Notas científicas
Selected body temperature of nine species of Mexican horned lizards ( Phrynosoma)
Temperatura corporal seleccionada de nueve especies mexicanas de lagartijas cornudas ( Phrynosoma)
Selected body temperature of nine species of Mexican horned lizards ( Phrynosoma)
Revista mexicana de biodiversidad, vol. 86, no. 1, 2015
Instituto de Biología
Received: 17 June 2014
Accepted: 17 September 2014
DOI: 10.7550/rmb.48028
Funding
Funding source: DGAPA-PAPIIT
Contract number: IN215011-3
Funding
Funding source: Conacyt
Contract number: 165066
Funding
Funding source: UC MEXUS-Conacyt
Contract number: FE-14-2
Abstract: Temperature plays an important role in the behavior, ecology, physiology, and evolution of reptiles. Herein, we present selected body temperatures on 9 species of Phrynosoma under laboratory conditions, and note congeneric differences related to altitude and reproductive mode. The results are discussed with those of other Phrynosomatid lizards.
Keywords: Thermal gradient, Phrynosomatids , Laboratory conditions, Preferred temperature, Reproductive mode, Altitude.
Resumen: La temperatura juega un rol importante en el comportamiento, ecología, fisiología y evolución de reptiles. Aquí, se presentan las temperaturas corporales seleccionadas de nueve especies de Phrynosoma bajo condiciones de laboratorio, tomando en cuenta la relación con la altitud y el modo reproductor. Se discuten los resultados con las temperaturas corporales de otros phrynosomátidos.
Palabras clave: Gradiente térmico, Phrynosomátidos, Condiciones de laboratorio, Temperatura preferida, Modo reproductor, Altitud.
In ectotherms most physiological mechanisms are affected by temperature, therefore they carefully regulate body temperature (T b) within a relatively narrow range by using behavioral adjustments ( Cowles & Bogert, 1944). Tb data collected in the field form the basis of most thermal biology reports in the literature ( Avery, 1982). In contrast, selected body temperatures (T sel) in laboratory conditions are rarely reported. T sel, also known as preferred body temperature, represents the range of body temperatures at which an ectothermic animal seeks to maintain itself by behavioral means in order to carry on their normal activities ( Brattstrom, 1965a). This temperature is commonly measured in laboratory conditions that provide an array of thermal environments free of physical and biotic constraints (e. g., predation, costs of thermoregulation; Clusella-Trullas, Terblanche, van Wyk, & Spotila, 2007). Tsel had been recorded for a wide range of diurnal lizard families, mostly in Scincidae, Agamidae, Lacertidae, Liolaemidae, and Phrynosomatidae ( Clusella-Trullas & Chown, 2014), nonetheless, within this last family the Tsel of horned lizards (genus Phrynosoma) is actually quite rare in the literature ( Lara-Reséndiz, Jezkova, Rosen, & Méndez-de la Cruz, 2014).
Horned lizards are one of the most distinctive lizards in North America. Most horned lizards have diets consisting primarily of native ants, dorsoventrally compressed bodies, cranial horns and body fringes, camouflaging coloration and sedentary habits, a constellation of unusual and interrelated traits including "relaxed" thermoregulation ( Pianka & Parker, 1975). According to the latest taxonomic revision, this genus comprises 17 species distributed from Canada to Guatemala ( Nieto-Montes de Oca, Arenas-Moreno, Beltrán-Sánchez, & Leaché, 2014). Among lizards, this genus has one of the largest altitudinal distributions (-200 to 3,440 m elev.) and 2 groups have evolved viviparity, apparently a characteristic associated with montane habitats ( Hodges, 2004). There have been several reports on T b in the field and critical temperatures in Phrynosoma ( Cowles & Bogert, 1944; Heath, 1962; Lemos-Espinal, Smith, & Ballinger, 1997; Pianka & Parker, 1975; Woolrich-Piña, Smith, & Lemos-Espinal, 2012, and others) and various other reports detailing aspects of its thermal ecology ( Brattstrom, 1965b; Christian, 1998; Guyer & Linder, 1985; Powell & Russell, 1985; Prieto & Whitford, 1971, and others). Nevertheless Tsel of horned lizards has attracted relatively little study even though it is essential to understanding behavior, natural history, ecophysiology ( Huey, 1982; Sherbrooke, 1997; Sherbrooke & Frost, 1989), and responses to the effects of global warming ( Sinervo et al., 2010). Therefore, our principal objective was to determine the selected body temperatures of Phrynosoma in a laboratory thermal gradient and to discuss the results with those of other Phrynosomatid lizards.
