Comunicaciones
Recepción: 16 Marzo 2017
Corregido: 07 Julio 2019
Aprobación: 09 Julio 2019
Abstract: We studied the effect of foliar applications (0, 0.4, 0.8, 1.2 and 1.6g·l-1) of boron on the concentrations of B, carbohydrates and total soluble amino acids in the leaf and on the thickness of the cell walls of buds in 2.5-year-old avocado cv. Hass trees. Penetration of foliar B application (0.8g·l-1) occurred, but due to the natural fluorescence of the leaf, it was not possible to distinguish the path that the fertilizer followed, nor the time needed for absorption. The increasing levels of B applied in three successive occasions with 20 days intervals did not affect the concentration of this element in leaves, sampled 15 days after the application. There was no effect of B on the concentration of carbohydrates. After the second sampling, there was a positive relationship between the concentration of B applied and the amino acids concentration. Boron increased the cell wall thickness of the buds; 1.2 and 1.6g·l-1 doses increased cell wall thickness in the cortex by 66% and 84% respectively, while in the pith this increase was 71% and 77%, respectively, compared with the control. Based on the enhanced cell wall thickness observed, a concentration of B at 1.2g·l-1 is recommended for foliar application in avocado cv. Hass trees.
Keywords: Carbohydrates, Cell Structure, Lauraceae, Persea americana .
Resumo: Foi estudado o efeito da aplicação foliar do boro (0; 0,4; 0,8; 1,2 e 1,6g·l-1) nas concentrações de B, carboidratos e aminoácidos solúveis totais na folha, bem como na espessura das paredes celulares de brotos em árvores de abacate cv. Hass de 2,5 anos de idade. Demonstrou-se que ocorreu penetração foliar de B, mas devido à fluorescência natural da folha, não foi possível distinguir o caminho que o fertilizante seguiu ou o tempo necessário para a absorção. Os níveis crescentes de B, aplicados sucessivamente com 20 dias de diferencia não afetaram a concentração deste elemento, medida 15 dias despois da aplicação. Não houve efeito de B sobre a nas folhas concentração de carboidratos. Aplicações e amostragem foram repetidas cada 15 dias e depois da segunda amostragem, houve uma relação positiva entre a concentração de B aplicada e a concentração de aminoácidos. O B aumentou a espessura da parede celular das gemas; 1,2 e 1,6g·l-1 doses aumentaram a espessura em 66% e 84% das paredes celulares no córtex, respectivamente, enquanto na medula este aumento foi de 71% e 77%, respectivamente, em comparação com o controle. Com base na maior espessura da parede celular observada, recomenda-se uma concentração de B de 1,2g·l-1 para aplicação foliar em árvores de abacate cv. Hass.
Resumen: Se estudió el efecto de la aplicación foliar de boro (0; 0,4; 0,8; 1,2 e 1,6g·l-1) en las concentraciones de B, carbohidratos y aminoácidos solubles totales en la hoja, así como en el grosor de la pared celular en brotes de árboles de aguacate cv. Hass de 2,5 años. Hubo penetración del B (0,8g·l-1) aplicado a las hojas, pero debido a la fluorescencia natural de la hoja no se pudo distinguir la vía de penetración o el tiempo requerido para su absorción. Niveles crecientes de B aplicado en tres ocasiones sucesivas separadas por 20 días no afectaron la concentración del elemento en las hojas, medido a los 15 días de cada aplicación. No se detectó efecto del B en la concentración de carbohidratos. Hubo una relación positiva entre la concentración de B aplicada después de dos y tres aspersiones foliares y la concentración de aminoácidos. El B aumentó el grosor de la pared celular de los brotes; dosis de 1,2 y 1,6g·l-1 de B aumentaron el grosor en 66% y 84% de la pared celular, respectivamente, mientras que la médula aumentó en 71% y 77%, respectivamente, en comparación con el control. Con base en el engrosamiento de la pared celular observado, se recomienda una concentración de B de 1,2g·l-1 para la aplicación foliar cerca del período de floración en árboles de aguacate cv. Hass.
Introduction
Avocado (Persea americana) is a member of the Lauraceae family, a tree native to South-Central Mexico. Its fruit is commercially valuable and is mainly produced in the Americas. In 2017, world production of avocados was 6.048×106t, with Mexico alone accounting for 33.6% of the total (2.029×106t). Other major producers include the Dominican Republic, Peru, Indonesia and Colombia, together totaling nearly 29.46% of world production (FAOSTAT, 2019). Nonetheless, the importance of the avocado goes beyond the economic aspect. For the human being, it represents a source of fiber, lipid-soluble antioxidants and monounsaturated fatty acids. In addition, it has important quantities of both folate and potassium. It represents a promising fruit that helps to cover the dietary needs of pregnant and lactating women, as well as of babies and young children (Comefor et al., 2016a, b).
