ARTICLES
Microbiology quality, detection of enterotoxin genes and antimicrobial resistance of Staphylococcus aureus isolated from milk and Coalho cheese
Qualidade microbiológica, detecção de genes de enterotoxinas e resistência antimicrobiana de Staphylococcus aureus isolados de leite e queijo de Coalho
Microbiology quality, detection of enterotoxin genes and antimicrobial resistance of Staphylococcus aureus isolated from milk and Coalho cheese
Semina: Ciências Agrárias, vol. 39, no. 5, pp. 1957-1967, 2018
Universidade Estadual de Londrina
Received: 30 November 2017
Accepted: 27 April 2018
Abstract: This study evaluated the microbiological quality of milk and Coalho cheese, the prevalence of enterotoxin genes, antimicrobial resistance and determined an inducible MLSB resistance phenotype by the D-test in strains of Staphylococcus aureus isolated from these products. Seventy samples of milk and Coalho cheese were analyzed. S. aureus strains were identified by biochemical tests. The presence of se genes (sea-see) was tested by polymerase chain reaction. The antimicrobial sensitivity of S. aureus strains was evaluated for 13 antimicrobial drugs using the disk diffusion technique and the double-disk diffusion test (D-test) was performed to determine inducible resistance to lincosamide phenotype. The amount of toxin sufficient to cause foodborne diseases is generally observed when Staphylococcus populations exceed 105 CFU mL-1 g-1. In this study, none of the milk samples analyzed showed these counts; however, 73.3% (22/30) of Coalho cheese samples exceeded this value. A total of 109 isolates were identified as S. aureus. The presence of enterotoxin genes was detected in 25.7% of these isolates and amplified only for the sec gene. Most of the isolates (78.5%) were resistant to one or more antimicrobial agents. The D test showed that 25.0% of erythromycin-resistant isolates had the constitutive resistance phenotype, and 3.8% had the inducible resistance phenotype to clindamycin. These results indicate that these dairy products represent a health risk since these bacteria can cause foodborne diseases or may be a possible route for the transfer of antimicrobial resistance to humans.
Key words: Antimicrobial resistance, Dairy products, Coalho cheese, Staphylococcal enterotoxins genes, Staphylococcus aureus.
Resumo: Este estudo avaliou a qualidade microbiológica de amostras de leite e queijo de Coalho, a prevalência de genes das enterotoxinas, a resistência antimicrobiana e o fenótipo de resistência MLSB por meio do D-teste, em cepas de Staphylococcus aureus isoladas destes produtos. Foram analisadas 70 amostras de leite e queijo de coalho. Cepas de S. aureus foram identificadas por meio de testes bioquímicos. A presença de genes se (sea-see) foi verificada pela técnica da reação em cadeia da polimerase (PCR). A sensibilidade antimicrobiana das cepas de S. aureus foi avaliada para 13 antimicrobianos, utilizando a técnica de difusão em disco e o ensaio de difusão de disco duplo (D-teste), para determinar o fenótipo de resistência induzível à lincosamidas. A quantidade de enterotoxina estafilocócica suficiente para causar a intoxicação alimentar é geralmente observada quando a população de estafilococos excede a contagem de 105 UFC mL-1 g-1. Neste estudo, nenhuma das amostras de leite analisadas apresentou esta contagem, entretanto, 73,3 % (22/30) das amostras de queijo de Coalho ultrapassaram este valor. Um total de 109 isolados foram identificados como S. aureus. A presença de genes de enterotoxinas foi detectada em 25,7% destas estirpes, ocorrendo amplificação apenas para o gene sec. A maioria destas bactérias (78,5 %) apresentaram resistência a um ou mais de dois agentes antimicrobianos. O D-teste demostrou que 25,0 % dos isolados resistentes à eritromicina tinham o fenótipo de resistência constitutiva e 3,8 % tinham o fenótipo de resistência induzível à clindamicina. Estes resultados indicam que estes produtos lácteos representam um risco para a saúde, desde que essas bactérias podem causar doenças de origem alimentar ou podem ser um possível caminho para a transferência de cepas resistentes aos antimicrobianos para os seres humanos.
Palavras-chave: Resistência antimicrobiana, Produtos lácteos, Queijo de coalho, Genes de enterotoxinas estafilocócicas, Staphylococcus aureus.
Introduction
Several reports have shown foodborne disease outbreaks caused by milk and other dairy products. These outbreaks can be associated with the poor microbiological quality of these products, which is influenced by the raw milk microbiota, processing conditions, and post-heat treatment contamination (CRETENET et al., 2011; EFSA, 2015; BRASIL, 2018; PAULIN et al., 2011).
