Investigación experimental o metaanalítica

Esta obra está bajo una Licencia Creative Commons Atribución-NoComercial-SinDerivar 4.0 Internacional.
Recepción: 11 Octubre 2024
Recibido del documento revisado: 21 Abril 2025
Aprobación: 23 Abril 2025
Publicación: 08 Mayo 2025
DOI: https://doi.org/10.15517/pensarmov.v23i1.62242
Abstract: The objective of this article was to assess the effect of hydrotherapy on the functional status, dyspnea, BODE index, and quality of life in patients with COPD. A randomized, controlled clinical trial was carried out with 24 COPD patients, randomly divided in two groups: 1) aquatic training group (ATG), participating in an aquatic training (AT) program of intermittent (moderate/high) intensity that took place three times a week, for 8 weeks; and 2) control group (CG), which received guidance on a healthy lifestyle and did not participate in supervised exercises. The variables (functional status, dyspnea, BODE index, and quality of life) were assessed before and after the intervention period. The level of significance was established in 5%. Results show that the ATG presented significant improvements in daily life activities (LCADL total Δ score: -10.1, p = .01), in functional status (PFSDQ-M, total Δ score: -16.9, p = .01), dyspnea (ΔMRC: -1.37, p = .003), BODE index (Δ: -1.25, p = .048) and quality of life (total Δ score: -7.4, p = 0.02). The CG did not show any significant changes. As a conclusion, intermittent aquatic aerobic training of moderate/high intensity fostered a clinically relevant improvement for most patients with COPD in terms of quality of life, limitations in daily life activities, dyspnea, and in the BODE mortality predictor index.
Keywords: lung diseases, hydrotherapy, quality of life, physical activity.
Resumen: El objetivo del artículo fue evaluar el efecto de la hidroterapia sobre el estado funcional, disnea, índice BODE y calidad de vida en pacientes con enfermedad pulmonar obstructiva crónica (EPOC). Se realizó un ensayo clínico aleatorizado y controlado con 24 pacientes con EPOC, asignados aleatoriamente a dos grupos: 1) grupo de entrenamiento acuático (GEA), que participó en un programa de entrenamiento acuático de intensidad intermitente (moderada/alta) 3 veces a la semana durante 8 semanas; 2) grupo control (GC), que recibió orientación sobre estilo de vida saludable y no realizó ejercicios supervisados. El estado funcional, disnea, índice BODE y calidad de vida fueron evaluados antes y después del período de intervención. El nivel de significancia se estableció en el 5%. Los resultados muestran que el GEA presentó una mejora significativa en las actividades de la vida diaria (Δ puntaje total de LCADL: -10.1, p = .01), en el estado funcional (PFSDQ-M, Δ puntaje total: -16.9, p = .01), disnea (ΔMRC: -1.37, p = .003), índice BODE (Δ: -1,25, p = .048) y calidad de vida (Δpuntaje total: -7.4, p = .02). El GC no presentó cambios significativos. Se concluye que el entrenamiento aeróbico acuático intermitente de intensidad moderada/alta promovió una mejora clínicamente relevante en los pacientes con EPOC en términos de calidad de vida, limitaciones en las actividades de la vida diaria, disnea, y en el índice BODE, predictor de mortalidad.
Palabras clave: enfermedades pulmonares, hidroterapia, calidad de vida, actividad física.
Resumo: O objetivo deste artigo foi avaliar o efeito da hidroterapia sobre o estado funcional, dispneia, índice BODE e qualidade de vida em pacientes com doença pulmonar obstrutiva crônica (DPOC). Um ensaio clínico randomizado e controlado foi realizado com 24 pacientes com DPOC, divididos aleatoriamente em dois grupos: 1) grupo de treinamento aquático (GEA), que participou de um programa de treinamento aquático de intensidade intermitente (moderada/alta) 3 vezes por semana durante 8 semanas; 2) grupo controle (GC), que recebeu orientações sobre estilo de vida saudável e não realizou exercícios supervisionados. O estado funcional, a dispneia, o índice BODE e a qualidade de vida foram avaliados antes e após o período de intervenção. O nível de significância adotado foi de 5%. Os resultados mostram que o GEA apresentou uma melhora significativa nas atividades de vida diária (Δ escore LCADL total: -10,1, p = 0,01), no status de desempenho (PFSDQ-M, Δ pontuação total: -16,9, p = 0,01), dispneia (ΔMRC: -1,37, p = 0,003), índice BODE (Δ: -1,25, p = 0,048) e qualidade de vida (Δ escore total: -7,4, p = 0,02). O GC não apresentou alterações significativas. Conclui-se que o treinamento aeróbio aquático intermitente de intensidade moderada/alta promoveu melhora clinicamente relevante em pacientes com DPOC em termos de qualidade de vida, limitações nas atividades de vida diária, dispneia e no índice BODE, preditor de mortalidade.
Palavras-chave: doenças pulmonares, hidroterapia, qualidade de vida, atividade física.
1. Introduction
Patients with chronic obstructive pulmonary disease (COPD) present systemic repercussions compromising their activities of daily living (ADL), designated as functional status (Kocks et al., 2011).
Scientific literature has reported that the assessment of dyspnea (Janssens et al., 2011) and the limitations of the ADL can be measured through the Modified Pulmonary Functional Status and Dyspnea Questionnaire (PFSDQ-M) (Carpes et al., 2008; Garrod et al., 2000; Kovelis et al., 2008; Lareau et al., 1998) and the London Chest Activity of Daily Living (LCADL) scale, which assesses the dyspnea during the ADL (Garrod et al., 2000).
Additionally, patients with COPD present reduced performance in BODE index (BMI, obstructed airways, dyspnea, and exercise capacity), which is associated with mortality, providing important prognosis information for clinical practice (Celli et al., 2004). Therefore, changes in its values after pulmonary rehabilitation programs provide valuable impacts on the prognosis (Cote & Celli, 2005).
Physical exercise is an important component in a pulmonary rehabilitation program for patients with COPD. According to McCarthy et al. (2015), pulmonary rehabilitation promotes a relevant clinical improvement of dyspnea and fatigue. Aquatic exercises feature among the exercise modalities employed in rehabilitation programs. Zampogna et al. (2023) reports that this exercise modality is efficient in improving the exercise capacity and the quality of life in people with COPD. Chen et al. (2021) also add that aquatic exercises deliver significant additional effects to this population. However, most studies use a Moderate Intensity Continuous Training (Chen et al., 2021) protocol. In this sense, considering that intermittent training on the land has been regarded as a feasible and interesting alternative for patients with COPD (Adolfo et al., 2019; Vogiatzis et al., 2002), the authors raised the hypothesis that intermittent exercises in an aquatic environment could improve the functional status, dyspnea, BODE mortality predictor index, and quality of life in patients with COPD. Thus, the purpose of this study was to assess the effects of an 8-week intermittent aquatic training program of moderate/high intensity on the functional status, activity of daily living, dyspnea, mortality prediction, and quality of life of patients with COPD.
