ARTICLES
Extract of Amburana cearensis maintains the survival of ovine preantral follicles during long-term ovarian tissue transport and promotes primordial follicle activation after in vitro culture
Extrato de Amburana cearensis mantém a sobrevivência de folículos pré-antrais ovinos durante longo período de transporte de tecido ovariano e promove a ativação de folículos primordiais após cultivo in vitro
Extract of Amburana cearensis maintains the survival of ovine preantral follicles during long-term ovarian tissue transport and promotes primordial follicle activation after in vitro culture
Semina: Ciências Agrárias, vol. 39, no. 5, pp. 2001-2016, 2018
Universidade Estadual de Londrina
Received: 11 August 2017
Accepted: 04 June 2018
Funding
Funding source: National Council for Scientific and Technological Development
Contract number: 562548/2010-6
Abstract: This study evaluated the effect of Amburana cearensis extract as a preservation or culture medium for ovine ovarian tissue. Ovarian fragments were fixed in 4% buffered formaldehyde for 18 h (fresh control), stored in Minimal Essential Medium (MEM) or in A. cearensis extract (0.1; 0.2 or 0.4 mg/mL) at a temperature of 4ºC for 6, 12 or 24 h (preservation - experiment 1) or cultured for 7 days in α-MEM+ or in A. cearensis extract without (0.1; 0.2 or 0.4 mg/mL) or with supplements (0.1+; 0.2+ or 0.4+ mg/mL; experiment 2). The percentages of morphologically normal follicles and follicular activation were submitted to analysis of variance (ANOVA) and Tukey´s test. The values of TUNEL-positive cells were submitted to Chi-square test (P < 0.05). The storage of fragments for 6 h in MEM showed higher percentages of normal follicles (62%) and a lower rate of TUNEL positive cells (36.17%) compared to other treatments (normal follicles: 46%; 43% and 52%; TUNEL positive cells: 58.57%; 55.30% and 55.63% for Amb 0.1; Amb 0.2 and Amb 0.4 mg/mL, respectively). However, after 12 or 24 h, MEM (12 h: 48%; 24 h: 45%) and Amb 0.2 mg/mL (12 h: 37%; 24 h: 39%) showed similar percentages of normal follicles and TUNEL positive cells (MEM - 12 h: 43.26%; 24 h: 58%; Amb 0.2 mg/mL - 12 h: 50%; 24 h: 61%). After culture, α-MEM+ recorded a higher percentage of normal follicles (58.25%) than A. cearensis treatments (32.8%; 25.4% and 34.2% for Amb 0.1; Amb 0.2 and Amb 0.4 mg/mL, and 22.25%; 20.0% and 36.6% for Amb 0.1+; Amb 0.2+ and Amb 0.4+ mg/mL, respectively) (P < 0.05). Follicular activation increased in all treatments (52.5%; 36.73%; 54.05%; 47.5% and 58.19% for α-MEM+; Amb 0.1; Amb 0.1+; Amb 0.2+ and Amb 0.4+ mg/mL, respectively) compared to the fresh control (11.65%), except for Amb 0.2 mg/mL (23.69%) and Amb 0.4 mg/mL (28.85%) (P > 0.05). Moreover, after in vitro culture, A. cearensis at a concentration of 0.1 mg/mL maintained the percentage of TUNEL positive cells (30.0%) in a way that is similar to that observed in the fresh control (22%) (P > 0.05). In conclusion, ovine preantral follicles can be preserved at 4°C in MEM for 6 h. For longer periods of preservation (24 h), MEM and 0.2 mg/mL A. cearensis are recommended. Moreover, after in vitro culture, A. cearensis extract (0.1 mg/mL) showed higher activation and lower DNA fragmentation in ovine preantral follicles.
Key words: Activation, Antioxidant, Medicinal plant, Oocyte, Ovary, Ovine.
Resumo: Este estudo avaliou o efeito do extrato de Amburana cearensis como meio de preservação ou de cultivo de tecido ovariano ovino. Fragmentos ovarianos foram fixados em formaldeído tamponado a 4% por 18 h (controle fresco), armazenados em Meio Essencial Mínimo (MEM) ou extrato de A. cearensis (0,1; 0,2 ou 0,4 mg/mL) a temperatura de 4 ºC durante 6, 12 ou 24 h (conservação - experimento 1) ou cultivados durante 7 dias em α-MEM + ou em extrato de A. cearensis sem (0,1; 0,2 ou 0,4 mg/mL) ou com suplementos (0,1+; 0,2+ ou 0,4+ mg/mL - experimento 2). As porcentagens de folículos normais e de ativação folicular foram submetidas às análises de variância (ANOVA) e teste de Tukey. Os valores das células TUNEL-positivas foram submetidos ao teste do qui-quadrado (P<0,05). A conservação de fragmentos por 6 h em MEM apresentou maiores percentagens de folículos normais (62%) e menor taxa de células TUNEL positivas (36,17%) comparado aos outros tratamentos (folículos normais - 46%; 43%; 52%; células TUNEL positivas - 58,57%; 55,30%; 55,63% em Amb 0,1 ;0,2 ou Amb 0,4 mg/mL, respectively). Entretanto, após 12 ou 24 h, MEM (12 h - 48%; 24 h - 45%) e Amb 0,2 (12 h - 37%; 24 h - 39%) apresentaram percentagens semelhantes de folículos normais e células TUNEL positivas (MEM: 12 h - 43,26%; 24 h -58%; Amb 0,2: 12 h - 50; 24 h - 61%). Após o cultivo, α-MEM+ (58,25%) apresentou maior percentual de folículos normais do que os tratamentos com A. cearensis (32,8%; 25,4%; 34,2% em Amb 0,1; Amb 0,2; Amb 0,4 mg/mL e 22,25%; 20,0%; 36,6% em Amb 0,1+; Amb 0,2+ e Amb 0,4+ mg/mL, respectivamente) (P < 0.05). A ativação folicular aumentou significativamente em todos os tratamentos (52,5%; 36,73%; 54.05%; 47,5% e 58,19% em α-MEM+; Amb 0,1; Amb 0,1+; Amb 0,2+ e Amb 0,4+ mg/mL, respectivamente) comparado ao controle fresco (11,65%), exceto em Amb 0,2 mg/mL (23,69%) e Amb 0,4 mg/mL (28,85%) (P > 0,05). Após o cultivo in vitro, a concentração de 0,1 mg/mL manteve a percentagem de células TUNEL positivas (30%) de modo similar ao observado no controle fresco (22%) (P > 0,05). Em conclusão, folículos pré-antrais ovinos podem ser preservados a 4 °C em MEM durante 6 h. Para períodos mais longos de transporte (até 24 h), MEM e 0,2 mg/mL do extrato de A. cearensis são recomendados. Além disso, após o cultivo in vitro, o extrato de A. cearensis (0,1 mg/mL) apresentou maior ativação e menor fragmentação de DNA em folículos pré-antrais de ovinos.
Palavras-chave: Ativação, Antioxidante, Planta Medicinal, Oócito, Ovário, Ovino.
