REVIEW
Received: 21 September 2021
Accepted: 10 May 2022
DOI: https://doi.org/10.1590/0103-1104202213422
ABSTRACT: Although the National Health Service (NHS) and the Unified Health System (SUS) are systems with similar universal principles, they can show different political measure patterns in the pharmaceutical field. This paper aimed to provide a comparative analysis of pharmaceutical policies highlighting strategies to guarantee access and sustainability to High-Price Medicines (HPMs) in Brazil and England. We performed an integrative literature review in electronic databases, supplemented by grey literature searched on governmental platforms (laws, decrees, ordinances, and resolutions). A total of Forty-seven articles and seven policies were selected and categorized for analysis. The results showed that both countries apply distinct policies to ensure access to HPMs, among them, policies to define price and reimbursement and actions to regulate the use inside the system. Also, these countries apply distinct policies to their sustainability as local partnerships for product development in Brazil and confidential managed agreements with multinational industries in the England. In conclusion, despite similarities in principles, these countries have been proposing and applying distinct pharmaceutical policies to maintain access and ensure the sustainability of their health systems.
KEYWORDS: Health policy, Technology, High-Cost, Public health.
RESUMO: Embora o National Health Service (NHS) e o Sistema Único de Saúde (SUS) sejam sistemas com princípios universais semelhantes, diferentes políticas no campo farmacêutico podem coexistir. O objetivo deste artigo foi fornecer uma análise comparativa destacando estratégias para garantir o acesso e a sustentabilidade a Medicamentos de Alto Preço (MAP) no Brasil e na Inglaterra. Foi realizada uma revisão integrativa da literatura em bases de dados eletrônicas, complementada por literatura cinzenta pesquisada em plataformas governamentais (leis, decretos, portarias e resoluções). Um total de 47 artigos e 7 políticas foram selecionados e categorizadas para análise. Os resultados demostraram que ambos os países aplicam distintas políticas para garantir o acesso aos MAP, entre elas, políticas para definição de preço e reembolso e ações para regular a utilização destes medicamentos dentro do sistema. Além disso, os países aplicam políticas distintas à sua própria sustentabilidade como as parcerias para o desenvolvimento produtivo local no Brasil e acordos confidenciais com indústrias multinacionais na Inglaterra. Em conclusão, apesar das semelhanças nos princípios, estes países têm proposto e aplicado políticas farmacêuticas distintas para manter o acesso e a sustentabilidade de seus sistemas de saúde.
PALAVRAS-CHAVE: Política de saúde, Tecnologia de alto custo, Saúde pública.
Introduction
Improving the provision of medicines is a critical component of any health service policy and one of the main priorities of governments that require comprehensive national strategies1. The Organization for Economic Co-operation and Development (OECD) showed that medicines account for 20 to 60% of health spending in Low Middle-Income Countries (LMIC), compared with 18% in High-Income Countries (HIC)2. The increase in health care spending can be associated with several determinants, such as population growth, population aging, service prices, disease prevalence or incidence, and service utilization3.
The most comprehensive picture of recent spending on medicines in England’s National Health Service (NHS) shows that costs rose from around £13.0 billion in 2010/11 to £17.4 billion in 2016/17 - a mean yearly growth of around 5 percent and a total of 32.1% in this period. Consequently, spending on hospital medicines nearly doubled, from around £4.2 billion to £8.3 billion - representing a mean yearly growth of 12.1%4. The spending amount has also increased in Brazil. From 2010 to 2016, the spending allocated to High-Price Medicines (HPMs) in the Brazilian Unified Health System (SUS) hiked from R$ 4.9 to 6.6 billion, with a mean yearly increase of around 5 percent and a total of 34.7% in this period, excluding the spending on medicines for oncological diseases5.
‘High-cost’, ‘high-priced’, or ‘premium price’ medicines definitions have not yet been defined internationally and can vary in the same country according to who defines them and how they are defined6. In most cases, HPMs are mostly new specialized medicines for complex conditions such as cancers and inflammatory diseases7. These medicines include biological agents, which selectively target specific molecular sites and are, therefore, effective with fewer side effects8. Sometimes, they are the only treatment options for diseases with a profound social impact or a high risk of mortality (e.g., Human Immunodeficiency Virus (HIV)/Acquired Immunodeficiency Syndrome (AIDS) or those used to treat conditions affecting only a minute portion of the population, such as for rare diseases9. The high price itself might not be the decisive criterion. Other determining factors may include the use of or demand for the product6. Nevertheless, the more contemporary definition was illustrated in a 2015 report by the World Health Organization (WHO) Regional Office for Europe, which defines medicine as high-priced if the therapy exceeds €10,000 per patient per year to be reimbursed by a public payer10.
Due to lack of a universal definition, all these elements - cost, exclusivity, novelty, and type of disease - converge to form the characteristics of these types of medicines leading to severe challenges to HPMs access and the definition of health policies9. Different patterns to access them between countries are interrelated, such as a health system level, regional/hospital, and patient-level8. While comparative studies about pharmaceutical policies have been conducted between European countries, the literature lacks analyses between low-middle income countries and high-income countries, such as Brazil and England, respectively. Although NHS and SUS are health systems with similar universal principles, analyzing their differences and similarities is essential for understanding the process of shaping policies in different contexts to help address the challenges and maintain sustainable systems. A similarity is that both systems face funding challenges. However, although the NHS has not had structural changes in its fiscal financing, incorporating precepts based on competition, originally from the market field, resulted in changes in the outpatient and hospital levels11. In the Brazilian case, SUS has faced systematic changes in legal frameworks, with reduced funding entailing lower provision of health services12. This backdrop supported the need for comparative studies assessing their pharmaceutical sectors and policies.
Thus, this study aims to provide a comparative analysis of pharmaceutical policies in the Brazilian and the English health systems, highlighting strategies to guarantee sustainability and access to HPMs.
Methodology
An integrative review approach was adopted13 and considered two types of literature: scientific reports (articles published in peer-reviewed journals) and grey literature, prioritizing laws, rules, or instructions by governmental authorities in Brazil and England from January 1, 2009, to December 31, 2019. The eligible inclusion and exclusion criteria adopted to select the studies and policies are described in table 1.
Selection and sorting of inclusion and exclusion criteria

Information sources and search strategies
The databases included Embase, PubMed, Scopus, Lilacs, Web of Science, and SciELO, selecting keywords in English, Portuguese or Spanish by the ‘Boolean Operator’ rules and terms ‘AND’ and ‘OR’. The keywords were combined and adapted to search individual databases. Also, web pages of relevant governmental platforms were searched to support the analysis (table 2). Databases and governmental platforms were selected based on regulatory themes to encompass different subjects in the health and policy areas. At the same time, due to the comparative analysis, national and international bases most used in both countries were included in the search strategy.