In Brazil, the majority of small dairy farms have poor technology and present deficiencies in hygiene and sanitary control, which results in raw milk with high levels of microbiological contamination (AZEVÊDO et al., 2014; DANTAS et al., 2013; RALL et al., 2008). It is under these conditions that many varieties of cheese are often produced, including Coalho cheese, increasing the risk of foodborne diseases.
Coalho cheese, which is very popular and widely consumed by the regional population, is mainly produced in the Northeastern States of Brazil. This production represents a significant source of income for milk suppliers, especially those who do not have access to milk processing plants (AZEVÊDO et al., 2014; DANTAS et al., 2013; OLIVEIRA et al., 2010; SOUSA et al., 2014; TIGRE; BORELLY, 2011).
Staphylococcal food poisoning (SFP) is one of the most common foodborne diseases related to the consumption of dairy products (CRETENET et al., 2011; EFSA, 2015; LE LOIR et al., 2003). It is caused by the ingestion of staphylococcal enterotoxins (SEs) preformed in food by enterotoxigenic strains of different Staphylococcus species, but mainly Staphylococcus aureus (ARGAW; ADDIS, 2015; HENNEKINNE et al., 2012).
SEs are potent superantigens whose biological activity is retained even after the pasteurization of milk (CRETENET et al., 2011; PAULIN et al., 2011). To date, 20 different SEs have been described and traditionally divided into two groups: classical (SEA-SEE) enterotoxins and non-classical enterotoxins (SEG-SEI, SEK-SEV) (FISHER et al., 2018; HENNEKINNE et al., 2012).
About 95.0% of SPF outbreaks worldwide are caused by classical SEs (ARGAW; ADDIS, 2015; CRETENET et al., 2011; LE LOIR et al., 2003). However, molecular studies suggest that non-classical enterotoxins play a prominent role in this disease, with the low availability of sensitive methods for confirming the involvement of these toxins in outbreaks having contributed to their real prevalence being underestimated (DENAYER et al., 2017; UMEDA et al., 2017).
In addition to the production of enterotoxins, S. aureus strains may have several mechanisms of antibiotic resistance (REYGAERT, 2013). Multidrug-resistant strains have been isolated from the food chain, including dairy products (DARWISH; HANNA, 2013; OLIVEIRA et al., 2011).
Macrolides are widely used in the therapy of mastitis infections (OLIVER; MURINDA, 2012). Macrolide resistance can be either constitutive or inducible. Strains with inducible resistance to clindamycin are difficult to detect in the routine laboratory as they appear to be clindamycin-sensitive and erythromycin-resistant when the disks are not placed adjacent to each other, but in vivo therapy with clindamycin may lead to clinical therapeutic failure (LI et al., 2015; STEWARD et al., 2005).
In this context, the aims of this work were to evaluate whether the Coagulase-Positive Staphylococci (CPS) counting meets the standards established by the Brazilian legislation in milk and Coalho cheeses (RDC/ANVISA n° 12), and, due to the importance of S. aureus among SCP, to determine which CPS are S. aureus, evaluate the presence of genes encoding classical enterotoxins and assess the antimicrobial resistance in these strains.
Material and Methods
Sampling procedure
Three small-scale dairy farms (A, B, C) situated in the rural area of Teresina City, Piauí State, Brazil were selected based on the ease of access. Forty samples of bulk tank milk and 30 samples of Coalho cheese were sampled weekly in the period from January to May 2013.
From farm A, which uses pasteurized cow milk for cheese making, 10 samples of raw milk, 10 samples of pasteurized milk and 10 cheese samples were collected. From the other farms, which prepare the cheese from raw cow milk, 10 raw milk samples and 10 cheese samples were collected. Each sample consisted of 250 g of cheese or 250 ml of milk and was collected aseptically in sterile flasks; these were immediately transported in coolers to the laboratory.
Isolation and counting of Staphylococcus Coagulase Positive
A portion of 25 g of cheese or 25 ml of milk was homogenized with 225 ml of 0.1% buffered peptone water (Himedia, India). Decimal serial dilutions were prepared, and each dilution was plated in Baird-Parker agar (Himedia, India) supplemented with egg yolk-tellurite emulsion (NewProv, Brazil). Plates were incubated at 37°C for 48 h. Then, 10 colonies, typical or atypical, were enumerated and subsequently taken for confirmatory testing (LAIRD et al., 2004).
