REVIEW ARTICLE
Received: 28 March 2022
Accepted: 11 April 2022
DOI: https://doi.org/10.21470/1678-9741-2022-0142
ABSTRACT:
Introduction
The second best conduit for coronary artery bypass grafting is uncertain. The
objective of this study is to determine the second best conduit according to
graft patency results from randomized controlled trials using a network
meta-analysis.
Methods
A systematic literature search was conducted for randomized controlled trials
comparing the angiographic patency rate of the no-touch saphenous vein
(NT-SV), the radial artery (RA), the right internal thoracic artery (RITA),
and the gastroepiploic artery (GEA) in reference to the conventionally
harvested saphenous vein (CON-SV). The primary outcome was graft occlusion,
and the secondary outcome was all-cause mortality.
Results
A total of 859 studies were retrieved, of which 18 were included. A total of
6,543 patients and 8,272 grafts were analyzed. The weighted mean
angiographic follow-up time was 3.5 years. Compared with CON-SV, RA
(incidence rate ratio [IRR] 0.56; 95% confidence interval [CI], 0.43-0.74)
and NT-SV (IRR 0.56; 95% CI, 0.44-0.70) demonstrated lower graft occlusion.
NT-SV and RA were ranked as the best conduits (rank score for NT-SV 0.88 vs.
0.87 for RA, 0.29 for GEA, 0.27 for CON-SV, and 0.20 for RITA). There was no
significant difference in late mortality between different conduit
types.
Conclusion
RA and NT-SV are associated with significantly lower graft occlusion rates
and are comparably ranked as the best conduit for patency.
Keywords: Coronary Artery Bypass, Coronary Artery Bypass Grafting, Angiography, Graft Patency, Coronary Artery Disease.

INTRODUCTION
The long-term benefit of coronary artery bypass grafting (CABG) is dependent on durable patency of the conduits used. The left internal thoracic artery (LITA) to left anterior descending (LAD) bypass is universally accepted as the gold-standard that confers the greatest survival benefit. Between a selection of arterial grafts and the saphenous vein, the second conduit of choice remains controversial[1].
Compared to the saphenous vein grafts, arterial grafts are advocated for long-term patency and resistance to progressive graft atherosclerosis[2]. However, minimal handling of the saphenous vein during harvesting has provided vein graft patency rates that are on par with their arterial counterparts[3]. A comprehensive network meta-analysis (NMA) of graft patency in randomized controlled trials (RCTs) was previously completed by our group[4]. The key findings were that the radial artery (RA) and no-touch saphenous vein (NT-SV) grafts were associated with significantly lower graft occlusion rates compared with the conventionally harvested saphenous vein (CON-SV), with RA demonstrating the best patency[4]. The systematic review of this study was completed in 2019. Since then, additional RCTs with pairwise comparisons of two or more conduit types have been published (including one very large study comparing CON-SV and NT-SV)[3], and previous studies have been updated with long-term results[2,5,6]. We have therefore updated the previously published NMA of the RCTs comparing graft patency of all conduit options in CABG, in an effort to provide high-level evidence to guide graft selection.
METHODS
No human subjects were involved; therefore, ethical approval of this analysis was not required. The data that support the findings of this study are available from the corresponding author upon request.
Search Strategy
For the previous NMA[4], a medical librarian (M.D.) had performed a comprehensive literature search, on November 11, 2019, of RCTs that compared CON-SV, NT-SV, RA, the right internal thoracic artery (RITA), or the gastroepiploic artery (GEA). For this NMA, the same librarian performed an updated search on December 22, 2021 in the following databases: Ovid® MEDLINE®, Ovid® EMBASE®, and the Cochrane Library. The search strategy included the terms “radial artery”, “internal mammary artery”, “internal thoracic artery”, “gastroepiploic artery”, and “saphenous vein”. The full search strategy is available in Table S1. This review was registered with the PROSPERO register of systematic reviews (CRD42022303553).
Search Strategy.

