ABSTRACT
Introduction: Transcatheter aortic valve replacement (TAVR) has revolutionized the management of patients with aortic valve disease. However, the need for pacemaker implantation remains a frequent complication. The objectives of this study were to estimate the incidence of permanent pacemaker implantation and to determine the associated risk factors.
Methods: This is a retrospective cohort study of adults who underwent TAVR, developed cardiac conduction disease, and required permanent pacemaker implantation during hospitalization. Groups were compared according to post procedure pacemaker implantation or not; and recognized preoperative and echocardiographically identified anatomic factors related to the procedure were evaluated. A predictive model was generated using multiple logistic regression.
Results: A total of 234 patients were included. The pacemaker implantation rate was 14%, and risk factors associated with this procedure were age (odds ratio [OR] 1.10, 95% confidence interval [CI] 1.01 - 1.22), female sex (OR 0.11, 95% CI 0.01 - 0.61), body surface area > 1.51 m2 (OR 9.78, 95% CI 2.13 - 73.6), right bundle branch block (OR 22.5, 95% CI 2.62 - 242), first-degree atrioventricular block (OR 18.8, 95% CI 3.04 - 150), and implantation depth measured via echocardiography (OR 1.76, 95% CI 1.26 - 2.64). The model demonstrated good predictive capability with an area under the receiver operating characteristic curve of 0.934 (P < 0.001, 95% CI 0.878 - 0.988).
Conclusion: A well-performing predictive model was developed with six independent risk factors for the need for pacemaker implantation after TAVR, based on factors related to anatomic echocardiographic measurements associated with classic risk factors.
Keywords: Atrioventricular Block, Transcatheter Aortic Valve Replacement, Artificial Pacemaker, Risk Factors.
ORIGINAL ARTICLE
Echocardiographic Anatomical Risk Factors for Permanent Pacemaker Implantation After Transcatheter Aortic Valve Replacement: A Retrospective Cohort Study
Sociedade Brasileira de Cirurgia Cardiovascular
Received: 01 April 2024
Accepted: 28 September 2024

Transcatheter aortic valve replacement (TAVR) represents a breakthrough in the field of interventional cardiology for the treatment of symptomatic aortic stenosis in high-risk surgical patients or elderly patients[1]. Currently, it also plays a fundamental role in the treatment of patients with severe or intermediate-risk aortic stenosis and will probably become an option for low-risk patients in the near future[2].
Given the current trend of performing TAVR in patients with a better prognosis, it is necessary to balance the risks related to artificial valve implantation with those of surgical aortic valve replacement[3,4]. One of these risks is the need for a permanent pacemaker (PPM), which depends on factors related to the patient and the valve used in TAVR. Balloon-expandable valves traditionally have lower rates of PPM implantation than self-expanding valves. However, the incidence of PPM implantation is still greater than that after surgical aortic valve replacement[5].
Multiple reviews have evaluated the risk factors for the need for PPM implantation, identifying prosthesis-related factors, preand post-procedure electrophysiological factors, and factors related to the experience of the operator. However, few studies present predictive scales for this outcome, as well as its impact on long-term mortality[6,7].
Electrophysiological factors related to the need for PPM implantation are widely known, but they only explain part of the pathophysiology of conduction disturbances after implantation. In the literature, two significant models stand out. Kiani et al.[8] found that a history of syncope, prior branch block, and oversizing were independently associated with the need PPM implantation after TAVR, particularly with balloon-expandable valves. On the other hand, Tsushima et al.[9] identified hypertension, right bundle branch block (RBBB), first-degree atrioventricular block (AVB), and the use of self-expanding valves as independent risk factors for PPM However, both studies did not consider anatomical factors in their risk assessment for pacemaker implantation. In recent years, research has focused mainly on associated anatomical factors, which are of special importance in populations with body surface area (BSA), and therefore anatomical structures, toward the lower limits of normal[10].
In this study, we sought to identify predictors of the need for PPM among patients undergoing TAVR. Furthermore, given its steady incidence and its possible impact on long-term outcomes, we sought a method for clinically stratifying this risk during hospitalization. Therefore, we proposed to develop a model based on anatomical factors that can be easily measured with echocardiography after TAVR.
