata: Laelapidae) parasitic of the sigmodontine rodent Oligoryzomys flavescens Waterhouse, 1837 (Rodentia: Cricetidae): Molecular and morphological characterization. Acta Tropica, 199, 105146. https://doi.org/10.1016/j.actatropica.2019.105146
Savchenko, E., & Lareschi, M. (2022) Integrative taxonomy reveals hidden diversity within the concept of laelapine mite species (Mesostigmata: Laelapidae) associated with sigmodontine rodents (Cricetidae): description of three new species of Laelaps Koch in the Neotropical region. Systematic and Applied Acarology, 27, 2426-2457. https://doi.org/10.11158/saa.27.12.5
Shultz, L., López-Pérez, A., Jasuja, R., Helman, S., Prager, K., Tokuyama, A., Quinn, N., Bucklin, D., Rudd, J., Clifford, D., et al. (2023) Vector-borne disease in wild mammals impacted by urban expansion and climate change. Ecohealth, 20, 286-299. https://doi.org/10.1007/s10393-023-01650-x
Shpynov, S., Fournier, P., Pozdnichenko, N., Gumenuk, A., & Skiba, A. (2018) New approaches in the systematics of rickettsiae. New Microbes and New Infections, 23, 93-102. https://doi.org/10.1016/j.nmni.2018.02.012
Sikes, R.S. (2016) Guidelines of the American Society of Mammalogists for the use of wild mammals in research and education. Journal of Mammalogy, 97, 663-688. https://doi.org/10.1093/jmammal/gyw078
Smit, F.G.A.M. (1987) An illustrated catalogue of the Rothschild collection of fleas (Siphonaptera) in the British Museum (Natural History): with keys and short descriptions for the identification of families, genera, species and subspecies of the order Malacopsylloidea and Rhopalopsyllidae (Volume 7). Oxford University Press, Oxford, USA.
Sponchiado, J., Melo, G., Landulfo, G., Jacinavicius, F.C., Barros-Battesti, D.M., & Cáceres, N.C. (2015) Interaction of ectoparasites (Mesostigmata, Phthiraptera, and Siphonaptera) with small mammals in Cerrado fragments, western Brazil. Experimental and Applied Acarology, 66, 369-381. https://doi.org/10.1007/s10493-015-9917-0
Stevenson, H.L., Labruna, M.B., Montenieri, J.A., Kosoy, M.Y., Gage, K.L., & Walker, D.H. (2005) Detection of Rickettsia felis in a new world flea species, Anomiopsyllus nudata (Siphonaptera: Ctenophthalmidae). Journal of Medical Entomology, 42, 163-167. https://doi.org/10.1093/jmedent/42.2.163
Tomassone, L., Portillo, A., Nováková, M., de Sousa, R., & Oteo, J. (2018) Neglected aspects of tick-borne rickettsioses. Parasites & Vectors, 11, 263. https://doi.org/10.1186/s13071-018-2856-y
Tsai, Y., Chang, C., Chuang, S., & Chomel, B. (2011) Bartonella species and their ectoparasites: selective host adaptation or strain selection between the vector and the mammalian host? Comparative Immunology, Microbiology and Infectious Diseases, 34, 299-314. https://doi.org/10.1016/j.cimid.2011.04.005
Urdapilleta, M., Cicuttin, G.L., De Salvo, M.N., Pech-May, A., Salomon, O.D., & Lareschi, M. (2020) Molecular detection and identification of Bartonella in the cat flea Ctenocephalides felis felis collected from cats and dogs in a border area in northeastern Argentina. Veterinary Parasitology: Regional Studies and Reports, 19, 100361 https://doi.org/10.1016/j.vprsr.2019.100361.
Urdapilleta, M., Pech-May, A., Lamattina, D., Burgos, F.E., Balcazar, E.D., Ferrari W.A., Lareschi, M., & Salomón, O.D. (2021) Ecology of fleas and their hosts in the trifinio of northeast Argentina: First detection of Rickettsia asembonensis in Ctenocephalides felis felis in Argentina. Medical and Veterinary Entomology, 36, 20-29. https://onlinelibrary.wiley.com/doi/10.1111/mve.12546
Venzal, J.M., Estrada-Peña, A., Portillo, A., Mangold, A.J., Castro, O., Souza, C.G.D., Felix, M.L., Perez Martinez, M.L., Santibáñez, S., & Oteo, J.A. (2012) Rickettsia parkeri: a rickettsial pathogen transmitted by ticks in endemic areas for spotted fever rickettsiosis in southern Uruguay. Revista do Instituto de Medicina Tropical de Sao Paulo, 54, 131-134. https://doi.org/10.1590/S0036-46652012000300003
Vitalone, C., & Delgado, A. (1994) Parque Pereyra Iraola: del territorio inalterado al territorio degradado. Repositorio Institucional Comisión de Investigaciones Científicas, Anales LINTA, La Plata, Argentina, 1, 21-36. https://digital.cic.gba.gob.ar/items/42d3b634-0506-475c-ac8e-7cba7af8f147
Yin, P.W., Guo, X.G., Jin, D.C., Fan, R., Zhao, C.F., Zhang, Z.W., Huang, X., & Mao, K. (2021) Distribution and host selection of tropical rat mite, Ornithonyssus bacoti, in Yunnan province of southwest China. Animals, 11, 110. https://doi.org/10.3390/ani11010110
Notas de autor
mlareschi@cepave.edu.ar
Información adicional
redalyc-journal-id: 3220