Introduction
The development of culture systems that maintain follicle architecture would not only permit studies on the earliest stages of follicle development but also enable the development of new technological approaches to fertility preservation (SHEA et al., 2014). The initiation of primordial follicle growth in vitro has been successful using in situ organ culture in farm animals: goat (CELESTINO et al., 2009), sheep (CAVALCANTE et al., 2016) and bovine (WANDJI et al., 1996). However, since the ovarian tissue is commonly collected at distances far from specialized laboratories, ovarian transport is still a limiting factor in the maintenance of follicular quality (FIGUEIREDO et al., 2011). Therefore, to allow ovarian tissue culture to be more widely employed for fertility preservation, it is necessary to establish an efficient system for the storage and transportation of harvested ovaries (BARBERINO et al., 2016a; KAMOSHITA et al., 2016).
The preservation of goat ovarian fragments in Minimal Essential Medium (MEM) at 4ºC for 4 h maintained the rates of morphologically normal follicles similar to those observed in fresh tissues and also maintained follicular viability after in vitro culture. Nevertheless, storage at a temperature close to physiological values (35°C) for 4 h affected the follicular morphology after 7 days of culture (CHAVES et al., 2008). Also, a temperature of 35°C was used to preserve ovine ovarian tissue without affecting the DNA fragmentation of preantral follicles and their ability to develop in vitro. However, the conservation medium was MEM supplemented with ITS (insulin, transferrin and sodium selenite), glutamine, hypoxanthine, ascorbic acid, bovine serum albumin, recombinant FSH, and antibiotics (GONÇALVES et al., 2015). Although with those good results, it is still important to emphasize that the use of synthetic media (e.g. MEM or α-MEM associated with commercial supplements) is expensive. Therefore, the discovery of an alternative cheaper medium would be important for the further successful growth of preantral follicles in vitro. In this context, the use of a medicinal plant for the preservation of ovarian tissue (GOUVEIA et al., 2015; CAVALCANTE et al., 2017) or in the in vitro culture of secondary follicles (BARBERINO et al., 2016b; GOUVEIA et al., 2016) was reported due to its antioxidant capacity. Among several extracts of medicinal plants, A. cearensis is noteworthy for its numerous cytoprotective characteristics.
A. cearensis (Allemão) A.C. Smith (Fabaceae) is a tree commonly found in Northeastern Brazil, where it is popularly known as "amburana" or "amburana-de-cheiro" (ALBUQUERQUE et al., 2007; LEAL et al., 2011). This plant has been reported to exhibit a variety of biological activities including anti-inflammatory, bronchodilator, antispasmodic (CANUTO; SILVEIRA, 2006), as well as antioxidant activity (LEAL et al., 2011). Studies performed by our team showed that the ethanolic extract of A. cearensis maintains follicular survival in a way similar to MEM after preservation of caprine ovarian tissue at 4ºC for 6 h (GOUVEIA et al., 2015) and similar to α-MEM+ after in vitro culture of caprine isolated secondary follicles (GOUVEIA et al., 2016). Moreover, by using terminal deoxynucleotidyl transferase (TdT) mediated dUTP nick-end labeling (TUNEL) assay, these studies showed that A. cearensis extract presented similar percentages of follicles with DNA fragmentation compared to those observed in the control medium (MEM or α-MEM). Both studies utilized the same A. cearensis extract in which the following compounds were identified: gallic acid, protocatechuic acid, epicatechin, p-coumaric acid and kaempferol (GOUVEIA et al., 2015). Overall, these compounds possess antioxidant properties, reducing oxidative stress, reactive oxygen species (ROS) levels, lipid peroxidation and DNA damage in different cell types (HUANG et al., 2014; WANG; SHAHID, 2014).
Therefore, to reduce the damage caused by ROS during ovaries transport, A. cearensis extract could be used as a preservative medium. Moreover, since oxidative stress may affect follicle growth (LIANG et al., 2017), the antioxidant properties of A. cearensis may be a useful and cheap substitute for a standard culture medium. However, there is no study that investigated the possible effects of A. cearensis extract as a preservation medium during ovaries transport or as a culture medium for the ovine ovarian tissue. Thus, the objectives of the present study were to evaluate the effectiveness of A. cearensis extract as a preservation medium for sheep ovaries transport at 4ºC, as well as to evaluate the effect of this extract, with or without supplement, on the survival, activation and DNA fragmentation of ovine preantral follicles after in vitro culture of ovarian tissue.
Materials and Methods
Plant extraction and preparation
Unless indicated, the chemicals used in the present study were purchased from Sigma Chemical Co. (St. Louis, MO, USA). Fresh leaves of A. cearensis were collected and a voucher specimen (5545) was deposited at the herbarium of the University. The leaves were dried in an oven at 40ºC, pulverized and extracted at room temperature using 95% ethanol (Vetec, Duque de Caxias-RJ, Brazil) for 72 h. Drying of the extract was done at a temperature of 45ºC using rotavapor and the yield was approximated at 10% to obtain the crude ethanolic extract of the leaves of A. cearensis, which was dissolved in 0.9% saline solution, corresponding to concentrations of 0.1; 0.2 or 0.4 mg/mL.
Source of ovarian tissue
This experiment was conducted according to the ethical guidelines of the Ethics Committee on Animal Use of Federal University of São Francisco Valley (protocol 005082012).
The animals used were non-pregnant and presumed to be undergoing normal estrous cycles due to the presence of corpora lutea in their ovaries. Ovarian cortical tissues from 18 ovaries were collected at a local slaughterhouse from nine adults (1-3 years old) mixed breed sheep for in vitro preservation (experiment 1) or culture (experiment 2). Immediately after slaughter, ovarian pairs were washed once in 70% alcohol (Dinâmica, São Paulo, Brazil) and twice in MEM buffered with HEPES (MEM-HEPES) and supplemented with antibiotics (100 µg/mL penicillin and 100 µg/mL streptomycin).
Experiment 1: In vitro preservation of ovarian tissue
The in vitro preservation was performed according to a previous study (CHAVES et al., 2008). While still in the slaughterhouse, ovarian cortical tissues from each animal (n = 08 ovaries) were divided into 13 fragments of approximately 3 × 3 mm (1 mm thickness) dimension. Therefore, one ovarian fragment was randomly taken and immediately fixed for histological and TUNEL analyses (fresh control). The other 12 fragments were randomly placed into tubes containing 5 mL MEM supplemented with antibiotics (100 µg/mL penicillin and 100 µg/mL streptomycin; control medium) or placed in a medium composed of different concentrations of A. cearensis extract, obtained from the dilution in 0.9% solution (0.1; 0.2 or 0.4 mg/mL) and stored at 4ºC for 6, 12 or 24 h, respectively (simulating transport). After the preservation periods, the ovarian fragments were fixed and used for histological and TUNEL analyses. Each treatment was repeated four times (Figure 1A).