2. Methods
Study design and population
This is a randomized controlled trial which was conducted in accordance with the Helsinki declaration and approved by the research ethics committee of the Methodist University of Piracicaba-São Paulo-Brazil under protocol #13/11. All volunteers signed an informed consent form prior to their inclusion in the study.
Sixty-two patients from a Regional Specialty Outpatient Clinic diagnosed with COPD were invited to participate in the study. The inclusion criteria consisted of having a diagnosis of COPD, according to the Global Initiative for Chronic Obstructive Lung Disease (GOLD) recommendations (GOLD, 2022), and a low level of physical activity according to the International Physical Activity Questionnaire (IPAQ), defined as engaging in less than 600 MET-minutes of total physical activity per week or walking less than 30 minutes per day (Craig et al., 2003). Participants were excluded if they were not clinically stable; had experienced a COPD exacerbation in the past three months; had participated in regular physical training programs within the past six months; or presented with any of the following conditions: diabetes mellitus, endocrine disorders, liver diseases, other pulmonary diseases, associated inflammatory diseases, heart conditions, musculoskeletal or neuromuscular impairments that could prevent the execution of the experimental protocols, dermatological conditions (e.g., skin fistulas, infected wounds, skin mycoses, varicose ulcers), hypersensitivity to products used in pool water treatment, and/or hydrophobia.
Twenty-four male patients, aged between 50 and 75 years, met the inclusion criteria and accepted to participate, being randomly allocated into the control group (CG, n=12) or aquatic training group (ATG, n=12). Randomization was performed by a researcher who was not involved in the study. A random assignment website (https://www.sealedenvelope.com/simple-randomiser/v1/lists) was used to generate a sequence of numbers indicating group allocation (CG or ATG). The group assignments were printed on paper and placed into opaque envelopes according to the generated order. These envelopes were opened by the researcher responsible for the rehabilitation program only after the baseline assessments had been completed. Due to exacerbation of the COPD or personal reasons, 3 volunteers were excluded from the CG and 4 from the ATG (Figure 1). The CG was instructed to maintain the conventional clinical treatment, characterized by drug therapy, receiving instructions on healthy lifestyle, and only participated in the assessments. On the other hand, the ATG was submitted to an aquatic intermittent aerobic physical exercise program (Figure 1).
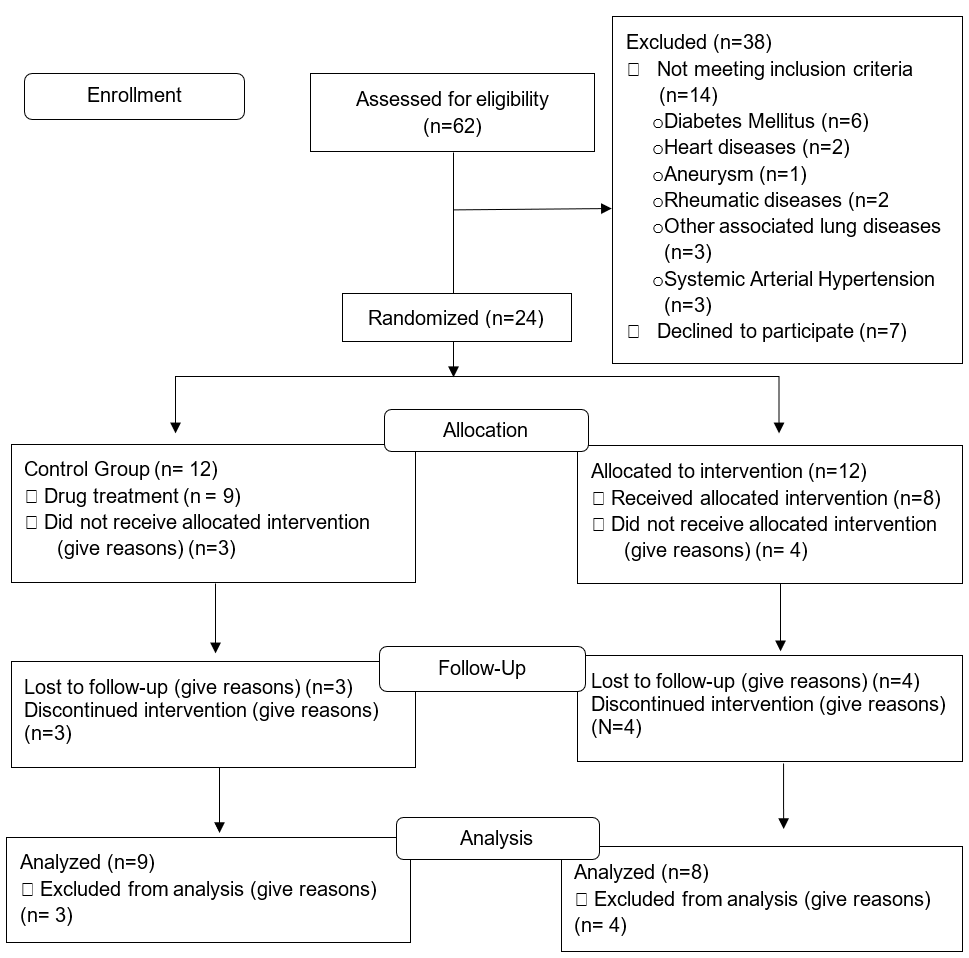
Figure 1
Inclusion criteria flowchart
Source: the authors.
Measurements and outcomes
Pulmonary function
The lung function was assessed through spirometry tests using a portable spirometer (Easy oneTM, ndd Medizintechnik AG, Zurich, Switzerland), in accordance with the American Thoracic Society (ATS) and European Respiratory Society (ERS) guidelines. Forced vital capacity (FVC), forced expiratory volume in the first second (FEV1) and the FEV1/FVC ratio were obtained as absolute and relative values (% of the predicted) (ATS, 2002; Miller et al., 2005; Pereira et al., 2007).
Quality of life
Quality of life was assessed through the application of the Brazilian version of the Saint George Respiratory Questionnaire (SGRQ) (Camelier et al., 2006). This is a self-administered questionnaire that comprises three domains: 1) “Symptoms”: which provided information about discomfort due to respiratory symptoms; 2) “Activities”: which assessed changes in physical activity; and 3) “Impact”: which assessed the global impact on the patient’s activities of daily living and well-being. In addition, the total score was also quantified. The results were expressed in percentages ranging from 0 to 100%, a higher score meaning worse performance in each domain.