We conducted fieldwork during 4 years (2010-2013) in 9 localities from Mexico, A) Zumpago de Neri, Guerrero ( P. asio and P. taurus; 17°36'11.73" N, 99°31'19.74" W, 1,130-1,400 m elev.) in a deciduous tropical forest; B) Ensenada, Baja California ( P. blainvilli; 31°55'14.94" N, 116°36'32.6" W, 235 m elev.) in a chaparral community; C) volcán La Malinche, Tlaxcala ( P. orbiculare; 19°14'38.49" N, 97°59'26.48" W, 3,140 m elev.) in a montane woodland and grassland; D) Chilapa de Álvarez, Guerrero ( P. sherbrookei; 17°33'10.85" N, 99°16'15.01" W, 2,020 m elev.) in an oak forest with patches of grassland and scrub; E) Zapotitlán de las Salinas, Puebla ( P. taurus; 18°19'39.68" N, 97°27'16.17" W, 1,400 m elev.) in a desert scrub with cacti and xerophilous plants; F) Hermosillo, Sonora ( P. solare; 28°57'49.82" N, 111°4'1.2" W, 160 m elev.) in a desert scrub habitat, some with large, columnar cacti and small trees; G) Altamirano, Janos, Chihuahua ( P. hernandesi; 30°20'58.24" N, 108°29'26.3" W, 1,830 m elev.) in an oak forest with patches of grassland and scrub; H) Samalayuca, Ciudad Juárez, Chihuahua ( P. modestum; 31°28'33.1" N, 106°28'33.6" W, 1,260 m elev.) in sand dunes and semi stabilized dunes; I) Janos, Chihuahua ( P. cornutum; 30°49'31.6" N, 108°28'26.6" W, 1,435 m elev.) in grasslands and scrublands.
All animals were captured by hand. Snout-vent length (SVL) was measured to the nearest 0.1 mm, and sex was determined for all individuals. Our data are based only on adult Phrynosoma ( Table 1). In the laboratory, the lizards were maintained at 20 °C in plastic containers. Laboratory experiments were conducted one day after capture using a thermal gradient located in a 20 °C controlled-temperature room. The thermal gradient consisted of a wood box 150 cm × 100 cm × 70 cm high divided into 10 lengthwise tracks separated by insulation barriers to prevent behavioral influence of adjacent lizards. The substrate was 2-3 cm of sandy soil. At one end, and at the center of the box, 3 lamps of 100 W hanging at 30 cm to generate a thermal gradient from 20 to 50 °C throughout the tracks. The T sel of individuals in the thermal gradient was taken manually each hour, using the digital thermometer Fluke model 51-II, according to the activity period observed in the field and previous records for each species. The 25% and 75% quartiles of each species' T sel range (T sel25 and T sel75) were calculated to obtain the lower and upper limits. After laboratory experiments, all lizards were released at the site of capture.
