Original articles
Association of CCR5 G59029A and RANTES -28 C/G polymorphisms in patients with chronic periodontitis and/or Type 2 Diabetes Mellitus, in a Southeastern Mexican population
Asociación de los polimorfismos CCR5 G59029A y RANTES -28 C/G en pacientes con periodontitis crónica y/o diabetes mellitus tipo 2 en una población del sureste mexicano
Association of CCR5 G59029A and RANTES -28 C/G polymorphisms in patients with chronic periodontitis and/or Type 2 Diabetes Mellitus, in a Southeastern Mexican population
Investigación Clínica, vol. 59, no. 2, pp. 135-145, 2018
Universidad del Zulia
Received: 10 January 2017
Accepted: 05 April 2018
Abstract: The aim of this study was to determine the association of poly morphisms CCR5G59029A and RANTES -28C/G in chronic periodontitis (CP) and type 2 diabetes mellitus (T2D). A case control study was carried out that included 163 patients with CP and/or T2D. A periodontal examination was done on all potential participants in order to diagnose CP. To confirm the diag nosis of T2D, peripheral blood samples were collected for laboratory analyses (blood glucose and glycosylated hemoglobin). Genotyping was performed by PCRRFLP. The Genotype AA of CCR5 G59029A increased the risk for CP 7.36 times (95% CI = 1.14-78.27, p = 0.03). This genotype’s A allele also increased T2D risk 2.23 times (95% CI = 1.10-4.54, p = 0.02). Genotype CG of RANTES -28 C/G increased CP risk 5.99 times (95% CI = 1.93-18.57, p = 0.002). Its G allele also raised CP risk 4.55 times (95% CI = 1.58-13.04, p = 0.002). In patients suffering both CP and T2D, the CG genotype was associated with the diseases, increasing their risk 4.68fold (95% CI = 1.60-14.03, p = 0.004). Its G allele raised CP and T2D risk 3.8 times (95% CI = 1.35-10.67, p = 0.008). The AA genotype and allele A of G59029A polymorphism of CCR5 gene is a risk factor for CP. The CG genotype and allele G polymorphism RANTES -28 C/G gene are risk factors for CP in the population studied. The CG genotype and G allele polymorphism RANTES -28 C/G showed a strong association and a subse quent risk factor for the coexistence of CP and T2D.
Keywords: polymorphism, periodontitis, RANTES, diabetes.
Resumen: El objetivo de este estudio fue determinar la asociación de los polimorfismos CCR5 G59029A y RANTES -28 C/G con la periodontitis crónica (PC) y la diabetes mellitus tipo 2 (DT2). Se realizó un estudio de casos y contro les en el que se incluyeron a 163 pacientes con PC y/o DT2. Se realizó un exa men periodontal a cada participante para confirmar el diagnóstico de PC. Para la confirmación de la DT2 fueron realizados análisis clínicos en muestras san guíneas (hemoglobina glucosilada y glucosa en sangre). La determinación de los genotipos se realizó por PCRRFLP. El genotipo AA del polimorfismo CCR5 G59029A fue un claro factor de riesgo para PC incrementando este en 7,36 veces (IC del 95% = 1,1478,27, p=0,03). El alelo A de este genotipo, también incrementó el riesgo de DT2 en 2,23 veces (IC del 95% = 1,10-4,54, p = 0,02). El genotipo CG del polimorfismo RANTES -28 C/G incrementó el riesgo de PC hasta 5,99 veces (IC del 95% = 1,93-18,57, p = 0,002). Su alelo G también aumentó el riesgo de PC 4,55 veces (IC del 95% = 1,5813,04, p = 0,002). El genotipo CG incrementó el riesgo de coexistencia de ambas enfermedades en 4,68 veces (IC del 95% CI = 1,60-14,03, p = 0,004). El alelo G aumentó el riesgo de PC y DT2 3,8 veces (IC del 95% = 1,35-10,67, p = 0,008). De acuerdo con los resultados de la presente investigación, el genotipo AA y el alelo A del polimorfismo G59029A del gen CCR5 se pueden considerar como factores de riesgo para PC. El genotipo CG y el alelo G del polimorfismo RANTES -28 C/G también son un factor de riesgo para PC en la población estudiada. El genotipo CG y el alelo G del polimorfismo -28 C/G del gen RANTES mostraron una fuer te asociación con PC y DT2 juntas, por lo que constituyen un factor de riesgo para la coexistencia de estas enfermedades.
Palabras clave: polimorfismo, periodontitis, RANTES, diabetes.
