Discussion papers
Using AUTO-ID Technologies in the Cuban Drug Logistics System to Increase its Safety and Efficiency
Uso de las tecnologías de AUTO-ID en el sistema logístico de medicamentos en Cuba para incrementar su seguridad y eficiencia
Uso de tecnologias AUTO-ID no sistema logístico de medicamentos em Cuba para aumentar sua segurança e eficiência
Using AUTO-ID Technologies in the Cuban Drug Logistics System to Increase its Safety and Efficiency
Sistemas & Telemática, vol. 16, no. 44, pp. 35-48, 2018
Universidad ICESI

This work is licensed under Creative Commons Attribution 4.0 International.
Received: 04 January 2018
Accepted: 02 February 2018
Abstract: This research was conducted involving five of the main supply chain of medicines in Cuba actors, including, since suppliers of medicines and the 3PL logistics operator, to healthcare centers and pharmacies. A diagnosis of the current state of the system of coding and classification of products was made. This activity showed, as main deficiencies, ineffectiveness on the products identification, since there are problems with codes, descriptions and units of measurements. A procedure to implement the AUTO-ID and EDI technologies proposed by GS1, was also designed, based on evaluating the effectiveness of these technologies in global logistics.
Keywords: AUTO-ID, EDI, GS1 international standards, coding, classifier, traceability, supply chain.
Resumen: La investigación que se reporta fue desarrollada involucrando cinco actores de la cadena de suministro de medicamentos en Cuba, que abarcan desde los proveedores de medicamentos y el operador logístico 3PL, hasta los centros asistenciales y las farmacias. Se realizó un diagnóstico del estado actual del sistema de codificación y clasificación de productos, lo que arrojó, como principales deficiencias, la inefectividad en la identificación de los productos, ya que existen problemas con los códigos, las descripciones y las unidades de medidas. Se diseñó además un procedimiento para implementar las tecnologías de AUTO-ID y EDI que propone GS1, a partir de evaluar la efectividad de estas tecnologías en la logística global.
Palabras clave: AUTO-ID, EDI, estándares internacionales GS1, codificación, clasificador, trazabilidad, cadena de suministro.
Resumo: A pesquisa relatada foi desenvolvida envolvendo cinco atores na cadeia de abastecimento de medicamentos em Cuba, que vão desde os fornecedores de medicamentos e o operador logístico 3PL, até os centros de saúde e farmácias. Foi elaborado um diagnóstico do estado atual do sistema de codificação e classificação dos produtos, o que mostrou, como principais deficiências, a ineficácia na identificação dos produtos, uma vez que existem problemas com os códigos, as descrições e as unidades de medidas. Foi desenhado um procedimento para implementar as tecnologias AUTO-ID e EDI propostas pela GS1, com base na avaliação da eficácia dessas tecnologias na logística global.
Palavras-chave: AUTO-ID, EDI, padrões internacionais GS1, codificação, classificador, rastreabilidade, cadeia de abastecimento.
I. Introduction
Supply chains, in general, are vulnerable to risks, especially regarding to traceability. In the pharmaceutical industry, it has generated facts all over the world that show the circulation of illicit medicines (WHO, 2017). Sub-Saharan Africa, India, Central, and South America, are the regions where a higher incidence is reported, it is stated that between 20% and 50% of medicines are illegal (“Comercio ilegal...”, 2010).
In Cuba, according to information from the regulatory agency of the sector, the Center for the State Control of Medicines, Equipment and Medical Devices [CECMED, Centro para el Control Estatal de Medicamentos, Equipos y Dispositivos Médicos], until 2016, has made several withdrawals from the market and communications of risks, due to quality problems in the medications (CECMED, 2017).
The traceability throughout the logistics system is a requirement in the drugs supply chains, then, it is necessary that products are correctly identified, which means that they can be identified in an unique way in each control point (ISO/IEC FDIS 15459-4:2014; NC-ISO 22005:2008; Krähenbühl & Haynes, 2012).
The unique identification is supported by the ISO/IEC FDIS 15459-4:2014 standard (Park, Zo, Ciganek, & Lim, 2011), which in session 4 standardizes the form of identification to all packaging levels and the relationship between the codes of each level. Its proposal focuses on all the actors in the chain using the standard to communicate and guarantee interoperability between information systems.
In the health field, Automatic Identification and Data Capture [AIDC] has been considered by the regulatory agencies as crucial in patient safety and in the accuracy and reliability of logistics. Since 1984, the Healthcare Industry Bar Code [HIBC] has been used as a way to capture data quickly and safely. Promoted by the International Medical Device Regulators Forum [IMDRF], with members such as the Food and Drug Administration [FDA] of the United States of America, the European Commission, an international project called Unique Device Identification [UDI] is started, which includes the AIDC as a center in the marking of the loads and as a data standard in the interoperable databases. The UDI was imposed as Federal Law in the United States in 2013, as the basis for its generalization from that year.