Search strategy and syntax by database

Study selection, data extraction, and analysis
A framework was adopted to categorize and define the studies and governmental reports selected. This conceptual framework guided the uptake of crucial measures implemented in these countries and effects on at least one relevant outcome classified into three categories: pre-launch, peri-launch, and post-launch activities. This framework considers the ‘lifecycle’ of a pharmaceutical product and the different regulatory levels and policy interventions over its course, from research and development to disinvestment10. The results from articles were summarized using content analysis in narrative synthesis examining the current evidence base across Brazil and England, supplemented by an interpretative analysis of policies between these two countries per categories defined in the framework (table 3). Data were extracted by a bibliometric framework. The extracted information was the author’s name and year of the publication, design, aims/objectives, methods, results, and conclusion findings. Two researchers extracted and compared these data and discussed any discrepancies with other researchers as necessary.
Categories of analyses based on WHO guidelines

Results
Search results and studies included
The search across all databases identified 3,288 papers. The selection process is shown in figure 1, totaling 47 articles. The documentary policy analysis also included seven normative instruments (laws, decrees, ordinances, and resolutions). As presented in figure 1, 54 documents were considered for review. The articles or normative documents were categorized into: Pre-launch: 4 (four); Peri-launch: 25 (twenty-five); Post-launch: 25 (twenty-five) per the WHO categories10 (table 4).
Overview of current pharmaceutical policies applied in Brazil and England

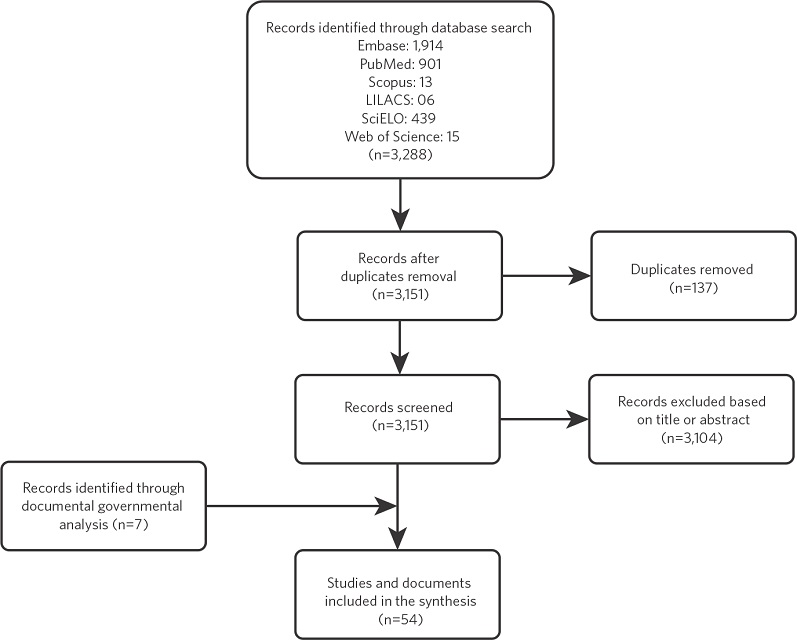
Figure 1
Flowchart of study and policy selection stages
Source: Elaborated by the authors.
Pre-launch category policies
HORIZON SCANNING ACTIVITIES
England has more than a decade of experience in Horizon Scanning (HS) through the English National Horizon Scanning Centre (NHSC). NHSC aims to supply timely information to key policy and decision-makers and research funders to the NHS about emerging health technologies (medicines and devices) that may significantly impact patients or the provision of health services in the future. A retrospective review used the NHSC information system and the list of National Institute for Health and Clinical Excellence (NICE) technology appraisals, estimating the false positive rate for NHSC identification in 10 years. The study showed a positive predictive value in terms of sensitivity over the past decade, meaning that almost 40% of topics reported to NICE were later selected for technology appraisal, and 60% were not. This data indicated that the filtration criteria used could be tightened for increased efficiency in the appraisal of NICE England15,16.
Brazil implemented HS in 2008. Currently, the legal attribution of HS activities at the federal level is incumbent on the National Committee for Health Technology Incorporation (CONITEC), and the methodological approach adopted is described by the EuroScan International Network17. Gomes et al.18, examined the significant repercussions generated by HS in Brazil, showing the developed outputs (internal/external) objectives range from helping build defense arguments in cases of judicialization. One of the internal reports on HPMs (metreleptin) for a rare disease was shared with other countries such as Argentina. CONITEC’s HS system has been structured, and its role as a tool to inform health managers has been relevant.
Peri-launch policies category
REGULATORY PRICE SETTING
In England, medicines’ prices are regulated by regulatory approaches vs. market force. The set of medicines’ prices is defined freely. However, a voluntary scheme - Voluntary Scheme for Branded Medicines Pricing and Access - occurs in cooperation with the Secretary of State for Health, ABPI (Association of the British Pharmaceutical Industry), and NHS England. Most large companies participate, and mainly two mechanisms are helpful to control spending: rate-of-return regulation for companies and an overall cap for NHS spending on branded medicines (2% per year in any of the next five years). The Voluntary Scheme commits the government to maintain the NICE cost-effectiveness threshold at between £20,000 and £30,000 per Quality-Adjusted Life Years (QALY) until 2025. If profits exceed a percentage agreed on after negotiations, the companies must reduce prices, delay price increases, or repay the excess to the Department of Health19.
In Brazil, an inter-ministerial federal entity is responsible for direct control over medicines prices in the market. The Medicine Market Regulation Chamber is responsible for regulating three market segments: factory price, maximum consumer price, and public sector price. The policy adopted is External Referencing Pricing (ERP) following the low price defined in countries such as Australia, Canada, Spain, the United States, France, Greece, Italy, New Zealand, and Portugal20. Furthermore, the government approves annually a metric to increase the price of medicines, including inflation and the competition level for each product, defining the price of medicines in each category21.
Recently, a study conducted by Vogler et al. across European Union Member States showed that some medicines are launched at different points in time, with delays of up to 3-5 years, between countries. Launch delays occur particularly in LMICs or small markets. The study finds that pharmaceutical companies have been applying this strategy in light of the widespread use of the EPR policy. If policymakers aim to apply the pricing policy of ERP for cost-containment purposes, as in Brazil, they are recommended to undertake continuous price revisions over the years22.
REIMBURSEMENT - AGENCIES AND THEIR WORK PROCESSES
The NICE was established in 1999 as an independent body applying different methodological approaches depending on the type of medicine evaluated (e.g., the program to appraise highly specialized technologies)23. Overall, a study conducted by Cowell et al.24, shows that it is difficult to determine whether the differences in appraisal programs/methodologies are likely to impact the efficiency of NHS spending significantly. However, they could lead to inefficient resource allocation. A study by Allen et al.25, comparing England’s national Health Technology Assessment (HTA) recommendations with other European HTA bodies shows how several factors can influence the HTA decision-making process and result in conflicting recommendations. The bodies share some similarities, focusing on clinical efficacy and Cost-Effectiveness (CE). The differences in recommendations could be due to an organizational approach to risk perception and the selected comparator. Harmful recommendations are linked to uncertainties surrounding factors such as CE, choice of comparator, clinical benefit, safety, trial design, and timing of submission. Chabot & Rocchi26 reviewed and compared England’s HTA oncological recommendation records and showed an extensive variation in the rate of positive recommendations (around 48 %). Data on survival was not mandatory for a favorable recommendation, and progression-free survival was found acceptable in some cases for a recommendation. Given that most medicines provided acceptable clinical benefits, CE was left as the most common criterion for a negative recommendation.