Functional Capacity
The functional capacity was assessed by the six-minute walk test (6MWT). The participants were requested to wear appropriate clothing (sportswear and trainers). Two tests were performed with a 30-minute interval and executed by two physical therapists. The repetition of two tests aimed at eliminating the learning effect and ensuring the reproducibility of the procedure (Hernandes et al., 2011). The best test was used in the data. The volunteers were instructed to walk at maximum speed as possible during the six minutes. During the walk, the volunteers were encouraged by the examiner through verbal stimuli at every minute, using the phrases recommended by the American Thoracic Society (ATS, 2002). The distance covered in meters was recorded at the end of the test. The blood pressure (BP), heart rate (HR), respiratory rate (RR), peripheral oxygen saturation (SpO2), and dyspnea and lower-limb (LL) fatigue score using the CR10 Borg scale were recorded at the beginning, during and at the end of the 6MWT. In case of desaturation (SpO2<90%), the test was interrupted, oxygen supplementation was provided, and another test was started (Borghi-Silva et al., 2010).
Functional status
The functional status was assessed using the modified version of the Pulmonary Functional Status and Dyspnea Questionnaire (PFSDQ-M). The PFSDQ-M consists of three domains: 1) influence of dyspnea on ADL, 2) influence of fatigue on ADL (5 general items and 10 specific items for each domain); and 3) change in ADL compared to the period prior to the disease (10 specific items). There are five general questions in the fatigue and dyspnea domains, which are informative and qualitative in nature and are not added to the questionnaire score. The participants describe how much dyspnea and fatigue interfere with the ten specific ADL items, scoring between 0 and 10 for each activity, being 0 (no interference), 1-3 (mild), 4-6 (moderate), 7-9 (severe) and 10 (very severe). In the third domain, the amount of change in ADL is reported in comparison to the period prior to the disease, choosing a value between 0-10 for each activity with 0 being as active as ever regarding this activity; 1-3 for small changes; 4-6 for moderate changes; 7-9 for extreme changes; and 10 for when no longer doing this activity. Subsequently, a partial score is calculated, ranging from 0 to 100 for each domain and a total score, which is composed of the sum of the partial scores of the three domains, ranging from 0 to 300. Higher values on the scale indicate greater limitations in ADL (Kovelis et al., 2008). A minimum change of 5 points in each component was considered as the minimum clinically important difference (MCID) (Regueiro et al., 2013).
London Chest Activity of Daily Living Scale
The level of dyspnea during the activities of daily living was assessed by the London Chest Activity of Daily Living Scale (LCADL), which is composed of four domains: 1) personal care, 2) domestic activities, 3) physical activities and 4) leisure activities, which includes 15 questions. The domains present items that receive a score ranging from 0 to 5, totaling a maximum score of 75 points, with higher values on the scale indicating greater limitations in ADL (Carpes et al., 2008). A minimum of 4 points change in the total score of the scale was considered as the MCID (Bisca et al., 2014).
Dyspnea
The dyspnea was also assessed using the Brazilian version of the Medical Research Council (MRC) Dyspnea Scale, which comprises five statements, with the patient reporting their subjective degree of dyspnea corresponding to how much it interferes with ADL as 1 (“only suffers from shortness of breath during intense exercise”); 2 (“suffers from shortness of breath when walking quickly or climbing a light slope”); 3 (“walks slower than people of the same age because of shortness of breath or has to stop to breathe even when walking slowly”); 4 (“stops walking to breathe after walking less than 100m or after a few minutes”); and 5 (“feels so short of breath that they no longer leave the house or when getting dressed”) (Kovelis et al., 2008).
BODE index
The BODE (B-body mass index; O-airflow obstruction; D-dyspnea; E-exercise capacity) mortality predictor index assesses the risk of mortality using the body mass index (BMI), FEV1, MRC scale score and the distance covered in the 6MWT. The score in the BODE index ranges from 0 to 10, with a higher score indicating greater severity (Celli et al., 2004).
Intermittent aquatic aerobic training protocol
The 8-week intermittent aquatic aerobic training, lasting 60 minutes, on alternate days, three times a week (American College of Sports Medicine Position Stand [ACSM], 1998; Nici et al., 2006) was performed based on a previously described protocol (Kim et al., 2010), adapted to the patients, which consisted of: 1) warm-up (10 minutes): stretching exercises and dynamic aerobic exercises; 2) aquatic physical conditioning: aerobic training consisting of aquatic exercises for the trunk, upper limbs (UL) and lower limbs (LL), with intensity prescribed at 4-6 on the CR10 Borg scale (Carvalho et al., 2009; Graef & Kruel, 2006; Horowitz et al., 1996), performed in an intermittent form, 1 minute of exercise and 1 minute of rest progressively increasing (Vogiatzis et al., 2002). The first eight sessions lasted 20 minutes, then the next eight sessions lasted 30 minutes and the last eight sessions lasted 40 minutes. 3) Cooling down (10 min): global stretching exercises. This training program was developed in a heated pool (32ºC). During all sessions, the patients were instructed to use pursed-lip breathing. Supplementary oxygen was offered if SpO2 levels were below 90%, during the exercise (Borghi-Silva et al., 2010; Moga, 2012). Prior to therapy, BP, HR, RR, SpO2, Borg dyspnea and Borg LL were measured and recorded. The sessions were carried out individually, and HR was monitored every 10 minutes of therapy using a FT1 model frequency meter (Polar® Electro Co.Ltda. Kempele, Oulu, Finland), SpO2 was measured by the wrist oximeter ONYX 9500 (Nonin®), and the subjective perception of the respiratory effort and LL effort by the Borg scale.
Statistical analysis
The data were analyzed using the SPSS 25.0 software (IBM, Amonk, USA). Normality of data distribution and homogeneity of variance were assessed using Shapiro-Wilk and Levene’s tests, respectively. A mixed twoway analysis of variance (ANOVA) was used to assess Group (CG vs ATG) and Time (Baseline vs Post) interaction. When interaction was significant, the Bonferroni’s post hoc pairwise comparison was performed, and the main effects results were disregarded. Partial eta squared (η²ₚ) was calculated to quantify the effect size for the ANOVA and interpreted as small (0.01), medium (0.06), or large (0.14). To assess the effect size for pairwise comparisons, Cohen’s d was calculated and classified as a small (0.2), medium (0.5), or large (0.8) effect, according to Cohen (1988). The significance level was established at 5% for all the analyses. Moreover, we analyzed the individual responses of each participant based on the available MCID values established in previous studies (Bisca et al., 2014; Regueiro et al., 2013).
3. Results
Table 1 presents the baseline values for the 2 studied groups. There were no significant differences observed between the groups (p>0.05). Table 2 presents the values for the SGRQ, LCADL, PFSDQ-M, BODE index, and MRC before and after intervention for the two studied groups (Gallo-Silva et al., 2025).