INTRODUCTION
Type 2 diabetes mellitus (T2D) is a wellknown risk factor for the severity and progression of chronic periodontitis (CP), a condition in which infections and/or injury lead to destruction of the soft tissues and bony support forming the tooth insertion (1). Diabetic patients with poor periodontal health are 2.8 times more likely to develop a destructive CP, and 4.2 times more likely to exhibit progressive loss of alveolar bone. Full understanding of the relationship between CP and T2D is vital, since control of blood glucose levels in conjunction with proper periodontal treatment can help control CP and reduce alveolar bone loss (2).
Chemokines are chemoattractants structurally related to cytokines, that activate specific leukocyte subsets (35) that in turn play an important role in focal immunity diseases such as CP (6). These molecules recruit and activate leukocytes in inflamed tissues, therefore playing a fundamental role in mediation and regulation of immune and inflammatory reactions (7, 8). The CCR5 chemokine receptor is expressed on the surface of white blood cells, macro phages/monocytes and activated T lymphocytes. Among its ligands is RANTES, which can be produced by fibroblasts, endothelial cells, monocytes/macrophages, osteoblasts and mastocytes (9). The RANTES chemokine is known to be present in the crevicular flu id of CP patients and absent in people with healthy gums; this suggests a relationship between this chemokine and periodontal disease.
The CCR5 receptor and its ligands are expressed in periodontal disease where they may contribute to migration of inflammatory cells, allowing more complete development of the disease and leading to greater bone loss by reabsorption (10). The G59029A polymorphism conditions up to a twofold increase in CRR5 expression (4), as well as an increase in CCR5 expression in mono nuclear cells in peripheral blood (11). The RANTES gene belongs to the CC chemokine family and contains the -28 C/G polymorphism. Located in the promoter region, it directly affects -28 C/G transcription, indicating that this polymorphism can increase expression of the RANTES cytokine. Host genetics are thought to modulate the se verity of disease caused by infection. Genes encoding chemokine ligands and receptors, have been found to play important role in inflammatory response, which most frequently involve the recruitment of leukocytes in or der to eliminate infectious agents (12). The objective of the present study was to identify any associations that CCR5 G59029A and RANTES -28C/G polymorphisms may have with CP and T2D.
MATERIAL AND METHODS
A case control study was carried out us ing the following selection criteria. Inclusion criteria: Patients 30 to 65 years of age, of either sex, unrelated, with a CP and/or T2D diagnosis, and patients 30 to 65 years of age, of either sex, unrelated, with no previous CP and/or T2D diagnosis and no family history of T2D. Exclusion criteria: Patients with known systemic diseases such as epilepsy, anemia, liver and/or thyroid diseases, those receiving chemotherapy or radiotherapy, active gum diseases, those that had received an antibiotics cycle in the three months prior to the study, those that had received periodontal treatment in the twelve months prior to the study, and pregnant or nursing women. All patients agreed to participate in the study via written consent. A periodontal examination was done on all potential par ticipants in order to diagnose CP, and inter views were conducted to determine if they met the inclusion criteria.
A dental history and complete physical examination were done of each patient, and a William’s periodontal probe was used to measure periodontal pocket depth and insertion loss. All teeth were probed following the clinical criteria proposed in the 1999 International Workshop for Periodontal Disease and Condition Classification and the consensus reported in the 5th European Workshop on Periodontology (13), and the findings were recorded on a periodontagram designed for this purpose. Anthropometric measurements (weight, height and BMI) were determined for all participants. To con firm T2D diagnosis, peripheral blood samples were collected for laboratory analyses (blood glucose and glycosylated hemoglo bin) following established guidelines (NOM 015SSA1994) (14) for diabetes mellitus prevention, treatment and control. Once the patients in each group were classified, sam ples were coded and stored at -70°C.
Polymorphism analysis was done using PCR and RFLP. Genomic DNA extraction was done using 1.5 mL blood with EDTA ac cording to Higuchi (15). Using established primers (Table I), genomic DNA was extract ed from blood for polymorphism analysis of CCR5 G59029A. The DNA was amplified with a 25 μL final PCR volume. Amplified fragment size was 268 bp, which has a cut site for the enzyme BSP1286I. Electrophoretic analysis of the digestion products was per formed on 12% polyacrylamide gel stained with silver nitrate. Genotype identification was done based on digestion product size. A similar process was used to analyze the RAN TES -28 C/G polymorphism (Table I). The fragment size was 173 bp, with two cut sites for MnlI for the C allele; one site was lost for the G allele. The amplified fragments were digested with the enzyme and the digestion products analyzed with electrophoresis, us ing 18% polyacrylamide gels stained with silver nitrate. Genotype identification was done based on digestion product size. Simple counts were used to quantify genotype and allele frequencies for the CCR5 G59029A and RANTES -28 C/G polymorphisms. This was done for both cases and controls, and the frequency distributions analyzed by applying population parameters for HardyWeinberg equilibrium. The association between the polymorphisms and CP and/or T2D risk was calculated using Chisquared and Fisher’s tests, considering a p < 0.05, and the odds ratio; confidence interval was 95% (OR, 95% CI). Each case group was compared to the control group.