In Brazil, the principles and related operational mechanisms in evaluating new innovative health technologies fall within CONITEC. The country has shown a Brazilian HTA system weakness, with some factors such as insufficient resources, the impact of court rulings, heavy dependence on foreign technologies, and incipient regional HTA processes and planning that make the backdrop complex and fragmented with different levels of maturity27. A study performed by the Ministry of Health (MoH) showed the spending per capita on HPMs was substantially higher compared with the overall per-capita spending on health care and associated with different access between region, age, and sex in Brazil, showing a substantial unbalanced resource distribution14. Therefore, Kuchenbecker and Polanczyk28 propose measures related to governance to foster Brazilian HTA’s institutionalization. It is necessary to promote broader policy-making approaches to assess the governance complexity, increasing the institutionalization in Brazil to contribute to HTA to provide better decision-making.
PHARMACOECONOMIC AND THRESHOLDS SETTING
Health Economic Evaluation (HEE) has a vital role in the HTA recommendations by NICE29. In a comparison performed by Schwarzer et al.30, evidence suggests the existence of an explicitly spending threshold known in England - ranging from £20,000 to £30,000 per QALY. The appraisal of end-of-life medicines entails a higher Incremental Cost-Effectiveness Ratio (ICER) of up to £50,000 per QALY. However, for rare diseases, recently, the ICER of £100,000-300,000/QALY can be accepted for these medicines23. Again, due to agreements with industries for most HPMs, the patient access schemes have disclosed price reductions, allowing the NHS to deny a few technologies based on cost-effectiveness in recent years30.
Brazil has not shown tradition in the production and use of HEE30. There are implicit thresholds identified representing gross domestic product per capita lower than 1 and 3 × (US$11,000-25,000). The science, technology, and innovation policy and the pharmaceutical sector economic market regulation - Law nº 10.742 deserve fostering applied research to encourage the application of methods that employ economic analyses. Although some advances were observed in the Brazilian HTA system, there is still room for improvement concerning the prioritization criteria and a more explicit and transparent process for how the HEE addresses the uncertainty parameters of technologies31.
VALUE-BASED PRICING
The introduction of Value-Based Pricing (VBP) in England was planned for the beginning of 2014, defined as ‘the price’ that ensures that the expected health benefits (of new technology) exceed the health predicted to be displaced elsewhere in the NHS due to its additional cost. However, implementing the VBP has been challenging, especially in therapeutic areas with no alternative treatment for a life-threatening and debilitating disease, such as oncology or rare diseases32. Managed-entry agreements are increasingly used for these high-priced medicines. The VBP is used in England to influence pricing decisions besides England assessing similar types of evidence from other European countries (although this is done only indirectly) as it will impact the ICER33.
In LMICs, such as Brazil, the value evaluation trend is primarily linked to the federal sphere. The body is responsible for defining the value of new medicines and clinical and economic evaluation to set limits on CE. Approaches designed for regulatory agencies include reimbursement based on economic evaluations, international reference pricing, significant volume discounts, rebates, or tax incentives34. A new way to define the value of new technologies has been discussed, such as implementing risk-sharing to increase responsibilities to the pharmaceutical industry. A new model for health care professionals and organizations in Brazil, the pay-for-performance (P4P) has shown promising results, shown by the private sector, where doctors receive extra financial incentives for each additional patient with controlled chronic disease. Brazil is headed to P4P by promoting prevention, updating guidelines, and engaging professionals, industry, and health institutions. That will be the key to genuinely improving viral liver disease-induced morbimortality in Brazil35. However, a study by Mathes et al.36, describes that it is uncertain whether payment for performance (P4P) - in England, compared to capitation-based payments without P4P for hospitals - in Brazil impacts patient outcomes, quality of care, equity, or resource sustainability. The effects on patient outcomes of P4P in hospitals were negligible, regardless of design factors and context/setting. It seems that only minor short-term but unsustainable effects can be achieved with additional payments.
MULTIPLE CRITERIA DECISION ANALYSIS
NICE is already using some approaches derived from Multiple Criteria Decision Analysis (MCDA). The degree of unmet clinical need is a formal criterion in healthcare decision-making for rare diseases. The severity and unmet need of the disease (i.e., the burden of illness), the clinical novelty and convenience to patients (i.e., innovative nature of technology), or the broader benefits to society (i.e., non-health objectives to the NHS) have often been perceived as essential considerations of value to decision-makers and have contributed to the debate on efficient resource allocation transparency in England37.
Brazil has not explicitly adopted an MCDA model. Consequently, no related studies have been found. However, a case conducted by Campolina et al.38, suggested applying the methodology in the CONITEC to clarify which criteria are relevant in decision-making, the importance attributed to each criterion, and how to use this information in a structure to evaluate the available alternatives. In doing so, even if the conclusion of a recommendation does not change, the method can increase the transparency and legitimacy of decisions in Brazil.
MANAGED ENTRY AGREEMENTS - MEAS
For several HPMs, arrangements designed as Managed Entry Agreements (MEAs) or Patient Access Schemes (PAS) have been conducted in England. The number of MEAs could be underestimated (around 210 PAS) because many contracts, especially the financial-based ones, are not public39. Ferrario et al.40, highlighted that MEA applications are increasing and have been heterogeneous across some countries, including England, which uses mainly discounts and free doses to influence prices. The health system-specific features relating to governance (the scope of the MEA policy and the mandate of the agency assessing medicines) explain some identified variations between European countries. In an analysis performed by Pauwels et al.41, the results showed that the application of MEA across European countries differs between the same indications. MEA is a common policy tool employed by public payers in European countries to ensure early access to high-price oncology medicines. Discounts and free stocks prevail in England. Nevertheless, according to Jarosławski & Toumi42, these schemes are driven by NICE and address uncertainty around HPMs. The study concluded that a more transparent process might be necessary to protect against a perverse impact of PAS on international reference pricing that uses list prices rather than the actual cost of purchasing medicines that the NHS incurs.
In 2019, Brazil discussed a hypothetical case of a high-cost assessment for a rare disease to identify critical considerations required to implement MEAs in Brazil. The primary considerations for enabling the use of MEAs were identified by the groups and classified into four dimensions: (i) economic; (ii) epidemiological; (iii) ethical; (iv) legal. There is a need to create a new regulation for the agreements in Brazil. However, the incorporation of nusinersen was implemented in the health system. The nusinersen is the most expensive medicine ever incorporated into the SUS. The incorporation has been described as a milestone in decision-making on new technologies, enabled through a risk-sharing agreement. However, due to two recommendations with divergent decisions (2018/2019), without any addition of new evidence or price reduction, the incorporation faced problems of transparency, accountability, and risks in the HTA institutionalization process in Brazil43.
Post-launch policies category
ORGANIZATIONAL POLICIES - GUIDELINES AND CLINICAL FORMULARIES
In England, the secondary specialist care services designed by Clinical Commission Groups (CCG) are the critical setting for the use of HPMs. Among the most important policy initiatives implemented support actions linked to the government’s plans was introducing Commissioning for Quality and Innovation (CQUIN), linked to rationalizing HPMs to define priorities per NICE guidelines and improving value for money procurement, reducing the unwarranted variation of HPMs spends44.