Clinical and demographic characteristics of both groups
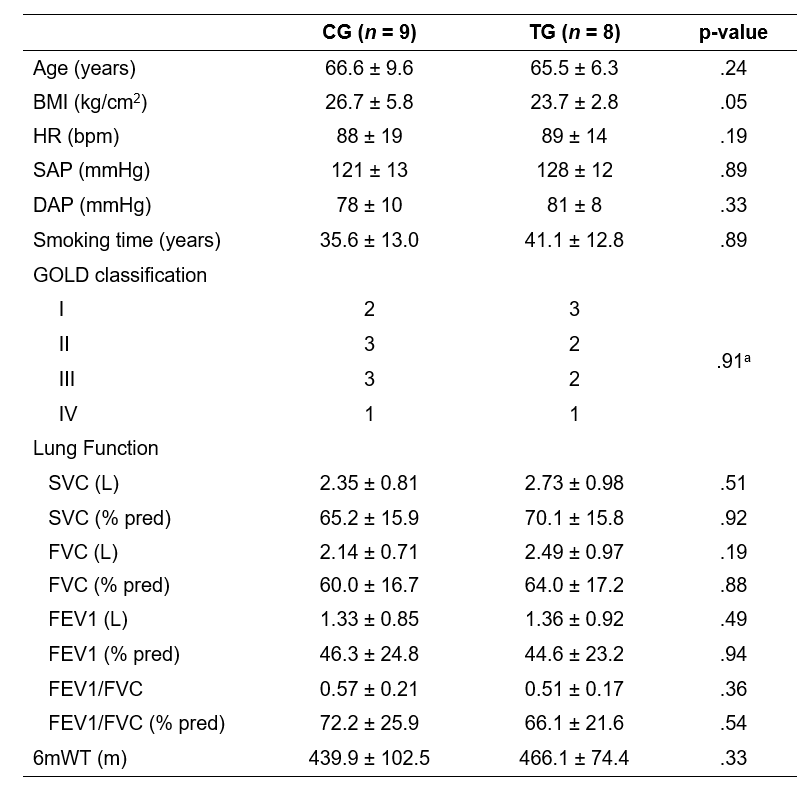
Source: the authors.Note. BMI (Body mass index); HR (Heart rate); SAP (systolic arterial pressure); DAP (diastolic arterial pressure); SVC (slow vital capacity); FVC (forced vital capacity); FEV1 (forced expiratory volume in first second); GOLD (Global Initiative for Lung Obstructive Pulmonary Disease); 6mWT (six-minute walk test). aFisher’s exact test.
Pre- and post-training comparison related to quality of life, LCADL, PFSDQ M, BODE index, and dyspnea
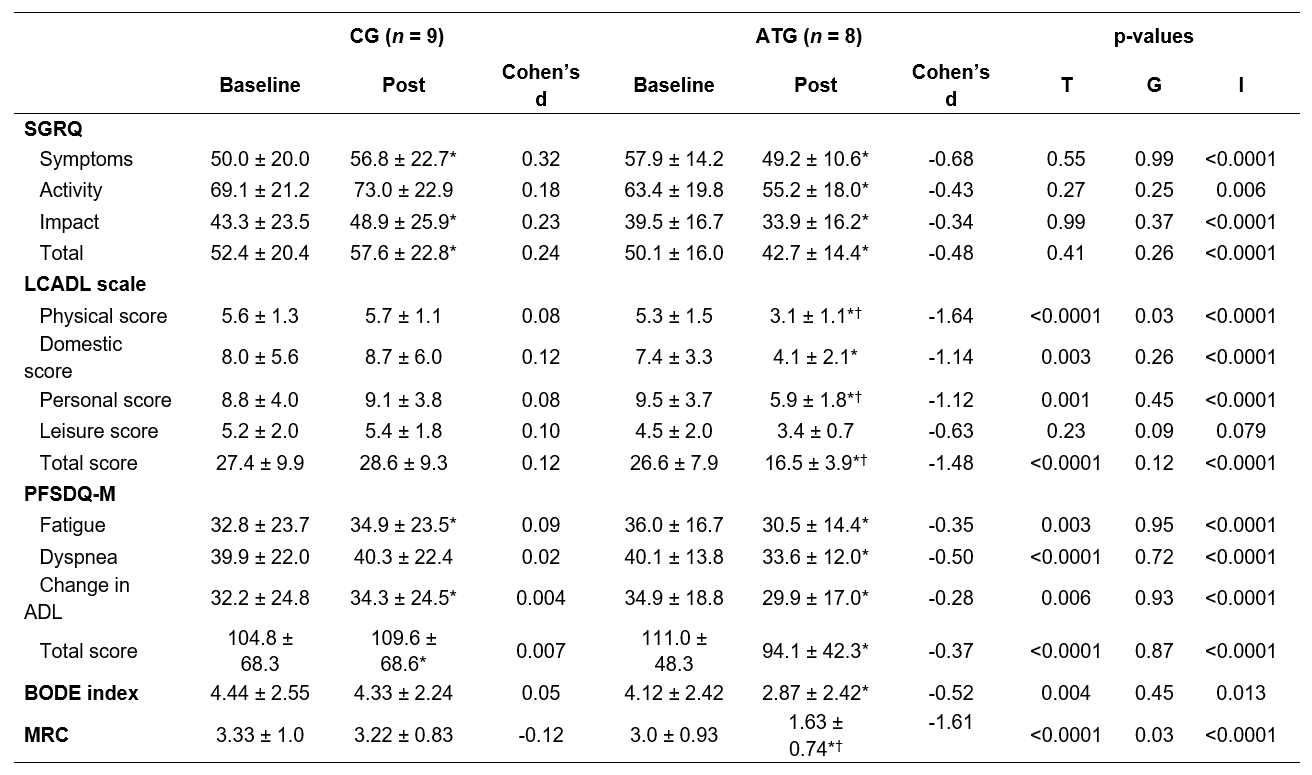
Source: the authors.Note. *p < .05 vs Baseline; †p < .05 vs CG Post; #p < .05 vs Δ CG. SGRQ (Saint George Respiratory Questionnaire), LCADL (London Chest Activity Daily Living), PFSDQ-M (Modified Pulmonary Functional Status and Dyspnea Questionnaire), BODE (B-body mass index; O- airflow obstruction; D-dyspnea; E-exercise capacity), and MRC (Medical Research Council).
Quality of Life
Regarding the quality of life assessed with the SGRQ (Table 2), a significant Group*Time interaction was found for Symptoms (F1,15 = 23.1; p<0.0001, ηp2 = 0.61), Activity (F1,15 = 10.3; p=0.006, ηp2 = 0.41) and Impact (F1,15 = 21.1; p<0.0001, ηp2 = 0.59) domains, as well as for the Total score (F1,15 = 20.8; p<0.0001, ηp2 = 0.58). Pairwise comparisons revealed that ATG significantly improved quality of life (total score, d= 0.48, p=0.02) and its domains, Symptoms (d=‑0.68, p=0.01), Activity (d=-0.43, p= 0.01) and Impact (d=-0.34, p=0.006). On the other hand, the CG experienced a worsening in overall quality of life (d=0.24, p=0.02), and in the domains Symptoms (d=0.32, p=0.01) and Impact (d=0.23, p=0.004).