RESULTS
A total of 163 patients were recruited from three health institutions in the state of Yucatan, Mexico. With regard to sex, we observed a higher frequency of women (63.2%) than men (36.8%). Based on existing condition, the patients were divided into four groups (three cases, one control) (Table II). Measurement of glycosylated hemoglobin (HbA1c < 7%) in T2D patients showed that 53% (n = 44) were apparently under control. This analysis included T2D and CP/T2D pa tients. No statistically significant differences were observed with respect to the BMI (Table II).
The most frequent genotypes for the CCR5 G59029A polymorphism for the CP group were GA (47.4%) and AA (47.4%). For the controls, the most frequent was GA (52.4%). The A allele was the most frequent (CP = 71.1%; controls = 52.4%) in both groups. The most frequent genotype of the RANTES -28 C/G polymorphism was CC (CP = 55.3%; controls = 88.1%), and the most frequent allele was C (CP = 77.6%; controls = 94.0%). Frequency distributions in both polymorphisms fitted the Hardy Weinberg equilibrium expectations of the control population. When comparing genotype AA of CCR5 G59029A between the cases and controls, a significant association (p = 0.03) with CP was identified, as well as a 7.36fold greater risk of CP (95% CI = 1.14-78.27). A significant difference (p = 0.02) in al lele frequencies between CP patients and healthy controls was found in a comparison of A allele frequencies, along with a 2.23 fold greater risk of CP (OR = 2.23; 95% CI = 1.10-4.54) (Table III).
| Primers 5’3’ | Annealing temperature (ºC) | Size of PCR product (bp) | |
| CCR5 G59029A | F: CAG TCA ACC TGG GCA AAG CC | 50 | 268 |
| CCR5 G59029A | R: AGC TTT GGT CCT GAG AGT CC | 50 | 268 |
| RANTES -28 C/G | F: ACA GAG ACT CGA ATT TCC GGA | 50 | 173 |
| RANTES -28 C/G | R: CCA CGT GCT GTC TTG ATC CTC | 50 | 173 |
| BMI Mean ± SD (Range) | Blood glucose Mean ± SD (Range) | Glycosylated hemoglobin Mean ± SD (Range) | ||
| Cases | CP n=38 | 29.06±0.78 (19.0139.78) | 97.92±2.83 (72.77188.40) | 5.32±0.19 (2.807.55) |
| Cases | T2D n=34 | 31.78±0.98 (22.6044.57) | 166.96±12.13 (70337.96) | 7.24±0.49 (2.3013.80) |
| Cases | CP/T2D n=49 | 30.30±0.66 (22.4441.14) | 170.45±9.57 (82.94362.64) | 18.17±0.39 (4.0415.42) |
| Controls | n=42 | 28.67±0.69 (19.4747.04) | 92.14±2.27 (60.14165) | 5.48±0.16 (2.106.63) |
Genotype frequencies of RANTES -28 C/G in CP patients, exhibited an association (p = 0.002) between CG and CP and a 5.99 times greater risk of CP (95% CI = 1.93- 18.57). In allele frequencies analysis, G allele distribution was found to be associated (p = 0.002) with CP, and the risk of CP was 4.55 times greater (OR = 4.55; 95% CI = 1.58-13.04) (Table III).