Study Selection and Quality Assessment
Searches across the aforementioned databases retrieved 859 studies. After citations were de-duplicated, two independent reviewers (M.X.D and H.L.) screened a total of 577 references. Discrepancies were resolved by consensus and opinion of a third author (S.E.F.). Titles and abstracts were reviewed against predefined inclusion and exclusion criteria. Articles were appraised for eligibility if they were written in English and were RCTs randomized by conduit type, comparing angiographic patency for at least two of the five conduits (RA, RITA, CON-SV, NT-SV, and GEA) in patients undergoing CABG. Animal studies, case reports, conference presentations, editorials, expert opinions, observational studies, literature review, abstract only publications, and studies not defining or reporting the outcomes of interest were excluded. Two references that were previously acknowledged in the original NMA were removed to avoid duplication.
Eligible abstracts proceeded to full-text review. The full flow diagram outlining the study selection process is shown in Figure S1. For overlapping studies involving the same study cohort with serial assessments over time, the study with the longest angiographic follow-up was included. The 13 studies reported in the original NMA were included in this updated review. The following variables were collected: study demographics (sample size, publication year, institution, country, and inclusion and exclusion criteria), patient demographics (age, sex, and comorbidities), procedure-related variables (number of grafts, distal anastomosis to the left circumflex artery, proximal anastomosis to the ascending aorta, and use of off-pump CABG), and angiographic-related variables (definition of graft occlusion, imaging modality, completeness of angiographic follow-up, and severity of the target vessel stenosis). The quality of the included trials was examined by the Cochrane Collaboration’s tool for assessing risk of bias[7].

Fig. S1
Preferred Reporting Items for Systematic Reviews and Meta-Analyses flow diagram.
The primary outcome was graft occlusion at the protocol-defined angiographic follow-up. The secondary outcome was all-cause mortality.
Statistical Analysis
The incidence rate with underlying Poisson process was used to account for different follow-up times among the studies, with the total number of events observed within a treatment group calculated out of the total person-time follow-up for that treatment group. Pooled crude graft patency results of the different graft types were performed using a random effects model and the generic inverse variance method. Random effects NMA using a frequentist approach was performed using the generic inverse variance method with CON-SV as reference. Pooled graft patency and late mortality were summarized as forest plots and league tables. Rank scores with probability ranks of different treatment groups were calculated for the primary outcome. Ranks closer to 1 indicate the probability that the treatment group leads to the greatest reduction in graft occlusion. Net graphs were constructed summarizing the numbers of direct comparisons of the included trials. Leave-one-out analysis for graft occlusion was done to assess for validity of the main analysis.
Subgroup analyses were performed for studies with target vessel stenosis ≥ 70% and studies that exclusively used computed tomography angiography (CTA) for postoperative graft assessment during follow-up.
The Cochran’s Q statistic was used to assess inconsistency using the decomposition approach. Inconsistencies were assessed based on separate indirect from direct evidence (or SIDE) using back-calculation method and decomposition of within-designs Q statistic. Net heat plot was used to evaluate for inconsistency in the network model. Heterogeneity was reported as low (I2 = 0-25%), moderate (I2 = 26-50%), or high (I2 > 50%).
Pairwise comparisons were also performed to assess the consistency of the network findings. Meta-regression was performed on the pairwise comparisons to explore the effect on the primary outcome of age, sex, hypertension, diabetes mellitus, dyslipidemia, target vessel stenosis, duration of follow-up, completeness of angiographic follow-up, percentage of proximal anastomoses on the ascending aorta, percentage of grafts to the circumflex coronary system, and use of off-pump CABG.
For hypothesis testing purposes, we built 95% confidence intervals (CI) without multiplicity adjustment. All statistical analyses were performed using the “meta” and “netmeta” packages of R (version 4.1.2, R Project for Statistical Computing using R Studio 2021.09.2).
RESULTS
After removal of duplicates, a total of 577 studies were retrieved from the literature search. Two additional studies not identified in the initial search were included after professional consultation (S.E.F.)[3,6]. Of the 579 studies, 13 abstracts proceeded to full-text screen. Ultimately, five additional RCTs were included in the final analysis[3,6,8-10]. Together with the 13 RCTs from the original meta-analysis[2,5,11-21], a total of 18 studies were included in this review (Table 1). The detailed inclusion and exclusion criteria of the individual trials are summarized in Table S2. Three trials were multicenter (two in Canada, one in the United States of America), two originated from Italy, two from Sweden, two from Korea, two from China, two from the United Kingdom, and one each from Belgium, Australia, Norway, Egypt, and Brazil. Two trials used within-patient randomization[12,14]. Both the RITA vs. RA (RAPCO-RITA) and the CON-SV vs. RA (RAPCO-SV) arms of the Radial Artery Patency and Clinical Outcomes (RAPCO) study were included[2]. In the 2005 trial by Gaudino et al.[15], results of graft randomization in the study cohort of patients with coronary in-stent restenosis and the control cohort of patent stents were included. In the 2021 parallel group by Angelini et al.[8], a factorial trial involving four treatment groups, only two of the groups were included - conventional harvest/high-pressure test and pedicled harvest/low-pressure test, representing CON-SV and NT-SV, respectively.
Characteristics of included randomized trials.