Dyspnea during activities of daily living
Regarding the level of dyspnea during activities of daily living, as assessed by LCADL (Table 2), a significant Group*Time interaction was found for the Total Score (F1,15 = 41.4; p<0.0001, ηp2 = 0.73) and the domains Physical (F1,15 = 32.5; p<0.0001, ηp2 = 0.68), Domestic (F1,15 = 29.1; p<0.0001, ηp2 = 0.66) and Personal (F1,15 = 26.3; p<0.0001, ηp2 = 0.64). No significant interaction or main effects were observed for the Leisure domain (F1,15 = 3.8; p=0.08, ηp2 = 0.19). Within group comparisons showed that ATG significantly improved total score (d=-1.48, p<0.0001), physical score (d=-1.64, p<0.0001), domestic score (d=-1.14, p<0.0001) and personal score (d =-1.12, p<0.0001) after the intervention. On the other hand, the CG did not present any significant changes (p>0.05). In the between group comparison, the ATG presented significantly lower values post intervention for the scores in the physical domain (d=-2.36, p<0.0001), personal domain (d=-1.08, p=0.04) and total score (d=-1.70, p=0.004), indicating an improvement in dyspnea in relation to the CG (Table 2).
Pulmonary functional status
Regarding pulmonary functional status, assessed by the PFSDQ-M (Table 2), a significant Group*Time interaction was found for Total Score (F1,15 = 82.2; p<0.0001, ηp2 = 0.81) and the domains Fatigue (F1,15 = 62.5; p<0.0001, ηp2 = 0.81), Dyspnea (F1,15 = 39.2; p<0.0001, ηp2 = 0.72) and Change in ADL (F1,15 = 63.1; p<0.0001, ηp2 = 0.81). Within group comparisons showed a significant difference in the total score (d=0.37, p<0.0001) and in all domains after the aquatic intervention, indicating an improvement in the impact of fatigue (d=-0.35, p<0.0001) and dyspnea (d=-0.5, p<0.0001) in activities of daily living, as well as in the perception of change in ADL (d=-0.28, p<0.04). On the other hand, the CG presented a significant increase, although with small effect on the total score (d=0.09, p=0.01), impact of fatigue (d=0.09, p=0.01) on ADL and the perception of change in ADL (d=0.004, p=0.004). There were no significant differences between groups for any of the variables concerning the PFSDQ-M (p > 0.05) (Table 2).
BODE index
A significant Group*Time interaction was also found for the BODE index (F1,15 = 7.93; p=0.01, ηp2 = 0.35). After the training, the ATG presented a significant improvement in the BODE index (d= 0.52, p=0.001), which was not observed in the CG (d=0.05, p=0.69). No significant difference was found for the between group comparisons (p>0.05).
Medical Research Council Dyspnea Scale
Finally, a significant Group*Time interaction was found for the MRC (F1,15 = 21.3; p<0.0001, ηp2 = 0.59, table 2). The ATG showed a significant improvement in dyspnea (d=-0.50, p<0.0001), assessed by the MRC, while no significant difference (d=-0.12, p=0.56) could be observed for the CG. Furthermore, the dyspnea observed in the ATG in the post-treatment period was significantly lower when compared to the dyspnea in the CG (d= 2.02, p=0.001, table 2).
Clinical improvement
Additionally, an individual analysis was carried out within each group to identify patients who achieved a clinically relevant improvement in the variables that presented MCID values reported in literature (Schünemann et al., 2003) (Figure 2). It was possible to identify clinically relevant improvement in 75% (6/8) of ATG patients in relation to the dyspnea domain of the PFSDQ-M; 50% (4/8) improved the fatigue and changes in ADL domains of the PFSDQ-M; and 87.5% improved the total LCADL score, indicating an improvement in dyspnea in ADL. In addition, patients classified as GOLD stages III and IV achieved the MCID across all the main outcomes (Figure 2).
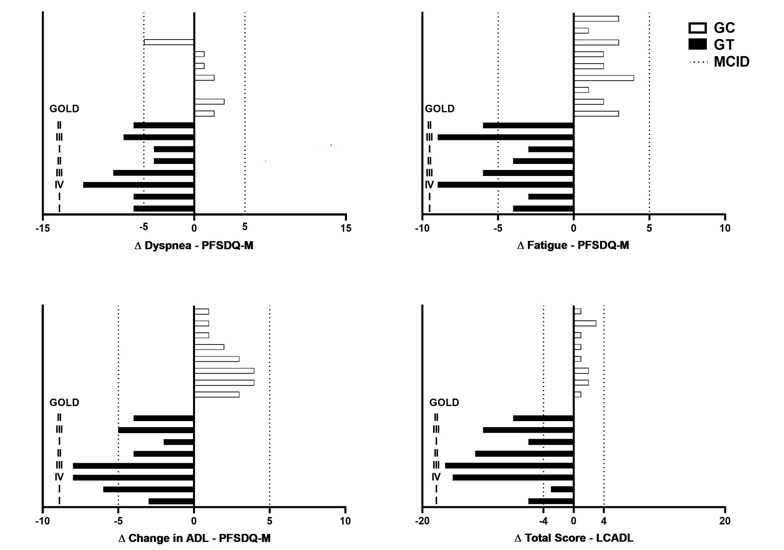
Figure 2.
Minimum clinically significant difference (MCID) delta chart. PFSDQ-M: Modified Pulmonary Functional Status and Dyspnea Questionnaire, SGRQ (Saint George Respiratory Questionnaire), LCADL (London Chest Activity Daily Living), BODE (B-body mass index; O- airflow obstruction; D-dyspnea; E-exercise capacity), and MRC (Medical Research Council).
Source: the authors.
4. Discussion
This study evaluated the effects of an aquatic intermittent aerobic training protocol in patients with COPD. The results show that the ATG presented significant improvements in quality of life, functional status in the activities of daily living, in the BODE index, and in general dyspnea, and during physical, domestic and personal activities, except for dyspnea in leisure activities. On the other hand, the control group, which maintained the drug treatment and received guidance on healthy lifestyle habits but did not participate in the supervised rehabilitation program during this period, showed a significant worsening of quality of life, fatigue and functional status.
These results are in accordance with the findings of previous studies. Wadell et al. (2004) proposed a 12-week program, three times a week for 45 minutes, of high-intensity aquatic training for patients with COPD. After training, the patients improved the activity component of the SGRQ. In the same sense, Felcar et al. (2018) evaluated the effects of a 6-month program (60 sessions) of water exercises compared to a land-based exercise program in patients with COPD. At the end of the program, both groups improved inspiratory, expiratory and peripheral muscle strength, as well as quality of life and functional status. Similar results were also observed by Mc Namara et al. (2013) and Lotshaw et al. (2007). Both authors observed a significant improvement in quality of life in COPD patients after an aquatic training program. This is extremely important, as the frequency of exacerbations in COPD is associated with impaired quality of life, decline in lung function and increased mortality (Ritchie & Wedzicha, 2020).