SCP identification was conducted by Gram staining, catalase test, coagulase test, and susceptibility to bacitracin (0.04 U; Laborclin, Brazil) and furazolidone (100 µg; Cecon, Brazil).
S. aureus identification
S. aureus was identified through tube coagulase reaction, aerobic and anaerobic fermentation of the sugars trehalose, maltose, and mannitol, acetoin production (Voges-Proskauer test), β-galactosidase activity and growth on P-agar with 7 µg mL-1 acriflavine dye (GANDRA et al., 2005; WINN et al., 2008).
Detection of classical staphylococcal enterotoxins genes
DNA from each S. aureus strain was extracted from 1.0 ml of BHI culture medium incubated at 37°C overnight (BD, New Jersey, USA) using a Wizard® kit (Promega®, USA), according to the manufacturer's instructions, except for the cell lysis, which was performed using 1.3 U of lysostaphin (Sigma®, USA). All of the DNA samples were stored at -20ºC.
All S. aureus species identified were screened for enterotoxin sea, seb, sec, sed and see genes by PCR. Table 1 shows the primers used for each gene studied.
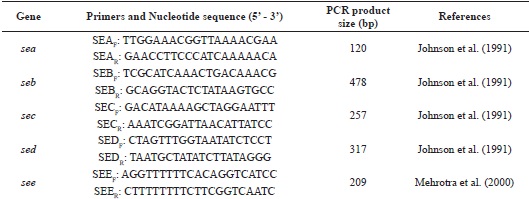
The PCR was performed in a 25 µl reaction mixture, as previously described (JOHNSON et al., 1991), by using 100 ng of DNA with the following amplification cycles: for the detection of the sea, seb and sec genes, initial denaturation at 94°C for 5 minutes followed by 27 cycles of denaturation for 2 minutes, annealing of primers at 52°C for 2 minutes and extension at 72°C for 1 minute. A final step at 72°C for 5 minutes was performed after completion of cycles. For amplification of the sed and see genes, the same parameters were used, except for annealing temperature, which was 51°C and 50°C, respectively (Table 1).
The following strains of S. aureus were used as positive controls for the presence of the genes evaluated: FRI 996 (sea, seb, and sed positive); INCQS 00080 (sec positive) and ATCC 27664 (see positive). Furthermore, strains of S. epidermidis ATCC 12228 and Escherichia coli ATCC 25922 were used as negative controls for the PCR reactions.
Antimicrobial susceptibility testing
The S. aureus strains were assessed for susceptibility to a panel of antimicrobial agents using the disc diffusion method on Mueller-Hinton agar (BD), according to the Clinical and Laboratory Standards Institute (CLSI, 2012). The following impregnated antibiotic discs (Cecon) were used: ampicillin (10 µg), doxycycline (30 µg), chloramphenicol (30 µg), linezolid (30 µg), trimethoprim-sulfamethoxazole (25 µg), tetracycline (30 µg), penicillin G (10 U), cephalothin (30 µg), enrofloxacin (5 µg) and gentamycin (10 µg). After incubation at 35 ± 2°C for 24 h, the susceptibility of the S. aureus isolates to each antimicrobial agent was assessed by measuring the halos of inhibition for each antimicrobial agent, and the results were interpreted in accordance with the standard criteria (CLSI, 2015). Oxacillin (1 µg) and cefoxitin (30 mg) disks were used for the detection of methicillin-resistant Staphylococcus aureus (MRSA).
To determine the inducible clindamycin resistance phenotype, the D-test was carried out by using erythromycin (15 µg) and clindamycin discs (2 µg), as recommended (FIEBELKORN et al., 2003). The plates were incubated at 35°C for 18 h, and the production of D-shaped zones between the erythromycin and clindamycin disks was visually evaluated.
The strains S. aureus ATCC 25923 and S. aureus ATCC BAA-977, which contain ermA, and S. aureus ATCC BAA-976, which contains msrA, were included in each trial as controls for the quality assessment of antimicrobial susceptibility and CLI-induction testing.
Results and Discussion
Isolation and counting of Staphylococcus Coagulase Positive
In this study, coagulase-positive staphylococci (CPS) were observed in 11 (27.5%) out of 40 milk samples and 22 (73.4%) out of 30 cheese samples (Table 2).

CPS - Coagulase Positive Staphylococci; CFU- Colony Forming Units.
The amount of toxin sufficient to cause foodborne diseases is generally observed when Staphylococcus counts exceed 105 CFU mL-1 g-1 (CRETENET et al., 2011). In this study, none of the milk samples analyzed showed these counts. However, 73.3% (22/30) of Coalho cheese samples exceeded this value. Furthermore, these samples do not conform to standards established by the National Agency of Sanitary Surveillance (ANVISA) for cheese, which determines maximum counts of 5.0 X 102 CFU/g (BRASIL, 2001).