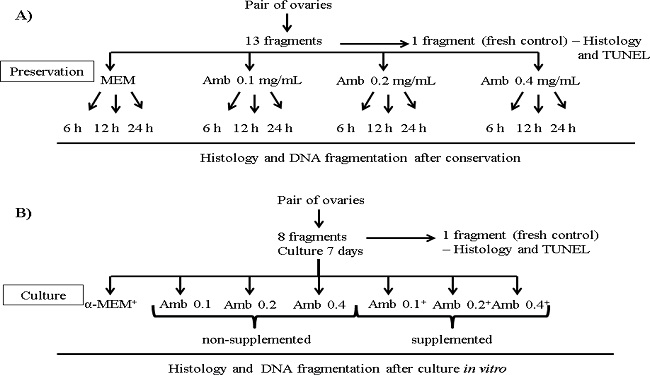
Figure 1
General experimental protocol for in vitro (A) preservation or (B) culture of ovine ovarian tissue in Amburana cearensis extract. Amb: Amburana cearensis.
Experiment 2: In vitro culture of ovarian tissue
A second experiment was performed to verify the effect of A. cearensis extract on the in vitro culture of preantral follicles enclosed in ovarian tissue. Based on the results of experiment 1 and the transportation period from the slaughterhouse to the laboratory (approximately 1 h), the entire ovaries (n=10) used in experiment 2 were transported in tubes containing MEM supplemented with antibiotics (control medium of experiment 1) at 4ºC.
The in vitro culture was performed according to a previous study (SANTOS et al., 2014). In the laboratory, the ovarian cortex from each ovarian pair were divided into 8 fragments of approximately 3 × 3 mm (1 mm thickness) dimension. Therefore, one ovarian fragment was taken randomly and immediately fixed in 4% buffered formaldehyde (Dinâmica, Diadema, São Paulo, Brazil) for 18 h, for histological and TUNEL analyses (fresh control). The remaining slices of ovarian cortex were individually cultured in 1 mL of culture medium in 24-well culture dishes for a period of 7 days at 39°C and 5% CO2. The base culture medium (control) consisted of α-MEM (pH 7.2-7.4; Gibco, Invitrogen, Karlsruhe, Germany) supplemented with 10 ng/mL insulin, 5.5 µg/mL transferrin, 5.0 ng/mL sodium selenite, 2 mM glutamine, 2 mM hypoxantine, 1.25 mg/mL bovine serum albumin, 50 µg/mL ascorbic acid, 100 µg/mL penicillin, 100 µg/mL streptomycin, and then referred as α-MEM+. To verify the influence of the A. cearensis extract medium (A. cearensis extract diluted in 0.9% saline solution), fragments of ovarian cortex were randomly distributed in α-MEM+ (control medium) or into different concentrations of A. cearensis extract without supplements (Amb 0.1; 0.2 or 0.4 mg/mL) or supplemented with the same substances earlier described for the α-MEM+ supplementation (Amb 0.1+; 0.2+ or 0.4+ mg/mL). Each treatment was repeated five times, using the ovaries of five different animals. The culture medium was replenished every second day. After in vitro culture for 7 days, the ovarian fragments from each treatment were fixed in 4% buffered formaldehyde (Dinâmica, Diadema, São Paulo, Brazil) for a period of 18 h and used for histological and TUNEL analyses (Figure 1B).
Morphological analysis of preantral follicles after in vitro preservation or culture
After fixation, the fragments were dehydrated in a graded series of ethanol (Dinâmica, Diadema, São Paulo, Brazil), clarified with xylene (Dinâmica, Diadema, São Paulo, Brazil) and embedded in paraffin wax (Dinâmica, Diadema, São Paulo, Brazil). Tissues were sectioned serially at a thickness of 5 µm and sections were stained using standard protocols with periodic acid-Schiff and hematoxylin (Vetec, Duque de Caxias-RJ, Brazil). Light microscopy (Nikon, Tokyo, Japan) at 400× magnification was used to examine sections.
Preantral follicles were counted and evaluated in the section where the oocyte nucleus was visible. The developmental stages of follicles have been previously defined (SILVA et al., 2004) as primordial (one layer of flattened granulosa cells around the oocyte) or growing follicles (intermediate: one layer of flattened to cuboidal granulosa cells; primary: one layer of cuboidal granulosa cells, and secondary: two or more layers of cuboidal granulosa cells around the oocyte). These follicles were classified individually as histologically normal when an intact oocyte was present and surrounded by granulosa cells that were well organized in one or more layers with the absence of pyknotic nuclei. Atretic follicles were defined as those with a retracted oocyte, pyknotic nucleus, and/or disorganized granulosa cells detached from the basement membrane.
Follicular activation (transition from primordial to growing follicles, when surrounding squamous pre-granulosa cells become cuboidal and begin to proliferate) was only evaluated in treatments submitted to in vitro culture (experiment 2). Only morphologically normal follicles with a visible oocyte nucleus were recorded, and the proportion of primordial and growing follicles was calculated at day 0 (fresh control) and after 7 days of culture.
TUNEL positive cell detection
Terminal deoxynucleotidyl transferase (TdT) mediated dUTP nick-end labeling (TUNEL) assay was used for a more in-depth evaluation of ovine preantral follicle quality before (fresh control) and after in vitro preservation or culture. TUNEL analysis was performed using a commercial kit (In Situ Cell Death Detection Kit, Roche Diagnostics Ltd., Indianapolis, USA) according to the manufacturer's protocol and as previously described (SANTOS et al., 2014), with some modifications. Briefly, sections of 3 µm from each block were cut on a microtome (EasyPath, São Paulo, Brazil) and mounted in Starfrost glass slides (Knittel, Braunschweig, Germany). The slides were incubated in citrate buffer (Dinâmica, Diadema, São Paulo, Brazil) at 95°C in a deckloaking chamber for 40 min to retrieve antigenicity, and endogenous peroxidase activity was prevented by incubation with 3% H2O2 (Dinâmica, Diadema, São Paulo, Brazil) and methyl ethanol (QEEL, São Paulo, Brazil) at room temperature for 10 min. After rinsing in phosphate-buffered saline (PBS; Sigma Aldrich Chemical Co.), the sections were incubated with TUNEL reaction mixture at 37ºC for 1 h. Following this step, the specimens were incubated with Converter-POD in a humidified chamber at 37ºC for 30 min. DNA fragmentation was revealed by incubation of the tissues with diaminobenzidine (DAB; Biocare, Concord, USA) for 1 min. Finally, sections were counterstained with Harry's haematoxylin (Vetec, Duque de Caxias-RJ, Brazil) in a dark chamber at room temperature for 1 min, dehydrated in ethanol, cleared in xylene, and mounted with balsam (Dinâmica, Diadema, São Paulo, Brazil). For negative controls (reaction controls), slides were incubated with label solution (without terminal deoxynucleotidyl transferase enzyme) instead of the TUNEL reaction mixture.
DNA fragmentation in preantral follicles was evaluated as previously described (SANTOS et al., 2014). Only follicles containing an oocyte nucleus were analyzed for TUNEL assay. The number of brown TUNEL positive cells (oocyte and granulosa cells) was counted in ten random fields per treatment using Image-Pro Plus® software. The percentage of TUNEL positive was calculated as the number of TUNEL cells obtained from the total number of cells (x 100).