Another important finding of this study is the improvement in the level of dyspnea in activities of daily living, except for leisure activities, assessed by the LCADL scale. These results partially corroborate the findings reported by Felcar et al. (2018), who observed improvements in all domains of the LCADL, except for the physical activity domain, in the group that underwent aquatic training. Furthermore, the current study also showed an improvement in all components of the PFSDQ-M scale, indicating a significant improvement in functional status.
Those findings are in accordance with the results obtained by Probst et al. (2011) who also observed an improvement in the functional status in patients with COPD after a training program on the land. In addition, it is important to emphasize that most patients presented an improvement of at least 5 units in each component, reaching the MCID for this outcome, while only 50% of the participants achieved the MCID for fatigue and activities of daily living. Improvement in dyspnea may be explained by the positive physiological adaptations induced by exercise training. It has been shown that the ventilatory requirements during submaximal exertion are reduced after an exercise program, by enhancing the aerobic capacity of skeletal muscles. In addition, exercise decreases blood lactate concentration and increases muscle mechanical efficiency, both of which may contribute to lower perception of dyspnea (Casaburi, 2001). On the other hand, activities of daily living and fatigue involve a more complex interplay between physiological systems, and the aquatic environment may not sufficiently replicate the demands of these activities in a land-based context. This could limit the transferability of improvements observed in water to daily life functioning (Benzo-Iglesias et al., 2023). Even so, the ATG showed a significant improvement in the BODE index, a predictor of mortality, with a moderate effect size, which is notable for this study population. These findings corroborate the results observed by de Souto Araujo et al. (2012). The authors observed a decrease in the BODE index after low-intensity training on the land and in water.
Another point worth mentioning regards the differences between responders and non-responders to the protocol, as illustrated in Figure 2. Our results showed that patients classified as GOLD stages III and IV achieved the MCID across all the main outcomes, whereas patients in stages I and II showed improvements in selected variables. This finding not unexpected and may be attributed to a “floor effect”, wherein patients with more severe baseline impairments have greater potential for improvement than patients with less compromise. These results are in accordance with previous studies reporting that individuals with higher levels of fatigue and poorer quality of life at baseline were more likely to respond favorably to rehabilitation programs (Baltzan et al., 2011, Van Herck et al., 2019).
The results of the study suggest that the intermittent training protocol in an aquatic environment is an effective therapeutic option for patients with COPD. In addition to the benefits of physical exercise itself, the physical properties of water can be highlighted among the possible mechanisms for the positive effects of aquatic training for this population. In this sense, hydrostatic pressure acts on the body and adjusts the position of the diaphragm muscle to a high and effective level. Thus, hydrostatic pressure exerts resistance to diaphragmatic contraction during inspiration, and facilitates expiration, consequently reducing the residual volume during immersion (Anstey & Roskell, 2000; Chen et al., 2021; Wadell et al., 2004). It is also observed that the increased capillarization of trained muscles reduces respiratory demand and dyspnea (Broman et al., 2006). This mechanism may have played an important role in reducing the sensation of dyspnea observed in this study. Buoyancy is another physical property worth mentioning. Buoyancy is the force that acts in the opposite direction to gravity, favoring flotation, reducing joint overload and facilitating the execution of movements against gravity. Therefore, the aquatic environment becomes suitable for this population, since, in addition to reducing the risk of falls in the elderly, it prevents impacts resulting from falls, which can lead to mild abrasions, mobility restrictions, limitation of activities of daily living, and loss of functional independence, which may compromise the quality of life of patients with COPD (Resende & Rassi, 2008).
Study limitations
Despite the promising results, the small size of the sample should be highlighted as a limitation of the study. Nevertheless, the analyses of the results were complemented by the effect size and the individual analysis in relation to the MDC, which are not size dependent. Furthermore, the inclusion of a group performing the same protocol on the land can also be seen as a limitation, as it would allow comparing the real advantages of training in the aquatic environment.
5. Conclusions
This study demonstrated that the intermittent aquatic aerobic training of moderate/high intensity promoted a significant and clinically relevant improvement for most patients with COPD in their quality of life, in the limitations of their activities of daily living, dyspnea, and in the BODE mortality predictor index.
6. References
Adolfo, J. R., Dhein, W., & Sbruzzi, G. (2019). Intensity of physical exercise and its effect on functional capacity in COPD: systematic review and meta-analysis. Jornal Brasileiro de Pneumologia, 45(6), e20180011. https://doi.org/10.1590/1806-3713/e20180011
American College of Sports Medicine Position Stand [ACSM]. (1998). The recommended quantity and quality of exercise for developing and maintaining cardiorespiratory and muscular fitness, and flexibility in healthy adults. Medicine and science in sports and exercise, 30(6), 975-991. https://doi.org/10.1097/00005768-199806000-00032
American Thoracic Society. (2002). ATS statement: guidelines for the six-minute walk test. American Journal of Respiratory and Critical Care Medicine, 166(1), 111-117. https://doi.org/10.1164/ajrccm.166.1.at1102
Anstey, K. H., & Roskell, C. (2000). Hydrotherapy: Detrimental or beneficial to the respiratory system? Physiotherapy, 86(1), 5-13. http://dx.doi.org/10.1016/S0031-9406(05)61320-X