Avocado fruit production may be influenced by several factors including rootstocks and cultivars, pest and disease management, fertilization, irrigation, and physical and chemical tree growth manipulation, among others (Könne, 1988). Nutritional deficiencies may lead to increased flower and fruit abscission (Garner and Lovatt, 2008). Hence, if any of the essential elements are deficient or in forms that are not bioavailable, the yield and quality of avocado decreases considerably.
In particular, boron (B) is a formative element of plant structures, and its deficiency causes developmental abnormalities in various tissues (Reguera et al., 2010). In terms of plant structures, the best-established role of B is as a covalent bridge between pectin molecules. Such linking decreases wall porosity and modifies the wall's biomechanical properties, thickness and growth (Caffal and Mohnen, 2009; Chormova and Fry, 2016).
Carbohydrate metabolism is possibly the most affected process under deficiency of this element. In B-deficient plants, the products of assimilation formed in the leaves are transported more slowly, such that a sugar deficit occurs in the meristematic tissues of the roots and in the upper parts of the plant, whereas in photosynthetically active leaves, these products accumulate. Under conditions of sufficient B, the assimilation products are transported as B complexes (sugar-borates; Alcántar et al., 2016).
The need for B varies considerably among plant species; amounts that can be suitable for some species may be toxic to others. The average concentration in plant tissues is ~20mg·kg-1 of dry material (Alcántar et al., 2016). According to Maldonado-Torres et al. (2007), in high-yield (>20t·ha-1) ‘Hass’ avocado trees, the optimal concentration of B in leaf tissues ranges from 126 to 352mg·kg-1. The participation of B in the formation and growth of pollen tubes in this species has been fully demonstrated. Thus, B deficiencies can result in great losses in avocado plantations by causing damage to the fruit such as deformation, browning, and hardening (Lovatt, 1999).
In this study we first aimed to determine the paths and times of B penetration in avocado leaves after foliar applications of B. Subsequently, we evaluated the effects of B application on the B, carbohydrate and amino acid concentrations in leaf tissues, as well as the thickness of the cell wall in leaves.
Materials and Methods
The work was conducted under greenhouse conditions with 2.5 year-old avocado cv. Hass trees from Uruapan, Michoacán, Mexico. The trees were transplanted into 40 liters black polyethylene bags containing 5kg of peat. The trees were irrigated with tap water every four days for 12 weeks. Subsequently, to ensure an adequate supply of nutrients, the trees were irrigated every eight days with 100% Steiner universal nutrient solution (Steiner, 1984) supplemented with micronutrients (except boron).
Experiment 1. Boron penetration paths
To identify the B penetration path in the leaf, Fluorescent Brightener 28 stain was used. The active ingredient in this stain is calcofluor M2R, at a concentration of 0.05%. The stain was mixed with H3BO3 at a B concentration of 0.8g·l-1, and supplemented with 0.1% Tween 20 as a surfactant. The pH of the solution was adjusted to 5 with NaOH. Foliar application was carried out at 7:00, and the leaves were sampled 15, 30, 120, 240, 360 and 480min after application.
Sections were taken from the collected leaves. Each section, cut from the middle of the leaf, measured ~0.5cm long by 0.3cm wide (Sandoval, 2005). The samples were placed at the bottom of a freezing microtome (AO Instrument CO, model 880, USA) where sections of 25µm thickness were cut. The sections were mounted between slide and cover slip and observed with a fluorescent microscope under an UV light number 2 filter (Nava-Sánchez et al., 2004). In order to measure the frequency of observed fluorescent signals in each of the evaluated treatments, we developed the following scale: +++ indicated fluorescence in at least 75% of the sections; ++ indicated fluorescence observed between 74 and 50% of the sections; and + indicated fluorescence observed in less than 50% of the sections.
Experiment 2. Foliar supply of boron
In a second stage of our study, three foliar sprayings of H3BO3 were carried out at 20 day intervals. The following concentrations of B were applied in the foliar sprays: 0, 0.4, 0.8, 1.2 and 1.6g·l-1. The prayed solutions were supplemented with 0.1%Tween 20, and pH was adjusted to 5 with NaOH in all cases.
Fifteen days after each foliar application of B, newly mature leaves were sampled to determine the total concentrations of B, carbohydrates and amino acids in the leaves. The foliar concentration of B was determined using the wet digestion method reported by Alcántar and Sandoval (1999). Readings of extracts obtained after digestion and filtration were determined by inductively coupled plasma atomic emission spectroscopy (ICP-AES, VARIAN™ model Liberty II, Australia).
The carbohydrate concentration was determined by the anthrone method (Southgate, 1976). The total soluble amino acid concentration was determined in leaves at the moment of slicing by means of ethanol extraction according to the methodology of Geiger et al. (1998) using the ninhydrin method (Moore and Stein, 1954). Leucine was used to prepare the standard curve for concentrations from 0 to 250μg·ml-1. Carbohydrate and amino acid extracts were read in a spectrophotometer (Spectronic, Genesys™ Series 10 UV-Vis, USA) at wavelengths of 600 and 570nm, respectively.
