Comunicaciones
BIOETHANOL PRODUCTION FROM BROKEN RICE GRAINS
PRODUCCIÓN DE BIOETANOL A PARTIR DE GRANOS DE ARROZ ROTOS
PRODUÇÃO DE BIOETANOL A PARTIR DE GRÃOS QUEBRADOS DE ARROZ
BIOETHANOL PRODUCTION FROM BROKEN RICE GRAINS
Interciencia, vol. 43, no. 12, pp. 846-851, 2018
Asociación Interciencia
Received: 11/04/2017
Corrected: 14/11/2018
Accepted: 16/11/2018
Funding
Funding source: Brazilian National Council for Scientific and Technological CNPq
Contract number: CNPq - 309652/ 2016-2
Funding
Funding source: Ministry of Science Technology Innovation and Comunication MCTIC
Contract number: MCTIC - 01. 0144.00/2010
Funding statement: The authors thank the Brazilian National Council for Scientific and Technological (CNPq - 309652/ 2016-2), and the Ministry of Science Technology Innovation and Comunication (MCTIC - 01. 0144.00/2010).
Abstract: The energy produced from biomass is an opportunity, not only to minimize the greenhouse effect, but also to reduce the dependence on fuel oil by diversifying the energy matrix. This study evaluates the production of bioethanol through the use of broken rice grains. To achieve this, the concentration of starch in the broken grain samples was quantified. Subsequently, the milled rice was hydrolyzed and fermented. Hydrolysis was performed with α-amylase and glucoamylase, and the yeast Saccharomyces cerevisiae was employed for the fermentation. The samples were analyzed by high-performance liquid chromatography (HPLC). The analysis indicated an average starch concentration of 80.1% in the broken rice. As a result of hydrolysis, a 70% conversion of starch to glucose was obtained. The entire content of glucose was converted to ethanol, yielding 29.2g of ethanol per 100g of broken rice grains. We conclude that the use of this residual feed crop material is promising to produce bioethanol.
Keywords: Bioethanol , Broken Rice , Fermentation , Hydrolysis.
Resumen: La energía producida a partir de biomasa es una oportunidad no solo para minimizar el efecto invernadero, sino para reducir la dependencia de los combustibles fósiles por medio de la diversificación de la matriz energética. Este estudio evalúa la producción de bioetanol por medio del uso de granos de arroz rotos. Inicialmente fue cuantificada la concentración de almidón en diversas muestras de granos. Subsecuentemente, el arroz molido fue hidrolizado y fermentado. La hidrólisis fue realizada con un complejo enzimático conteniendo actividades de α-amilasa y glucoamilasa. Para la fermentación se utilizó la levadura Saccharomyces cerevisiae. Las muestras de los cultivos fueron analizadas por cromatografía líquida de alta eficiencia (HPLC). El análisis indicó una concentración media de almidón de 80,1% (p/p) en los granos de arroz rotos. Como resultado de la hidrólisis se obtuvo la conversión del 70% de almidón a glucosa. En la fermentación, toda la glucosa fue convertida estequiométricamente en etanol, obteniéndose 29,2g de etanol por 100g de granos de arroz rotos. Por tanto, se constató que el arroz residual de la producción de alimentos puede ser utilizado para la producción de bioetanol.
Resumo: A energia produzida a partir de biomassa é uma oportunidade não somente para minimizar o efeito estufa, mas também para reduzir a dependência dos combustíveis fósseis através da diversificação da matriz energética. Este estudo avalia a produção de bioetanol mediante a utilização de grãos quebrados de arroz. Inicialmente foi quantificada a concentração de amido em diversas amostras de grãos. Subsequentemente, o arroz moído foi hidrolisado e fermentado. A hidrólise foi realizada com um complexo enzimático contendo atividades de α-amilase e glucoamilase. Para a fermentação se utilizou levedura Saccharomyces cerevisiae. As amostras dos cultivos foram analisadas por cromatografia líquida de alta eficiência (HPLC). A análise indicou concentração média de amido de 80,1% (p/p) nos grãos quebrados de arroz. Como resultado da hidrólise se obteve a conversão de 70% de amido para glicose. Na fermentação, toda a glicose foi convertida estequiometricamente em etanol, obtendo-se 29,2g de etanol por 100g de grãos quebrados de arroz. Por tanto, foi constatado que o arroz residual da produção de alimentos pode ser utilizado para a produção de bioetanol.