Baltzan, M. A., Scott, A. S., Wolkove, N., Bailes. S., Bernard, S., Bourbeau, J., Maltais F. & Canadian COPD Pulmonary Rehabilitation Research Group (2011). Fatigue in COPD: prevalence and effect on outcomes in pulmonar rehabilitation. Chronic Respiratory Disease, 8(2),119-128. https://doi.org/10.1177/1479972310396737
Benzo-Iglesias, M. J., Rocamora-Pérez, P., Valverde-Martínez, M. A., Garcia-Luengo, A. V., & Lopes-Lira R. (2023). Effectiveness of water-Based exercise in patients with chronic obstructive pulmonar disease: systematic review and meta-analysis. Sensors, 23(20), 8557. https://doi.org/10.3390/s23208557
Bisca, G. W., Proença, M., Salomão, A., Hernandes, N. A., & Pitta, F. (2014). Minimal detectable change of the London chest activity of daily living scale in patients with COPD. Journal of Cardiopulmonary Rehabilitation and Prevention, 34(3), 213-216. https://doi.org/10.1097/hcr.0000000000000047
Borghi-Silva, A., Mendes, R.G., Toledo, A. C., Sampaio, L. M. M., da Silva, T. P., Kunikushita, L. N., Sousa, H. C. D, Salvini, T. F., & Costa, D. (2010). Adjuncts to physical training of patients with severe COPD: oxygen or noninvasive ventilation? American Journal of Respiratory and Critical Care Medicine, 181(7), 885-894. https://doi.org/10.1164/ajrccm-conference.2010.181.1_meetingabstracts.a6502
Broman, G., Quintana, M., Lindberg, T., Jansson, E., & Kaijser, L. (2006). High intensity deep water training can improve aerobic power in elderly women. European Journal Applied Physiolology, 98, 117-123. https://doi.org/10.1007/s00421-006-0237-2
Camelier, A., Warken Rosa, F., Salmi, C., Nascimento, O.A, Cardoso, F., & Jardim, J.R. (2006). Avaliação da qualidade de vida pelo questionário do hospital Saint George na doença respiratória em portadores de doença pulmonar obstrutiva crônica: validação de uma nova versão para o Brasil. Jornal Brasileiro Pneumologia, 32(2), 114-122. https://doi.org/10.1590/S1806-37132006000200006
Carpes, M. F., Mayer, A. F., Simon, K. M., Jardim, J. R., & Garrod, R. (2008). Versão brasileira da escala London Chest Activity of Daily Living para uso em pacientes com doença pulmonar obstrutiva crônica. Jornal Brasileiro de Pneumologia, 34(3), 143-151. https://doi.org/10.1590/s1806-37132008000300004
Carvalho, V. O., Boccchi, E. A., & Guimarães, G. V. (2009). The borg scale as an important tool of self-monitoring and self-regulation of exercise prescription in heart failure patients during hydrotherapy. Circulation Journal, 73(10), 1871-1876. https://doi.org/10.1253/circj.cj-09-0333
Casaburi, R. (2001). Skeletal muscle dysfunction in chronic obstructive pulmonary disease. Medicine Science Sports Exercise, 33(7), S662-S670. https://doi.org/10.1097/00005768-200107001-00004
Celli, B. R., Cote, C. G., Marin, J. N., Casanova, C., Montes de Oca, M., Mendez, R. A., Pinto Plata, V., & Cabral, H. J. (2004). The body-mass index, airflow obstruction, dyspnea, and exercise capacity index in chronic obstructive pulmonary disease. New England Journal of Medicine, 350(10), 1005-1012. https://doi.org/10.1056/nejmoa021322
Chen, H., Li, P., Li, N., Wang, Z., Wu, W., & Wang, J. (2021). Rehabilitation effects of land and water based aerobic exercise on lung function, dyspnea, and exercise capacity in patients with chronic obstructive pulmonary disease A systematic review and meta-analysis. Medicine, 100(33), e26976. https://doi.org/10.1097/md.0000000000026976
Cohen, J. (1988). Statistical Power Analysis for the Behavioral Sciences. (2nd ed.). Routledge. https://doi.org/10.4324/9780203771587
Cote, C. G., & Celli, B. R. (2005). Pulmonary rehabilitation and the BODE index in COPD. European Respiratory Journal, 26(4), 630-636. https://doi.org/10.1183/09031936.05.00045505
Craig, C. L., Marshall, A. L., Sjöström, M., Baumann, A. E., Booth, M. L., Ainsworth, B. E., Pratt, M., Ekelund, U., Yngve, A., Sallis, J., & Oja, P. (2003). International physical activity questionnaire: 12-country reliability and validity. Medicine Science Sports Exercise, 35(8), 1381–1395. https://doi.org/10.1249/01.mss.0000078924.61453.fb
de Souto Araujo, Z. T., de Miranda Silva Nogueira, P. A., Alves Cabral, E. E., de Paula dos Santos, L., Soares da Silva, I., & Holanda Ferreira, G. M. (2012). Effectiveness of low-intensity aquatic exercise on COPD: A randomized clinical trial. Respiratory Medicine, 106(11), 1535-1543. https://doi.org/10.1016/j.rmed.2012.06.022
Felcar, J. M., Probst, V. S., de Carvalho, D. R., Merli, M. F., Mesquita, R., Vidotto, L. S., Ribeiro, L. R. G. & Pitta, F. (2018). Effects of exercise training in water and on land in patients with COPD: a randomised clinical trial. Physiotherapy, 104(4), 408-416. https://doi.org/10.1016/j.physio.2017.10.009
Gallo-Silva, B., Zamunér, A. R., Cerezer-Silva, V., Gullo Ferreira, D., Sakabe, D. I., Kel de Souza, L. D., Bertholo, V. C., Ferreira Brasil, M. T., & Moreno, M. A. (2025). Data base of Benefits of hydrotherapy in patients with chronic obstructive pulmonary disease: additional results from a randomized controlled trial. Pensar en Movimiento: Revista de Ciencias del Ejercicio y la Salud, 25(1). https://doi.org/10.15517/pensarmov.v23i1.65042
Garrod, R., Bestall, J. C., Paul, E. A., Wedzicha, J. A., & Jomes, P. W. (2000). Development and validation of a standardized measure of activity of daily living in patients with severe COPD: the London Chest Activity of Daily Living scale (LCADL). Respiratory Medicine, 94(6), 589-596. https://doi.org/10.1053/rmed.2000.0786
Global Initiative for Chronic Obstructive Lung Disease (GOLD). (2022). Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. https://goldcopd.org
Graef, F. I., & Kruel, F. M. (2006). Frequência cardíaca e percepção subjetiva do esforço no meio aquático: diferenças em relação ao meio terrestre e aplicações na prescrição do exercício-uma revisão. Revista Brasileira de Medicina do Esporte, 12(4), 228-232. https://doi.org/10.1590/S1517-86922006000400011
Hernandes, N. A., Wouters, E. F. M., Meijer, K., Annegarn, J., Pitta, F., & Spruit, M. A. (2011). Reproducibility of 6-minute walking test in patients with COPD. European Respiratory Journal, 38(2), 261-267. https://doi.org/10.1183/09031936.00142010
Horowitz, M. B., Littenberg, B., & Mahler, D.A. (1996). Dyspnea ratings for prescribing exercise intensity in patients with COPD. Chest, 109(5), 1169-1175. https://doi.org/10.1378/chest.109.5.1169