Inclusion and exclusion criteria of the included trials.

A total of 6,543 randomized patients were included in the final analysis. Demographics of the included patients are presented in Table S3. The number of patients in the trials ranged from 50 to 2,655. The mean age range was 58.0 to 76.9 years in the CON-SV group, 61.0 to 77.6 years in the NT-SV group, 55.7 to 77.3 years in the RA group, 59.5 to 63.5 years in the RITA group, and 56.1 to 61.9 years in the GEA group. Female patients ranged from 1% to 46% in the CON-SV group, 7% to 44% in the NT-SV group, 0% to 51% in the RA group, 5% to 19% in the RITA group, and 12% to 13% in the GEA group. The prevalence of diabetes mellitus ranged from 4% to 84% in the CON-SV group, 2% to 76% in the NT-SV group, 11% to 49% in the RA group, were 11% in the RITA group, and ranged from 20% to 27% in the GEA group. The details of procedure- and angiography-related variables are shown in Tables S4 and S5, respectively.
Demographics of included patients.

Procedure-related variables by trial.

Angiography-related variables by trial.

A total of 8,272 grafts were analyzed across the 18 included trials: 3,732 CON-SV grafts, 2,647 NT-SV grafts, 1,223 RA grafts, 549 RITA grafts, and 121 GEA grafts. The weighted mean angiographic follow-up time was 3.5 years (95% CI 1.5-5.4). The crude patency rates of the analyzed conduits were as follows: RA 94.1% (95% CI 90.0-97.6); NT-SV 91.4% (95% CI 87.3-94.3); RITA 89.2% (95% CI 71.2-96.5); CON-SV 86.3% (95% CI 81.2-90.2); and GEA 61.2% (95% CI 52.2-69.4). Details of patency rates are given in Table 2.
Pooled patency of different grafts.

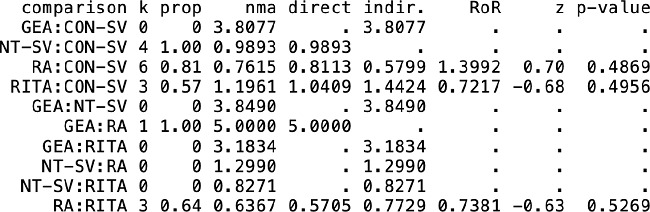
With CON-SV as reference, only RA (incidence rate ratio [IRR] 0.56; 95% CI 0.43-0.74) and NT-SV (IRR 0.56; 95% CI 0.44-0.70) were associated with significantly lower rate of graft occlusion, whereas RITA (IRR 1.06; 95% CI 0.73-1.54) and GEA (IRR 0.98; 95% CI 0.64-1.52) were not (Table 3, Figure 1, Figure 2A). The width of the CI supports a clinically meaningful benefit of RA and NT-SV in comparison to CON-SV. NT-SV was ranked as the best conduit with a rank score of 0.88 vs. 0.87 for RA, 0.29 for GEA, 0.27 for CON-SV, and 0.20 for RITA. These results were confirmed in the individual pairwise meta-analyses (Figure S2 and Table S6A).
League tables summarizing the results of the network meta-analysis (expressed as incidence rate ratio with 95% confidence interval) for graft occlusion using random effects model.


Fig. 1
Forest plot for graft occlusion for the different conduits. CI=confidence interval; CON-SV=conventionally harvested saphenous vein; GEA=gastroepiploic artery; IRR=incidence rate ratio; NT-SV=no-touch saphenous vein; RA=radial artery; RITA=right internal thoracic artery.