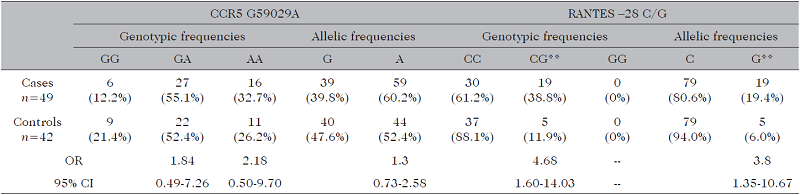
When comparing CCR5 G59029A frequencies between the T2D and control groups, GA was the most frequent genotype (T2D = 53.0%; control = 52.4%), and the A allele the most frequent (T2D = 64.7%; control = 52.4%). For RANTES -28 C/G, the CC genotype was most frequent (T2D = 82.4%; control = 88.1%), and the C allele was the most frequent (T2D = 91.2%; control = 94.0%) (Table IV). In the comparison between the CP/T2D group and the control for CCR5 G59029A, the GA genotype was the most frequent (CP/T2D = 55.1%; control = 52.4%), and the A allele the most frequent (CP/T2D = 60.2%; control = 52.4%). For RANTES -28 C/G, the most frequent geno type was CC (CP/T2D = 61.2%; control = 88.1%), and the most frequent allele was C (CP/T2D = 80.6%; control = 94.0%) (Table V). An association (p = 0.004) was identified in this group between the CG genotype of RANTES -28 C/G and CP/T2D, with a 4.68 times greater risk (95% CI = 1.60-14.03). There was also an association (p = 0.008) between the G allele and CP/T2D, with a 3.8 times greater risk (95% CI = 1.35-10.67).
DISCUSSION
Previous studies suggest, that the chemokinereceptor complex operates in periodontal tissue; this complex consists of the chemokines CCL3, CCL4 and CCL5 (RANTES) and their receptors, CCR1, CCR3 and CCR5. The CCR5 receptor has been observed to mediate chemoattraction of pro leukocytes and osteoclasts, reinforcing the role of cytokines in CP pathogenesis (16). RANTES expression may cause an increase in the CCR5 receptor; this could lead to greater destruction of periodontal tissues, since the CCR5 carrier cell population includes osteoclast precursors from a monocyte lineage. This has a relationship with an increased bone reabsorption activity during CP (16). In mononuclear blood cells such as lymphocytes and monocytes, the AA genotype of the CCR5 G59029A polymorphism, has been reported to augment CCR5 ex pression as well as alterations to the ligand binding properties (17, 18). An increase in this RANTES receptor, could lead to great er destruction of periodontal tissues in the presence of CP, because the CCR5 receptor is linked to the production of osteoclastic cells, which act directly upon the bone tis sue supporting the tooth (19). The RANTES-28 C/G polymorphism is located in the re gion promoting its transcription, leading to an increase in the RANTES chemokine in the organism (20,21). This increase may directly affect the development of CP, since RANTES has been reported in CP patients (6), and regulates the equilibrium between pro and antiinflammatory Th1 response cells. Oral cancer studies have reported that patients with the G allele are 2.84 times more likely to have oral squamous cell carcinoma (22).
| CCR5 G59029A | CCR5 G59029A | CCR5 G59029A | CCR5 G59029A | CCR5 G59029A | RANTES -28 C/G | RANTES -28 C/G | RANTES -28 C/G | RANTES -28 C/G | RANTES -28 C/G | |
| Genotypic frequencies | Genotypic frequencies | Genotypic frequencies | Allelic frequencies | Allelic frequencies | Genotypic frequencies | Genotypic frequencies | Genotypic frequencies | Allelic frequencies | Allelic frequencies | |
| GG | GA | AA* | G | A* | CG | CG** | GG | C | G** | |
| Cases | 2 | 18 | 18 | 22 | 54 | 21 | 17 | 0 | 59 | 17 |
| n=38 | (5.2%) | (47.4%) | (47.4%) | (28.9%) | (71.1%) | (55.3%) | (44.7%) | (0%) | (77.6%) | (22.4%) |
| Controls | 9 | 22 | 11 | 40 | 44 | 37 | 5 | 0 | 79 | 5 |
| n=42 | (21.4%) | (52.4%) | (26.2%) | (47.6%) | (52.4%) | (88.1%) | (11.9%) | (0%) | (94.0%) | (6.0%) |
| OR | OR | 3.68 | 7.36 | 2.23 | 2.23 | 2.23 | 5.99 | 4.55 | ||
| 95% CI | 95% CI | 0.6238.39 | 1.1478.27 | 1.104.54 | 1.104.54 | 1.104.54 | 1.9318.57 | 1.5813.04 |
| CCR5 G59029A | CCR5 G59029A | CCR5 G59029A | CCR5 G59029A | CCR5 G59029A | RANTES - 28 C/G | RANTES - 28 C/G | RANTES - 28 C/G | RANTES - 28 C/G | RANTES - 28 C/G | |
| Genotypic frequencies | Genotypic frequencies | Genotypic frequencies | Allelic frequencies | Allelic frequencies | Genotypic frequencies | Genotypic frequencies | Genotypic frequencies | Allelic frequencies | Allelic frequencies | |
| GG | GA | AA | G | A | CC | CG | GG | C | G | |
| Cases | 3 | 18 | 13 | 24 | 44 | 28 | 6 | 0 | 62 | 6 |
| n=34 | (8.8%) | (53.0%) | (38.2%) | (35.3%) | (64.7%) | (82.4%) | (17.6%) | (0%) | (91.2%) | (8.8%) |
| Controls | 9 | 22 | 11 | 40 | 44 | 37 | 5 | 0 | 79 | 5 |
| n=42 | (21.4) | (52.4%) | (26.2%) | (47.6%) | (52.4%) | (88.1%) | (11.9%) | (0%) | (94.0%) | (6.0%) |
| OR | 2.45 | 3.52 | 1.66 | 1.58 | 1.52 | |||||
| 95% CI | 0.5015.92 | 0.6324.61 | 0.823.39 | 0.435.72 | 0.445.24 |
GG: wild type genotype. GA: heterozygote genotype. AA: mutated homozygote genotype. CC: wild type genotype. CG: heterozygote genotype. GG: mutated homozygote genotype. OR: odds ratio, *p<0.05, **p<0.01.