Introduction
Bioenergy production is an environmental and economic benefit, and Brazil has experience and worldwide recognition in this area. The country has demonstrated the viability of the large-scale production of ethanol (Pereira et al., 2012).
Sustainable use of agricultural biomass will be necessary in the future, and there is a large diversity of vegetable raw materials used in the industry, food processing and others (Gonzalez-Salazar et al., 2014; Aditiya et al., 2016). This will contribute to the decentralization of biofuels production and is attractive to the economy of the regions, considering the use of regional productive capacities and the waste or by-products of different sources of agricultural or industrial production. Among the fuels, bioethanol stands out for having great potential to boost regional economy.
Ethanol can be produced from any polysaccharide that contains sugars or starches, even when structured in larger molecules. Sugar-based raw materials, such as sugar cane, are commonly used in Brazil and in other countries and represent the biomass most widely used for the production of ethanol (Teetor et al., 2011; Dias et al., 2015; Gumienna et al., 2016; Moraes et al., 2016). Starchy materials such as corn, cassava, sweet potato, potato, wheat and rice can also be raw materials used to produce ethanol in high yield (Shanavas et al., 2011; Zhang et al., 2013; Cinelli et al., 2015), and cellulosic materials, which after pretreatment and saccharification, can also be used for alcohol production (Ishola et al., 2015; Tewfik et al., 2015; Ko et al., 2016).
Regarding the potential use of rice for bioethanol production, it is important to consider that the annual grain production is ~606×106t. In this scenario, Brazil contributes with 13,140,900t (2.17% of world production) and stands out as the only non-Asian country among the 10 largest producers (FAO, 2015). The largest consumption of rice is as food in the form of grains that can be cooked or fried, sake and extruded snack foods (Juliano, 2016).
Rice is an excellent source of energy. Due to its high concentration of starch, rice is suitable for the production of bioethanol (Kennedy, 2002; Chu-Ky et al., 2016). Typically, ethanol production is an exothermic reaction in which 51.14g of ethanol and 48.86g of CO2 are generated from 100g of glucose fermentation, which represent the theoretical 100% ethanol yield (Fujieda et al., 2012).
The southern region of Brazil accounts for about 80% of the country’s rice production. The state of Rio Grande do Sul, the largest national producer, yields over 65% of the harvested grain, with an average productivity of 7500 kg·ha-1. This is well above the national average, which is of 4500kg·ha-1 (IRGA, 2015).
New varieties of rice have been studied as energy sources. Some of these varieties have characteristics different from those required for human consumption, such as that known as giant rice, which is hardy but adaptable to the soil and climatic conditions in southern Brazil. Another source is broken rice or grits, which comes from industrial waste processing of grains (Carvalho et al., 2012; EMBRAPA, 2015). In 2008, broken rice accounted for 14% of rice production (Limberger et al., 2009). The use of this waste, which is rich in starch, is mainly as animal feed and may contain contaminants such as rice husks, barnyard-grass seeds (Equinocloa spp.) and angiquinho (Aeschinomene spp.) (Irineo et al., 2007).
This study aims to analyze an alternative for the production of bioethanol fuel in the state of Rio Grande do Sul, through the use of broken rice and waste from the rice production system not intended for human consumption.
Methodology
The rice used in the experiments consisted of 50kg of polished white broken rice grains, types 3 and 4. The samples were ground in a hammer mill to form a flour. Particle size was determined through the use of a sieve shaker with intermittent beats (APBIH7 - PHD equipment) using sieves of 28, 32, 48, 60, 100 and 200 mesh.
Starch content was determined by mass difference after the removal of proteins. To remove the proteins 15g of the sample were mixed with 6 volumes of NaOH 0.16% and allowed to stand for 24h. The remaining solid was collected and mixed again with the same basic solution. The procedure was repeated until the biuret test (1ml of the supernatant over 4ml of biuret solution) showed no positive signs for the presence of proteins (Zaia et al., 1998). The reduction of proteins in the supernatant was observed after about six washes, when the color the solution changed from the initial purple to blue.