Fig. 2
Net graph of the different comparisons for A) the primary outcome of graft occlusion and B) the secondary outcome of late mortality. Width of the lines indicate the number of studies comparing each pair of treatment. In the network plots, colored polygons indicate the presence of multi-arm (3 or more) trials, whereas line shading and thickness are inversely proportional to standard errors of the fixed effect estimate stemming from direct between-arm comparisons. CON-SV=conventionally harvested saphenous vein; GEA=gastroepiploic artery; NT-SV=no-touch saphenous vein; RA=radial artery; RITA=right internal thoracic artery.

Fig. S2
Forest plot for the pairwise comparison of graft occlusion for A) radial artery (RA) vs. conventionally harvested saphenous vein (CON-SV); B) no-touch saphenous vein (NT-SV) vs. CON-SV; C) right internal thoracic artery (RITA) vs. CON-SV; D) RA vs. RITA; E) and NT-SV vs. RA. CI=confidence interval; IRR=incidence rate ratio; RAPCO=Radial Artery Patency and Clinical Outcomes; RAPS=Radial Artery Patency Study; RSVP=Radial Artery Versus Saphenous Vein Patency; SAVE-RITA=Saphenous Vein versus Right Internal Thoracic Artery; seTE=standard error of treatment estimate; SV=saphenous vein; TE=estimate of treatment effect, e.g., log hazard ratio or risk difference.
The results of the sensitivity analysis for target vessel stenosis ≥ 70% showed superiority of RA (IRR, 0.49; 95% CI, 0.30-0.82) to CON-SV, but no significant difference between NT-SV (IRR, 0.58; 95% CI, 0.25-1.31) and CON-SV (Figure S3). Studies using CTA for graft assessment were consistent with the primary analysis (Figure S4).

Fig. S3
Subgroup analysis for the primary outcome in studies with target vessel stenosis ≥ 70%. CI=confidence interval; CON-SV=conventionally harvested saphenous vein; GEA=gastroepiploic artery; IRR=incidence rate ratio; NT-SV=no-touch saphenous vein; RA=radial artery; RITA=right internal thoracic artery.

Fig. S4
Sensitivity analyses for studies using computed tomography angiography exclusively for postoperative graft assessment. There were not enough studies reporting data for the right internal thoracic artery and the gastroepiploic artery. CI=confidence interval; CON-SV=conventionally harvested saphenous vein; IRR=incidence rate ratio; NT-SV=no-touch saphenous vein; RA=radial artery.
Late mortality was comparable between conduits at a weighted mean follow-up time of 3.5 years (Figures 2B and 3, Tables 4 and S6B). The network RA vs. GEA comparison appeared to favor RA, with limited data - although only one study directly compared the two conduits[20].
League tables summarizing the results of the network meta-analysis (expressed as incidence rate ratio with 95% confidence interval) for late mortality using random effects model.

Summary of different pairwise comparisons using random effects modeling for A) graft occlusion and B) late mortality. For each pairwise comparison, the second group is the reference arm.


Fig. 3
Forest plot for late mortality for the different conduits. CI=confidence interval; CON-SV=conventionally harvested saphenous vein; GEA=gastroepiploic artery; IRR=incidence rate ratio; NT-SV=no-touch saphenous vein; RA=radial artery; RITA=right internal thoracic artery.
Heterogeneity/inconsistency estimates and net split are shown in Tables S7 and S8, and in the net heat plot shown in Figure S5. Overall heterogeneity was low (I2 < 5%) for graft patency and late mortality (Table S8). Risk of bias was low for most of the trials (Table 5).
Assessment of inconsistency based on separate indirect from direct evidence (or SIDE) using back-calculation method and random effects model.

Quantifying heterogeneity.

Assessment of risk of bias using the Cochrane Collaboration’s tool.


Fig. S5
Net heat plot evaluating for inconsistency (i.e., disagreement between direct and indirect evidence) in the network model for A) graft patency and B) late mortality. The areas of gray squares represent the relative contributions of designs listed in the columns to the network estimate of designs listed in the rows. The colors are associated with changes in inconsistency between direct and indirect evidence in designs listed in the rows after detaching the effect of designs listed in the columns. Yellow colors indicate a decrease (the stronger the intensity of the color, the stronger the change). CON-SV=conventionally harvested saphenous vein; GEA=gastroepiploic artery; NT-SV=no-touch saphenous vein; RA=radial artery; RITA=right internal thoracic artery.
Leave-one-out analysis and funnel plot did not identify strong evidence of invalidity of the main analysis (Figures S6 and S7).