Statistical analysis
Percentages of morphologically normal follicles (follicular survival), primordial and growing follicles (follicular activation) were submitted to ANOVA and Tukey´s test was applied for comparison among treatments. Values of TUNEL-positive cells were submitted to Chi-square test. The differences were considered to be statistically significant when P < 0.05. The results of follicular survival and growth were expressed as mean ± SD and the results of follicular activation expressed as mean ± SEM.
Results
Experiment 1
Effect of storage conditions on follicular morphology
Among the 1,560 preantral follicles analyzed, 1,056 were primordial, 456 intermediate, 23 primary and 25 secondary follicles. Morphologically normal preantral follicles showed centrally located oocytes and organized granulosa cells surrounded by normal intact basement membranes (Figure 2A, B, and C). Nevertheless, after storage in Amb 0.1 for 24 h, atretic follicles with a retracted oocyte, pyknotic nucleus, and disorganized granulosa cells were observed (Figure 2D).
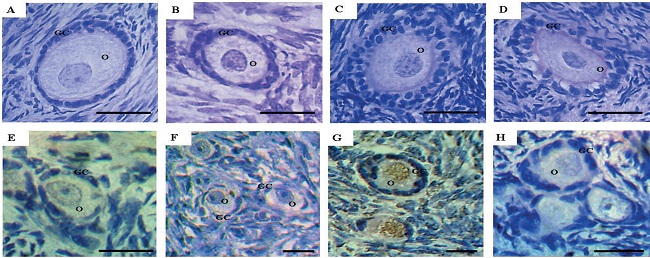
Figure 2
Histological sections and TUNEL positive cell detection in ovine ovarian tissue after in vitro preservation. Normal primary follicle in the fresh control (A), normal intermediate follicle after 6 h of preservation in MEM (B), normal secondary follicle after 24 h of preservation in 0.2 mg/mL A. cearensis extract (C), atretic intermediate follicle after 24 h of preservation in 0.1 mg/mL A. cearensis extract (D), normal primordial follicle after preservation for 6 h in MEM (E) and for 24 h in 0.2 mg/mL A. cearensis extract (F); follicle preserved in 0.4 mg/mL A. cearensis extract for 24 h, showing DNA fragmentation in brown (G) and negative control (H). O: oocyte; GC: granulosa cells. Scale bar: 30 µm (400x).
Storage of ovarian fragments for 6 h in MEM (62%: control medium) was the only treatment in which the percentage of morphologically normal follicles was maintained (P >0.05) similar to that observed in the fresh control (78%) (Table 1). Moreover, this treatment had higher (P<0.05) percentage of normal follicles than all A. cearensis treatments after 6 h of preservation (46%; 43% and 52% for Am b 0.1; Amb 0.2 and Amb 0.4 mg/mL, respectively). After all preservation periods (6, 12 or 24 h), the different concentrations of A. cearensis extract showed similar (P > 0.05) percentages of morphologically normal follicles (6 h: 46%; 43% and 52%; 12 h: 39%; 37% and 45%; 24 h: 35%; 39% and 32% for Amb 0.1; Amb 0.2 and Amb 0.4 mg/mL, respectively). However, after long-term conservation (24 h), only Amb 0.2 mg/mL showed follicular survival rates (39%) similar to those observed in the control medium (MEM: 45%; P>0.05). After comparing the same treatment in the different preservation periods, only the storage of ovine ovarian tissue in Amb 0.2 mg/mL, maintained (P>0.05) follicular survival throughout the periods (6, 12 or 24 h). Nevertheless, after preservation in MEM, a significant decrease was observed in follicular survival from 12 h onwards.
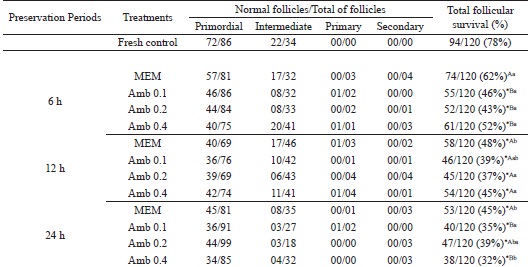
* Differs significantly from fresh control (P < 0.05). (A, B) Different letters denote significant differences among treatments (different media) in the same period (P < 0.05). (a,b) Different letters denote significant differences among periods in the same media (P < 0.05).
TUNEL positive cell detection
Analysis of TUNEL positive cells was performed in the fresh control and in the different treatments preserved at 4ºC for 6, 12 or 24 h (Figure 3). There was an increase in the rate of TUNEL positive cells (oocytes and granulosa cells) (P<0.05) after all periods of preservation in all treatments (MEM - 6 h: 36.17%; 12 h: 43.36%; 24 h: 58%; Amb 0.1 - 6 h: 58.17%; 12 h: 52.73%; 24 h: 75%; Amb 0.2 - 6 h: 55.39%; 12 h: 50%; 24 h: 61%; Amb 0.4 mg/mL - 6 h: 55.63%; 12 h: 58.08%; 24 h: 79%) compared to the fresh control (21%) (Figure 3). Moreover, storage of ovarian tissue for 6 h in the different concentrations of A. cearensis extract increased (P < 0.05) the percentage of follicular cells with DNA fragmentation (Amb 0.1: 59%; Amb 0.2: 55% and Amb 0.4 mg/mL: 56%) compared to MEM (36%). After 12 h of preservation, no differences (P>0.05) were observed in the percentage of TUNEL positive cells between Amb 0.1 (53%) or Amb 0.2 (50%) compared to MEM (43%). However, after long-term storage (24 h), only Amb 0.2 mg/mL kept the rates of TUNEL positive cells (61%) similar to those observed in the control medium (58%)(P>0.05). After comparing the same treatment in the different conservation periods, only the storage of ovine ovarian tissue in Amb 0.2 mg/mL maintained (P>0.05) similar rates of cells with DNA fragmentation throughout the preservation periods (6 h: 55,39%; 12 h: 50% or 24 h: 61%).
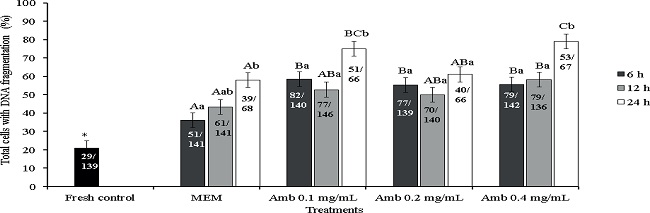
Figure 3
Percentage of TUNEL positive cells (cells showing DNA fragmentation) in the fresh control and after preservation in MEM or A. cearensis extract. *Differs significantly from fresh control (P < 0.05). (A, B) Different letters denote significant differences among treatments (different media) in the same period (P < 0.05). (a,b) Different letters denote significant differences among periods in the same media (P < 0.05).
Figure 2 shows the histological sections after TUNEL analysis. Low apoptotic cells were observed after preservation for 6 h in MEM (Figure 2E) or 24 h in Amb 0.2 mg/mL (Figure 2F). Nevertheless, oocytes with DNA fragmentation were observed in Amb 0.4 mg/mL, which was highlighted by brown staining (Fig 2G). Negative control did not show any staining (Figure 2H).