Cuts to evaluate the cell wall thickness were made on 8 buds per treatment, collected 15 days after the final application. The techniques for fixation, dehydration, inclusion in paraffin and staining of the cuts in fast green Safranin for light microscopy were carried out as described by López et al. (2005). Cuts of 10µm thickness were observed at 40X under a light microscope (Carl Zeiss, Photomicroscope III, Germany), and photographs were obtained. The cell wall thickness was measured with the software program Image Tool 3.0.
A completely randomized experimental design was used. Each treatment testing B effects had eight replications, and a ‘Hass’ avocado tree was used as the experimental unit. The recorded data were statistically analyzed using analysis of variance and Tukey’s range test (α=0.05).
Results and Discussion
Foliar penetration paths of boron
Leaf cuts observed under the microscope (Figure 1) showed differences in fluorescence levels at 15 and 30min between the leaves sprayed only with water (a and b) and those treated with the calcofluor-boron solution (A and B). There were no differences observed between leaves sprayed with and without calcofluor in cuts made 120min after treatment application (Figure 1). Indeed, we could observe that foliar B penetration occurred, but due to the natural fluorescence of the leaf, it was not possible to distinguish the path that the fertilizer followed, or the time needed for absorption.
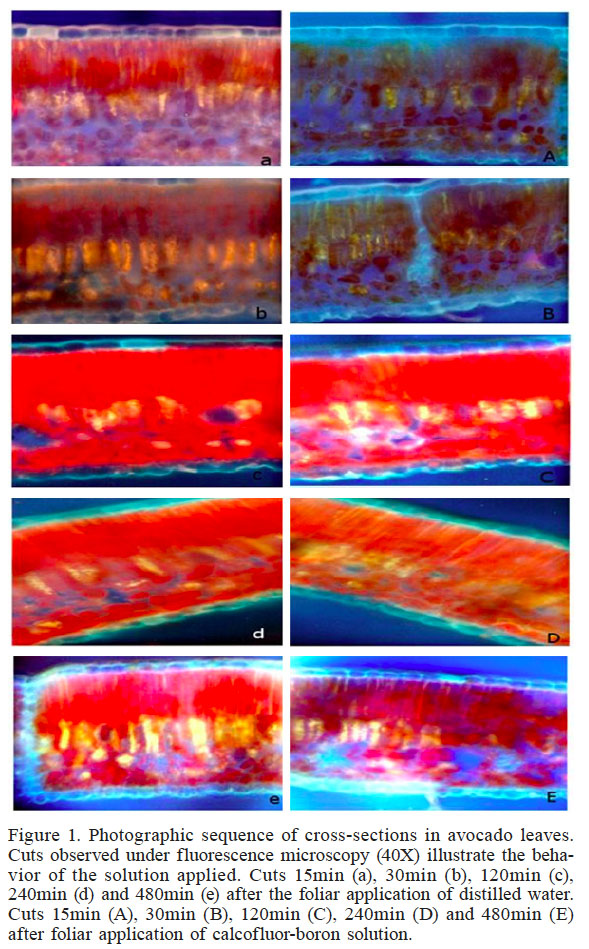
Figure 1.
Photographic sequence of cross-sections in avocado leaves. Cuts observed under fluorescence microscopy (40X) illustrate the behavior of the solution applied. Cuts 15min (a), 30min (b), 120min (c), 240min (d) and 480min (e) after the foliar application of distilled water. Cuts 15min (A), 30min (B), 120min (C), 240min (D) and 480min (E) after foliar application of calcofluor-boron solution.
Table I shows the fluorescence scale used, displaying an average of 36 cuts for each time interval of leaves sprayed only with distilled water (control) and leaves sprayed with B solution. In the first group of leaves, fluorescence is observed in more than 75% of the cuts, both in the palisade and the spongy parenchyma cells. The same pattern was observed following treatment with calcofluor 30min after application. In general, the fluorescence was very similar between the solution with B and calcofluor and the control where only distilled water was applied at 120min, as confirmed by the images in Figure 1. In onion leaves, Nava-Sánchez et al. (2004) observed foliar absorption of urea using the same stain. The concentration of urea in the foliar solution was 0.5%, while in this study the concentration of B was 0.05%. This concentration was very low because B is a micronutrient, and a narrow concentration-range between boron deficiency and excess has been reported (Reid et al., 2004; Chormova and Fry, 2016). Consequently, because of the low B concentration applied, we were not able to detect differences among treatments.
