ABSTRACT
Introduction: Cardiac surgery is a frequent surgical procedure and may present a high risk of complications. Among the prophylactic strategies studied to decrease the rates of negative outcomes, respiratory care seems to reduce pulmonary complications. Incentive spirometry (IS) is a low-cost, respiratory exercise technique, used for the prevention and treatment of postoperative pulmonary complications (PPC). The aim of this review was to evaluate whether IS is superior to respiratory care, mobilization exercises, and noninvasive ventilation on PPC, and clinical outcomes.
Methods: Systematic review. Medical Literature Analysis and Retrieval System Online (or MEDLINE®), Embase®, Cochrane Central Register of Controlled Trials (or CENTRAL), Physiotherapy Evidence Database (or PEDro), Cumulative Index of Nursing and Allied Health (or CINAHL®), Latin American and Caribbean Health Sciences Literature (or LILACS), Scientific Electronic Library Online (or SciELO), Allied, Scopus®, and OpenGrey databases, clinical trial registration sites, conferences, congresses, and symposiums were searched.
Results: Twenty-one randomized trials and one quasi-randomized trial (1,677 participants) were included. For partial pressure of oxygen (PaO2), IS was inferior to respiratory care (mean difference [MD] -4.48; 95% confidence interval [CI] -8.32 to -0.63). Flow-oriented IS was inferior to respiratory care on PaO2 (MD -4.53; 95% CI -8.88 to -0.18). However, compared to respiratory care, flow-oriented IS was superior on recovery vital capacity.
Conclusions: This meta-analysis revealed that IS was not superior to standard respiratory care for PPCs and clinical outcomes, therefore its use should not be widely recommended until further studies with high quality be performed to ensure this clinical guidance.
Keywords: Cardiac Surgical Procedures, Postoperative Care, Noninvasive Ventilation, Systematic Review
REVIEW ARTICLE
Is Incentive Spirometry Superior to Standard Care in Postoperative Cardiac Surgery on Clinical Outcomes and Length of Hospital and Intensive Care Unit Stay? A Systematic Review with Meta-Analysis
Recepción: 05 Abril 2023
Aprobación: 16 Mayo 2023

Cardiac surgery is a frequent surgical procedure. Each year, Australian hospitals perform > 12,000 cardiac surgeries, and a single Brazilian hospital has already performed > 2,900 of these procedures[1,2]. In the United States of America, the cost of cardiac surgery is approximately 1% to 2% of the health budget[3]. The majority of patients undergo coronary artery bypass grafting (CABG), and 74.6% of surgeries are scheduled[4]. Complex cardiac surgery and prolonged hospital length of stay (LOS) may present a high risk of complications and mortality; postoperative mortality has been documented at 4% (valve operations) within the first seven days and 6.4% (overall mortality) within the first postoperative month[4].
Approximately 10.2% to 27.3% of CABG patients present at least one complication, 70.6% after valve surgery, and 84.2% after combined surgery (CABG + valve surgery)[5,6]. Regarding the complications, 2.2% are major adverse cardiovascular events[7], 7.5% are reintubated during the intensive care unit (ICU) stay, which increases the rate of complications[8], 23.2% remain hospitalized in an ICU for more than two days after surgery, and 59.7% remain hospitalized for more than seven days[6]. It seems that when the complication rate increases, hospital LOS and mortality also increase (12% in the ICU and 15.1% in the 30-day period), mainly in older adults[5,9].
Among the prophylactic strategies to decrease these rates of negative outcomes, respiratory care seems to reduce pulmonary complications and minimize postoperative pulmonary dysfunction[10]. As one of the respiratory care techniques, incentive spirometry (IS) is a low-cost, widespread, respiratory exercise technique, used for the prevention and treatment of postoperative pulmonary complications (PPC) in patients undergoing cardiac surgery[11]. IS is a device that provides visual feedback when the patient inhales at a predetermined flow or volume. The patient is required to place the lips firmly around the mouthpiece and to inhale slowly to raise the ball (flow-oriented) or piston/plate (volume-oriented) in the chamber toward the defined target[12].
It has been suggested that patients undergoing cardiac surgery who are more adherent to IS therapy may benefit from a reduced LOS and a reduction in the mortality rate[13]. On the other hand, scientific evidence has suggested that IS does not improve clinical outcomes in different surgical patients[14]. In order to strengthen the scientific findings, our systematic review, performed with strict methodological criteria, is intended to clarify these specific gaps, exclusively in patients undergoing cardiac surgery and assist clinicians in decision making. Our aim was to assess whether IS is superior to respiratory care, mobilization exercises, and noninvasive ventilation (NIV) on PPC, adverse events, mortality, hospital and/or ICU LOS, lung function, oxygenation, and maximal inspiratory pressure (MIP) in patients undergoing cardiac surgery.
We conducted a systematic review following the reporting recommendations proposed by the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (or PRISMA)[15]. The protocol was registered in the International Prospective Register of Systematic Reviews (or PROSPERO) (#CRD42020161009), is available online at https://www.crd.york.ac.uk/prospero/export_record_pdf.php), and was previously published[16].
We searched for randomized and quasi-randomized controlled trials published in any year, in any language. The studies included in this review were required to have enrolled patients aged 18 years or older, who were breathing spontaneously, undergoing cardiac surgeries, and which evaluated the effects of postoperative flow or volume-oriented IS on our pre-defined clinical outcomes. The treatment comparison was made with standard care, such as respiratory care (maximal inspiratory breathing exercises, coughing and deep breathing, supported/assisted coughing, huffing technique, diaphragmatic breathing, fractional inspiration, active cycle of breathing, and autogenic drainage), NIV, and other therapies (mobilization exercise, blow bottles, and verbal encouragement). The mobilization exercises considered in this review were early mobilization programs, active/passive exercises of upper/lower limbs, and physical therapy.
The controlled trials had to have evaluated at least one of the following outcomes:
PPC: For this systematic review, atelectasis and pneumonia were considered.
Adverse events: Any reaction, harm, or complication associated with IS reported in the included studies.
Mortality: All reported deaths were accepted, regardless of cause.
LOS: The number of days spent in hospital after cardiac surgical procedure.
Length of ICU stay: The number of days spent in the ICU after cardiac surgical procedure.
Lung function: Variables evaluated were peak of expiratory flow (PEF), forced expiratory volume in one second (FEV₁), forced vital capacity (FVC), and vital capacity (VC).
Oxygenation: Arterial partial pressure of oxygen (PaO2) and peripheral and central arterial oxygen saturation (SO2) were accepted.
MIP (cmH₂O): MIP measured with digital or analog manovacuometer or manometer was accepted.
The search strategy was sensitive (Supplement 1) to capture all potentially qualifying studies through the Medical Literature Analysis and Retrieval System Online (or MEDLINE®), Embase®, Cochrane Central Register of Controlled Trials (or CENTRAL), Physiotherapy Evidence Database (PEDro), Cumulative Index of Nursing and Allied Health (or CINAHL®), Latin American and Caribbean Health Sciences Literature (or LILACS), Scientific Electronic Library Online (or SciELO), and Scopus® databases, as well as in the OpenGrey database, the main clinical trial registration sites, conferences, congresses, and symposiums in the area described in the protocol[16]. When necessary, we contacted the authors of the clinical trials to request additional data. The snowball technique, which consists of searching the reference lists of the included studies, was used to optimize the search. The search was performed on July 22 and 24, 2022.