This is a historical cohort study involving patients from a university hospital. All patients who underwent TAVR for the first time between January 2009 and March 2022 at the Fundación Cardioinfantil - Instituto de Cardiología, Colombia, were included. Patients with previous PPM implantation, any other device implanted for rhythm control, or indications for such a device prior to the procedure were excluded. We determined that enrolling 222 patients in the exposed group with 80% power would allow us to detect a relative risk of three for each of the risk factors at a two-sided statistical significance level of 0.05. This calculation was based on an expected event rate of 10% for the exposed group.
Using the database of the interventional cardiology group, we collected the clinical characteristics of the patients, including patient history, electrocardiograms (ECGs), preoperative images (computerized axial tomography and echocardiography), and data related to the TAVR procedure. The main outcome was the incidence of PPM implantation. Data on several secondary outcomes, such as major vascular complications, intensive care unit stay, and mortality, were also collected according to the definitions of the Second Valve Academic Research Consortium (VARC-2). Data collection was approved by the institutional research and clinical research ethics committees.
We evaluated the images according to their quality, and those with technical problems were excluded. We reviewed the long axis of the aortic valve in the parasternal projections (for measuring implantation depth) and an apical four-chamber projection (for measuring the length of the perimembranous septum). The annular plane was defined as the lowest point at which the aortic valve leaflets were implanted.
Demographic, clinical, and paraclinical characteristics are described with means and standard deviations for continuous variables and counts and proportions for discrete variables.
For the bivariate analysis, we used logistic regression to determine candidate variables for the predictive model, selecting those with a level of statistical significance < 20% (P < 0.200). We included age among the candidate variables given its clinical relevance and its potential confounding role. The predictive model was constructed using multiple logistic regression with a backward selection algorithm and set the statistical significance level to 5% as a criterion for retaining variables according to the likelihood ratio test. We evaluated the goodness of fit of the model with the Hosmer-Lemeshow test and its predictive capacity by estimating the area under the curve (AUC) of the receiver operating characteristic (ROC) curve. Due to missing data from echocardiographic measurements (22.5%) because of random loss of complete exams for certain patients, we evaluated models with and without this predictor, as well as the predictive capacity of other factors.
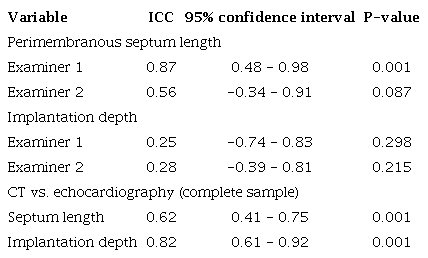
An exploratory analysis was performed in a 5% subsample to assess the agreement between observers and the expert grader using an intraclass correlation coefficient (ICC) for measurements of implantation depth and septal length as assessed by echocardiography. In addition, an ICC was calculated to assess the agreement between computed tomography (CT) angiography measurements of implantation depth and septal length compared with echocardiographic measurements as a novel and reproducible predictor. Complete sample data were used for this purpose.
Finally, we described some of the secondary outcomes of the procedure via exploratory analysis. All the analyses were carried out in the statistical program R.
We identified a total of 309 patients in the database of the interventional cardiology service during the observation period. Seventy patients were excluded after a review of the study eligibility criteria (Figure 1). The most frequent reasons for exclusion were valve-in-valve procedures, the implantation of a previous pacemaker or similar device, and the need for circulatory support during TAVR. The final cohort consisted of 239 participants for the proposed analyses. The main demographic, clinical, imaging, and surgical characteristics are presented in Table 1.
Baseline characteristics of patients according to permanent pacemaker implantation.