In contrast, Brazil developed the National Pharmaceutical Policy, implementing a solid expansion and regulation of access to HPMs mostly by actions for financing and guaranteeing access through Authorization to High Complexity Procedures Cost (AHCP)20,45. However, some problems are cognized, such as the determinants of the use of health services and the bureaucracy that limits access, which increases the demand, exceeding the capacity of the healthcare services network. These assumptions generated the search for other means of access, such as the judicialization of health45,46.
ORPHAN DISEASES POLICIES
The European Medicines Agency (EMA) orphan designation - Regulation (EC) nº 141/2000 of the European Parliament and Council - has helped to approve in the market upwards new therapies for rare diseases47. However, the authorization does not reflect more significant access to these medicines that are still variable in Europe despite all implementing similar standards for approving medicines48. Nevertheless, in England, more patients can access medicines for rare diseases in less time than other European countries. In the organizational settings, the CCGs are responsible for diseases with an incidence of fewer than 400 cases, providing at a regional level specific services designed for Specialized Commissioning Groups. Very few highly specialized services are provided in units across England, and the medicines are most commonly dispensed from hospital pharmacies49. A study conducted by Gammi et al.50, identified six broad categories of regulation and policy instruments to enable patient access, including England. High prices and insufficient evidence often limit orphan medicines from meeting the traditional HTA, especially CE, influencing access. England had the highest number of MEAs for orphan medicines in European countries.
In Brazil, the Rare Diseases Comprehensive Care Policy for the SUS was introduced in 201451. The MoH has defined the annual plan of action and procurement and financing of medicines. Currently, 46 protocols covered by the SUS have the care and medicines dispensation guaranteed in 19 specialized units (hospital) or via ministerial order programs52. However, some treatments are not officially covered by the Brazilian government, and patients can receive treatment after recourse to the courts53. Thus, Brazil also struggles in establishing a health policy for each rare disease, especially new rational models to address this growing challenge on the role of HTA in decision-making for integrated approaches that combine aspects of (bio) ethics, law, the health sciences, and economics54.
POLICIES FOR ONCOLOGICAL MEDICINES
Among European countries with the lowest reimbursement rate for licensed cancer medicines, England appears with 38% of 48 medicines licensed in a study conducted by Cheema et al.55, and the argument most accepted in the cause of rejection is lack of CE55. In 2011, to overcome this precept and increase access to innovative medicines, the Cancer Drugs Fund (CDF) was established as a financing source for HPMs without CE. Many MEAs have been performed between companies and the NHS to guarantee cost-effective prices during other data collection. A total of 783 recommendations were published from March 2000 to October 2017. The overall rate of approval for all technologies was 81% (513 of 634 recommendations), and the cancer treatment rate was 64% (119 of 185 recommendations)56. However, Aggarwal et al. affirm that there is no indication that substantial funds allocated to this program have brought significant benefits to patients. In 2016, a clear reform of the CDF was proposed, which will soon impact access and spending on HPMs57. Furthermore, a Long-Term Plan, published in 2019, is a consonance policy related to improving cancer outcomes and services in England over the next ten years. The Cancer Vanguard was an alliance derived from the plan resulting in the introduction of biosimilars into the NHS. We observe the growing use of biosimilars across England, and significant policy changes are related to more effective access to treatment options determined by pharmacogenetics58.
In Brazil, access to oncological medicines occurs only in high-complexity care. The MoH is responsible for the oncological network and financial policy established. The centers denominated High-Complexity Oncology Centers (CACON) and High-Complexity Oncology Units (UNACON) are responsible for comprehensive care, including patient’s access to oncological medicines59. Besides legislation creating comprehensive insurance systems, a study by Barrios and Werutsky60 highlights that access to innovative oncological HPMs is low in Brazil and, in general, is lagging many years behind concerning the date approval in developed countries. Recently, expanding on this discrepancy, the Brazilian Clinical Oncology Society reported that there were 37 therapeutic indications available in the private but not the public health system. A study by Lopes and Barrios61 showed how LMICs use multiple mechanisms to increase access to cancer medicines. They highlight the need to expand generic and biosimilar policies for oncological medicines, participate in clinical trials, compulsory licensing schemes, and increase multiple-stakeholder public-private partnerships.
COMPULSORY LICENSING AND LOCAL PRODUCT DEVELOPMENT POLICY
In Brazil, the Industrial Property Law nº 9.279 was approved in 1996, becoming compliant with the Agreement on Trade-Related Aspects of Intellectual Property Rights (TRIPS). All members of the World Trade Organization had to adapt their industrial property to comply with the new legal framework, which meant granting patents in the pharmaceutical sector. However, the agreement had negative reverberations, with fertile ground for HPMs created by changes in medicine lifecycle dynamics62. Adopting compulsory licenses was one of the strategies to increase access to HPMs in LMICs as Brazil63.
In 2007, using the safeguards defined in the TRIPS Agreement and the Doha Declaration, the Brazilian government adopted the compulsory license to produce the antiretroviral Efavirenz. The license was established by Presidential Decree nº 6.108/2007 and stemmed from the unsuccessful negotiations to reduce the price, which was consumed by about 40% of people living with HIV in Brazil. After the successful experience of the national production of the Efavirenz, the Partnerships for Productive Development (PDP) established, the instrument was implemented within the health policy to stimulate to the Health Industrial-Economic Complex (HEIC) and generate positive impacts against SUS demands64. In this regard, the Brazilian experience was the launch of the PDP in 2008, which placed the HEIC on the priorities of the new Brazilian industrial policy. The tool designed was that of the PDP as a specific action coordinated by the MoH. The basic PDP model involves central procurement of products (usually of high cost and greater technological complexity) purchased in the market (with a significant share of imports) to stimulate local production, involving technology transfer. In this context, the Bio-Manguinhos and Farmanguinhos Institutes (FIOCRUZ/MH) and Butantan were the prominent institutions involved in the technology transfer processes of interest to SUS65. However, from a global health viewpoint, this topic is widely discussed since, like the World Trade Organization member countries, the Brazilian government must enact legislation that facilitates compulsory licensing to meet legitimate public health needs, public funding of research, and disciplining investments in innovation, which always returns to a confrontation between health and trade that is increasingly evident and polarized66.
ACCELERATED ACCESS COLLABORATIVE POLICY (AAC)
In 2016, Accelerated Access Collaborative (AAC) was created as an initiative to select and simplify the development and approval process, set pricing, NICE assessment, and commercial negotiation, bringing forward access to innovative medicines. The aim was to allow the NHS to work positively and collaboratively and provide reassurance that system-wide, innovation-led changes will have significant benefits in streamlining clinical pathways, improving patient outcomes, and generating efficiencies in the system. Initially, England invested around £2 million, which is still being implemented and has covered 50,000 patients in seven strategic technology areas67.