Experiment 2
Follicular morphology and development after in vitro culture
Among the 1,200 preantral follicles analyzed, 721 were primordial, 527 intermediate, 28 primary and 23 secondary follicles. There was a significant decrease in the percentage of morphologically normal follicles after 7 days of culture in all treatments (78%) compared to the fresh control (Table 2). In addition, the percentage of normal follicles (follicular survival) was significantly higher in the control medium (α-MEM+: 58.25%) compared to other treatments. Moreover, no difference was observed in the rate of follicular survival among the different concentrations of A. cearensis with or without supplements (P>0.05).
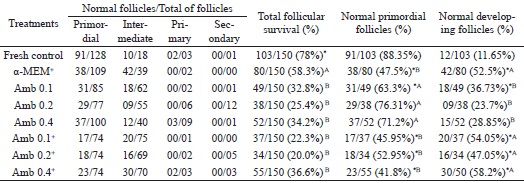
* Differs significantly from fresh control (P < 0.05). (A, B) Different letters denote significant differences among cultured treatments.
After in vitro culture, it was observed a decrease in the percentage of primordial follicles (α-MEM+: 47.5%; Amb 0.1: 63.27%; Amb 0.1+: 45.95%; Amb 0.2+: 52.95%; Amb 0.4+ mg/mL: 41.81%) and a concomitant increase in the percentage of developing follicles in all treatments (α-MEM+: 52.5%; Amb 0.1: 36.73%; Amb 0.1+ : 54.05%; Amb 0.2+ : 47.5%; Amb 0.4+ mg/mL: 58.19 %) compared to fresh control (primordial follicles: 88.35%; developing follicles: 11.65%; P < 0.05), except in both Amb 0.2 mg/mL(primordial follicles: 76.31%; developing follicles: 23.69%) and Amb 0.4 mg/mL (primordial follicles: 71.15%; developing follicles: 28.85%; Table 2) treatments. Furthermore, there was similarity in the percentage of growing follicles between the control medium (α-MEM+: 52.5%) and all supplemented A. cearensis treatments (Amb 0.1+: 54.05%; Amb0,2+: 47.05% and Amb 0.4+ mg/mL: 58.19%), but the value was higher (P < 0.05) than that in the non-supplemented treatments (Amb 0.1: 36.73%; Amb 0.2: 23.69% or Amb 0.4 mg/mL: 28.85%).
TUNEL positive cell detection
Culture in Amb 0.1 mg/mL maintained the percentage of TUNEL positive cells similar to that observed in the fresh control (29% for Amb 0.1 and 22% for fresh control, respectively; P > 0.05) and lower (P < 0.05) than that in the control medium (α-MEM+: 72%), Amb 0.2 mg/mL (59%) and Amb 0.4 mg/mL (76%; Figure 4). Figure 5 shows normal follicles cultured in Amb 0.1 mg/mL (Figure 5A) and a follicle with DNA fragmentation cultured in α-MEM+ (Figure 5B). Negative control did not show staining for TUNEL analysis (Figure 5C).
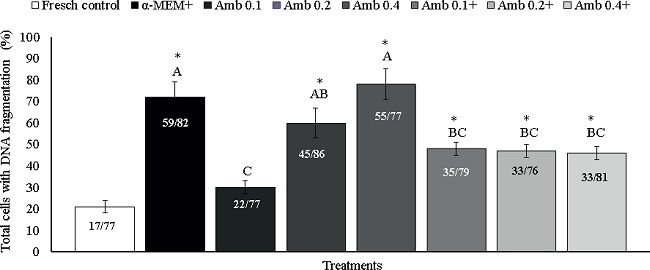
Figure 4
Percentage of TUNEL positive cells (cells showing DNA fragmentation) in fresh tissues (fresh control) and after in vitro culture in α-MEM+ or different concentrations of Amburana cearensis extract in the absence (Amb 0.1; 0.2 and Amb 0.4 mg/mL) or presence of supplements (Amb 0.1+; Amb 0.2+ and Amb 0.4+ mg/mL). *Differs significantly from fresh control (P < 0.05). (A, B) Different letters denote significant differences among cultured treatments.
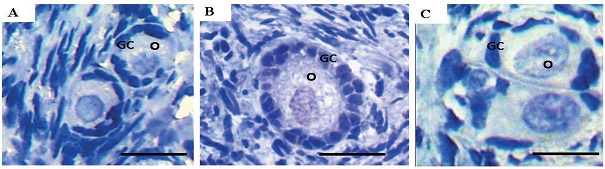
Figure 5
DNA fragmentation detection in ovine ovarian tissue cultured in vitro for 7 days. Normal ovarian follicles after culture in Amb 0.1 mg/mL (A); Apoptotic follicle cultured in α-MEM+ (B) and negative control (C). Note the DNA damage in the oocyte and granulosa cells (brown) in figure B. O: oocyte; GC: granulosa cells. Scale bar: 25 µm (400x).
Discussion
This study demonstrated that 0.2 mg/mL A. cearensis extract can be used to maintain ovine follicles during long-term ovarian tissue transport (24 h). Moreover, supplemented A. cearensis extract can be used to activate primordial follicles in vitro with less DNA damage.
In this study, MEM was efficient to preserve ovine ovarian fragments at 4ºC for up to 6 h. In caprine species, similar results were obtained with the preservation of ovarian fragments in MEM at 4ºC for up to 4 h, including the maintenance of follicular viability after in vitro culture of these fragments (CHAVES et al., 2008). MEM is a commercial medium rich in substances compatible with the nutritional requirements for cell function, such as amino acids (L-methionine, L-tryptophane, L-threonine, L-phenylalanine), glucose (anidro dextrose) and vitamins (folic acid, riboflavin, niacinamide). According to Wongsrikeao et al. (2005), the occlusion of blood flow during ovaries transport to the laboratory reduces the supply of oxygen and energy, which causes accumulation of lactic acid and decreases the pH. It is likely that in the first 6 h of preservation (initial stage of hypoxia), the ovarian tissue may need more metabolic precursors (amino acids and glucose) and vitamins that were found in MEM. On the other hand, the nutritional sources found in the A. cearensis extract may not have been sufficient to reduce follicular loss in the first 6 h of ovaries preservation. Nevertheless, from 12 h onward, the nutrients of MEM may have been insufficient to maintain follicular survival, thus leading to a decrease in the percentage of morphologically normal follicles.