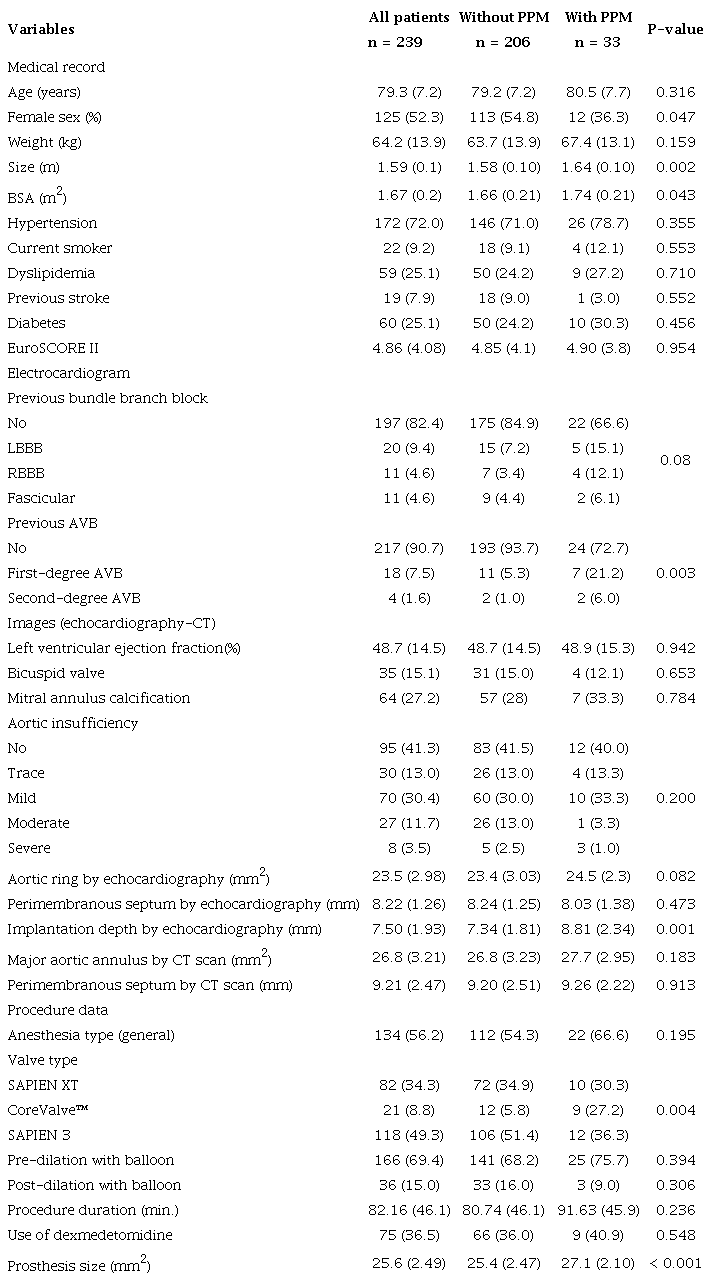
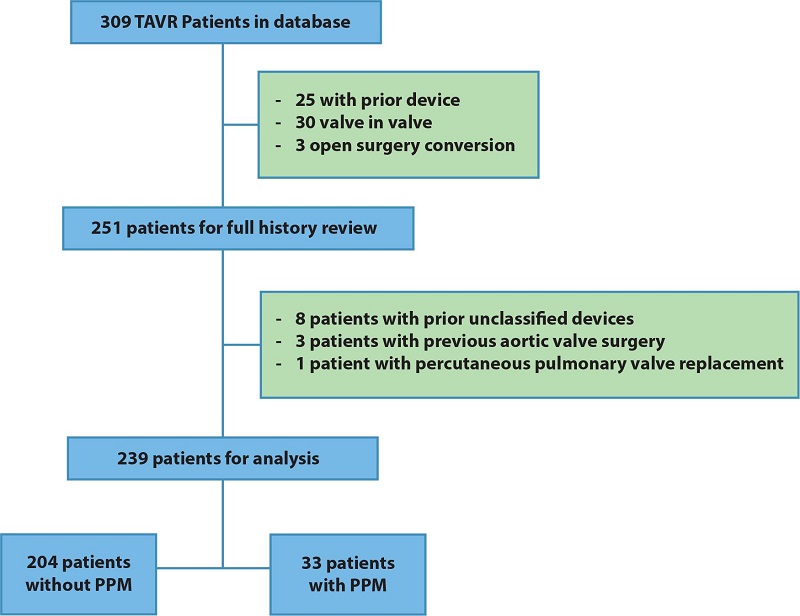
Fig. 1
Patient selection flowchart for the cohort - number of patients and the reasons for exclusion. PPM=permanent pacemaker; TAVR=transcatheter aortic valve replacement.
In 33 of the 239 participants, a PPM was implanted after transcatheter aortic valve implantation (14%, 95% confidence interval [CI] 10 - 19) with no differences in incidence observed during the time the cohort was formed (2009 - 2022) even though several types of valves were used (SAPIEN XT, SAPIEN 3, CoreValve™, and Evolut™), with SAPIEN 3 being the most frequently implanted (51.0%).