Information and communication technologies have revolutionized the supply chains, have made them safer and more efficient, that is why this article aims to evaluate the present conditions and the requirements for implementing a procedure to introduce technologies Auto-ID [Automatic Identification] and EDI [Electronic Data Interchange] technologies in the drug supply chain in Cuba.
The use of these technologies is widely addressed in the literature, but also, among other regulations have identified the following as support for the proposal: ISO/IEC 15424:2008 (Ferrer-Roca, 2011), ISO 17367:2013 and ISO/FDIS 29161:2016, all supporting the advent of enabling technologies for logistics 4.0.
II. Materials and Methods
The present research covers five actors in the supply chain: the importing units, the national producers, the 3PL logistics operator of the chain, and the health care centers and community pharmacies.
The Module for Coding and Classification of the products of the Reference Model of the Inventories is used for the appraisal of the codification system, with the application of the MRInv tool (Martínez, 2013). This module constitutes a checklist with thirty descriptors whose objective is to evaluate aspects related to the level of standardization of the coding systems that companies must use. The use of classification and coding systems regulated in the country and the correct use of the units of measurement in these systems is oriented. Each descriptor is evaluated using a 0 to 3 points scale, according to its level in the logistical system. “0” is assigned when the described practice is not exist and “3” when there is a high level of application. This structure lets determine the individual aspects and the modules constituting weaknesses or strengths, to then define the action plans that will allow the improvement of inventory management.
The main tools used during this research were: direct observation, interviews with experts, benchmarking, analysis of databases extracted from the information systems of the entities, the use of the MS Excel and MS Visio, and the bibliographic analysis.
III. Results
The use of automatic identification to obtain the data from products is a weakness of Cuban drugs logistic system. Its main cause is the low level of use of codification standards in the levels of packing (Martínez, 2013). In order to contribute to the solution of that problem, we propose the use of, at least, two codices for each product, one for classification, other for unique identification. For classification, the propose is supported in the need to organize the products in a way that allows managing the inventory in groups within a logical meaning for the chain of medicines, which is not guaranteed with the direct application of the national classifier proposed by the National Office of Statistics and Information [ONEI, Organización Nacional de Estadística e Información). For the coding system, the proposal is based on GS1 standards to be carried out for all packaging levels.
A. Use of Auto-ID y EDI Technologies in the Health Sector
The fact evidences that when there is not a data standardization needed by the computer systems through the whole drugs supply chain, and there isa manual introduction of the product data, these systems won´t be efficient. It may be issuing late, unreal, and ambiguous information about the state of the products.
To face this issue, the drug supply chain must adopt a system of coding and classification of its products, supported by an efficient technological infrastructure. There are only 39 agencies which emit Universal Device Identifier[UDI] codes, which can answer this need, including:
- Health Industry Business Communications Council [HIBCC];
- International Council for Commonality in Blood Banking Automation [ICCBBA];
- Eurodata Council [EDC];
- AIM [Association for Automatic Identification and Mobility];
- CEFIC [Chemical Industries Association];
- EDIFICE, for B2B business;
- ODETTE, for automotive industry;
- DIN [Deutshes Institut for Cooperation]; and
- GS1.
These companies or groups develop their own identification, classification, and data capture systems; based on standards and the conception that a product or a device is identified in a single way globally. Others use standards that are only functional within the confines of a single industry or a single country. The system of standards proposed by GS1 is one of the most used because it is global, solid, and multisectorial. It is also generated for the user and is scalable (GSI, 2011).
The GS1 standards system is a flexible architecture that ensures maximum efficiency. It is built around and on two secure elements: GS1 automatic identification standards and communication standards. This organization counts with 107 member organizations that serve 150 countries (GSI, 2011). In Cuba, its representation is located in the Chamber of Commerce and is named GS1 Cuba.
There are several traceability systems supported in the GS1 standards, such as the National Drug Traceability System in Argentina, the National Tracking and Pharmaceutical Tracking System [ITS] in Turkey, and some pilot tests supported by these standards are being implemented to implement systems national traceability in Brazil, Colombia, and Egypt (Gonzalez, 2013; Bulaon, 2016; Ünal, 2016; Afify & Mabrouk, 2014; Derecho & Sánchez, 2014; Filho, Blanco, & Ferreira, 2011; GS1, 2013; Brommeyer, 2013).
The technologies that allow the implementation of these systems based on the GS1 standards are the automatic identification of product data [Auto-ID] and electronic data interchange [EDI].