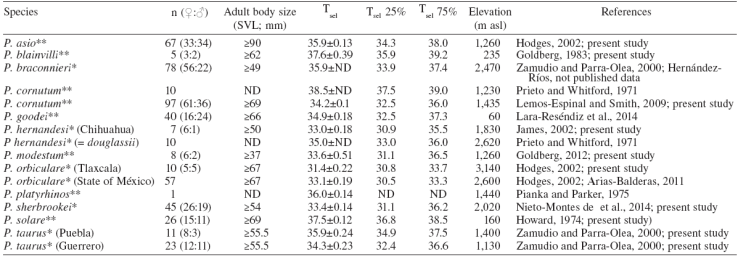
The results in Table 1 show the T sel average ± standard error (mean±SE) and sample size ( n) of each species studied and previous published data. The means of T sel of the Phrynosoma species reported here are within the range of T sel observed in other species of Phrynosoma ( Table 1). Furthermore, Sinervo et al. (2010) reported an average of Tb of 35.2±0.20 °C for the family Phrynosomatidae considering 210 populations and a T b range of 26.8-41.5 °C, which is similar to other diurnal lizards ( Andrews, 1998). Similarly, Clusella-Trullas and Chown (2014) reported an average of T sel of 35.1±2.2 °C considering 20 species of phynosomatids. Moreover, Phrynosoma cornutum had the highest T sel (38.5 °C) and P. orbiculare had the lowest (31.4 °C) ( Fig. 1). T sel of oviparous species varied from 33.5 to 38.5 °C and viviparous species from 31.4 to 35.9 °C ( Fig. 1).
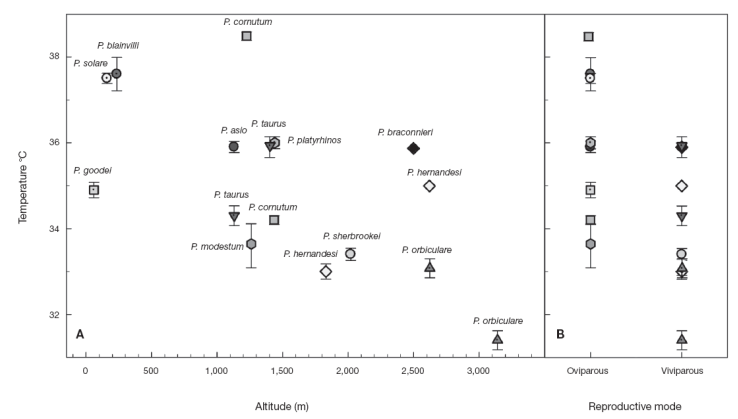
Figure 1
Mean selected body temperature (Tsel ) of horned lizards ( Phrynosoma) as a function of elevation (A) and reproductive mode (B).
Although the Ts el have a genetic basis, in many cases this T sel of lizards can be altered due to selective pressures of ambient temperatures, as an adaptation to habitat. This means that the T sel of individuals may be influenced by local environment temperatures ( Huey & Bennett, 1987; Sinervo, 1990). Many studies have addressed questions regarding labile or static aspects of lizard's thermal biology (see Clusella-Trullas & Chown, 2014 and references therein). Recently, it has been suggested that T b and Tsel tend to be similar and conserved within closely related species, even at the level of genus, despite obvious morphological differences, such as size, coloration, reproductive mode or different environmental conditions ( Grigg & Buckley, 2013). However, we found that the mean T sel of horned lizard species seems to be related to the type of habitat, therefore not being overall conserved within the genus Phrynosoma. At localities with higher ambient temperatures horned lizard species tend to have higher average T sel, while in cooler habitats horned lizard species have lower average T sel. Again within the genus, higher and lower mean T sel appear related, respectively, with oviparous and viviparous modes of reproduction ( Fig. 1), as noted by Hodges (2004).
Future phylogenetic comparative analysis of thermal preferences of horned lizards should refine issues related to elevation, reproductive mode and other life history traits. In addition, the direction and magnitude of labile shifts in T sel should be incorporated into mechanistic models ( Lara-Reséndiz et al., 2014; Sinervo et al., 2010), to improve predictions of the impact of global climate change on populations of Phrynosoma.
In conclusion, the results presented above suggest that Phrynosoma maintain similar T sel to other Phrynosomatids and relatively higher T sel at low elevations than at high elevations. However, this conclusion should be considered preliminary, pending broader data and incorporating biophysical models and phylogenetic analysis on their thermal biology.