Discussion
This integrative review summarizes pharmaceutical policies and practices and how similar health systems have adopted different ways to guarantee HPMs access and sustainability in each country. As mentioned in The High-Level Panel, access to medicines is not limited to LMICs, but this issue similarly affects rich countries67. In this way, countries must learn from each other to address the difficulties to define the best pharmaceutical policies to access these medicines and maintain the system’s sustainability. In literature, some policies such as patent reform legislation, reference pricing, outcome-based pricing, and incentivizing physicians to prescribe low-cost medicines are among the most promising short-term policy options brought to addressing the challenge of access to HPMs16,62. A study conducted in OECD member states showed that the policies most often evaluated and used in these countries were payment reforms, managed care, and cost-sharing. However, despite the importance of this topic to the countries, for many widely-used policies, especially in LMICs, minimal evidence is available on their effectiveness in containing healthcare costs68.
Remarkable differences are observed between the policies applied in Brazil and England. Among them, the Brazilian government has adopted the ERP to define medicines prices, while England has increased the agreements with industries (e.g., a voluntary scheme for branded medicines), showing different systems-price that ideally form a part of other policy control options. We cannot confirm that different mechanisms lead to different outcomes due to the lack of direct comparison of these systems’ price results. However, some results suggest that prices adopted in Brazil are higher than international benchmark prices21. The problem with ERP is the undisclosed rebates that most purchasers currently adopt for HPMs. Furthermore, the delay in launching the medicines in LMICs suggests that this system fails from the viewpoint of sustainability40,22. Likewise, England’s system agreement also fails to address the underlying factors, such as the lack of transparency in negotiations and several other negative repercussions of the current research and development model69,70.
As illustrated in the peri-launch category, both countries draw similar HTA criteria for selecting medicines for reimbursement. However, the use of HTA in Brazil can also be limited due to the need for high-level HTA skills (pharmacoeconomic approaches and definition of thresholds) and the involvement of different stakeholders, their roles and responsibilities, and also transparency in evaluation results27,28,30,31. Notwithstanding, a recent study identified that both HTA agencies (CONITEC and NICE) operate in a context of uncertainty in evaluating these medicines, which is expressed in the lack of critical assessment criteria in the final report published for their reimbursement (or not)71.
A new mechanism has been introduced in the field of HTA in the last few years. The MEAs in England to approve a new medicine for reimbursement are growing, and NICE has been adopting a work process that includes uncertainty in decision-making72. Negotiated discounts or confidential price sharing costs can provide the most efficient approach to achieving the right price and access to HPMs. However, these private arrangements can lead to acceptable uncertainty regarding a medicine’s efficacy, effectiveness, and safety. Because of the confidential discounts, a fair process that aids the assessment of these schemes is also required44,56. The lack of adequate evidence, unrealistic expectations, and pressure on stakeholders represent an appropriate backdrop for funding medicines without clear sustainability criteria in health care. As a result, Brazil introduced the first MEAs for nusinersen43. However, there is no evidence that the Brazilian health network is ready to evaluate the health outcomes independently to provide the outcome evidence. Therefore, the risk is likely to be only for the SUS, not the industry. Also, a new risk agreement may drastically affect the SUS principles, resulting in ineffective medicine payments that could adversely affect investments in preventive actions, especially those aimed at reducing prevalent health inequalities. This policy change points out to Brazil a new model that no longer gives priority to the development of national technology itself, in favor of using strategies like England, aiming only at market negotiation with industries. This Brazilian policy change has been aligned with significant economic policy changes involving decreased public investment.
Lastly, in post-launch policies, from a broader policy viewpoint, the CDF launch in 2010 gave the government in England the opportunity to build political capital, although it might also be described as a deregulation policy due to the very limited negative impact56,57. This review suggests that the judicialization of health in Brazil is similar to the CDF context that considers it acceptable for the courts to incorporate these uncertainties into available medicines. One Brazilian primary policy measure, which is different from the approach taken in England, is the local production and development capacity by HEIC64,65. A broader policy, ‘Medicines for the Many: Public Health before Private Profit’, has been discussed by the current English government, which addresses the issue of HPMs and recognizes that the current innovation system in the UK is bankrupt. It considers Brazil as an example to be analyzed about the use of flexibilities of the TRIPS agreement and a new local technology development model to bring economic opportunity and sustainability to the NHS69. However, the policy applied nowadays in England is the AAC, which has been implemented to accelerate innovative medicines access. Meanwhile, it has not yet been measured how much access this plan has produced - from uptake, clinical outcomes, and return on investment for this plan67.
Conclusions
Based on a variety of pharmaceutical policies related to this review, both governments have proposed different measures to ensure access to HPMs sustainably. Among the key differences between these countries, the Brazilian government has adopted the ERP policy, which can be seen as problematic, coupled with a weak HTA system without limit to reimburse for HPMs. These results suggest a long-term commitment by recessive policies adopted in the economic policy that will, for sure, impact the pharmaceutical field and people’s access to medicines. On the other hand, Brazil has a favorable policy, facing the barriers to access, with partnership proposals by intellectual property for public-private agreements. England has an apparent influence on the market with increasing managed agreements with industries, rate-of-return regulation for companies, and an overall limit for NHS spending, combined with a capital policy. Besides these differences, they are facing low success in controlling national pharmaceutical expenditure with considerable implications for the system’s overall sustainability. The fact is that access and sustainability to medicines is no longer just a problem for LMICs but has become a global problem. A comprehensive approach appears necessary to secure policy initiatives, reconsidering regulatory assessment requirements to address the paucity of true innovation observed among newly approved medicines. Contextualizing evaluations in each health system, culture, and society is a factor that may be evaluated, valued, and encouraged to balance global consensus and local needs to define what pharmaceutical policies can be helpful in each country.
References
Correa CM, Hilty RM, editors. Access to Medicines and Vaccines. Cham: Springer International Publishing; 2022.
World Health Organization. WHO guideline on country pharmaceutical pricing policies [access in 2019 Feb 4]. Available in: https://www.who.int/publications/i/item/9789240011878.
Dieleman JL, Squires E, Bui AL, et al. Factors associated with increases in US health care spending, 1996-2013. JAMA - J Am Med Assoc. 2017 [access in 2019 Jan 2]; 318(17):1668-78. Available in: https://jamanetwork.com/journals/jama/fullarticle/2661579.
Ewbank L, Omojomolo D, Sullivan K, et al. The Kings Fund. The rising cost of medicines to the NHS What’s the story? 2018. [access in 2019 Jun 23]. Available in: https://www.kingsfund.org.uk/sites/default/files/2018-04/Rising-cost-of-medicines.pdf.
Vieira FS. Evolução do gasto com medicamentos do sistema único de saúde no período de 2010 a 2016. Rio Janeiro. 2018. [access in 2019 Mar 23]; 7-10. Available in: https://www.ipea.gov.br/portal/images/stories/PDFs/TDs/180117_td_2356.pdf.
Hasan SS, Lu CY. Chapter 1 - Access to High-Cost Medicines: An Overview. In: Equitable Access to High-Cost Pharmaceuticals. Amsterdam: Elsevier; 2018. p. 1-10.
Hasan SS, Kow CS, Babar Z-U-D. High-Cost Medicines: Access, Affordability, and Prices. Enc. Pharm. Pract. Clin. Pharm. 2019 [access in 2019 Jul 25]; (2):20-6. Available in: https://pure.hud.ac.uk/en/publications/high-cost-medicines-access-affordability-and-prices.