In twelve scientific papers reviewed on case studies in the field, it is generally observed that the data carrier DataMatrix GS1 is frequently used for the healthcare sector (Boden et al., 2014; Callan & Bailey, 2015; Heydrich, 2013; Izquierdo, 2016; Krähenbühl & Haynes, 2012; Wang & Chien 2013; Majois, 2013; Marechal & Jost, 2016; Martínez, 2014; Susuki, 2012; Webb, 2015; Wimmers, 2014). The advantage is that it allows the registration of several data (GTIN, No. Series, expiration date, etc.), necessary to perform the traceability. This two-dimensional carrier, embodied in very small formats of the primary packaging, enables hospitals and other health institutions to carry the exact control for each unit in the process of administering the medication to the patient. These centers need the technology of automatic data collection in their main processes (reception, re-labeling, distribution, administration to the patient). The GS1 DataMatrix can also be used in the secondary packaging, although it is recommended that it also be accompanied by the linear code to guarantee negotiations with the trading partners that use it.
The use of Radio Frequency Identification [RFID] technology in the health sector has also increased in recent years. The integration of RFID technology for pharmaceutical monitoring allows to optimize the delivery and logistics of the product, its security and the supervision of its administration (Jones, Henry, Cochran, & Frailey, 2010). In manufacturing and logistics, the main limitations of optical technology (linear or two-dimensional bar codes), compared to that of RFID (Guido, Mainetti, & Patron, 2012), are focused on limiting the speed of operations of the packaging lines and in lectures in logistics operations.
In some studies, it is suggested that the use of RFID (Rotunno, Cesarotti, Bellman, Introna, & Benedetti, 2014) may be negative in the physical characteristics of medicines, although Maffia, Mainetti, Patron, and Urso (2012) propose safe ways to use them and take advantage of their potential for the sector. Despitethis, the main limitation to the use of RFID tags are the high costs they represent, the customization of software and the integration systems with technology (Guido et al., 2012).
Izquierdo (2016), Callan and Bailey (2015), and Boden et al., (2014) highlight the use of the Global Data Synchronization Network [GDSN] to share product master data among the actors in the supply chain. The system is known as the National Product Catalog [NPC], which according to Callan and Bailey (2015) is a register for all products marketed in the health care sector.
The NPC is the only source of master data for health institutions seeking to buy medicines, medical devices and other necessary items in health care. For example, the NPC in Ireland is managed by GS1 Ireland for data synchronization, that is, a certified GDSN database. The existence of this communication standard allows, together with GS1 XML technology and electronic exchange, to optimize the negotiation processes between supply chain partners, as shown in Figure 1, where the electronic contracting model is presented developed by Callan and Bailey (2015).
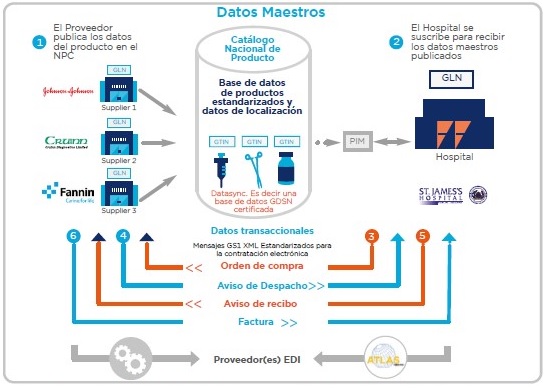
Figure 1
Electronic contracting modeling
B. Identification of Pharmaceutical Products in Cuba Drug Supply Chain
As a concept, a product classifier is a system of categories and subcategories of products that allow them to be grouped according to the needs of the activity (OMS, 2007), they have defining attributes that are similar to each other (internally homogeneous), but at the same time, allow to differentiate them from the other categories (externally heterogeneous) (INE, 2013). The above allows us to order and categorize characteristics of the economic and social reality of a country, region, city or locality (INEC, 2012).
In order to obtain the development of a product classification system, it should be considered, by definition, that the codes for identification are different from the codes for classification. The first ones are used to make an unambiguous identification of things, while the second ones are used to group similar goods into common categories (CED, 2007).
In Cuba drug supply chain, the Uniform Product Classifier [CUP, Clasificador Uniforme de Productos] is used as an identification code for pharmaceutical products. This code was elaborated in the first years of the decade of 1970 grouping goods of industrial and agricultural origin. Its structure was presented at a generic, sub-generic, and specific level. This classifier was discontinued by the National ONEI at the beginning of the 90s, due to limitations in its nomenclature and methodological specifications became obsolete.
Currently in the health sector continues to be used with a level called assortment. This level is granted by the Ministry of Public Health [MINSAP, Ministerio de Salud Pública] and corresponds to the combinations of digits that are reserved for a specific family to code the product.