Two authors independently selected the studies identified by the search strategy based on eligibility criteria. Duplicate publications were excluded, after which the authors selected the studies by titles and abstracts. Non-randomized trials and studies lacking predefined outcomes were excluded. In some cases, it was necessary to read the full texts. Where reports with the same participants but different outcome measurements or using different time points for the assessments were found, both reports were included. However, the two reports were considered as parts of only one study.
The Rayyan app was used to optimize the process of screening and selecting the studies[17]. Disagreements between authors regarding the inclusion of the study were resolved by a third author. Two authors extracted data independently, and disagreements were also resolved by a third author.
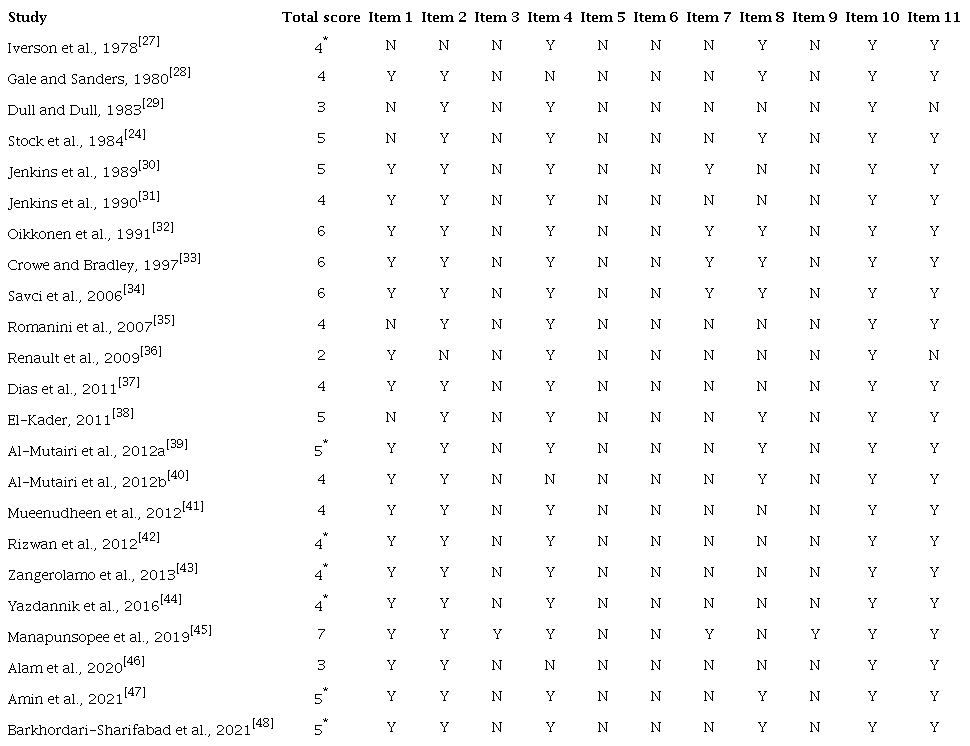
We assessed the methodological characteristics of the trials using the PEDro scale[18]. We used PEDro scores available at https://pedro.org.au/. Where PEDro scores were not available, two previously trained authors evaluated the clinical trials using the PEDro scale. The PEDro methodological rigor scale ranges between 1 and 10, with higher scores indicating higher quality studies. The studies are classified according to the scores as follows: < 4 are considered “poor”, 4 to 5 are considered “fair”, 6 to 8 are considered “good”, and 9 to 10 are considered “excellent”[19]. We assessed the certainty of evidence using the Classification of Recommendations, Assessment, Development and Evaluation (GRADE)[20], through the software GRADEpro in the main outcomes[21].
When at least two studies were sufficiently homogeneous in terms of participants, interventions, and outcome measures, we pooled their results in a meta-analysis. Meta-analyses were performed using an inverse variance method and random effects model in Review Manager version 5.3 (The Nordic Cochrane Center, Copenhagen, Denmark)[22]. Continuous variables were analyzed using the weighted mean differences (MD) and for studies that evaluated the same outcome with different instruments, we used the standardized mean differences (SMD) with 95% confidence interval (CI)[23]. Dichotomous variables were analyzed using risk ratios (RR) with 95% CI.
Trials were pooled according to similarity of intervention, populations, and the outcomes measured. Separate meta-analyses were conducted to examine the effects of IS in the following comparisons:
IS vs. respiratory care.
IS vs. NIV.
IS vs. other therapies.
In case of trials that examined the effects of multiple interventions that were of interest for this review, to avoid double counting the participants, we included two reasonably independent comparisons. However, we split the “shared” group sample size (respiratory care) into two smaller sample sizes. For example, Stock et al. (1984)[24] had three groups in its clinical trial: intervention group (with 12 participants), control group 1 (with 13 participants), and control group 2 (with 13 participants). In this situation, the analysis was performed twice; in the first analysis, the intervention group (with six participants [half the original sample size]) was analyzed vs. control group 1. In the second analysis, the intervention group (with six participants [half the original sample size]) was compared with control group 2.
Therefore, in the included clinical trials with three comparison groups (flow-IS group vs. volume-IS vs. respiratory care), and where data were analyzed twice in our study, we initially identified the name of the main author, and then the year of publication, followed by the letter “a” (Amin et al 2021a: flow-IS group vs. respiratory care) and in the second mention, we identified the name of the main author, and then the year of publication, followed by the letter “b” (Amin et al 2021b: volume-IS group vs. second standard care)[25].
As planned, where appropriate data were available, we carried out subgroup analyses so as to investigate the influence of each comparison on the size of the treatment. Among the preplanned subgroup analyses, it was possible to perform subgroup analyses considering the type of device used (flow-oriented or volume-oriented) in the main comparisons (IS vs. respiratory care; IS vs. NIV; and IS vs. other therapies).
To estimate the heterogeneity across the studies in each meta-analysis, the I2 statistic was used. As suggested in the Cochrane Handbook for Systematic Reviews of Interventions, if heterogeneity was substantial (I2 ≥ 50%), a sensitivity analysis was considered[26]. Although we intended to perform separate analyses for studies with no blinding or deficiency in blinding of outcome assessors, with inappropriate randomization methods, with a large number (> 20%) of patients lost to follow-up, with imputation of standard deviation, or when adherence was not reported, we could not perform sensitivity analyses because we did not find enough studies with appropriate blinding, randomization, or follow-up.