FOLIAR PENETRATION OF DISTILLED WATER AND FOLIAR B SOLUTION WITH CALCOFLUOR IN AVOCADO LEAVES
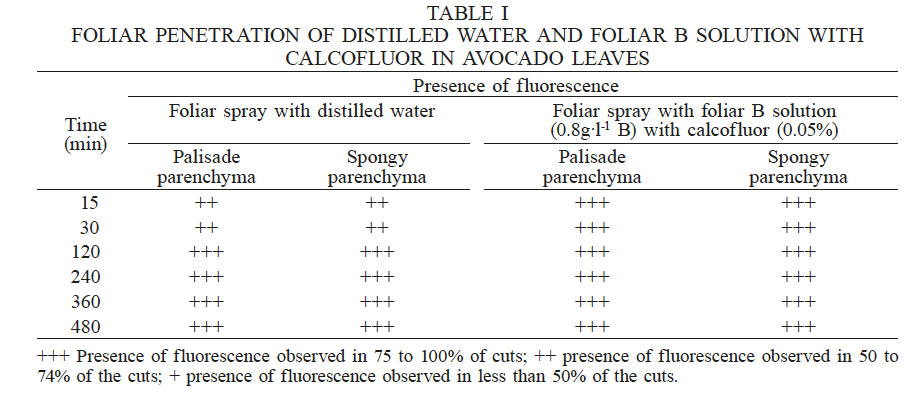
+++ Presence of fluorescence observed in 75 to 100% of cuts; ++ presence of fluorescence observed in 50 to 74% of the cuts; + presence of fluorescence observed in less than 50% of the cuts.
Foliar concentration of boron
The concentrations of B recorded in the leaves ranged between 0.148 and 0.203mg·g-1 of dry material (Figure 2), which is considered optimal in orchards of avocado cv. Hass with yields exceeding 20t·ha-1, according to Maldonado-Torres et al. (2007).
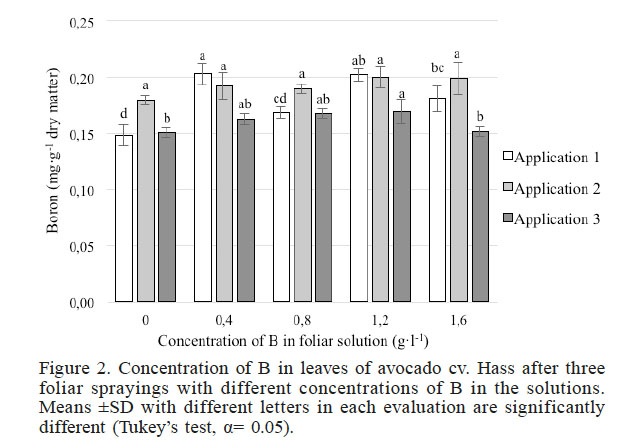
Figure 2
Concentration of B in leaves of avocado cv. Hass after three foliar sprayings with different concentrations of B in the solutions. Means ±SD with different letters in each evaluation are significantly different (Tukey’s test, α= 0.05).
In the first sampling, performed after the first B spray application, analysis of variance indicated that the B concentration in the leaves was significantly different among treatments. Means comparison showed the lowest foliar B concentration in the control treatment, whereas the highest concentrations occurred in treatments with a foliar supply of 0.4 and 1.2g·l-1 B. According to the results of the first sampling (Figure 2), it is evident that foliar penetration occurred because the treatments that received B had a higher concentration of this element compared with the control. However, we were unable to demonstrate that penetration (Figure 1) was positively correlated with the concentration of B in the foliar solution.
In the second sampling, after foliar spray of B solution 20 days later, there were no significant differences among treatments. However, the greatest concentration of B in leaves of plants treated with this element is clear (Figure 2). In this sampling, an increase in foliar B concentration was observed only in the control treatment compared with the first sampling.
Foliar B concentrations determined after three foliar sprays showed a positive relationship between foliar B concentration and foliar concentration supplied within the range of 0 to 1.2g·l-1. Leaves treated with the highest dose of B in this study (1.6g·l-1) had concentrations of B lower than the control leaves. Similarly, in sunflower (Helianthus annuus L.), different foliar B spray rates (0, 0.03, 0.07, 0.13 and 1.3kg·ha-1 of B) increased the B concentration in various parts of the plant tops, even those that were developed subsequent to the spray applications; however, the B concentration in the roots did not increase (Asad et al., 2003).
All the treatments had concentrations that were within the optimum range according to Maldonado-Torres et al. (2007). In samplings 2 and 3, the concentrations of B in the leaves increased with foliar application of B. Nevertheless, differences in the concentrations of B in leaves between samples (i.e. samples 1, 2 and 3) within the same treatment group were not observed (Figure 2). These results may be due to the rapid transport of B to other parts of the plant; the B sprayed in the last two samples likely migrated towards the developing floral buds, as mentioned by Sotomayor et al. (2010), who found that the maximum concentrations of B in kiwi trees occurred 24 and 48h after treatment applications in the treated leaves, and an increase in B in flower buds was observed between 24 and 96h after treatment, demonstrating the rapid migration of boron from leaves to flowers. Moreover, our results confirm the hypothesis stated by Minchin et al. (2012), who established that the existence of a polyol (perseitol) in the phloem sap of avocado causes B to be phloem-mobile. This demonstrates that mature leaves act as sources of B for both vegetative and reproductive growth.