Fig. S6
Leave-one-out analysis for graft occlusion in A) radial artery (RA) vs. conventionally harvested saphenous vein (CON-SV); B) no-touch saphenous vein (NT-SV) vs. CON-SV; C) right internal thoracic artery (RITA) vs. CON-SV; D) RA vs RITA; E) RA vs. NT-SV. CI=confidence interval; IRR=incidence rate ratio; RAPCO=Radial Artery Patency and Clinical Outcomes; RAPS=Radial Artery Patency Study; RSVP=Radial Artery Versus Saphenous Vein Patency; SV=saphenous vein.

Fig. S7
Funnel plot for all studies. CON-SV=conventionally harvested saphenous vein; GEA=gastroepiploic artery; NT-SV=no-touch saphenous vein; RA=radial artery; RITA=right internal thoracic artery.
Meta-regression
Comparing RA and CON-SV, the percentage of off-pump technique use was directly associated, and the percentage of female patients was inversely associated with the IRR for the primary outcome of graft occlusion. There was no significant association between the variables and other graft comparisons in the meta-regression (Table S9).
Meta-regression for the primary outcome of graft occlusion.

DISCUSSION
In this NMA of 18 RCTs (8,272 grafts), we found that compared with CON-SV, RA and NT-SV have significantly lower occlusion rate at a mean weighted follow-up time of 3.5 years. NT-SV and RA ranked as the best conduits, whereas there was no strong evidence for greater patency in RITA and right GEA when compared to CON-SV.
Currently, there is still a lack of consensus on the second best conduit after the LITA to LAD bypass for non-LAD targets. Meta-analysis of angiographic RCTs allows a robust understanding of patency rates of various conduits while minimizing confounding and risk of bias. By amalgamating the randomized trials, a meta-analysis is the highest level of evidence available. Additionally, NMA provides the advantage of facilitating indirect comparisons of multiple interventions, thereby increasing the power of the analysis.
The comparison between NT-SV and CON-SV was assessed by the largest RCT included in our NMA, with 2,655 randomized patients[3]. Tian et al.[3] reported a lower rate of graft occlusion at 12 months compared to CON-SV, with an odds ratio (OR) of 0.56 (95% CI, 0.41-0.76; P<0.001); however, there was no difference in major adverse cardiac and cerebrovascular events. The caveat of NT-SV is a higher rate of leg wound surgical intervention at three months of follow-up (OR 2.55; 95% CI, 1.85-3.52; P<0.001)[3]. Deb et al.[13] also showed an over two fold increase in the rate of leg infections (P<0.01) and more severe infection with NT-SV (P=0.004) at 30 days, compared to CON-SV. Due to an increased risk of harvest-site complications, guidelines recommend NT-SV harvest technique only in patients with low risk of wound complications[22]. The NT-SV received a Class IIa recommendation in the 2018 European Revascularization guidelines[23] and was a Best Practice in the 2021 American College of Cardiology/American Heart Association revascularization guidelines[22].
Several large RCTs support the long-term patency of RA over CON-SV[2,11,12]. The Radial Artery Database International Alliance (RADIAL) database also reported lower 10-year composite outcome of death, myocardial infarction, or repeat revascularization for patients who received RA relative to CON-SV[24]. Conversely, the Arterial Revascularization Trial (ART) did not find a difference in survival and event-free survival at 10 years among patients randomized to receive RITA[25]. However, the ART trial is criticized for its high crossover between single and bilateral internal thoracic artery (BITA) groups and confounding from RA use, which may have diminished the clinical benefit of RITA. In an as-treated analysis of the ART trial, non-randomized data showed a meaningful difference in mortality in favor of multiple arterial grafts. The merit of multiple vs. single arterial grafting in improving cardiovascular events and death in patients after CABG is currently being investigated in the ROMA trial (Randomized Comparison of the Outcome of Single versus Multiple Arterial Grafts. ClinicalTrials.gov registration number: 1703018094)[26].