In decreasing order of frequency, the indications for PPM implantation were complete AVB (54.5%), alternating trifascicular block (15.1%), first-degree AVB plus left bundle branch block (LBBB) (15.1%), electrophysiological findings indicative of a high risk of blockade (9.1%), and Mobitz II second-degree AVB (6.0%). Patients who required PPM implantation after TAVR were predominantly male (63.7% vs. 45.2%, respectively; P = 0.047) and had a greater BSA than those who did not (1.74 m2 vs. 1.66 m2, respectively;
P = 0.043). The baseline left ventricular ejection fraction, the European System for Cardiac Operative Risk Evaluation (or EuroSCORE) II, and the remaining clinical characteristics as measured prior to the procedure were not significantly different between patients who did and did not undergo PPM implantation.
The reproducibility of the echocardiographic measurements was moderate for the length of the perimembranous septum (ICC 0.87 for evaluator 1 and 0.56 for evaluator 2) but poor for the depth of valve implantation (ICC 0.25 for evaluator 1 and 0.56 for evaluator 2), taking the expert measurements as the gold standard. The ICCs for the echocardiographic and CT measurements for these variables were 0.62 and 0.82, respectively (Supplementary Table 1).
Regarding initial ECG findings, RBBB and first-degree AVB were more common in the PPM group (12.1% vs. 3.4%; P < 0.05; 21.2% vs. 5.3%; P < 0.001, respectively). LBBB and bifascicular block (defined by concurrent RBBB and left anterior or posterior fascicular block) did not significantly differ between the groups. There was a very marginal difference in the incidence of Mobitz I second-degree AVB between the two groups, given its low incidence in the cohort.
Among the variables related to the procedure, self-expanding prosthetic valves were more commonly used in the group that required PPM implantation (27.2% vs. 5.8%, P < 0.005). The size of the prosthesis was also relevant, as more patients in the PPM group required implantation of the largest valve (25.4% vs. 27.1%, P < 0.001). There were no significant differences in other characteristics of the procedure or in the use of dexmedetomidine during anesthesia.
The valve implantation depth was only measured in 181 patients (161 who did not undergo PPM implantation vs. 20 who did); the remaining patients either lacked post-TAVR echocardiography images (n = 40) or had images of poor quality (n = 18). A significant difference was found between the study groups: 8.81 mm for the PPM implantation group vs. 7.26 mm for the nonimplantation group (P = 0.002).
In summary, the candidate variables for the construction of the multivariate model were age, sex, BSA, previous AVB, previous bundle branch block, annulus size, valve type, prosthesis size, presence of aortic regurgitation, and implantation depth (Table 2). The variables that independently and statistically contributed to predicting the need for PPM post-TAVR were subsequently included in the multiple logistic regression model (Table 3).
Candidate predictors according to permanent pacemaker implantation.

Final model for predicting permanent pacemaker implantation after TAVR.
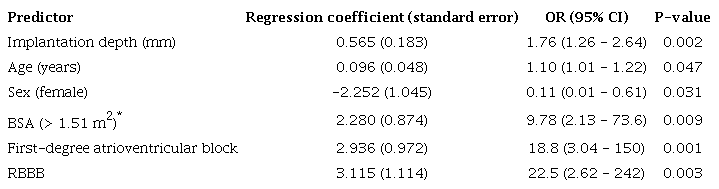
* This cut-off point was identified as having the greatest discriminatory capacity against the outcome using ROC analysis.
In the predictive model, the small sample size posed a significant limitation in evaluating factors such as first-degree AVB (95% CI 3.04 - 150), and RBBB (95% CI 2.62 - 242), which substantially reduced the precision of these estimates, resulting in wide CIs.
To improve the interpretability of the model, the BSA was dichotomized using 1.51 m2 as the cutoff point, as this value showed the greatest discriminability for the outcome according to ROC curve analysis. In general, the model had a high goodness-of-fit (P = 0.941 for the Hosmer-Lemeshow test) as well as high precision, with an AUC of 0.93 (95% CI 0.88 - 0.99) (Figure 2).