Foliar concentration of carbohydrates
The concentrations of carbohydrates in the leaves did not show any significant differences among the treatments in any of the three samplings, as seen in Table II.
TOTAL CARBOHYDRATE CONCENTRATIONS IN LEAVES OF AVOCADO CV. HASS WITH DIFFERENT CONCENTRATIONS OF FOLIAR B FERTILIZER
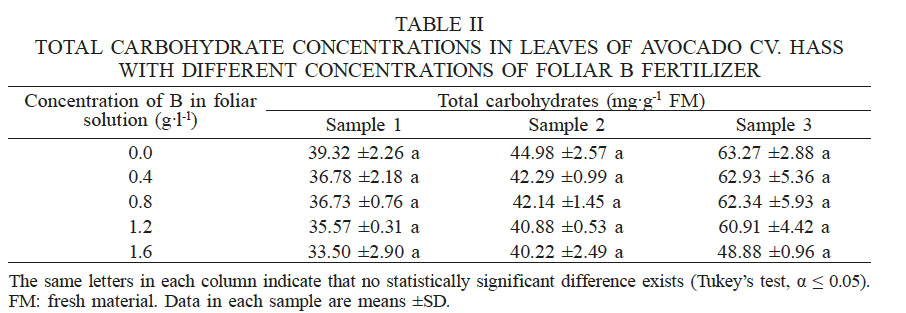
T The same letters in each column indicate that no statistically significant difference exists (Tukey’s test, α ≤ 0.05). FM: fresh material. Data in each sample are means ±SD.
Despite the lack of significant differences, the highest concentrations were recorded in the control treatment. In the third sampling, the carbohydrate concentration obtained in plants treated with 1.6g·l-1 B represented 77.3% of concentration determined for the control. These results may be a consequence of the transport of carbohydrates for flower bud formation, which is of great importance in avocado. Boldingh et al. (2016) showed that in the case of the cultivar Hass, carbohydrate and B contents of flowers at anthesis have a negative effect on the potential of flowers to set fruit, while successful fruit set represented higher levels of starch and all individual sugars in the styles.
Foliar concentration of amino acids
The analysis of variance of the amino acid concentrations obtained in the first and third samplings showed no statistically significant differences among the treatments (Tukey’s test, α ≤0.05). In sampling 2, differences between the 1.6g·l-1 B treatment and the control were observed (Table III).
TOTAL CONCENTRATIONS OF AMINO ACIDS IN LEAVES OF AVOCADO CV. HASS TREATED WITH FOLIAR FERTILIZER WITH DIFFERENT CONCENTRATIONS OF B
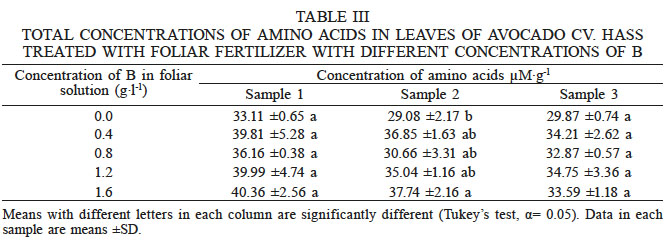
Means with different letters in each column are significantly different (Tukey’s test, α= 0.05). Data in each sample are means ±SD
The concentrations of amino acids decreased throughout each sampling, behaving in the same way as the B concentration, suggesting that the amino acids were transported to demand tissues (i.e. floral buds), similarly to carbohydrates.
The influence of B on the N assimilation process in higher plants has been reported in various species. In tobacco, B increases the assimilation of ammonium and consequently causes a progressive accumulation of amino acids and proteins (López-Lefebre et al., 2002). In contrast, Cakmak et al. (1995) showed a decrease in the amino acid content in sunflower leaves deficient in B. Likewise, toxic effects of B (0.5 and 2.0mM) on tomato plants significantly decreased leaf biomass, relative growth rate, organic N, soluble proteins, as well as nitrate reductase (NR) and nitrite reductase (NiR) activities (Cervilla et al., 2008).
Thickness of the cell wall
Cell wall thickness in the pith was significantly different (Tukey’s test, α ≤0.05) among treatments. Plants treated with 1.2 and 1.6g·l-1 had the thickest walls, 15.17 and 15.77µm thick, respectively; the increase compared with the control was 71% with 1.2g·l-1 B and 77% with 1.6g·l-1 B. Plants treated with 0.8g·l-1 B showed intermediate thickness (10.94µm), while in the treatment with 0.4g·l-1 B it was 8.99µm thick, not statistically different from the control treatment, which yielded an average thickness of 8.89µm (Figure 3).