Similar results were found in Brazil, which verified CPS counts of between 105 and 106 CFU g-1 in 35.0% of samples of Coalho cheese analyzed and marketed in Salvador City (TIGRE; BORELY, 2011). Another study found CPS counts ranging from 1.6 x 103 to 2.0 x 105 CFU g-1 in 76.2% of Coalho cheese samples analyzed (SOUSA et al., 2014; OLIVEIRA et al., 2010). Unlike these studies, Borges et al. (2008) did not demonstrate high counts of these agents, with results ranging from <2.0 x 101 to 102 CFU g-1 for Coalho cheese from the production line in Fortaleza City, Ceará State.
The differences observed in the CPS counts between milk and cheese samples may be explained by the capacity of these bacteria to grow in milk and cheese during the cheese making process. Another possible source of cheese contamination could be unhygienic milk processing conditions or handling practices in cheese manufacturing. These staphylococci could be introduced directly to milk from the cow with clinical or subclinical staphylococcal mastitis, from the environment or by human handling, since coagulase positive species such as S. aureus belong to the microbiota (AZEVÊDO et al., 2014; JØRGENSEN et al., 2005; PAULIN et al., 2011).
The consumption of Coalho cheese in the North and Northeast of the Brazil is vast. It has also achieved international projection by being reported as a major functional food (SILVA et al., 2012). Therefore, it is important to obtain a higher quality product with the adoption of Good Manufacturing Practices (GMP) and the heat treatment of raw milk by producers.
S. aureus identification
Out of 479 Staphylococcus spp. isolates obtained from milk and Coalho cheese samples, 148 (30.9%) were characterized as CPS; of these, 109 (73.6%) were identified as S. aureus. These isolates were biochemically identified by positive coagulase 4+ or 3+ in rabbit plasma, the ability to ferment mannitol, maltose and trehalose under aerobic conditions and mannitol under anaerobic conditions, the production of acetoin, the absence of β-galactosidase activity and acriflavine resistance.
In Brazil, this species was associated with 798 outbreaks (19.5%) from 2000 to 2014, with more than 356 being linked to dairy products (BRASIL, 2018).
Detection of classical staphylococcal enterotoxins genes
None of the S. aureus strains were positive for the sea, seb, sed and see genes, while 28 (25.7%) were positive for the sec gene, with 3 (10.7%) being reported in milk and 25 (89.3%) in Coalho cheese (Table 3).
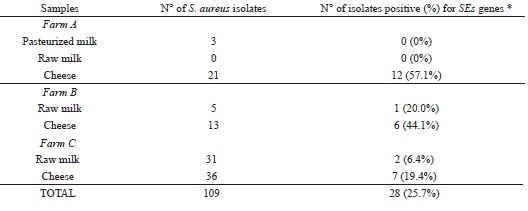
SE: staphylococcal enterotoxins.* All strains positives for SEs genes amplified only for the sec gene.
Akindolire et al. (2015) and Khudor et al. (2012) found similar results, with the sec gene detected in 24.5% and 32.7%, respectively, of the S. aureus isolates from bovine milk samples; none of the other SE genes were detected. Also, Tsegmed et al. (2007) found the sec gene in 19.0% of the S. aureus isolates in raw milk, and none of these isolates had the sea, seb, sed or see genes. Other authors observed that S. aureus strains isolated from milk and dairy products from ruminants had mainly sec and sea genes; in humans, the sea gene was the most frequently identified (JØRGENSEN et al., 2005; NORMANNO et al., 2007; RALL et al., 2008).
The prevalence of enterotoxin genes in S. aureus strains often depends on the environment from which the strain was isolated (PAULIN et al., 2011; JØRGENSEN et al., 2005). This variation is so great that the frequencies reported for strains of S. aureus carrier or enterotoxin producers described for milk, cheese, and both products ranged from 12.0% to 77.7%, 9.0% to 100% and 8.8% to 93.3%, respectively (PAULIN et al., 2011). Therefore, the results obtained in this study agree with other reports related to the prevalence of S. aureus strains isolated from dairy products with regards to their diversity as well as their enterotoxigenicity.
Antimicrobial susceptibility testing
Resistance to antimicrobial compounds used in veterinary and human therapy is a major concern worldwide, and S. aureus is known for its ability to become resistant to antimicrobials. Our study assessed the antimicrobial susceptibility patterns of S. aureus isolates (Table 4).