Moreover, it is important to note that all treatments with A. cearensis extract showed follicular survival rates similar to MEM after 12 h of preservation. The ethanolic extract of A. cearensis used was the same from previous studies (GOUVEIA et al., 2015, 2016; BARBERINO et al., 2016b) in which the following compounds were identified: phenolic acids (protocatechuic and gallic acid), flavonoids (epicatechin and kaempferol) and p-coumaric acid. It was demonstrated that the antioxidant action of the protocatechuic acid is related to the metal ion chelating and ROS scavenging activities (LI et al., 2011). This acid also had a protective effect on the male reproductive system of rat after oxidative stress (BEYTUR et al., 2012) and reduced apoptosis in ganglion cells from oxidative damage induced by H2O2 (WANG et al., 2015). Furthermore, a study demonstrated that protocatechuic acid, as the sole antioxidant added to the base medium for ovine secondary follicle culture (replacing the association of transferrin, selenium and ascorbic acid) maintained follicular survival, GSH and active mitochondria levels, meiotic developmental competence and DNA integrity of cultured oocytes (MENEZES et al., 2017). Other studies have shown that treatment with kaempferol increased the levels of catalase, superoxide dismutase and glutathione peroxidase after the oxidative stress induced by alcohol and thermally oxidized polyunsaturated fatty acid in rats (SHAKYA et al., 2014) and significantly suppressed lipopolysaccharide-induced ROS production in rat macrophages (HUANG et al., 2014). Moreover, some studies have shown that pretreatment with gallic acid ameliorates cerebral ischemia/reperfusion injury, and enhances antioxidant defense (FARBOOD et al., 2013). Both epicatechin and p-coumaric acid have radical scavenging effects (DAVID et al., 2002; WANG; SHAHID, 2014) and p-coumaric acid reduced the levels of DNA cleavage (WAND; SHAHID, 2014). Therefore, the presence of these substances in the A. cearensis extract, especially protocatechuic acid and gallic acid, may have decreased oxidative damage on follicular cells after ovary preservation for 12 h.
After increasing the preservation period to 24 h, only A. cearensis with a concentration of 0.2 mg/mL maintained follicular survival and DNA integrity similar to those observed in MEM. It is likely that 0.1 mg/mL did not have sufficient antioxidant levels to maintain the percentage of normal follicles for a long period, probably causing an increase of DNA fragmentation. Additionally, it can be suggested that 0.4 mg/mL may have a cytotoxic effect since high kaempferol levels inhibit the development of sea urchin egg (COSTA-LOTUFO et al., 2003). This maintenance of follicular survival after 24 h with 0.2 mg/mL of A. cearensis would be useful in reducing costs for longer periods of ovaries transport.
According to the results of experiment 1 and in an attempt to integrate preantral follicle preservation and culture, an in situ organ culture was conducted in experiment 2. In this approach, α-MEM maintained preantral follicle survival after in vitro culture. This medium is the most enriched variation of the MEM formulation (containing 21 non-essential amino acids, vitamins, inorganic salts, dextrose, sodium pyruvate, etc.), and has been used as a base culture medium for caprine and ovine ovarian tissue, as well as to maintain the rates of follicular survival similar to those described in this study: 48% (SANTOS et al., 2014) and 58% (MAGALHÃES-PADILHA et al., 2012). Nevertheless, follicular survival was not maintained by A. cearensis after culture. Contrary to this result, 0.1 mg/mL A. cearensis extract without supplements maintained the survival and promoted the in vitro development of ovine isolated secondary follicles after 18 days of culture (BARBERINO et al., 2016b). These differences in results may be related to the lowest numbers of cells in the isolated secondary follicles compared to the cells present in the ovarian tissue. Moreover, the fact that ovarian cortex has follicles in different stages of development, with different metabolic needs, besides the cells of the germinal epithelium and stroma, should be taken into account. Furthermore, a comparison can be made with our preservation results, in which 0.2 mg/mL A. cearensis extract was effective to maintain follicular survival after 24 h of storage (similar to MEM), possibly due to the decreased of the metabolic rate at 4ºC.
An addictive effect of medium supplementation on primordial follicle activation was observed in both media (α-MEM or A. cearensis extract) compared to the fresh control and other non-supplemented treatments. Therefore, although there were lower rates of follicular survival in the different concentrations of A. cearensis, the extract promoted early primordial follicle growth. These results may be explained by the presence of protocatechuic acid in A. cearensis extract, associated with supplements. It was demonstrated that protocatechuic acid increased proliferation of rats nervous cells (GUAN et al., 2009).
Some studies have demonstrated that protocatechuic acid and p-coumaric acid reduced apoptosis (WANG et al., 2015) and DNA fragmentation (WANG; SHAHIDI, 2014) in different cell types. Therefore, in the lowest concentration of the extract, these compounds may be sufficient to prevent or reduce DNA fragmentation in the follicles. Moreover, due to the low rates of morphologically normal follicles, it can be suggested that the follicles died through the necrosis pathway in this treatment (0.1 mg/mL), instead of apoptosis because cell swelling and vacuolated oocytes were observed. However, the presence of these compounds in the highest extract concentrations (0.2 and 0.4 mg/mL) without supplements may have a pro-oxidant effect, increasing apoptosis (SHAILASREE et al., 2015) due to oxidative stress which can lead to DNA fragmentation (JOHNSON, 2003). It is also important to emphasize that all concentrations of supplemented A. cearensis extract showed less DNA damage compared to the control medium. Among the antioxidants added to the media, selenium (sodium selenite) improved the in vitro development of ovarian follicles by reducing the ROS concentration, increasing the total antioxidant capacity and glutathione peroxidase activity (ABEDELAHI et al., 2010), while ascorbic acid enhanced the in vitro development of embryos by reducing ROS levels and the rates of TUNEL positive cells in the swine blastocyst (HU et al., 2012). Moreover, ascorbic acid prevented the formation of hydroxyl radical by preventing them from participating in ROS reactions. Selenium can participate in the decomposition of peroxides by converting them to nontoxic products (ZIMMERMAN et al., 2015; SURRAI et al., 2016).
Conclusions
In conclusion, ovine preantral follicles can be preserved enclosed in ovarian tissue at 4°C in MEM for 6 h. For longer periods of transportation of ovarian tissue (24 h), both MEM and 0.2 mg/mL A. cearensis extract can be used for the maintenance of follicular survival. Moreover, α-MEM or supplemented A. cearensis extract can be used to stimulate primordial follicles activation in vitro. However, the supplemented A. cearensis extract showed less DNA damage than α-MEM after culture. Therefore, A. cearensis extract could be recommended due to the lower cost (about 10 times less that MEM and 40 times less than α-MEM). The use of in vitro activation of primordial follicles represents an opportunity to maximize the number of oocytes available for fertility preservation.
Acknowledgments
This work was supported by the National Council for Scientific and Technological Development (CNPq; Process 562548/2010-6).
References
ABEDELAHI, A.; SALEHNIA, A.; ALLAMEH, A. A.; DAVOODI, D. Sodium selenite improves the in vitro follicular development by reducing the reactive oxygen species level and increasing the total antioxidant capacity and glutathione peroxide activity. Human Reproduction, Oxford v. 25, n. 4, p. 977-985, 2010.
ALBUQUERQUE, U. P.; MEDEIROS, P. M.; ALAMEIDA, A. L. S.; MONTEIRO, J. M.; LIN-NETO, E. M. F.; MELO, J. G.; SANTOS, J. P. Medicinal plants of the caatinga (semi-arid) vegetation of NE Brazil: a quantitative approach. Journal of Ethnopharmacol, Limerick v. 114, n. 3, p. 325-354, 2007.