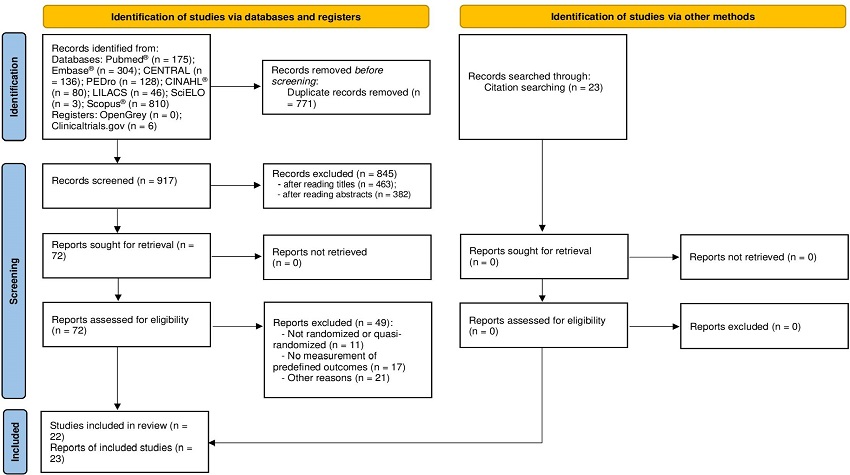
Twenty-three reports of 22 studies were included in this systematic review[27-48]. Twenty-two publications were reported in full; from one clinical trial, only the abstract was reported. One study with two publications was included in this systematic review. The reports of this study were named as Jenkins et al. (1989)[30] and Jenkins et al. (1990)[31], however, as planned, they were considered as part of only one study. The authors of the clinical trial published in abstract format were contacted in an attempt to request additional data[39], however, we did not receive any answers. In this case, we used the data available in the abstract. Twenty-one studies were randomized controlled trials (RCTs) and one was a quasi-randomized trial. The flow chart of this systematic review is shown in Figure 1.
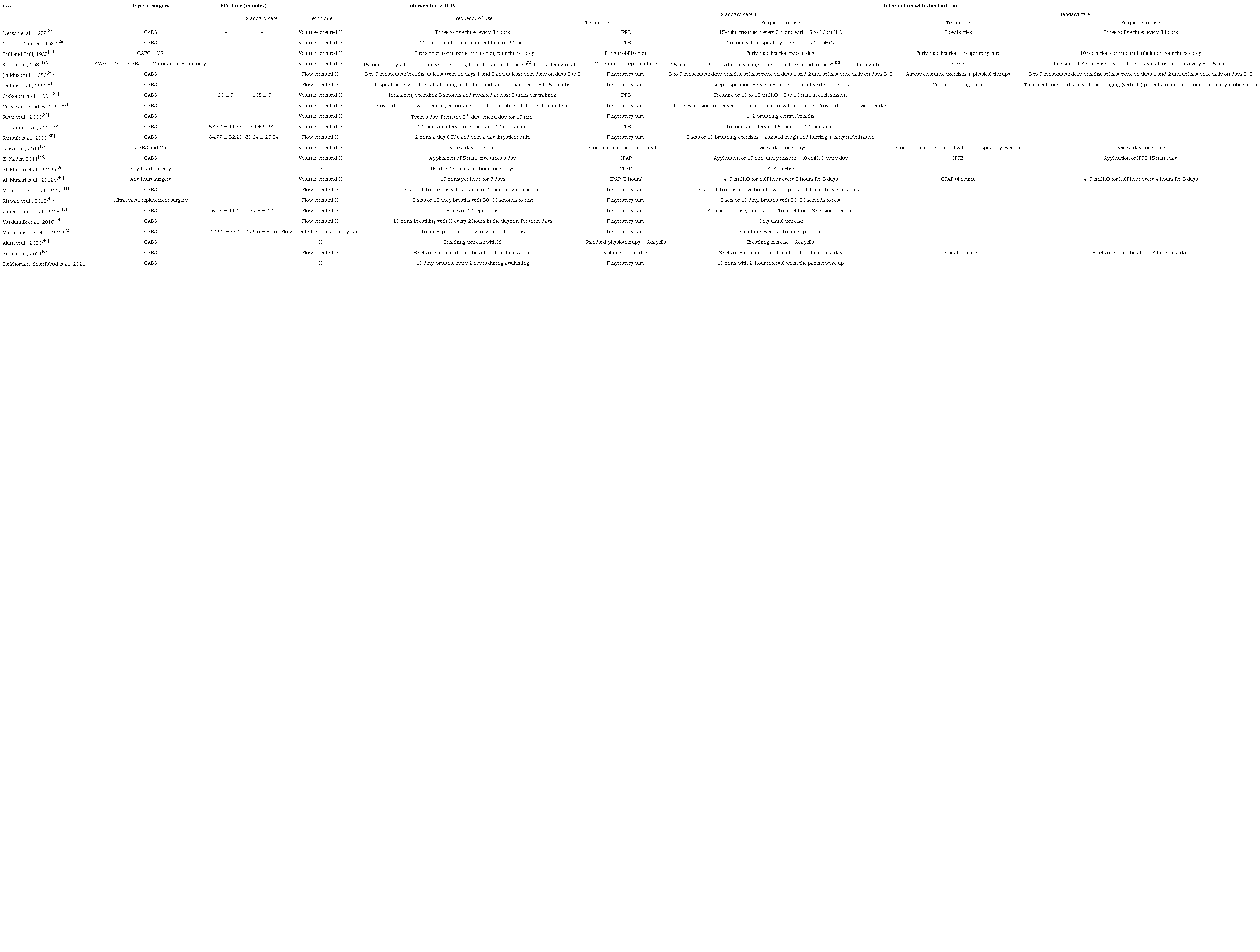
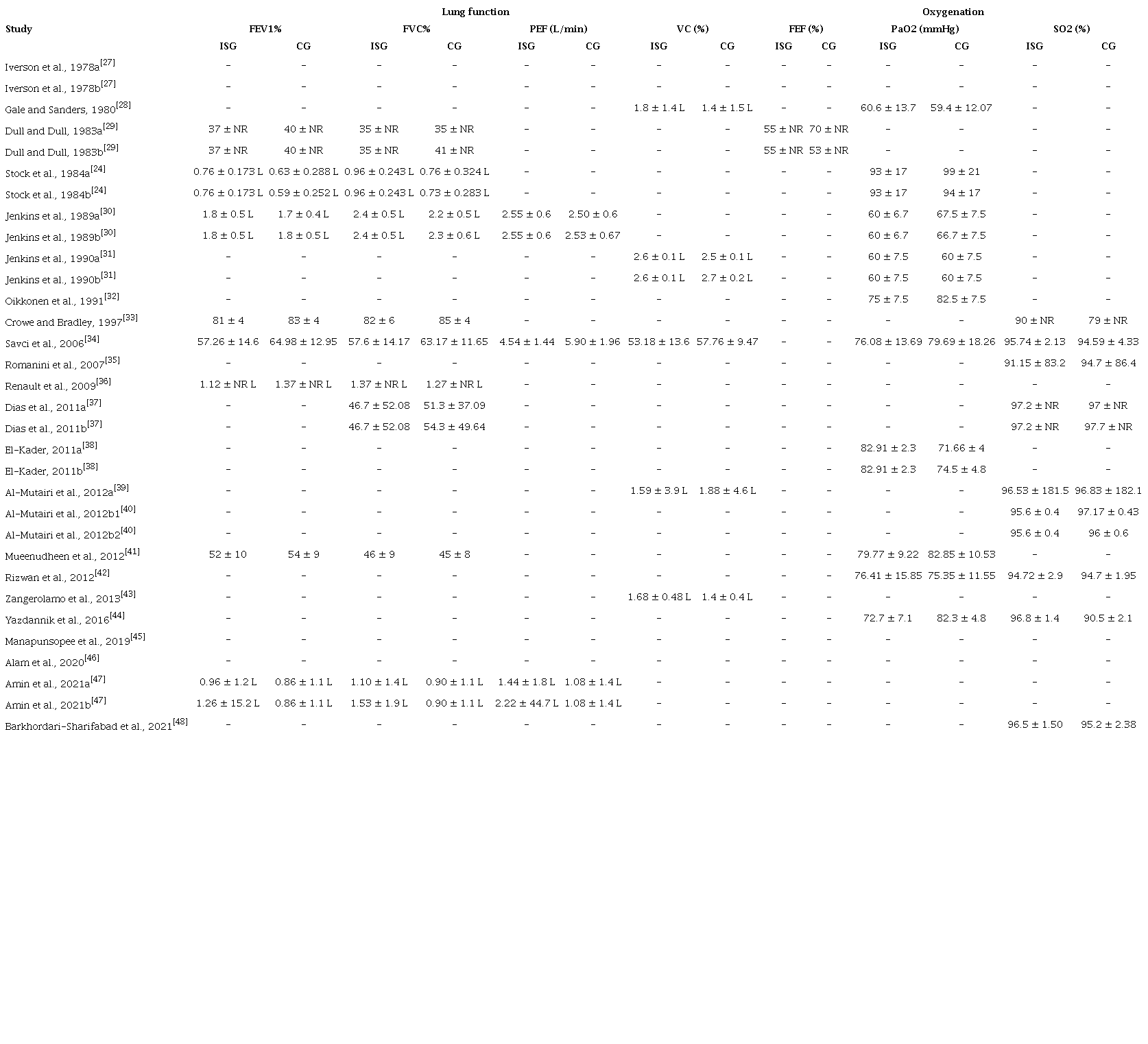
Overall, we included 21 randomized trials and one quasi-randomized controlled trial in this systematic review. The studies involved 1,677 patients, with ages ranging from 38.3 to 65 years[31,45], sample sizes ranging from 16 to 270 participants[43,46], and study follow-up time ranging from two