Hydrolysis / Saccharification
For the liquefaction and saccharification an α-amylase (trade name Liquozyme Supra 2.2X) obtained from Bacillus licheniformis, and the exo-1,4-α-D-glucosidase enzyme (glucoamylase; 1,4-α-D-glucan glucohydrolase; commercially known as AMG 300L) obtained from Aspergillus niger were used. The methodology followed the basic instructions provided by the product manufacturer, which contained information on the temperature optimization and enzyme concentration to be applied in the hydrolysis (Novozymes, 2008).
The hydrolysis experiments were performed with a 5% load (w/v) in aqueous medium with controlled pH levels. Initially, the sample was pretreated by boiling for 0.5h. Hydrolyses were conducted in a water bath under stirring, at pH 6, for 2h, with α-amylase at concentrations of 0.378 and 1.134 KNU (α-amylase units with activity of 300 KNU/g) at 70 and 90ºC. Afterwards, the pH was adjusted to 4.5 and the temperature changed to 50 and 60ºC, and second enzyme was added at concentrations of 0.45 and 0.75 AGU (amyloglucosidase units with activity of 300 AGU/ml). The second stage was also conducted for 2h in a water bath under stirring. Samples were taken at regular time intervals for sugar analysis using high performance liquid chromatography (HPLC).
Fermentation
After saccharification of the samples, the hydrolyzate was fermented with Saccharomyces cerevisiae (Angel Thermal Resistance Alcohol Yeast) with the purpose of producing bioethanol, according to the methodology of Vancov et al. (2015). Fermentation was performed in 100ml autoclaved bottles, to which 50ml of the filtered hydrolyzate, 0.1g monobasic potassium phosphate, 0.025g of yeast extract and 0.05g magnesium sulfate were added. The vials were autoclaved at 121ºC for 30min. After cooling, 0.25g of yeast was added, and the containers were incubated in a shaker (Marconi, MA420) at 30ºC and 100rpm for 24h. Samples were taken at regular time intervals for sugars and bioethanol determination by HPLC.
Sugars and bioethanol determination
The identification and quantification of sugar and bioethanol were conducted on a high performance liquid chromatograph (Shimadzu) with refractive index detector (RID-A10), RHM (H+Monosaccharides) column (300×7.8mm). The analysis was conducted at 85ºC using an ultrapure water flow of 0.6ml·min-1 as the mobile phase, i.e., a maximum pressure of 210kg·cm-2 and analysis time of 20min. The volume injected sample was 5μl. The samples were previously centrifuged at 14,100rpm in a centrifuge (Mini Spin - Eppendorf) and filtered through a 0.45μm filter (Chromafil Xtra - RC 45/25).
Statistical analysis
Statistical analysis of the data was performed using the Kruskal-Wallis test and Assistat Software version 7.7 Beta. Choosing the test was based on comparing groups, with data being categorical, non-parametric, ordinal, unpaired, independent and with a population >2.
Results and Discussion
Hydrolysis
The hydrolysis was conducted with a broken rice sample that showed 80.1 ±1.2% of starch. These starch contents were close to those found by Frei et al. (2003) for whole grain rice (74.12%), polished white rice (87.58%) and polished parboiled rice (85.08%). The starch content was regarded as the total to be converted in the hydrolysis to glucose. The broken rice sample was hydrolyzed with two enzymes in sequence as shown in Table I.
According to de la Hera et al. (2013) α-amylase affinity for native starches from rice flour is dependent on the particle size of starch. The particle size diminishes when milled and the enzyme actions are favored due to the exposure and superficial interaction between starch and enzyme. For this reason, the broken rice samples were milled in a hammer mill and evaluated for particle size distribution. Particle size distribution of rice flour showed mainly two particle size fractions: 500-300μm and 300-200μm, comprising 75.9 and 21.9%, respectively, of the total mass of milled broken rice. These particle size ranges are close to those used by Gohel and Duan (2012a) and lower than that used by Gohel and Duan (2012b), and are the ones that are applied to facilitate gelatinization of the starch.