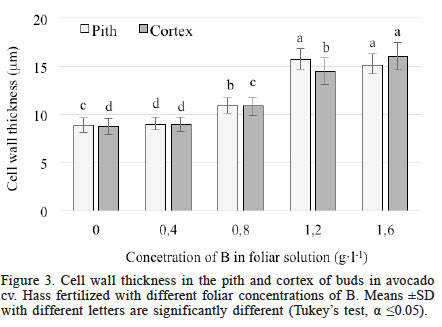
Figure 3
Cell wall thickness in the pith and cortex of buds in avocado cv. Hass fertilized with different foliar concentrations of B. Means ±SD with different letters are significantly different (Tukey’s test, α ≤0.05)
In the case of the cortex, the analysis of variance also showed significant differences (Tukey’s test, α ≤0.05). The treatment with the highest B concentration (1.6g·l-1) had the thickest cell wall (16.02µm), an increase of 84% compared with the control, followed by treatment with a foliar B concentration of 1.2g·l-1 with a thickness of 14.47µm, an increase of 66% compared with the control. The treatment with 0.8g·l-1 B was the third lowest, with 10.86µm. The lowest foliar concentration of B supplied in this study (0.4g·l-1) was not significantly different from the control, with values of 8.97 and 8.72µm, respectively (Figure 3).
The images in Figure 4 show the similarities in the cell wall thicknesses between the treatment with the lowest B concentration (0.4g·l-1 B) and the control, which means that a foliar supply of B at a concentration of 0.4g·l-1 has no effect on the cell wall thickness of the pith. Pink color changes can be clearly observed in cuts fertilized with 1.2 and 1.6g·l-1B, indicating the presence of phenols.
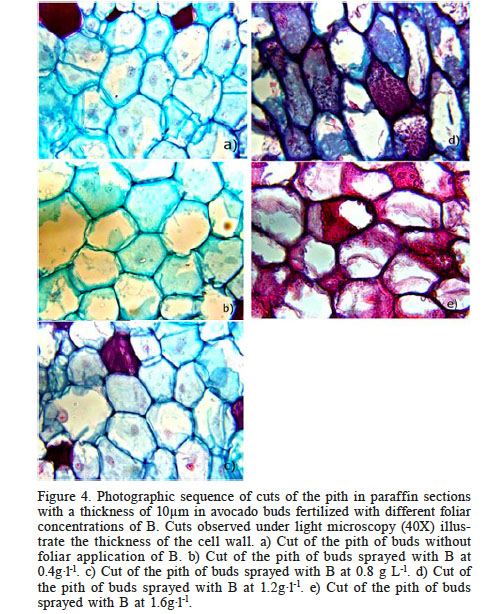
Figure 4.
Photographic sequence of cuts of the pith in paraffin sections with a thickness of 10µm in avocado buds fertilized with different foliar concentrations of B. Cuts observed under light microscopy (40X) illustrate the thickness of the cell wall. a) Cut of the pith of buds without foliar application of B. b) Cut of the pith of buds sprayed with B at 0.4g·l-1. c) Cut of the pith of buds sprayed with B at 0.8 g L-1. d) Cut of the pith of buds sprayed with B at 1.2g·l-1. e) Cut of the pith of buds sprayed with B at 1.6g·l-1.
The effect of B on the cell wall of the cortex is evident, and a positive relationship can be observed: as the foliar concentration of B supplied increased, the thickness of the cell wall of the cortex also increased (Figure 5).
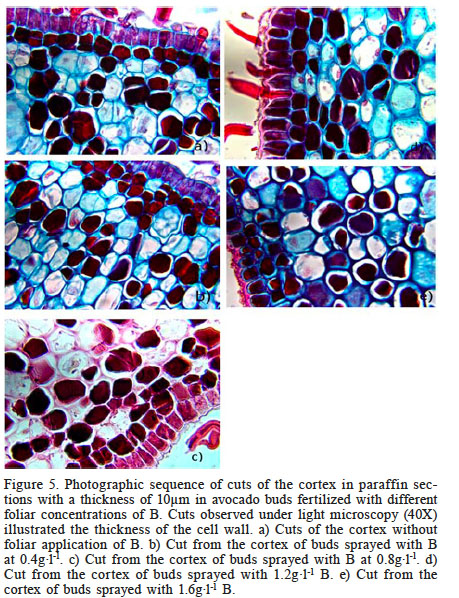
Figure 5.
Photographic sequence of cuts of the cortex in paraffin sections with a thickness of 10µm in avocado buds fertilized with different foliar concentrations of B. Cuts observed under light microscopy (40X) illustrated the thickness of the cell wall. a) Cuts of the cortex without foliar application of B. b) Cut from the cortex of buds sprayed with B at 0.4g·l-1. c) Cut from the cortex of buds sprayed with B at 0.8g·l-1. d) Cut from the cortex of buds sprayed with 1.2g·l-1 B. e) Cut from the cortex of buds sprayed with 1.6g·l-1 B.
The Pearson correlation coefficient was 0.6979, indicating a highly positive relationship between the thickness of the cell wall of the pith and the cortex at a significance level of less than 0.0001, i.e., increasing the thickness of the pith also increases the thickness of the cortex. It should be mentioned that some of the leaves began to develop minor burns starting from treatments with B at 1.2g·l-1.