The CUP allocation for national products is done when these are registered in the CECMED; for imported products, the allocation is done by their importers, as shown in Figure 2, where the current formation of the product code in the drug system is illustrated.
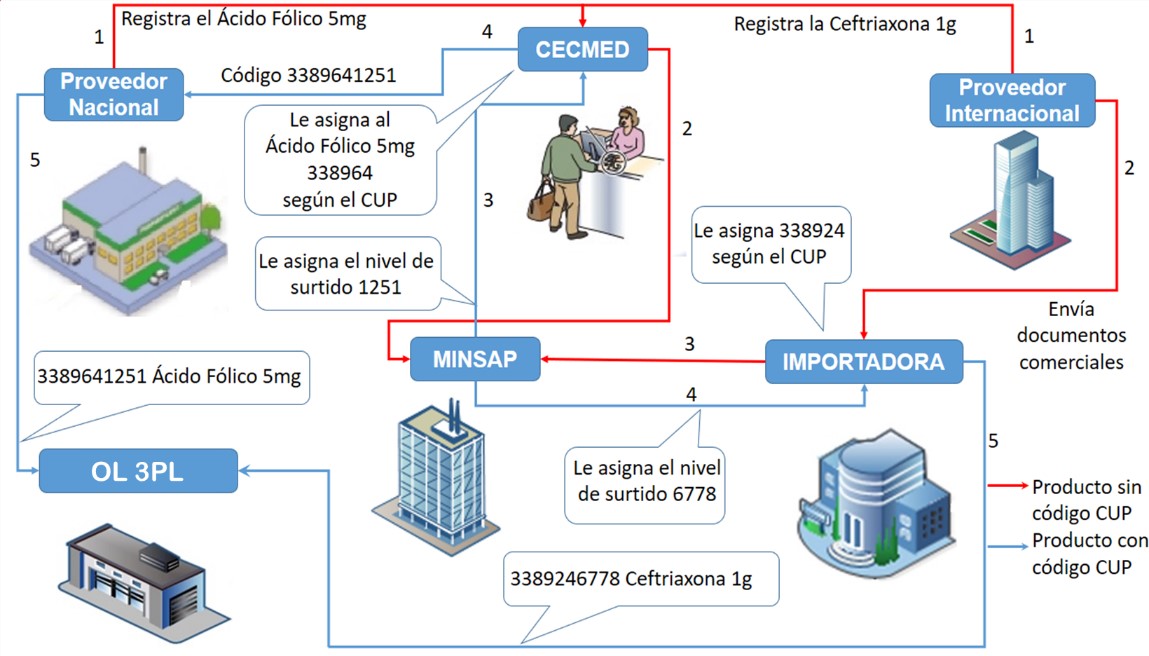
Figure 2
Example of the CUP code assignment for a national and an imported product
To evaluate the current situation in the identification of items, the Coding System and Classification System of the Inventory Reference Model designed by Martínez (2013) was applied to the 3PL Logistic Operator of the drug supply chain. The result yielded an evaluation of 2.47 points, considered acceptable; but the aspects evaluated are met by 70%, which means that there are still deficient aspects, among them the obsolete use of the CUP as the only code in the chain.
The main errors found in the product identification from the evaluation of the Coding System and Classifier of Products are:
- Products with the same code and different codes;
- products with different packing and the same presentation;
- products identified on their packaging with an international standard but not in commercial documents;
- absence of standardization in descriptions;
- use of measurement units that do not correspond to the International System of Units [ISU] and that are not standardized;
- introduction of information to computer systems not centered on the 3PL logistics operator; and
- poor use of automatic identification technologies.
In Figure 3 appears examples of the above errors.