According to Syahariza et al. (2013), when the starch is heated, granule gelatinization occurs, which breaks the hydrogen bonds of the molecule, and disintegrates the semi-crystalline grain structure. Consequently, the starch molecules are susceptible to the activity of the enzymes, which have easier access to the inside of the granules.
Thus, preliminary studies defined the hydrolysis temperature according to the enzyme manufacturer’s directions and the condition of thermal pretreatment of the rice to gelatinize and facilitate the hydrolysis (Amaral et al., 2007; Denardin and da Silva, 2008). The reaction temperatures of the first enzyme (α-amylase) were pre-tested to optimize the proper temperature for the liquefaction of starch. The result of these tests showed a better uniformity of the sample before the addition of the enzymes, at the boiling point of water. Table I shows that experiments with the first enzyme at 90ºC exhibited a higher conversion as compared to the experiments conducted at 70ºC. However, there was no improvement in yield, involving the effect of the temperature on the second enzyme, when compared at 50ºC and 60ºC. Both comparisons were made in the same hydrolysis conditions, except for the change in temperature. Moreover, there are indications that the effect of the concentration on the two enzymes influences the yield of the hydrolysis. However, these performance improvements are not significant.
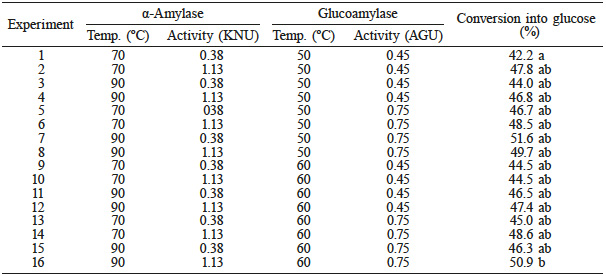
a and b= indicate similarity between results.
Statistical analysis showed that, considering as a null hypothesis (H0) that the treatments come from the same population and at the level of 5% probability, rejecting H0 and with H and H-crit defined, there were two large groups with similarities among the 16 entries in the experimental design. Of the statistical analysis of multiple comparisons, only experiments 1 and 16, which are the extremes of concentration and temperature in the experimental design, are significantly different. Thus, for the continuity of the experiments, experiment 7 was selected, since it apparently showed a higher conversion to glucose (51.6%). This hydrolysis condition was carried out with the lowest concentration of α-amylase and the highest concentration of glucoamylase, which can be an important factor in ensuring a higher conversion of starch to glucose, since the effect of the enzymes to form glucose complement each other. This reaction condition, although not significantly different from other conditions studied, was chosen for the production of bioethanol.
The results found are due to the α-amylase action, which hydrolyzes starch to disaccharides, transforming polysaccharides by breaking α-1,4-glycosidic and glucoamylase bonds which hydrolyses α-1, 4 and the α-1,6 bonds in the liquefied starch. Thus, rice starch, which is equivalent to glucose bound by α-1,4-linked, is liquefied and saccharified by amylases (Fujieda et al., 2012).
The ratio of sugars along the hydrolysis at controlled pH can be seen in Figures 1 and 2. The conversion of glucose was mainly observed after the addition of the glucoamylase. It is noted that the hydrolysis rate depended on the type of bond broken and the size of the polysaccharide chain; for example, α-1,4 bonds are hydrolysed faster than α-1,6 bonds; and therefore, maltotriose and maltose formed in the first stage tend to be hydrolyzed at a lower rate than the larger oligosaccharide (Denardin and da Silva, 2008; Jonathan et al., 2015; Jonathan et al., 2016). In this regard, it is noted in Figure 1 that the compounds formed in the hydrolysis with α-amylase do not include glucose and form mainly maltose.