Wahlster P, Scahill S, Lu CY, et al. Barriers to access and use of high-cost medicines: A review. Health Policy and Technology. 2015 [access in 2019 mar 23]; 4(3):191-214. Available in: https://www.sciencedirect.com/science/article/abs/pii/S2211883715000465.
Pan American Health Organization. Access to High-Cost Medicines in the Americas: Situation, Challenges and Perspectives. In: Pan American Health Organization. Technical Series - Essential Medicine, Access, and Innovation. Washington, D.C.: PAHO; 2010. p. 81. [access in 2019 Apr 24]. Available in: https://www.paho.org/hq/dmdocuments/2010/High-cost-Med-Tech-Series-No-1-Sep-15-10.pdf.
World Health Organization. Access to new medicines in Europe: technical review of policy initiatives and opportunities for collaboration and research. 2015. [access in 2019 Apr 24]. Available in: https://apps.who.int/iris/bitstream/handle/10665/159405/WHO-EURO-2015-3211-42969-60044-eng.pdf?sequence=3&isAllowed=y.
Ferreira MRJ, Mendes AN. Commodification in the reforms of the German, French and British health systems. Ciênc. Saúde Colet. 2018 [access in 2019 Apr 24]; 23(7):2159-70. Available in: http://www.cienciaesaudecoletiva.com.br/en/articles/commodification-in-the-reforms-of-the-german-french-and-british-health-systems/16838?id=16838.
De Souza LEPF, Paim JS, Teixeira CF, et al. The current challenges of the fight for a universal right to health in brazil. Ciênc. Saúde Colet. 2019 [access in 2020 Jul 28]; 24(8):2783-92. Available in: https://www.scielo.br/j/csc/a/HXCBkzpnQ7LbLKWqvXd3bGt/?lang=en.
Whittemore R, Knafl K. The integrative review: updated methodology. J. Adv. Nurs. 2005 [access in 2020 Jul 28]; 52(5):546-53. Available in: https://onlinelibrary.wiley.com/doi/10.1111/j.1365-2648.2005.03621.x.
Brandão CMR, Guerra Júnior AA, Cherchiglia ML, et al. Gastos do Ministério da Saúde do Brasil com Medicamentos de Alto Custo: Uma Análise Centrada no Paciente. Value in Health. 2011 [access in 2020 Jul 28]; 14(5):S71-7. Available in: https://www.sciencedirect.com/science/article/pii/S1098301511014446.
Packer C, Fung M, Stevens A. Analysing 10 years of early awareness and alert activity in the United Kingdom. Int J Technol Assess Health Care. 2012 [access in 2019 Nov 24]; 28(3):308-14. Available in: https://www.cambridge.org/core/journals/international-journal-of-technology-assessment-in-health-care/article/abs/analyzing-10-years-of-early-awareness-and-alert-activity-in-the-united-kingdom/88741A540C44A141530B5EA32AFAD73E.
Godman B, Bucsics A, Vella Bonnano P, et al. Barriers for Access to New Medicines: Searching for the balance between rising costs and limited budgets. Front Public Heal. 2018 [access 2019 Nov 24]; (6):328. Available in: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6290038/.
Brasil. Ministério da Saúde, Hospital Alemão Oswaldo Cruz. Monitoramento do Horizonte Tecnológico no Brasil: Avanços e Desafios. Ministério da Saúde: Brasília-DF; 2018. [access in 2022 Aug 24]. Available in: https://bvsms.saude.gov.br/bvs/publicacoes/monitoramento_horizonte_tecnologico_brasil.pdf.
Gomes PTC, Mata VE, Borges TC, et al. Horizon scanning in Brazil: Outputs and repercussions. Rev Saude Publica. 2019 [access in 2020 Jul 24]; 53(111):1-10. Available in: http://old.scielo.br/scielo.php?pid=S0034-89102019000100295&script=sci_arttext&tlng=en.
England. Department of Health & Social Care. The 2019 Voluntary Scheme for Branded Medicines Pricing and Access. [access in 2019 Dec 10]. Available in: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/761834/voluntary-scheme-for-branded-medicines-pricing-and-access-chapters-and-glossary.pdf.
Luiza VL, Oliveira MA, Chaves GC, et al. Pharmaceutical Policy in Brazil. In: Babar ZD, Editor. Pharmaceutical policy in countries with developing healthcare systems. Rio de Janeiro. Springer International Publishing; 2017. p. 23-149.
Bangalee V, Suleman F. A Comparative Study on Medicine Pricing in Brazil, Russia, India, China and South Africa (BRICS). Glob J Health Sci. 2018 [access in 2019 Jul 26]; 10(10):152. Available in: https://www.ccsenet.org/journal/index.php/gjhs/article/view/0/36973.
Vogler S, Schneider P, Zimmermann N. Evolution of Average European Medicine Prices: Implications for the Methodology of External Price Referencing. Pharm. Econ. 2019 [access 2019 jul 24]; 3(3):303-9. Available in: https://link.springer.com/article/10.1007/s41669-019-0120-9.
Charlton V. NICE and Fair? Health Technology Assessment Policy Under the UK’s National Institute for Health and Care Excellence, 1999-2018. Health Care Anal. 2019 [access in 2019 Jul 10]; 28(3):193-227. Available in: https://link.springer.com/article/10.1007/s10728-019-00381-x.
Cowles E, Marsden G, Cole A, Devlin N. A Review of NICE Methods and Processes Across Health Technology Assessment Programmes: Why the Differences and What is the Impact? Appl Health Econ Health Pol. 2017 [access in 2019 Jul 11]; 15(4):469-77. Available in: https://link.springer.com/article/10.1007/s40258-017-0309-y.
Allen N, Walker SR, Liberti L, et al. Health Technology Assessment (HTA) - Case Studies: Factors Influencing Divergent HTA Reimbursement Recommendations. Value Heal. 2017 [access in 2019 Jul 20]; 20(3):320-8. Available in: https://www.valueinhealthjournal.com/article/S1098-3015(16)30019-5/fulltext?_returnURL=https%3A%2F%2Flinkinghub.elsevier.com%2Fretrieve%2Fpii%2FS1098301516300195%3Fshowall%3Dtrue.
Chabot I, Rocchi A. Oncology drug health technology assessment recommendations: Canadian versus UK experiences. Clin Outcomes Res. 2014 [access in 2019 Jul 21]; 6(1):357-67. Available in: https://www.dovepress.com/oncology-drug-health-technology-assessment-recommendations-canadian-ve-peer-reviewed-fulltext-article-CEOR.
Silva HP, Elias FTS. Incorporação de tecnologias nos sistemas de saúde do Canadá e do Brasil: perspectivas para avanços nos processos de avaliação. Cad. Saúde Pública. 2019 [access in 2019 Nov 24]; (35):1-14. Available in: https://www.scielo.br/j/csp/a/6swM97RtM3jwggWMQLYVL3S/?lang=pt.
Kuchenbecker R, Polanczyk CA. Institutionalizing Health Technology Assessment in Brazil: Challenges Ahead. Value Heal Reg Issues. 2012 [access in 2019 Apr 24]; 1(2):257-61.