Other studies have investigated the dis tributions of RANTES and CCR5 genotypes in HIV1 infected patients and had different conclusions. Gong et al. (23) suggested that the RANTES -28G allele might play a role in resistance to HIV1 infection among Asians and Gupta et al. (24) did not observe an as sociation between G59029A of the CCR5 gene and HIV infection in the Indian population. On the other hand, Fang et al. (25) suggested an association between the -28G allele and asthma risk in Asian children, but not in Caucasian children.
A number of studies in different populations have identified an association be tween T2D and CCR5 G59029A. In one project in India, independent studies were done on populations in the country’s north and south (26). The northern population ex hibited a strong association (p = 0.0001) between the polymorphism and T2D, with a risk of T2D that was 2.22 times (95% CI 1.71-2.87) greater than in controls. An as sociation (p < 0.001) was also found in the southern population, with a 2.17fold greater (95% CI 1.43-3.29) risk of T2D. This differs from the present results in which no association (p > 0.05) between CCR5 G59029A and risk of T2D was observed. In a study of T2D patients with chronic renal insufficiency (CRI) in the presence of CCR5 G59029A, an association (p = 0.028) was found between CRI and presence of the AG genotype in T2D patients. No association was identified in the present study, between the AG genotype of CCR5 G59029A and T2D; like Ting et al. (27) who observed no link between CCR5 G59029A and coronary artery disease. It is possible that one conclu sion drawn in one population or racial group may be totally divergent from another. Since racial differences are common in polymor phic systems (28), various alleles or genotypes or genes interacting with one another, may exert their influences on the manifestations of the clinical phenotype (29) in different populations. In the present study, a strong association was found between this polymorphism and CP in that it led to a 4.9 times greater risk of suffering more severe CP. This is the first study of this association and therefore establishes a base for future research into the role of CCR5 G59029A in CP pathogenesis. No known research has been done on the relationship between the RANTES -28 C/G polymorphism and CP, although reports exist on the association between RANTES and CP; this chemokine was found in CP patients and was eliminated after root cleaning and polishing (6). Stud ies in other diseases support the participation of these polymorphisms in different pathologies such as those reported by Weng et al. (30) who studied the impact of these polymorphisms on the risk and clinical pathological characteristics of oral cancer, reporting a 2.8 times greater risk (95% CI 1.43-5.4) of oral cancer in the presence of the genotype CG of polymorphism -28 C/Gof the RANTES gene compared to patients without the polymorphism. They found no association between disease and polymorphism G59029A of the CCR5 gene. In the present study, a significant association was identified between genotype CG and allele G of polymorphism RANTES -28 with a 5.99 (95% CI 1.93-18.57) and 4.55 (95% CI 1.58-13.04) times greater risk respectively for CP but not in CCR5 G59029A. There are no reports associating both polymorphisms with CP, but Qidwai (22), who observed an increased risk of oral cancer due to the presence of the genotype CG of polymorphism -28 in combination with polymorphism -403 of the gene RANTES. There are other reports where the presence of these poly morphisms is not associated with various pathologies such as the reports of Tahara et al. (31) found no association between this polymorphism and ulcerative colitis in the Chinese population. Also, AlQhatani et al.