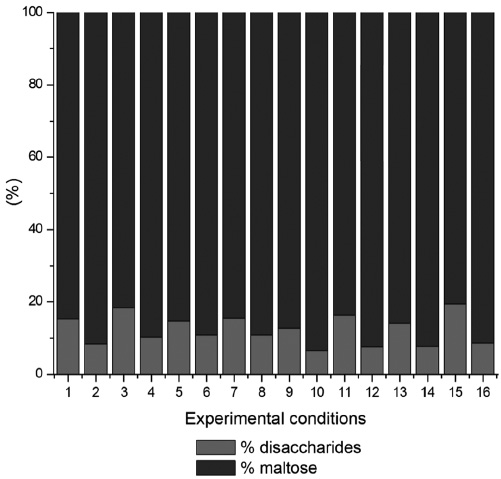
Figure 1
Relationship of disaccharides formed by the hydrolysis with α-amylase at pH 6
In Figure 2 it can be seen that with the addition of glucoamylase, glucose was formed, it being understood that this enzyme performs the breaking of disaccharides such as maltose. However, being free of transglicosidade activity, this enzyme may cause the formation of isomaltose and panose by transferring glucose molecules in α-1,4 to α 1,6-position, which may result in lower yields of reaction (Novozymes, 2002). In some experiments carried out in a buffered medium at the indicated pH for each enzyme, the conversion was shown to decrease.
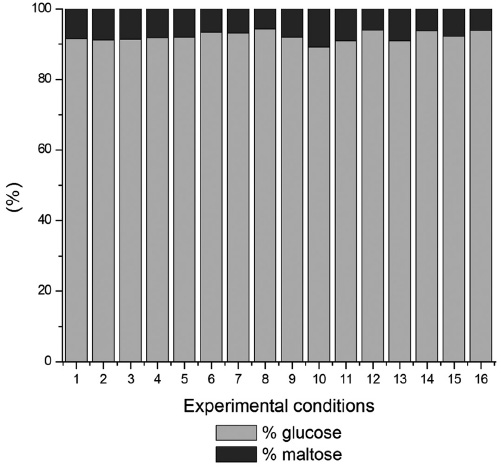
Figure 2
Relationship of the compounds formed in the hydrolysis with α-amylase (pH 6) and glucoamylase (pH 4.5) in sequence
Whereas the addition of buffer in the medium can be a determining factor for the economic viability of the process, pH was adjusted to 6 and 4.5 for the α-amylase and glucoamylase, respectively, with the aid of a phosphoric solution. The results are shown in Figure 3 and correspond to the temperatures and enzyme concentrations according to experiment 7 of Table I. In this condition, a higher conversion of starch to glucose was observed. No buffer was used.
The maximum conversion achieved was 70% within 5h of hydrolysis, in which the entire disaccharide content had been converted into glucose. Furthermore, it was observed that the average conversion of starch to glucose at the time of addition of the glucoamylase was 5.2% and after 1h it reached 48.7% of glucose. The particle size of broken rice flour can be a factor that hinders the complete conversion of starch to glucose, since the larger sample fraction is 500-300μm.
The relationship between glucose and maltose during hydrolysis (Figure 3) is also a factor which shows that the conversion rate was high. It was observed that the addition of glucoamylase in the medium nearly quadrupled the formation of glucose from maltose, passing a ratio of 0.42 to 1.51. These results are consistent with those of Surmely et al. (2001) with a yield of 110%, due to the formation of dextrose, which also corresponds to the percentage of water molecules entering the starch chains.
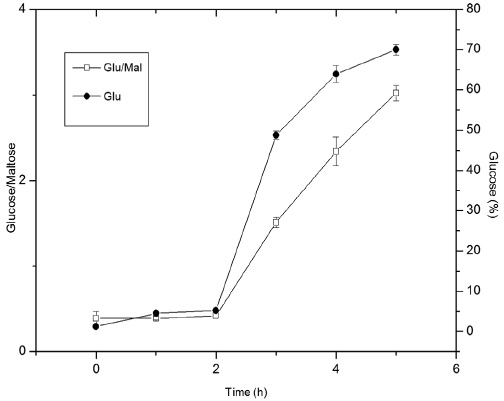
Figure 3
Conversion (%) of starch present in broken rice into glucose, by employing α-amylase (90ºC / 0.38 KNU / pH6) and glucoamylase (50° C / 0.75 AGU / pH4.5) in sequence, and the relationship between the content of glucose and maltose along hydrolysis employing α-amylase and glucoamylase enzymes in sequence (n = 6)
As for the activity of enzymes for hydrolysis, Yamada et al. (2011) suggest that, compared with other raw materials, the activity of amylase and glucoamylases are high in whole grain rice. Since broken rice is from polished rice, there are no benefits from metals that aid in stabilizing the α-amylase, as noted by the author.