The use of RA received a Class I indication and is preferred to saphenous vein as the second most important conduit for a significantly stenosed, non-LAD vessel in the 2021 American revascularization guidelines[22]. Although RA is a versatile graft, calcium channel blockers are routine adjuncts to prevent vasospasm. RA should only be used to bypass severely stenotic target vessels due to the risk of string sign in the setting of competitive flow.
These findings challenge the previously accepted belief that RITA is the natural second conduit of choice due to its biophysiological similarity with LITA. The explanation is multifactorial. Firstly, there are less randomized evidence regarding RITA and CON-SV when compared to RA and CON-SV (three trials including a total of 353 patients for RITA, seven trials including a total of 841 patients for RA). Secondly, the RAPCO trial used RITA as a free graft, which may affect graft patency. Thirdly, BITA surgery is more technically challenging than using RA and LITA, with successful application of RITA reliant on surgeon experience. This may partly explain the 14% crossover from BITA to the single internal thoracic artery in the ART trial[25]. Even though the ART trial recruited surgeons with over 50 BITA cases of experience, there was still a wide variation of intraoperative BITA conversion rates across surgeons, which highlights the technical demand of successful BITA grafting[27].
There were no differences in late mortality for any of the second conduits, including RA, compared to the control saphenous vein graft. The association between graft patency and survival is biologically sound and demonstrated by the five-year results of the RADIAL database, where there is a concordant association between improved patency of RA compared to the control saphenous vein and reduction of myocardial infarction and repeat revascularization[28]. These results are further substantiated in the RADIAL 10-year extension study’s post-hoc analysis for survival[24]. In the NMA and pairwise comparisons, survival in RA patients was greater than in RITA patients, but it did not cross the threshold for statistical significance (95% CI of 1.00). The data for RA vs. GEA comparison was limited.
In the previous NMA, RA was ranked as the best conduit[4]. In this updated NMA, the introduction of five additional trials has led NT-SV to achieve a higher patency ranking than RA, albeit by a very small margin. Of the five RCTs, three investigated NT-SV and CON-SV (n=2,805)[3,8,9], one compared RA and CON-SV (n=50)[10], and one assessed RITA and RA (n=224)[6]. The increased sample size in NT-SV and CON-SV enhanced the power of analysis in favor of NT-SV. Many of the newly added trials reported early-term results, which likely inflated pooled saphenous vein patency and decreased the weighted mean follow-up time of the NMA from 5.1 to 3.5 years. In keeping with the 2021 NMA findings[4], no conduit provided a statistically significant mortality benefit over CON-SV. Meta-regression for IRR of graft occlusion continued to suggest a positive association with off-pump CABG use (i.e., increased graft occlusion) and inverse association with increased proportion of female patients (i.e., decreased graft occlusion)[4].
Limitations
Limitations of this meta-analysis included a small sample size causing certain pairwise analyses to be underpowered, varying quality of the RCTs included, and no data collected on renal disease, secondary prevention, and antispasmodic therapy, which are additional factors that influence graft patency. It is worthwhile to note that the included studies involving NT-SV grafts used pedicle harvest technique with[8,19,21] or without manual dilatation with a syringe[3,5,9,13,14]. The factorial trial by Angelini et al. involving CON-SV vs. NT-SV and low- vs. high-pressure graft dilation reported that low-pressure distention of CON-SV can achieve wall thickening comparable to NT-SV[8].
CONCLUSION
In this NMA of 18 angiographic RCTs, the current randomized evidence shows significantly better patency rates for RA and NT-SV compared with CON-SV, while all conduits were associated with similar rates of late mortality compared with CON-SV. NT-SV and RA were identified as the second best conduits using data from this NMA of angiographic trials.