(32) and Lian et al. (33) found no association between polymorphism -28 C/G of the RANTES gene in hepatitis B infection and systemic lupus erythematosus, respectively. In the same way, an association between polymorphism G59029A of the CCR5 gene and CP was observed. Zhao et al. (34) reported an increased risk of diabetic nephropathy in the presence of G59020A and Oliveira et al. (35), also reported that the AA genotype of polymorphism 59029 from the CCR5 gene was related to the develop ment of chronic heart problems in Chagas disease. These results are similar to those observed in our study, where a significant association was found between the AA geno type with a risk of 7.36 (95% CI 1.14-78.27) and a more destructive CP.
In terms of T2D, one study did report an association between the GG genotype of RANTES -28 C/G and diabetic nephropathy in T2D patients, with a 2.2fold increase in risk (36). No association was identified be tween this polymorphism and T2D in the present results. Ting et al. (27) did not observe an association between this polymer phism and the coronary artery disease. No literature was found associating polymorphism with CP; however, Shih et al. (6) studied del ta 32 polymorphism of the CCR5 gene in CP and aggressive periodontitis; they found no association between them, contrary to the result observed in this study, where we found an association between genotype AA and risk allele A (p<0.05) with a risk of 7.36 (95% CI 1.14-78.27) and 2.23 (95% CI 1.10-4.54)
respectively. When analyzing the genotypic and allelic frequencies of polymorphism -28 C/G of the RANTES gene and G59029A of the CCR5 gene, no association with T2D was found. Borkar et al. (11) reported as sociation between this polymorphism and endstage renal disease, this is attributed to genotypes AG y GG, which is contrary to the report of the present work. Mokubo et al. (20) conducted a study in the Japanese population associated with polymorphism -28 C/G of the RANTES gene with diabetic nephropathy, finding no association between this disease and said polymorphism, a result similar to the one reported in our study. Periodontal disease and T2D is related, with CP classified as the sixth complication of T2D. No association has been reported to date of these two diseases together with a poly morphism. This is the first time that such association has been observed. In the comparison of patients with the RANTES -28 C/G polymorphism to those in the control group, a clear association (p < 0.05) was identified, with a 3.8 (95% IC 1.35-10.67) times greater risk of suffering the CP/T2D combination. Bagci et al. (37) studied the association of CCR5 G59029A in patients with chronic renal failure (CRF) and hyper tension, T2D and atherosclerosis (AS) find ing association between CRF and AS with a risk of 2.58 (95% CI 1.21-5.51) and T2D with a risk of 1.69 (95% CI 1.21-5.51) (39); this result diverges from our study where no association was found between CP/T2D and this polymorphism. Mlynarsky et al. (38), studied the risk of diabetic nephropathy in male patients with T1D, and found no association with the presence of polymorphism G59029A of the CCR5 gene. This result is similar to observed in our study. In conclusion, the CCR5 G59029A polymorphism of the CCR5 gene, is a risk factor for developing CP in patients carrying the mutated homozygote or heterozygote genotype. The RANTES -28 C/G polymorphism is a risk factor for CP and the CP/T2D combination in patients carrying the mutated homozygote or heterozygote genotype. The relationship between CP and T2D, and the resulting periodontal destruction, may involve more than an interaction between immunological reactions. It may be a genetic component that predisposes patients to more extreme responses to etiological factors of CP, and thus place them at higher risk of developing more acute forms of this disease.
REFERENCES
1. Southerland JH, Taylor GW, Offenbacher S. Diabetes and periodontal infection: Mak ing the connection. Clin Diabetes 2005; 23(4): 171178.
2. Mealey BL, Ocampo GL. Diabetes mellitus and periodontal disease. Periodontol 2000 2007; 44(1): 127153.
3. Kaur G, Holtfreter B, Rathmann W, Schwahn C, Wallaschofski H, Schipf S, Nauck M, Kocher T. Association between type 1 and type 2 diabetes with periodontal disease and tooth loss. J Clin Periodontol 2009; 36(9): 765774.
4. Gamonal J, Acevedo A, Bascones A, Jorge O, Silva A. Characterization of cellular in filtrate, detection of chemokine receptor CCR5 and interleukin8 and RANTES che mokines in adult periodontitis. J Periodon tal Res 2001; 36(3): 194203.
5. Yu HH, Liu PH, Yang YH, Lee JH, Wang LC, Chen WJ, Chiang BL. Chemokine MCP1/ CCL2 and RANTES/CCL5 gene polymor phisms influence HenochSchönlein purpu ra susceptibility and severity. J Formos Med Assoc 2015; 114 (4): 347352.
6. Shih YS, Fu E, Fu MM, Lin FG, Chiu HC, Shen EC, Chiang CY. Association of CCL5 and CCR5 gene polymorphisms with peri odontitis in Taiwanese. J Periodontol 2014; 85(11): 15961602.