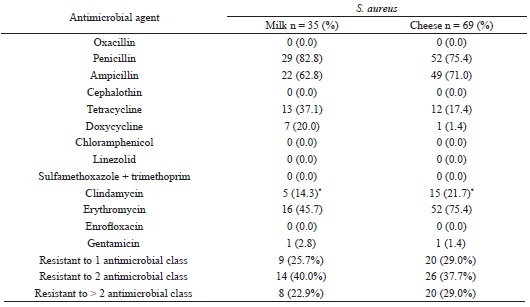
* Three strains of S. aureus obtained from of milk and one from Coalho cheese displayed inducible clindamycin resistance phenotype.
A high prevalence of β-lactam (77.9% for penicillin and 68.3% for ampicillin), erythromycin (65.4%), tetracycline (24.0%), and clindamycin (19.2%) resistance was observed. Other studies found similar results (AKINDOLIRE et al., 2015; COSTA et al., 2013; JAMALI et al., 2015).
One possible reason for the phenotypes presented in staphylococcal isolates from milk and cheese could be due to the frequent use of antibiotics for the treatment of animal infections, with β-lactam antibiotics being the most often used in mastitis therapy and other staphylococcal infections, and tetracycline and erythromycin being commonly used in animal diseases caused by several bacteria (JAMALI et al., 2015; SUDAGIDAN; AYDIN, 2013).
In our study, we found 68 (65.4%) erythromycin-resistant and 20 (19.2%) clindamycin-resistant isolates. For clindamycin, 3.8% (3/104) of isolates showed an inducible resistance phenotype to clindamycin (D-test positive) and 15.4% (17/104) isolates showed constitutive resistance. Also, 51 isolates showed both erythromycin resistance and clindamycin sensitivity without an inducible phenotype, suggesting that the resistance phenotype occurs by efflux pump, which allows the safe and effective use of clindamycin therapy (LI et al., 2015).
Few studies have verified the presence of the inducible clindamycin resistance phenotype of S. aureus isolated from food (JAMALI et al., 2015; SUDAGIDAN; AYDIN, 2013). Infections caused by the transmission of these strains in the food chain can result in an inappropriate clindamycin therapy.
Fortunately, in this study, none of the isolates were identified as MRSA, differing from other studies that have described the detection of this microorganism in dairy products, at levels varying from 1.1% to 60.5% worldwide and from 10.0% to 25.4% in Brazil (COSTA et al., 2013; DIAS et al., 2011; PEXARA et al., 2013).
However, isolates with a multidrug phenotype were found in the current study. The multidrug-resistant (MDR) isolates were detected in 22.8% of milk and 29.0% of Coalho cheese samples. The prevalence of MDR S. aureus isolates (26.9%) in this study, agrees with earlier findings (AKINDOLIRE et al., 2015; JAMALI et al., 2015).
Conclusion
This study showed a high prevalence of milk and cheese contaminated with S. aureus. The higher counts and numbers of contaminated samples in cheese compared to milk could indicate failures in the hygienic conditions applied during milking and the manufacture of Coalho cheese.
The results provided evidence that the presence of enterotoxigenic and antimicrobial-resistant strains of S. aureus has become remarkably widespread in foods, presenting a potential risk of foodborne diseases. None of the isolates were methicillin resistant. In this study, a low prevalence of the inducible clindamycin resistance phenotype was found among S. aureus strains; however, 26.9% of isolates were multidrug-resistant.
In conclusion, sanitary measures are needed to guarantee the quality of these products and prevent the spread of antimicrobial resistance organisms, since food may act as a vehicle for the transfer of toxins and antibiotic-resistant microorganisms to humans.
Acknowledgments
The authors would like to thanks the farmers for their contribution, and Antônio Aécio de Carvalho Bezerra for the help in performing technical parts of the project. This study was supported by the Federal University of Piauí; Foundation for Research of Piauí State (FAPEPI), and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenadoria de Aperfeiçoamento de Pessoal de Nível Superior (Capes), Brazil.
References
AKINDOLIRE, M. A.; BABALOLA, O. O.; ATEBA, C. N. Detection of antibiotic resistant Staphylococcus aureus from milk: a public health implication. International Journal of Environmental Research and Public Health, Basel v. 12, n. 9, p. 10254-10275, 2015.
ARGAW, S.; ADDIS, M. A review on staphylococcal food poisoning. Food Science and Quality Management, Hong Kong S.A.R, v. 40, n. 2015, p. 59-72, 2015.