ACKNOWLEDGEMENTS
We would like to thank Dr. Angelini, Dr. Chris Rogers, and Dr. Rebecca Evans for allowing us to include supplementary data[8] in this NMA.
REFERENCES
Gaudino M, Antoniades C, Benedetto U, Deb S, Di Franco A, Di Giammarco G, et al. Mechanisms, consequences, and prevention of coronary graft failure. Circulation. 2017;136(18):1749-64. doi:10.1161/CIRCULATIONAHA.117.027597.
Buxton BF, Hayward PA, Raman J, Moten SC, Rosalion A, Gordon I, et al. Long-term results of the RAPCO trials. Circulation. 2020;142(14):1330-8. doi:10.1161/CIRCULATIONAHA.119.045427.
Tian M, Wang X, Sun H, Feng W, Song Y, Lu F, et al. No-touch versus conventional vein harvesting techniques at 12 months after coronary artery bypass grafting surgery: multicenter randomized, controlled trial. Circulation. 2021;144(14):1120-9. doi:10.1161/CIRCULATIONAHA.121.055525.
Gaudino M, Hameed I, Robinson NB, Ruan Y, Rahouma M, Naik A, et al. Angiographic patency of coronary artery bypass conduits: a network meta-analysis of randomized trials. J Am Heart Assoc. 2021;10(6):e019206. doi:10.1161/JAHA.120.019206.
Samano N, Geijer H, Liden M, Fremes S, Bodin L, Souza D. The no-touch saphenous vein for coronary artery bypass grafting maintains a patency, after 16 years, comparable to the left internal thoracic artery: a randomized trial. J Thorac Cardiovasc Surg. 2015;150(4):880-8. doi:10.1016/j.jtcvs.2015.07.027.
Kim MS, Kim KB. Saphenous vein versus right internal thoracic artery as a y-composite graft: ten-year angiographic and long-term clinical results of the SAVE RITA trial. Circulation. 2021;144(14):1186-8. doi:10.1161/CIRCULATIONAHA.121.056438.
Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. doi:10.1136/bmj.l4898.
Angelini GD, Johnson T, Culliford L, Murphy G, Ashton K, Harris T, et al. Comparison of alternate preparative techniques on wall thickness in coronary artery bypass grafts: the HArVeST randomized controlled trial. J Card Surg. 2021;36(6):1985-95. doi:10.1111/jocs.15477.
Hou X, Zhang K, Liu T, Li Y, Zhao Y, Song B, et al. The expansion of no-touch harvesting sequential vein graft after off-pump coronary artery bypass grafting. J Card Surg. 2021;36(7):2381-8. doi:10.1111/jocs.15577.
Hagras M, Elsharkawy A, Toure O. Comparative study between radial artery versus saphenous vein grafts patency after 5 years. J Card Disease Res. 2021;12(5):187-93.
Collins P, Webb CM, Chong CF, Moat NE; Radial Artery Versus Saphenous Vein Patency (RSVP) Trial Investigators. Radial artery versus saphenous vein patency randomized trial: five-year angiographic follow-up. Circulation. 2008;117(22):2859-64. doi:10.1161/CIRCULATIONAHA.107.736215.
Deb S, Cohen EA, Singh SK, Une D, Laupacis A, Fremes SE, et al. Radial artery and saphenous vein patency more than 5 years after coronary artery bypass surgery: results from RAPS (radial artery patency study). J Am Coll Cardiol. 2012;60(1):28-35. doi:10.1016/j.jacc.2012.03.037.
Deb S, Singh SK, de Souza D, Chu MWA, Whitlock R, Meyer SR, et al. SUPERIOR SVG: no touch saphenous harvesting to improve patency following coronary bypass grafting (a multi-centre randomized control trial, NCT01047449). J Cardiothorac Surg. 2019;14(1):85. doi:10.1186/s13019-019-0887-x.
Dreifaldt M, Mannion JD, Geijer H, Lidén M, Bodin L, Souza D. The no-touch saphenous vein is an excellent alternative conduit to the radial artery 8 years after coronary artery bypass grafting: a randomized trial. J Thorac Cardiovasc Surg. 2021;161(2):624-30. doi:10.1016/j.jtcvs.2019.09.177.
Gaudino M, Cellini C, Pragliola C, Trani C, Burzotta F, Schiavoni G, et al. Arterial versus venous bypass grafts in patients with in-stent restenosis. Circulation. 2005;112(9 Suppl):I265-9. doi:10.1161/CIRCULATIONAHA.104.512905.