Raftery J, Powell J. Health Technology Assessment in the UK. Lancet. 2013 [access in 2019 Jul 5]; 382(9900):1278-85. Available in: https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(13)61724-9/fulltext.
Schwarzer R, Rochau U, Saverno K, et al. Systematic overview of cost-effectiveness thresholds in ten countries across four continents. J Comp Eff Res. 2015 [access in 2019 Jul 1]; 4(5):485-504. Available in: https://www.futuremedicine.com/doi/10.2217/cer.15.38.
Elias FTS, Araújo DV. How health economic evaluation (HEE) contributes to decision-making in public health care: The case of Brazil. Z Evid Fortbild Qual Gesundhwes. 2014 [access in 2019 Jul 1]; 108(7):405-12. Available in: https://pubmed.ncbi.nlm.nih.gov/25444299/.
Vogler S, Paris V, Ferrario A, et al. How Can Pricing and Reimbursement Policies Improve Affordable Access to Medicines? Lessons Learned from European Countries. Appl Health Econ Health Pol. 2017 [access in 2019 Jul 2]; 15(3):307-21. Available in: https://link.springer.com/article/10.1007/s40258-016-0300-z.
Angelis A, Lange A, Kanavos P. Using health technology assessment to assess the value of new medicines: results of a systematic review and expert consultation across eight European countries. Eur J Heal Econ. 2018 [access in 2019 Jul 2]; 19(1):123-52. Available in: https://link.springer.com/article/10.1007/s10198-017-0871-0.
Mejía A, Gilardino R, Kristensen FB, et al. Value-Based Pricing in Latin America: How Far Away Are We?. Value Heal Reg Issues. 2018 [access in 2019 Jul 02]; (7):1-5. Available in: https://www.sciencedirect.com/science/article/pii/S2212109918303625.
Okumura LM, Rosim M, Riveros BS, et al. A glance in hepatitis C policy in Brazil: Access and performance. Ciênc. Saúde Colet. 2018 [access in 2019 Jul 3]; 23(2):675-676. Available in: https://www.scielo.br/j/csc/a/N8WwyqMmbjmLHQvhBS4dJtw/?lang=pt.
Mathes T, Pieper D, Morche J, et al. Pay for performance for hospitals. Cochrane Database of Systematic Reviews. 2019 [access in 2019 Dec 1]; (7). Available in: https://www.cochranelibrary.com/cdsr/doi/10.1002/14651858.CD011156.pub2/full#:~:text=The%20aim%20of%20this%20Cochrane,care%2C%20resource%20use%20and%20equity.
Angelis A, Kanavos P. Multiple Criteria Decision Analysis (MCDA) for evaluating new medicines in Health Technology Assessment and beyond: The Advance Value Framework. Soc Sci Med. 2017 [access in 2019 Jul 6]; (188):137-56. Available in: https://www.sciencedirect.com/science/article/pii/S0277953617303933?via%3Dihub.
Campolina AG, Soárez PC, Amaral FV, et al. Análise de decisão multicritério para alocação de recursos e avaliação de tecnologias em saúde: Tão longe e tão perto?. Cad. Saúde Pública. 2017 [access in 2019 Aug 24]; 33(10):1-15. Available in: https://www.scielo.br/j/csp/a/Qgrxx6hCsCqR4JVndQZz3vS/?lang=pt.
Bouvy JC, Sapede C, Garner S. Managed Entry Agreements for Pharmaceuticals in the Context of Adaptive Pathways in Europe. Front Pharmacol. 2018 [access in 2019 Jan 24]; 279:280. Available in: https://www.frontiersin.org/articles/10.3389/fphar.2018.00280/full.
Ferrario A, Kanavos P. Dealing with uncertainty and high prices of new medicines: A comparative analysis of the use of managed entry agreements in Belgium, England, the Netherlands and Sweden. Soc Sci Med. 2015 [access in 2020 Jul 24]; (124):39-47. Available in: https://www.sciencedirect.com/science/article/abs/pii/S0277953614007266?via%3Dihub.
Pauwels K, Huys I, Vogler S, et al. Managed entry agreements for oncology drugs: Lessons from the European experience to inform the future. Front Pharmacol. 2017 [access in 2020 Jul 24]; 8:1-8. Available in: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5378787/.
Jarosławski S, Toumi M. Design of Patient Access Schemes in the UK: Influence of health technology assessment by the National Institute for Health and Clinical Excellence. Appl Health Econ Health Pol. 2011 [access in 2020 Jul 2]; 9(4):209-15. Available in: https://link.springer.com/article/10.2165/11592960-000000000-00000.
Caetano R, Hauegen RC, Osorio-de-Castro CGS. A incorporação do nusinersena no Sistema Único de Saúde: uma reflexão crítica sobre a institucionalização da avaliação de tecnologias em saúde no Brasil. Cad. Saúde Pública. 2019 [access in 2019 Jul 19]; 35(8):1-15. Available in: https://www.scielo.br/j/csp/a/Fv7D7nRF8DfRXFzCvW7KWbN/?lang=pt.
Baines D. Equitable Access to High-Cost Pharmaceuticals. ScienceDirect. [access in 2019 Jan 24]. Available in: https://www.sciencedirect.com/book/9780128119457/equitable-access-to-high-cost-pharmaceuticals 23-34.
Acurcio FA, Brandão CMR, Guerra Júnior AA, et al. Perfil demográfico e epidemiológico dos usuários de medicamentos de alto custo no Sistema Único de Saúde. Rev Bras Estud Popul. 2009 [access in 2019 Jul 24]; (26):263-82. Available in: https://www.scielo.br/j/rbepop/a/zDNWnTBNfhTW7GSZB3nMcfs/?lang=pt.
Rover MRM, Vargas-Pelaez CM, Rocha Farias M, et al. Acceso a medicamentos de alto precio en Brasil: La perspectiva de médicos, farmacéuticos y usuarios. Gac Sanit. 2016 [access in 2019 Jul 20]; (30):110-6. Available in: https://www.sciencedirect.com/science/article/pii/S0213911115002514.
Europe. European Parliament. Regulation (EC) nº 141/2000 of the European Parliament and the Council. Off J Eur Communities. 1999. [access in 2022 Jul 15]. Available in: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A32000R0141.
Blankart CR, Stargardt T, Schreyögg J. Availability of and Access to Orphan Drugs. Pharmacoeconomics. 2011 [access in 2019 Jul 24]; 29(1):63-82. Available in: https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0140002.
Detiček A, Locatelli I, Kos M. Patient Access to Medicines for Rare Diseases in European Countries. Value Heal. 2018 [access in 2020 Jul 24]; 21(5):553-60. Available in: https://www.valueinhealthjournal.com/article/S1098-3015(18)30189-X/fulltext?_returnURL=https%3A%2F%2Flinkinghub.elsevier.com%2Fretrieve%2Fpii%2FS109830151830189X%3Fshowall%3Dtrue.
Gammie T, Lu CY, Ud-Din Babar Z. Access to orphan drugs: A comprehensive review of legislations, regulations and policies in 35 countries. PLoS One. 2015 [access in 2020 Jul 24]; 10(10):1-24. Available in: https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0140002.