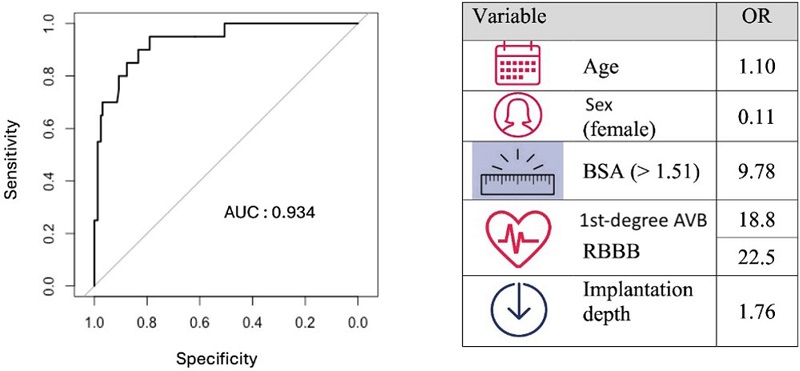
Fig. 2
Receiver operating characteristic curve of the predictive model and table showing the independent variables and their odds ratio (OR). The area under the curve (AUC) was 0.934 (95% confidence interval: 0.878 - 0.988). AVB=atrioventricular block; BSA=body surface area; RBBB= right bundle branch block.
Among the important secondary outcomes, 44 patients had LBBB after the procedure, and the incidence of complete AVB was 7.5%. The hospital stay was greater in the PPM group (5.79 vs. 9.91 days; P < 0.001). There was no difference in mortality or in the other outcomes of the procedure according to VARC-2 definitions (Supplementary Table 2).
Conduction disorders are common after TAVR, the most clinically relevant include high-grade AVB and new-onset LBBB. These conduction disturbances may improve in the first hours after TAVR in half of patients due to the resolution of inflammation secondary to the procedure[5,6,11]. However, 90% of patients who present with permanent or intermittent alterations 24 hours after the procedure or during hospitalization will require the implantation of a PPM in the first three days after the procedure[12]. Nai Fovino et al.[13] showed that PPM implantation was required in 44.6% of TAVR patients after 30 days and in 46.7% after one year; in all cases, PPM implantation has been related to higher mortality and more frequent hospitalizations for heart failure[11].
The main indication for PPM is complete AVB, which we identified in 50% of our cohort; the other indications were new LBBB associated with first-degree block or intermittent AVB. The incidence of PPM implantation during hospitalization was 14%, close to that previously reported for patients who received balloon-expandable valves[7,14]. This incidence is a reflection of the greater number of SAPIEN 3 and XT valves used in our institution compared to self-expanding valves, which have been associated with a greater incidence of AVB and a greater need for PPM implantation (37.6% vs. 17.3%; P < 0.001) due to the continuous effect of radial compression on the left ventricular outflow tract[15]. In our study, a greater incidence of PPM implantation was demonstrated in patients with self-expanding valves. According to bivariate analysis, CoreValve™ was more likely to be implanted in patients later requiring PPM (27.2 vs. 5.8, P = 0.004); however, this relationship was not maintained when confounding factors were controlled for in the multivariate analysis.
The implantation of the latest generation of balloon-expandable valves (SAPIEN 3) has been associated with a greater rate of PPM implantation than previous versions[16], with rates as high as 11% to 17%[4,17]. In our work, this relationship could not be demonstrated despite the large proportion of patients implanted with the latest generation valves (SAPIEN 3 vs. XT). The main risk factors for the implantation of a PPM were similar between the different types of valves, reflecting the impact of other factors on the incidence of PPM implantation, such as electrophysiological and anatomical factors related to the implant.
Our study revealed that age, male sex, BSA > 1.51 m2, a preexisting bundle branch block or first-degree block, and a low prosthesis implantation depth were independent predictors of the need for PPM after TAVR implantation, regardless of the type of valve implanted, the length of the perimembranous septum, the size of the annulus measured by echocardiography, and oversizing of the implant. These findings, which were evaluated as possible predictors prior to the analysis, are supported by previous studies and biological plausibility due to the anatomical and physiological relationships of the conduction system[18].