7. Sahingur SE, Yeudall WA. Chemokine function in periodontal disease and oral cavity cancer. Front Immunol 2015; 6: 214.
8. SahinCalapoglu N, Demirci S, Calapo glu M, Yasar B. A casecontrol association study of RANTES (-28C>G) polymorphism as a risk factor for Parkinson's disease in Isparta, Turkey. Parkinson´s Dis 2016; 2016: 5042604.
9. Silva TA, Garlet GP, Fukada SY, Silva JS, Cunha FQ. Chemokines in oral inflamma tory diseases: apical periodontitis and peri odontal disease. J Dent Res 2007; 86(4): 306319.
10. Venza I, Visalli M, Cucinotta M, De Gra zia G, Teti D, Venza M. Proinflammatory gene expression at chronic periodontitis and periimplantitis sites in patients with or without type 2 diabetes. J Periodontol 2010; 81(1): 99108.
11. Borkar M, Tripathi G, Sharma RK, Sankh war SN, Agrawal S. Chemokine (CCR) and fractalkine (CX3CR) receptors and end stage renal disease. Inflamm Res 2011; 60(4): 399407.
12. Qidwai T, Khan MY. Impact of genetic variations in CC chemokine receptors and ligands on infectious diseases. Hum Immu nol 2016; 77(10): 961971.
13. Armitage GC. Development of a classifica tion system for periodontal diseases and conditions. Ann Periodontol 1999; 4(1): 16.
14. Norma Oficial Mexicana NOM015 SSA1994, "Para la prevención, tratamien to y control de la diabetes mellitus en la atención primaria," Diario Oficial de la Fe deración.
15. Higuchi R. Simple and rapid preparation of samples for PCR. En: Erlich HA, Ed. PCR Technology. New: Stockton Press 1989; 318.
16. Ferreira SB Jr, Repeke CE, Raimundo FM, Nunes IS, AvilaCampos MJ, Ferreira BR, Santana da Silva J, Campanelli AP, Garlet GP. CCR5 mediates proosteoclastic and os teoclastogenic leukocyte chemoattraction. J Dent Res 2011; 90(5): 632637.
17. Henckaerts L, Fevery J, Van Steenbergen W, Verslype C, Nevens F, Yap P, Roskams T, Rutgeerts P, Vermeire S. The RANTES -28 G polymorphism is associated with primary sclerosing cholangitis. Gut 2007; 56(6): 891892.
18. Carpenter D, Taype C, Goulding J, Levin M, Eley B, Anderson S, Shaw MA, Armour JA. CCL3L1 copy number, CCR5 genotype and susceptibility to tuberculosis. BMC Med Genet 2014; 15: 5.
19. Savarrio L, Donati M, Carr C, Kinane DF, Berglundh T. Interleukin24, RANTES and CCR5 gene polymorphisms are not asso ciated with chronic adult periodontitis. J Periodontal Res 2007; 42(2): 152158.
20. Mokubo A, Tanaka Y, Nakajima K, Wata da H, Hirose T, Kawasumi M, Sakai K, Kanazawa A, Maeda S, Hosokawa K, At sumi Y, Matsuoka K, Kawamori R. Che motactic cytokine receptor 5 (CCR5) gene promoter polymorphism (59029A/G) is as sociated with diabetic nephropathy in Jap anese patients with type 2 diabetes: a 10 year longitudinal study. Diabetes Res Clin Pract 2006; 73(1): 8994.
21. Prasad P, Tiwari AK, Kumar KM, Ammini AC, Gupta A, Gupta R, Thelma BK. Asso ciation of TGFbeta1, TNFalpha, CCR2 and CCR5 gene polymorphisms in type2 dia betes and renal insufficiency among Asian Indians. BMC Med Genet 2007; 8: 20.
22. Qidwai T. Chemokine genetic polymor phism in human health and disease. Immu nol Lett 2016; 176: 128138.
23. Gong Z, Tang J, Xiang T, Zhang L, Liao Q, Liu W, Wang Y. Association between regulated upon activation, normal T cells expressed and secreted (RANTES) -28C/G polymorphism and susceptibility to HIV1 infection: a metaanalysis. PLoS One 2013; 8(4): e60683.
24. Gupta A, Padh, H. Analysis of CCR5 and SDF1 genetic variants and HIV infection in Indian population. Int J Immunogenet 2015; 42(4): 270278.
25. Fang Q, Wang F, Zhao D. Association be tween regulated upon activation, normal T cells expressed and secreted (RANTES) -28C/G polymorphism and asthma risk -a metaanalysis. Int J Med Sci 2010; 7(1): 55-61.