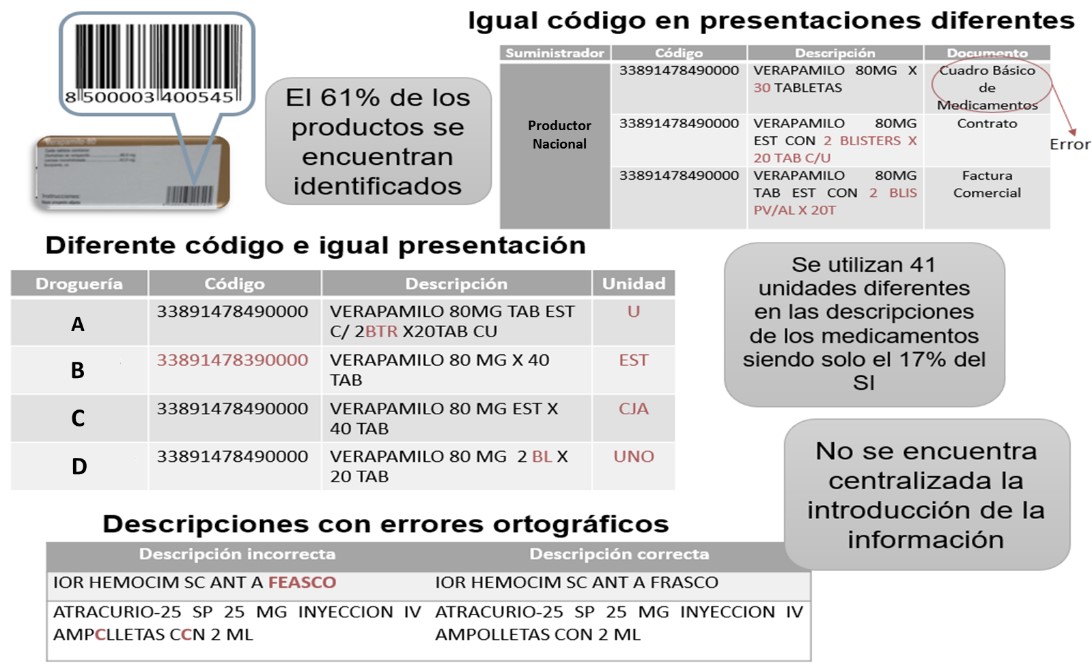
Figure 3
Issues in the coding and classification system
When the pharmaceutical product arrives at the healthcare centers, it is given an entry in the inventory through the introduction of its data in the computer system. The above are entered manually and through the name that already exists in the system, not through the code, which brings as a consequence that the same product, with the same dosage, but with a different code (is expressed in the bill), is reflected in the system with the same code, a situation that affects traceability.
One of the final actors in this chain are community pharmacies. Only those located in three municipalities of Havana have computerized media and scanners at the Point of Sale [POS], while the rest do not have any technology.
In both types of pharmacies, the inventory management is carried out, with the main identifier as the name of the product and not its code, which is because the same situations occur in health care centers. In the case of pharmacies without any type of technology, this issue is only reflected in the stowage or kardex cards, and not in the computer system.
In general, the perception is that the financial aspect is the restriction to implement automatic identification in the drugs supply chains. However, considering the opinion of these authors, is a priority to consider the implementation of global coding standards, such as Global Trade Identification Number [GTIN] codes or commercial items identification numbers. In Cuba, this issue affects all the economy sectors (Martínez, 2014).
C. Procedure for Auto-ID and EDI Technologies Implementation in Cuba Drugs Supply Chain
Based in the characteristics of GS1 standards, which support the use of Auto ID and EDI technologies, a procedure is designed to be implemented in Cuba drugs supply chain, in order to increase their safety and efficiency. The procedure has a scope throughout the supply chain and consists of four stages, as shown in Figure 4.
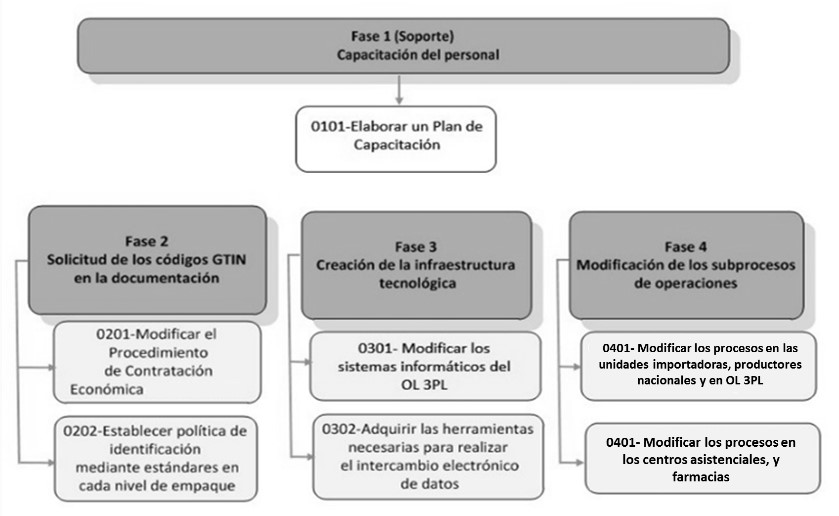
Figure 4.
Procedure stages for the implementation of Auto-ID and EDI
The proposal of the standards codes to be used for the identification of each level of pharmaceutical products packaging is shown in Figure 5, all supported by ISO 15459:2014.