The relevance of the BSA within the model may reflect its relationship with the use of larger valves, themselves associated with a greater risk of deep implantation. This finding has not been previously described as an associated factor in systematic reviews[18]. In contrast, sex has previously been associated with PPM implantation. According to a meta-analysis carried out in 2021 involving 46 studies published in PubMed (n = 70,313), the cumulative rate of PPM implantation in women was lower than that in men (14.9%, 95% CI 12-6 - 17.6 vs. 16.6%, 95% CI 14.2 - 19.4), and so the risk of PPM implantation after TAVR was lower in women (odds ratio [OR] 0.90, 95% CI 0.84 - 0.96; P = 0.0022)[19]. We found similar results in our study; women had a lower probability of needing PPM implantation (OR 0.47, 95% CI 0.21 - 0.99).
Several studies decided to exclude anatomical variables from their prediction models because very specific imaging protocols and trained personnel are required to interpret the images correctly. Therefore, despite the high diagnostic precision reported, it can be complicated to integrate these variables into predictive scales without compromising their generalizability and applicability[8,9]. That said, it is precisely these reasons that allowed us to generate our hypotheses from the measurement of anatomical variables in less complex images, closer to the patient's bed and with little or no potential damage, as is the case with echocardiography.
Studies that have used anatomical predictors are based on high-resolution CT[10,13]. However, in addition to its high complexity and expensiveness, CT can potentially injure the patient via the administration of contrast medium and radiation exposure, which limits its use immediately after the procedure. In comparison, echocardiography can be conducted immediately after the procedure and has high reproducibility for expert operators (Figure 3).
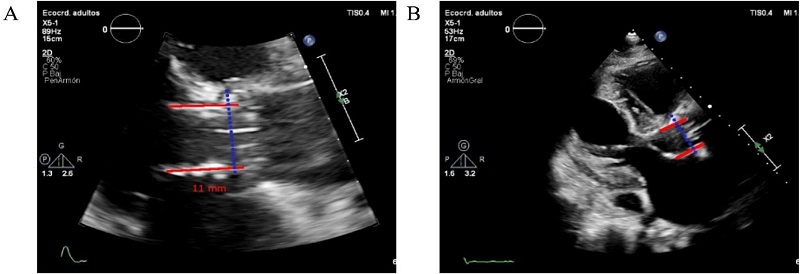
Fig. 3
2D transthoracic echocardiography images in the aortic longitudinal plane showing the measurement of the valve implantation depth. A) Patient with a low valve implant ratio (30:70). B) Approach with measurements for a patient with a very low implant ratio (10:90) and the need for postoperative permanent pacemaker implantation (11 mm from the annulus line [dashed line]).
Some previous studies have used echocardiographic measures to evaluate LBBB as a primary outcome[20]. However, at the time of this study, we were unable to identify other models that use the implantation depth measured with echocardiography as a predictor of PPM implantation. According to our model, patients with greater implantation depths had a greater risk of needing a PPM during hospitalization (OR 1.468, 95% CI 1.15 - 1.90) for each mm of greater valve implantation depth.
The goal of our practice for valve implantation is to achieve a 90/10 ratio with respect to the annular plane (90% of the valve above the annulus, 10% below) as a strategy to reduce the mechanical impact on the conduction system according to the recommendations from the literature[21,22]. Generally, an attempt is made not to exceed a 70/30 implantation ratio; however, the main objective for a successful outcome is the valve function and not the final height, which is why it was decided to integrate the role of implantation depth in our model in the analysis. One of the main limitations of anatomical measurements of implantation depth is that a major part of these measurements is performed after the procedure, which limits the ability to determine the pre-procedure probability and the risk of PPM implantation a priori. However, given the magnitude of its effect in our model (accounting for approximately 20% of the prediction effect), the post-procedure measurement of the implantation depth can help to identify patients in need of prolonged rhythm observation and monitoring or additional electrophysiological examinations.
Kiani et al.[8] previously reported that a history of syncope, a previous bundle branch block, and oversizing were independently associated with the implantation of a PPM after TAVR using mainly balloon-expandable valves, which contrasts with studies using other types of valves, in which implantation height alone was independently associated with the implantation of a PPM[23,24]. This suggests that there are multiple patient-specific factors, such as preexisting conduction disease, particular anatomy, and procedural factors, that also predict the need for a PPM.
Tsushima et al.[9] also described a risk scale using logistic regression in a retrospective cohort of 888 patients in which hypertension, RBBB, first-degree AVB, and the use of self-expanding valves were found to be independent risk factors associated with the implantation of a PPM after TAVR.
Among the most studied electrophysiological parameters prior to the procedure, first-degree AVB and bundle branch blocks have been most associated with the need for a PPM, with RBBB having shown the strongest relationship in the literature[25].