High conversions can also be found at temperatures and from various starchy raw materials. In these cases, capital and operational costs are approximately 41% and 51% lower, respectively. For rice, 51% of hydrolysis efficiency with pancreatic α-amylase and glucoamylase from A. niger (37 C, pH 6.0, 24h) has been reported (Cinelli et al., 2015).
Fermentation
Fermentation was carried out from the broken rice hydrolyzate and the results were shown in Figure 4. For 100g of initial biomass, 29.2g of ethanol were obtained, corresponding to a yield of 104% of bioethanol produced from glucose. After 6h of fermentation, the glucose concentration reached near zero from total consumption, and the formation of the maximum concentration of ethanol was reached at 16h. Sugar was totally converted to bioethanol by yeast through the glycolytic pathway (Fujieda et al., 2012).
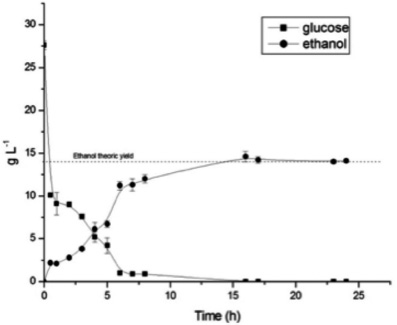
Figure 4
Glucose and bioethanol curve in the fermentation of broken rice hydrolyzate
The conversion of bioethanol into glucose reached the theoretical yield. The conversion percentage found was comparable to those in other studies of rice. According to Fujieda et al. (2012) a yield of 89.5% ethanol was possible from rice, and Saga et al. (2010) reached efficiencies of 95 and 90% yields in hydrolysis and fermentation, respectively, in a high-yield rice plant.
It is interesting that the limiting step in ethanol production is not fermentation, but saccharification of starch. For fermentation there is no formation of inhibitors and saccharification conduces to glucose production, sugar used for the production of ethanol by the yeast selected in the relation previously presented at 0.51 (ethanol / glucose).
To make improvements, it is still possible to change the process to make it more sustainable and economically competitive, employing some innovations such as fermentation with the removal of ethanol in situ, through a stripping column and immobilization of yeast cells (Bai et al., 2008).
Conclusion
We conclude that broken rice can be used as an alternative for the production of bioethanol. The conditions to produce bioethanol from broken rice were optimized with the use of two enzymes, α-amylase (90ºC / 0.38 KNU / pH 6) and glucoamylase (50ºC / 0.75 AGU / pH 4.5). The optimized conditions led to 70% conversion of starch to glucose and some disaccharides. The hydrolyzed starch was fully converted into ethanol (104% theoretical yield).
The results obtained at laboratory-scale are promising if applied at a pilot scale, which could assess the economic feasibility of using broken rice for the production of bioethanol.
It is emphasized that this proposal looks to diversify the production of biofuel in rice-producing regions, as the volume of broken rice is large and bioethanol production may lead to profits for rice producers and promote decentralization in energy production.
Acknowledgements
The authors thank the Rio Grande Institute of Rice (Instituto Rio-grandense do Arroz; IRGA), the Novozymes Latin America Ltda., the Brazilian National Council for Scientific and Technological (CNPq - 309652/ 2016-2), the Mixed Cooperative of Tobacco Growers Brazil Ltd. (Cooperfumos), the Ministry of Science Technology Innovation and Comunication (MCTIC - 01. 0144.00/2010), the Science and Technology Regional Park (CEPPOB-TecnoUnisc), Science, Technology and Economic Development Secretary (SDECT-RS) and the FAP - UNISC.
REFERENCES
Aditiya HB, Chong WT, Mahlia TMI, Sebayang AH, Berawi MA, Nur H (2016) Second generation bioethanol potential from selected Malaysia’s biodiversity biomasses: A review. Waste Manag. 47A: 46-61.
Amaral LD, Gaspar M, Costa PMF, Aidar MPM, Buckeridge MS (2007) Novo método enzimático rápido e sensível de extração e dosagem de amido em materiais vegetais. Hoehnea 34: 425-431.