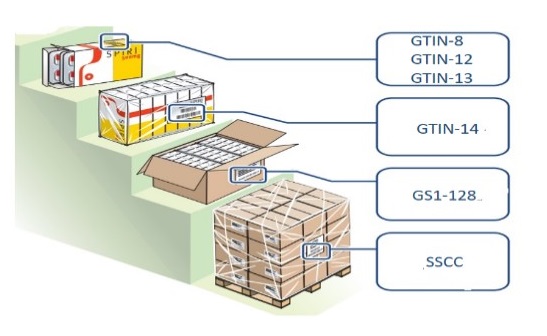
Figura 5
Codes according to packing level
In Phase 3 it is important to consider that the right selection of code scanners depends on the correspondence level achieved between the use conditions, the process requirements, and the technical characteristics of the scanner.
IV. Discussion
When analyzing the status of the coding and classification system of the drug supply chain in Cuba, the ineffectiveness of drug identification is seen as an important weakness. This situation can affect the withdrawals of products from the market or the monitoring that can be done to certain products with different purposes, both commercial and investigative, with respect to quality alerts.
These systems supported by the Auto-ID and EDI technologies allow safer and more efficient supply chains, since they guarantee the implementation of efficient traceability systems, such as the cases seen in section III. A of this article.
According to the proposed procedure, the implementation of the Auto-ID and EDI in Cuba drugs supply will allow obtaining benefits such as:
- Increase the security of the service provided to the population;
- improve the stocks management;
- decrease illegal activities;
- control costs and logistics cycles; and
- improve the evaluation of quality control and productivity.
The implementation of these technologies represents high costs for the sector, which is why it is necessary to perform analyzes to evaluate and contrast the risks and benefits, both economic and social, that will allow the Auto-ID and the EDI, based on the premise that at a higher technological level the chain will present lower security risks and will be more efficient.
V. Conclusions
The analysis of the current state of the coding and classification system allows us to identify that the main deficiencies in the identification of the product are: the use of an obsolete classifier, such as an encoder, that has brought various issues as the assignment of two codes for the same product, and that two products with different presentations have the same code; 41 different units of measurement are used in the descriptions of medicines, but of them, only 17% corresponding to the international measurement system; and it is not centralized the introduction of information to the system that allows the existence of several descriptions for the same product and with different units of measures, as well as encouraging in the health centers and pharmacies the stock introduction in wrong codes.
The non-use of the coding standards by the actors of the chain considerably limits the implementation of Auto-ID technologies. For the efficient implementation of Auto-ID and EDI technologies, a four-stage procedure, supported in the modification of actual process.
It is recommended that the processes in the drug system be designed including the implementation of automatic identification, in order to guarantee its traceability, from its origin to the patient. Although this is stated in the literature and in the case studies analyzed, it is considered that there is a lack of standardization of a procedure that can be implemented in a generalized manner in different types of health services.
Another element to be developed in future researches is the design of a tool to decide, from the analysis of logistical process, the class of scanners to be used, because it is hard for the logistics to decide just based in technical aspects and prices.
References
Afify, A. M. & Mabrouk, M. (2014). Safemed mantiene los medicamentos falsificados fuera de la cadena de abastecimiento en Egipto. In Guía de referencia GS1Healthcare 2013/2014, (pp. 17-22). México: GS1.
Boden, R. Wagener, S., & Sinha, I. (2014). Las Pequeñas y Medianas Empresas lideran el camino con los Estándares GS1. In Guía de referencia GS1Healthcare 2013/2014, (pp. 11-15). México: GS1.
Brommeyer, M. (2013). Healthcare suppliers benefiting from Global Data Synchronisation. In: GS1 Healthcare Reference Book 2012/2013, (pp. 4-10). Brussels, Belgium: GS1.
Bulaon, V. (2016). NSW Health continúa beneficiándose de la implementación de los estándares GS1. In: Manual de Referencia GS1 Healthcare 2015-2016: implementaciones exitosas de los estándares GS1, (pp. 42-45). Buenos Aires, Argentina: GS1.
Callan, P. V. & Bailey, P. (2015). La contratación electrónica (eProcurement) en el Hospital St. James, Dublin. In: Manual de Referencia GS1 Healthcare 2015-2016: implementaciones exitosas de los estándares GS1, (pp. 23-28). Buenos Aires, Argentina: GS1.
Centro para el Control Estatal de Medicamentos, Equipos y Dispositivos Médicos [CECMED]. (2017). Vigilancia postcomercialización. Retrieved from: http://www.cecmed.cu/tipo-de-funcion/vigilancia-postcomercializacion
Comercio ilegal de medicamentos, un peligro mortal para la salud. (2010). Retrieved from: http://boletinseps.blogspot.com/2010/02/comercio-ilegal-de-medicamentos-un.html
Consultorías Empresariales Delfos [CED]. (2007). Sistemas de clasificación internacional.