AZEVÊDO, M. C.; SANTOS, G. B.; ZOCCHE, F.; HORTA, M. C.; DIAS, F. S.; COSTA, M. M. Microbiological evaluation of raw milk and Coalho cheese commercialized in the semi-arid region of Pernambuco, Brazil. African Journal of Microbiology Research, Lagos v. 8, n. 3, p. 222-229, 2014.
BORGES, M. F.; NASSU, R. T.; PEREIRA, J. L.; ANDRADE, A. P. C.; KUAYE, A. Y. Perfil de contaminação por Staphylococcus e suas enterotoxinas e monitorização das condições de higiene em uma linha de produção de queijo de Coalho. Ciência Rural, Santa Maria v. 38, n. 5, p. 1431-1438, 2008.
BRASIL. Ministério da Saúde. Agência Nacional de Vigilância Sanitária. Resolução RDC nº 12. 2001. Diário Oficial [da]República Federativa do Brasil, Brasília, 10, jan., 2001. Seção I, p. 45-53.
______. ______. Secretaria de Vigilância em Saúde. Surtos de doenças transmitidas por alimentos no Brasil. Brasília, 2018. Available at: <http://portalarquivos2.saude.gov.br/images/pdf/2018/janeiro/17/Apresentacao-Surtos-DTA-2018.pdf>. Accessed at: 03 maio 2018.
CLINICAL AND LABORATORY STANDARDS INSTITUTE - CLSI. Performance standards for antimicrobial disk and dilution susceptibility testing; twenty-third informational supplement. CLSI document M100-S25 Informational Supplement. Wayne: CLSI, 2015.
______. Performance standards for antimicrobial disk susceptibility tests, CLSI document M02-A11. 11th ed. Wayne: CLSI, 2012.
COSTA, G. M.; BARROS, R. A.; CUSTÓDIO, D. A. C.; PEREIRA, U. P.; FIGUEIREDO, D. J.; SILVA, N. Resistência a antimicrobianos em Staphylococcus aureus isolados de mastite em bovinos leiteiros de Minas Gerais, Brasil. Arquivos do Instituto Biológico, São Paulo v. 80, n. 3, p. 297-302, 2013.
CRETENET, M.; EVEN, S.; LE LOIR, Y. Unveiling Staphylococcus aureus enterotoxin production in dairy products: a review of recent advances to face new challenges. Dairy Science & Technology, Rennes, v. 91, n. 2, p. 127-150, 2011.
DANTAS, D. S.; ARAÚJO, A. M.; SANTOS, J. O.; SANTOS, M. S.; RODRIGUES, O. G. Qualidade microbiológica do queijo de Coalho comercializado no município de Patos, Estado da Paraíba. ACSA - Agropecuária Científica no Semiárido, Patos, v. 9, n. 3, p. 110-118, 2013.
DARWISH, S. F.; HANNA, A. E. A. Investigation of biofilm forming ability in Staphylococci causing bovine mastitis using phenotypic and genotypic assays. The Scientific World Journal, Newbury v. 2013, n. 2013, p. 1-9, 2013.
DENAYER, S.; DELBRASSINNE, L.; NIA, Y.; BOTTELDOORN, N. Food-borne outbreak investigation and molecular typing: high diversity of Staphylococcus aureus strains and importance of toxin detection. Toxins, Basel v. 9, n. 13, p. 407-420, 2017.
DIAS, N. L.; SILVA, D. C. B.; OLIVEIRA, D. C. B. S.; FONSECA JUNIOR, A. A.; SALES, M. L.; SILVA, N. Detecção dos genes de Staphylococcus aureus, enterotoxinas e de resistência à meticilina em leite. Arquivos Brasileiros de Medicina Veterinária e Zootecnia, Belo Horizonte v. 63, n. 6, p. 1547-1552, 2011.
EUROPEAN FOOD SAFETY AUTHORITY; EUROPEAN CENTRE FOR DISEASE PREVENTION AND CONTROL - EFSA; ECDC. EU Summary Report on antimicrobial resistance in zoonotic and indicator bacteria from humans, animals and food in 2013. EFSA Journal, Parma v. 13, n. 5, p. 4036-4042, 2015.
FIEBELKORN, K. R.; CRAWFORD, S. A.; MCELMEEL, M. L.; JORGENSEN, J. H. Practical disk diffusion method for detection of inducible clindamycin resistance in Staphylococcus aureus and coagulase-negative Staphylococci. Journal of Clinical Microbiology, Washington v. 41, n. 10, p. 4740-4744, 2003.