Brasil. Ministério da Saúde. Diretrizes para Atenção Integral às Pessoas com Doenças Raras no Sistema Único de Saúde - SUS. 2014. [access in 2020 Jul 10]. Available in: http://conitec.gov.br/images/Protocolos/Diretrizes_Atencao-DoencasRaras.pdf.
Schwartz IVD, Souza MV, Gilberto P, et al. Clinical genetics and public policies: how should rare diseases be managed? Clin Biomed Res. 2014 [access in 2020 Jul 24]; 34(2). Available in: https://seer.ufrgs.br/hcpa/article/view/47988.
Boy R, Schwartz IVD, Krug BC, et al. Ethical issues related to the access to orphan drugs in Brazil: the case of mucopolysaccharidosis type I. J Med Ethics. 2011 [access in 2020 Jul 24]; 37(4):233-9. Available in: https://jme.bmj.com/lookup/pmidlookup?view=long&pmid=21266385.
Souza MV, Krug BC, Picon PD, et al. Medicamentos de alto custo para doenças raras no Brasil: o exemplo das doenças lisossômicas. Ciênc. Saúde Colet. 2010 [access in 2020 Jul 24]; (15):3443-54. Available in: https://www.scielo.br/j/csc/a/r6Tsb5nQ6fW3HtQCLnHY7Kx/?lang=pt.
Cheema PK, Gavura S, Migus M, et al. International variability in the reimbursement of cancer drugs by publicly funded drug programs. Curr Oncol. 2012 [access in 2020 Jul 11]; 19(3):165-76. Available in: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3364777/.
Dillon A, Landells LJ. NICE, the NHS, and Cancer Drugs. JAMA. 2018 [access in 2020 Jul 24]; 319(8):767-768. Available in: https://jamanetwork.com/journals/jama/article-abstract/2671713.
Aggarwal A, Fojo T, Chamberlain C, et al. Do patient access schemes for high-cost cancer drugs deliver value to society? lessons from the NHS Cancer Drugs Fund. Ann Oncol. 2017 [access in 2020 Jul 24]; 28(8):1738-50. Available in: https://www.annalsofoncology.org/article/S0923-7534(19)32125-8/fulltext.
England. National cancer transformation programme, NHS. Achieving World-Class Cancer Outcomes: A strategy for England 2015-2020. Progress report 2016-17. 2017. [access in 2020 Jun 27]. Available in: https://www.england.nhs.uk/wp-content/uploads/2017/10/national-cancer-transformation-programme-2016-17-progress.pdf.
Madi MR, Cerri GG. Organisation of the cancer network in SUS: evolution of the care model. Clinics. 2018 [access in 2020 Jul 24]; 73(suppl1):e430s. Available in: https://www.scielo.br/j/clin/a/FscQY4j9nczynThgZfd8bWC/?lang=en.
Barrios CH, Reinert T, Werutsky G. Access to high-cost drugs for advanced breast cancer in Latin America, particularly trastuzumab. ecancermedicalscience. 2019 [access in 2020 Jul 24]; (13):898. Available in: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6372298/.
Lopes GDL, Souza JA, Barrios C. Access to cancer medications in low- and middle-income countries. Nat Rev Clin Oncol. 2013 [access in 2020 Jul 24]; 10(6):314-22. Available in: https://www.nature.com/articles/nrclinonc.2013.55.
Gronde TV, Uyl-de Groot CA, Pieters T. Addressing the challenge of high-priced prescription drugs in the era of precision medicine: A systematic review of drug life cycles, therapeutic drug markets, and regulatory frameworks. PLoS One. 2017 [access in 2020 Jul 24]; 12(8):e0182613. Available in: https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0182613.
World Trade Organization. Agreement on Trade-Related Aspects of Intellectual Property Rights (TRIPS). 1994. [access in 2020 Jun 27]. Available in: https://www.wto.org/english/docs_e/legal_e/27-trips_01_e.htm.
Gadelha CAG, Temporão JG. Development, innovation, and health: The theoretical and political perspective of the health economic-industrial complex. Ciênc. Saúde Colet. 2018 [access in 2020 Jul 24]; 23(6):1891-902. Available in: https://www.arca.fiocruz.br/handle/icict/47528.
Chaves GC, Hasenclever L, Osorio-de-Castro CGS, et al. Strategies for price reduction of HIV medicines under a monopoly situation in Brazil. Rev. Saúde Pública. 2015 [access in 2020 Jul 24]; 49(0). Available in: https://www.scielo.br/j/rsp/a/DmLdYsvnyLLdvfXMSj69Bnn/?lang=en.
Bermudez JAZ. Acesso a medicamentos: impasse entre a saúde e o comércio! Cad. Saúde Pública. 2017 [access in 2020 Jul 24]; 33(9):e00123117. Available in: http://cadernos.ensp.fiocruz.br/csp/artigo/228/acesso-a-medicamentos-impasse-entre-a-saude-e-o-comercio.
England. Accelerated Access Review: Final Report - Review of innovative medicines and medical Technologies. 2016. [access in 2020 Jul 24]. Available in: https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/565072/AAR_final.pdf.
Stadhouders N, Kruse F, Tanke M, et al. Effective healthcare cost-containment policies: A systematic review. Health Policy. 2019 [access in 2020 Jul 24]; 123(1):71-9. Available in: https://linkinghub.elsevier.com/retrieve/pii/S0168-8510(18)30634-1.
England. Public Health before Private Profit. 2020. [access in 2020 Jul 24]. Available in: https://labour.org.uk/wp-content/uploads/2019/09/Medicines-For-The-Many.pdf.
Abdel Rida N, Mohamed Ibrahim MI, Babar ZUD, et al. A systematic review of pharmaceutical pricing policies in developing countries. J Pharm Heal Serv Res. 2017 [access in 2020 Jul 24]; 8(4):213-26. Available in: https://onlinelibrary.wiley.com/doi/abs/10.1111/jphs.12191.
Vicente G, Cunico C, Leite SN. Transforming uncertainties into legitimate regulation? NICE and CONITEC agencies’ decisions on rare diseases. Ciênc. Saúde Colet. 2021 [access in 2020 Jul 24]; 26(11):5533-5546. Available in: https://www.scielo.br/j/csc/a/n9bDQMSzVLcdMZcnRF6Qqhm/?lang=en.
Brown P, Calnan M. NICE technology appraisals: working with multiple levels of uncertainty and the potential for bias. Med Health Care Philos. 2013 [access in 2020 Jul 24]; 16(2):281-93. Available in: https://link.springer.com/article/10.1007/s11019-011-9376-2.
Notes
Author notes
Vicente G (0000-0002-2758-4320)*, contributed to conception and planning, acquisition, data analysis and interpretation, structuring and final revision of the paper. Calnan M (0000-0002-7239-6898)* contributed to data analysis and interpretation of the paper and critical review of important intellectual content. Rech N (0000-0003-4808-4277)* contributed to critical review of important intellectual content. Leite S (0000-0002-5258-9684)* contributed to the structuring and review of draft and final versions of the paper.
Conflict of interest declaration