Derecho, M. & Sánchez, M. J. (2014). ANMAT marcando el camino. In: Guía de referencia GS1 Healthcare 2013/2014, (pp. 5-7). México: GS1.
Ferrer-Roca, O. (2011). Standards in telemedicine. In: E-Health systems quality and reliability: Models and standards: Models and Standards, (pp. 220-243). Hershey, PA: IGI GLobal.
Filho, A. F. M., Blanco, P., & Ferreira, L. (2011). Proyecto piloto de sistema de trazabilidad de productos farmacéuticos en Brasil. In: Guía de Referencia de GS1 Healthcare 2010/2011, (pp. 9-12). Buenos Aires, Argentina: GS1.
Gonzalez, J. E. (2013). Deployment of a traceability system by a pharmaceutical wholesaler leveraging GS1 Standards. In: GS1 Healthcare Reference Book 2012/2013, (pp. 19-22). Brussels, Belgium: GS1.
GS1. (2011). El valor y los beneficios del sistema de estándares GS1. Buenos Aires, Argentina: GS1.
GS1. (2013). Mount Waverley, Australia: SG1.
Guido, A. L., Mainetti, L., & Patrono, L. (2012). Evaluating potential benefits of the use of RFID, EPCglobal, and ebXML in the pharmaceutical supply chain. International Journal of Healthcare Technology and Management, 13(4), 198-222. doi:10.1504/IJHTM.2012.050625.
Heydrich, J. (2013). Implementing GS1 datamatrix at Moinhos de Vento Hospital: Ensuring drug traceability and patient safety. In: GS1 Healthcare Reference Book 2012/2013, (pp. 15-18). Brussels, Belgium: GS1.
Instituto Nacional de Estadísticas [INE]. (2013). Manual de codificación y supervisión. Santiago de Chile: INE.
Instituto Nacional de Estadísticas y Censos [INEC]. (2012). Clasificación nacional central de productos. Quito, Ecuador: INEC.
ISO/IEC FDIS 15459-4:2014 - Information technology – Automatic identification and data capture techniques – Unique identification - Part 4: Individual products and product packages. Geneva, Switzerland: ISO
ISO/IEC 15424:2008 - Information technology – Automatic identification and data capture techniques — Data Carrier Identifiers (including Symbology Identifiers). Geneva, Switzerland: ISO
ISO 17367:2013- Supply chain applications of RFID – Product tagging. Geneva, Switzerland: ISO
ISO/FDIS 29161:2016 - Information technology – Data structure – Unique identification for the Internet of Things. Geneva, Switzerland: ISO.
Izquierdo, D. E. (2016). El Sanatorio Güemes mejora la seguridad del paciente mediante la iniciativa de trazabilidad. In: Manual de Referencia GS1 Healthcare 2015-2016: implementaciones exitosas de los estándares GS1, (pp. 8-11). Buenos Aires, Argentina: GS1.
Jones, E., Henry, M., Cochran, D., & Frailey, T. (2010). RFID pharmaceutical tracking: From manufacturer through in vivo drug delivery. Journal of Medical Devices, 4(1). doi:10.1115/1.4000495
Krähenbühl, C., & Haynes, I. (2012). Uso de Estándares GS1 para combatir la falsificación y mejorar la seguridad del paciente. . In: Guía de Referencia de GS1 Healthcare 2011/2012, (pp. 51-53). Buenos Aires, Argentina: GS1.
Maffia, M., Mainetti, L., Patrono, L., & Urso, E. (2012). Potential impact of RFID-based tracing systems on the integrity of pharmaceutical products Advanced RFID Systems. In: Advanced RFID systems, security, and applications, (pp. 241-263): Hershey, PA: IGI Global.
Majois, P. (2013). Baxter endorses GS1 standards as a building block of brand integrity programme. In: GS1 Healthcare Reference Book 2012/2013, (pp. 13-14). Brussels, Belgium: GS1.
Marechal, B. & Jost, V. (2016). Permitiendo la trazabilidad en el Hospital Universitario Dijon mediante la identificación de todas las habitaciones y localizaciones. In: Manual de Referencia GS1 Healthcare 2015-2016: implementaciones exitosas de los estándares GS1, (pp. 19-22). Buenos Aires, Argentina: GS1.
Martínez, I. L. (2013). Modelo de referencia para la evaluación de la gestión de inventarios en los sistemas logísticos [thesis]. Universidad tecnológica de La Habana: Cuba.
Martínez, I. L. (2014). Problemas de codificación de productos que afectan la gestión de inventarios: Caso de estudio en empresas cubanas. DYNA, 81(187), 64-72. doi: 10.15446/dyna.v81n186.40070
NC-ISO 22005:2008: Trazabilidad de la cadena alimentaria — principios generales y requisitos fundamentales para el diseño y la implementación del sistema. La Habana, Cuba: Oficina Nacional de Normalización [NC].