Glineur D, D'hoore W, El Khoury G, Sondji S, Kalscheuer G, Funken JC, et al. Angiographic predictors of 6-month patency of bypass grafts implanted to the right coronary artery a prospective randomized comparison of gastroepiploic artery and saphenous vein grafts. J Am Coll Cardiol. 2008;51(2):120-5. doi:10.1016/j.jacc.2007.09.030.
Goldman S, Sethi GK, Holman W, Thai H, McFalls E, Ward HB, et al. Radial artery grafts vs saphenous vein grafts in coronary artery bypass surgery: a randomized trial. JAMA. 2011;305(2):167-74. doi:10.1001/jama.2010.1976.
Muneretto C, Bisleri G, Negri A, Manfredi J, Carone E, Morgan JA, et al. Left internal thoracic artery-radial artery composite grafts as the technique of choice for myocardial revascularization in elderly patients: a prospective randomized evaluation. J Thorac Cardiovasc Surg. 2004;127(1):179-84. doi:10.1016/j.jtcvs.2003.08.004.
Pettersen Ø, Haram PM, Winnerkvist A, Karevold A, Wahba A, Stenvik M, et al. Pedicled vein grafts in coronary surgery: perioperative data from a randomized trial. Ann Thorac Surg. 2017;104(4):1313-7. doi:10.1016/j.athoracsur.2017.03.076.
Santos GG, Stolf NA, Moreira LF, Haddad VL, Simões RM, Carvalho SR, et al. Randomized comparative study of radial artery and right gastroepiploic artery in composite arterial graft for CABG. Eur J Cardiothorac Surg. 2002;21(6):1009-14. doi:10.1016/s1010-7940(02)00180-x.
Song SW, Sul SY, Lee HJ, Yoo KJ. Comparison of the radial artery and saphenous vein as composite grafts in off-pump coronary artery bypass grafting in elderly patients: a randomized controlled trial. Korean Circ J. 2012;42(2):107-12. doi:10.4070/kcj.2012.42.2.107.
Writing Committee Members, Lawton JS, Tamis-Holland JE, Bangalore S, Bates ER, Beckie TM, et al. 2021 ACC/AHA/SCAI guideline for coronary artery revascularization: a report of the American college of cardiology/American heart association joint committee on clinical practice guidelines. J Am Coll Cardiol. 2022;79(2):e21-e129. Erratum in: J Am Coll Cardiol. 2022;79(15):1547. doi:10.1016/j.jacc.2021.09.006.
Neumann FJ, Sousa-Uva M, Ahlsson A, Alfonso F, Banning AP, Benedetto U, et al. 2018 ESC/EACTS guidelines on myocardial revascularization. Eur Heart J. 2019;40(2):87-165. Erratum in: Eur Heart J. 2019;40(37):3096. doi:10.1093/eurheartj/ehy394.
Gaudino M, Benedetto U, Fremes S, Ballman K, Biondi-Zoccai G, Sedrakyan A, et al. Association of radial artery graft vs saphenous vein graft with long-term cardiovascular outcomes among patients undergoing coronary artery bypass grafting: a systematic review and meta-analysis. JAMA. 2020;324(2):179-87. doi:10.1001/jama.2020.8228.
Taggart DP, Benedetto U, Gerry S, Altman DG, Gray AM, Lees B, et al. Bilateral versus single internal-thoracic-artery grafts at 10 years. N Engl J Med. 2019;380(5):437-46. doi:10.1056/NEJMoa1808783.
Gaudino M, Alexander JH, Bakaeen FG, Ballman K, Barili F, Calafiore AM, et al. Randomized comparison of the clinical outcome of single versus multiple arterial grafts: the ROMA trial-rationale and study protocol. Eur J Cardiothorac Surg. 2017;52(6):1031-40. doi:10.1093/ejcts/ezx358.
Benedetto U, Altman DG, Flather M, Gerry S, Gray A, Lees B, et al. Incidence and clinical implications of intraoperative bilateral internal thoracic artery graft conversion: insights from the arterial revascularization trial. J Thorac Cardiovasc Surg. 2018;155(6):2346-55.e6. doi:10.1016/j.jtcvs.2018.02.012.
Gaudino M, Benedetto U, Fremes S, Biondi-Zoccai G, Sedrakyan A, Puskas JD, et al. Radial-artery or saphenous-vein grafts in coronary-artery bypass surgery. N Engl J Med. 2018;378(22):2069-77. doi:10.1056/NEJMoa1716026.
Notes
Author notes
Correspondence Address: Stephen E. Fremes, https://orcid.org/0000-0003-1723-3049 University of Toronto, Schulich Heart Centre, Sunnybrook Health Sciences Centre, 2075 Bayview Ave., Room H4 05, Toronto, ON, M4N 3M5, Canada, E-mail: stephen.fremes@sunnybrook.ca
Conflict of interest declaration