days to hospital discharge (Table 1)[33,39,40,42]. Regarding the characteristics of the surgery and intervention, 74% of patients underwent CABG, 48% of patients received treatment using volume-oriented IS, 39% of patients used flow-oriented IS, and three studies did not have enough information to determine whether the type of spirometer was flowor volume-oriented (Table 2)[39,46,48]. The hospital LOS ranged from 6.5 to 12.5 days, and the length of ICU stay ranged from 2.61 to 6.87 days (Table 3). PaO2 ranged on average from 59.4 mmHg to 99 mmHg[24,28], and SO2 from 79 to 97.7%[35,39].

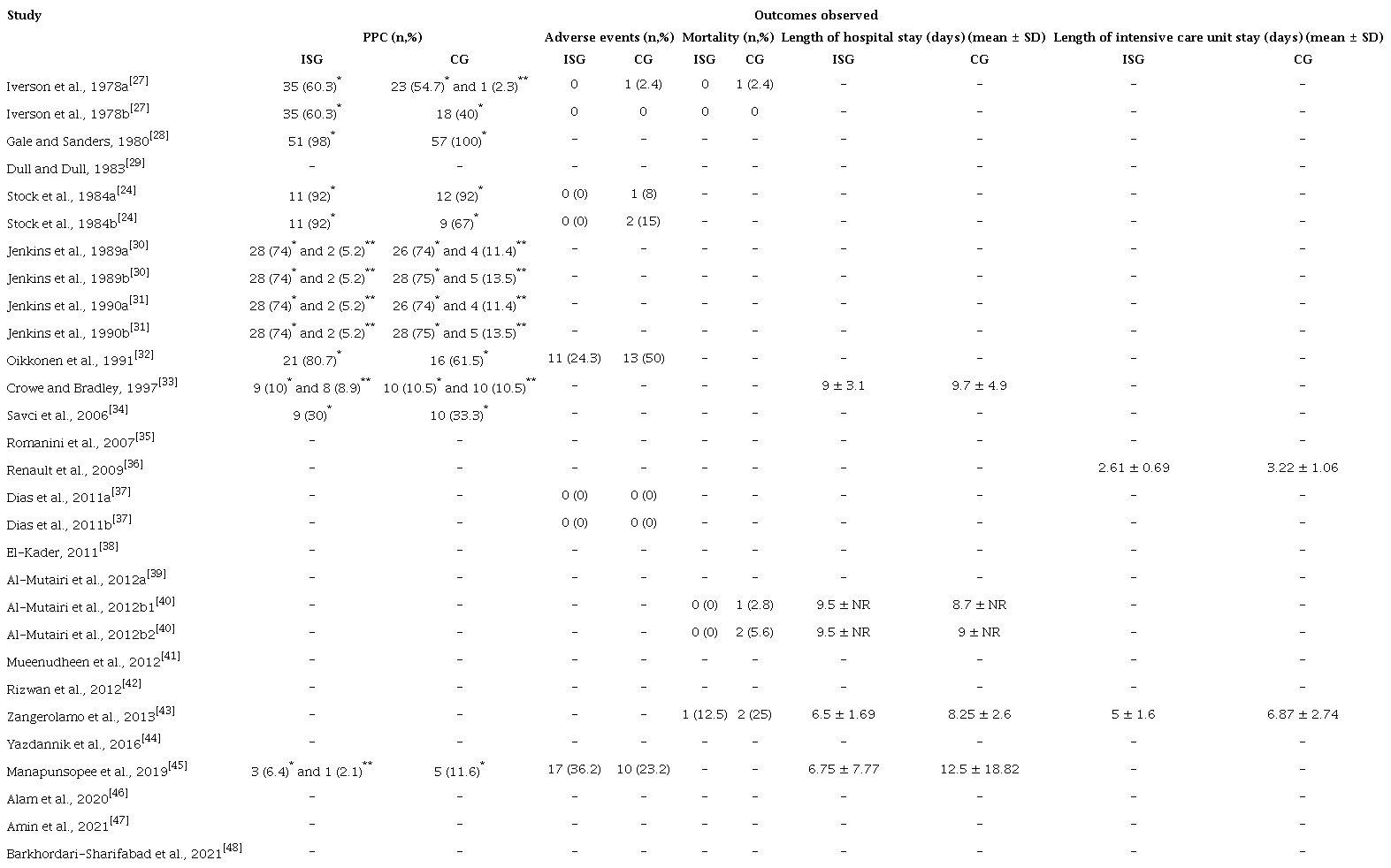
Considering the primary outcomes analysis, among the included studies, nine clinical trials reported PPC rate[24,27,28,30-34,45], five reported adverse events rate[24,27,32,37,45], and three reported mortality rate (Table 3)[26,40,43].
With respect to the secondary outcomes analysis, four trials reported LOS[33,40,43,45], two reported ICU LOS[36,43], eight reported parameters of lung function[24,29,30,33,34,36,41,47], ten reported PaO2, nine reported SO2, and one reported reintubation rate. No trials evaluated the use of antibiotics (which was an outcome of interest for this review[16]).