Organización Mundial de la Salud [OMS]. (2007). Proyecto de elaboración de una clasificación internacional para la seguridad del paciente: informe de los resultados de la encuesta Delfos sobre la introducción a la clasificación internacional para la seguridad del paciente. Ginebra, Suiza: OMS.
Park, S., Zo, H., Ciganek, A. P., & Lim, G. G. (2011). Examining success factors in the adoption of digital object identifier systems. Electronic Commerce Research and Applications, 10(6), 626-636.
Rotunno, R., Cesarotti, V., Bellman, A., Introna, V., & Benedetti, M. (2014). Impact of track and trace integration on pharmaceutical production systems. International Journal of Engineering Business Management, 6(25). doi:10.5772/58934
Suzuki, R. (2012). La administración de logística de productos farmacéuticos con códigos de barras y etiquetas RFID, TBC Tokio, Toho Pharmaceutical Co. Ltda. In: Guía de Referencia de GS1 Healthcare 2011/2012, (pp. 35-39). Buenos Aires, Argentina: GS1.
Ünal, P. Ö. (2016). Turquía implementa el primer sistema nacional exitoso de rastreo y seguimiento farmacéutico (ITS) para una cadena de abastecimiento segura y confiable. In: Manual de Referencia GS1 Healthcare 2015-2016: implementaciones exitosas de los estándares GS1, (pp. 46-49). Buenos Aires, Argentina: GS1.
Wang, L-Ch & Chien, S-Y. (2013). TFDA, CCH and GS1 Taiwan advancing GS1 barcodes to improve patient safety. In: GS1 Healthcare Reference Book 2012/2013, (pp. 30-32). Brussels, Belgium: GS1.
Webb, J. (2015). Kent Pharmaceuticals: una serialización farmacéutica y un sistema de verificación en vivo. In: Manual de Referencia GS1 Healthcare 2014-2015: doce historias de implementación exitosas, (pp. 41-44). Buenos Aires, Argentina: GS1.
Wimmers, H. (2014). Implementación de la trazabilidad de medicamentos en el Hospital Alemán en Argentina. In: Guía de referencia GS1 Healthcare 2013/2014, (pp. 8-10). México: GS1.
World Health Organization [WHO]. (2017). Global surveillance and monitoring system for substandard and falsified medical products. Geneva, Switzerland: WHO.
Author notes
Doctor en Ciencias Técnicas. Graduado de Ingeniería Industrial, especializado en Organización de Empresas. Profesor de la Facultad de Ingeniería Industrial de la CUJAE, Jefe del Departamento de Ingeniería Industrial. Miembro del grupo de investigación de Logística y Gestión de la Producción [LOGESPRO]. Profesor titular en temas de logística y cadena de suministros en pregrado y posgrado.
.
Industrial Engineer, professor at the Faculty of Industrial Engineering of the CUJAE (Havana, Cuba). Member of the Logistics and Production Management research group [LOGESPRO]. Instructor professor in logistics and supply chain in undergraduate and postgraduate courses.
Ingeniera Industrial, profesora de la Facultad de Ingeniería Industrial de la CUJAE. Miembro del grupo de investigación de Logística y Gestión de la Producción [LOGESPRO]. Profesora Instructora en temas de logística y cadena de suministros en pregrado y posgrado.
Ingeniera Industrial, profesora de la Facultad de Ingeniería Industrial de la CUJAE. Miembro del grupo de investigación de Logística y Gestión de la Producción [LOGESPRO]. Profesora adiestrada en temas de logística y cadena de suministros en pregrado.
Ingeniera Industrial, profesora de la Facultad de Ingeniería Industrial de la CUJAE. Miembro del grupo de investigación de Logística y Gestión de la Producción (LOGESPRO). Profesora Adiestrada en temas de logística y cadena de suministros en pregrado.
Doctora en Ciencias Técnicas. Graduada de Ingeniería Industrial. Profesor de la Facultad de Ingeniería Industrial de la CUJAE. Coordinadora Ejecutiva de Laboratorio de Logística y Gestión de Producción [LOGESPRO]. Miembro de la Centro Europeo-Latinoamericano de Logística y Proyectos Ecológicos [CELALE]. Profesora titular en temas de logística y cadena de suministros en pregrado y posgrado.
Máster en Psicología Social. Licenciada en Psicología por la Universidad de La Habana [UH] (Cuba). Profesora de la Facultad de Psicología de la UH. Segunda jefa del Departamento de Ejercicio de la Profesión y profesora auxiliar de temas de psicología organizacional y comunicación organizacional.