Anatomically, the left branch of His bundle is sensitive to mechanical damage from valve deployment due to the proximity of its perforating portion to the space between the noncoronary and right coronary leaflets of the aortic valve[9,11]. This relationship is in accordance with the findings obtained from the regression model, which revealed both first-degree AVB and RBBB as independent factors in our cohort.
We believe our study offers a new approach by incorporating anatomical predictors of PPM, potentially improving the accuracy of predictions compared to prior models that focus primarily on electrophysiological factors. By utilizing easily obtainable preoperative and postoperative variables, our model can help assess the risk of PPM implantation during hospitalization.
Our study has several limitations. The observational design restricts our ability to establish causality, though the predictors identified are consistent with previous research and are physiologically plausible. Data limitations prevented the assessment of certain risk factors, such as the presence of calcium distal to the implantation site. The single-center nature of the study may limit the generalizability of our findings to broader populations undergoing TAVR. Additionally, the lack of external validation weakens the robustness of the model. Incomplete echocardiographic data affected the precision of our model, and issues with the reproducibility of these measurements, particularly implantation depth, raise concerns about their reliability in clinical practice. Furthermore, potential confounding factors like comorbidities and medication use were not fully explored, which further weakens the model's robustness. These limitations should be considered when interpreting the results and highlight the need for further prospective studies to validate and refine our findings.
This study is notable for its inclusion of a significant number of local patients, capturing clinical and demographic characteristics not well represented in previous studies of predictors of PPM implantation risk. It represents a first step in risk prediction specific to our population. A novel aspect is that it overcomes the shortcomings of measuring valve implantation depth, a key predictor. Echocardiography proved to be a reliable alternative to CT for measurements. Several strategies were employed to mitigate information bias and design confounding, including a broad design with minimal exclusion criteria and sensitivity analyses to account for missing data and subgroup differences. Logistic regression was selected for robust analysis, accounting for interactions and potential confounding variables, with checks to ensure adherence to underlying assumptions.
The need for PPM implantation after TAVR is a frequent and potentially deleterious complication. Stratifying the specific risk for each patient is necessary, especially with the expansion of recommendations for TAVR to patients with lower risk and greater life expectancy. Our suggested model, which uses easily measurable preoperative and postoperative variables, can estimate the risk of the need for PPM implantation during hospitalization and differentiate between those at low risk who can be safely discharged and those at higher risk who require closer monitoring or additional electrophysiology study. The model requires external validation in larger, multicenter cohorts to improve its generalizability, clinical applicability, and effectiveness across diverse populations and various clinical settings.
We thank Drs. Yimy Santana and Laura Gutiérrez for their contributions to the data collection and Laura Catalina Peña for her contribution to drafting manuscript.
Correspondence Address:Hugo Andrés Mantilla, https://orcid.org/0000-0002-8998-1348, Anesthesiology Department, Fundación Cardioinfantil - Instituto de Cardiología, Cl. 163a #13B-60, Bogotá, Colombia, Zip Code: 111111, E-mail: hamgzero@gmail.com

Baseline characteristics of patients according to permanent pacemaker implantation.


Fig. 1
Patient selection flowchart for the cohort - number of patients and the reasons for exclusion. PPM=permanent pacemaker; TAVR=transcatheter aortic valve replacement.
Candidate predictors according to permanent pacemaker implantation.

Final model for predicting permanent pacemaker implantation after TAVR.

* This cut-off point was identified as having the greatest discriminatory capacity against the outcome using ROC analysis.

Fig. 2
Receiver operating characteristic curve of the predictive model and table showing the independent variables and their odds ratio (OR). The area under the curve (AUC) was 0.934 (95% confidence interval: 0.878 - 0.988). AVB=atrioventricular block; BSA=body surface area; RBBB= right bundle branch block.

Fig. 3
2D transthoracic echocardiography images in the aortic longitudinal plane showing the measurement of the valve implantation depth. A) Patient with a low valve implant ratio (30:70). B) Approach with measurements for a patient with a very low implant ratio (10:90) and the need for postoperative permanent pacemaker implantation (11 mm from the annulus line [dashed line]).
Exploratory assessment of the reproducibility of imaging-based measurements between observers and experts.
Procedure outcomes.