For these continuous outcomes, results were reported differently across studies, and we performed transformations where it was adequate. In two clinical trials, PaO2 was converted from kilopascals to millimeters of mercury and in one clinical trial the standard deviation was estimated using the Revman calculator[28,31,32]. For some studies the standard deviation was also estimated using the Revman calculator[24,37,39,47]. In one clinical trial, LOS was registered as median, with minimum and maximum, and this was converted to mean and standard deviation for our analysis[45,49] (Table 4). For some studies, transformations were not possible. For instance, one clinical trial recorded forced expiratory flow without standard deviation[29], and insufficient information to estimate the standard deviation. Therefore, we did not pool the results in the meta-analysis. When results were presented using different measures, such as those from studies reporting lung function, which reported values both as a percentage of predicted values and as absolute values in liters, then results were pooled using the SMD.
Among the included studies, in general, the PEDro score ranged from 2 to 7 points, with a mean and standard deviation of 4.5±1.1. For seven trials, the scores were not available on the PEDro platform, therefore, the scores were independently graded by two authors[27,39,42-44,47,48]. After the evaluation of the two authors, three inconsistencies were observed, one on item 11 and two on item 8[27,42,44]. In this situation, a third author was consulted to arbitrate. Considering the PEDro scale, the following percentages of studies did not meet the criteria: on item 1, 21.7%; on item 2, 8.7%; on item 3, 95.7%; on item 4, 13%; on items 5 and 6, 100%; on item 7, 78.2%; on item 8, 52.1%; on item 9, 95.7%; and on item 11, 8.7%. On item 10, all studies met the criteria. In the classification of the PEDro scale, 16 (69.6%) studies were judged as having “fair”[24,27,28,30,31,35,37-44,47,48], four (17.4%) as “good”[32-34,45], three (13%) as “poor”[29,36,46], and zero (0%) as “excellent” quality (Table 5). Considering the low methodological rigor of the studies included in this review, we were not able to perform sensitivity analysis including only high-quality studies.
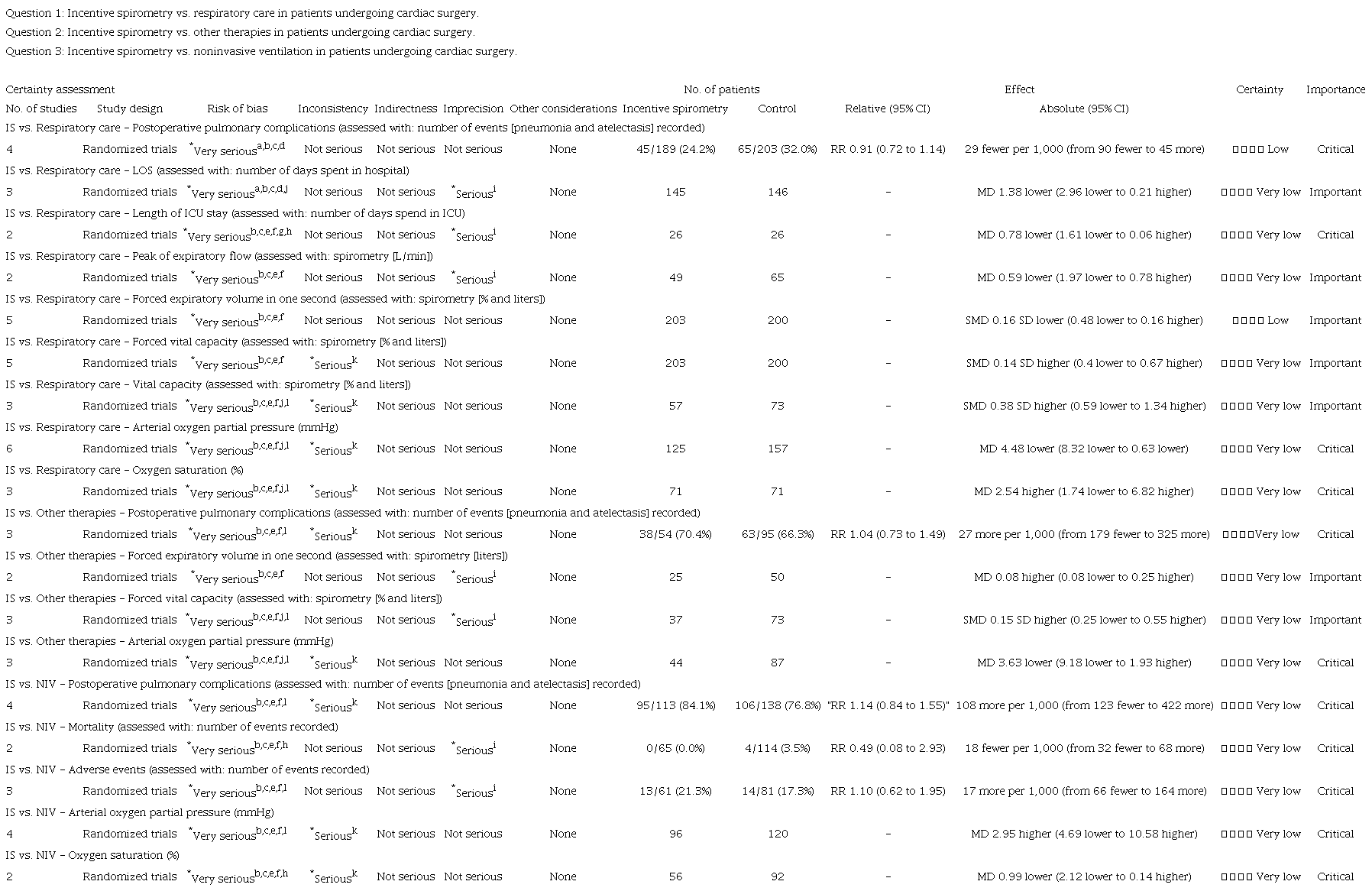
We rated the certainty of the evidence for each outcome in all comparisons using the GRADE approach[21]. The details of each evaluation can be found in Supplement 2.
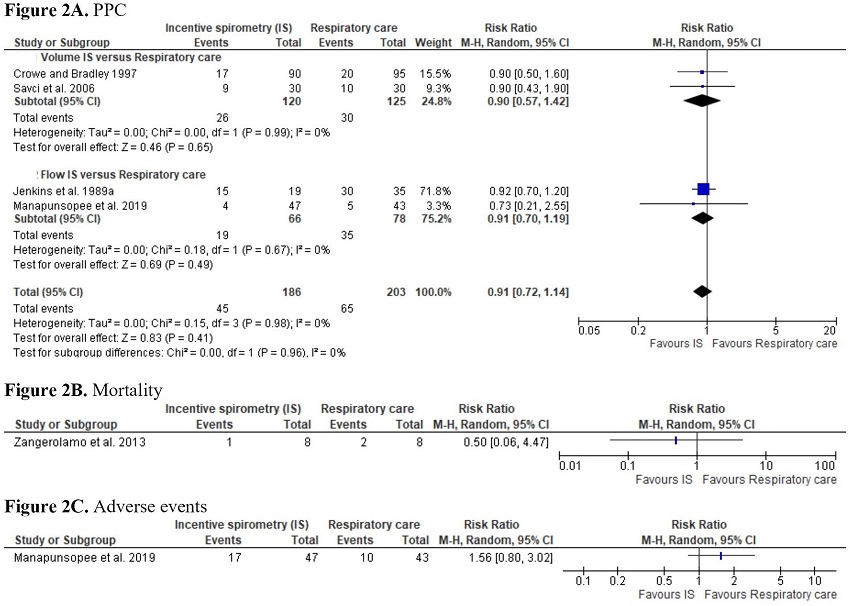
There may be a small difference or no difference on PPC rate between IS and respiratory care (RR 0.91; 95% CI 0.72 to 1.14) (low certainty of evidence) (Supplement 3 - Figure 2A). The evidence is of very low certainty for the other primary outcomes. Only one trial evaluated the mortality rate[43]. This trial also used flow IS and compared its effects to the effects of respiratory care (Supplement 3 - Figure 2B). In the same way, only one trial evaluated the adverse events[45]. This trial used flow IS and compared its effects to the effects of respiratory care (Supplement 3 - Figure 2C).