FISHER, E. L.; OTTO, M.; CHEUNG, G. Y. C.. Basis of virulence in enterotoxin-mediated Staphylococcal food poisoning. Frontiers in Microbiology, Lausanne v. 9, n. 436, p. 1-18, mar. 2018.
GANDRA, E. A.; SILVA, J. A.; MACEDO, M. R. P.; ARAÚJO, M. R.; MATA, M. M.; SILVA, W. P. Biochemical differentiation among S. aureus, S. intermedius, and S. hyicus isolated from bovines with subclinical mastitis. Archives of Veterinary Science, Curitiba v. 10, n. 1, p. 75-87, 2005.
HENNEKINNE, J. A.; DE BUYSER, M. L.; DRAGACCI, S. Staphylococcus aureus and its food poisoning toxins: characterization and outbreak investigation. FEMS Microbiology Reviews, Amsterdam v. 36, n. 4, p. 815-836, 2012.
JAMALI, H.; PAYDAR, M.; RADMEHR, B.; ISMAIL, S.; DADRASNIA, A. Prevalence and antimicrobial resistance of Staphylococcus aureus isolated from raw milk and dairy products. Food Control, UK v. 54, p. 383-388, 2015.
JOHNSON, W. M.; TYLER, S. D.; EWAN, E. P.; ASHTON, F. E.; POLLARD, D. R.; ROZEE, K. R. Detection of genes for enterotoxins, exfoliative toxins, and toxic shock syndrome toxin in Staphylococcus aureus by the polymerase chain reaction. Journal of Clinical Microbiology, Washington v. 29, n. 3, p. 426-430, 1991.
JØRGENSEN, H. J.; MØRK, T.; HØGA, H. R.; RØRVIK, L. M. Enterotoxigenic Staphylococcus aureus in bulk milk in Norway. Journal of Applied Microbiology, Oxford v. 99, n. 1, p. 158-166, 2005.
KHUDOR, M. H.; ABBAS, B. A.; IDBEIS, H. I. Detection of enterotoxin genes of Staphylococcus aureus isolates from raw milk. Basrah Journal of Veterinary Research, Basra v. 11, n. 1, p. 254-264, 2012.
LAIRD, D. T.; GAMBREL-LENARS, S. A.; SCHER, F. M.; GRAHAM, T. E.; REDDY, R. Microbiological count methods. In: WEHR, H. M.; FRANK, J. F. (Ed.). Standard methods for the examination of dairy products. 17th ed. Washington: American Public Health Association, 2004. p 153-86.
LE LOIR, Y.; BARON, F.; GAUTIER, M. Staphylococcus aureus and food poisoning. Genetics and Molecular Research, Ribeirão Preto v. 31, n. 2, p. 63-76, 2003.
LI, L.; FENG, W.; ZHANG, Z.; XUE, H.; ZHAO, X. Macrolide-lincosamide-streptogramin resistance phenotypes and genotypes of coagulase-positive Staphylococcus aureus and coagulase-negative staphylococcal isolates from bovine mastitis. BMC Veterinary Research, London v. 11, n. 168, p. 168-172, 2015.
MEHROTRA, M.; WANG, G.; JOHNSON, W. M. Multiplex PCR for detection of genes for Staphylococcus aureus enterotoxins, exfoliative toxins, toxic shock syndrome toxin 1, and methicillin resistance. Journal of Clinical Microbiology, Washington v. 38, n. 3, p. 1032-1035, 2000.
NORMANNO, G.; LA SALANDRA, G.; DAMBROSIO, A.; QUAGLIA, N. C. Occurrence, characterization and antimicrobial resistance of enterotoxigenic Staphylococcus aureus isolated from meat and dairy products. International Journal of Food Microbiology, Amsterdam v. 115, n. 3, p. 290-296, 2007.
OLIVEIRA, K. D.; EVÊNCIO NETO, J.; PAIVA, J. E. Qualidade microbiológica do queijo de Coalho comercializado no Município do Cabo de Santo Agostinho, Pernambuco, Brasil. Arquivos do Instituto Biológico, São Paulo v. 77, n. 3, p. 435-440, 2010.
OLIVEIRA, L.; RODRIGUES, A. C.; HULLAND, C.; RUEGG, P. L. Enterotoxin production, enterotoxin gene distribution, and genetic diversity of Staphylococcus aureus recovered from milk of cows with subclinical mastitis. American Journal of Veterinary Research, Chicago v. 72, n. 10, p. 1361-1368, 2011.