Bai FW, Anderson WA, Moo-Young M (2008) Ethanol fermentation technologies from sugar and starch feedstocks. Biotechnol. Adv. 26: 89-105.
Carvalho AV, Bassinello PZ, Mattietto RDA, Carvalho RN, Rios ADO, Seccadio LL (2012) Processamento e caracterização de snack extrudado a partir de farinhas de quirera de arroz e de bandinha de feijão. Braz. J. Food Technol. 15: 72-83.
Chu KS, Pham TH, Bui KLT, Nguyen TT, Pham KD, Nguyen HDT, Luong HN, Tu VP, Nguyen TH, Ho PH, Le TM (2016) Simultaneous liquefaction, saccharification and fermentation at very high gravity of rice at pilot scale for potable ethanol production and distillers dried grains composition. Food Bioprod. Proc. 98: 79-85.
Cinelli BA, Castilho LR, Freire DMG, Castro AM (2015) A brief review on the emerging technology of ethanol production by cold hydrolysis of raw starch. Fuel 150: 721-729.
De La Hera E, Gomez M, Rosell CM (2013) Particle size distribution of rice flour affecting the starch enzymatic hydrolysis and hydration properties. Carbohydr. Polym. 98: 421-427.
Denardin CC, Da Silva LP (2008) Estrutura dos grânulos de amido e sua relação com propriedades fisicoquímicas. Ciênc. Rural 39: 945-954.
Dias MODS, Maciel Filho R, Mantelatto PE, Cavalett O, Rossell CEV, Bonomi A, Leal MRLV (2015) Sugarcane processing for ethanol and sugar in Brazil. Environ. Devel. 15: 35-51.
EMBRAPA (2015) Empresa Brasileira de Pesquisa Agropecuária. http://www.embrapa.br (Cons. 08/11/2015).
FAO (2015) Statistical databases. Food and Agriculture Organization. United Nations. http://www.fao.org (Cons. 15/06/2015).
Frei M, Siddhuraju P, Becker K (2003) Studies on in vitro starch digestibility and the glycemic index of six different indigenous rice cultivars from the Philippines. Food Chem. 83:395-402.
Fujieda T, Kitamura Y, Yamasaki H, Furuishi A, Motobayashi K (2012) An experimental study on whole paddy saccharification and fermentation for rice ethanol production. Biomass Bioenergy 44: 35-141.
Gohel V, Duan G (2012a) Conventional process for ethanol production from Indian broken rice and pearl millet. Bioproc. Biosyst. Eng. 35: 1297-1308.
Gohel V, Duan G (2012b) No-cook process for ethanol production using Indian broken rice and pearl millet. Int. J. Microbiol. 2012:680232. doi: 10.1155/2012/680232
Gonzalez-Salazar MA, Morini M, Pinelli M, Spina PR, Venturini M, Finkenrath M, Poganietz WR (2014) Methodology for estimating biomass energy potential and its application to Colombia. Appl. Energy 136: 781-796.
Gumienna M, Szwengiel A, Szczepańska-Alvarez A, Szambelan K, Lasik-Kurdyś M, Czarnecki Z, Sitarski A (2016) The impact of sugar beet varieties and cultivation conditions on ethanol productivity. Biomass Bioenergy 85: 228-234.
IRGA (2015) Instituto Rio Grandense do Arroz. http://www.irga.rs.gov.br (Cons. 26/06/2015).
Irineo BDSBJ, De Toledoii ZGSP, Vieiraii EGXTA, Gonçalvesii EC (2007) Dietas para frangos de corte contendo quirera de arroz. Ciênc. Rural 37: 1423-1429.
Ishola MM, Brandberg T, Taherzadeh MJ (2015) Simultaneous glucose and xylose utilization for improved ethanol production from lignocellulosic biomass through SSFF with encapsulated yeast. Biomass Bioenergy 77: 192-199.
Jonathan MC, Van Brussel M, Scheffers MS, Kabel MA (2015) Characterisation of branched gluco-oligosaccharides to study the mode-of-action of a glucoamylase from Hypocrea jecorina. Carbohydr. Polym. 132: 59-66.