Acknowledgments
This research was supported by a DGAPA-PAPIIT grant to F. R. Méndez-De la Cruz (IN215011-3). R. A. Lara-Reséndiz was funded by a Conacyt doctoral scholarship (165066) and a UC MEXUS-Conacyt postdoctoral fellowship (FE-14-2) to complete this manuscript. Collection permits (03699/09 and 05570/10) were given by Semarnat. We thank H. Gadsden, G. Castañeda, R. Huitrón, V. Jiménez, A. Díaz de la Vega, L.Rivera, S. González-Guzmán, L. López-Damián, M. García, D. Brindis, R. Santos, L. Florentino, and Solis-Ramírez family for field and laboratory assistance. Finally, we thank La Malinche Scientific Station, Helia Bravo Hollis Botanical Garden, and El Uno Ecological Reserve (TNC) for logistics support.
References
Andrews, R.M. (1998). Geographic variation in field body temperature of Sceloporus lizards. Journal of Thermal Biology, 23, 329-334.
Arias-Balderas, S.F. (2011). Termorregulación y comportamiento de forrajero de Phrynosoma orbiculare en El Arenal, Nicolás Romero, Estado de México. (M.Sc. Thesis). México, D.F.: Instituto de Biología, Universidad Nacional Autónoma de México.
Avery, R.A. (1982). Field studies of body temperatures and thermoregulation. In C. Gans & F.H. Pough (Eds.), Biology of the Reptilia (pp. 93-166). New York: Academic Press.
Brattstrom, B.H. (1965a). Body temperatures of reptiles. American Midland Naturalist, 73, 376-422.
Brattstrom, B.H. (1965b). Temperature regulation and diurnal activity in horned lizards. Berkeley: University of California Publications in Zoology (pp. 97-136).
Christian, K.A. (1998). Thermoregulation by the short-horned lizard ( Phrynosoma douglassi) at high elevation., Journal of Thermal Biology 23, 395-399.
Clusella-Trullas, S., & Chown, S.L. (2014). Lizard thermal trait variation at multiple scales: a review. Journal of Comparative Physiology B-Biochemical Systemic and Environmental Physiology, 184, 5-21.
Clusella-Trullas, S., Terblanche, J.S., van Wyk, J.H., & Spotila, J.R. (2007). Low repeatability of preferred body temperature in 4 species of cordylid lizards: temporal variation and implications for adaptive significance. Evolutionary Ecology, 21, 63-79.
Cowles, R.B., & Bogert, C.M. (1944). A preliminary study of the thermal requirements of desert reptiles. Bulletin of the American Museum of Natural History, 83, 263-296.
Goldberg, S.R. (1983). Reproduction of the coast horned lizard, Phrynosoma coronatum, in Southern California. Southwestern Naturalist, 28, 478-479.
Goldberg, S.R. (2012). Notes on reproduction in roundtail horned lizards, Phrynosoma modestum (Squamata: Phrynosomatidae) from New Mexico. Bulletin of the Chicago Herpetological Society, 47, 34-36.
Grigg, J.W., & Buckley, L.B. (2013). Conservatism of lizard thermal tolerances and body temperatures across evolutionary history and geography. Biology Letters, 9, 20121056.
Guyer, C., & Linder, A.D. (1985). Thermal ecology and activity patterns of the short-horned lizard ( Phrynosoma douglassi) and the sagebrush lizard ( Sceloporus graciosus) in southeastern Idaho. Great Basin Naturalist, 45, 607-614.
Heath, J.E. (1962). Temperature-independent morning emergence in lizards of the genus Phrynosoma. Science, 138, 891-892.
Hodges, W.L. (2002). Phrynosoma systematics, comparative reproductive ecology, and conservation of a Texas native. (Ph.D. Thesis). Austin: University of Texas.
Hodges, W.L. (2004). Evolution of viviparity in horned lizards ( Phrynosoma): testing the cold-climate hypothesis. Journal of Evolutionary Biology, 17, 1230-1237.