OLIVER, S. P.; MURINDA, S. E.. Antimicrobial resistance of mastitis pathogens. Veterinary Clinics of North America: Food Animal Practice, Maryland Heights, v. 28, n. 2, p. 165-185, 2012
PAULIN, S.; HORN, B.; HUDSON, J. A. Factors influencing staphylococcal enterotoxin production in dairy products. Wellington: Institute of Environmental Science and Research Limited, 2011. 83 p.
PEXARA, A.; SOLOMAKOS, N.; GOVARIS, A. Prevalence of methicillin-resistant Staphylococcus aureus in milk and dairy products. Journal of the Hellenic Veterinary Medical Society, Athena v. 1, n. 64, p. 17-23, 2013.
RALL, V. L. M.; VIEIRA, F. P.; RALL, R.; VIEITIS, R. L.; FERNANDES, A. J.; CARDOSO, K. F.; ARAÚJO, J. P. J. PCR detection of staphylococcal enterotoxin genes in Staphylococcus aureus strains isolated from raw and pasteurized milk. Veterinary Microbiology, Amsterdam v. 132, n. 3-4, p. 408-413, 2008.
REYGAERT, W. C. Antimicrobial resistance mechanisms of Staphylococcus aureus. In: MÉNDEZ-VILAS, A. (Ed.). Microbial pathogens and strategies for combating them: science, technology and education. Badajoz: Formatex Research Center, 2013. vol. 1, p. 297-305.
SILVA, R. A.; LIMA, M. S. F.; VIANA, J. B. M.; BEZERRA, V. S.; PIMENTEL, M. C.; PORTO, A. L.; CAVALCANTI, M. T.; LIMA FILHO, J. L. Can artisanal Coalho cheese from Northeastern Brazil be used as a functional food? Food Chemistry, Amsterdam v. 135, n. 3, p. 1533-1538, 2012.
SOUSA, A. Z. B.; ABRANTES, M. R.; SAKAMOTO, S. M.; SILVA, J. B. A.; LIMA, P. O.; LIMA, R. N.; ROCHA, M. O. C.; PASSOS, Y. D. B.. Aspectos físico-químicos e microbiológicos do queijo tipo Coalho comercializado em estados do nordeste do Brasil. Arquivos do Instituto Biológico, São Paulo v. 81, n. 1, p. 30-35, 2014.
STEWARD, C. D.; RANEY, P. M.; MORRELL, A. K.; WILLIAMS, P. P.; MCDOUGAL, L. K.; JEVITT, L.; MCGOWAN JUNIOR, J. E.; TENOVER, F. C. Testing for induction of clindamycin resistance in erythromycin-resistant isolates of Staphylococcus aureus. Journal of Clinical Microbiology, Washington v. 43, n. 4, p. 1716-1721, 2005.
SUDAGIDAN, M.; AYDIN, A. Presence of inducible clindamycin resistance phenotype and erm genes in foodborne Staphylococcus aureus isolates. Foodborne Pathogens and Disease, New Rochelle v. 10, n. 6, p. 555-558, 2013.
TIGRE, D. M.; BORELLY, M. A. D. N. Pesquisa de Estafilococos coagulase-positiva em amostras de queijo Coalho comercializadas por ambulantes na praia de Itapuã (Salvador-BA). Revista de Ciências Médicas e Biológicas, Salvador v. 10, n. 2, p. 162-166, 2011.
TSEGMED, U.; NORMANNO, G.; PRINGLE, M.; KROVACEK, K. Occurrence of enterotoxic Staphylococcus aureus in raw milk from yaks and cattle in Mongolia. Journal of Food Protection, Des Moines v. 70, n. 7, p. 1726-1729, 2007.
UMEDA, K.; NAKAMURA, H.; YAMAMOTO, K.; NISHINA, N.; YASUFUKU, K.; HIRAI, Y.; HIRAYAMA, T.; GOTO, K.; HASE, A.; OGASAWARA, J. Molecular and epidemiological characterization of staphylococcal foodborne outbreak of Staphylococcus aureus harboring seg, sei, sem, sen, seo, and selu genes without production of classical enterotoxins. International Journal of Food Microbiology, Amsterdam v. 256, n. 1, p. 30-35, 2017.
WINN, W. C.; ALLEN, S.; JANDA, W.; KONEMAN, E.; PROCOP, G.; SCHRECKENBERGER, P.; WOODS, G. Koneman, diagnóstico microbiológico: texto e atlas colorido. 6. ed. Rio de Janeiro: Guanabara Koogan, 2008. 1760 p.
Author notes
*Author for correspondence
Conflict of interest declaration
No competing financial interests exist.