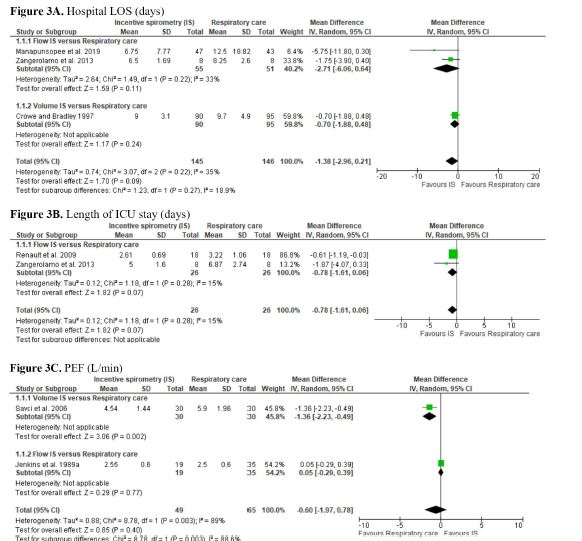
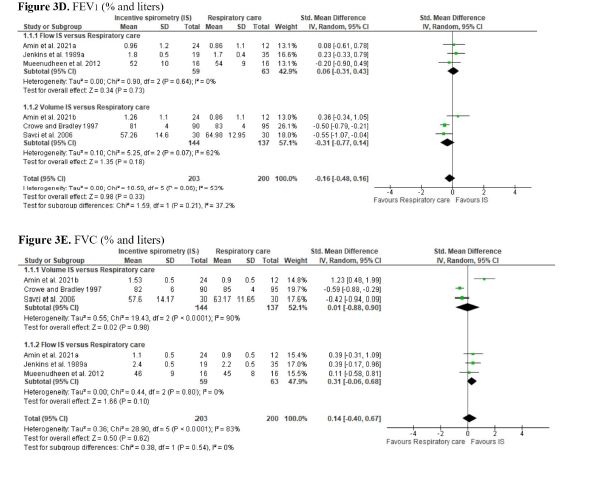
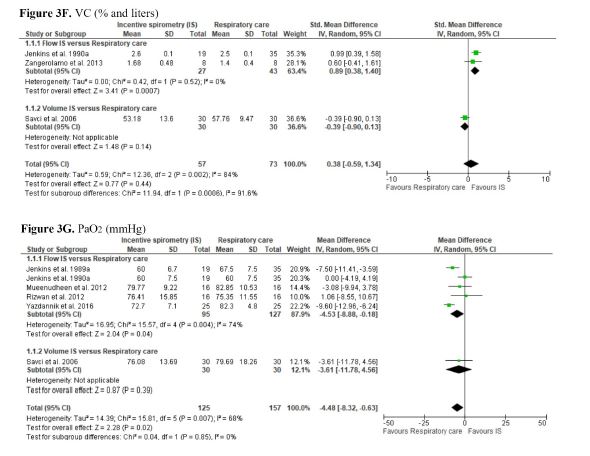
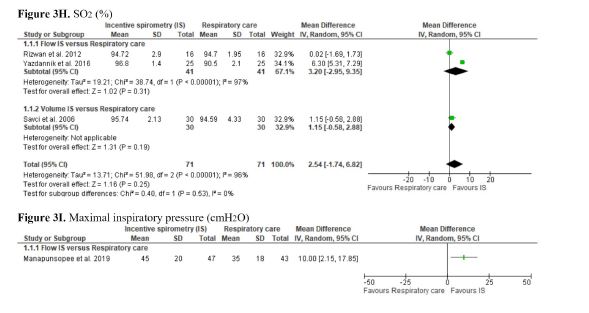
We found low certainty of evidence that there may be a small or no difference on FEV1 between IS and respiratory care (SMD -0.16; 95% CI -0.48 to 0.16) (Supplement 3 - Figure 3D). The evidence is of very low certainty for all the other secondary outcomes of this comparison. For these outcomes, no differences in LOS (MD -1.38; 95% CI -2.96 to 0.21), length of ICU stay (MD -0.78; 95% CI -1.61 to 0.06), PEF (MD -0.60; 95% CI -1.97 to 0.78), FVC (SMD 0.14; 95% CI -0.40 to 0.67), VC (SMD 0.38; 95% CI -0.59 to 1.34), and SO2 (MD 2.54; 95% CI -1.74 to 6.82) were observed, comparing IS and respiratory care (Supplement 3 - Figures 3A, 3B, 3C, 3E, 3F, and 3H). However, in the subgroup analysis of VC, flow IS was superior compared to respiratory care. The Amin et al. (2021)[47] study was not included in the PEF meta-analysis as it did not have sufficient extractable data. The Barkhordari-Sharifabad et al. (2021)[48] study was not included in the SO2 meta-analysis as it was unclear whether it used flow-oriented IS or volume-oriented IS.
The meta-analysis showed that IS leads to lower recovery of PaO2 than respiratory care (MD -4.48; 95% CI -8.32 to -0.63) (very low certainty of evidence). In the subgroup analyses, flow-oriented IS was inferior to recovery PaO2 compared to respiratory care (Supplement 3 - Figure 3G). Two trials evaluated MIP[36,45], however, only one had sufficient extractable data (Supplement 3 - Figure 3I)[45].
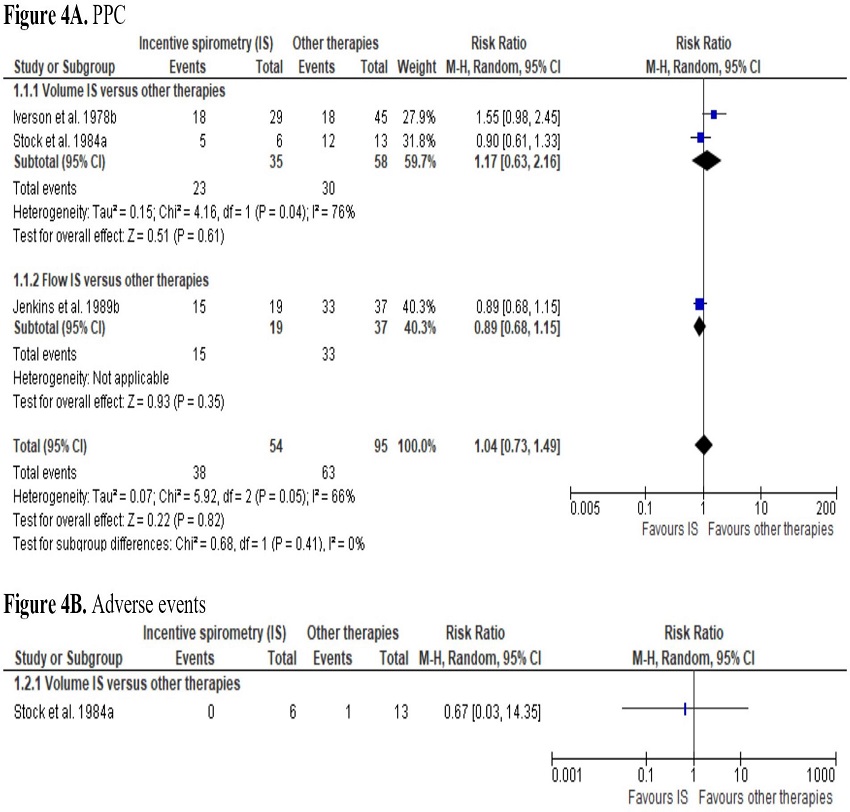
The evidence for the primary outcomes of IS vs. other therapies is of very low certainty. We found no differences on PPC between IS and other therapies (RR 1.04; 95% CI 0.73 to 1.49) (Supplement 3 - Figure 4A). Only one study evaluated adverse events (Supplement 3 - Figure 4B).