Janssens, M. A. T., De Peuter, S., Stans, L., Verleden, G., Troosters, T., Decramer, M., & Van den Bergh, O. (2011). Dyspnea Perception in COPD Association between anxiety, dyspnea-related fear, and dyspnea in a pulmonary rehabilitation program. Chest, 140(3), 618-625. https://doi.org/10.1378/chest.10-3257
Kim, I. S., Chung, S. H., Park, Y. J., & Kang, H. Y. (2010). The effectiveness of an aquarobic exercise program for patients with osteoarthritis. Applied Nursing Research, 25(3),181-189. https://doi.org/10.1016/j.apnr.2010.10.001
Kocks, J. W. H., Asijee, G. M., Tsiligianni, I. G., Kerstjens, H. A., & van der Molen, T. (2011). Functional status measurement in COPD: a review of available methods and their feasibility in primary care. Primary Care Respiratory Journal, 20, 269-275. https://doi.org/10.4104/pcrj.2011.00031
Kovelis, D., Oldemberg Segretti, N., Probst, V.S., Lareau. S.C., Brunetto, A. F., & Pitta, F. (2008). Validação do Modified Pulmonary Functional Status and Dyspnea Questionnaire e da escala do Medical Research Council para o uso em pacientes com doença pulmonar obstrutiva crônica no Brasil. Jornal Brasileiro de Pneumologia, 34(12), 1008-1018. https://doi.org/10.1590/s1806-37132008001200005
Lareau, S. C., Meek, P.M., & Roos, P. J. (1998). Development and Testing of a Modified Version of Pulmonary Functional Status & Dyspnea Questionnaire (PFSDQ-M). Heart & Lung, 27(3), 159-168. https://doi.org/10.1016/s0147-9563(98)90003-6
Lotshaw, A. M., Thompson, M., Sadowsky, H.S., Hart, M.K., & Millard, M.W. (2007). Quality of life and physical performance in land and water based pulmonary rehabilitation. Journal of Cardiopulmonary Rehabilitation Prevention, 27(4), 247-251. https://doi.org/10.1097/01.hcr.0000281772.28394.30
McCarthy, B., Casey, D., Devane, D., Murphy, K., Murphy, E., & Lacasse, Y. (2015). Pulmonary rehabilitation for chronic obstructive pulmonary disease. Cochrane Database Systematic Reviews, 2015(2), CD003793. https://doi.org/10.1002/14651858.CD003793.pub3
Mc Namara, R. J, Mc Keough, Z. J., Mc Kenzie, D. K., & Alison, J. A. (2013). Water-based exercise in COPD with comorbidities: a randomised controlled trial. European Respiratory Journal, 41(6),1284-1291. https://doi.org/10.1183/09031936.00034312
Miller, M. R., Hankinson, J., Brusasco, V., Burgos, F., Casaburi, R., Coates, A., Crapo, R., Enright, P., van der Grinten, C. P. M., Gustafsson, P., Jensen, R., Johnson, D. C., Macintyre, N., McKay, R., Navajas, D., Pedersen, O.F., Pellegrino, R., Viegi, G., & Wanger, J. (2005). Standardisation of spirometry. European Respiratory Journal, 26(2), 319-338. https://doi.org/10.1183/09031936.05.00034805
Moga, A. M., de Marchie, M., Saey, D., & Spahija, J. (2012). Mechanisms of non-pharmacologic adjunct therapies used during exercise in COPD. Respiratory Medicine, 106(5), 614-626. https://doi.org/10.1016/j.rmed.2012.01.006
Nici, L., Donner, C., Wouters, E., Zuwallack, R., Ambrosino, N., Bourbeau, J., Carone, M., Celli, B., Engelen, M., Fahy, B., Garvey, C., Goldstein, R., Gosselink, R., Lareau, S., MacIntyre, N, Maltais, F., Morgan, M., O´Donnell, D., Prefault, C., Reardon, J., Rochester, C., Schols, A., Singh, S., & Troosters, T. (2006). American Throracic Society/ European Respiratory Society Statement on Pulmonary Rehabilitation. American Journal of Respiratory Critical Care Medicine, 173(12), 1390-1413. https://doi.org/10.1164/rccm.200508-1211st
Pereira, C. A., Sato, T., & Rodrigues, S. C. (2007). Novos valores de referência para espirometria forçada em brasileiros adultos de raça branca. Jornal Brasileiro de Pneumologia, 33(4), 397-406. https://doi.org/10.1590/s1806-37132007000400008
Probst, V. S., Kovelis, D., Hernandes, N. A., Camillo, C. A., Cavalheri, V., & Pitta, F. (2011). Effects of 2 Exercise Training Programs on Physical Activity in Daily Life in Patients With COPD. Respiratory Care, 56(11), 1799-1807. https://doi.org/10.4187/respcare.01110
Regueiro, E. M. G., Burtin, C., Baten, P., Langer, D., Van Remoortel, H., Pires Di Lorenzo, V. A., Costa, D., Janssens, W., Decramer, M., Gosselink, R., & Troosters, T. (2013). The minimal important difference of the pulmonary functional and dyspnea questionnaire in patients with severe chronic obstructive pulmonary disease. Respiratory Research, 14(58). https://doi.org/10.1186/1465-9921-14-58
Resende, S. M., & Rassi, C. M. (2008). Efeitos da hidroterapia na recuperação do equilíbrio e prevenção de quedas em idosas. Revista Brasileira de Fisioterapia, 12(1), 57-63. https://doi.org/10.1590/S1413-35552008000100011
Ritchie, A. I., & Wedzicha, J. A. (2020). Definitions, causes, pathogenesis, and consequences of chronic obstructive pulmonary disease exacerbations. Clinics in Chest Medicine, 41(3),421-438. https://doi.org/10.1016/j.ccm.2020.06.007
Schünemann, H. J., Griffith, L., Jaeschke, R., Goldstein, R., Stubbing, D., & Guyatt, G.H. (2003). Evaluation of the minimal important difference for the feeling thermometer and the St. George’s Respiratory Questionnaire in patients with chronic airflow obstruction, Journal of Clinical Epidemiology, 56(12),1170-1176. https://doi.org/10.1016/S0895-4356(03)00115-X
Van Herck, M., Antons, J., Vercoulen, J., Goërtz, Y., Ebadi, Z, Burtin, C., Janssen, D., Thong, M., Otker, J., Coors, A., Sprangers., M., Muris, J., Wouters, E., Prins, J., Spruit, M., & Peters, J. (2019). Pulmonary rehabilitation reduces subjective fatigue in COPD: a responder analysis. European Respiratory Journal, 54(63), PA573. https://doi.org/10.1183/13993003.congress-2019.PA573
Vogiatzis, I., Nanas, S., & Roussos, C. (2002). Interval training as an alternative modality to continuous exercise in patients with COPD. European Respiratory Journal, 20(1),12-19. https://doi.org/10.1183/09031936.02.01152001
Wadell, K., Sundelin, G., Henriksson-Larsén, K., & Lundgren, R. (2004). High intensity physical group training in water—an effective training modality for patients with COPD. Respiratory Medicine, 98(5),428-438. https://doi.org/10.1016/j.rmed.2003.11.010
Zampogna, E., Ferriero, G., Visca, D., Patrini, M., Negrini, S., & Arienti, C. (2023). An overview of Cochrane systematic reviews for pulmonar rehabilitation interventions in people with chronic obstructive pulmonary disease: a mapping synthesis. Panminerva Medica, 65(2), 234-243. https://doi.org/10.23736/s0031-0808.22.04757-7
Notes
Notas de autor
Información adicional
Associate editor in charge:: Ph.D Pedro Carazo Vargas
Información adicional
redalyc-journal-id: 4420