Howard, C.W. (1974). Comparative reproductive ecology of horned lizards (Genus Phrynosoma) in southwestern United States and northern Mexico. Journal of the Arizona Academy of, Science 9, 108-116.
Huey, R.B. (1982). Temperature, physiology, and ecology of reptiles. In C. Gans & F. H. Pough (Eds.), Biology of the Reptilia (pp. 25-91).. New York: Academic Press
Huey, R.B., & Bennett, A.F. (1987). Phylogenetic studies of coadaptation: preferred temperatures versus optimal performance temperatures of lizards. Evolution, 41, 1098-1115.
James, J.D. (2002). A survey of short-horned lizard ( Phrynosoma hernandesi hernandesi) populations in Alberta. Alberta Species at Risk Report, 29, 1-25.
Lara-Reséndiz, R.A., Jezkova, T., Rosen, P.C., & Méndez-de la Cruz, F.R. (2014). Thermoregulation during the summer season in the Goode's horned lizard Phrynosoma goodie (Iguania: Phrynosomatidae) in Sonoran Desert. Amphibia-Reptilia, 35, 161-172.
Lemos-Espinal, J.A., & Smith, H.M. (2009). Anfibios y reptiles del estado de Chihuahua, México/Amphibians and reptiles of the state of Chihuahua, Mexico. México, D. F.: Universidad Nacional Autónoma de México-Comisión para el Conocimiento y Uso de la Biodiversidad.
Lemos-Espinal, J.A., Smith, G.R., & Ballinger, R.E. (1997). Observations on the body temperatures and natural history of some Mexican reptiles. Bulletin of the Maryland Herpetological Society, 33, 159-164.
Nieto-Montes de Oca A., Arenas-Moreno, D., Beltrán-Sánchez E., & Leaché, A. (2014). A new species of horned lizard (genus Phrynosoma) from Guerrero, México, with an updated multilocus phylogeny. Herpetologica, 70, 241-257.
Pianka, E.R., & Parker, W.S. (1975). Ecology of horned lizards: review with special reference to Phrynosoma platyrhinos. Copeia, 1975, 141-162.
Powell, G.L., & Russell, A.P. (1985). Field thermal ecology of the eastern short-horned lizard ( Phrynosoma douglassi brevirostre) in southern Alberta. Canadian Journal of Zoology, 63, 228-238.
Prieto, A.A. Jr., & Whitford, W.G. (1971). Physiological responses to temperature in horned lizards, Phrynosoma cornutum and Phrynosoma douglassii, Copeia 1971, 498-504.
Sherbrooke, W. C. (1997). Physiological (rapid) change of color in horned lizards ( Phrynosoma) of arid habits: hormonal regulation, effects of temperature, and role in nature., Amphibia-Reptilia 18, 155-175.
Sherbrooke, W. C., & Frost, S. K. (1989). Integumental chromatophores of a color-change thermoregulating lizard Phrynosoma modestum (Iguanidae; Reptilia). American Museum Novitates, 2943, 1-14.
Sinervo, B. (1990). Evolution of thermal physiology and growth rate between populations of the western fence lizard ( Sceloporus occidentalis). Oecologia, 83, 228-237.
Sinervo, B., Méndez-de-la-Cruz, F., Miles, D.B., Heulin, B., Bastiaans, E., Villagrán-Santa-Cruz, M., et al. (2010). Erosion of lizard diversity by climate change and altered thermal niches., Science 328, 894-899.
Woolrich-Piña, G. A., Smith, G.R., & Lemos-Espinal, J.A. (2012). Body temperatures of 3 species of Phrynosoma from Puebla, Mexico. Herpetology Notes, 5, 361-364.
Zamudio, K.R., & Parra-Olea, G. (2000). Reproductive mode and female reproductive cycles of 2 endemic Mexican horned lizards ( Phrynosoma taurus and Phrynosoma braconnieri)., Copeia 2000, 222-229.
Author notes
*Corresponding author. E-mail address: rafas.lara@gmail.com (R.A. Lara-Reséndiz).