BARBERINO, R. S.; BARROS, V. R. P.; MENEZES, V. G.; SANTOS, L. P.; ARAÚJO, V. R.; QUEIROZ, M. A. A.; ALMEIDA, J. R. G. S.; PALHETA-JUNIOR, R. C.; MATOS, M. H. T. Amburana cearensis leaf extract maintains survival and promotes in vitro development of ovine secondary follicles. Zygote, Cambridge v. 24, n. 2, p. 277-285, 2016b.
BARBERINO, R. S.; GONÇALVES, R. J. S.; MENEZES, V. G.; BARROS, V. R. P.; LINS, T. L. B. G.; GOUVEIA, B. B.; MACEDO, T. J. S.; SANTOS, L. P.; MATOS, M. H. T. Influence of the ovarian fragmentation before storage at 4oC on the apoptosis rates and in vitro development of ovine preantral follicles. Animal Reproduction, Belo Horizonte v. 13, n. 1, p. 28-35, 2016a.
BEYTUR, A.; CIFTCI, O.; AYDIN, M.; CAKIR, O.; TIMURKAAN, N.; YILMAZ, F. Protocatechuic acid prevents reproductive damage caused by 2, 3, 7, 8-tetrachlorodibenzo-p-dioxin (TCDD) in male rats. Andrologia, Berlin v. 44, p. 454-461, 2012. Supplement 1.
CANUTO, K. M.; SILVEIRA, E. R. Constituintes químicos da casca do caule de Amburana cearensis A. C. Smith. Química Nova, São Paulo v. 29, n. 6, p. 1241-1243, 2006.
CAVALCANTE, A. Y. P.; BARBERINO, R. S.; GOUVEIA, B. B.; BEZERRA, M. E. S.; MENEZES, V. G.; SOUZA, G. R.; ROLIM, L. A.; ROLIM NETO, P. J.; ALMEIDA, J. R. G. S.; MATOS, M. H. T. Effect of ovarian tissue storage in Morus nigra extract on the morphology and DNA fragmentation of ovine preantral follicles. Semina: Ciências Agrárias, Londrina, v. 38, n. 4, p. 1973-1986, 2017
CAVALCANTE, A. Y. P.; GOUVEIA, B. B.; BARBERINO, R. S.; LINS, T. L. B. G.; SANTOS, L. P.; GONÇALVES, R. J. S.; CELESTINO, J. J. H.; MATOS, M. H. T. Kit ligand promotes the transition from primordial to primary follicles after in vitro culture of ovine ovarian tissue. Zygote, Cambrigde v. 24, n. 4, p. 578-582, 2016.
CELESTINO, J. J. H.; BRUNO, J. B.; LIMA-VERDE, I. B.; MATOS, M. H. T.; SARAIVA, M. V.; CHAVES, R. N.; MARTINS, F. S.; LIMA, L. F.; NAME, K. P. O.; CAMPELLO, C. C.; SILVA, J. R. V.; BÁO, S. N.; FIGUEIREDO, J. R. Recombinant epidermal growth factor maintains follicular ultrastructure and promotes the transition to primary follicles in caprine ovarian tissue cultured in vitro. Reproduction Science, Amsterdam v. 16, n. 3, p. 239-246, 2009.
CHAVES, R. N.; MARTINS, F. S.; SARAIVA, M. V. A.; CELESTINO, J. J. H.; LOPES, C. A. P.; CORREIA, J. C.; VERDE, I. B.; MATOS, M. H.; BÁO, S. N.; NAME, K. P.; CAMPELLO, C. C.; SILVA, J. R.; FIGUEIREDO, J. R. Chilling ovarian fragments during transportation improves viability and growth of goat preantral follicles cultured in vitro. Reproduction, Fertility and Development, East Melbourne, v. 20, n. 5, p. 640-647, 2008.
COSTA-LOTUFO, L. V.; JIMENEZ, P. C.; WILKE, D. V.; LEAL, L. K. A. M.; CUNHA, G. M.; SILVEIRA, E. R. Antiproliferative effects of several compounds isolated from Amburana cearensis A. C. Smith. Zeitschrift fur Naturforschung - Section C - Journal of Biosciences, Tubingen, v. 58, n. 9-10, p. 675-680, 2003.
DAVID, J. P.; SANTOS, E. O.; MIRANDA, M. S.; BARREIROS, A. L. B. S.; SANTOS, I. D.; DAVID, J. M. Atividade antioxidante da epicatequina-(2-7,4-8)-epicatequina isolada de Dioclea lasiophylla. Revista Brasileira de Farmacognosia, São Paulo v. 12, p. 5-6, 2002. Suplemento.
FARBOOD, Y.; SARKAKIZ, A.; HASHEMI, S.; MANSOURI, M. T.; DIANAT, M. The effects of gallic acid on pain and memory following transient global ischemia/reperfusion in Wistar rats. Avicenna Journal of Phytomedicine, Mashhad v. 3, n. 4, p. 329-340, 2013.
FIGUEIREDO, J. R.; RODRIGUES, A. P. R.; SILVA, J. R. V.; SANTOS, R. R. Cryopreservation and in vitro culture of caprine preantral follicles. Reproduction, Fertility and Development, East Melbourne, v. 23, n. 1, p. 40-47, 2011.
GONÇALVES, R. J. S.; CAVALCANTE, A. Y. P.; GOUVEIA, B. B.; LINS, T. L. B.; BARBERINO, R. S.; MENEZES, V. G.; BARROS, V. R. P.; MACEDO, T. J. S.; FIGUEIREDO, J. R.; MATOS, M. H. T. Lower apoptosis rate in ovine preantral follicles from ovaries stored in supplemented preservation media. Zygote, Cambrigde v. 23, n. 6, p. 1-8, 2015.
GOUVEIA, B. B.; BARROS, V. P.; GONÇALVES, R. J. S.; BARBERINO, R. S.; MENEZES, V. G.; LINS, T. L. B.; MACEDO, T. J. S.; SANTOS, J. M. S.; ROLIM, L. A.; ROLIM NETO, P. J.; ALMEIDA, J. R. G. S.; MATOS, M. H. T. Effect of ovarian tissue transportation in Amburanacearensis extract on the morphology and apoptosis of goat preantral follicles. Animal Reproduction, Belo Horizonte v. 12, n. 2, p. 316-323, 2015.
GOUVEIA, B. B.; MACEDO, T. J. S.; SANTOS, J. M. S.; BARBERINO, R. S.; MENEZES, V. G.; MÜLLER, M. C.; ALMEIDA, J. R. G. S.; FIGUEIREDO, J. R.; MATOS, M. H. T. Supplemented base medium containing Amburana cearensis associated with FSH improves in vitro development of isolated goat preantral follicles. Theriogenology, Stoneham v. 86, n. 5, p. 1275-1284, 2016.
GUAN, S.; GE, D.; LIU, T. Q.; MA, X.; CUI, Z. F. Protocatechuic acid promotes cell proliferation and reduces basal apoptosis in cultured neural stem cells. Toxicology In Vitro, New York v. 23, n. 2, p. 201-208, 2009.