Boron deprivation initially reduces the elongation of growing points due to restricted cell wall deposition and then, in more extreme cases, induces necrosis of these tissues due to cell death (Wang et al., 2015). Thus, the application of B in avocado trees in order to increase the thickness of the cell wall in the pith is beneficial, mainly in the branches that may provide greater strength to support the weight of future fruits.
A thicker cortex cell wall can help trees because it increases their resistance to pathogens. B increases cell wall thickness and is important in the synthesis of phenols and lignin. Acute and prolonged B deficiencies in ‘Hass’ avocado have been associated with the occurrence of bacterial canker, which commonly attacks trunks and branches of the plants (Salazar, 2002).
In this study it is demonstrated that foliar B application increases cell wall thickness in avocado. This finding is of pivotal importance for avocado plants, since the cell size and shape are determined by the mechanical control of cell expansion, governed by the plant cell wall. Hence, the cell wall ultimately determines tissue and organ morphology (Le Gall et al., 2015). It has been demonstrated that both B and Ca affect in vitro organogenesis of Eucalyptus grandis (Brondani et al., 2012). Here, B allowed the induction of well-developed buds. It has also been reported that under B deficiency there is a disorganization of coffee plantlets, causing thinner walls in xylem tissues. In B-deficient leaves there were fewer and deformed stomata (Rosolem and Leite, 2007). However, B deficiency can be related to a thicker cell wall, known as the swollen cell wall phenotype. The reason for the increase in such thickness is not the deposition of more carbohydrate polymers, but an increase of the pore size of the cell wall (Fleischer et al., 1999).
Conclusions
Foliar penetration of B occurred in avocado leaves, but due to the natural fluorescence of the leaf, it was not possible to distinguish the path followed by the fertilizer, nor the time needed for absorption. Such penetration may be passive, given that the H3BO3 molecule is neutral.
An increase in the concentration of B in the leaf spray solution increased the cell wall thickness in both the pith and cortex, which confirms the structural function of B in the cell wall.
There were no significant effects of foliar application of B on the concentration of carbohydrates in any of the three samplings. With respect to the amino acid concentrations, there were differences only between the control and the 1.6g·l-1 B concentration in the second sampling.
Based on the enhanced cell wall thickness observed, a concentration of B at 1.2g·l-1 is recommended for foliar application in avocado cv. Hass.
FOLIAR BORON FERTILIZATION IN ‘HASS’ AVOCADO INCREASES AMINO ACID CONCENTRATIONS AND CELL WALL THICKNESS
Alcántar GG, Sandoval VM (1999) Manual de Análisis Químico de Tejido Vegetal. Publicación Especial 10. Sociedad Mexicana de la Ciencia del Suelo. Chapingo, México. 156 pp.
Alcántar GG, Trejo-Téllez LI, Fernández PL, Rodríguez MMN (2016) Elementos esenciales. In Alcántar GG, Trejo-Téllez LI, Gómez-Merino FC (Eds.) Nutrición de Cultivos. 2nd ed. Colegio de Postgraduados, México, pp. 22-55.
Asad A, Blamey FPC, Edwards DG (2003) Effects of boron foliar applications on vegetative and reproductive growth of sunflower. Ann. Bot. 92: 565-570.
Boldingh HI, Alcaraz ML, Thorp TG, Minchih PEH, Gould N, Hormaza JI (2016) Carbohydrate and boron content of styles of ‘Hass’ avocado (Persea americana Mill.) flowers at anthesis can affect final fruit set. Sci. Hotic. 198: 125-131.
Brondani GE, De Araujo MA, De Alcântara BK, De Carvalho JG, Gonçalves AN, De Almedia M (2012) In vitro organogenesis of Eucalyptus grandis: effects of boron and calcium. Acta Sci. Agron. 34: 403-411.
Caffall KH, Mohnen D (2009) The structure, function, and biosynthesis of plant cell wall pectin polysaccharides. Carbohyd. Res. 344: 1879-1900.
Cakmak I, Kurz H, Marschner H (1995) Short-term effects of boron, germanium and high light intensity on membrane permeability in boron deficient leaves of sunflower. Physiol. Plant. 95: 11-18.
Cervilla LM, Blasco B, Ríos JJ, Rosales MA, Rubio-Wilhelmi MM, Sánchez-Rodríguez E, Romero L, Ruíz JM (2008) Response of nitrogen metabolism to boron toxicity in tomato plants. Plant Biol. 11: 671-677.
Chormova D, Fry SC (2016) Boron bridging of rhamnogalacturonan-II is promoted in vitro by cationic chaperones, including polyhistidine and wall glycoproteins. New Phytol. 219: 241-251.
Comerfor KB, Ayoob KT, Murray RD, Atkinson SA (2016a) The role of avocados in complementary and transitional feeding. Nutrients 8: 316.