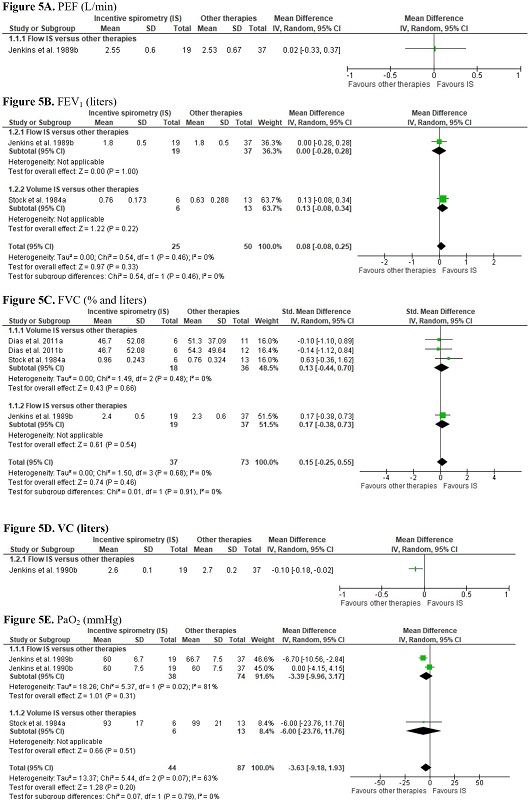
The evidence for the secondary outcomes is also of very low certainty. No difference was observed between IS and other therapies regarding FEV1 (MD 0.08; 95% CI -0.08 to 0.25), FVC (SMD 0.15; 95% CI -0.25 to 0.55), and PaO2 (MD -3.63; 95% CI -9.18 to 1.93) (very low certainty of evidence) (Supplement 3 - Figures 5B, 5C, and 5E). Only one study evaluated PEF[30], and another study evaluated VC (Supplement 3 - Figure 5A, 5D)[31].
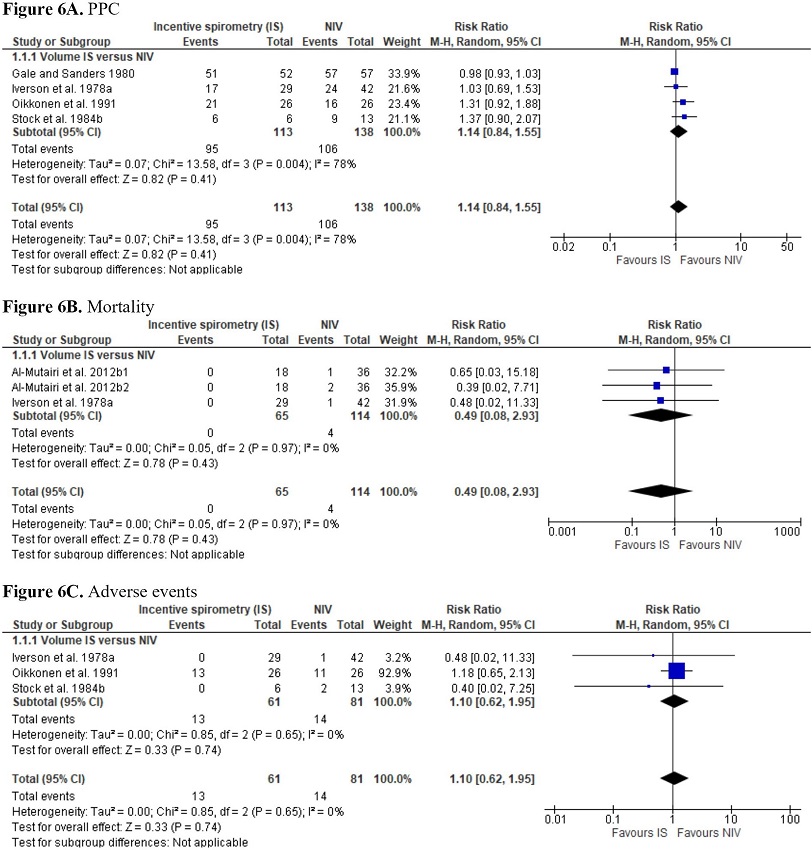
Four trials compared the effects of IS vs. NIV on PPC, and three trials on mortality and adverse events. The evidence for the primary outcomes of IS vs. NIV is of very low certainty. All trials used volume-oriented IS. No differences were found between volume-oriented IS and NIV on PPC (RR 1.14; 95% CI 0.84 to 1.55), mortality (RR 0.49; 95% CI 0.08 to 2.93), and adverse events (RR 1.10; 95% CI 0.62 to 1.95) (Supplement 3 - Figures 6A, 6B, and 6C).
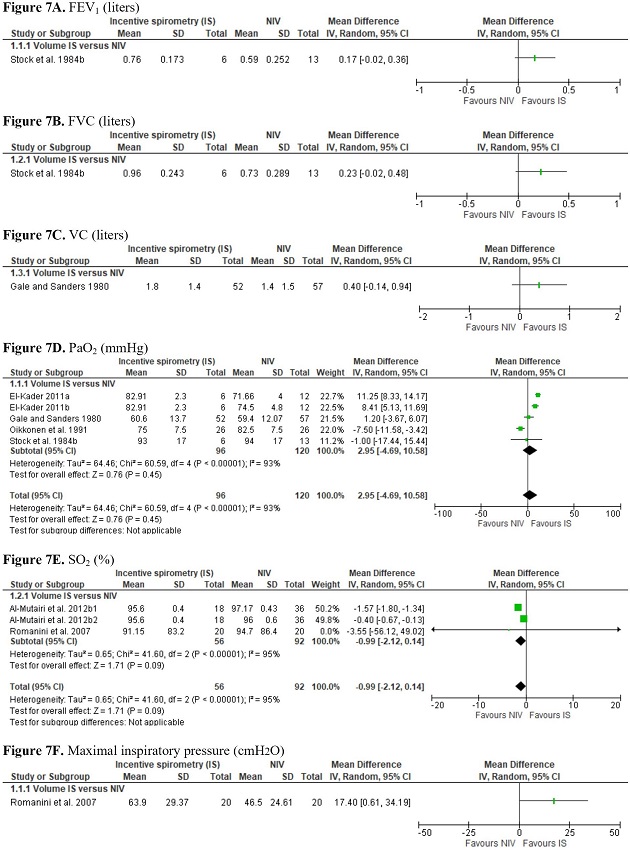
The evidence for secondary outcomes is also of very low certainty. No differences were found between IS and NIV on PaO2 (MD 2.95; 95% CI -4.69 to 10.58) or on SO2 (MD -0.99; 95% CI -2.12 to 0.14) (Supplement 3 - Figures 7D, 7E). Only one trial compared the effects of IS and NIV on FEV1, FVC, VC, and MIP. All trials used volume-oriented IS (Supplement 3 - Figures 7A, 7B, 7C, and 7F). A single study recorded the reintubation rate, with zero reintubation in the IS group and one reintubation in the standard care group[32].