26. Ahluwalia TS, Khullar M, Ahuja M, Kohli HS, Bhansali A, Mohan V, Venkatesan R, Rai TS, Sud K, Singal PK. Common vari ants of inflammatory cytokine genes are associated with risk of nephropathy in type 2 diabetes among Asian Indians. PLoS One 2009; 4(4): e5168.
27. Ting KH, Ueng KC, Chiang WL, Chou YE, Yang SF, Wang PH. Relationship of genetic polymorphisms of the chemokine, CCL5, and its receptor, CCR5, with coronary ar tery disease in Taiwan. Evid Based Comple ment Alternat Med 2015; 2015: 851683.
28. Pandey BN, Jahangeer MD, Kumar D, Vatsa S, Verma DK. Genetic structure and microgenetic differentiation among popu lations of Terai belt of Bihar, India. Euro J Exp Bio 2013; 3(6): 8087.
29. Yonekura S, Okamoto Y, Horiguchi S, Sakurai D, Chazono H, Hanazawa T, Okawa T, Aoki S, Konno A. Effects of aging on the natural history of seasonal allergic rhini tis in middleaged subjects in South chiba, Japan. Int Arch Allergy Immunol 2012; 157(1): 7380.
30. Weng CJ, Chien MH, Lin CW, Chung TT, Zavras AI, Tsai CM, Chen MK, Yang SF. Ef fect of CC chemokine ligand 5 and CC che mokine receptor 5 genes polymorphisms on the risk an clinicopathological devel opment of oral cancer. Oral Oncol 2010; 46(10): 767772.
31. Tahara T, Shibata T, Okubo M, Ishizuka T, Kawamura T, Yamashita H, Nakamura M, Nakagawa Y, Nagasaka M, Arisawa T, Ohmiya N, Hirata I. Effect of RANTES gene promoter genotypes in patients with ulcer ative colitis. Biomed Rep 2014; 2(4): 602 606.
32. AlQahtani A, Alarifi S, AlOkail M, Hus sain Z, Abdo A, Sanai F, AlAnazi M, Khalaf N, AlHumaidan H, AlAhdal M, Almajhdi FN. RANTES gene polymorphisms (-403G>A and -28C>G) associated with hepatitis B virus infection in a Saudi pop ulation. Genet Mol Res 2012; 11(2): 855 862.
33. Lian LH, Kee BP, Ng HL, Chua KH. Lack of association between RANTES -28, SDF 1 gene polymorphisms and systemic lupus erythematosus in the Malaysian population. Genet Mol Res 2011; 10(4): 2841-2850.
34. Zhao L, Zou L, Han N, Qiu J, Li F, Sun X, Zhang L, Li, C. The metaanalysis of CCR5 genetic variants and diabetic nephropathy risk. Int J Clin Exp Med 2016; 9(8): 15582 15588.
35. de Oliveira AP, Bernardo CR, Camargo AV, Ronchi LS, Borim AA, de Mattos CC, de Campos Júnior E, Castiglioni L, Netinho JG, Cavasini CE, Bestetti RB, de Mattos LC. Genetic susceptibility to cardiac and di gestive clinical forms of chronic chagas dis ease: involvement of the CCR5 59029 A/G polymorphism. PLoS One 2015: 10(11): e0141847.
36. Lima G, SotoVega E, AtishaFregoso Y, SánchezGuerrero J, Vallejo M, Vargas Alarcón G, Llorente L. MCP1, RANTES, and SDF1 polymorphisms in Mexican pa tients with systemic lupus erythematosus. Hum Immunol 2007; 68(12): 980985.
37. Bagci B, Bagci G, Huzmeli C, Sezgin I, Oz demir O. Associations of fractalkine recep tor (CX3CR1) and CCR5 gene variants with hypertension, diabetes and atherosclerosis in chronic renal failure patients undergoing hemodialysis. Int Urol Nephrol 2016; 48(7): 11631170.
38. Mlynarski WM, Placha GP, Wolkow PP, Bochenski JP, Warram JH, Krolewski AS. Risk of diabetic nephropathy in type 1 dia betes is associated with functional polymor phisms in RANTES receptor gene (CCR5): a sexspecific effect. Diabetes 2005; 54(11): 33313335.
Author notes
Corresponding author: Nina Valadez González. Regional Research Center “Dr. Hideyo Noguchi”. Hematology Lab., Autonomous University of Yucatán, Mérida, Yucatán, México. Email: valadez@correo.uady.mx