Comerfor KB, Ayoob KT, Murray RD, Atkinson SA (2016b) The role of avocados in maternal diets during the periconceptional period, pregnancy, and lactation. Nutrients 8: 313.
FAOSTAT (2019) Statistic Crops. Avocado. Food and Agriculture Organization of the United Nations. http://www.fao.org/faostat/en/#data/QC (Cons. 07/07/2019).
Fleischer A, O'Neill MA, Ehwald R (1999) The pore size of non-graminaceous plant cell walls is rapidly decreased by borate ester cross-linking of the pectic polysaccharide rhamnogalacturonan I. Plant Physiol. 121: 829-838.
Garner LC, Lovatt CJ (2008) The relationship between flower and fruit abscission and alternate bearing of Hass avocado. J. Am. Soc. Hortic. Sci. 133: 3-10.
Geiger M, Walch-Liu P, Engels C, Harnecker J, Schulze E, Ludewig F, Sonnewald U, Scheible W, Stitt M (1998) Enhanced carbon dioxide leads to a modified diurnal rhythm of nitrate reductase activity in older plants, and a large stimulation of nitrate reductase activity and higher levels of amino acids in young tobacco plants. Plant Cell Environ. 21: 253-268.
Könne JS (1988) Methods of increasing avocado fruit production. South African Avocado Growers’ Association Yearbook 11: 53-55.
Le Gall H, Philippe F, Domon J-F, Gillet F, Pelloux J, Rayon C (2015) Cell wall metabolism in response to abiotic stress. Plants 4: 112-166.
López CMdL, Márquez G, Murguía GS (2005) Técnicas para el Estudio del Desarrollo de las Angiospermas. Libro de Laboratorio. 2nd ed. UNAM. México. 178 pp.
López-Lefebre LR, Ruiz JM, Rivero RM, García PC, Sánchez E, Romero L (2002) Supplemental boron stimulates ammonium assimilation in leaves of tobacco plants (Nicotiana tabacum L.). Plant Growth Regul. 36: 231-236.
Lovatt CL (1999) Timing citrus and avocado foliar nutrient applications to increase fruit set and size. HortTechnology 9: 607-612.
Maldonado-Torres R, Álvarez-Sánchez ME, Almaguer-Vargas G, Barrientos-Priego EF, García-Mateos R (2007) Estándares nutrimentales para aguacatero ‘Hass’. Rev. Chapingo Ser. Hortic. 13: 103-108.
Minchin PE, Thorp TG, Bolding HL, Gould N, Cooney JM, Negm FB, Focht E, Arpaia ML, Hu H, Brown P (2012) A possible mechanism for phloem transport of boron in “Hass” avocado (Persea americana Mill.) trees. J. Hortic. Sci. Biotech. 87: 23-28.
Moore S, Stein WH (1954) Procedures for the chromatographic determination of amino acids on four per cent cross-linked sulfonated polystyrene resins. J. Biol. Chem. 211: 893-906.
Nava-Sánchez R, Almaguer-Vargas G, Pérez-Grajales M, Maldonado-Torres R, Cárdenas-Soriano E (2004) Fertilización foliar en cebolla. Rev. Chapingo Ser. Hortic. 10: 159-163.
Reguera M, Wimmer M, Bustos P, Goldbach HE, Bolaños L, Bonilla I (2010) Ligands of boron in Pisum sativum nodules are involved in regulation of oxygen concentration and rhizobial infection. Plant Cell Environ. 33: 1039-1048.
Reid RJ, Hayes JE, Post A, Stangoulis JCR, Graham RD (2004) A critical analysis of the causes of boron toxicity in plants. Plant Cell Environ. 27: 1405-1414.
Rosolem CA, Leite VM (2007) Coffee leaf and stem anatomy under boron deficiency. Rev. Bras. Ciênc. Solo 31: 477-483.
Salazar GS (2002) Nutrición del Aguacate. Principios y Aplicaciones. INPOFOS, INIFAP. Querétaro, México. 165 pp.
Sandoval ZE (2005) Técnicas Aplicadas al Estudio de la Anatomía Vegetal. Colección Cuadernos del Instituto de Biología. UNAM. México. 278 pp.
Sotomayor C, Norambuena P, Ruiz R (2010) Boron dynamics related to fruit growth and seed production in kiwifruit (Actinidia deliciosa cv. Hayward). Cienc. Inv. Agrar. 37: 133-141.
Southgate DA (1976) Determination of Food Carbohydrates. Applied Science. London, UK. 105 pp.
Steiner AA (1984) The universal nutrient solution. Proc. 6th Int. Cong. On Soilless Culture, Lunteren 1984. International Society for Soilless Culture. Wageningen, The Netherlands. pp. 633-649.
Wang N, Yang C, Pan Z, Liu Y, Peng S (2015) Boron deficiency in woody plants: various responses and tolerance mechanisms. Front. Plant Sci. 6: Art. 916.