To the best of our knowledge, this is the first systematic review with meta-analysis to investigate the effects of IS exclusively in patients undergoing cardiac surgery, performing sub-analysis to pool the studies according to the type of IS used as respiratory care. The results showed that the use of IS was not superior to respiratory care, other therapies, and NIV on the outcomes evaluated. On the other hand, IS was inferior to respiratory care for recovery PaO2. In the subgroup analysis, flow-oriented IS was inferior to respiratory care on recovery PaO2. However, flow-oriented IS was superior to respiratory care on VC. Overall, the methodological rigor of the clinical trials included in this review was “fair” and the certainty of evidence ranged from “very low” to “low”.
In general, although our meta-analysis showed that IS is not different from respiratory care, other therapies, or NIV, except for PaO2 (in IS vs. respiratory care) for which we cannot make any positive or negative statements about effectiveness after cardiac surgery, the majority of the included studies present severe methodological problems and inadequate sample size. In addition, over the years studies have investigated the effects of IS on PPC, adverse events, and mortality after surgical procedures on the thorax, showing different results, some in agreement with and others contrary to our findings[11,50,51].
Our results are in line with a previous Cochrane systematic review that included seven RCTs with a total of 592 patients to assess the effects of IS for preventing pulmonary complications after CABG[51]. This review found no evidence of a benefit from IS in reducing pulmonary complications and in decreasing the negative effects on pulmonary function in patients undergoing CABG. Of note, besides including only patients that had undergone CABG, this review is outdated and did not perform the certainty of evidence evaluation. The inclusion of a broader and updated body of knowledge and GRADE assessments in our review is of particular importance, as it facilitates decision making of physiotherapists working in the frontline.
A clinical trial investigated the effects of IS after cardiac surgery in 90 patients; 47 patients were treated with flow-oriented IS + deep breathing exercise, and 43 patients received only deep breathing exercise (control group)[45]. Patients who received IS + deep breathing exercise had no reduction in atelectasis, pneumonia, pneumothorax, and pleural effusion. However, the control group had fewer adverse events (dyspnea) (P-value = 0.03)[45]. On the other hand, one thing is certain, although, to date, the clinical efficacy on PPC is not proven, IS is widely used and investigated[52].
A preliminary trial[53] that investigated the effectiveness of IS (flow-oriented device) on respiratory motion in healthy subjects suggested that two weeks of respiratory training using IS is useful for improving respiratory motion and pulmonary function. A clinical trial[54] with 260 surgical patients (non-cardiac patients) showed that IS (flow-oriented and volume-oriented) and diaphragmatic breathing exercise better preserve pulmonary function and diaphragm excursion. If these findings are also demonstrated in patients after cardiac surgery using IS, this method will represent an easily accessible and low-cost device to be used in the treatment of these patients.
A broad range of different types of IS devices and treatment protocols were used in the studies included in this review. However, we were unable to determine which of them is more effective. Although we planned to perform other subgroup analyses, we were also unable to identify whether the type of surgery, the severity of the disease, or details of the intervention, such as frequency, duration, and time the intervention started could influence the effect of intervention. Due to the heterogeneity of the RCTs regarding the combinations of interventions and comparisons, different comparisons had to be made, and we were only able to perform a few comprehensive meta-analyses. Therefore, the precision of effect estimates was jeopardized.
Furthermore, due to several methodological limitations in the included studies and conflicting results, further well-designed trials, with long-term follow-up, and which report the rate of core outcome results, such as PPC, adverse events, mortality, lung function, and LOS, are needed, as well as in the ICU. New RCTs should be standardized to provide more homogeneous and reliable data to properly compare the results. For example, studies should evaluate the same IS device, delivered using standardized protocols, for treating similar types of surgeries.
Of note, some limitations should be underscored. In addition, there is a need for clear and complete reporting of outcome data for the interventions being compared. All trials included in this review had important methodological limitations. Although blinding of participants and personnel may be very difficult from a practical perspective, several other factors such as the lack of blinding of outcome assessors, loss to follow-up, and the absence of intention-to-treat analyses were common methodological limitations in the available studies.
Overall, due to the serious risk of bias and imprecision, the overall certainty of the available evidence is very low, and several questions persist. Thus, it is unclear whether IS used alone or in combination with other therapies is effective when compared to other interventions used alone or in combination.
Moreover, although some studies concluded that IS was safe, the available information on adverse events was insufficient to perform a comprehensive meta-analysis that could provide more accurate results on the safety of IS. The evidence is currently insufficient to support or refute the routine use of IS after cardiac surgeries. The results of the six ongoing RCTs are necessary to provide more precise and reliable information on which to base further trials and protocols, and to guide clinical decision-making processes on the use of IS after cardiac surgeries.
We believe the strengths of this systematic review include transparency, rigid methods, assessment of the quality of evidence for each outcome, and extensive and careful searches, with no restrictions on language or publication date. We searched the gray literature database and ongoing studies and performed a rigorous critical assessment of the current body of evidence. Furthermore, the assessment of certainty of evidence using the GRADE approach is paramount in pointing out limitations in current trials and upon which to base further high quality RCTs. Another strong point of this review was the separate analysis by the type of IS (flowor volume-oriented IS), when possible. This high-quality review underlines that there is an urgent need to conduct high-quality RCTs in this field.
We consider as limitations of this systematic review the inclusion of biased clinical trials, such as those with lack of blinding of outcome assessors, or without adequate randomization; substantial heterogeneity among studies that made them unsuitable for meta-analysis; or studies with small samples that do not allow us to provide accurate estimates of the effects. As another limitation, we were unable to explain the heterogeneity in the meta-analysis of the PaO2 and SO2 outcomes.
This meta-analysis revealed that IS was not superior to standard respiratory care for PPC and clinical outcomes, therefore its use should not be widely recommended until high-quality further studies are performed to ensure this clinical guidance.

Correspondence Address: Elinaldo da Conceição dos Santos, https://orcid.org/0000-0002-4039-2980, Department of Biological and Health Sciences, Universidade Federal do Amapá, Rodovia Josmar Chaves Pinto, S/N, Jardim Marco Zero, Macapá, AP, Brazil, Zip Code: 68902-280, E-mail: drelinaldo@yahoo.com.br

