HU, J.; CHEG, D.; GAO, X.; BAO, J.; MA, X.; WANG, H. Vitamin C enhances the in vitro development of porcine pre-implantation embryos by reducing oxidative stress. Reproduction in Domestic Animals, Berlin v. 47, n. 6, p. 873-879, 2012.
HUANG, Y. B.; LIN, M. W.; CHAO, Y.; HUANG, C. T.; TSAI, Y. H.; WU, P. C.. Anti-oxidant activity and attenuation of bladder hyperactivity by the flavonoid compound kaempferol. International Journal of Urology, Tokyo v. 21, n. 1, p. 94-98, 2014.
JOHNSON, A. L. Intracellular mechanisms regulating cell survival in ovarian follicles. Animal Reproduction Science, Amsterdam v. 78, n. 3-4, p. 185-201, 2003.
KAMOSHITA, K.; OKAMOTO, N.; NAKAJIMA, M.; HAINO, T.; SUGIMOTO, K.; OKAMOTO, A.; SUGISHITA, Y.; SUZUKI, N. Investigation of in vitro parameters and fertility of mouse ovary after storage at an optimal temperature and duration for transportation. Human Reproduction, England v. 31, n. 4, p. 774-781, 2016.
LEAL, L. K. A. M.; PIERDONÁ, T. M.; GÓES, J. G. S.; FONSÊCA, K. S.; CANUTO, K. M.; SILVEIRA, E. R.; BEZERRA, A. M.; VIANA, G. S. A comparative chemical and pharmacological study of standardized extracts and vanillic acid from wild and cultivated Amburanacearensis A. C. Smith. Phytomedicine, Stuttgart v. 18, n. 2-3, p. 230-233, 2011.
LI, X.; WANG, X.; CHEN, D.; CHEN, S. Antioxidant activity and mechanism of protocatechuic acid in vitro. Functional Foods in Health and Disease, San Diego v. 1, n. 7, p. 232-244, 2011.
LIANG, L. F.; QI, S. T.; XIAN, Y. X.; HUANG, L.; SUN, X. F.; WANG, W. H.. Protective effect of antioxidants on the pre-maturation aging of mouse oocytes. Scientific Reports, London v. 7, n. 1, p 1-10, 2017.
MAGALHÃES-PADILHA, D. M.; FONSECA, G. R.; HAAG, K. T.; WISCHRAL, A.; GASTAL, M. O.; JONES, K. L.; GEISLER-LEE, J.; F IGUEIREDO, J. R.; GASTAL, E. L. Long-term in vitro culture of ovarian cortical tissue in goats: effects of FSH and IGF-I on preantral follicular development and FSH and IGF-I receptor mRNA expression. Cell and Tissue Research, Berlin v. 350, n. 3, p. 503-511, 2012.
MENEZES, V. G.; SANTOS, J. M. S.; MACEDO, T. J. S.; LINS, T. L. B.; BARBERINO, R. S.; GOUVEIA, B. B.; BEZERRA, M. E. S.; CAVALCANTE, A. Y. P.; QUEIROZ, M. A. A.; PALHETA-JÚNIOR, R. C.; MATOS, M. H. T. Use of protocatechuic acid as the sole antioxidant in the base medium for in vitro culture of ovine isolated secondary follicles. Reproduction in Domestic Animals, Berlin v. 52, n. 5, p. 890-898, 2017.
SANTOS, J. M. S.; MENEZES, V. G.; BARBERINO, R. S.; MACEDO, T. J. S.; LINS, T. L. B.; GOUVEIA, B. B.; BARROS, V. R. P.; SANTOS, L. P.; GONÇALVES, R. J. S.; MATOS, M. H. T. Immunohistochemical localization of fibroblast growth factor-2 in the sheep ovary and its effects on pre-antral follicle apoptosis and development in vitro. Reproduction in Domestic Animals, Berlin v. 49, n. 3, p. 522-528, 2014.
SHAILASREE, S.; VENKATARAMANA, M.; NIRANJANA, S. R.; PRAKASH, H. S. Cytotoxic effect of p -coumaric acid on neuroblastoma, N2a cell via generation of reactive oxygen species leading to dysfunction of mitochondria inducing apoptosis and autophagy. Molecular Neurobiology, New York v. 51, n. 1, p. 119-130, 2015.
SHAKYA, G.; MANJINI, S.; HODA, M.; RAJAGOPALAN, R.. Hepatoprotective role of kaempferol during alcohol- and DPUFA-induced oxidative stress. Journal of Basic and Clinical Physiology and Pharmacology, London v. 25, n. 1, p. 73-79, 2014.
SHEA, L. D.; WOODRUFF, T. K.; SHIKANOV, A. Bioengineering the ovarian follicle microenvironment. Annual Review of Biomedical Engineering, Palo Alto v. 16, p. 29-52, 2014,
SILVA, J. R. V.; VAN DEN HURK, R.; COSTA, S. H. F.; ANDRADE, E. R.; NUNES, A. P. A.; FERREIRA, F. V. A.; LOBO, R. N. B.; FIGUEIREDO, J. R. Survival and growth of goat primordial follicles after in vitro culture of ovarian cortical slices in media containing coconut water. Animal Reproduction Science, Amsterdam v. 81, n. 3, p. 273-286, 2004.
SURRAI, P. F.; FISININ, V. I.; KARADAS, F. Antioxidant systems in chick embryo development. Part 1. Vitamin E, carotenoids and selenium. Archives of Animal Nutrition, Oxford v. 2, n. 1, p. 1-11, 2016.
WANG, J.; SHAHID, F. Acidolysis of p-Coumaric acid with Omega-3 oils and antioxidant activity of phenolipid products in in vitro and biological model systems. Journal of Agricultural and Food Chemistry, Washington v. 62, n. 2, p. 454-461, 2014.
WANG, Z.; PAN, X.; WANG, D.; SUN, H.; HAN, F.; LV, C.; ZHANG, X. Protective effects of protocatechuic acid on retinal ganglion cells from oxidative damage induced by H2O2. Neurological Research, New York v. 37, n. 2, p. 150-166, 2015.
WANDJI, S. A.; SRSEN, V.; VOSS, A. K.; EPPIG, J. J.; FORTUNE, J. E. Initiation in vitro of growth of bovine primordial follicles. Biology of Reproduction, New York v. 55, n. 5, p. 942-948, 1996.
WONGSRIKEAO, P.; KANESHIGE, Y.; OOKI, R.; TANIGUCHI, M.; AGUNG, B.; NII, M.; OTOI, T. Effect of the removal of cumulus cells on the nuclear maturation, fertilization and development of porcine oocytes. Reproduction in Domestic Animals, Berlin v. 40, n. 2, p. 166-170, 2005.
ZIMMERMAN, M. T.; BAYSE, C. A.; RAMOUTAR, R. B.; BRUMAGHIM, J. L.. Sulfur and selenium antioxidants: challenging radical scavenging mechanisms and developing structure-activity relationships based on metal binding. Journal of Inorganic Biochemistry, New York v. 145, p. 30-40, 2015.
Author notes
*Author for correspondence