Jonathan MC, Van Brussel M, Scheffers MS, Kabel MA (2016) Different action patterns of glucoamylases on branched gluco-oligosaccharides from amylopectin. Carbohydr. Polymers 143: 198-203.
Juliano BO (2016) Rice: Overview. In Encyclopedia of Food Grains. 2nd ed. Academic Press. Oxford, RU. pp. 125-129.
Kennedy G (2002) Nutrient impact assessment of rice in major rice-consuming countries. Int. Rice Commiss. Newslett. 51: 33-42.
Ko JK, Um Y, Woo HM, Kim KH, Lee SM (2016). Ethanol production from lignocellulosic hydrolysates using engineered Saccharomyces cerevisiae harboring xylose isomerase-based pathway. Bioresource Technol. 209: 290-296.
Limberger VM, Comarela CG, Patias LD, Brum FB, Emanuelli T, Silva LPD (2009) Produção de salgadinho extrusado de quirera de arroz para uso na indústria de alimentos. Ciênc. Rural 39: 2590-2594.
Moraes MaFDD, Bacchi MRP, Caldarelli CE (2016). Accelerated growth of the sugarcane, sugar, and ethanol sectors in Brazil (2000-2008): Effects on municipal gross domestic product per capita in the south-central region. Biomass Bioenergy 91: 116-125.
Novozymes (2002) Ficha Técnica - Enzima AMG 300L.
Novozymes (2008) Ficha de informação do produto - Liquozyme Supra 2.2X.
Pereira MG, Camacho CF, Freitas MaV, Silva NFD (2012) The renewable energy market in Brazil: Current status and potential. Renew. Sustain. Energy Rev. 16: 3786-3802.
Saga K, Imou K, Yokoyama S, Minowa T (2010) Net energy analysis of bioethanol production system from high-yield rice plant in Japan. Appl. Energy 87: 2164-2168.
Shanavas S, Padmaja G, Moorthy SN, Sajeev MS, Sheriff JT (2011) Process optimization for bioethanol production from cassava starch using novel eco-friendly enzymes. Biomass Bioenergy 35: 901-909.
Surmely R, Alvarez H, Cereda MP, Vilpoux OF (2001) Hidrólise do amido. Culturas de tuberosas amiláceas latino americanas. Em Tecnologia, usos e potencialidades de tuberosas amiláceas Latino Americanas: Fundação Cargil. pp. 377-448.
Syahariza ZA, Sar S, Hasjim J, Tizzotti MJ, Gilbert RG (2013) The importance of amylose and amylopectin fine structures for starch digestibility in cooked rice grains. Food Chem. 136: 742-749.
Teetor VH, Duclos DV, Wittenberg ET, Young KM, Chawhuaymak J, Riley MR, Ray DT (2011) Effects of planting date on sugar and ethanol yield of sweet sorghum grown in Arizona. Indust. Crops Prod. 34: 1293-1300.
Tewfik SR, Abulnour AGMG, Sorour MH (2015) Techno-economic and environmental aspects of the production of medium scale ligno-cellulosic ethanol under Egyptian conditions. Egyp. J. Petrol. 24: 375-381.
Vancov T, Schneider RCS, Palmer J, Mcintosh S, Stuetz R (2015) Potential use of feedlot cattle manure for bioethanol production. Bioresource Technol. 183: 120-128.
Yamada R, Yamakawa S-I, Tanaka T, Ogino C, Fukuda H, Kondo A (2011) Direct and efficient ethanol production from high- yielding rice using a Saccharomyces cerevisiae strain that express amylases. Enz. Microb. Technol. 48: 393-396.
Zaia DA, Zaia C, Lichtig J (1998) Determinação de proteínas totais via espectrofometria: vantagens e desvantagens dos métodos existentes. Quím. Nova 21: 787-793.
Zhang P, Chen C, Shen Y, Ding T, Ma D, Hua Z, Sun D (2013) Starch saccharification and fermentation of uncooked sweet potato roots for fuel ethanol production. Bioresource Technol. 128: 835-838.
Author notes
Alternative link
https://www.interciencia.net/wp-content/uploads/2018/12/846-SCHNEIDER-43_12.pdf (pdf)